Comparative Influence of Biochar and Zeolite on Soil Hydrological Indices and Growth Characteristics of Corn (Zea mays L.)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Procedure
2.2. Soil Measurements
2.3. Plant Measurements
2.4. Data Analysis
3. Results
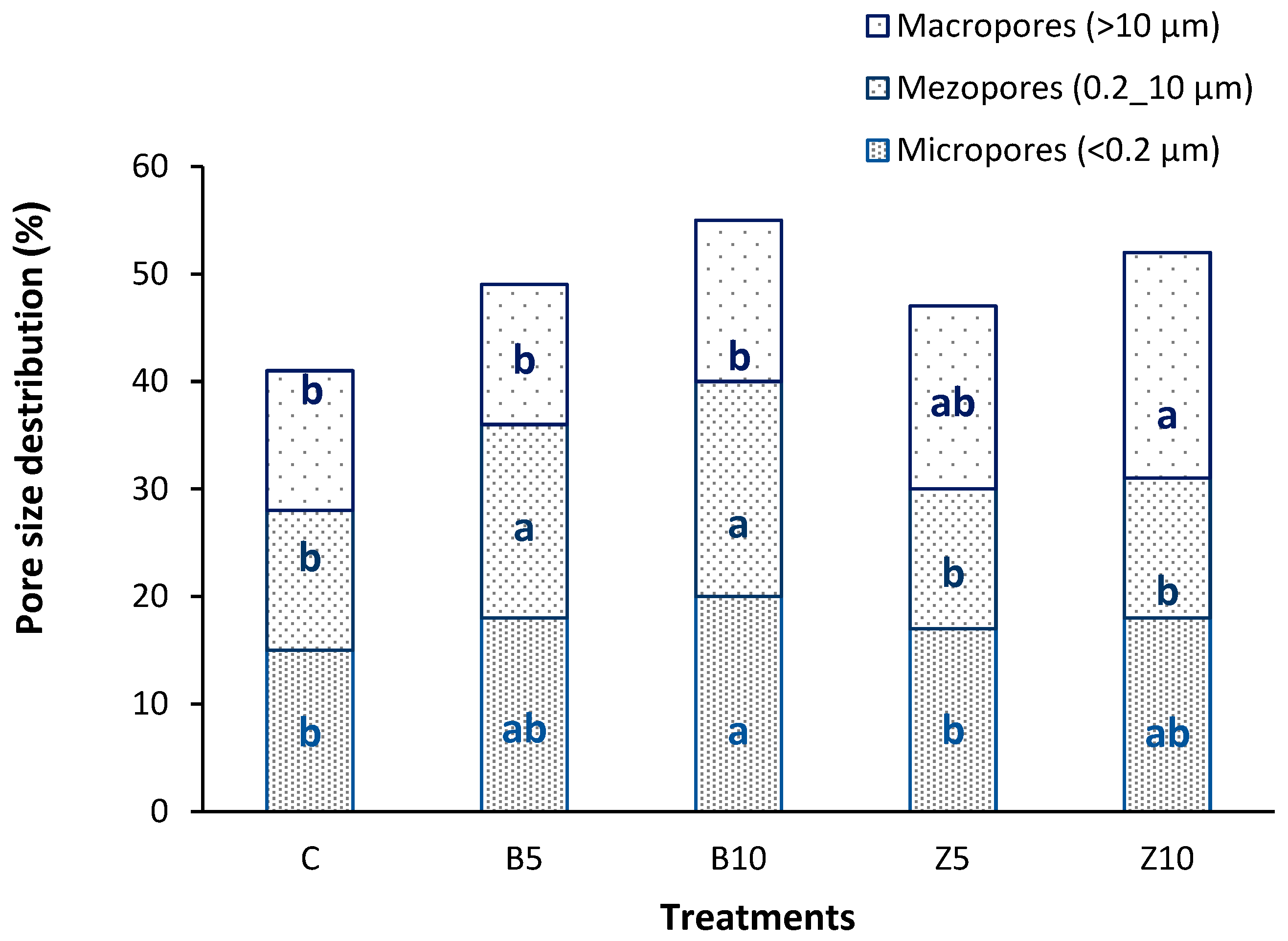
3.1. Changes in Soil Characteristics
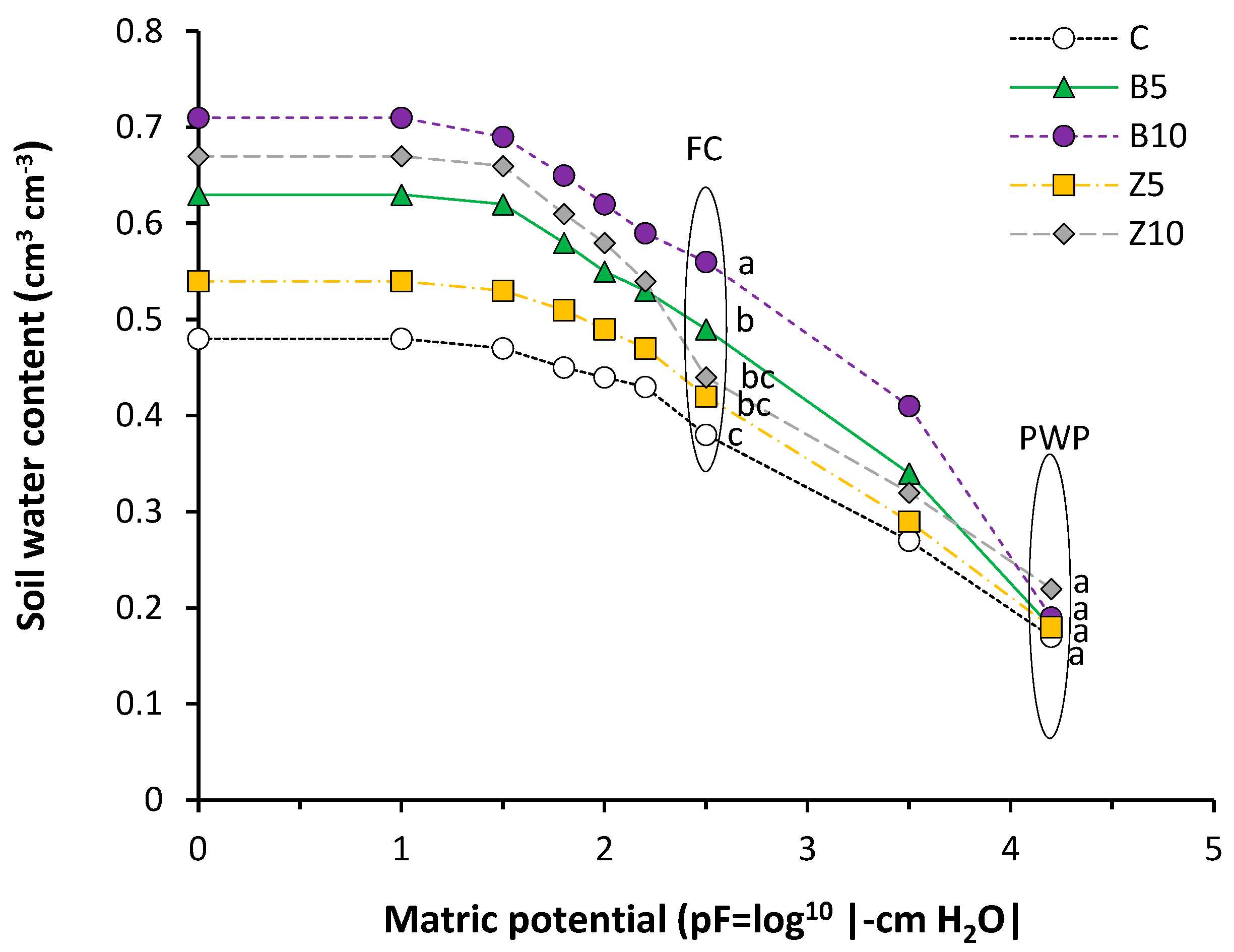
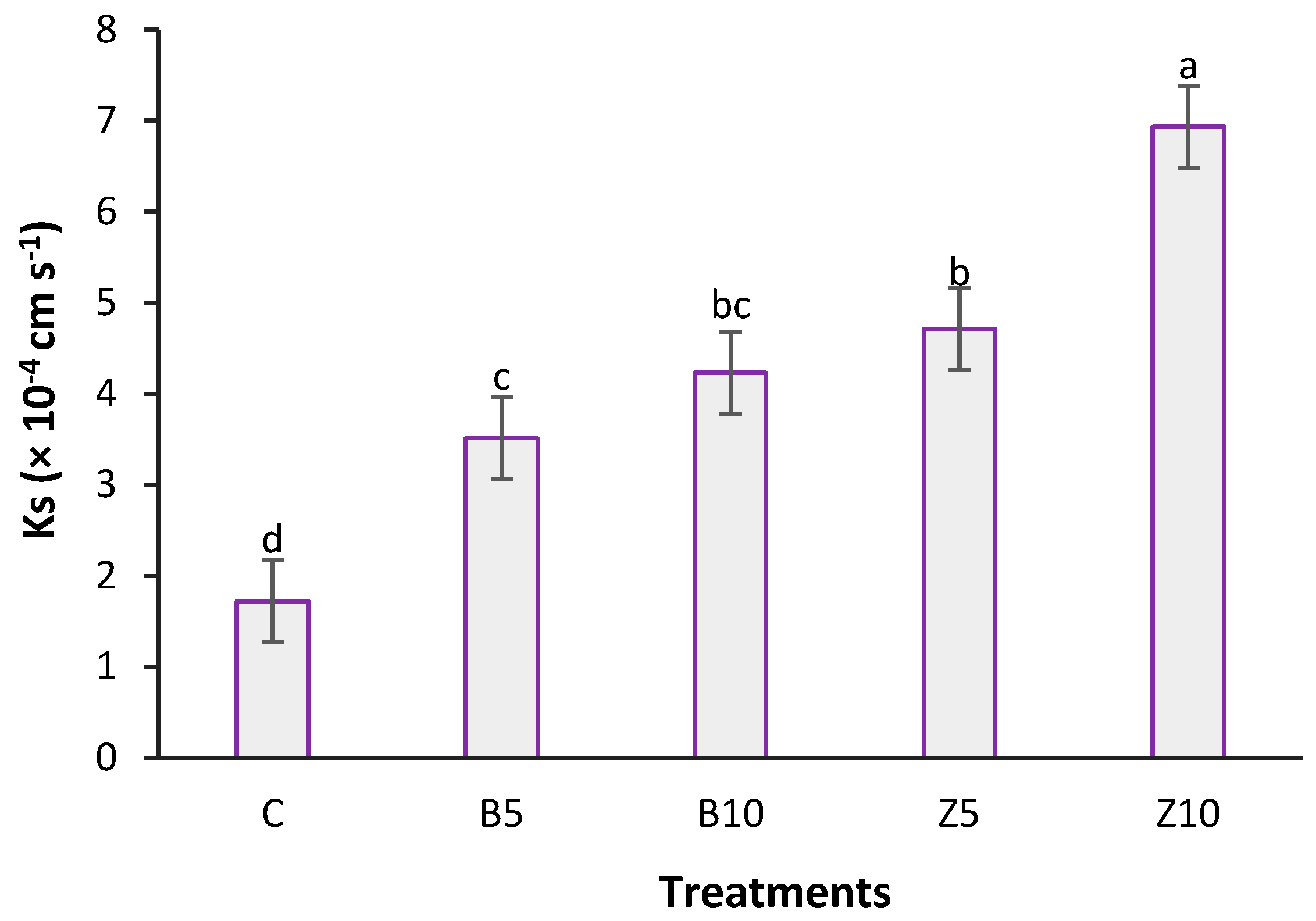
3.2. Changes in Soil Hydrologycal Indics
3.3. Changes in Plant Growth
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yavitt, J.B.; Pipes, G.T.; Olmos, E.C.; Zhang, J.; Shapleigh, J.P. Soil Organic Matter, Soil Structure, and Bacterial Community Structure in a Post-Agricultural Landscape. Front. Earth Sci. 2021, 9, 590103. [Google Scholar] [CrossRef]
- George, P.B.L.; Fidler, D.B.; Van Nostrand, J.D.; Atkinson, J.A.; Mooney, S.J.; Creer, S.; Griffiths, R.I.; McDonald, J.E.; Robinson, D.A.; Jones, D.L. Shifts in Soil Structure, Biological, and Functional Diversity Under Long-Term Carbon Deprivation. Front. Microbiol. 2021, 12, 735022. [Google Scholar] [CrossRef] [PubMed]
- Sessitsch, A.; Weilharter, A.; Gerzabek, M.H.; Kirchmann, H.; Kandeler, E. Microbial Population Structures in Soil Particle Size Fractions of a Long-Term Fertilizer Field Experiment. Appl. Environ. Microbiol. 2001, 67, 4215–4224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, W.; Hodgkinson, L.; Jin, K.; Watts, C.W.; Ashton, R.W.; Shen, J.; Ren, T.; Dodd, I.C.; Binley, A.; Phillips, A.L.; et al. Deep Roots and Soil Structure. Plant. Cell Environ. 2016, 39, 1662–1668. [Google Scholar] [CrossRef] [Green Version]
- Lucas, M.; Schlüter, S.; Vogel, H.-J.; Vetterlein, D. Roots Compact the Surrounding Soil Depending on the Structures They Encounter. Sci. Rep. 2019, 9, 16236. [Google Scholar] [CrossRef] [Green Version]
- Fu, Y.; Tian, Z.; Amoozegar, A.; Heitman, J. Measuring Dynamic Changes of Soil Porosity during Compaction. Soil Tillage Res. 2019, 193, 114–121. [Google Scholar] [CrossRef]
- Singh, N.; Kumar, S.; Udawatta, R.P.; Anderson, S.H.; de Jonge, L.W.; Katuwal, S. X-Ray Micro-Computed Tomography Characterized Soil Pore Network as Influenced by Long-Term Application of Manure and Fertilizer. Geoderma 2021, 385, 114872. [Google Scholar] [CrossRef]
- Satoh, Y.; Yoshimura, K.; Pokhrel, Y.; Kim, H.; Shiogama, H.; Yokohata, T.; Hanasaki, N.; Wada, Y.; Burek, P.; Byers, E.; et al. The Timing of Unprecedented Hydrological Drought under Climate Change. Nat. Commun. 2022, 13, 3287. [Google Scholar] [CrossRef]
- Hassan, W.; Li, Y.; Saba, T.; Jabbi, F.; Wang, B.; Cai, A.; Wu, J. Improved and Sustainable Agroecosystem, Food Security and Environmental Resilience through Zero Tillage with Emphasis on Soils of Temperate and Subtropical Climate Regions: A Review. Int. Soil Water Conserv. Res. 2022, 10, 530–545. [Google Scholar] [CrossRef]
- Liang, B.; Ma, C.; Fan, L.; Wang, Y.; Yuan, Y. Soil Amendment Alters Soil Physicochemical Properties and Bacterial Community Structure of a Replanted Apple Orchard. Microbiol. Res. 2018, 216, 1–11. [Google Scholar] [CrossRef]
- Larkin, R.P. Effects of Selected Soil Amendments and Mulch Type on Soil Properties and Productivity in Organic Vegetable Production. Agronomy 2020, 10, 795. [Google Scholar] [CrossRef]
- Simioniuc, D.P.; Simioniuc, V.; Topa, D.; van den Berg, M.; Prins, U.; Bebeli, P.J.; Gabur, I. Assessment of Andean Lupin (Lupinus Mutabilis) Genotypes for Improved Frost Tolerance. Agriculture 2021, 11, 155. [Google Scholar] [CrossRef]
- Amirahmadi, E.; Mohammad Hojjati, S.; Kammann, C.; Ghorbani, M.; Biparva, P. The Potential Effectiveness of Biochar Application to Reduce Soil Cd Bioavailability and Encourage Oak Seedling Growth. Appl. Sci. 2020, 10, 3410. [Google Scholar] [CrossRef]
- Asadi, H.; Ghorbani, M.; Rezaei-Rashti, M.; Abrishamkesh, S.; Amirahmadi, E.; Chengrong, C.; Gorji, M. Application of Rice Husk Biochar for Achieving Sustainable Agriculture and Environment. Rice Sci. 2021, 28, 325–343. [Google Scholar] [CrossRef]
- Ghorbani, M.; Asadi, H.; Abrishamkesh, S. Effects of Rice Husk Biochar on Selected Soil Properties and Nitrate Leaching in Loamy Sand and Clay Soil. Int. Soil Water Conserv. Res. 2019, 7, 258–265. [Google Scholar] [CrossRef]
- Szatanik-Kloc, A.; Szerement, J.; Adamczuk, A.; Józefaciuk, G. Effect of Low Zeolite Doses on Plants and Soil Physicochemical Properties. Materials 2021, 14, 2617. [Google Scholar] [CrossRef]
- Mortazavi, N.; Bahadori, M.; Marandi, A.; Tangestaninejad, S.; Moghadam, M.; Mirkhani, V.; Mohammadpoor-Baltork, I. Enhancement of CO2 Adsorption on Natural Zeolite, Modified Clinoptilolite with Cations, Amines and Ionic Liquids. Sustain. Chem. Pharm. 2021, 22, 100495. [Google Scholar] [CrossRef]
- Kubů, M.; Millini, R.; Žilková, N. 10-Ring Zeolites: Synthesis, Characterization and Catalytic Applications. Catal. Today 2019, 324, 3–14. [Google Scholar] [CrossRef]
- Moazeni, M.; Parastar, S.; Mahdavi, M.; Ebrahimi, A. Evaluation Efficiency of Iranian Natural Zeolites and Synthetic Resin to Removal of Lead Ions from Aqueous Solutions. Appl. Water Sci. 2020, 10, 60. [Google Scholar] [CrossRef] [Green Version]
- Belviso, C. Zeolite for Potential Toxic Metal Uptake from Contaminated Soil: A Brief Review. Processes 2020, 8, 820. [Google Scholar] [CrossRef]
- Tehrani, R.M.A.; Salari, A.A. The Study of Dehumidifying of Carbon Monoxide and Ammonia Adsorption by Iranian Natural Clinoptilolite Zeolite. Appl. Surf. Sci. 2005, 252, 866–870. [Google Scholar] [CrossRef]
- Martelletti, S.; Meloni, F.; Freppaz, M.; Viglietti, D.; Lonati, M.; Ravetto Enri, S.; Motta, R.; Nosenzo, A. Effect of Zeolitite Addition on Soil Properties and Plant Establishment during Forest Restoration. Ecol. Eng. 2019, 132, 13–22. [Google Scholar] [CrossRef]
- Behzadfar, M.; Sadeghi, S.H.; Khanjani, M.J.; Hazbavi, Z. Effects of rates and time of zeolite application on controlling runoff generation and soil loss from a soil subjected to a freeze-thaw cycle. Int. Soil. Water. Conserv. Res. 2017, 5, 95–101. [Google Scholar] [CrossRef]
- Boros-Lajszner, E.; Wyszkowska, J.; Kucharski, J. Use of zeolite to neutralise nickel in a soil environment. Environ. Monit. Assess. 2018, 190, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azogh, A.; Marashi, S.K.; Babaeinejad, T. Effect of Zeolite on Absorption and Distribution of Heavy Metal Concentrations in Roots and Shoots of Wheat under Soil Contaminated with Weapons. Toxin Rev. 2021, 40, 1301–1307. [Google Scholar] [CrossRef]
- Hamidpour, M.; Kalbasi, M.; Afyuni, M.; Shariatmadari, H.; Holm, P.E.; Hansen, H.C.B. Sorption Hysteresis of Cd(II) and Pb(II) on Natural Zeolite and Bentonite. J. Hazard. Mater. 2010, 181, 686–691. [Google Scholar] [CrossRef]
- Hwang, S.Y.; Yoo, E.S.; Im, S.S. Effects of TS-1 Zeolite Structures on Physical Properties and Enzymatic Degradation of Poly (Butylene Succinate) (PBS)/TS-1 Zeolite Hybrid Composites. Polymer 2011, 52, 965–975. [Google Scholar] [CrossRef]
- Yang, X.; Yang, S.; Yang, S.; Hu, J.; Tan, X.; Wang, X. Effect of PH, Ionic Strength and Temperature on Sorption of Pb(II) on NKF-6 Zeolite Studied by Batch Technique. Chem. Eng. J. 2011, 168, 86–93. [Google Scholar] [CrossRef]
- Amirahmadi, E.; Ghorbani, M.; Moudrý, J. Effects of Zeolite on Aggregation, Nutrient Availability, and Growth Characteristics of Corn (Zea Mays L.) in Cadmium-Contaminated Soils. Water Air Soil Pollut. 2022, 233, 436. [Google Scholar] [CrossRef]
- Ghorbani, M.; Amirahmadi, E.; Zamanian, K. In-situ Biochar Production Associated with Paddies: Direct Involvement of Farmers in Greenhouse Gases Reduction Policies besides Increasing Nutrients Availability and Rice Production. L. Degrad. Dev. 2021, 32, 3893–3904. [Google Scholar] [CrossRef]
- Ghorbani, M.; Konvalina, P.; Neugschwandtner, R.W.; Kopecký, M.; Amirahmadi, E.; Moudrý, J.; Menšík, L. Preliminary Findings on Cadmium Bioaccumulation and Photosynthesis in Rice (Oryza Sativa L.) and Maize (Zea Mays L.) Using Biochar Made from C3- and C4-Originated Straw. Plants 2022, 11, 1424. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Zhao, S.; Geng, R.; Zhao, Y.; Wang, Y.; Yu, F.; Zhang, J.; Ma, X. Does Biochar Application Enhance Soil Salinization Risk in Black Soil of Northeast China (a Laboratory Incubation Experiment)? Arch. Agron. Soil Sci. 2021, 67, 1566–1577. [Google Scholar] [CrossRef]
- Ding, Y.; Liu, Y.; Liu, S.; Li, Z.; Tan, X.; Huang, X.; Zeng, G.; Zhou, L.; Zheng, B. Biochar to Improve Soil Fertility. A Review. Agron. Sustain. Dev. 2016, 36, 36. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.; Li, C.; Parikh, S.J.; Scow, K.M. Impact of Biochar on Water Retention of Two Agricultural Soils—A Multi-Scale Analysis. Geoderma 2019, 340, 185–191. [Google Scholar] [CrossRef]
- Chang, Y.; Rossi, L.; Zotarelli, L.; Gao, B.; Shahid, M.A.; Sarkhosh, A. Biochar Improves Soil Physical Characteristics and Strengthens Root Architecture in Muscadine Grape (Vitis Rotundifolia L.). Chem. Biol. Technol. Agric. 2021, 8, 7. [Google Scholar] [CrossRef]
- Shetty, R.; Prakash, N.B. Effect of Different Biochars on Acid Soil and Growth Parameters of Rice Plants under Aluminium Toxicity. Sci. Rep. 2020, 10, 12249. [Google Scholar] [CrossRef]
- Abbruzzini, T.F.; Davies, C.A.; Toledo, F.H.; Cerri, C.E.P. Dynamic Biochar Effects on Nitrogen Use Efficiency, Crop Yield and Soil Nitrous Oxide Emissions during a Tropical Wheat-Growing Season. J. Environ. Manag. 2019, 252, 109638. [Google Scholar] [CrossRef]
- Joseph, U.E.; Toluwase, A.O.; Kehinde, E.O.; Omasan, E.E.; Tolulope, A.Y.; George, O.O.; Zhao, C.; Hongyan, W. Effect of Biochar on Soil Structure and Storage of Soil Organic Carbon and Nitrogen in the Aggregate Fractions of an Albic Soil. Arch. Agron. Soil Sci. 2020, 66, 1–12. [Google Scholar] [CrossRef]
- Saha, R.; Galagedara, L.; Thomas, R.; Nadeem, M.; Hawboldt, K. Investigating the Influence of Biochar Amendment on the Physicochemical Properties of Podzolic Soil. Agriculture 2020, 10, 471. [Google Scholar] [CrossRef]
- Ahmed, R.; Li, Y.; Mao, L.; Xu, C.; Lin, W.; Ahmed, S.; Ahmed, W. Biochar Effects on Mineral Nitrogen Leaching, Moisture Content, and Evapotranspiration after 15N Urea Fertilization for Vegetable Crop. Agronomy 2019, 9, 331. [Google Scholar] [CrossRef]
- Ahmad Bhat, S.; Kuriqi, A.; Dar, M.U.D.; Bhat, O.; Sammen, S.S.; Towfiqul Islam, A.R.M.; Elbeltagi, A.; Shah, O.; AI-Ansari, N.; Ali, R.; et al. Application of Biochar for Improving Physical, Chemical, and Hydrological Soil Properties: A Systematic Review. Sustainability 2022, 14, 11104. [Google Scholar] [CrossRef]
- Toková, L.; Igaz, D.; Horák, J.; Aydin, E. Effect of Biochar Application and Re-Application on Soil Bulk Density, Porosity, Saturated Hydraulic Conductivity, Water Content and Soil Water Availability in a Silty Loam Haplic Luvisol. Agronomy 2020, 10, 1005. [Google Scholar] [CrossRef]
- Ayaz, M.; Feizienė, D.; Tilvikienė, V.; Akhtar, K.; Stulpinaitė, U.; Iqbal, R. Biochar Role in the Sustainability of Agriculture and Environment. Sustainability 2021, 13, 1330. [Google Scholar] [CrossRef]
- Ameur, D.; Zehetner, F.; Johnen, S.; Jöchlinger, L.; Pardeller, G.; Wimmer, B.; Rosner, F.; Faber, F.; Dersch, G.; Zechmeister-Boltenstern, S.; et al. Activated Biochar Alters Activities of Carbon and Nitrogen Acquiring Soil Enzymes. Pedobiologia 2018, 69, 1–10. [Google Scholar] [CrossRef]
- Hussain, S.; Sharma, V.; Arya, V.M.; Sharma, K.R.; Rao, C.S. Total Organic and Inorganic Carbon in Soils under Different Land Use/Land Cover Systems in the Foothill Himalayas. CATENA 2019, 182, 104104. [Google Scholar] [CrossRef]
- Bu, H.; Liu, D.; Yuan, P.; Zhou, X.; Liu, H.; Du, P. Ethylene Glycol Monoethyl Ether (EGME) Adsorption by Organic Matter (OM)-Clay Complexes: Dependence on the OM Type. Appl. Clay Sci. 2019, 168, 340–347. [Google Scholar] [CrossRef]
- Xu, L.; He, N.; Yu, G. Methods of Evaluating Soil Bulk Density: Impact on Estimating Large Scale Soil Organic Carbon Storage. CATENA 2016, 144, 94–101. [Google Scholar] [CrossRef]
- Besalatpour, A.A.; Ayoubi, S.; Hajabbasi, M.A.; Mosaddeghi, M.R.; Schulin, R. Estimating Wet Soil Aggregate Stability from Easily Available Properties in a Highly Mountainous Watershed. CATENA 2013, 111, 72–79. [Google Scholar] [CrossRef] [Green Version]
- Robertson, B.B.; Gillespie, J.D.; Carrick, S.T.; Almond, P.C.; Payne, J.; Chau, H.W.; Smith, C.M.S. Measuring the Water Retention Curve of Rock Fragments: A Novel Repacked Core Methodology. Eur. J. Soil Sci. 2022, 73, e13181. [Google Scholar] [CrossRef]
- Sakai, K. Determination of Pore Size and Pore Size Distribution. J. Memb. Sci. 1994, 96, 91–130. [Google Scholar] [CrossRef]
- Klute, A. Laboratory Measurement of Hydraulic Conductivity of Saturated Soil; American Society of Agronomy, Inc.: Madison, MI, USA, 2015; pp. 210–221. [Google Scholar] [CrossRef] [Green Version]
- Tsakaldimi, M.N.; Ganatsas, P.P. Effect of Chemical Root Pruning on Stem Growth, Root Morphology and Field Performance of the Mediterranean Pine Pinus Halepensis Mill. Sci. Hortic. 2006, 109, 183–189. [Google Scholar] [CrossRef]
- Wojciechowski, K.L.; Barbano, D.M. Modification of the Kjeldahl Noncasein Nitrogen Method to Include Bovine Milk Concentrates and Milks from Other Species. J. Dairy Sci. 2015, 98, 7510–7526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, M.; Han, G.; Zhang, Q. Effects of Soil Aggregate Stability on Soil Organic Carbon and Nitrogen under Land Use Change in an Erodible Region in Southwest China. Int. J. Environ. Res. Public Health 2019, 16, 3809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramakrishnan, B.; Maddela, N.R.; Venkateswarlu, K.; Megharaj, M. Organic Farming: Does It Contribute to Contaminant-Free Produce and Ensure Food Safety? Sci. Total Environ. 2021, 769, 145079. [Google Scholar] [CrossRef]
- Uzinger, N.; Takács, T.; Szili-Kovács, T.; Radimszky, L.; Füzy, A.; Draskovits, E.; Szűcs-Vásárhelyi, N.; Molnár, M.; Farkas, É.; Kutasi, J.; et al. Fertility Impact of Separate and Combined Treatments with Biochar, Sewage Sludge Compost and Bacterial Inocula on Acidic Sandy Soil. Agronomy 2020, 10, 1612. [Google Scholar] [CrossRef]
- Mukherjee, A.; Zimmerman, A.R.; Hamdan, R.; Cooper, W.T. Physicochemical Changes in Pyrogenic Organic Matter (Biochar) after 15 Months of Field Aging. Solid Earth 2014, 5, 693–704. [Google Scholar] [CrossRef] [Green Version]
- Ghorbani, M.; Konvalina, P.; Kopecký, M.; Kolář, L. A Meta-analysis on the Impacts of Different Oxidation Methods on the Surface Area Properties of Biochar. L. Degrad. Dev. 2022, in press. [Google Scholar] [CrossRef]
- Hafeez, A.; Pan, T.; Tian, J.; Cai, K. Modified Biochars and Their Effects on Soil Quality: A Review. Environments 2022, 9, 60. [Google Scholar] [CrossRef]
- Osman, A.I.; Fawzy, S.; Farghali, M.; El-Azazy, M.; Elgarahy, A.M.; Fahim, R.A.; Maksoud, M.I.A.A.; Ajlan, A.A.; Yousry, M.; Saleem, Y.; et al. Biochar for Agronomy, Animal Farming, Anaerobic Digestion, Composting, Water Treatment, Soil Remediation, Construction, Energy Storage, and Carbon Sequestration: A Review. Environ. Chem. Lett. 2022, 20, 2385–2485. [Google Scholar] [CrossRef]
- Bakshi, S.; Aller, D.M.; Laird, D.A.; Chintala, R. Comparison of the Physical and Chemical Properties of Laboratory and Field-Aged Biochars. J. Environ. Qual. 2016, 45, 1627–1634. [Google Scholar] [CrossRef]
- Wang, S.; Yao, X.; Zhang, Z.; He, X.; Ye, S. Soil Aggregation and Aggregate-Related Exchangeable Base Cations under Different Aged Tea (Camellia Sinensis L.) Plantations in the Hilly Regions of Southern Guangxi, China. Soil Sci. Plant Nutr. 2020, 66, 636–644. [Google Scholar] [CrossRef]
- Munera-Echeverri, J.L.; Martinsen, V.; Strand, L.T.; Zivanovic, V.; Cornelissen, G.; Mulder, J. Cation Exchange Capacity of Biochar: An Urgent Method Modification. Sci. Total Environ. 2018, 642, 190–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berihun, T.; Tadele, M.; Kebede, F. The Application of Biochar on Soil Acidity and Other Physico-chemical Properties of Soils in Southern Ethiopia. J. Plant Nutr. Soil Sci. 2017, 180, 381–388. [Google Scholar] [CrossRef]
- Ren, X.; Sun, H.; Wang, F.; Zhang, P.; Zhu, H. Effect of Aging in Field Soil on Biochar’s Properties and Its Sorption Capacity. Environ. Pollut. 2018, 242, 1880–1886. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Tong, L.; Zhao, N.; Wang, X.; Yang, X.; Lv, Y. Key Factors and Microscopic Mechanisms Controlling Adsorption of Cadmium by Surface Oxidized and Aminated Biochars. J. Hazard. Mater. 2020, 38, 121002. [Google Scholar] [CrossRef]
- Almeida, D.M.; Oliveira, M.M.; Saibo, N.J.M. Regulation of Na+ and K+ Homeostasis in Plants: Towards Improved Salt Stress Tolerance in Crop Plants. Genet. Mol. Biol. 2017, 40 (Suppl. 1), 326–345. [Google Scholar] [CrossRef] [Green Version]
- Al-Busaidi, A.; Yamamoto, T.; Inoue, M.; Eneji, A.E.; Mori, Y.; Irshad, M. Effects of Zeolite on Soil Nutrients and Growth of Barley Following Irrigation with Saline Water. J. Plant Nutr. 2008, 31, 1159–1173. [Google Scholar] [CrossRef]
- Mirzaei Aminiyan, M.; Safari Sinegani, A.A.; Sheklabadi, M. Aggregation Stability and Organic Carbon Fraction in a Soil Amended with Some Plant Residues, Nanozeolite, and Natural Zeolite. Int. J. Recycl. Org. Waste Agric. 2015, 4, 11–22. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Zhang, Y.-J.; Novak, A.; Yang, Y.; Wang, J. Role of Biochar in Improving Sandy Soil Water Retention and Resilience to Drought. Water 2021, 13, 407. [Google Scholar] [CrossRef]
- Wallace, D.; Almond, P.; Carrick, S.; Thomas, S. Targeting Changes in Soil Porosity through Modification of Compost Size and Application Rate. Soil Res. 2020, 58, 268. [Google Scholar] [CrossRef]
- Fan, Q.; Sun, J.; Chu, L.; Cui, L.; Quan, G.; Yan, J.; Hussain, Q.; Iqbal, M. Effects of Chemical Oxidation on Surface Oxygen-Containing Functional Groups and Adsorption Behavior of Biochar. Chemosphere 2018, 207, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Dokoohaki, H.; Miguez, F.E.; Laird, D.; Horton, R.; Basso, A.S. Assessing the Biochar Effects on Selected Physical Properties of a Sandy Soil: An Analytical Approach. Commun. Soil Sci. Plant Anal. 2017, 48, 1387–1398. [Google Scholar] [CrossRef]
- Rahmati, M.; Pohlmeier, A.; Abasiyan, S.M.A.; Weihermüller, L.; Vereecken, H. Water Retention and Pore Size Distribution of a Biopolymeric-Amended Loam Soil. Vadose Zone J. 2019, 18, 1–13. [Google Scholar] [CrossRef]
- Sepaskhah, A.R.; Yousefi, F. Effects of Zeolite Application on Nitrate and Ammonium Retention of a Loamy Soil under Saturated Conditions. Soil Res. 2007, 45, 368. [Google Scholar] [CrossRef]
- Ghorbani, M.; Konvalina, P.; Neugschwandtner, R.W.; Kopecký, M.; Amirahmadi, E.; Bucur, D.; Walkiewicz, A. Interaction of Biochar with Chemical, Green and Biological Nitrogen Fertilizers on Nitrogen Use Efficiency Indices. Agronomy 2022, 12, 2106. [Google Scholar] [CrossRef]
- Peng, J.; Han, X.; Li, N.; Chen, K.; Yang, J.; Zhan, X.; Luo, P.; Liu, N. Combined Application of Biochar with Fertilizer Promotes Nitrogen Uptake in Maize by Increasing Nitrogen Retention in Soil. Biochar 2021, 3, 367–379. [Google Scholar] [CrossRef]
- Bergmann, J.; Verbruggen, E.; Heinze, J.; Xiang, D.; Chen, B.; Joshi, J.; Rillig, M.C. The Interplay between Soil Structure, Roots, and Microbiota as a Determinant of Plant-Soil Feedback. Ecol. Evol. 2016, 6, 7633–7644. [Google Scholar] [CrossRef]
- Sepaskhah, A.R.; Barzegar, M. Yield, Water and Nitrogen-Use Response of Rice to Zeolite and Nitrogen Fertilization in a Semi-Arid Environment. Agric. Water Manag. 2010, 98, 38–44. [Google Scholar] [CrossRef]
- Gholizadeh-Sarabi, S.; Sepaskhah, A.R. Effect of Zeolite and Saline Water Application on Saturated Hydraulic Conductivity and Infiltration in Different Soil Textures. Arch. Agron. Soil Sci. 2013, 59, 753–764. [Google Scholar] [CrossRef]
- Saadat, S.; Sepaskhah, A.R.; Azadi, S. Zeolite Effects on Immobile Water Content and Mass Exchange Coefficient at Different Soil Textures. Commun. Soil Sci. Plant Anal. 2012, 43, 2935–2946. [Google Scholar] [CrossRef]
- Shah, G.A.; Shah, G.M.; Rashid, M.I.; Groot, J.C.J.; Traore, B.; Lantinga, E.A. Bedding Additives Reduce Ammonia Emission and Improve Crop N Uptake after Soil Application of Solid Cattle Manure. J. Environ. Manag. 2018, 209, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Karami, S.; Hadi, H.; Tajbaksh, M.; Modarres-Sanavy, S.A.M. Effect of Zeolite on Nitrogen Use Efficiency and Physiological and Biomass Traits of Amaranth (Amaranthus Hypochondriacus) Under Water-Deficit Stress Conditions. J. Soil Sci. Plant Nutr. 2020, 20, 1427–1441. [Google Scholar] [CrossRef]

| pH | CEC (cmol(+) kg−1) | OC (%) | Ca2+ (cmol(+) kg−1) | Mg2+ (cmol(+) kg−1) | Na+ (cmol(+) kg−1) | Available P (mg kg−1) | Available K (mg kg−1) | Ntot (%) | SSA (m2 g−1) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Soil | 6.61 | 25.6 | 0.51 | 4.61 | 6.12 | 3.26 | 2.87 | 2.51 | 0.88 | 118 |
| Biochar | 9.42 | 185.2 | 52.4 | 25.9 | 38.5 | 13.5 | 10.1 | 28.7 | 1.51 | 278 |
| Zeolite | 8.11 | 148.1 | - | 17.7 | 12.3 | 45.8 | 1.31 | 2.12 | 0.31 | 78.9 |
| pH | CEC (cmolc kg−1) | OM (%) | SAR (meq |−1)0.5 | BD (g cm−3) | TP (%) | MWD mm | SSA (m2 g−1) | |
|---|---|---|---|---|---|---|---|---|
| C | 6.53 c | 24.5 e | 0.87 c | 3.12 c | 1.42 a | 41.1 c | 0.68 c | 116 d |
| B5 | 7.35 b | 115.1 b | 1.31 b | 3.39 c | 1.33 b | 49.3 b | 1.22 b | 251 b |
| B10 | 7.89 a | 139.2 a | 1.68 a | 3.88 c | 1.21 b | 54.8 a | 1.81 a | 315 a |
| Z5 | 6.68 c | 38.5 d | 0.91 c | 5.16 b | 1.41 a | 47.1 b | 0.61 c | 170 c |
| Z10 | 6.73 c | 59.7 c | 0.95 c | 7.59 a | 1.38 a | 50.2 ab | 0.35 d | 281 b |
| Root Analysis | Shoot Analysis | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Root Dry Weight (g) | Root Total Length (cm) | Root Area (cm2) | Root Diameter (mm) | Shoot Dry Weight (g) | Shoot Length (cm) | N (mg g−1) | P (mg g−1) | K (mg g−1) | |
| C | 0.75 c | 532 d | 645 c | 0.38 a | 141 c | 72 c | 63.2 d | 23.6 d | 95.1 c |
| B5 | 0.88 b | 664 b | 689 b | 0.39 a | 156 b | 85 b | 134.4 b | 61.8 b | 147.5 b |
| B10 | 0.96 a | 692 a | 764 a | 0.41 a | 168 a | 94 a | 176.5 a | 75.7 a | 181.2 a |
| Z5 | 0.78 c | 581 c | 685 b | 0.37 a | 143 c | 75 bc | 85.2 c | 39.2 c | 101.5 c |
| Z10 | 0.85 b | 605 c | 694 b | 0.38 a | 144 c | 77 bc | 66.5 d | 25.7 d | 108 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghorbani, M.; Amirahmadi, E.; Konvalina, P.; Moudrý, J.; Bárta, J.; Kopecký, M.; Teodorescu, R.I.; Bucur, R.D. Comparative Influence of Biochar and Zeolite on Soil Hydrological Indices and Growth Characteristics of Corn (Zea mays L.). Water 2022, 14, 3506. https://doi.org/10.3390/w14213506
Ghorbani M, Amirahmadi E, Konvalina P, Moudrý J, Bárta J, Kopecký M, Teodorescu RI, Bucur RD. Comparative Influence of Biochar and Zeolite on Soil Hydrological Indices and Growth Characteristics of Corn (Zea mays L.). Water. 2022; 14(21):3506. https://doi.org/10.3390/w14213506
Chicago/Turabian StyleGhorbani, Mohammad, Elnaz Amirahmadi, Petr Konvalina, Jan Moudrý, Jan Bárta, Marek Kopecký, Răzvan Ionuț Teodorescu, and Roxana Dana Bucur. 2022. "Comparative Influence of Biochar and Zeolite on Soil Hydrological Indices and Growth Characteristics of Corn (Zea mays L.)" Water 14, no. 21: 3506. https://doi.org/10.3390/w14213506
APA StyleGhorbani, M., Amirahmadi, E., Konvalina, P., Moudrý, J., Bárta, J., Kopecký, M., Teodorescu, R. I., & Bucur, R. D. (2022). Comparative Influence of Biochar and Zeolite on Soil Hydrological Indices and Growth Characteristics of Corn (Zea mays L.). Water, 14(21), 3506. https://doi.org/10.3390/w14213506











