Thermally Enhanced Biodegradation of TCE in Groundwater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material Characterization
2.2. Monitoring
2.3. Lab Test Set-Up
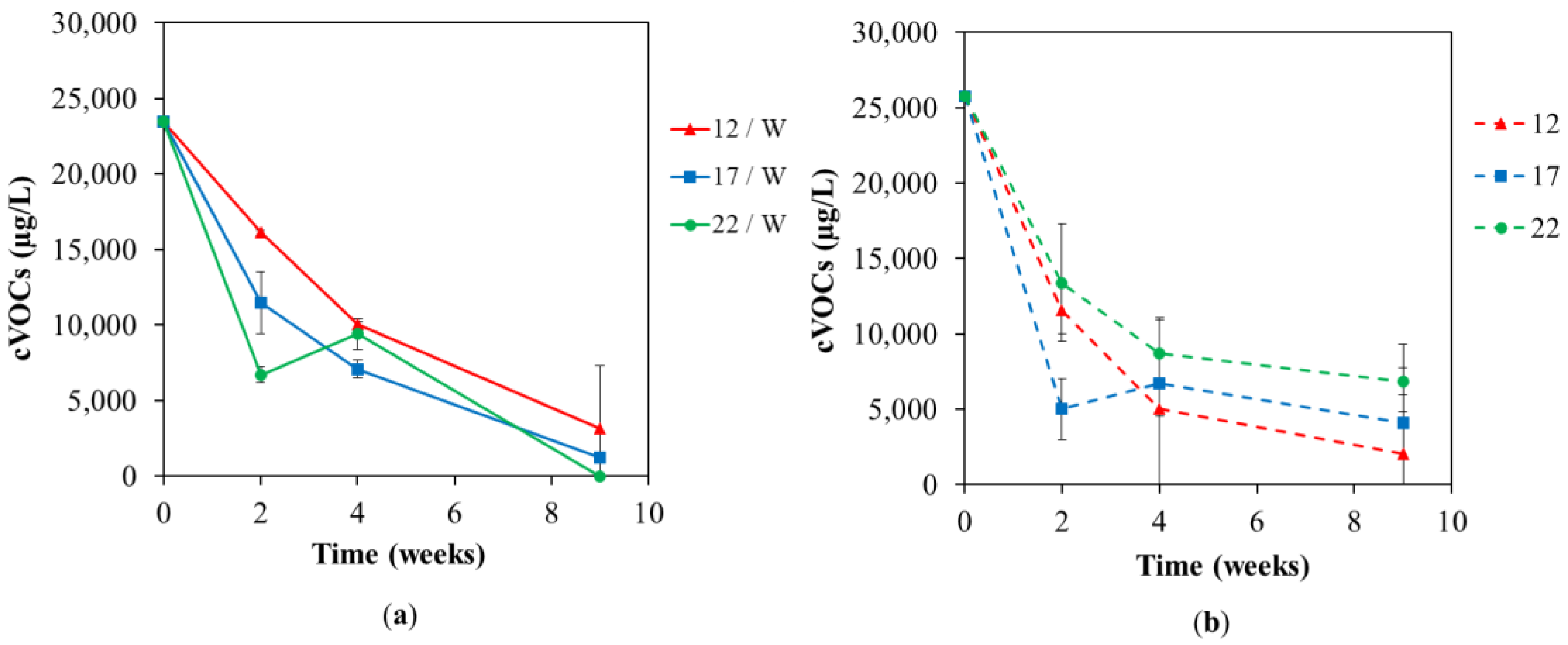
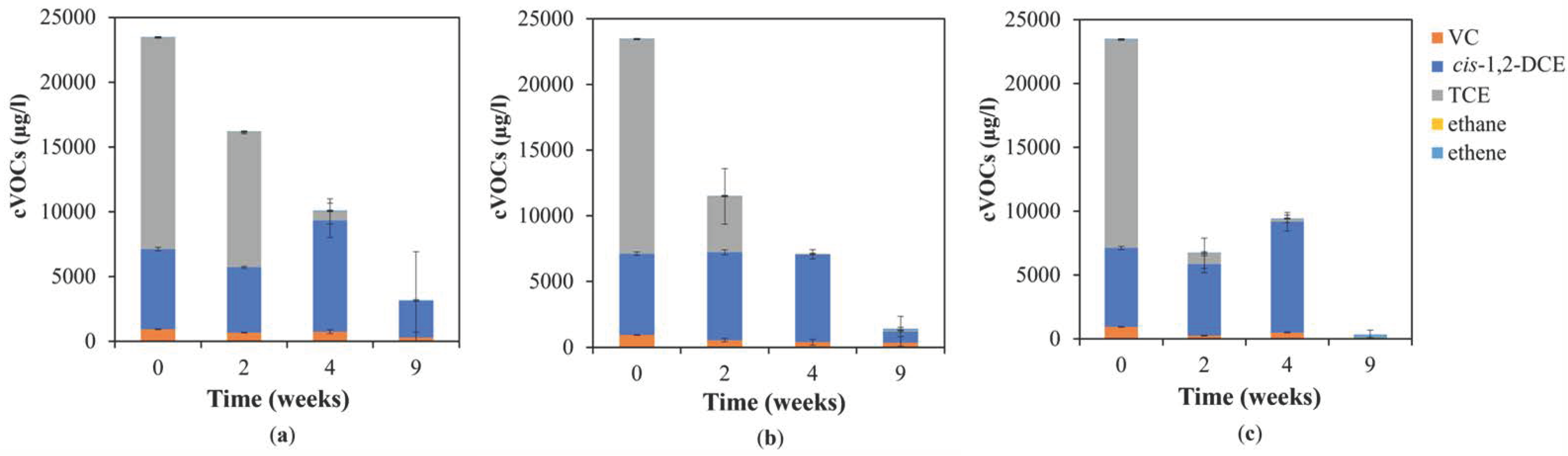
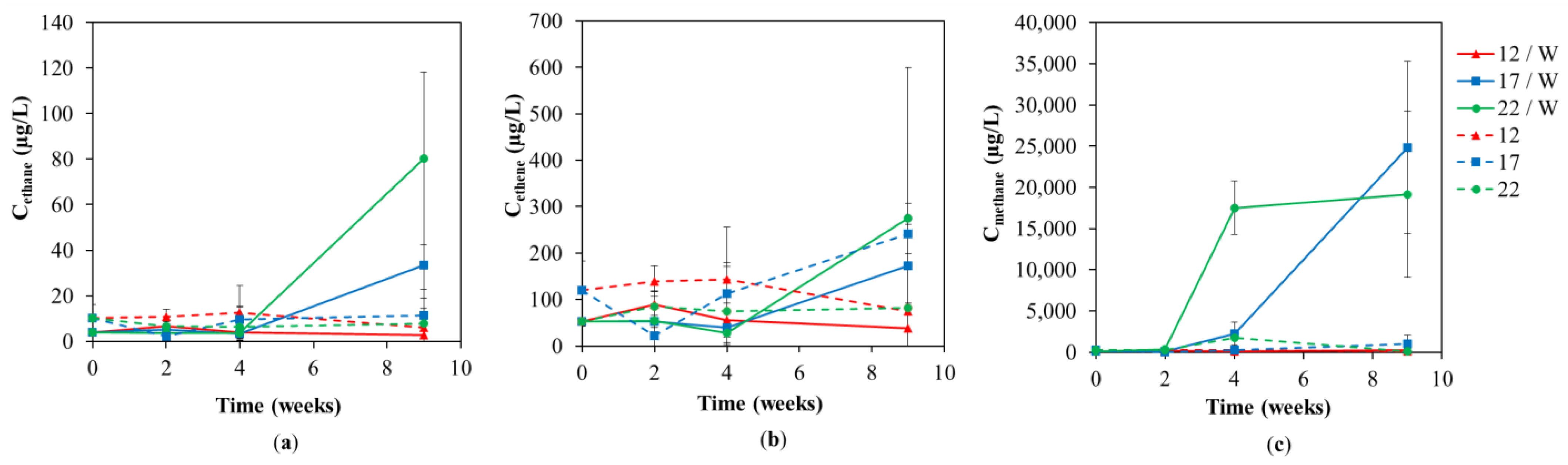
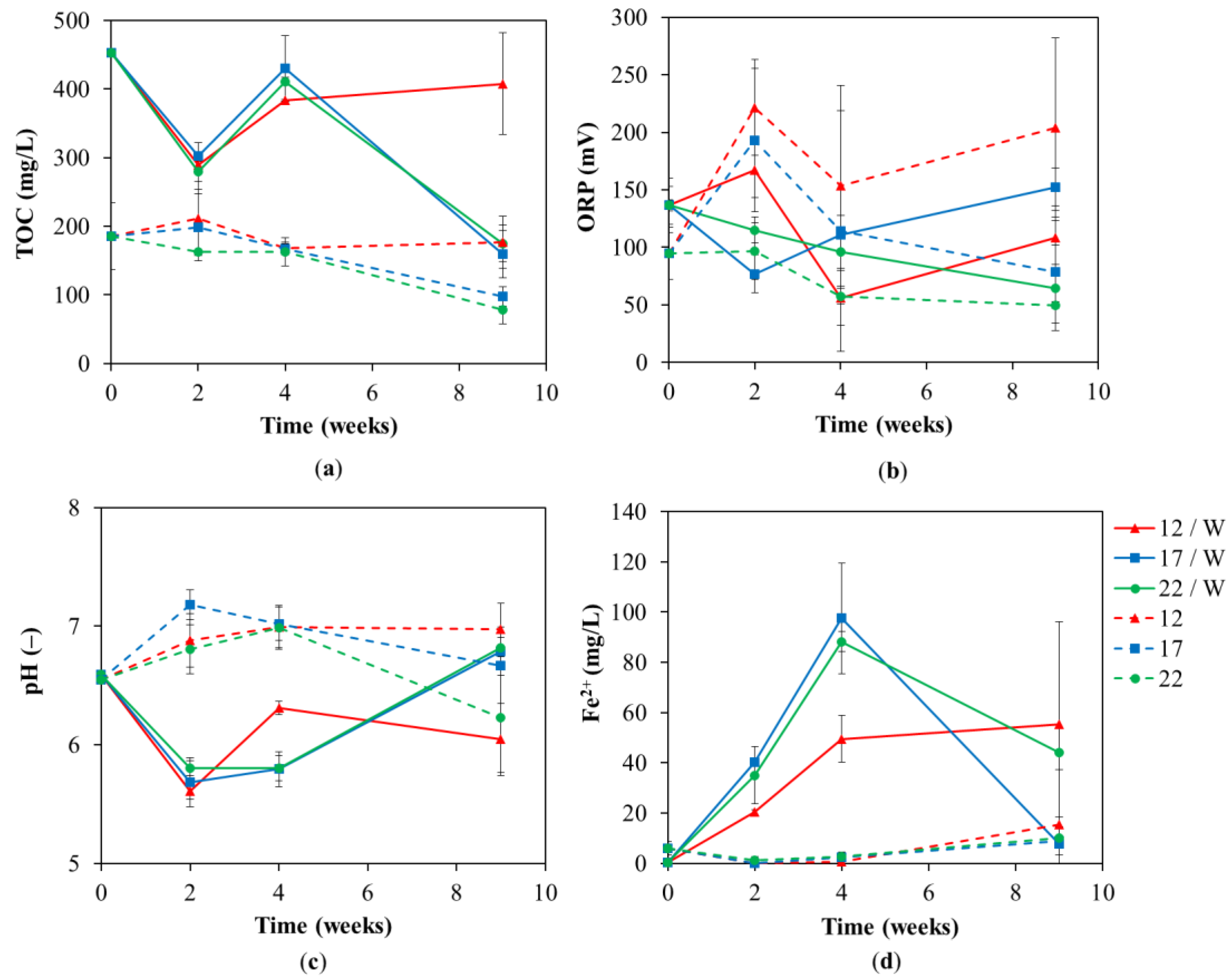
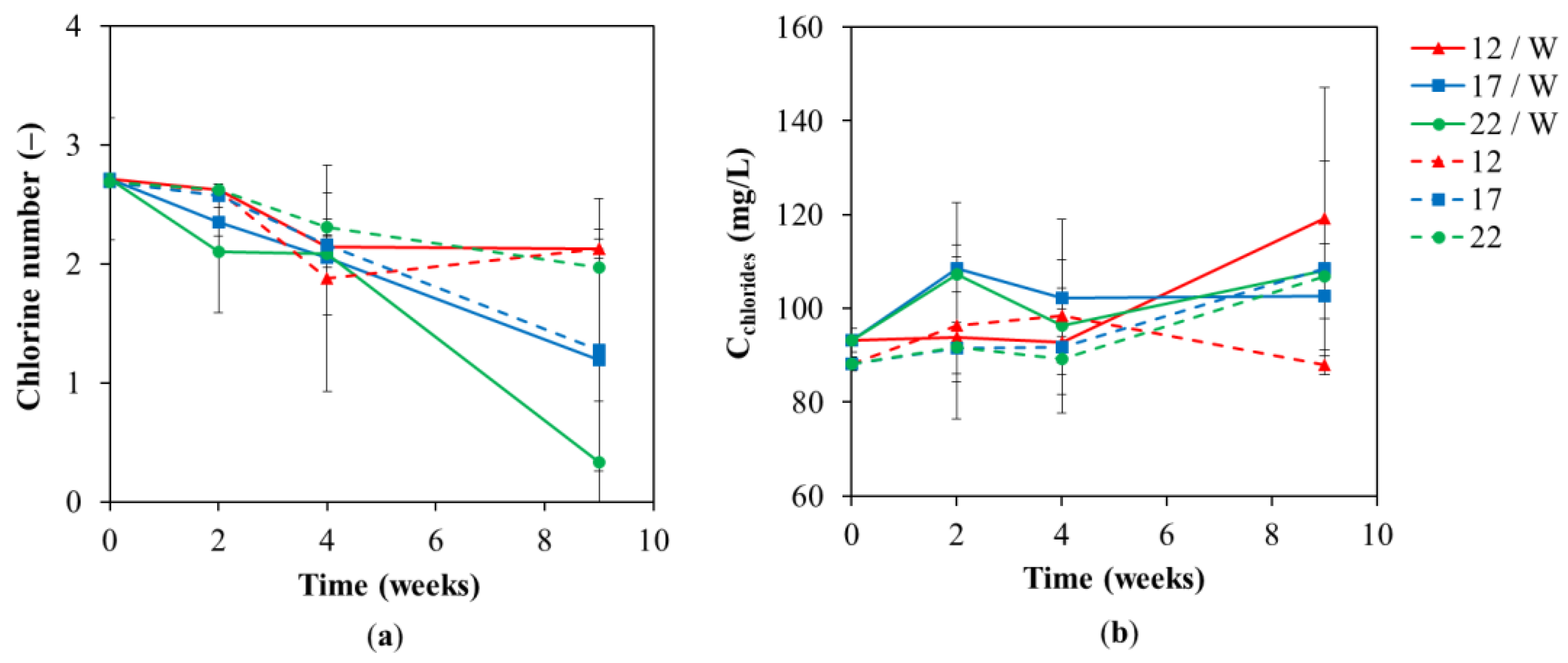
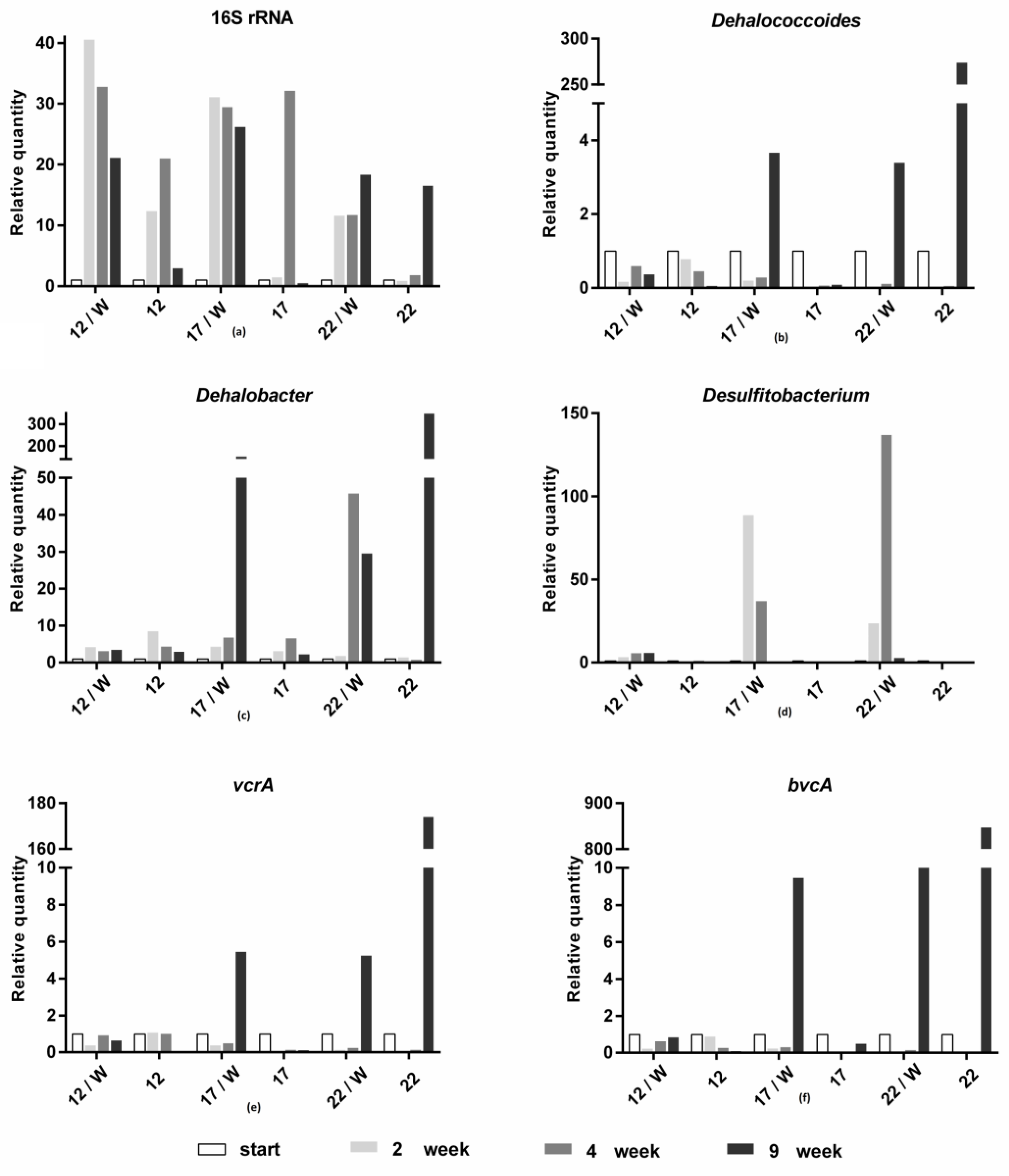
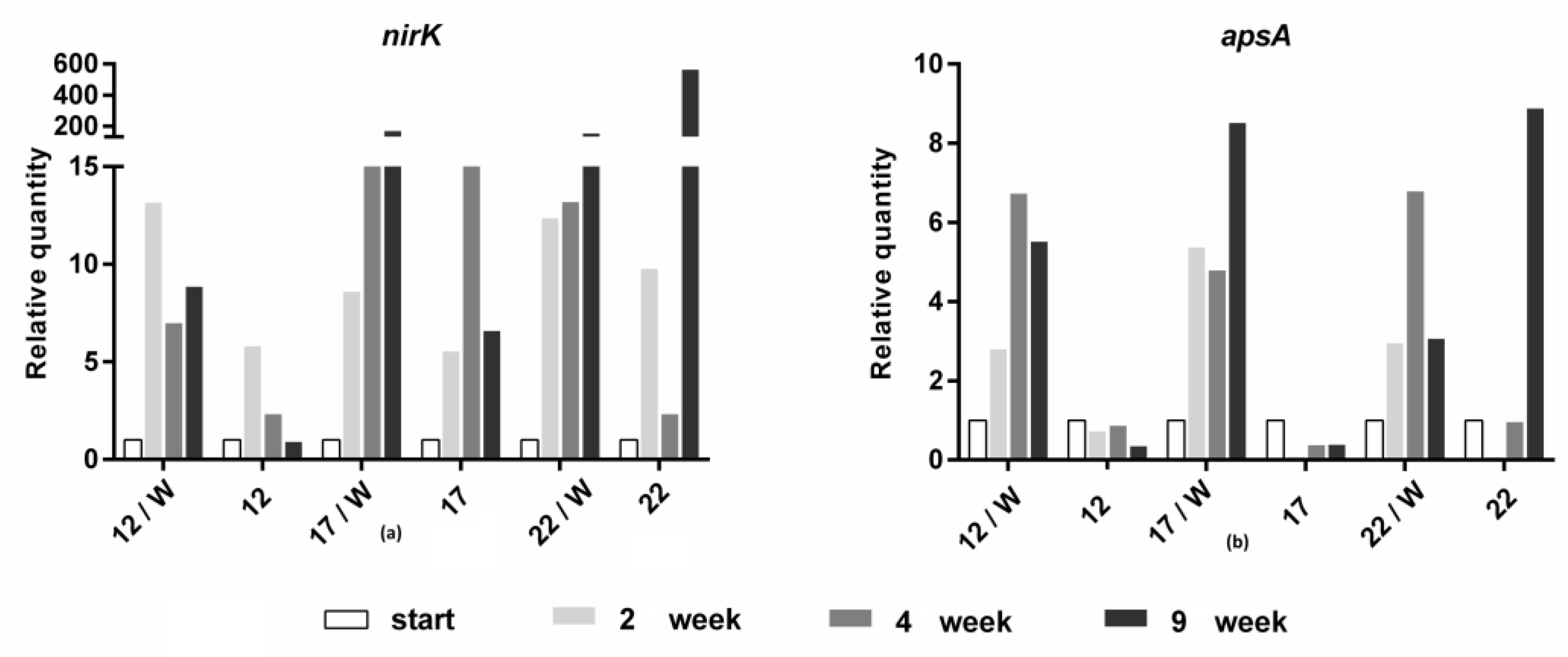
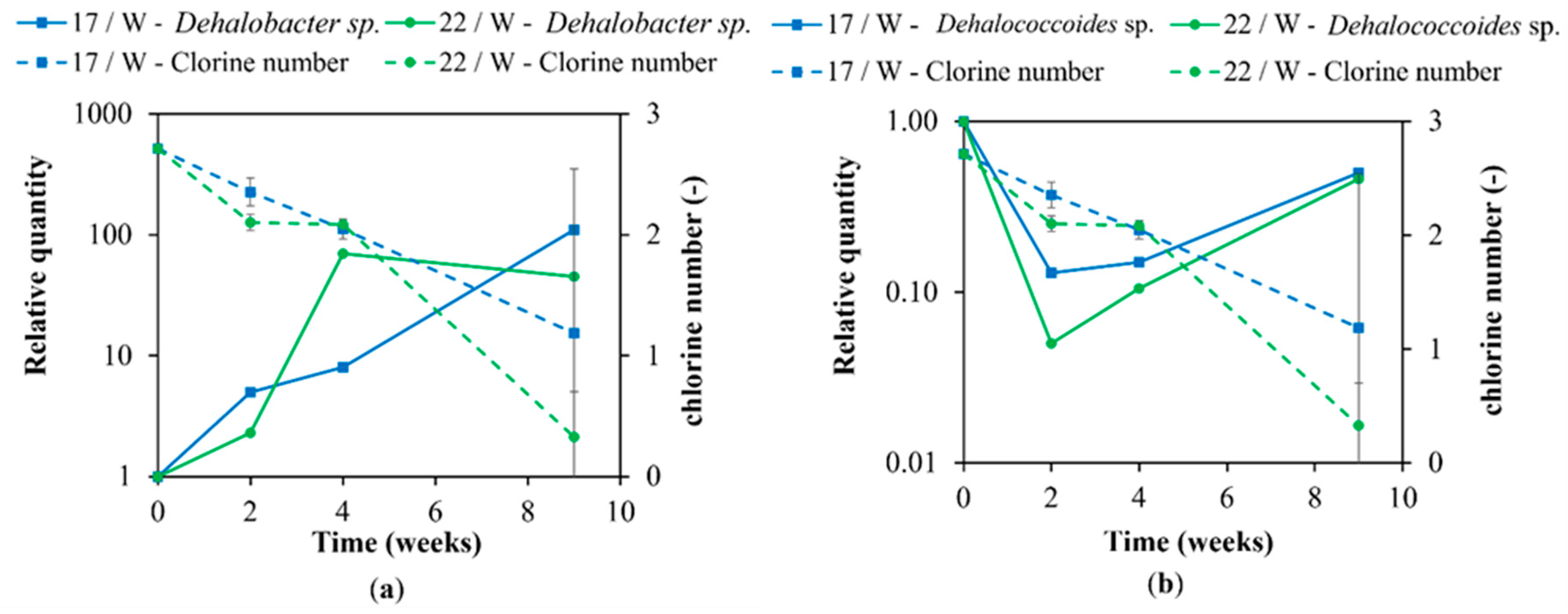
3. Results and Discussion
3.1. Chemical Analyses
3.2. Molecular Biological Analyses
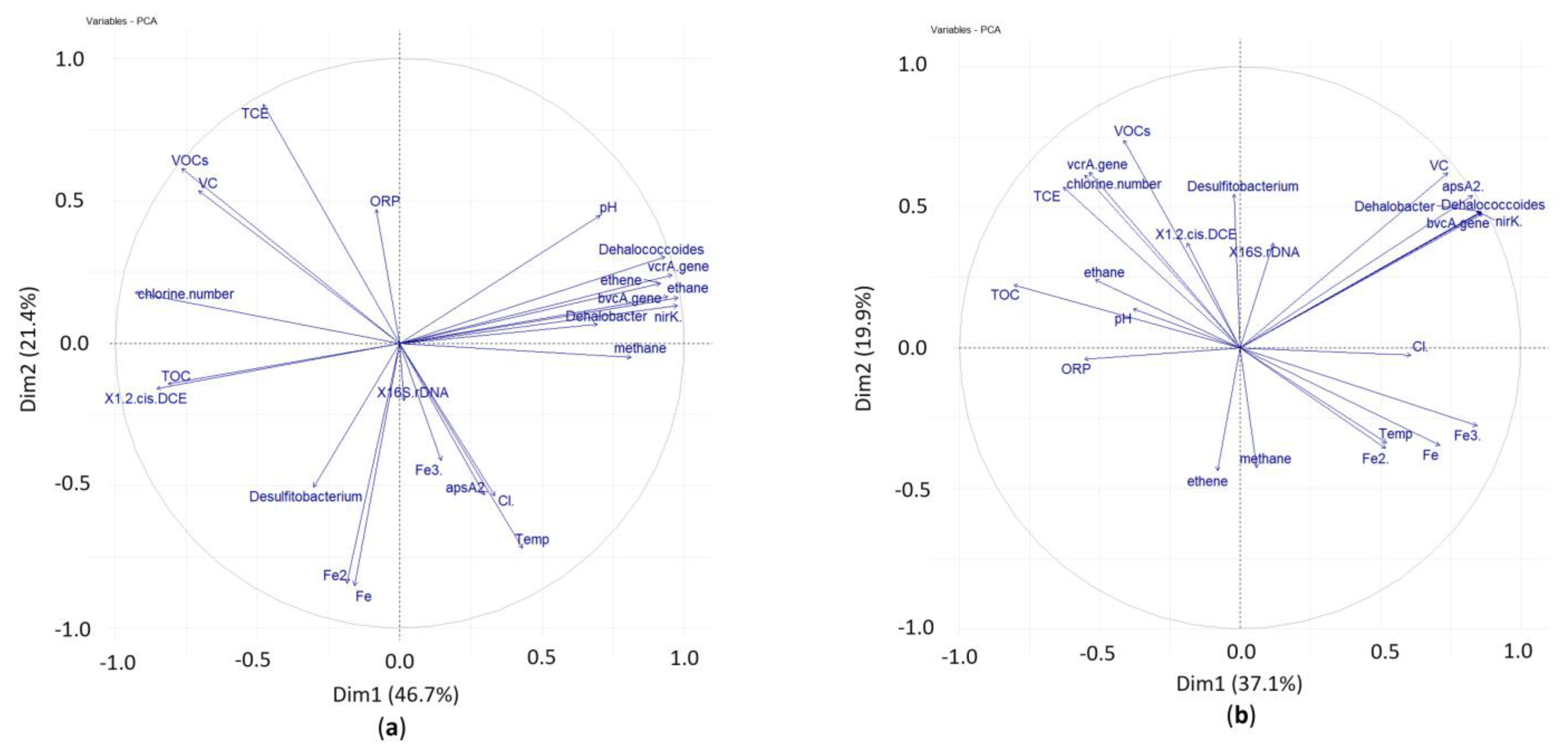
3.3. Principal Component Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rusyn, I.; Chiu, W.A.; Lash, L.H.; Kromhout, H.; Hansen, J.; Guyton, K.Z. Trichloroethylene: Mechanistic, epidemiologic and other supporting evidence of carcinogenic hazard. Pharmacol. Ther. 2014, 141, 55–68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bradley, P.M.; Chapelle, F.H. Biodegradation of chlorinated ethenes. In In Situ Remediation of Chlorinated Solvent Plumes; Stroo, H.F., Ward, C.H., Eds.; Springer: New York, NY, USA, 2010; pp. 39–67. [Google Scholar]
- Vogel, T.M.; McCarty, P.L. Biotransformation of tetrachloroethylene to trichloroethylene, dichloroethylene, vinyl chloride, and carbon dioxide under methanogenic conditions. Appl. Environ. Microbiol. 1985, 49, 1080–1083. [Google Scholar] [CrossRef] [Green Version]
- Bouwer, E.J.; Norris, R.; Hinchee, R.; Brown, R.; McCarty, P.; Semprini, L.; Wilson, J.; Kampbell, D.; Reinhard, M.; Borden, R. Bioremediation of chlorinated solvents using alternate electron acceptors. In Handbook of Bioremediation; CRC Press: Boca Raton, FL, USA, 1994; pp. 149–175. [Google Scholar]
- Gibson, S.A.; Sewell, G.W. Stimulation of reductive dechlorination of tetrachloroethene in anaerobic aquifer microcosms by addition of short-chain organic acids or alcohols. Appl. Environ. Microbiol. 1992, 58, 1392–1393. [Google Scholar] [CrossRef] [Green Version]
- McCarty, P.L. Biotic and abiotic transformations of chlorinated solvents in ground water. In Symposium on Natural Attenuation of Chlorinated Organics in Ground Water; US Environmental Protection Agency: Washington, DC, USA, 1996; pp. 5–9. [Google Scholar]
- Vainberg, S.; Condee, C.W.; Steffan, R.J. Large-scale production of bacterial consortia for remediation of chlorinated solvent-contaminated groundwater. J. Ind. Microbiol. Biotechnol. 2009, 36, 1189–1197. [Google Scholar] [CrossRef]
- Schaefer, C.E.; Towne, R.M.; Vainberg, S.; McCray, J.E.; Steffan, R.J. Bioaugmentation for treatment of dense non-aqueous phase liquid in fractured sandstone blocks. Environ. Sci. Technol. 2010, 44, 4958–4964. [Google Scholar] [CrossRef]
- Stroo, H.F.; West, M.R.; Kueper, B.H.; Borden, R.C.; Major, D.W.; Ward, C.H. In situ bioremediation of chlorinated ethene source zones. In Chlorinated Solvent Source Zone Remediation; Kueper, B.H., Stroo, H.F., Vogel, C.M., Ward, C.H., Eds.; Springer: New York, NY, USA, 2014; pp. 395–457. [Google Scholar]
- Dang, H.Y.; Kanitkar, Y.H.; Stedtfeld, R.D.; Hatzinger, P.B.; Hashsham, S.A.; Cupples, A.M. Abundance of chlorinated solvent and 1,4-dioxane degrading microorganisms at five chlorinated solvent contaminated sites determined via shotgun sequencing. Environ. Sci. Technol. 2018, 52, 13914–13924. [Google Scholar] [CrossRef] [PubMed]
- IARC. Vinyl chloride. In Chemical Agents and Related Occupations; World Health Organization: Lyon, France, 2012; p. 454. [Google Scholar]
- Chambon, J.C.; Bjerg, P.L.; Scheutz, C.; Baelum, J.; Jakobsen, R.; Binning, P.J. Review of reactive kinetic models describing reductive dechlorination of chlorinated ethenes in soil and groundwater. Biotechnol. Bioeng. 2013, 110, 1. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, P.; Pavlostathis, S.G. Effect of temperature, pH and electron donor on the microbial reductive dechlorination of chloroalkenes. Chemosphere 1995, 31, 3537–3548. [Google Scholar] [CrossRef]
- Friis, A.K.; Heimann, A.C.; Jakobsen, R.; Albrechtsen, H.J.; Cox, E.; Bjerg, P.L. Temperature dependence of anaerobic TCE-dechlorination in a highly enriched Dehalococcoides-containing culture. Water Res. 2007, 41, 355–364. [Google Scholar] [CrossRef]
- Ni, Z.B.; van Gaans, P.; Smit, M.; Rijnaarts, H.; Grotenhuis, T. Biodegradation of cis-1,2-dichloroethene in simulated underground thermal energy storage systems. Environ. Sci. Technol. 2015, 49, 13519–13527. [Google Scholar] [CrossRef]
- Atlas, R.M.; Bartha, R. Biotechnological aspects of microbial ecology. In Microbial Ecology: Fundamentals and Applications; Benjamin-Cummings Pub. Co.: Menlo Park, CA, USA, 1987; pp. 364–459. [Google Scholar]
- Najmanová, P.; Knytl, V.; Steinová, J.; Macháčková, J.; Němeček, J. Anaerobic biodegradation of chlorinated ethenes enhanced by elevated temperature – lab test result. In Conference Remediation Technologies XIX; Ekomonitor: Třeboň, Czech Republic, 2016. [Google Scholar]
- Bewley, R.; Hick, P.; Rawcliffe, A. Meeting the challenges for bioremediation of chlorinated solvents at operational sites: A comparison of case studies. In Proceedings of the 13th International UFZ-Deltarez Conference on Sustainable Use and Management of Soil, Sediment and Water Resources, Copenhagen, Denmark, 9–12 June 2015. [Google Scholar]
- Němeček, J.; Marková, K.; Špánek, R.; Antoš, V.; Kozubek, P.; Lhotský, O.; Černík, M. Hydrochemical conditions for aerobic/anaerobic biodegradation of chlorinated ethenes—A multi-site assessment. Water 2020, 12, 322. [Google Scholar] [CrossRef] [Green Version]
- Smits, T.H.M.; Devenoges, C.; Szynalski, K.; Maillard, J.; Holliger, C. Development of a real-time PCR method for quantification of the three genera Dehalobacter, Dehalococcoides, and Desulfitobacterium in microbial communities. J. Microbiol. Methods 2004, 57, 369–378. [Google Scholar] [CrossRef]
- Yoshida, N.; Takahashi, N.; Hiraishi, A. Phylogenetic characterization of a polychlorinated-dioxin-dechlorinating microbial community by use of microcosm studies. Appl. Environ. Microbiol. 2005, 71, 4325–4334. [Google Scholar] [CrossRef] [Green Version]
- Behrens, S.; Azizian, M.F.; McMurdie, P.J.; Sabalowsky, A.; Dolan, M.E.; Semprini, L.; Spormann, A.M. Monitoring abundance and expression of “Dehalococcoides” species chloroethene-reductive dehalogenases in a tetrachloroethene-dechlorinating flow column. Appl. Environ. Microbiol. 2008, 74, 5695–5703. [Google Scholar] [CrossRef] [Green Version]
- Ben-Dov, E.; Brenner, A.; Kushmaro, A. Quantification of sulfate-reducing bacteria in industrial wastewater, by real-time polymerase chain reaction (PCR) using dsrA and apsA genes. Microb. Ecol. 2007, 54, 439–451. [Google Scholar] [CrossRef]
- Henry, S.; Baudoin, E.; Lopez-Gutierrez, J.C.; Martin-Laurent, F.; Brauman, A.; Philippot, L. Quantification of denitrifying bacteria in soils by nirK gene targeted real-time PCR. J. Microbiol. Methods 2004, 59, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Clifford, R.J.; Milillo, M.; Prestwood, J.; Quintero, R.; Zurawski, D.V.; Kwak, Y.I.; Waterman, P.E.; Lesho, E.P.; Mc Gann, P. Detection of bacterial 16S rRNA and identification of four clinically important bacteria by real-time PCR. PLoS ONE 2012, 7, e48558. [Google Scholar] [CrossRef] [PubMed]
- Kengen, S.W.M.; Breidenbach, C.G.; Felske, A.; Stams, A.J.M.; Schraa, G.; de Vos, W.M. Reductive dechlorination of tetrachloroethene to cis-1,2-dichloroethene by a thermophilic anaerobic enrichment culture. Appl. Environ. Microbiol. 1999, 65, 2312–2316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fletcher, K.E.; Costanza, J.; Cruz-Garcia, C.; Ramaswamy, N.S.; Pennell, K.D.; Loffler, F.E. Effects of elevated temperature on Dehalococcoides dechlorination performance and DNA and RNA biomarker abundance. Environ. Sci. Technol. 2011, 45, 712–718. [Google Scholar] [CrossRef]
- Marcet, T.F.; Capiro, N.L.; Yang, Y.; Loffler, F.E.; Pennell, K.D. Impacts of low-temperature thermal treatment on microbial detoxification of tetrachloroethene under continuous flow conditions. Water Res. 2018, 145, 21–29. [Google Scholar] [CrossRef]
- Yamazaki, Y.; Hasegawa, A.; Tian, X.W.; Suzuki, I.; Kobayashi, T.; Shimizu, T.; Inoue, D.; Ike, M. Effect of elevated temperature on cis-1,2-dichloroethene dechlorination and microbial community structure in contaminated soils—A biostimulation approach. J. Environ. Chem. Eng. 2020, 8, 8. [Google Scholar] [CrossRef]
- Yamazaki, Y.; Kitamura, G.; Tian, X.W.; Suzuki, I.; Kobayashi, T.; Shimizu, T.; Inoue, D.; Ike, M. Temperature dependence of sequential chlorinated ethenes dechlorination and the dynamics of dechlorinating microorganisms. Chemosphere 2022, 287, 9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hu, M.; Li, P.F.; Wang, X.; Meng, Q.J. Trichloroethylene removal and bacterial variations in the up-flow anaerobic sludge blanket reactor in response to temperature shifts. Appl. Microbiol. Biotechnol. 2015, 99, 6091–6102. [Google Scholar] [CrossRef]
- He, J.Z.; Ritalahti, K.M.; Aiello, M.R.; Loffler, F.E. Complete detoxification of vinyl chloride by an anaerobic enrichment culture and identification of the reductively dechlorinating population as a Dehalococcoides species. Appl. Environ. Microbiol. 2003, 69, 996–1003. [Google Scholar] [CrossRef] [Green Version]
- Chan, A.L.; Tseng, C.Y.; Chen, T.W.; Lo, Y.H.; Chang, S.C. Microbiome reengineering by heat selection for rapid biodegradation of trichloroethylene with minimal vinyl chloride formation. Water Air Soil Pollut. 2021, 232, 21. [Google Scholar] [CrossRef]
- Riedel, T. Temperature-associated changes in groundwater quality. J. Hydrol. 2019, 572, 206–212. [Google Scholar] [CrossRef]
- Marcet, T.F.; Capiro, N.L.; Morris, L.A.; Hassan, S.M.; Yang, Y.; Loffler, F.E.; Pennell, K.D. Release of electron donors during thermal treatment of soils. Environ. Sci. Technol. 2018, 52, 3642–3651. [Google Scholar] [CrossRef]
- Meng, B.Y.; Yang, Y.; Huang, Y.H.; Kolditz, O.; Shao, H.B. Remediation potential of borehole thermal energy storage for chlorinated hydrocarbon plumes: Numerical modeling in a variably-saturated aquifer. Front. Earth Sci. 2021, 9, 17. [Google Scholar] [CrossRef]
- Horst, J.; Flanders, C.; Klemmer, M.; Randhawa, D.S.; Rosso, D. Low-temperature thermal remediation: Gaining traction as a green remedial alternative. Ground Water Monit. Remediat. 2018, 38, 18–27. [Google Scholar] [CrossRef]
- Ni, Z.B.; van Gaans, P.; Rijnaarts, H.; Grotenhuis, T. Combination of aquifer thermal energy storage and enhanced bioremediation: Biological and chemical clogging. Sci. Total Environ. 2018, 613, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Friis, A.K.; Kofoed, J.L.L.; Heron, G.; Albrechtsen, H.J.; Bjerg, P.L. Microcosm evaluation of bioaugmentation after field-scale thermal treatment of a TCE-contaminated aquifer. Biodegradation 2007, 18, 661–674. [Google Scholar] [CrossRef]
- Nozhevnikova, A.N.; Simankova, M.V.; Parshina, S.N.; Kotsyurbenko, O.R. Temperature characteristics of methanogenic archaea and acetogenic bacteria isolated from cold environments. Water Sci. Technol. 2001, 44, 41–48. [Google Scholar] [CrossRef]
- Wen, L.L.; Zhang, Y.; Chen, J.X.; Zhang, Z.X.; Yi, Y.Y.; Tang, Y.; Rittmann, B.E.; Zhao, H.P. The dechlorination of TCE by a perchlorate reducing consortium. Chem. Eng. J. 2017, 313, 1215–1221. [Google Scholar] [CrossRef]
- Robles, A.; Yellowman, T.L.; Joshi, S.; Rangan, S.M.; Delgado, A.G. Microbial chain elongation and subsequent fermentation of elongated carboxylates as H2-producing processes for sustained reductive dechlorination of chlorinated ethenes. Environ. Sci. Technol. 2021, 55, 10398–10410. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, K.E.; Costanza, J.; Pennell, K.D.; Loffler, F.E. Electron donor availability for microbial reductive processes following thermal treatment. Water Res. 2011, 45, 6625–6636. [Google Scholar] [CrossRef] [PubMed]
- Kruse, S.; Turkowsky, D.; Birkigt, J.; Matturro, B.; Franke, S.; Jehmlich, N.; von Bergen, M.; Westermann, M.; Rossetti, S.; Nijenhuis, I.; et al. Interspecies metabolite transfer and aggregate formation in a co-culture of Dehalococcoides and Sulfurospirillum dehalogenating tetrachloroethene to ethene. ISME J. 2021, 15, 1794–1809. [Google Scholar] [CrossRef]
- Li, J.W.; Hu, A.Y.; Bai, S.J.; Yang, X.Y.; Sun, Q.; Liao, X.; Yu, C.P. Characterization and performance of lactate-feeding consortia for reductive dechlorination of trichloroethene. Microorganisms 2021, 9, 751. [Google Scholar] [CrossRef]
- Anam, G.B.; Choi, J.; Ahn, Y. Reductive dechlorination of perchloroethene (PCE) and bacterial community changes in a continuous-flow, two-stage anaerobic column. Int. Biodeterior. Biodegrad. 2019, 138, 41–49. [Google Scholar] [CrossRef]
- Dutta, N.; Thomsen, K.; Ahring, B.K. Degrading chlorinated aliphatics by reductive dechlorination of groundwater samples from the Santa Susana Field Laboratory. Chemosphere 2022, 298, 10. [Google Scholar] [CrossRef]
- Chen, P.P.; Liu, H.; Xing, Z.L.; Wang, Y.Q.; Zhang, X.P.; Zhao, T.T.; Zhang, Y.R. Cometabolic degradation mechanism and microbial network response of methanotrophic consortia to chlorinated hydrocarbon solvents. Ecotoxicol. Environ. Saf. 2022, 230, 9. [Google Scholar] [CrossRef]
- Cao, L.F.; Sun, W.H.; Zhang, Y.T.; Feng, S.M.; Dong, J.Y.; Zhang, Y.M.; Rittmann, B.E. Competition for electrons between reductive dechlorination and denitrification. Front. Environ. Sci. Eng. 2017, 11, 10. [Google Scholar] [CrossRef]
- Hermon, L.; Hellal, J.; Denonfoux, J.; Vuilleumier, S.; Imfeld, G.; Urien, C.; Ferreira, S.; Joulian, C. Functional genes and bacterial communities during organohalide respiration of chloroethenes in microcosms of multi-contaminated groundwater. Front. Microbiol. 2019, 10, 16. [Google Scholar] [CrossRef] [Green Version]
- Han, K.; Hong, U.; Park, S.; Lee, G.; Kwon, S.; Kim, Y. Long-term monitoring and evaluating biological activity of in situ anaerobic reductive dechlorination at a highly recharged and TCE-contaminated aquifer. Desalination Water Treat. 2016, 57, 24085–24095. [Google Scholar] [CrossRef]
- Lo, K.H.; Lu, C.W.; Chien, C.C.; Sheu, Y.T.; Lin, W.H.; Chen, S.C.; Kao, C.M. Cleanup chlorinated ethene-polluted groundwater using an innovative immobilized Clostridium butyricum column scheme: A pilot-scale study. J. Environ. Manag. 2022, 311, 13. [Google Scholar] [CrossRef]
- Chen, W.T.; Chen, K.F.; Surmpalli, R.Y.; Zhang, T.C.; Ou, J.H.; Kao, C.M. Bioremediation of trichloroethylene-polluted groundwater using emulsified castor oil for slow carbon release and acidification control. Water Environ. Res. 2022, 94, 15. [Google Scholar] [CrossRef]
- Florey, J.; Adams, A.; Michel, L. Reductive dechlorination of a chlorinated solvent plume in Houston, Texas. Remediat. J. Environ. Cleanup Costs Technol. Tech. 2017, 28, 45–54. [Google Scholar] [CrossRef]
- Liu, P.W.G.; Wu, Y.J.; Whang, L.M.; Lin, T.F.; Hung, W.N.; Cho, K.C. Molecular tools with statistical analysis on trichloroethylene remediation effectiveness. Int. Biodeterior. Biodegrad. 2020, 154, 10. [Google Scholar] [CrossRef]
- Schaefer, C.E.; Lavorgna, G.M.; Haluska, A.A.; Annable, M.D. Long-term impacts on groundwater and reductive dechlorination following bioremediation in a highly characterized trichloroethene DNAPL source area. Ground Water Monit. Remediat. 2018, 38, 65–74. [Google Scholar] [CrossRef]
- Park, C.W.; Bastian, E.; Farkas, B.; Drake, M. The effect of acidification of liquid whey protein concentrate on the flavor of spray-dried powder. J. Dairy Sci. 2014, 97, 4043–4051. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.Y.; Wu, J.H.; Chu, S.C. Deciphering microbiomes in anaerobic reactors with superior trichloroethylene dechlorination performance at low pH conditions. Environ. Pollut. 2020, 257, 12. [Google Scholar] [CrossRef]
- Jacome, L.A.P.; Wang, P.H.; Molenda, O.; Li, Y.X.; Islam, M.A.; Edwards, E.A. Sustained dechlorination of vinyl chloride to ethene in Dehalococcoides-enriched cultures grown without addition of exogenous vitamins and at low pH. Environ. Sci. Technol. 2019, 53, 11364–11374. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Capiro, N.L.; Marcet, T.F.; Yan, J.; Pennell, K.D.; Loffler, F.E. Organohalide respiration with chlorinated ethenes under low pH conditions. Environ. Sci. Technol. 2017, 51, 8579–8588. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.G.; Chien, C.C.; Sheu, Y.T.; Verpoort, F.; Chen, S.C.; Kao, C.M. Enhanced bioremediation of trichloroethene-contaminated groundwater using modified gamma-PGA for continuous substrate supplement and pH control: Batch and pilot-scale studies. J. Clean. Prod. 2021, 278, 15. [Google Scholar] [CrossRef]
- Xu, Y.; Xue, L.; Ye, Q.; Franks, A.E.; Zhu, M.; Feng, X.; Xu, J.; He, Y. Inhibitory effects of sulfate and nitrate reduction on reductive dechlorination of PCP in a flooded paddy soil. Front. Microbiol. 2018, 9, 567. [Google Scholar] [CrossRef]
- Murray, A.; Maillard, J.; Rolle, M.; Broholm, M.; Holliger, C. Impact of iron- and/or sulfate-reduction on a cis-1,2-dichloroethene and vinyl chloride respiring bacterial consortium: Experiments and model-based interpretation. Environ. Sci.-Process. Impacts 2020, 22, 740–750. [Google Scholar] [CrossRef]
- Němeček, J.; Steinová, J.; Špánek, R.; Pluhař, T.; Pokorný, P.; Najmanová, P.; Knytl, V.; Černík, M. Thermally enhanced in situ bioremediation of groundwater contaminated with chlorinated solvents—A field test. Sci. Total Environ. 2018, 622–623, 743–755. [Google Scholar] [CrossRef]
- Hellal, J.; Joulian, C.; Urien, C.; Ferreira, S.; Denonfoux, J.; Hermon, L.; Vuilleumier, S.; Imfeld, G. Chlorinated ethene biodegradation and associated bacterial taxa in multi-polluted groundwater: Insights from biomolecular markers and stable isotope analysis. Sci. Total Environ. 2021, 763, 10. [Google Scholar] [CrossRef]
- Baelum, J.; Scheutz, C.; Chambon, J.C.; Jensen, C.M.; Brochmann, R.P.; Dennis, P.; Laier, T.; Broholm, M.M.; Bjerg, P.L.; Binning, P.J.; et al. The impact of bioaugmentation on dechlorination kinetics and on microbial dechlorinating communities in subsurface clay till. Environ. Pollut. 2014, 186, 149–157. [Google Scholar] [CrossRef]
- Lee, P.K.H.; Macbeth, T.W.; Sorenson, K.S.; Deeb, R.A.; Alvarez-Cohen, L. Quantifying genes and transcripts to assess the in situ physiology of “Dehalococcoides” spp. in a trichloroethene-contaminated groundwater site. Appl. Environ. Microbiol. 2008, 74, 2728–2739. [Google Scholar] [CrossRef] [Green Version]
- Němeček, J.; Dolinová, I.; Machačková, J.; Špánek, R.; Ševců, A.; Lederer, T.; Černík, M. Stratification of chlorinated ethenes natural attenuation in an alluvial aquifer assessed by hydrochemical and biomolecular tools. Chemosphere 2017, 184, 1157–1167. [Google Scholar] [CrossRef]
- Wan, J.X.; Chen, C.; Chen, J.W.; Miao, Q.Y.; Liu, Y.D.; Ye, J.X.; Chen, K.Z.; Jin, Y.Y.; Tang, X.J.; Shen, C.F. Acceleration of perchloroethylene dechlorination by extracellular secretions from Microbacterium in a mixed culture containing Desulfitobacterium. Environ. Pollut. 2019, 245, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Q.; Chen, C.; Zhao, S.Y.; He, J.Z. Microbial synergistic interactions for reductive dechlorination of polychlorinated biphenyls. Sci. Total Environ. 2019, 666, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Nagymate, Z.; Jurecska, L.; Romsics, C.; Toth, F.; Bodai, V.; Meszaros, E.; Szabo, A.; Erdelyi, B.; Marialigeti, K. Preparation and characterization of site-specific dechlorinating microbial inocula capable of complete dechlorination enriched in anaerobic microcosms amended with clay mineral. World J. Microbiol. Biotechnol. 2020, 36, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antoniou, K.; Mamais, D.; Pantazidou, M. Reductive dechlorination of trichloroethene under different sulfate-reducing and electron donor conditions. J. Contam. Hydrol. 2019, 226, 10. [Google Scholar] [CrossRef]
- Fan, T.T.; Yang, M.; Li, Q.; Zhou, Y.; Xia, F.Y.; Chen, Y.; Yang, L.; Ding, D.; Zhang, S.T.; Zhang, X.D.; et al. A new insight into the influencing factors of natural attenuation of chlorinated hydrocarbons contaminated groundwater: A long-term field study of a retired pesticide site. J. Hazard. Mater. 2022, 439, 9. [Google Scholar] [CrossRef]
- Zhang, X.; Luo, M.; Deng, S.; Long, T.; Yu, L.S.R. Field study of microbial community structure and dechlorination activity in a multi-solvents co-contaminated site undergoing natural attenuation. J. Hazard. Mater. 2022, 423, 127010. [Google Scholar] [CrossRef]
- Freeborn, R.A.; West, K.A.; Bhupathiraju, V.K.; Chauhan, S.; Rahm, B.G.; Richardson, R.E.; Alvarez-Cohen, L. Phylogenetic analysis of TCE-dechlorinating consortia enriched on a variety of electron donors. Environ. Sci. Technol. 2005, 39, 8358–8368. [Google Scholar] [CrossRef]
- Lu, Q.; Liu, J.; He, H.; Liang, Z.; Qiu, R.; Wang, S. Waste activated sludge stimulates in situ microbial reductive dehalogenation of organohalide-contaminated soil. J. Hazard. Mater. 2021, 411, 125189. [Google Scholar] [CrossRef]
| Parameter | Unit | Concentration at Time 0 |
|---|---|---|
| 1,1-dichloroethene | µg/L | <20 |
| cis-1,2-dichloroethene | µg/L | 7430 |
| trans-1,2-dichloroethene | µg/L | <20 |
| Trichloroethene | µg/L | 19,450 |
| Tetrachloroethene | µg/L | <20 |
| Vinyl chloride | µg/L | 1310 |
| Σ cVOCs | µg/L | 28,190 |
| Acetylene | µg/L | <1.0 |
| Ethane | µg/L | 10.1 |
| Ethene | µg/L | 119.6 |
| Methane | µg/L | 234.5 |
| TOC | mg/L | 145 |
| Chlorides | mg/L | 98.6 |
| pH | - | 6.48 |
| ORP | mV | 75.0 |
| Fe2+ | mg/L | 7.89 |
| Fe3+ | mg/L | 0 |
| Primer | Primer Sequences 5′-3′ | Gene | References |
|---|---|---|---|
| Dre441F | GTTAGGGAAGAACGGCATCTGT | Dehalobacter spp.; gene 16S rRNA | [20] |
| Dre645R | CCTCTCCTGTCCTCAAGCCATA | ||
| DHC793f | GGGAGTATCGACCCTCTCTG | Dehalococcoides spp.; gene 16S rRNA | [21] |
| DHC946r | CGTTYCCCTTTCRGTTCACT | ||
| Dsb406F | GTACGACGAAGGCCTTCGGGT | Desulfitobacterium spp.; gene 16S rRNA | [20] |
| Dsb619R | CCCAGGGTTGAGCCCTAGGT | ||
| bvcA277F | TGGGGACCTGTACCTGAAAA | functional gene of VC reduction, Dehalococcoides spp. strain BAV-1 | [22] |
| bvcA523R | CAAGACGCATTGTGGACATC | ||
| vcrA880F | CCCTCCAGATGCTCCCTTTA | functional gene of VC reduction, Dehalococcoides spp. strain VS | [22] |
| vcrA1018R | ATCCCCTCTCCCGTGTAACC | ||
| RH1-aps-F | CGCGAAGACCTKATCTTCGAC | sulfate-reducing bacteria—functional gene apsA | [23] |
| RH2-aps-R | ATCATGATCTGCCAGCGGCCGGA | ||
| nirK876 | ATYGGCGGVCAYGGCGA | denitrifying bacteria—functional gene nirK | [24] |
| nirK1040 | GCCTCGATCAGRTTRTGGTT | ||
| 16SqPCR-F | TCCTACGGGAGGCAGCAGT | gene for 16S rRNA | [25] |
| 16SqPCR-R | GGACTACCAGGGTATCTAATCCTGTT |
| Designation of Test Variant | Temperature | Groundwater | Soil (Dry Matter) | Whey Addition |
|---|---|---|---|---|
| 12/W | 12 °C | 200 mL | 100 g | 1 g/L |
| 17/W | 17 °C | 200 mL | 100 g | 1 g/L |
| 22/W | 22 °C | 200 mL | 100 g | 1 g/L |
| 12 | 12 °C | 200 mL | 100 g | - |
| 17 | 17 °C | 200 mL | 100 g | - |
| 22 | 22 °C | 200 mL | 100 g | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Najmanová, P.; Steinová, J.; Czinnerová, M.; Němeček, J.; Špánek, R.; Knytl, V.; Halecký, M. Thermally Enhanced Biodegradation of TCE in Groundwater. Water 2022, 14, 3456. https://doi.org/10.3390/w14213456
Najmanová P, Steinová J, Czinnerová M, Němeček J, Špánek R, Knytl V, Halecký M. Thermally Enhanced Biodegradation of TCE in Groundwater. Water. 2022; 14(21):3456. https://doi.org/10.3390/w14213456
Chicago/Turabian StyleNajmanová, Petra, Jana Steinová, Marie Czinnerová, Jan Němeček, Roman Špánek, Vladislav Knytl, and Martin Halecký. 2022. "Thermally Enhanced Biodegradation of TCE in Groundwater" Water 14, no. 21: 3456. https://doi.org/10.3390/w14213456
APA StyleNajmanová, P., Steinová, J., Czinnerová, M., Němeček, J., Špánek, R., Knytl, V., & Halecký, M. (2022). Thermally Enhanced Biodegradation of TCE in Groundwater. Water, 14(21), 3456. https://doi.org/10.3390/w14213456






