Assessment of Artificial Sweeteners as Wastewater Co-Tracers in an Urban Groundwater System of Mexico (Monterrey Metropolitan Area)
Abstract
:1. Introduction
2. Materials and Methods
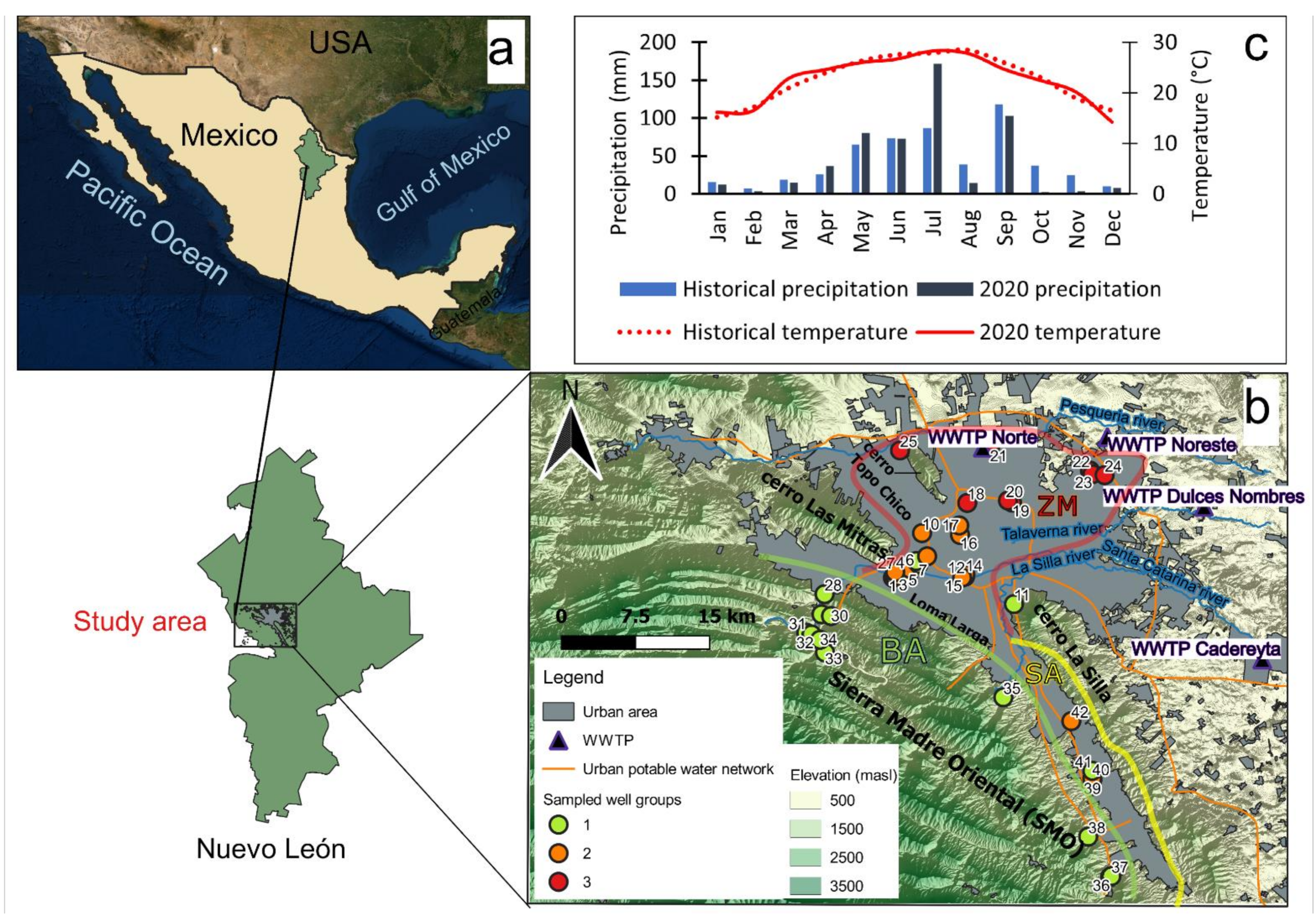
2.1. Study Area
2.2. Field and Laboratory Work
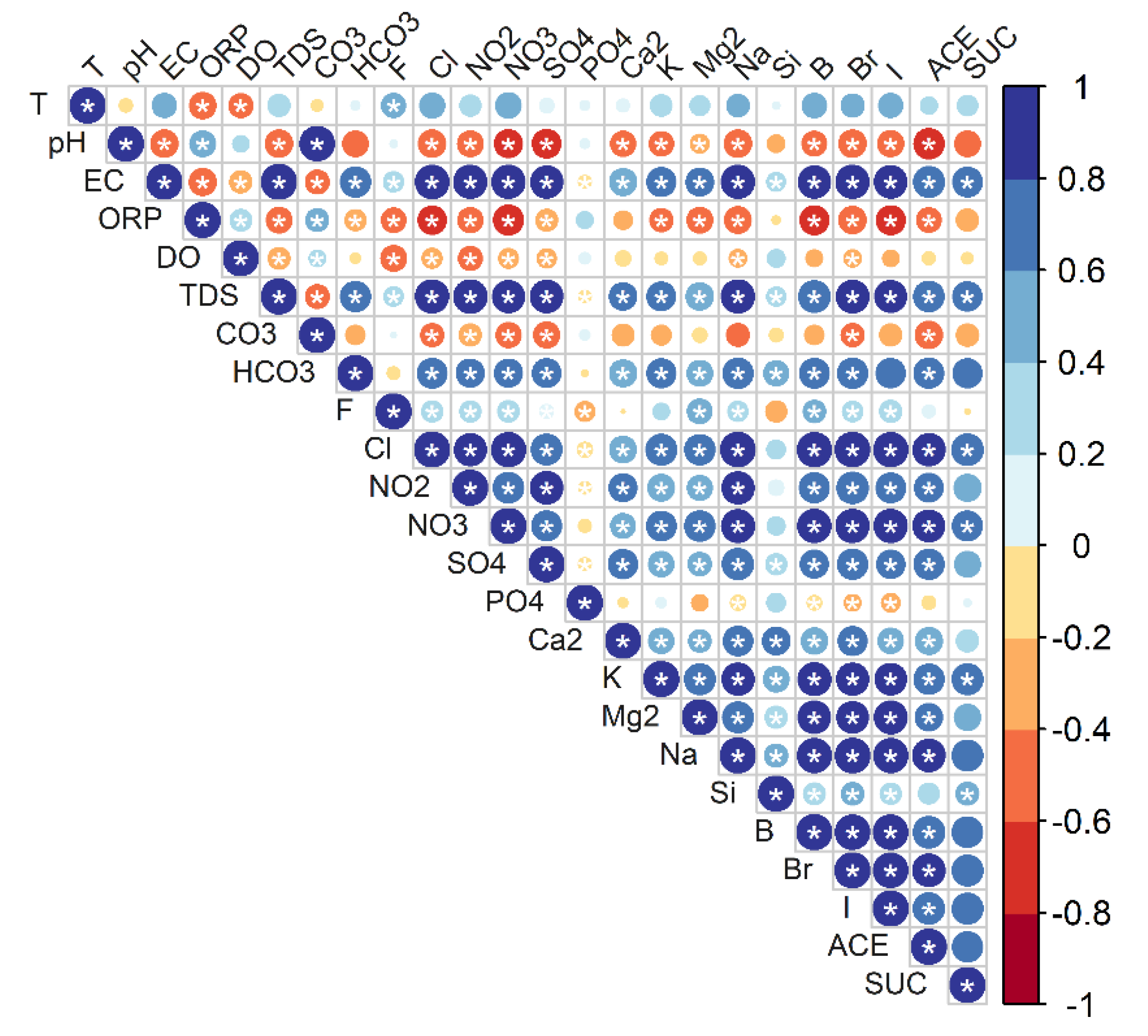
2.3. Statistical Analysis and Interpretation Techniques
3. Results
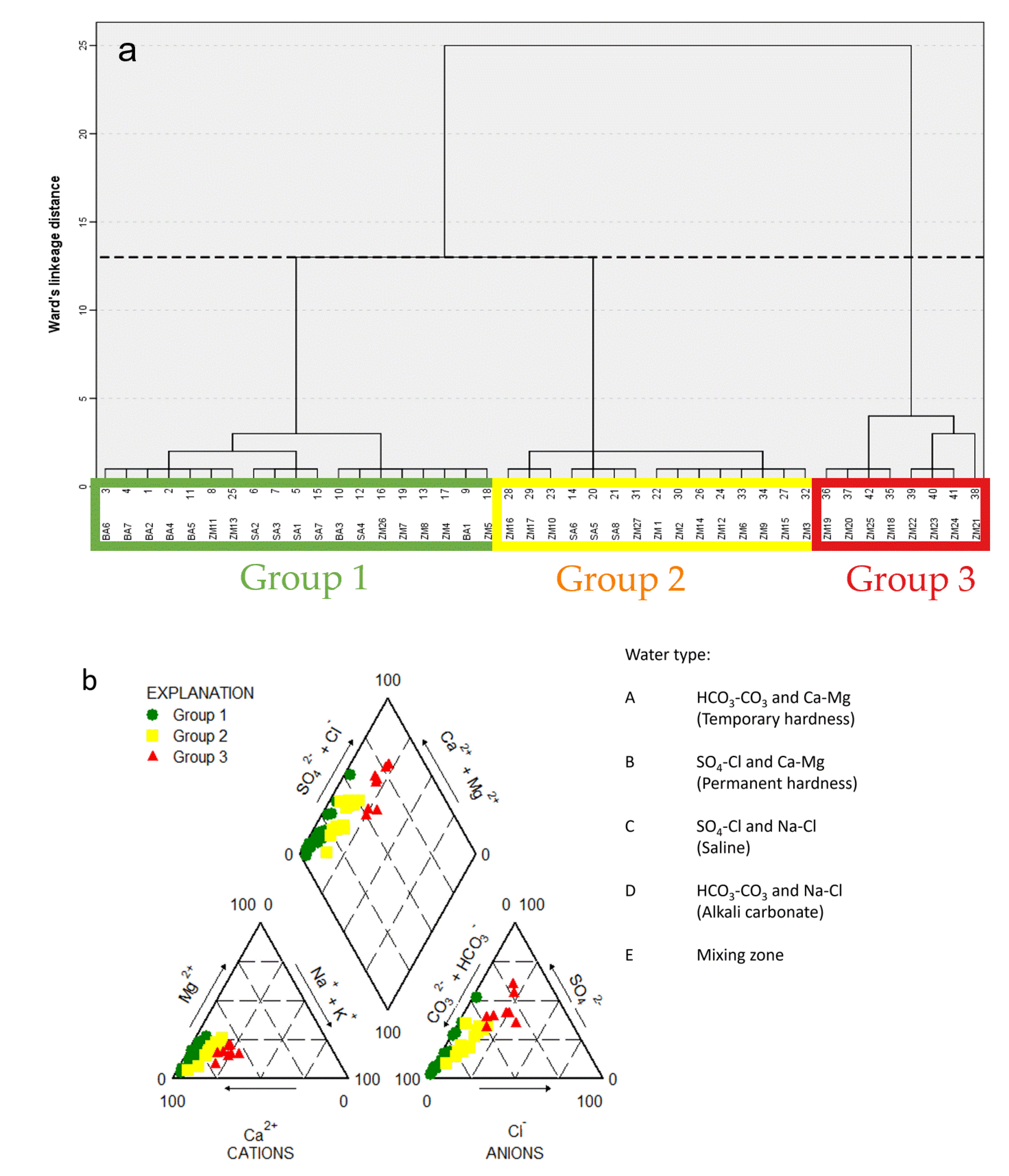
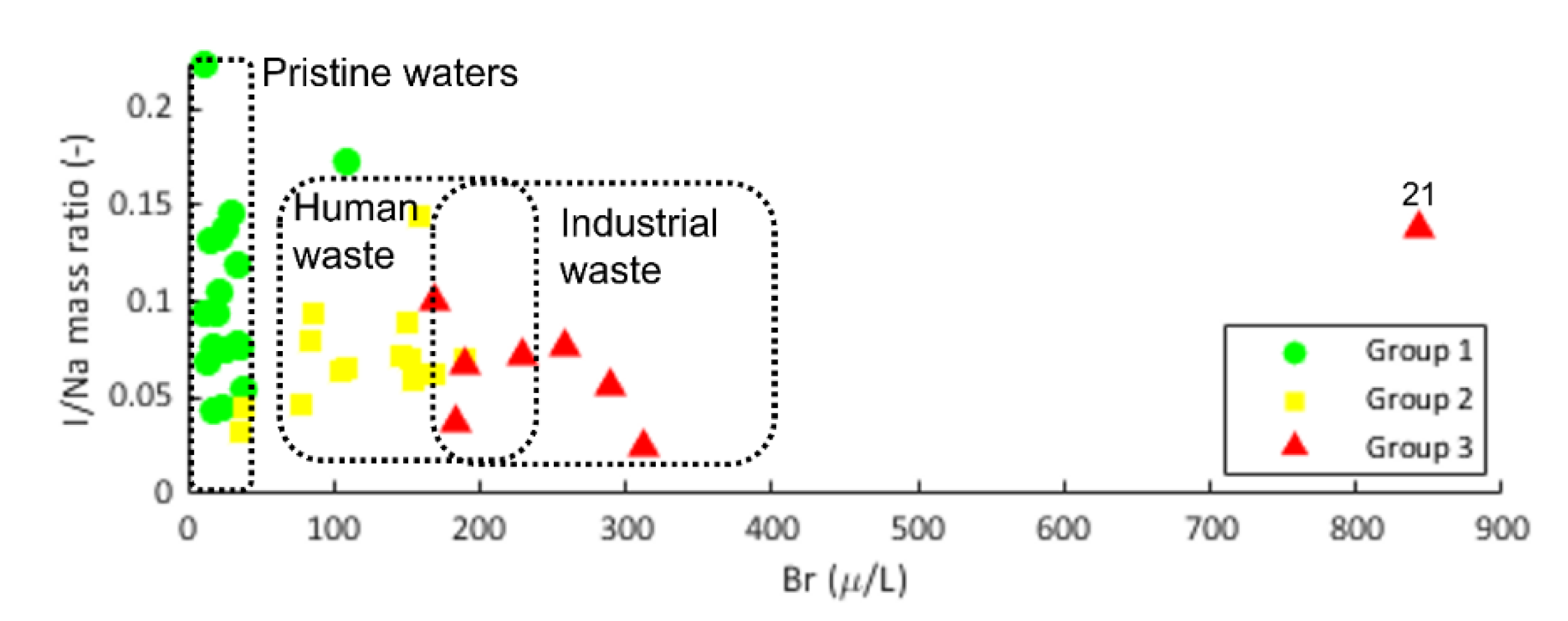
3.1. Groundwater Sampling Groups and Hydrochemical Description
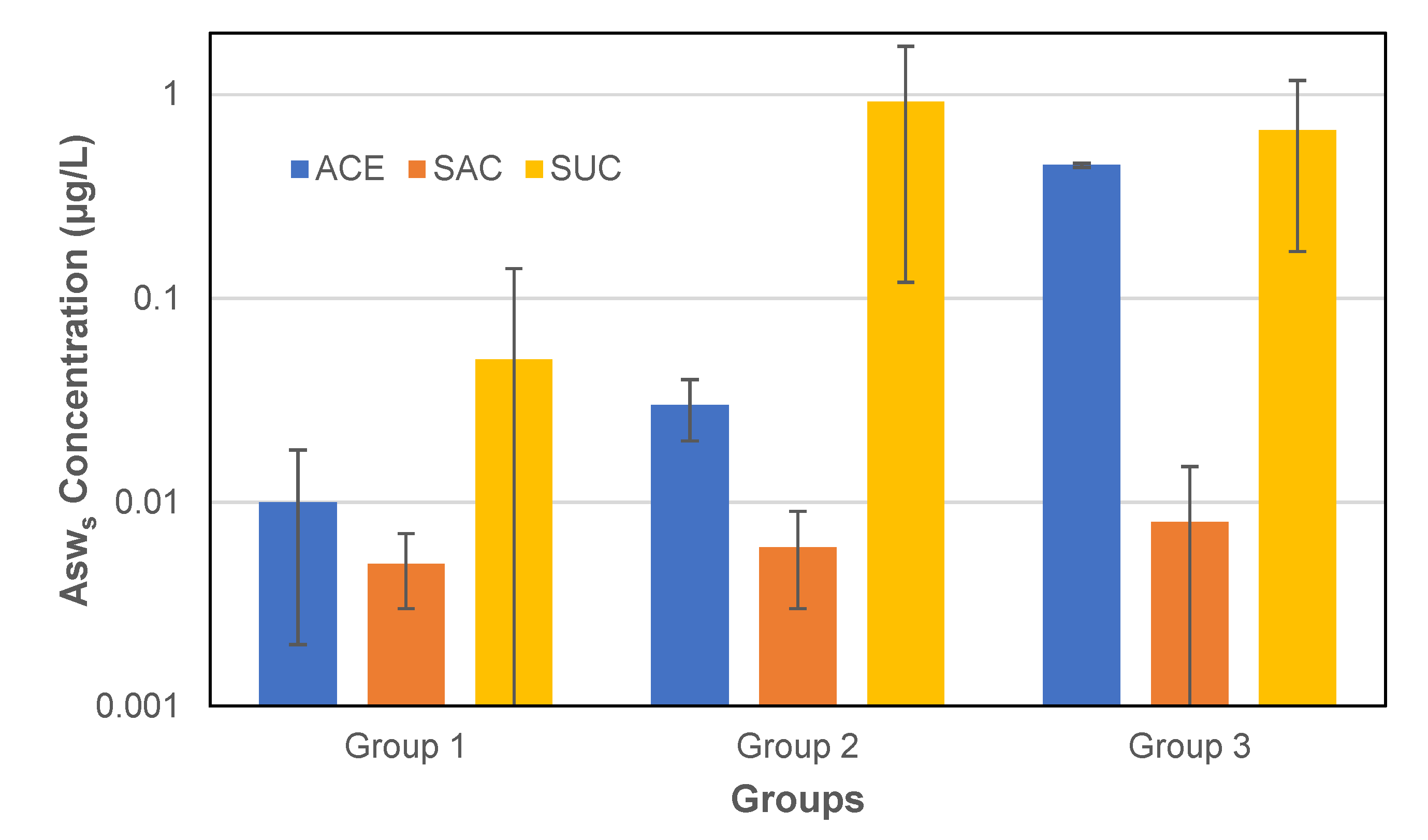
3.2. Concentration of Asws in Groundwater
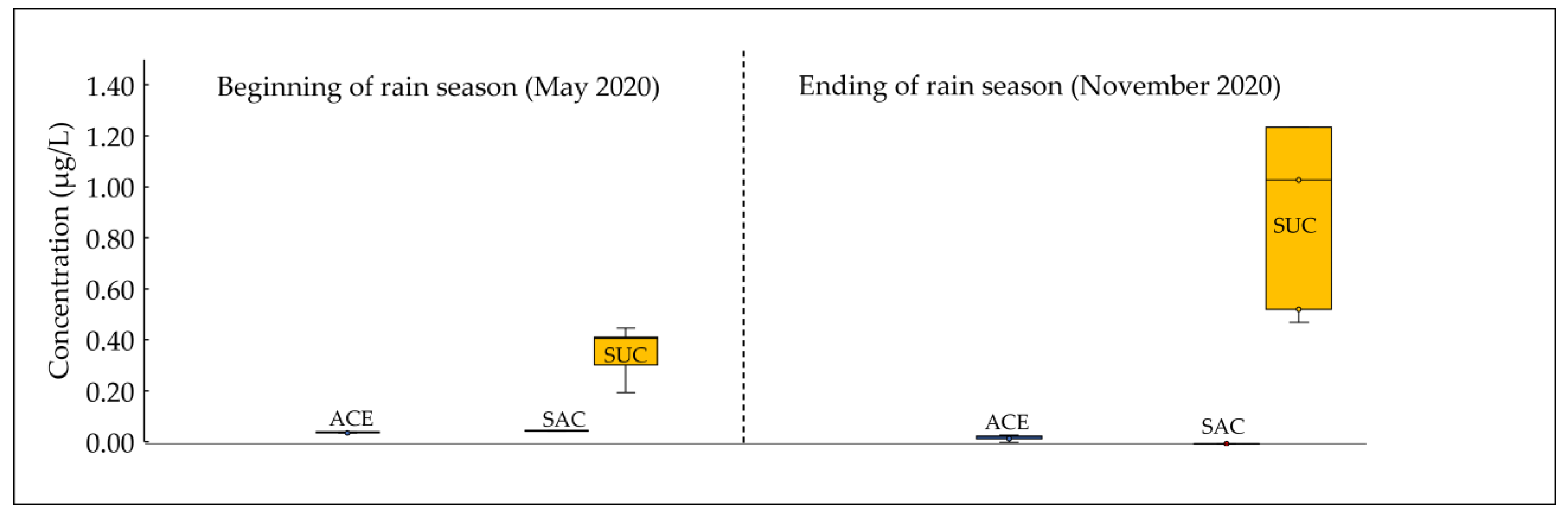
3.3. Temporal Variation in Asws Concentrations
4. Discussion
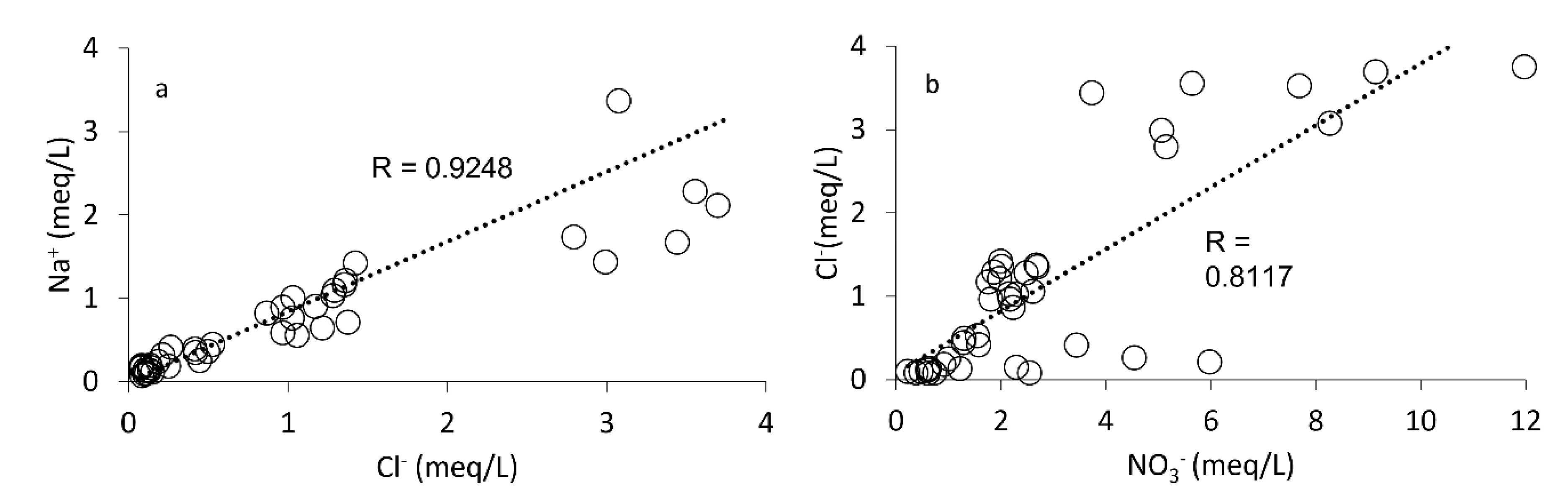
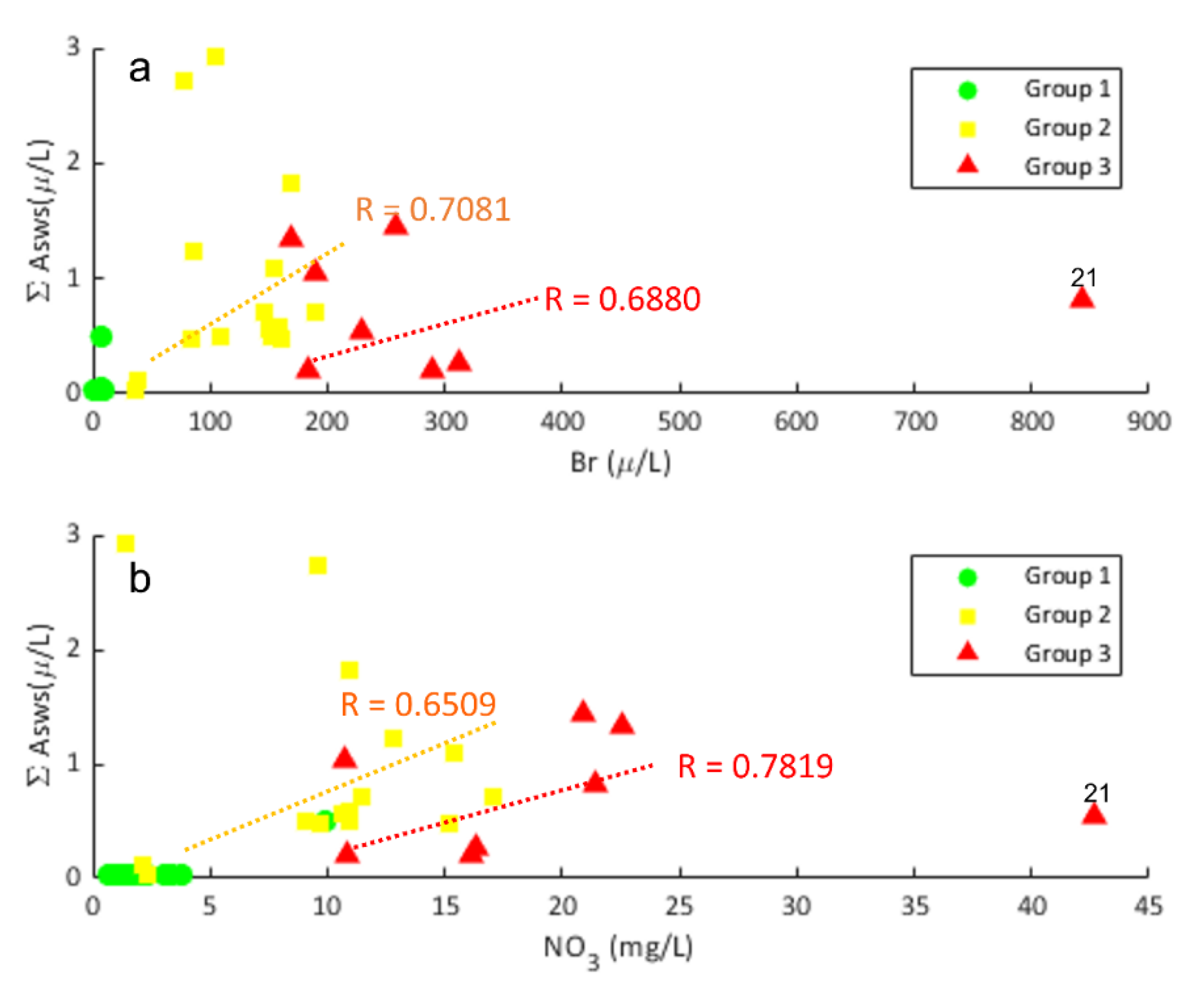
4.1. Association of Asws with Hydrochemistry
4.2. Suitability and Applicability of Asws as Wastewater Co-Tracers
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de la Peña, C. Artificial sweetener as a historical window to culturally situated health. Ann. N. Y. Acad. Sci. 2010, 1190, 159–165. [Google Scholar] [CrossRef]
- Lange, F.T.; Scheurer, M.; Brauch, H.J. Artificial sweeteners—A recently recognized class of emerging environmental contaminants: A review. Anal. Bioanal. Chem. 2012, 403, 2503–2518. [Google Scholar] [CrossRef] [PubMed]
- Belton, K.; Schaefer, E.; Guiney, P.D. A Review of the Environmental Fate and Effects of Acesulfame-Potassium. Integr. Environ. Assess. Manag. 2020, 16, 421–437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buerge, I.J.; Buser, H.R.; Kahle, M.; Muller, M.D.; Poiger, T. Ubiquitous occurrence of the artificial sweetener acesulfame in the aquatic environment: An ideal chemical marker of domestic wastewater in groundwater. Environ. Sci. Technol. 2009, 43, 4381–4385. [Google Scholar] [CrossRef] [PubMed]
- Scheurer, M.; Brauch, H.J.; Lange, F.T. Analysis and occurrence of seven artificial sweeteners in German wastewater and surface water and in soil aquifer treatment (SAT). Anal. Bioanal. Chem. 2009, 394, 1585–1594. [Google Scholar] [CrossRef]
- Gan, Z.; Sun, H.; Feng, B.; Wang, R.; Zhang, Y. Occurrence of seven artificial sweeteners in the aquatic environment and precipitation of Tianjin, China. Water Res. 2013, 47, 4928–4937. [Google Scholar] [CrossRef] [PubMed]
- van Stempvoort, D.R.; Roy, J.W.; Brown, S.J.; Bickerton, G. Artificial sweeteners as potential tracers in groundwater in urban environments. J. Hydrol. 2011, 401, 126–133. [Google Scholar] [CrossRef]
- Tran, N.H.; Hu, J.; Li, J.; Ong, S.L. Suitability of artificial sweeteners as indicators of raw wastewater contamination in surface water and groundwater. Water Res. 2014, 48, 443–456. [Google Scholar] [CrossRef] [PubMed]
- Spoelstra, J.; Senger, N.D.; Schiff, S.L. Artificial sweeteners reveal septic system effluent in rural groundwater. J. Environ. Qual. 2017, 46, 1434–1443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mawhinney, D.B.; Young, R.B.; Vanderford, B.J.; Borch, T.; Snyder, S.A. Artificial sweetener sucralose in US drinking water systems. Environ. Sci. Technol. 2011, 45, 8716–8722. [Google Scholar] [CrossRef] [PubMed]
- Wolf, L.; Zwiener, C.; Zemann, M. Tracking artificial sweeteners and pharmaceuticals introduced into urban groundwater by leaking sewer networks. Sci. Total Environ. 2012, 430, 8–19. [Google Scholar] [CrossRef]
- Gasser, G.; Rona, M.; Voloshenko, A.; Shelkov, R.; Tal, N.; Pankratov, I.; Elhanany, S.; Lev, O. Quantitative evaluation of tracers for quantification of wastewater contamination of potable water sources. Environ. Sci. Technol. 2010, 44, 3919–3925. [Google Scholar] [CrossRef] [PubMed]
- van Stempvoort, D.R.; Roy, J.W.; Grabuski, J.; Brown, S.J.; Bickerton, G.; Sverko, E. An artificial sweetener and pharmaceutical compounds as co-tracers of urban wastewater in groundwater. Sci. Total Environ. 2013, 461, 348–359. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.H.; Reinhard, M.; Khan, E.; Chen, H.; Nguyen, V.T.; Li, Y.; Goh, S.G.; Nguyen, Q.B.; Saeidi, N.; Gin, K.Y.H. Emerging contaminants in wastewater, stormwater runoff, and surface water: Application as chemical markers for diffuse sources. Sci. Total Environ 2019, 676, 252–267. [Google Scholar] [CrossRef]
- Yang, Y.-Y.; Liu, W.-R.; Liu, Y.-S.; Zhao, J.-L.; Zhang, Q.-Q.; Zhang, M.; Zhang, J.-N.; Jiang, L.-J.; Ying, G.G. Suitability of pharmaceuticals and personal care products (PPCPs) and artificial sweeteners (ASs) as wastewater indicators in the Pearl River Delta, South China. Sci. Total Environ. 2017, 590, 611–619. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spoelstra, J.; Schiff, S.L.; Brown, S.J. Septic systems contribute artificial sweeteners to streams through groundwater. J. Hydrol. 2020, 7, 100050. [Google Scholar] [CrossRef]
- Fu, K.; Wang, L.; Wei, C.; Li, J.; Zhang, J.; Zhou, Z.; Liang, Y. Sucralose and acesulfame as an indicator of domestic wastewater contamination in Wuhan surface water. Ecotoxicol. Environ. Saf. 2020, 189, 109980. [Google Scholar] [CrossRef]
- Oppenheimer, J.; Eaton, A.; Badruzzaman, M.; Haghani, A.W.; Jacangelo, J.G. Occurrence and suitability of sucralose as an indicator compound of wastewater loading to surface waters in urbanized regions. Water Res. 2011, 45, 4019–4027. [Google Scholar] [CrossRef]
- Scheurer, M.; Storck, F.R.; Graf, C.; Brauch, H.J.; Ruck, W.; Lev, O.; Lange, F.T. Correlation of six anthropogenic markers in wastewater, surface water, bank filtrate, and soil aquifer treatment. J. Environ. Monit. 2011, 13, 966–973. [Google Scholar] [CrossRef]
- Glória, M.B.A. Intense sweeteners and synthetic colorants. In Food Analysis by HPLC; Marcel Dekker Inc.: New York, NY, USA, 2000; pp. 523–573. [Google Scholar]
- Buerge, I.J.; Keller, M.; Buser, H.R.; Müller, M.D.; Poiger, T. Saccharin and other artificial sweeteners in soils: Estimated inputs from agriculture and households, degradation, and leaching to groundwater. Environ. Sci. Technol. 2011, 45, 615–621. [Google Scholar] [CrossRef]
- Subedi, B.; Lee, S.; Moon, H.B.; Kannan, K. Emission of artificial sweeteners, select pharmaceuticals, and personal care products through sewage sludge from wastewater treatment plants in Korea. Environ. Int. 2014, 68, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; O’Brien, J.W.; Tscharke, B.J.; Okoffo, E.D.; Mueller, J.F.; Sun, H.; Thomas, K.V. Artificial sweeteners in end-use biosolids in Australia. Water Res. 2021, 200, 117237. [Google Scholar] [CrossRef] [PubMed]
- Majewsky, M.; Cavalcanti, C.B.; Cavalcanti, C.P.; Horn, H.; Frimmel, F.H.; Abbt-Braun, G. Estimating the trend of micropollutants in lakes as decision-making support in IWRM: A case study in Lake Paranoá, Brazil. Environ. Earth Sci. 2014, 72, 4891–4900. [Google Scholar] [CrossRef]
- Alves, P.D.C.C.; Rodrigues-Silva, C.; Ribeiro, A.R.; Rath, S. Removal of low-calorie sweeteners at five Brazilian wastewater treatment plants and their occurrence in surface water. J. Environ. Manag. 2021, 289, 112561. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; Hawkes, C. Sweetening of the global diet, particularly beverages: Patterns, trends, and policy responses. Lancet Diabetes Endocrinol. 2016, 4, 174–186. [Google Scholar] [CrossRef] [Green Version]
- INEGI—Instituto Nacional de Estadística y Geografía. 2021. Censo de Población y Vivienda 2020. Available online: https://censo2020.mx/ (accessed on 6 June 2022).
- Martinez, M.A.R.; Werner, J. Research into the Quaternary sediments and climatic variations in NE Mexico. Quat. Int. 1997, 43, 145–151. [Google Scholar] [CrossRef]
- Montalvo-Arrieta, J.C.; Cavazos-Tovar, P.; de León, I.N.; Alva-Niño, E.; Medina-Barrera, F. Mapping seismic site clases in Monterrey metropolitan area, northeast Mexico. Bol. Soc. Geol. Mex. 2008, 60, 147–157. [Google Scholar] [CrossRef]
- Aguilar-Barajas, I.; Sisto, N.P.; Ramirez, A.I.; Magaña-Rueda, V. Building urban resilience and knowledge co-production in the face of weather hazards: Flash floods in the Monterrey Metropolitan Area (Mexico). Environ. Sci. Policy 2019, 99, 37–47. [Google Scholar] [CrossRef]
- CONAGUA. Reporte del Clima en México. Reporte Anual (2020). Estado de Nuevo León, México. 2020. Available online: https://smn.conagua.gob.mx/tools/DATA/Climatolog%C3%ADa/Diagn%C3%B3stico%20Atmosf%C3%A9rico/Reporte%20del%20Clima%20en%20M%C3%A9xico/Anual2020.pdf (accessed on 16 February 2022).
- Mahlknecht, J.; Alonso-Padilla, D.; Ramos, E.; Reyes, L.M.; Álvarez, M.M. The presence of SARS-CoV-2 RNA in different freshwater environments in urban settings determined by RT-qPCR: Implications for water safety. Sci. Total Environ. 2021, 784, 147183. [Google Scholar] [CrossRef]
- Torres-Martínez, J.A.; Mora, A.; Knappett, P.S.; Ornelas-Soto, N.; Mahlknecht, J. Tracking nitrate and sulfate sources in groundwater of an urbanized valley using a multi-tracer approach combined with a Bayesian isotope mixing model. Water Res. 2020, 182, 115962. [Google Scholar] [CrossRef]
- Jasso, J.A.S. Estudio Geotécnico-Geofísico del Comportamiento Dinámico del Subsuelo para el Área Metropolitana de Monterrey, Nuevo León, México. Ph.D. Thesis, Universidad Autónoma de Nuevo León, San Nicolás de los Garza, NL, Mexico, 2014. [Google Scholar]
- Mora, A.; Mahlknecht, J.; Rosales-Lagarde, L.; Hernández-Antonio, A. Assessment of major ions and trace elements in groundwater supplied to the Monterrey metropolitan area, Nuevo León, Mexico. Environ. Monit. Assess. 2017, 189, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DIN 38407-47:2017-07; German Standard Methods for the Examination of Water, Waste Water and Sludge—Jointly Determinable Substances (Group F)—Part 47: Determination of Selected Active Pharmaceutical Ingredients and Other Organic Substances in Water and Waste Water—Method Using High Performance Liquid Chromatography and Mass Spectrometric Detection (HPLC-MS/MS or -HRMS) after Direct Injection (F 47). German Institute for Standardization: Berlin, Germany, 2017.
- IBM Corp. IBM SPSS Statistics for Windows; Version 26.0; IBM Corp: Armonk, NY, USA, 2019. [Google Scholar] [CrossRef]
- Kynčlová, P.; Hron, K.; Filzmoser, P. Correlation between compositional parts based on symmetric balances. Math. Geosci. 2017, 49, 777–796. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum; World Health Organization, Ed.; World Health Organization: Geneva, Switzerland, 2017; Available online: https://www.who.int/publications/i/item/9789241549950 (accessed on 22 May 2022).
- Norma Oficial Mexicana NOM-086-SSA1-1994, Bienes y Servicios. Alimentos y Bebidas No Alcohólicas con Modificaciones en su Composición. Especificaciones Nutrimentales. Available online: https://www.dof.gob.mx/nota_detalle.php?codigo=4890075&fecha=26/06/1996#gsc.tab=0 (accessed on 3 March 2022).
- Pastén-Zapata, E.; Ledesma-Ruiz, R.; Harter, T.; Ramírez, A.I.; Mahlknecht, J. Assessment of sources and fate of nitrate in shallow groundwater of an agricultural area by using a multi-tracer approach. Sci. Total Environ. 2014, 470, 855–864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stefania, G.A.; Rotiroti, M.; Buerge, I.J.; Zanotti, C.; Nava, V.; Leoni, B.; Fumagalli, L.; Bonomi, T. Identification of groundwater pollution sources in a landfill site using artificial sweeteners, multivariate analysis and transport modeling. Waste Manag. 2019, 95, 116–128. [Google Scholar] [CrossRef] [PubMed]
- van Stempvoort, D.R.; Robertson, W.D.; MacKay, R.; Collins, P.; Brown, S.J.; Danielescu, S.; Pascoe, T. The role of groundwater in loading of nutrients to a restricted bay in a Precambrian Shield lake. Part 1.–Conceptual model and field observations. J. Great Lakes Res. 2021, 47, 1259–1272. [Google Scholar] [CrossRef]
- Martínez-Quiroga, G.E.; de León-Gómez, H.; Yépez-Rincón, F.D.; López-Saavedra, S.; Benavides, A.C.; Cruz-López, A. Alluvial Terraces and Contaminant Sources of the Santa Catarina River in the Monterrey Metropolitan Area, Mexico. J. Maps 2021, 17, 247–256. [Google Scholar] [CrossRef]
- Zirlewagen, J.; Licha, T.; Schiperski, F.; Nödler, K.; Scheytt, T. Use of two artificial sweeteners, cyclamate and acesulfame, to identify and quantify wastewater contributions in a karst spring. Sci. Total Environ. 2016, 547, 356–365. [Google Scholar] [CrossRef]
- Fetter, C.W. Applied Hydrogeology; Waveland Press: Long Grove, IL, USA, 2018. [Google Scholar]
- Jalali, M. Geochemistry characterization of groundwater in an agricultural area of Razan, Hamadan, Iran. Environ. Geol. 2019, 56, 1479–1488. [Google Scholar] [CrossRef]
- Meybeck, M. Global chemical weathering of surficial rocks estimated from river dissolved loads. Am. J. Sci. 1987, 287, 401–428. [Google Scholar] [CrossRef]
- Rahman, M.A.T.; Saadat, A.H.M.; Islam, M.; Al-Mansur, M.; Ahmed, S. Groundwater characterization and selection of suitable water type for irrigation in the western region of Bangladesh. Appl. Water Sci. 2017, 7, 233–243. [Google Scholar] [CrossRef] [Green Version]
- Panno, S.V.; Hackley, K.C.; Hwang, H.H.; Greenberg, S.E.; Krapac, I.G.; Landsberger, S.; O’kelly, D.J. Characterization and identification of Na-Cl sources in ground water. Ground Water 2006, 44, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.N.; Fabryka-Martin, J.T.; Wolfsberg, L.E. Variations of bromide in potable ground water in the United States. Ground Water 2004, 42, 902–909. [Google Scholar] [PubMed]
- Romo-Romo, A.; Almeda-Valdés, P.; Brito-Córdova, G.X.; Gómez-Pérez, F.J. Prevalencia del consumo de edulcorantes no nutritivos (ENN) en una población de pacientes con diabetes en México. Gac. Med. Mex. 2017, 153, 61–74. [Google Scholar]
- INEGI—Instituto Nacional de Estadística y Geografía, 2021. Directorio Estadístico Nacional de Unidades Económicas: DENUE Interactivo. Available online: https://www.inegi.org.mx/app/mapa/denue/default.aspx (accessed on 22 May 2022).
- Labare, M.P.; Alexander, M. Microbial cometabolism of sucralose, a chlorinated disaccharide, in environmental samples. Appl. Microbiol. Biotechnol. 1994, 42, 173–178. [Google Scholar] [CrossRef]
- Dickenson, E.R.; Snyder, S.A.; Sedlak, D.L.; Drewes, J.E. Indicator compounds for assessment of wastewater effluent contributions to flow and water quality. Water Res. 2011, 45, 1199–1212. [Google Scholar] [CrossRef]
- Vázquez-Tapia, I.; Salazar-Martínez, T.; Acosta-Castro, M.; Meléndez-Castolo, K.A.; Mahlknecht, J.; Cervantes-Avilés, P.; Capprelli, M.V.; Mora, A. Occurrence of emerging organic contaminants and endocrine disruptors in different water compartments in Mexico–A review. Chemosphere 2022, 308, 136285. [Google Scholar] [CrossRef] [PubMed]
- Sedlak, D.L.; Huang, C.H.; Pinkston, K. Strategies for selecting pharmaceuticals to assess attenuation during indirect potable water reuse. In Pharmaceuticals in the Environment; Springer: Berlin/Heidelberg, Germany, 2004; pp. 107–120. [Google Scholar]
- Ma, R.; Li, L.; Zhang, B. Impact assessment of anthropogenic activities on the ecological systems in the Xiongan New Area in the North China Plain. Integr. Environ. Assess. Manag. 2021, 17, 866–876. [Google Scholar] [CrossRef]
- Ferrer, I.; Thurman, E.M. Analysis of sucralose and other sweeteners in water and beverage samples by liquid chromatography/time-of-flight mass spectrometry. J. Chromatogr. A 2010, 1217, 4127–4134. [Google Scholar] [CrossRef]
- Richards, L.A.; Kumari, R.; White, D.; Parashar, N.; Kumar, A.; Ghosh, A.; Kumar, S.; Chakravorty, B.; Lu, C.; Civil, W.; et al. Emerging organic contaminants in groundwater under a rapidly developing city (Patna) in northern India dominated by high concentrations of lifestyle chemicals. Environ. Poll. 2021, 268, 115765. [Google Scholar] [CrossRef]
- Zhao, Z.; Yin, H.; Xu, Z.; Peng, J.; Yu, Z. Pin-pointing groundwater infiltration into urban sewers using chemical tracer in conjunction with physically based optimization model. Water Res. 2020, 175, 115689. [Google Scholar] [CrossRef]
- Hunter, B.; Walker, I.; Lassiter, R.; Lassiter, V.; Gibson, J.M.; Ferguson, P.L.; Deshusses, M.A. Evaluation of private well contaminants in an underserved North Carolina community. Sci. Total Environ. 2021, 789, 147823. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Kim, K.Y.; Hamm, S.Y.; Kim, M.; Kim, H.K.; Oh, J.E. Occurrence and distribution of pharmaceutical and personal care products, artificial sweeteners, and pesticides in groundwater from an agricultural area in Korea. Sci. Total Environ. 2019, 659, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Biel-Maeso, M.; González-González, C.; Lara-Martín, P.A.; Corada-Fernández, C. Sorption and degradation of contaminants of emerging concern in soils under aerobic and anaerobic conditions. Sci. Total Environ. 2019, 666, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Roy, J.W.; van Stempvoort, D.R.; Bickerton, G. Artificial sweeteners as potential tracers of municipal landfill leachate. Environ. Pollut. 2014, 184, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Propp, V.R.; Brown, S.J.; Collins, P.; Smith, J.E.; Roy, J.W. Artificial Sweeteners Identify Spatial Patterns of Historic Landfill Contaminated Groundwater Discharge in an Urban Stream. Groundw. Monit. Remediat. 2021, 42, 50–64. [Google Scholar] [CrossRef]
- Edwards, Q.A.; Sultana, T.; Kulikov, S.M.; Garner-O’Neale, L.D.; Metcalfe, C.D. Micropollutants related to human activity in groundwater resources in Barbados, West Indies. Sci. Total Environ. 2019, 671, 76–82. [Google Scholar] [CrossRef]
- Ishii, E.; Watanabe, Y.; Agusa, T.; Hosono, T.; Nakata, H. Acesulfame as a suitable sewer tracer on groundwater pollution: A case study before and after the 2016 Mw 7.0 Kumamoto earthquakes. Sci. Total Environ. 2020, 754, 142409. [Google Scholar] [CrossRef]
- Sharma, B.M.; Bečanová, J.; Scheringer, M.; Sharma, A.; Bharat, G.K.; Whitehead, P.G.; Klánová, J.; Nizzetto, L. Health and ecological risk assessment of emerging contaminants (pharmaceuticals, personal care products, and artificial sweeteners) in surface and groundwater (drinking water) in the Ganges River Basin, India. Sci. Total Environ. 2019, 646, 1459–1467. [Google Scholar] [CrossRef]
- Close, M.E.; Humphries, B.; Northcott, G. Outcomes of the first combined national survey of pesticides and emerging organic contaminants (EOCs) in groundwater in New Zealand 2018. Sci. Total Environ. 2021, 754, 142005. [Google Scholar] [CrossRef]

| Group | T (°C) | pH | EC (µS/cm) | ORP (mV) | DO (mg/L) | CO3− (mg/L) | HCO3− (mg/L) | F− (mg/L) | Cl− (mg/L) | NO2-N (mg/L) | NO3−-N (mg/L) | SO4− (mg/L) | PO4− (mg/L) | Ca2+ (mg/L) | K+ (mg/L) | Mg2+ (mg/L) | Na+ (mg/L) | Si (mg/L) | B (µg/L) | I (µg/L) | ACE (µg/L) | ASP (µg/L) | CYC (µg/L) | SAC (µg/L) | SUC (µg/L) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Detection limit | - | - | - | - | - | - | - | 0.01 | 0.03 | 0.02 | 0.01 | 0.03 | 0.04 | 10 | 0.6 | 0.2 | 1 | 4 | 3 | 0.2 | 0.01 | 0.01 | 0.01 | 0.01 | 0.05 | |
| 1 (n = 19) | 24 a | 8.3 a | 424.8 a | 374.1 a | 4.5 a | 150.8 a | 184.4 a | 0.24 a | 8.9 a | 0.01 a | 2.3 a | 66.1 a | 0.02 a | 78.5 a | 0.6 a | 8.8 a | 5.5 a | 4.7 a | 20.1 a | 2.7 a | 0.01 a | BQL | BQL | 0.005 a | 0.05 a | |
| σx | 3.6 | 0.5 | 81.3 | 102.2 | 1.1 | 114.6 | 19.2 | 0.2 | 8.5 | 0.0 | 2.1 | 62.6 | 0.0 | 33.7 | 0.2 | 3.5 | 3.7 | 1.9 | 14.1 | 1.4 | 0.1 | BQL | BQL | 0.003 | 0.1 | |
| Min. | 17.3 | 6.9 | 256.9 | 208 | 2.95 | 5.21 | 160 | 0 | 2.78 | 0.01 | 0.66 | 10.6 | 0.02 | 35.5 | 0.3 | 2.22 | 1.7 | 1.1 | 7 | 1.2 | BQL | BQL | BQL | BQL | BQL | |
| Max. | 30.9 | 9.1 | 549 | 572.1 | 6.56 | 549 | 230 | 0.61 | 36.4 | 0.02 | 9.94 | 287 | 0.04 | 197 | 0.9 | 15.3 | 17.6 | 9.1 | 56 | 7.8 | 0.4 | BQL | BQL | 0.017 | 0.5 | |
| 2 (n = 15) | 24.6 a | 7.7 b | 760.1 b | 317.9 a | 4.3 a | 86.6 a | 244.9 b | 0.14 a | 37.9 b | 0.02 b | 9.9 b | 116.3 a | 0.04 b | 102.3 a | 1.5 b | 14.4 a,b | 19.7 b | 7.9 b | 75.3 b | 7.5 a | 0.032 b | BQL | BQL | 0.006 a | 0.92 b | |
| σx | 1.0 | 0.6 | 61.5 | 82.5 | 13.7 | 84.1 | 61.6 | 0.1 | 12.2 | 0.0 | 4.8 | 35.3 | 0.0 | 33.1 | 0.6 | 7.0 | 7.0 | 2.4 | 46.5 | 3.7 | 0.0 | BQL | BQL | 0.004 | 0.9 | |
| Min. | 22.9 | 6.8 | 690 | 211.1 | 1.94 | 4.52 | 200 | 0 | 9.3 | 0.02 | 1.36 | 84.1 | 0.04 | 51.6 | 0.62 | 4.19 | 9 | 4.8 | 15 | 2.2 | BQL | BQL | BQL | BQL | BQL | |
| Max. | 26.6 | 8.4 | 903 | 451.1 | 6.42 | 314.9 | 460 | 0.21 | 50.4 | 0.02 | 17.1 | 218 | 0.04 | 189 | 2.3 | 25.8 | 32.8 | 11.2 | 180 | 13.7 | 0.081 | BQL | BQL | 0.022 | 2.9 | |
| 3 (n = 8) | 25.6 a | 7.4 b | 1410.9 c | 249.3 a | 3.3 a | 49.6 a | 247.5 b | 0.4 b | 118.9 c | 0.05 c | 20.2 c | 340 b | 0.01 c | 148.5 b | 1.6 b | 25.1 b | 64.8 c | 6.6 a,b | 179.8 c | 27 b | 0.45 b | BQL | BQL | 0.008 a | 0.67 b | |
| σx | 2.3 | 0.6 | 284.1 | 33.8 | 0.9 | 61.2 | 32.4 | 0.1 | 12.6 | 0.0 | 10.2 | 129.8 | 0.0 | 62.2 | 0.8 | 16.8 | 33.8 | 6.5 | 105.5 | 28.1 | 0.1 | BQL | BQL | 0.5 | 0.5 | |
| Min. | 23.8 | 6.8 | 1150 | 229.4 | 1.5 | 3.9 | 195 | 0.16 | 99.0 | 0.05 | 10.7 | 179 | 0.01 | 81.9 | 0.6 | 9 | 33.1 | 0.5 | 57 | 8 | 0.029 | BQL | BQL | BQL | 0.098 | |
| Max. | 29.3 | 8.3 | 1978 | 332 | 4.66 | 160.2 | 295 | 0.7 | 113 | 0.05 | 42.7 | 575 | 0.01 | 275 | 3.1 | 62.9 | 125 | 17.8 | 389 | 95.6 | 0.73 | BQL | BQL | 1.4 | 1.4 | |
| Guideline/Standard | 6.5–8.5 *,+ | 1500 * | - | 6.5–8 * | - | 250 */500 + | 1.5 *,+ | 250 *,+ | 3 */0.9 + | 50 */11.0 + | 250 */400 + | 0.04 * | 300 */500 + | 12 * | 150 * | 200 *,+ | 50 * | 2400 * | 18 * | Not regulated | Not regulated | Not regulated | Not regulated | Not regulated |
| Group | ACE | ASP | CYC | SAC | SUC |
|---|---|---|---|---|---|
| 1 | 10.5% (0.5) | BQL (n/a) | BQL (n/a) | 5.3% (0.5) | 5.3% (0.5) |
| 2 | 93.3% (3) | BQL (n/a) | BQL (n/a) | 13.3% (0.5) | 93.3% (10.8) |
| 3 | 100.0% (4) | BQL (n/a) | BQL (n/a) | 25.0% (0.5) | 100.0% (12.6) |
| Country | Location | Well Samples (n) | Concentration (µg/L) | References | ||||
|---|---|---|---|---|---|---|---|---|
| ACE | SUC | CYC | SAC | ASP | ||||
| Mexico | MMA, Nuevo León | 42 | <QL–0.1 | <QL–2.9 | <QL–0.009 | <QL–0.0052 | <QL | This study |
| United States | Wake County, North Carolina | 12 | - | 0.002–0.15 | - | - | - | [62] |
| Various locations | 8 | - | 0.6–2.4 | - | N.D. | N.D. | [59] | |
| Canada | Poplar Bay, Ontario | 55 | 0.004–11.3 | N.D.–7.8 | - | - | - | [43] |
| Barrie and Jasper sites | 53 | 2.5 | - | 0.046 | 0.035 | - | [13] | |
| Southern Ontario | 188 | 0.225 | 0.291 | 0.204 | 0.38 | - | [16] | |
| Various locations | 48 | 5.7 | - | 0.02 | 0.009 | - | [65] | |
| Dyment’s Creek, Barrie, Ontario | 60 | 0.1 | 0.05 | 0.1 | 0.05 | - | [66] | |
| West Indies | Barbados | 10 | 0.12 | 0.006 | <QL | 0.005 | - | [67] |
| Germany | Rastatt urban area | 50 | 0.170–2.9 | <QL–0.005 | 0.006–1.2 | <QL–0.001 | <QL–0.002 | [11] |
| Karlsruhe | 12 | 0.007 | 0.006 | - | - | - | [5] | |
| Switzerland | Zurich | 100 | 4.7 | - | - | - | - | [4] |
| Italy | Aosta Plain | 37 | 0.68–9.69 | 1.75 | 0.14–29.56 | 0.68–5.44 | - | [42] |
| China | Xiongan New Area, Beijing | 44 | 0.005–1.34 | 0.003–3.16 | <QL–0.30 | <QL–0.32 | - | [58] |
| Dongiang River Basin | 11 | 0.012–4.5 | 0.054–2.4 | 0.002–0.110 | 0.015–0.385 | - | [15] | |
| Tianjin | 3 | 0.68 | 0.46 | 0.9 | 2.26 | - | [6] | |
| Japan | Kumamoto area | 49 | 0.003 | 0.002 | - | 0.001 | - | [68] |
| Singapore | Urban catchment | 138 | <QL–0.09 | <QL | <QL–0.087 | <QL–0.021 | <QL | [8] |
| South Korea | Geumjeong, Kyungsang | 4 | 0.090–1.3 | N.D. | N.D. | 0.005–0.025 | N.D. | [63] |
| India | Ganges River Basin | 14 | <QL–0.002 | 0.005–0.002 | <QL–0.003 | N.D. | - | [69] |
| Patna and Ballia districts | 42 | 0.051–0.76 | 0.019–1.2 | - | 0.001–0.061 | - | [60] | |
| New Zealand | Various locations | 18 | - | 0.1 | - | - | - | [70] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramos, E.; Padilla-Reyes, D.; Mora, A.; Barrios-Piña, H.; Kant, S.; Mahlknecht, J. Assessment of Artificial Sweeteners as Wastewater Co-Tracers in an Urban Groundwater System of Mexico (Monterrey Metropolitan Area). Water 2022, 14, 3210. https://doi.org/10.3390/w14203210
Ramos E, Padilla-Reyes D, Mora A, Barrios-Piña H, Kant S, Mahlknecht J. Assessment of Artificial Sweeteners as Wastewater Co-Tracers in an Urban Groundwater System of Mexico (Monterrey Metropolitan Area). Water. 2022; 14(20):3210. https://doi.org/10.3390/w14203210
Chicago/Turabian StyleRamos, Edrick, Diego Padilla-Reyes, Abrahan Mora, Hector Barrios-Piña, Shashi Kant, and Jürgen Mahlknecht. 2022. "Assessment of Artificial Sweeteners as Wastewater Co-Tracers in an Urban Groundwater System of Mexico (Monterrey Metropolitan Area)" Water 14, no. 20: 3210. https://doi.org/10.3390/w14203210








