A Sequential Anammox Zeolite-Biofilter for the Removal of Nitrogen Compounds from Drinking Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Pre-Treatment and Filter Set-Up
2.1.1. Zeolite Characteristics and Pre-Treatment
2.1.2. Sludge Inoculum
2.2. Anammox Zeolite-Biofilter Operation
2.3. Evaluation
2.4. Analytical Methods
3. Results and Discussion
3.1. Preliminary Investigations
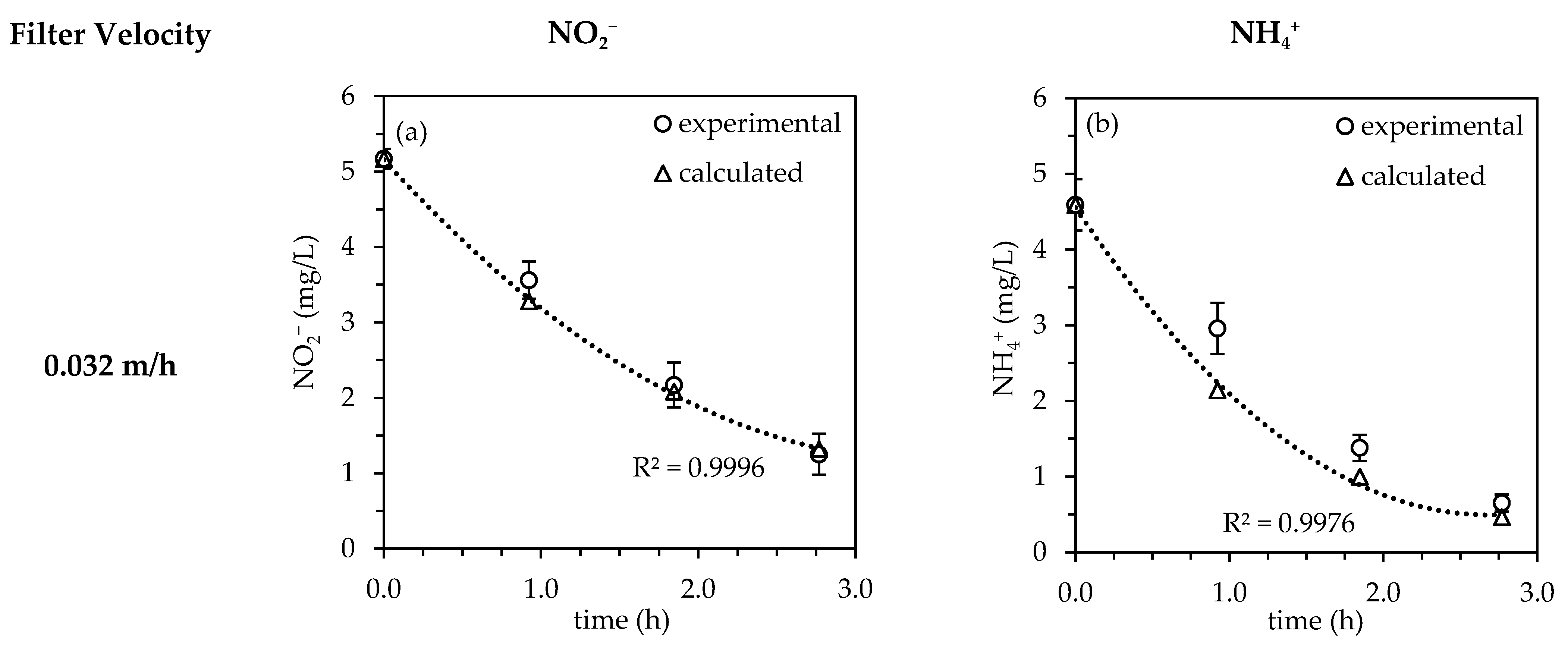
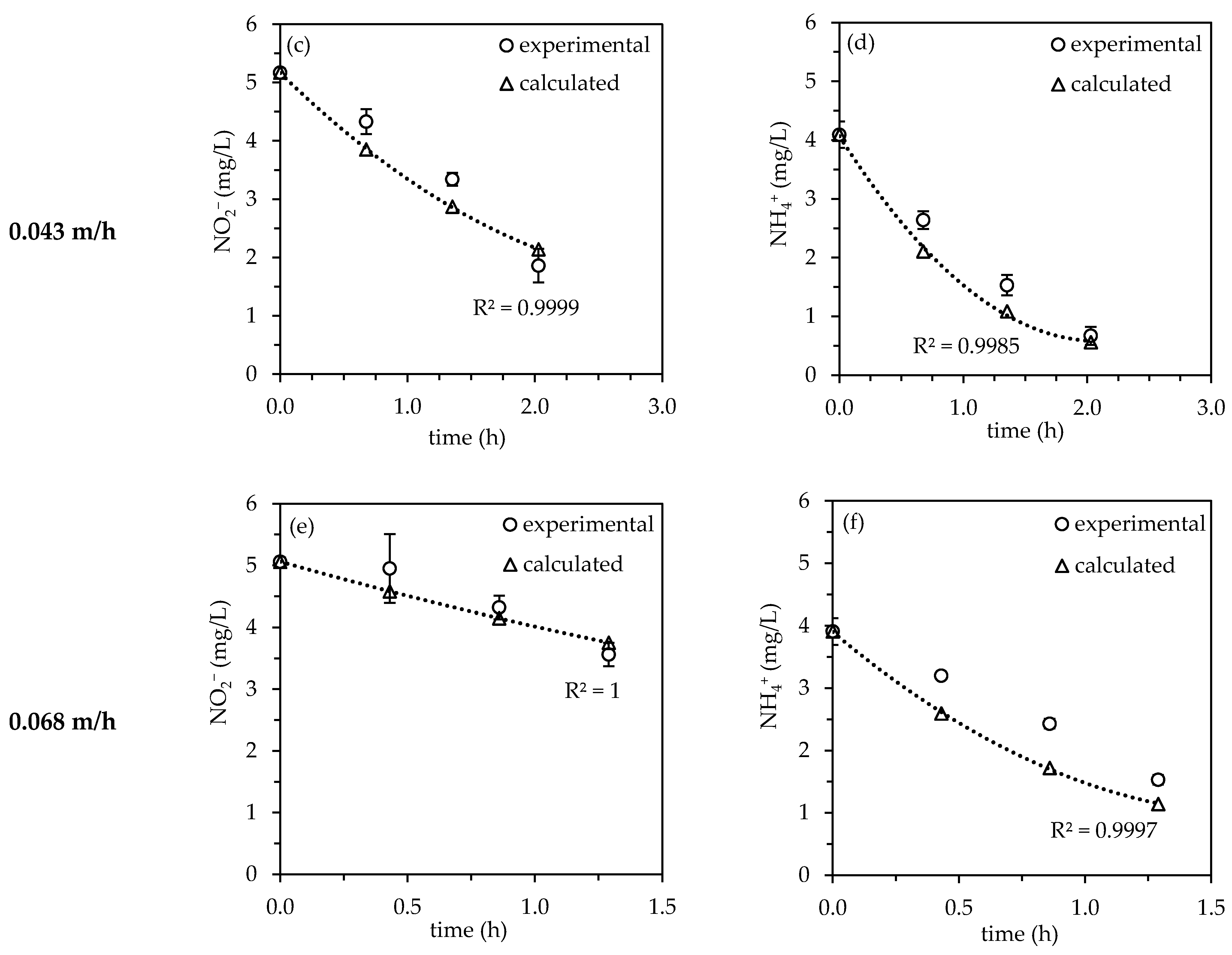
3.2. Influence of Filter Velocity
3.2.1. Investigation of the Nitrite/Ammonium-Ratio and Half-Life
3.2.2. Kinetic Evaluation
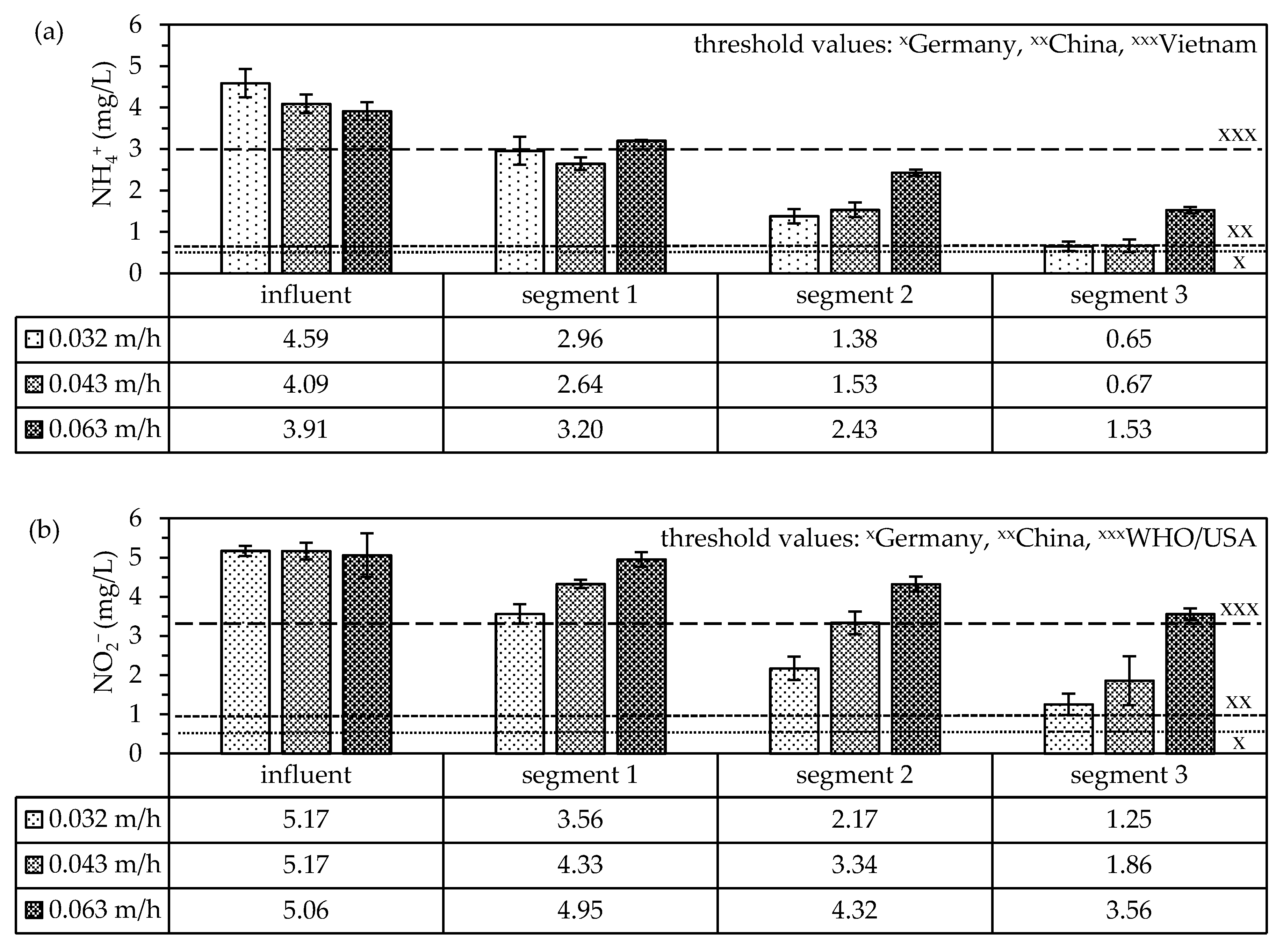
3.2.3. Nitrogen Compound Removal Efficiencies
3.2.4. Compliance with Local Threshold Values
3.3. Simplified Process Monitoring
Correlation of Nitrogen Compounds and Electrical Conductivity
4. Conclusions
- Adapting the Anammox sludge to lower substrate concentrations and temperatures decreased its specific activity.
- An ever-increasing deviation from the ideal NO2−/NH4+-ratio for Amx over the course of the filter bed is coupled with a higher rate of NH4+ and a suboptimal NO2−/NH4+-ratio for the following filter segment.
- In a pseudo-first-order kinetic model, the half-life and reaction rate constants for NH4+ and NO2− removal are strongly affected by filter velocity.
- NH4+ and NO2− removal efficiencies of the Anammox zeolite-biofilter are comparable with those reported in other studies. These other studies were generally carried out at higher concentrations and temperatures; therefore, the change to drinking water conditions did not significantly affect removal efficiencies.
- The WHO’s threshold values for NH4+ and NO2− could be met using our system.
- Simplified process monitoring by correlating electrical conductivity and the concentration of nitrogen compounds is a cost-effective tool and a promising basis for process prediction by ANN in decentralized applications.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bohnet, M.; Ullmann, F. Ullmann’s Encyclopedia of Industrial Chemistry, 6th ed.; Wiley-VCH: Weinheim, Germany, 2003; ISBN 9783527303854. [Google Scholar]
- Roser, M.; Ritchie, H. Nitrogen Fertilizers. Available online: https://ourworldindata.org/fertilizers#nitrogen-fertilizer-production (accessed on 14 November 2021).
- Elshorbagy, W.; Chowdhury, R. (Eds.) Water Treatment; InTech: London, UK, 2013; ISBN 978-953-51-0928-0. [Google Scholar]
- Carrera, J.; Baeza, J.A.; Vicent, T.; Lafuente, J. Biological nitrogen removal of high-strength ammonium industrial wastewater with two-sludge system. Water Res. 2003, 37, 4211–4221. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum; World Health Organisation: Geneva, Switzerland, 2017. [Google Scholar]
- Berg, M.; Tran, H.C.; Nguyen, T.C.; Pham, H.V.; Schertenleib, R.; Giger, W. Arsenic contamination of groundwater and drinking water in Vietnam: A human health threat. Environ. Sci. Technol. 2001, 35, 2621–2626. [Google Scholar] [CrossRef] [PubMed]
- Norrman, J.; Sparrenbom, C.J.; Berg, M.; Dang, D.N.; Jacks, G.; Harms-Ringdahl, P.; Pham, Q.N.; Rosqvist, H. Tracing sources of ammonium in reducing groundwater in a well field in Hanoi (Vietnam) by means of stable nitrogen isotope (δ15N) values. Appl. Geochem. 2015, 61, 248–258. [Google Scholar] [CrossRef]
- Jiao, J.J.; Wang, Y.; Cherry, J.A.; Wang, X.; Zhi, B.; Du, H.; Wen, D. Abnormally high ammonium of natural origin in a coastal aquifer-aquitard system in the Pearl River Delta, China. Environ. Sci. Technol. 2010, 44, 7470–7475. [Google Scholar] [CrossRef] [PubMed]
- Uhlmann, W.; Kreuziger, Y.; Kruspe, R.; Neumann, J.; Berg, T. Untersuchungen zu Ammonium in ostsächsischen Bergbaufolgeseen. Available online: https://www.wasser.sachsen.de/download/Abschlussbericht_TP_05_online.pdf (accessed on 4 August 2021).
- Gupta, V.K.; Sadegh, H.; Yari, M.; Shahryari Ghoshekandi, R.; Maazinejad, B.; Chahardori, M. Removal of ammonium ions from wastewater: A short review in development of efficient methods. Glob. J. Environ. Sci. Manag. 2015, 1, 149–158. [Google Scholar] [CrossRef]
- Ward, M.H.; Jones, R.R.; Brender, J.D.; de Kok, T.M.; Weyer, P.J.; Nolan, B.T.; Villanueva, C.M.; van Breda, S.G. Drinking Water Nitrate and Human Health: An Updated Review. Int. J. Environ. Res. Public Health 2018, 15, 1557. [Google Scholar] [CrossRef]
- Strous, M.; Heijnen, J.J.; Kuenen, J.G.; Jetten, M.S.M. The sequencing batch reactor as a powerful tool for the study of slowly growing anaerobic ammonium-oxidizing microorganisms. Appl. Microbiol. Biotechnol. 1998, 50, 589–596. [Google Scholar] [CrossRef]
- Mulder, A. Anaerobic ammonium oxidation discovered in a denitrifying fluidized bed reactor. FEMS Microbiol. Ecol. 1995, 16, 177–183. [Google Scholar] [CrossRef]
- Strous, M.; Fuerst, J.A.; Kramer, E.H.; Logemann, S.; Muyzer, G.; van de Pas-Schoonen, K.T.; Webb, R.; Kuenen, J.G.; Jetten, M.S. Missing lithotroph identified as new planctomycete. Nature 1999, 400, 446–449. [Google Scholar] [CrossRef]
- Driessen, W.; Hendrickx, T. Two Decades of Experience with the Granular Sludge-Based ANAMMOX® Process Treating Municipal and Industrial Effluents. Processes 2021, 9, 1207. [Google Scholar] [CrossRef]
- Wang, C.-C.; Kumar, M.; Lan, C.-J.; Lin, J.-G. Landfill-leachate treatment by simultaneous partial nitrification, anammox and denitrification (SNAD) process. Desalin. Water Treat. 2011, 32, 4–9. [Google Scholar] [CrossRef]
- Lackner, S.; Gilbert, E.M.; Vlaeminck, S.E.; Joss, A.; Horn, H.; van Loosdrecht, M.C.M. Full-scale partial nitritation/anammox experiences -an application survey. Water Res. 2014, 55, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Waki, M.; Abe, K.; Yasuda, T.; Fukumoto, Y. Tolerance of anammox reactor packed with zeolite to partial supply of nitrite or ammonium using purified livestock wastewater. Environ. Technol. 2020, 41, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Yapsakli, K.; Aktan, C.K.; Mertoglu, B. Anammox-zeolite system acting as buffer to achieve stable effluent nitrogen values. Biodegradation 2017, 28, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Collison, R.; Grismer, M. Upscaling the Zeolite-Anammox Process: Treatment of Secondary Effluent. Water 2018, 10, 236. [Google Scholar] [CrossRef]
- Collison, R.; Grismer, M. Upscaling the Zeolite-Anammox Process: Treatment of Anaerobic Digester Filtrate. Water 2018, 10, 1553. [Google Scholar] [CrossRef]
- Robert, C. Self-Regenerating Zeolite Reactor for sustainable Ammonium Removal. Available online: https://patentimages.storage.googleapis.com/d1/40/48/e4d2ed8436e8f0/US20150151996A1.pdf (accessed on 9 May 2022).
- Weatherley, L.R.; Miladinovic, N.D. Comparison of the ion exchange uptake of ammonium ion onto New Zealand clinoptilolite and mordenite. Water Res. 2004, 38, 4305–4312. [Google Scholar] [CrossRef]
- Kwakye-Awuah, B.; Sefa-Ntiri, B.; Von-Kiti, E.; Nkrumah, I.; Williams, C. Adsorptive Removal of Iron and Manganese from Groundwater Samples in Ghana by Zeolite Y Synthesized from Bauxite and Kaolin. Water 2019, 11, 1912. [Google Scholar] [CrossRef]
- Erdem, E.; Karapinar, N.; Donat, R. The removal of heavy metal cations by natural zeolites. J. Colloid Interface Sci. 2004, 280, 309–314. [Google Scholar] [CrossRef]
- Belviso, C. Zeolite for Potential Toxic Metal Uptake from Contaminated Soil: A Brief Review. Processes 2020, 8, 820. [Google Scholar] [CrossRef]
- Kotoulas, A.; Agathou, D.; Triantaphyllidou, I.; Tatoulis, T.; Akratos, C.; Tekerlekopoulou, A.; Vayenas, D. Zeolite as a Potential Medium for Ammonium Recovery and Second Cheese Whey Treatment. Water 2019, 11, 136. [Google Scholar] [CrossRef]
- Eberle, S.; Börnick, H.; Stolte, S. Granular Natural Zeolites: Cost-Effective Adsorbents for the Removal of Ammonium from Drinking Water. Water 2022, 14, 939. [Google Scholar] [CrossRef]
- Zeolite Products. Home-Zeolite Products. Available online: https://www.zeolite-products.com/ (accessed on 15 March 2022).
- Virta, R. Minerals Yearbook 2011. Available online: https://s3-us-west-2.amazonaws.com/prd-wret/assets/palladium/production/mineral-pubs/zeolites/myb1-2011-zeoli.pdf (accessed on 12 December 2021).
- Grismer, M.; Collison, R. The Zeolite-Anammox Treatment Process for Nitrogen Removal from Wastewater—A Review. Water 2017, 9, 901. [Google Scholar] [CrossRef]
- Montalvo, S.; Huiliñir, C.; Borja, R.; Sánchez, E.; Herrmann, C. Application of zeolites for biological treatment processes of solid wastes and wastewaters—A review. Bioresour. Technol. 2020, 301, 122808. [Google Scholar] [CrossRef] [PubMed]
- Güven, D.; Dapena, A.; Kartal, B.; Schmid, M.C.; Maas, B.; van de Pas-Schoonen, K.; Sozen, S.; Mendez, R.; Op den Camp, H.J.M.; Jetten, M.S.M.; et al. Propionate oxidation by and methanol inhibition of anaerobic ammonium-oxidizing bacteria. Appl. Environ. Microbiol. 2005, 71, 1066–1071. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.-Y.; Chung, Y.-C.; Shin, H.-S.; Son, D.-H. Enhanced ammonia nitrogen removal using consistent biological regeneration and ammonium exchange of zeolite in modified SBR process. Water Res. 2004, 38, 347–354. [Google Scholar] [CrossRef]
- Gisvold, B.; Ödegaard, H.; Föllesdal, M. Enhancing the removal of ammonia in nitrifying biofilters by the use of a zeolite containing expanded clay aggregate filtermedia. Water Sci. Technol. 2000, 41, 107–114. [Google Scholar] [CrossRef]
- Fernández, I.; Vázquez-Padín, J.R.; Mosquera-Corral, A.; Campos, J.L.; Méndez, R. Biofilm and granular systems to improve Anammox biomass retention. Biochem. Eng. J. 2008, 42, 308–313. [Google Scholar] [CrossRef]
- Worch, E. Adsorption Technology in Water Treatment: Fundamentals, Processes, and Modeling, 2nd ed.; De Gruyter: Berlin, Germany, 2021; ISBN 9783110715507. [Google Scholar]
- Van der Star, W.R.L.; Abma, W.R.; Blommers, D.; Mulder, J.-W.; Tokutomi, T.; Strous, M.; Picioreanu, C.; van Loosdrecht, M.C.M. Startup of reactors for anoxic ammonium oxidation: Experiences from the first full-scale anammox reactor in Rotterdam. Water Res. 2007, 41, 4149–4163. [Google Scholar] [CrossRef]
- Jin, R.-C.; Yang, G.-F.; Yu, J.-J.; Zheng, P. The inhibition of the Anammox process: A review. Chem. Eng. J. 2012, 197, 67–79. [Google Scholar] [CrossRef]
- Laureni, M.; Falås, P.; Robin, O.; Wick, A.; Weissbrodt, D.G.; Nielsen, J.L.; Ternes, T.A.; Morgenroth, E.; Joss, A. Mainstream partial nitritation and anammox: Long-term process stability and effluent quality at low temperatures. Water Res. 2016, 101, 628–639. [Google Scholar] [CrossRef] [PubMed]
- Soliman, M.; Eldyasti, A. Ammonia-Oxidizing Bacteria (AOB): Opportunities and applications—A review. Rev. Environ. Sci. Biotechnol. 2018, 17, 285–321. [Google Scholar] [CrossRef]
- De Mulder, C. Impact of intrinsic and extrinsic parameters on the oxygen kinetic parameters of Ammonia and Nitrite Oxidizing Bacteria. LabMET Master 2014, 104. [Google Scholar] [CrossRef]
- Kartal, B.; Geerts, W.; Jetten, M.S.M. Cultivation, detection, and ecophysiology of anaerobic ammonium-oxidizing bacteria. Methods Enzymol. 2011, 486, 89–108. [Google Scholar] [CrossRef]
- Jetten, M.; Schmid, M.; van de Pas-Schoonen, K.; Sinninghe Damsté, J.; Strous, M. Anammox Organisms: Enrichment, Cultivation, and Environmental Analysis. In Environmental Microbiology, Online-Ausg; Leadbetter, J.R., Ed.; Elsevier Academic Press: Amsterdam, The Netherlands, 2005; pp. 34–57. ISBN 9780121828028. [Google Scholar]
- Van de Graaf, A.A.; de Bruijn, P.; Robertson, L.A.; Jetten, M.S.M.; Kuenen, J.G. Autotrophic growth of anaerobic ammonium-oxidizing micro-organisms in a fluidized bed reactor. Microbiology 1996, 142, 2187–2196. [Google Scholar] [CrossRef]
- Ding, C.; Enyi, F.O.; Adrian, L. Anaerobic Ammonium Oxidation (Anammox) with Planktonic Cells in a Redox-Stable Semicontinuous Stirred-Tank Reactor. Environ. Sci. Technol. 2018, 52, 5671–5681. [Google Scholar] [CrossRef]
- Egli, K.; Fanger, U.; Alvarez, P.J.; Siegrist, H.; van der Meer, J.R.; Zehnder, A.J. Enrichment and characterization of an anammox bacterium from a rotating biological contactor treating ammonium-rich leachate. Arch. Microbiol. 2001, 175, 198–207. [Google Scholar] [CrossRef]
- Mulder, A.; Versprille, A.I.; van Braak, D. Sustainable nitrogen removal by denitrifying anammox applied for anaerobic pre-treated potato wastewater. Water Sci. Technol. 2012, 66, 2630–2637. [Google Scholar] [CrossRef]
- Fu, W.; Zhu, R.; Lin, H.; Zheng, Y.; Hu, Z. Effect of organic concentration on biological activity and nitrogen removal performance in an anammox biofilm system. Water Sci. Technol. 2021, 84, 725–736. [Google Scholar] [CrossRef]
- Blackburne, R.; Yuan, Z.; Keller, J. Demonstration of nitrogen removal via nitrite in a sequencing batch reactor treating domestic wastewater. Water Res. 2008, 42, 2166–2176. [Google Scholar] [CrossRef]
- Qin, Y.; Cao, Y.; Ren, J.; Wang, T.; Han, B. Effect of glucose on nitrogen removal and microbial community in anammox-denitrification system. Bioresour. Technol. 2017, 244, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Xiao, P.; Zhang, D.; Lu, P.; Yao, Z.; He, Q. The kinetics for ammonium and nitrite oxidation under the effect of hydroxylamine. Water Sci. Technol. 2016, 73, 1067–1073. [Google Scholar] [CrossRef] [PubMed]
- Jetten, M.S.M.; Wagner, M.; Fuerst, J.; van Loosdrecht, M.; Kuenen, G.; Strous, M. Microbiology and application of the anaerobic ammonium oxidation (‘anammox’) process. Curr. Opin. Biotechnol. 2001, 12, 283–288. [Google Scholar] [CrossRef]
- Lackner, S.; Welker, S.; Gilbert, E.M.; Horn, H. Influence of seasonal temperature fluctuations on two different partial nitritation-anammox reactors treating mainstream municipal wastewater. Water Sci. Technol. 2015, 72, 1358–1363. [Google Scholar] [CrossRef]
- Gilbert, E.M.; Agrawal, S.; Karst, S.M.; Horn, H.; Nielsen, P.H.; Lackner, S. Low temperature partial nitritation/anammox in a moving bed biofilm reactor treating low strength wastewater. Environ. Sci. Technol. 2014, 48, 8784–8792. [Google Scholar] [CrossRef]
- Laureni, M.; Weissbrodt, D.G.; Szivák, I.; Robin, O.; Nielsen, J.L.; Morgenroth, E.; Joss, A. Activity and growth of anammox biomass on aerobically pre-treated municipal wastewater. Water Res. 2015, 80, 325–336. [Google Scholar] [CrossRef]
- Lotti, T.; Kleerebezem, R.; Abelleira-Pereira, J.M.; Abbas, B.; van Loosdrecht, M.C.M. Faster through training: The anammox case. Water Res. 2015, 81, 261–268. [Google Scholar] [CrossRef]
- Pereira, A.D.; Cabezas, A.; Etchebehere, C.; Chernicharo, C.A.d.L.; de Araújo, J.C. Microbial communities in anammox reactors: A review. Environ. Technol. Rev. 2017, 6, 74–93. [Google Scholar] [CrossRef]
- Rütting, T.; Boeckx, P.; Müller, C.; Klemedtsson, L. Assessment of the importance of dissimilatory nitrate reduction to ammonium for the terrestrial nitrogen cycle. Biogeosciences 2011, 8, 1779–1791. [Google Scholar] [CrossRef]
- World Health Organization. Nitrate and Nitrite in Drinking-Water. Available online: https://cdn.who.int/media/docs/default-source/wash-documents/wash-chemicals/nitrate-nitrite-background-jan17.pdf?sfvrsn=1c1e1502_4 (accessed on 10 May 2022).
- Verordnung über die Qualität von Wasser für den Menschlichen Gebrauch (Trinkwasserverordnung-TrinkwV). Available online: http://www.gesetze-im-internet.de/trinkwv_2001/BJNR095910001.html#BJNR095910001BJNG000201310 (accessed on 3 August 2021).
- Ministery of Health Vietnam. National Technical Regulation on Demestic Water Quality. Available online: https://waterhealth.phad.org/wp-content/uploads/2020/07/QCVN-02-2009-BYT.pdf (accessed on 10 May 2022).
- Jin, Y.; E, X.; Chen, C.; Chen, X.-P.; Zhang, L.; Chen, Y.; Cai, Z. National Standard of the People’s Republic of China: Standards for Drinking Water Quality. Available online: http://tradechina.dairyaustralia.com.au/wp-content/uploads/2018/08/GB-5749-2006-Standards-for-Drinking-Water-Quality.pdf (accessed on 10 May 2022).
- U.S. Environmental Protection Agency. National Primary Drinking Water Regulations. Available online: https://www.epa.gov/sites/default/files/2016-06/documents/npwdr_complete_table.pdf (accessed on 10 May 2022).
- Wesoly, I. Betriebsstrategien für den Anammox-Prozess in Sequencing Batch Reaktoren. 2009. Available online: https://depositonce.tu-berlin.de/handle/11303/2571 (accessed on 15 September 2022).
- Fröba, L.; Pettigrew, L.; Vega, M.; Groß, F.; Delgado, A. Messtechnische Datenerfassung und–Analyse zur Prozessregelung einer Dezentralen Abwasseraufbereitungsanlage. Available online: https://www.gala-ev.org/images/beitraege/beitraege%202014/pdf/46.pdf (accessed on 16 May 2022).
- Antwi, P.; Zhang, D.; Xiao, L.; Kabutey, F.T.; Quashie, F.K.; Luo, W.; Meng, J.; Li, J. Modeling the performance of Single-stage Nitrogen removal using Anammox and Partial nitritation (SNAP) process with backpropagation neural network and response surface methodology. Sci. Total Environ. 2019, 690, 108–120. [Google Scholar] [CrossRef]
- Vega-De Lille, M.; Groß, F.; Delgado, A. Modeling, simulation and adaptive process control of biotechnological processes in decentralized anaerobic treatment of domestic wastewater. Proc. Appl. Math. Mech. 2014, 14, 893–894. [Google Scholar] [CrossRef]




| Composition | Value (%) | Characteristics | |
|---|---|---|---|
| SiO2 | 65.00–71.30 | Exchange capacity | 1.2–1.5 mol/kg |
| Al2O3 | 11.50–13.10 | Selectivity | NH4+ > K+ > Na+ > Ca2+ > Mg2+ |
| CaO | 2.70–5.20 | Mean pore diameter | 0.4 nm |
| K2O | 2.20–3.40 | Specific surface | 30–60 m2/g |
| Fe2O3 | 0.70–1.90 | Si/Al | 4.80–5.40 (−) |
| MgO | 0.60–1.20 | Grain size | 8–16 mm |
| Na2O | 0.20–1.30 | ||
| TiO2 | 0.10–0.30 | ||
| Zeolite-Biofilter | Specification | Each Segment | Specification |
|---|---|---|---|
| Total height | 525 mm | Height | 150 mm |
| Filter material | PA x | Zeolite bed height | 80 mm |
| Tube material | PVC xx | Sampling point | 115 mm |
| Pneumatic connector | Nickel plated brass, PA x | Inner diameter | 80 mm |
| Nitrogen Compound | Start Concentration (mg/L) | End Concentration (mg/L) | Removal Efficiency (%) |
|---|---|---|---|
| NH4+ | 4.16 | 0.45 | 89 |
| NO2− | 5.56 | 0.56 | 90 |
| NO3− | 2.41 | 1.17 | 51 |
| TN | 5.46 | 0.98 | 82 |
| Filter Velocity (m/h) | Effective Hydraulic Retention Time (h) | Half-Life (NH4+) (h) | Half-Life (NO2−) (h) |
|---|---|---|---|
| 0.032 | 2.77 | 0.90 | 1.68 |
| 0.048 | 2.03 | 1.04 | 1.85 |
| 0.063 | 1.29 | 1.19 | 3.08 |
| Nitrogen Compound | Start Concentration (mg/L) | Removal Efficiency (%) | Temperature (°C) | Reference |
|---|---|---|---|---|
| NH4+ | 4.3 | 86 | 22 | This study |
| NO2− | 5.7 | 76 | ||
| NH4+ | 100–700 | 95.7 | 20 | Waki et al. [18] |
| NO2− | 110–800 | 96.2 | ||
| NH4+ | 48–301 | 65.5–87.5 | 34 | Yapsakli et al. [19] |
| NO2− | 44–304 | 48–100 | ||
| NH4+ | 479–511 | 82.6–96.1 | 20 | Collison & Grismer [21] |
| NO2− | - x | - x |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eberle, S.; Börnick, H.; Stolte, S. A Sequential Anammox Zeolite-Biofilter for the Removal of Nitrogen Compounds from Drinking Water. Water 2022, 14, 3512. https://doi.org/10.3390/w14213512
Eberle S, Börnick H, Stolte S. A Sequential Anammox Zeolite-Biofilter for the Removal of Nitrogen Compounds from Drinking Water. Water. 2022; 14(21):3512. https://doi.org/10.3390/w14213512
Chicago/Turabian StyleEberle, Stephan, Hilmar Börnick, and Stefan Stolte. 2022. "A Sequential Anammox Zeolite-Biofilter for the Removal of Nitrogen Compounds from Drinking Water" Water 14, no. 21: 3512. https://doi.org/10.3390/w14213512
APA StyleEberle, S., Börnick, H., & Stolte, S. (2022). A Sequential Anammox Zeolite-Biofilter for the Removal of Nitrogen Compounds from Drinking Water. Water, 14(21), 3512. https://doi.org/10.3390/w14213512





