The Application of Passive Sampling Devices in Wastewater Surveillance
Abstract
:1. Introduction
2. Materials and Methods
3. Results
| Sampler | Sampling Device or Material | Configuration | Target | Reference |
|---|---|---|---|---|
| DGT 1,* | Device | Binding agent: AG MP-1 | Platinum group elements | [12] |
| Binding agent: Chelex® and TiO2 | Metals | [55] | ||
| Binding agent: Chelex® resin; filter: polysulfone membrane; diffusive agent: open pore gel | Silver nanoparticles | [104] | ||
| Binding agent: mixed cation exchange gel; filter: nylon filter membrane; diffusive agent: polyacrylamide with agarose derivative cross linker gel | Melamine and related triazines | [105] | ||
| Binding agent: mixed cation exchange gel; filter: PES, PTFE, PVDF, polycarbonate and nylon; diffusive agent: polyacrylamide gel | Denatonium benzoate | [106] | ||
| o-DGT 3 | Device | Binding phase: HLB binding gel Diffusive agent: agarose gel | Pharmaceuticals | [18] |
| Binding phase: HLB binding gel Diffusive agent: agarose gel | Pesticides | [18] | ||
| Binding phase: HLB containing binding gel Diffusive agent: diffusive gel | Polar organic contaminants | [54] | ||
| Binding phase: HLB, XAD 18 or XDA-1 resin Diffusive agent: agarose gel Filter: PES membrane | Illicit drugs | [70] | ||
| Binding phase: HLB, XAD 18 or XDA-1 resin Diffusive agent: agarose gel Filter: PES membrane | Antibiotics | [70] | ||
| Binding phase: XAD18 resin Diffusive agent: agarose gel Filter: Hydrophilic Millipore membrane Binding phase: Sepra-ZT binding gel Diffusive agent: agarose gel | Estrogen and estrogen-like compounds | [80] | ||
| Binding phase: Sepra-ZT binding gel Diffusive agent: agarose gel | Pharmaceuticals | [16] | ||
| Binding phase: XAD 18 resin Diffusive agent: agarose gel Filter: PES membrane | Pesticides | [16] | ||
| Antibiotics | [14] | |||
| Binding agent: XAD18 resin; filter: PES membrane; diffusive agent: agarose gel | Per and polyfluoroalkyls substances | [42] | ||
| Binding agent: weak anion exchanger; filter: hydrophilic PES membrane; diffusive agent: agarose gel | Per and polyfluoroalkyls substances | [40] | ||
| Binding agent: XAD18 resin; filter: membrane filter; diffusive agent: agarose gel | Estrogens | [43] | ||
| Binding agent: porous carbon material gel | Antibiotics | [48] | ||
| Binding agents: HLB, XAD18, or Strata-XL-A; diffusive agent: polyacrylamide and agarose gel | Endocrine disrupting chemicals | [39,79] | ||
| Binding agents: HLB, XAD18, or Strata-XL-A; diffusive agent: polyacrylamide and agarose gel | Household and personal care products | [107] | ||
| Binding agents: Sigma-MIP resin; filter membranes: PES, PTFE, PVDF, polycarbonate and nylon; diffusive agent: polyacrylamide | Fluoroquinolone | [108] | ||
| POCIS 2 | Device | Oasis HLB sorbent between two PES membranes | Pharmaceuticals | [1,9,11,25,56,65,67,69,71,72,75,76] |
| Anticancer drugs | [74] | |||
| Illicit drugs | [85] | |||
| Fluoroquinolone antibiotics | [68] | |||
| Antibiotic resistant genes | [68] | |||
| Pathogenic viral genomes | [68] | |||
| Polar organic contaminants | [54,62] | |||
| Polar organic compounds | [55] | |||
| Organic pollutants | [59,61] | |||
| Hydrophilic contaminants of emerging concern | [57] | |||
| Estrogens | [49,78,82,84] | |||
| Methamphetamine | [10] | |||
| Pesticides | [9,56,65] | |||
| Endocrine disrupting compounds | [65] | |||
| Chemical contaminants | [15] | |||
| Beta-blockers and hormones | [9] | |||
| Steroid hormones | [109] | |||
| Photosystem II inhibitors | [52] | |||
| Emerging contaminants | [51] | |||
| Immobilized ionic liquid between two PES membranes | Per-fluorinated substances | [52] | ||
| Oasis HLB sorbent between two polysulfone membranes | Various emerging contaminants | [24] | ||
| Triphasic admixture of hydroxylated polystyrene-divinylbenzene resin and adsorbent dispersed on a styrene divinylbenzene co-polymer | Pharmaceuticals and illicit drugs | [77] | ||
| Home synthesized sorbents and nylon membranes | Organic contaminants | [63] | ||
| Chemcatcher® | Device | Teflon; diffusion limiting membrane; Ion-exchange receiving disk | Nitrate and phosphate | [24] |
| SDB-RPS disks | Micropollutants | [88] | ||
| HLB-L disks as receiving phase covered by PES membranes | Pharmaceuticals | [1,5] | ||
| Receiving phase: SDB-XC, or SDB-RPS, or C18FF; diffusion phase: PES membrane or Omnipore membrane | Trace organic chemicals | [17] | ||
| Empore disks | Estrogens | [82] | ||
| C18 Empore disks | Organotin compounds | [3] | ||
| SDB-RPS disk | Pharmaceuticals or pharmaceutical ingredients | [50] | ||
| HLB disk covered by PES membrane | Personal care products | [5] | ||
| Illicit drugs | [5] | |||
| SDB-RPS disks | Estrogenic activity | [50] | ||
| SDB-RPS 4 | Material | SDB-RPS disks | Polar organic contaminants | [45] |
| PES membranes | Estrogenic activity | [60] | ||
| Organic micropollutants | [60] | |||
| Empore disks | Estrogens | [82] | ||
| Endocrine disrupting compounds | [19] | |||
| SDK-RPS disks mounted on an aluminium alloy plate | Micropollutants | [87] | ||
| PASSIL 5 | Device | Ionic liquid between two PES membranes held between two screwed together plexiglass disks | Pharmaceuticals | [25] |
| MESCO 6 | Device | PDMS or PES or POM | Polar organic contaminants | [47] |
| Tampons | Material | Optical brightener free tampons | Optical brighteners | [38] |
| Hollow fibre silicone membranes | Material | Polar organic contaminants | [46] | |
| Agarose hydrogel diffusion-based sampler | Material | HLB sorbent between diffusive hydrogel disks | Chemical and biological contaminants | [103] |
| Strata-X SPE sorbent | Licit and illicit drugs | [2] | ||
| Microporous PE tube 7 | Material | Strata-X sorbent and agarose gel | Pharmaceuticals and personal care products | [2] |
| Multi-armed polyethene strip | Material | Industry discharge | [13] | |
| SR sheets 8 | Material | Non-polar organic compounds | [55] | |
| Polyethylene | Material | Poly- and per fluorinated alkyl substances | [89,90] | |
| SPMD 9 | Device | Hydrophobic contaminants of emerging concern | [57] | |
| Chemical contaminants | [65] | |||
| Pesticides | [65] | |||
| Pharmaceuticals | [65] | |||
| PE tubing | Pharmaceuticals | [69] | ||
| Silicone rubber | Material | Chemical characterisation | [66] | |
| Silicone sampler | Material | Translucent silicone sheets | Hydrophobic organic compounds | [58] |
| MPS 10 | Device | Combined PDMS and Oasis HLB | Organic contaminants | [62] |
| Microporous ceramic sampler | Device | Diffusion phase: water membrane; reverse phase: Sepra ZT; retaining phase; pyrrolidone modified SDB polymer | Anticancer drugs | [73] |
| Ceramic toximeters | Device | Dissolved dioxin-like PCBs | [37] | |
| Polymer inclusion membrane | Material | NaCl receiving solution | Sulfamethoxazole | [41] |
| Partitioned based sampler | Device | Silicone rubber sheets | Chemical status (toxicity and chemical analysis) | [64] |
| HLB embedded cellulose acetate membrane | Material | Organophosphate flame retardants | [91] | |
| Zetapore filter | Filter | Norovirus and ostreid herpesvirus type 1 | [36] | |
| Low-density PE | Material | Norovirus and ostreid herpesvirus type 1 | [36] | |
| Nylon nets | Material | Norovirus and ostreid herpesvirus type 1 | [36] | |
| Polyvinylidene difluoride immobilon | Material | Norovirus and ostreid herpesvirus type 1 | [36] | |
| Gauze pads | Material | Norovirus and ostreid herpesvirus type 1 | [36] | |
| Medical gauze | Material | Paratyphoid bacilli | [21] | |
| Polio virus | [22] | |||
| COSCa 11 | Device | 3D printed acrylonitrile butadiene styrene hollowed sphere containing either electronegative filters, medical gauze, a cheesecloth or a cellulose sponge. | SARS-CoV-2 | [28] |
| 3D printed torpedo style sampler | Device | Contained medical gauze, swabs, electronegative filter membranes and cotton buds. Sampler wrapped in shade cloth. | SARS-CoV-2 | [20] |
| 3D printed matchbox style sampler | Device | Contained cotton buds, hot glued into location. Wrapped in shade cloth. | SARS-CoV-2 | [20] |
| 3D printed boat style sampler | Device | Contained medical gauze, swabs, electronegative filter membranes and cotton buds. Sampler wrapped in shade cloth. | SARS-CoV-2 | [20] |
| Colander sampler | Device | Made from readily available colander from IKEA containing gauze swabs, electronegative filter membranes and cotton buds. Sampler wrapped in shade cloth. | SARS-CoV-2 | [20] |
| Moore Swab | Material | Medical gauze | Salmonella typhi | [94,95,96,97,98] |
| Salmonella spp. | [93] | |||
| Paratyphoid B | [92] | |||
| Enteric organisms | [99] | |||
| Vibrio cholerae | [100] | |||
| Coxsackievirus | [96] | |||
| Mycobacterium tuberculosis | [96] | |||
| SARS-CoV-2 | [27,102] | |||
| Organic cotton tampon | Material | SARS-CoV-2 | [101] | |
| 3D torpedo style sampler | Device | Contained medical gauze, swabs, and electronegative filter membranes. | SARS-CoV-2 | [101] |
| 3D printed sampler | Device | Contained electronegative filters. | SARS-CoV-2 | [30] |
| Cotton tampon-based sampler | Material | SARS-CoV-2 | [31] | |
| Ion exchange filter papers | Material | SARS-CoV-2 | [31] | |
| 3D printed torpedo style sampler | Device | Contained electronegative membranes. | SARS-CoV-2 | [32] |
| Zetapore membrane sampler | Material | SARS-CoV-2 | [33] | |
| Nylon membrane sampler | Material | SARS-CoV-2 | [33] | |
| 3D printed torpedo style sampler | Device | Contained cotton swabs and electronegative filter membranes. | SARS-CoV-2 | [34] |
3.1. Sampling Sites
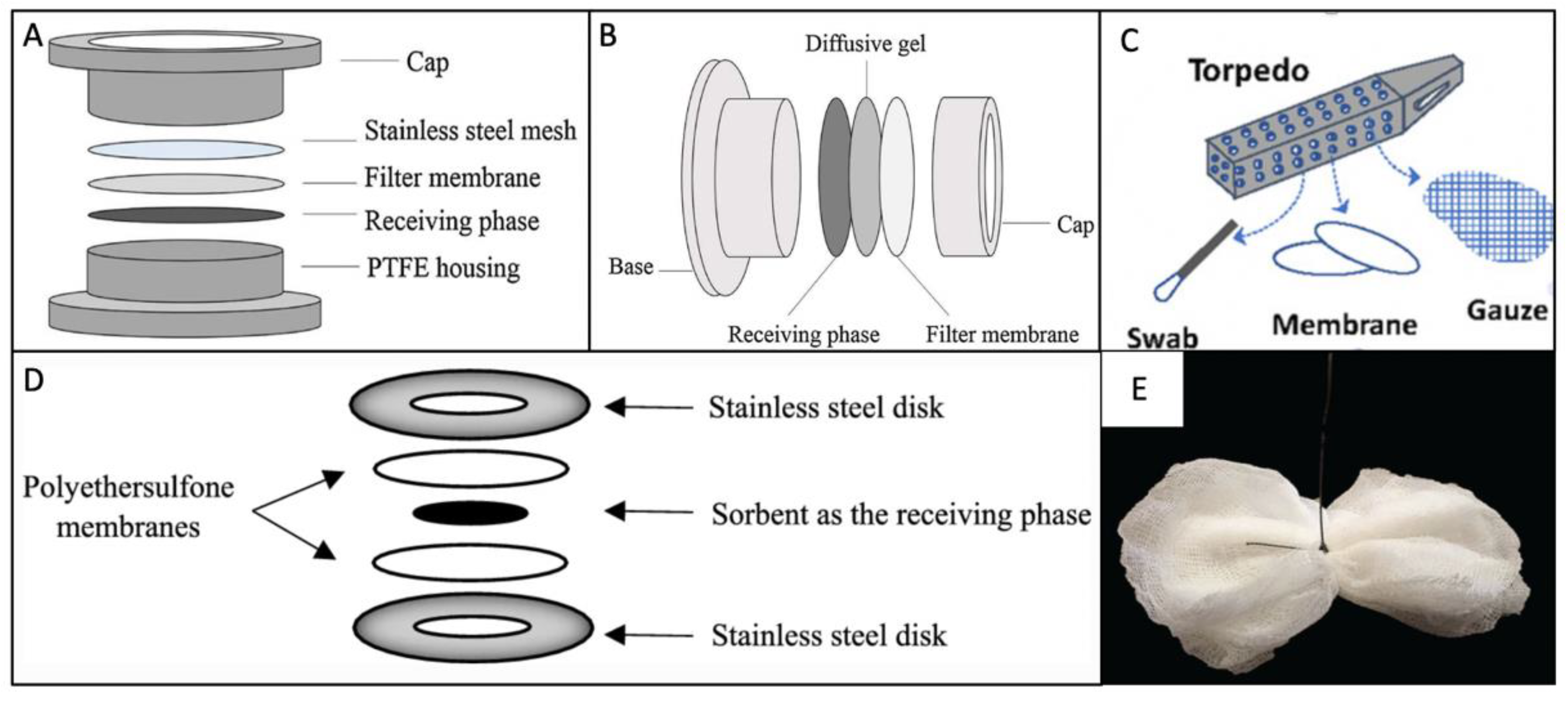
3.2. Passive Sampling Devices Used in Wastewater Surveillance
3.2.1. The Polar Organic Chemical Integrative Sampler (POCIS)
3.2.2. Diffusive Gradients in Thin Films (DGT) and Organic-Diffusive Gradients in Thin Films (o-DGT)
3.2.3. Chemcatcher®
3.2.4. Semipermeable Membrane Device (SPMD)
3.2.5. Other Sampling Devices
3.3. Method Design
3.4. Comparison to Grab Sampling
3.5. Extraction Techniques
3.6. Analysis of Extracts
4. Discussion
4.1. Use of Passive Samplers in Wastewater
4.2. Current Limitations in Passive Sampling
4.3. Sampling Method Design
4.4. Sampling Extraction and Analysis
4.5. Future Studies
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gravell, A.; Fones, G.R.; Greenwood, R.; Mills, G.A. Detection of pharmaceuticals in wastewater effluents-a comparison of the performance of Chemcatcher (R) and polar organic compound integrative sampler. Environ. Sci. Pollut. Res. 2020, 27, 27995–28005. [Google Scholar] [CrossRef] [PubMed]
- McKay, S.; Tscharke, B.; Hawker, D.; Thompson, K.; O’Brien, J.; Mueller, J.F.; Kaserzon, S. Calibration and validation of a microporous polyethylene passive sampler for quantitative estimation of illicit drug and pharmaceutical and personal care product (PPCP) concentrations in wastewater influent. Sci. Total Environ. 2020, 704, 135891. [Google Scholar] [CrossRef] [PubMed]
- Ahkola, H.; Juntunen, J.; Krogerus, K.; Huttula, T.; Herve, S.; Witick, A. Suitability of Chemcatcher® passive sampling in monitoring organotin compounds at a wastewater treatment plant. Environ. Sci. Water Res. Technol. 2016, 2, 769–778. [Google Scholar] [CrossRef]
- Macleod, S.L.; McClure, E.L.; Wong, C.S. Laboratory calibration and field deployment of the polar organic chemical integrative sampler for pharmaceuticals and personal care products in wastewater and surface water. Environ. Toxicol. Chem. 2007, 26, 2517–2529. [Google Scholar] [CrossRef]
- Petrie, B.; Gravell, A.; Mills, G.A.; Youdan, J.; Barden, R.; Kasprzyk-Hordern, B. In situ calibration of a new chemcatcher configuration for the determination of polar organic micropollutants in wastewater effluent. Environ. Sci. Technol. 2016, 50, 9469–9478. [Google Scholar] [CrossRef] [Green Version]
- Sims, N.; Kasprzyk-Hordern, B. Future perspectives of wastewater-based epidemiology: Monitoring infectious disease spread and resistance to the community level. Environ. Int. 2020, 139, 105689. [Google Scholar] [CrossRef]
- Daughton, C.G. Emerging pollutants, and communicating the science of environmental chemistry and mass spectrometry: Pharmaceuticals in the environment. J. Am. Soc. Mass Spectrom. 2001, 12, 1067–1076. [Google Scholar] [CrossRef] [Green Version]
- Aguiar-Oliveira, M.d.L.; Campos, A.; Matos, A.R.; Rigotto, C.; Sotero-Martins, A.; Teixeira, P.F.P.; Siqueira, M.M. Wastewater-based epidemiology (Wbe) and viral detection in polluted surface water: A valuable tool for covid-19 surveillance—A brief review. Int. J. Environ. Res. Public Health 2020, 17, 9251. [Google Scholar] [CrossRef]
- Bartelt-Hunt, S.L.; Snow, D.D.; Damon-Powell, T.; Brown, D.L.; Prasai, G.; Schwarz, M.; Kolok, A.S. Quantitative evaluation of laboratory uptake rates for pesticides, pharmaceuticals, and steroid hormones using POCIS. Environ. Toxicol. Chem. 2011, 30, 1412–1420. [Google Scholar] [CrossRef]
- Boles, T.H.; Wells, M.J.M. Pilot survey of methamphetamine in sewers using a Polar Organic Chemical Integrative Sampler. Sci. Total Environ. 2014, 472, 9–12. [Google Scholar] [CrossRef]
- Bailly, E.; Levi, Y.; Karolak, S. Calibration and field evaluation of polar organic chemical integrative sampler (POCIS) for monitoring pharmaceuticals in hospital wastewater. Environ. Pollut. (1987) 2013, 174, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Trommetter, G.; Dumoulin, D.; Billon, G. Development and validation of DGT passive samplers for the quantification of Ir, Pd, Pt, Rh and Ru: A challenging application in waters impacted by urban activities. Talanta 2021, 223, 121707. [Google Scholar] [CrossRef] [PubMed]
- Aydin, M.E.; Beduk, F.; Aydin, S.; Koyuncu, S.; Genuit, G.; Bahadir, M. Development of biofilm collectors as passive samplers in sewerage systems—A novel wastewater monitoring method. Environ. Sci. Pollut. Res. 2020, 27, 8199–8209. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.E.; Zhang, H.; Ying, G.G.; Jones, K.C. Evidence and Recommendations to Support the Use of a Novel Passive Water Sampler to Quantify Antibiotics in Wastewaters. Environ. Sci. Technol. 2013, 47, 13587–13593. [Google Scholar] [CrossRef]
- Jacquet, R.; Miège, C.; Bados, P.; Schiavone, S.; Coquery, M. Evaluating the polar organic chemical integrative sampler for the monitoring of beta-blockers and hormones in wastewater treatment plant effluents and receiving surface waters. Environ. Toxicol. Chem. 2012, 31, 279–288. [Google Scholar] [CrossRef]
- Stroski, K.M.; Luong, K.H.; Challis, J.K.; Chaves-Barquero, L.G.; Hanson, M.L.; Wong, C.S. Wastewater sources of per- and polyfluorinated alkyl substances (PFAS) and pharmaceuticals in four Canadian Arctic communities. Sci. Total Environ. 2020, 708, 134494. [Google Scholar] [CrossRef]
- Allinson, M.; Kadokami, K.; Shiraishi, F.; Nakajima, D.; Zhang, J.; Knight, A.; Gray, S.R.; Scales, P.J.; Allinson, G. Wastewater recycling in Antarctica: Performance assessment of an advanced water treatment plant in removing trace organic chemicals. J. Environ. Manag. 2018, 224, 122–129. [Google Scholar] [CrossRef]
- Challis, J.K.; Hanson, M.L.; Wong, C.S. Development and Calibration of an Organic-Diffusive Gradients in Thin Films Aquatic Passive Sampler for a Diverse Suite of Polar Organic Contaminants. Anal. Chem. 2016, 88, 10583–10591. [Google Scholar] [CrossRef]
- Tan, B.L.L.; Hawker, D.W.; Müller, J.F.; Leusch, F.D.L.; Tremblay, L.A.; Chapman, H.F. Comprehensive study of endocrine disrupting compounds using grab and passive sampling at selected wastewater treatment plants in South East Queensland, Australia. Environ. Int. 2007, 33, 654–669. [Google Scholar] [CrossRef]
- Schang, C.; Crosbie, N.D.; Nolan, M.; Poon, R.; Wang, M.; Jex, A.; John, N.; Baker, L.; Scales, P.; Schmidt, J.; et al. Passive Sampling of SARS-CoV-2 for Wastewater Surveillance. Environ. Sci. Technol. 2021, 55, 10432–10441. [Google Scholar] [CrossRef]
- Moore, B. The Detection of Enteric Carriers in Towns By Means of Sewage Examination. J. R. Sanit. Inst. 1951, 71, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Sattar, S.A.; Westwood, J.C. Isolation of apparently wild strains of poliovirus type 1 from sewage in the Ottawa area. Can. Med. Assoc. J. 1977, 116, 25–27. [Google Scholar] [PubMed]
- de Melo Cassemiro, K.M.; Burlandy, F.M.; Barbosa, M.R.; Chen, Q.; Jorba, J.; Hachich, E.M.; Sato, M.I.; Burns, C.C.; da Silva, E.E. Molecular and Phenotypic Characterization of a Highly Evolved Type 2 Vaccine-Derived Poliovirus Isolated from Seawater in Brazil, 2014. PLoS ONE 2016, 11, e0152251. [Google Scholar] [CrossRef]
- Knutsson, J.; Rauch, S.; Morrison, G.M. Performance of a passive sampler for the determination of time averaged concentrations of nitrate and phosphate in water. Environ. Sci.-Process. Impacts 2013, 15, 955–962. [Google Scholar] [CrossRef] [Green Version]
- Lis, H.; Stepnowski, P.; Caban, M. Static renewal and continuous-flow calibration of two types of passive samplers for the monitoring of pharmaceuticals in wastewater. Microchem. J. 2021, 165, 106121. [Google Scholar] [CrossRef]
- Habtewold, J.; McCarthy, D.; McBean, E.; Law, I.; Goodridge, L.; Habash, M.; Murphy, H.M. Passive sampling, a practical method for wastewater-based surveillance of SARS-CoV-2. Environ. Res. 2022, 204, 112058. [Google Scholar] [CrossRef]
- Corchis-Scott, R.; Geng, Q.; Seth, R.; Ray, R.; Beg, M.; Biswas, N.; Charron, L.; Drouillard, K.D.; D’Souza, R.; Heath, D.D.; et al. Averting an outbreak of SARS-CoV-2 in a university residence hall through wastewater surveillance. Microbiol. Spectr. 2021, 9, e00792-21. [Google Scholar] [CrossRef]
- Hayes, E.K.; Sweeney, C.L.; Anderson, L.E.; Li, B.; Erjavec, G.B.; Gouthro, M.T.; Krkosek, W.H.; Stoddart, A.K.; Gagnon, G.A. A novel passive sampling approach for SARS-CoV-2 in wastewater in a Canadian province with low prevalence of COVID-19. Environ. Sci. Water Res. Technol. 2021, 7, 1576–1586. [Google Scholar] [CrossRef]
- Hayes, E.K.; Stoddart, A.K.; Gagnon, G.A. Adsorption of SARS-CoV-2 onto granular activated carbon (GAC) in wastewater: Implications for improvements in passive sampling. Sci. Total Environ. 2022, 847, 157548. [Google Scholar] [CrossRef]
- Hayes, E.K.; Sweeney, C.L.; Fuller, M.; Erjavec, G.B.; Stoddart, A.K.; Gagnon, G.A. Operational Constraints of Detecting SARS-CoV-2 on Passive Samplers using Electronegative Filters: A Kinetic and Equilibrium Analysis. ACS EST Water 2022. [Google Scholar] [CrossRef]
- Kevill, J.L.; Lambert-Slosarska, K.; Pellett, C.; Woodhall, N.; Richardson-O’Neill, I.; Pântea, I.; Alex-Sanders, N.; Farkas, K.; Jones, D.L. Assessment of two types of passive sampler for the efficient recovery of SARS-CoV-2 and other viruses from wastewater. Sci. Total Environ. 2022, 838, 156580. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ahmed, W.; Metcalfe, S.; Smith, W.J.M.; Tscharke, B.; Lynch, P.; Sherman, P.; Vo, P.H.N.; Kaserzon, S.L.; Simpson, S.L.; et al. Monitoring of SARS-CoV-2 in sewersheds with low COVID-19 cases using a passive sampling technique. Water Res. 2022, 218, 118481. [Google Scholar] [CrossRef] [PubMed]
- Vincent-Hubert, F.; Wacrenier, C.; Desdouits, M.; Jousse, S.; Schaeffer, J.; Le Mehaute, P.; Nakache-Danglot, F.; Guyader, F.S.L.; Bertrand, I.; Boni, M.; et al. Development of passive samplers for the detection of SARS-CoV-2 in sewage and seawater: Application for the monitoring of sewage. Sci. Total Environ. 2022, 833, 155139. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.; Qiu, Y.; Yu, J.; Lee, B.E.; McCarthy, D.T.; Pang, X. Comparison of Auto Sampling and Passive Sampling Methods for SARS-CoV-2 Detection in Wastewater. Pathogens 2022, 11, 359. [Google Scholar] [CrossRef]
- Münze, R.; Hannemann, C.; Orlinskiy, P.; Gunold, R.; Paschke, A.; Foit, K.; Becker, J.; Kaske, O.; Paulsson, E.; Peterson, M.; et al. Pesticides from wastewater treatment plant effluents affect invertebrate communities. Sci. Total Environ. 2017, 599–600, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Vincent-Hubert, F.; Morga, B.; Renault, T.; Le Guyader, F.S. Adsorption of norovirus and ostreid herpesvirus type 1 to polymer membranes for the development of passive samplers. J. Appl. Microbiol. 2017, 122, 1039–1047. [Google Scholar] [CrossRef] [Green Version]
- Addeck, A.; Croes, K.; Van Langenhove, K.; Denison, M.S.; Afify, A.S.; Gao, Y.; Elskens, M.; Baeyens, W. Time-integrated monitoring of dioxin-like polychlorinated biphenyls (dl-PCBs) in aquatic environments using the ceramic toximeter and the CALUX bioassay. Talanta 2014, 120, 413–418. [Google Scholar] [CrossRef] [Green Version]
- Chandler, D.M.; Lerner, D.N. A low cost method to detect polluted surface water outfalls and misconnected drainage. Water Environ. J. 2015, 29, 202–206. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Pan, S.; Cheng, H.; Sweetman, A.J.; Zhang, H.; Jones, K.C. Diffusive gradients in thin-films (DGT) for in situ sampling of selected endocrine disrupting chemicals (EDCs) in waters. Water Res. 2018, 137, 211–219. [Google Scholar] [CrossRef] [Green Version]
- Fang, Z.; Li, Y.; Li, Y.; Yang, D.; Zhang, H.; Jones, K.C.; Gu, C.; Luo, J. Development and Applications of Novel DGT Passive Samplers for Measuring 12 Per- And Polyfluoroalkyl Substances in Natural Waters and Wastewaters. Environ. Sci. Technol. 2021, 55, 9548–9556. [Google Scholar] [CrossRef]
- Garcia-Rodriguez, A.; Fontas, C.; Matamoros, V.; Almeida, M.; Cattrall, R.W.; Kolev, S.D. Development of a polymer inclusion membrane-based passive sampler for monitoring of sulfamethoxazole in natural waters. Minimizing the effect of the flow pattern of the aquatic system. Microchem. J. 2016, 124, 175–180. [Google Scholar] [CrossRef]
- Guan, D.X.; Li, Y.Q.; Yu, N.Y.; Yu, G.H.; Wei, S.; Zhang, H.; Davison, W.; Cui, X.Y.; Ma, L.Q.; Luo, J. In situ measurement of perfluoroalkyl substances in aquatic systems using diffusive gradients in thin-films technique. Water Res. 2018, 144, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Van Langenhove, K.; Denison, M.S.; Baeyens, W.; Elskens, M.; Gao, Y. Estrogenic Activity Measurements in Water Using Diffusive Gradients in Thin-Film Coupled with an Estrogen Bioassay. Anal. Chem. 2017, 89, 13357–13364. [Google Scholar] [CrossRef] [PubMed]
- Li, H.X.; Vermeirssen, E.L.M.; Helm, P.A.; Metcalfe, C.D. Controlled field evaluation of water flow rate effects on sampling polar organic compounds using polar organic chemical integrative samplers. Environ. Toxicol. Chem. 2010, 29, 2461–2469. [Google Scholar] [CrossRef]
- Mutzner, L.; Vermeirssen, E.L.M.; Ort, C. Passive samplers in sewers and rivers with highly fluctuating micropollutant concentrations—Better than we thought. J. Hazard. Mater. 2019, 361, 312–320. [Google Scholar] [CrossRef] [Green Version]
- Nyoni, H.; Chimuka, L.; Vrana, B.; Cukrowska, E.; Tutu, H. Optimisation of the membrane-assisted passive sampler and its comparison with solid phase extraction technique. Water SA 2010, 36, 501–508. [Google Scholar] [CrossRef] [Green Version]
- Posada-Ureta, O.; Olivares, M.; Zatón, L.; Delgado, A.; Prieto, A.; Vallejo, A.; Paschke, A.; Etxebarria, N. Uptake calibration of polymer-based passive samplers for monitoring priority and emerging organic non-polar pollutants in WWTP effluents. Anal. Bioanal. Chem. 2016, 408, 3165–3175. [Google Scholar] [CrossRef]
- Ren, S.Y.; Tao, J.; Tan, F.; Cui, Y.; Li, X.N.; Chen, J.W.; He, X.; Wang, Y. Diffusive gradients in thin films based on MOF-derived porous carbon binding gel for in-situ measurement of antibiotics in waters. Sci. Total Environ. 2018, 645, 482–490. [Google Scholar] [CrossRef]
- Rujiralai, T.; Bull, I.D.; Llewellyn, N.; Evershed, R.P. In situ polar organic chemical integrative sampling (POCIS) of steroidal estrogens in sewage treatment works discharge and river water. J. Environ. Monit. 2011, 13, 1427–1434. [Google Scholar] [CrossRef]
- Vermeirssen, E.L.M.; Asmin, J.; Escher, B.I.; Kwon, J.H.; Steimen, I.; Hollender, J. The role of hydrodynamics, matrix and sampling duration in passive sampling of polar compounds with Empore (TM) SDB-RPS disks. J. Environ. Monit. 2008, 10, 119–128. [Google Scholar] [CrossRef]
- Wang, L.; Gong, X.; Wang, R.; Gan, Z.; Lu, Y.; Sun, H. Application of an immobilized ionic liquid for the passive sampling of perfluorinated substances in water. J. Chromatogr. A 2017, 1515, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Hibberd, A.; Zhou, J.L. Analysis of emerging contaminants in sewage effluent and river water: Comparison between spot and passive sampling. Anal. Chim. Acta 2008, 607, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.T.; Fang, Z.; Li, Y.; Wang, R.M.; Zhang, H.; Jones, K.C.; Cui, X.Y.; Shi, X.Y.; Yin, D.X.; Li, C.; et al. Novel Method for in Situ Monitoring of Organophosphorus Flame Retardants in Waters. Anal. Chem. 2018, 90, 10016–10023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Challis, J.K.; Almirall, X.O.; Helm, P.A.; Wong, C.S. Performance of the organic-diffusive gradients in thin-films passive sampler for measurement of target and suspect wastewater contaminants. Environ. Pollut. 2020, 261, 114092. [Google Scholar] [CrossRef] [PubMed]
- De Baat, M.L.; Van der Oost, R.; Van der Lee, G.H.; Wieringa, N.; Hamers, T.; Verdonschot, P.F.M.; De Voogt, P.; Kraak, M.H.S. Advancements in effect-based surface water quality assessment. Water Res. 2020, 183, 116017. [Google Scholar] [CrossRef]
- Jarosova, B.; Blaha, L.; Vrana, B.; Randak, T.; Grabic, R.; Giesy, J.P.; Hilscherova, K. Changes in concentrations of hydrophilic organic contaminants and of endocrine-disrupting potential downstream of small communities located adjacent to headwaters. Environ. Int. 2012, 45, 22–31. [Google Scholar] [CrossRef]
- Sultana, T.; Murray, C.; Ehsanul Hoque, M.; Metcalfe, C.D. Monitoring contaminants of emerging concern from tertiary wastewater treatment plants using passive sampling modelled with performance reference compounds. Environ. Monit. Assess. 2016, 189, 1. [Google Scholar] [CrossRef]
- Vrana, B.; Rusina, T.; Okonski, K.; Prokeš, R.; Carlsson, P.; Kopp, R.; Smedes, F. Chasing equilibrium passive sampling of hydrophobic organic compounds in water. Sci. Total Environ. 2019, 664, 424–435. [Google Scholar] [CrossRef]
- Gallé, T.; Koehler, C.; Plattes, M.; Pittois, D.; Bayerle, M.; Carafa, R.; Christen, A.; Hansen, J. Large-scale determination of micropollutant elimination from municipal wastewater by passive sampling gives new insights in governing parameters and degradation patterns. Water Res. 2019, 160, 380–393. [Google Scholar] [CrossRef]
- Ganser, B.; Bundschuh, M.; Werner, I.; Homazava, N.; Vermeirssen, E.L.M.; Moschet, C.; Kienle, C. Wastewater alters feeding rate but not vitellogenin level of Gammarus fossarum (Amphipoda). Sci. Total Environ. 2019, 657, 1246–1252. [Google Scholar] [CrossRef]
- Iparraguirre, A.; Prieto, A.; Vallejo, A.; Moeder, M.; Zuloaga, O.; Etxebarria, N.; Paschke, A. Tetraphasic polar organic chemical integrative sampler for the determination of a wide polarity range organic pollutants in water. The use of performance reference compounds and in-situ calibration. Talanta 2017, 164, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.; Schäffer, A.; Smith, K. A comparison of equilibrium and kinetic passive sampling for the monitoring of aquatic organic contaminants in German rivers. Water Res. 2018, 145, 248–258. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.; Yan, Q.; Xiong, J.; Li, H.; Ma, X.; You, J. Point or non-point source: Toxicity evaluation using m-POCIS and zebrafish embryos in municipal sewage treatment plants and urban waterways. Environ. Pollut. 2022, 292, 118307. [Google Scholar] [CrossRef] [PubMed]
- Hamers, T.; Legradi, J.; Zwart, N.; Smedes, F.; de Weert, J.; van den Brandhof, E.J.; van de Meent, D.; de Zwart, D. Time-Integrative Passive sampling combined with TOxicity Profiling (TIPTOP): An effect-based strategy for cost-effective chemical water quality assessment. Environ. Toxicol. Pharmacol. 2018, 64, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Jálová, V.; Jarošová, B.; Bláha, L.; Giesy, J.P.; Ocelka, T.; Grabic, R.; Jurčíková, J.; Vrana, B.; Hilscherová, K. Estrogen-, androgen- and aryl hydrocarbon receptor mediated activities in passive and composite samples from municipal waste and surface waters. Environ. Int. 2013, 59, 372–383. [Google Scholar] [CrossRef]
- Ouyang, X.; Leonards, P.; Legler, J.; van der Oost, R.; de Boer, J.; Lamoree, M. Comprehensive two-dimensional liquid chromatography coupled to high resolution time of flight mass spectrometry for chemical characterization of sewage treatment plant effluents. J. Chromatogr. A 2015, 1380, 139–145. [Google Scholar] [CrossRef]
- Baz-Lomba, J.A.; Reid, M.J.; Thomas, K.V. Target and suspect screening of psychoactive substances in sewage-based samples by UHPLC-QTOF. Anal. Chim. Acta 2016, 914, 81–90. [Google Scholar] [CrossRef] [Green Version]
- Cristovao, M.B.; Tela, S.; Silva, A.F.; Oliveira, M.; Bento-Silva, A.; Bronze, M.R.; Crespo, M.T.B.; Crespo, J.G.; Nunes, M.; Pereira, V.J. Occurrence of Antibiotics, Antibiotic Resistance Genes and Viral Genomes in Wastewater Effluents and Their Treatment by a Pilot Scale Nanofiltration Unit. Membranes 2021, 11, 9. [Google Scholar] [CrossRef]
- Hoque, M.E.; Cloutier, F.; Arcieri, C.; McInnes, M.; Sultana, T.; Murray, C.; Vanrolleghem, P.A.; Metcalfe, C.D. Removal of selected pharmaceuticals, personal care products and artificial sweetener in an aerated sewage lagoon. Sci. Total Environ. 2014, 487, 801–812. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, R.; Cheng, H.; Khorram, M.S.; Zhao, S.; Tham, T.T.; Tran, T.M.; Minh, T.B.; Jiang, B.; Jin, B.; et al. Field evaluation of diffusive gradients in thin-film passive samplers for wastewater-based epidemiology. Sci. Total Environ. 2021, 773, 145480. [Google Scholar] [CrossRef]
- MacLeod, S.L.; Wong, C.S. Loadings, trends, comparisons, and fate of achiral and chiral pharmaceuticals in wastewaters from urban tertiary and rural aerated lagoon treatments. Water Res. 2010, 44, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Turja, R.; Lehtonen, K.K.; Meierjohann, A.; Brozinski, J.M.; Vahtera, E.; Soirinsuo, A.; Sokolov, A.; Snoeijs, P.; Budzinski, H.; Devier, M.H.; et al. The mussel caging approach in assessing biological effects of wastewater treatment plant discharges in the Gulf of Finland (Baltic Sea). Mar. Pollut. Bull. 2014, 97, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Franquet-Griell, H.; Pueyo, V.; Silva, J.; Orera, V.M.; Lacorte, S. Development of a macroporous ceramic passive sampler for the monitoring of cytostatic drugs in water. Chemosphere 2017, 182, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Cristóvão, M.B.; Bento-Silva, A.; Bronze, M.R.; Crespo, J.G.; Pereira, V.J. Detection of anticancer drugs in wastewater effluents: Grab versus passive sampling. Sci. Total Environ. 2021, 786, 147477. [Google Scholar] [CrossRef] [PubMed]
- Cukrowska, E.; Chimuka, L.; Amdany, R. Determination of naproxen, ibuprofen and triclosan in wastewater using the polar organic chemical integrative sampler (POCIS): A laboratory calibration and field application. Water SA 2014, 40, 407–414. [Google Scholar] [CrossRef] [Green Version]
- De Vargas, J.P.R.; Bastos, M.C.; Al Badany, M.; Gonzalez, R.; Wolff, D.; Santos, D.R.D.; Labanowski, J. Pharmaceutical compound removal efficiency by a small constructed wetland located in south Brazil. Environ. Sci. Pollut. Res. Int. 2021, 28, 30955–30974. [Google Scholar] [CrossRef]
- Fedorova, G.; Randak, T.; Golovko, O.; Kodes, V.; Grabicova, K.; Grabic, R. A passive sampling method for detecting analgesics, psycholeptics, antidepressants and illicit drugs in aquatic environments in the Czech Republic. Sci. Total Environ. 2014, 487, 681–687. [Google Scholar] [CrossRef]
- Balaam, J.L.; Grover, D.; Johnson, A.C.; Jürgens, M.; Readman, J.; Smith, A.J.; White, S.; Williams, R.; Zhou, J.L. The use of modelling to predict levels of estrogens in a river catchment: How does modelled data compare with chemical analysis and in vitro yeast assay results? Sci. Total Environ. 2010, 408, 4826–4832. [Google Scholar] [CrossRef]
- Challis, J.K.; Stroski, K.M.; Luong, K.H.; Hanson, M.L.; Wong, C.S. Field Evaluation and in Situ Stress Testing of the Organic-Diffusive Gradients in Thin-Films Passive Sampler. Environ. Sci. Technol. 2018, 52, 12573–12582. [Google Scholar] [CrossRef]
- Guo, W.; Van Langenhove, K.; Vandermarken, T.; Denison, M.S.; Elskens, M.; Baeyens, W.; Gao, Y. In situ measurement of estrogenic activity in various aquatic systems using organic diffusive gradients in thin-film coupled with ERE-CALUX bioassay. Environ. Int. 2019, 127, 13–20. [Google Scholar] [CrossRef]
- Liscio, C.; Magi, E.; Di Carro, M.; Suter, M.J.F.; Vermeirssen, E.L.M. Combining passive samplers and biomonitors to evaluate endocrine disrupting compounds in a wastewater treatment plant by LC/MS/MS and bioassay analyses. Environ. Pollut. 2009, 157, 2716–2721. [Google Scholar] [CrossRef] [PubMed]
- Škodová, A.; Prokeš, R.; Šimek, Z.; Vrana, B. In situ calibration of three passive samplers for the monitoring of steroid hormones in wastewater. Talanta 2016, 161, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Vallejo, A.; Prieto, A.; Moeder, M.; Usobiaga, A.; Zuloaga, O.; Etxebarria, N.; Paschke, A. Calibration and field test of the Polar Organic Chemical Integrative Samplers for the determination of 15 endocrine disrupting compounds in wastewater and river water with special focus on performance reference compounds (PRC). Water Res. 2013, 47, 2851–2862. [Google Scholar] [CrossRef] [PubMed]
- Vermeirssen, E.L.M.; Eggen, R.I.L.; Escher, B.I.; Suter, M.J.F. Estrogens in Swiss rivers and effluents—Sampling matters. Chimia 2008, 62, 389–394. [Google Scholar] [CrossRef] [Green Version]
- Bishop, N.; Jones-Lepp, T.; Margetts, M.; Sykes, J.; Alvarez, D.; Keil, D.E. Wastewater-based epidemiology pilot study to examine drug use in the Western United States. Sci Total Environ. 2020, 745, 140697. [Google Scholar] [CrossRef] [PubMed]
- Gallé, T.; Bayerle, M.; Pittois, D.; Huck, V. Allocating biocide sources and flow paths to surface waters using passive samplers and flood wave chemographs. Water Res. 2020, 173, 115533. [Google Scholar] [CrossRef]
- Mutzner, L.; Bohren, C.; Mangold, S.; Bloem, S.; Ort, C. Spatial Differences among Micropollutants in Sewer Overflows: A Multisite Analysis Using Passive Samplers. Environ. Sci. Technol. 2020, 54, 6584–6593. [Google Scholar] [CrossRef]
- Tlili, A.; Hollender, J.; Kienle, C.; Behra, R. Micropollutant-induced tolerance of in situ periphyton: Establishing causality in wastewater-impacted streams. Water Res. 2017, 111, 185–194. [Google Scholar] [CrossRef]
- Dixon-Anderson, E.; Lohmann, R. Field-testing polyethylene passive samplers for the detection of neutral polyfluorinated alkyl substances in air and water. Environ. Toxicol. Chem. 2018, 37, 3002–3010. [Google Scholar] [CrossRef]
- Gardiner, C.; Robuck, A.; Becanova, J.; Cantwell, M.; Kaserzon, S.; Katz, D.; Mueller, J.; Lohmann, R. Field Validation of a Novel Passive Sampler for Dissolved PFAS in Surface Waters. Environ. Toxicol. Chem. 2022, 41, 2375–2385. [Google Scholar] [CrossRef]
- Xu, Y.; Qing, D.; Xie, R.; Zhu, F.; Gao, X.; Rao, K.; Ma, M.; Wang, Z. Integrated passive sampling and fugacity model to characterize fate and removal of organophosphate flame retardants in an anaerobic-anoxic-oxic municipal wastewater treatment system. J. Hazard. Mater. 2022, 424, 127288. [Google Scholar] [CrossRef]
- Moore, B.; Perry, E.L.; Chard, S.T. A survey by the sewage swab method of latent enteric infection in an urban area. J. Hyg. 1952, 50, 137–156. [Google Scholar] [CrossRef] [Green Version]
- Bloom, H.H.; Mack, W.N.; Mallmann, W.L. Enteric Viruses and Salmonellae Isolation: II. Media Comparison for Salmonellae. Sew. Ind. Wastes 1958, 30, 1455–1460. [Google Scholar]
- Greenberg, A.E.; Wickenden, R.W.; Lee, T.W. Tracing typhoid carriers by means of sewage. Sew. Ind. Wastes 1957, 29, 1237–1242. [Google Scholar]
- Jones, A.C. A hospital outbreak of typhoid fever bacteriological and serological investigations. J. Hyg. 1951, 49, 335–348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, S.M.; Clark, M.E.; Coleman, M.B. Demonstration of infectious agents in sewage. Am. J. Public Health Nations Health 1955, 45, 1438–1446. [Google Scholar] [CrossRef]
- Sears, S.D.; Ferreccio, C.; Levine, M.M. Sensitivity of Moore sewer swabs for isolating Salmonella typhi. Appl. Environ. Microbiol. 1986, 51, 425–426. [Google Scholar] [CrossRef] [Green Version]
- Shearer, L.A.; Browne, A.S.; Gordon, R.B.; Hollister, A.C., Jr. Discovery of typhoid carrier by sewage sampling. J. Am. Med. Assoc. 1959, 169, 1051–1055. [Google Scholar] [CrossRef]
- Robinson, R.G. The isolation of enteric organisms from sewage and the development of the sewage pad technique. J. Med. Lab. Technol. 1958, 15, 79–95. [Google Scholar]
- Barrett, T.J.; Blake, P.A.; Morris, G.K.; Puhr, N.D.; Bradford, H.B.; Wells, J.G. Use of Moore swabs for isolating Vibrio cholerae from sewage. J. Clin. Microbiol. 1980, 11, 385–388. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Verhagen, R.; Ahmed, W.; Metcalfe, S.; Thai, P.K.; Kaserzon, S.L.; Smith, W.J.M.; Schang, C.; Simpson, S.L.; Thomas, K.V.; et al. In Situ Calibration of Passive Samplers for Viruses in Wastewater. ACS EST Water 2022. [Google Scholar] [CrossRef]
- Rafiee, M.; Isazadeh, S.; Mohseni-Bandpei, A.; Mohebbi, S.R.; Jahangiri-rad, M.; Eslami, A.; Dabiri, H.; Roostaei, K.; Tanhaei, M.; Amereh, F. Moore swab performs equal to composite and outperforms grab sampling for SARS-CoV-2 monitoring in wastewater. Sci. Total Environ. 2021, 790, 148205. [Google Scholar] [CrossRef] [PubMed]
- Alygizakis, N.A.; Urík, J.; Beretsou, V.G.; Kampouris, I.; Galani, A.; Oswaldova, M.; Berendonk, T.; Oswald, P.; Thomaidis, N.S.; Slobodnik, J.; et al. Evaluation of chemical and biological contaminants of emerging concern in treated wastewater intended for agricultural reuse. Environ. Int. 2020, 138, 105597. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, C.D.; Sultana, T.; Martin, J.; Newman, K.; Helm, P.; Kleywegt, S.; Shen, L.; Yargeau, V. Silver near municipal wastewater discharges into western Lake Ontario, Canada. Environ. Monit. Assess. 2018, 190, 55. [Google Scholar] [CrossRef]
- Liu, S.S.; Cai, Q.S.; Li, C.L.; Cheng, S.M.; Wang, Z.N.; Yang, Y.N.; Ying, G.G.; Sweetman, A.J.; Chen, C.E. In situ measurement of an emerging persistent, mobile and toxic (PMT) substance—Melamine and related triazines in waters by diffusive gradient in thin-films. Water Res. 2021, 206, 117752. [Google Scholar] [CrossRef]
- Liu, S.S.; Chen, S.B.; Li, X.H.; Yue, Y.B.; Li, J.L.; Williams, P.N.; Wang, Z.Y.; Li, C.L.; Yang, Y.Y.; Ying, G.G.; et al. Development and application of diffusive gradients in thin-films for in situ sampling of the bitterest chemical-denatonium benzoate in waters. J. Hazard. Mater. 2021, 418, 126393. [Google Scholar] [CrossRef]
- Chen, W.; Li, Y.; Chen, C.-E.; Sweetman, A.J.; Zhang, H.; Jones, K.C. DGT Passive Sampling for Quantitative in Situ Measurements of Compounds from Household and Personal Care Products in Waters. Environ. Sci. Technol. 2017, 51, 13274–13281. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.-S.; Li, J.-L.; Ge, L.-K.; Li, C.-L.; Zhao, J.-L.; Zhang, Q.-Q.; Ying, G.-G.; Chen, C.-E. Selective diffusive gradients in thin-films with molecularly imprinted polymer for measuring fluoroquinolone antibiotics in waters. Sci. Total Environ. 2021, 790, 148194. [Google Scholar] [CrossRef]
- Vermeirssen, E.L.; Hollender, J.; Bramaz, N.; Van Der Voet, J.; Escher, B.I. Linking toxicity in algal and bacterial assays with chemical analysis in passive samplers deployed in 21 treated sewage effluents. Environ. Toxicol. Chem. 2010, 29, 2575–2582. [Google Scholar] [CrossRef]
- Gong, X.; Li, K.; Wu, C.; Wang, L.; Sun, H. Passive sampling for monitoring polar organic pollutants in water by three typical samplers. Trends Environ. Anal. Chem. 2018, 17, 23–33. [Google Scholar] [CrossRef]
- Godlewska, K.; Stepnowski, P.; Paszkiewicz, M. Pollutant analysis using passive samplers: Principles, sorbents, calibration and applications. A review. Environ. Chem. Lett. 2021, 19, 465–520. [Google Scholar] [CrossRef]
- Sikorski, M.J.; Levine, M.M. Reviving the Moore Swab: A Classic Environmental Surveillance Tool Involving Filtration of Flowing Surface Water and Sewage Water To Recover Typhoidal Salmonella Bacteria. Appl. Environ. Microbiol. 2020, 86, e00060-20. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hou, D.; Qi, S.; O’Connor, D.; Luo, J. High stress low-flow (HSLF) sampling: A newly proposed groundwater purge and sampling approach. Sci. Total Environ. 2019, 664, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.; Zamyadi, A.; Deere, D.; Vanrolleghem, P.A.; Crosbie, N.D. SARS-CoV-2 known and unknowns, implications for the water sector and wastewater-based epidemiology to support national responses worldwide: Early review of global experiences with the COVID-19 pandemic. Water Qual. Res. J. 2021, 56, 57–67. [Google Scholar] [CrossRef]
- Aymerich, I.; Acuña, V.; Ort, C.; Rodríguez-Roda, I.; Corominas, L. Fate of organic microcontaminants in wastewater treatment and river systems: An uncertainty assessment in view of sampling strategy, and compound consumption rate and degradability. Water Res. 2017, 125, 152–161. [Google Scholar] [CrossRef] [Green Version]
- Jones, L.; Ronan, J.; McHugh, B.; Regan, F. Passive sampling of polar emerging contaminants in Irish catchments. Water Sci. Technol. 2019, 79, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Zhu, P.; Fan, H.; Piao, S.; Xu, L.; Sun, T. Effect of Biofilm on Passive Sampling of Dissolved Orthophosphate Using the Diffusive Gradients in Thin Films Technique. Anal. Chem. 2016, 88, 6836–6843. [Google Scholar] [CrossRef]
- Taylor, A.C.; Fones, G.R.; Vrana, B.; Mills, G.A. Applications for Passive Sampling of Hydrophobic Organic Contaminants in Water—A Review. Crit. Rev. Anal. Chem. 2021, 51, 20–54. [Google Scholar] [CrossRef]
- Vincent-Hubert, F.; Wacrenier, C.; Morga, B.; Lozach, S.; Quenot, E.; Mège, M.; Lecadet, C.; Gourmelon, M.; Hervio-Heath, D.; Le Guyader, F.S. Passive Samplers, a Powerful Tool to Detect Viruses and Bacteria in Marine Coastal Areas. Front. Microbiol. 2021, 12, 631174. [Google Scholar] [CrossRef]
- Wang, P.; Challis, J.K.; He, Z.X.; Wong, C.S.; Zeng, E.Y. Effects of biofouling on the uptake of perfluorinated alkyl acids by organic-diffusive gradients in thin films passive samplers. Environ. Sci. Process. Impacts 2022, 24, 242–251. [Google Scholar] [CrossRef]
- Mechelke, J.; Vermeirssen, E.L.M.; Hollender, J. Passive sampling of organic contaminants across the water-sediment interface of an urban stream. Water Res. 2019, 165, 114966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elkayar, K.; Park, J.-A.; Pineda, M.; Westlund, P.; Yargeau, V. Passive sampling and in vitro assays to monitor antiandrogens in a river affected by wastewater discharge. Sci. Total Environ. 2022, 804, 150067. [Google Scholar] [CrossRef] [PubMed]
| Search Terms Used to Identify Relevant Articles |
|---|
| “passive sample **” AND Wastewater OR sewage AND Exclude “reviews” |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shakallis, A.G.; Fallowfield, H.; Ross, K.E.; Whiley, H. The Application of Passive Sampling Devices in Wastewater Surveillance. Water 2022, 14, 3478. https://doi.org/10.3390/w14213478
Shakallis AG, Fallowfield H, Ross KE, Whiley H. The Application of Passive Sampling Devices in Wastewater Surveillance. Water. 2022; 14(21):3478. https://doi.org/10.3390/w14213478
Chicago/Turabian StyleShakallis, Andreana G., Howard Fallowfield, Kirstin E. Ross, and Harriet Whiley. 2022. "The Application of Passive Sampling Devices in Wastewater Surveillance" Water 14, no. 21: 3478. https://doi.org/10.3390/w14213478






