Modification of Disinfection Process at a Local Water Treatment Plant—Skawina (Poland)
Abstract
:1. Introduction
2. Materials and Methods
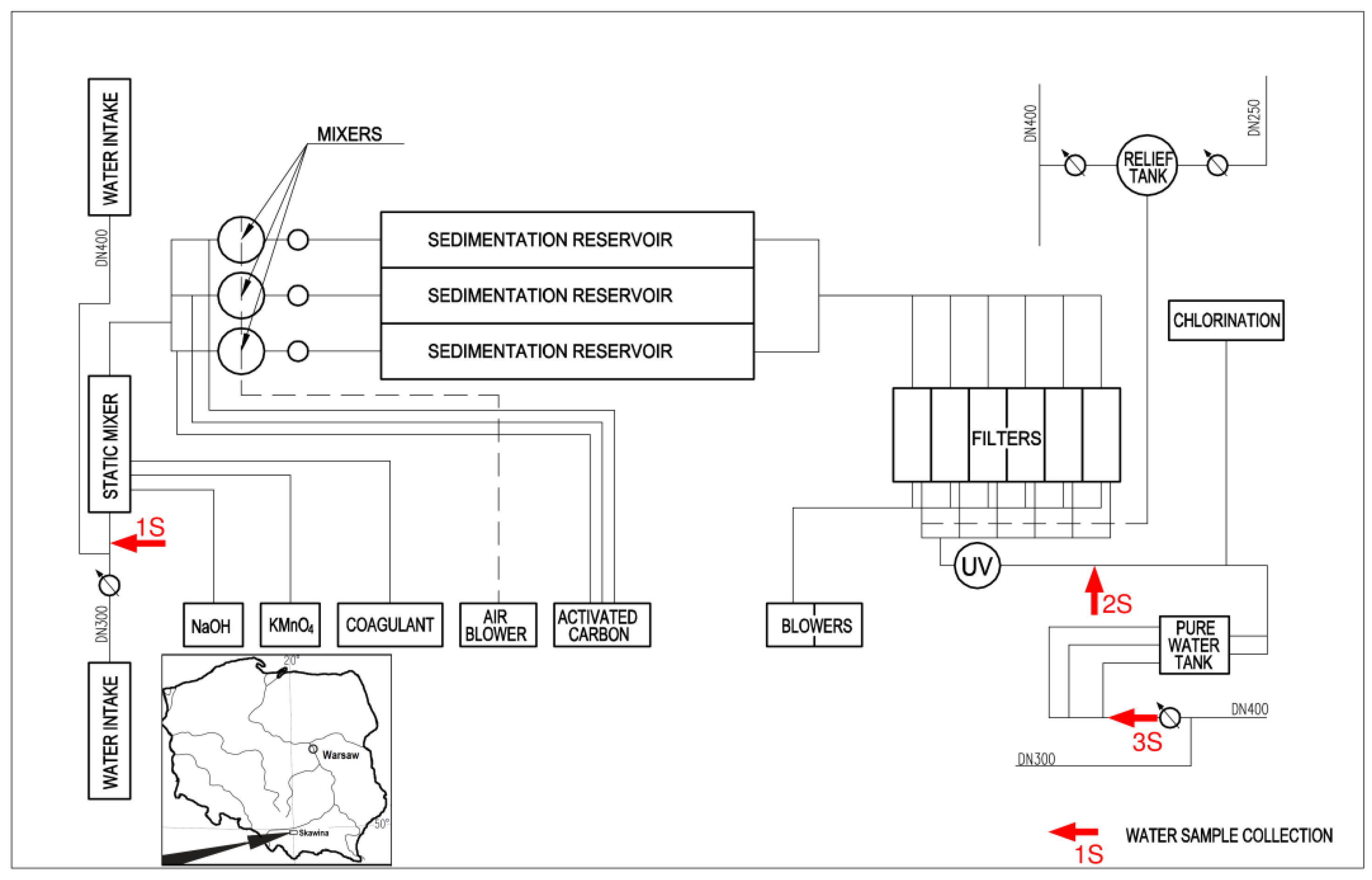
2.1. Water Treatment System at the WTP Skawina
2.2. Raw Water Quality at WTP Skawina
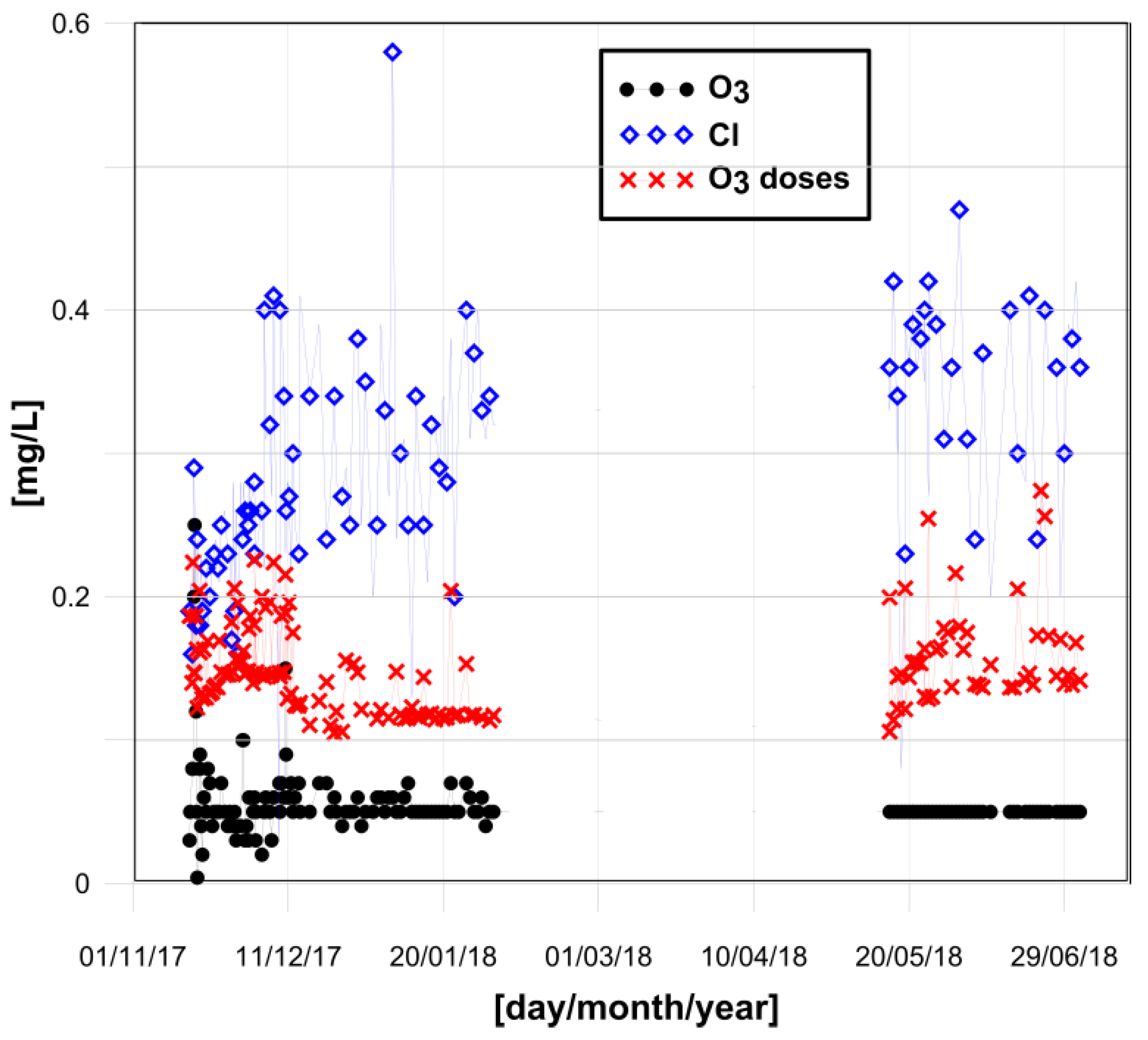
2.3. Ozonation Step as a Modification to the Water Disinfection Process
- To enhance the bacteriostatic action within the public water supply system;
- To minimize the dosage of chlorine added to water;
- To remove any odors remaining after the water treatment processes;
- To ensure a better taste of water supplied to the end users.
2.4. Water Quality Monitoring at the WTP Skawina Prior to and after Modifications of the Disinfection Process
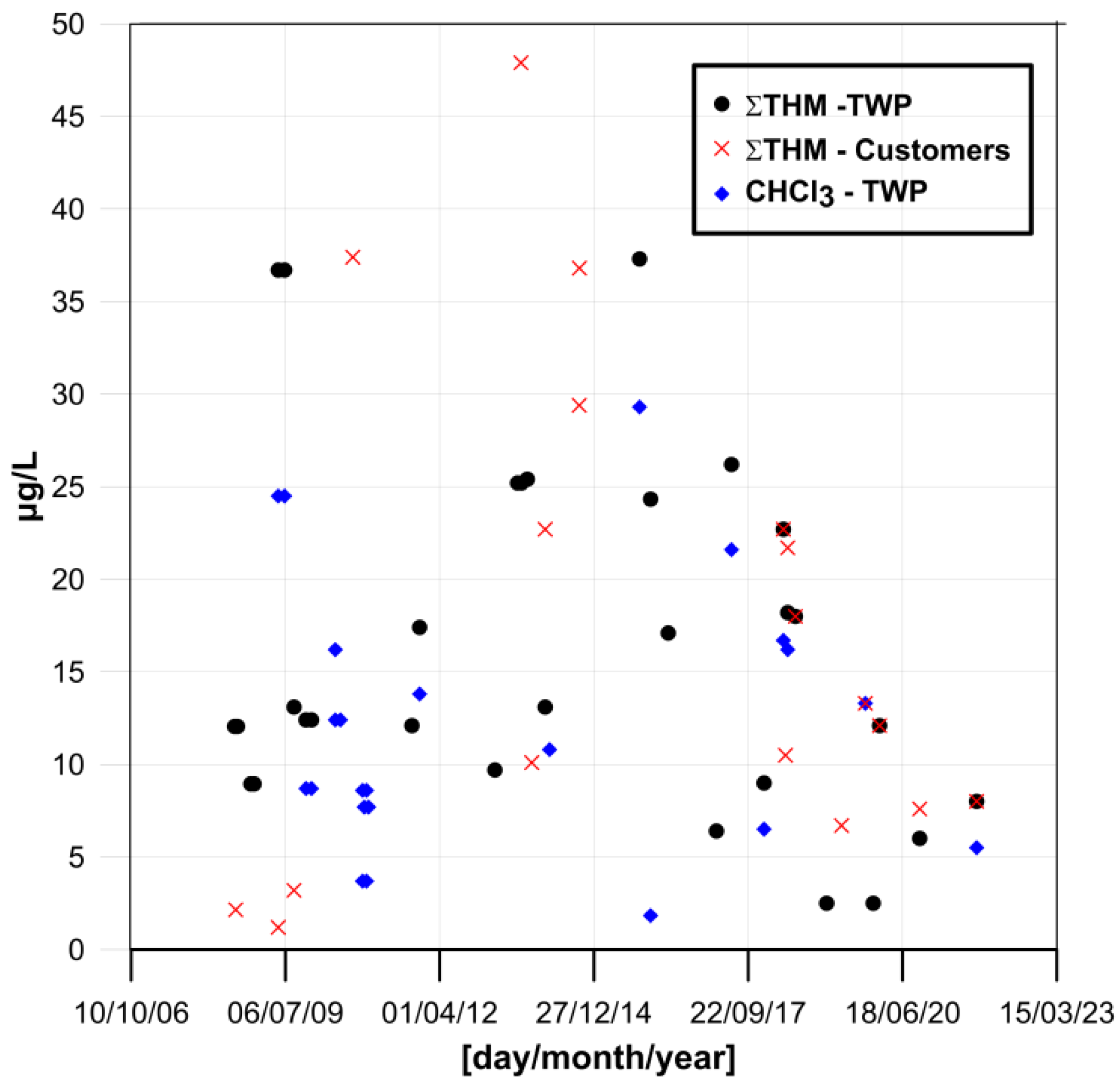
3. Results and Discussion
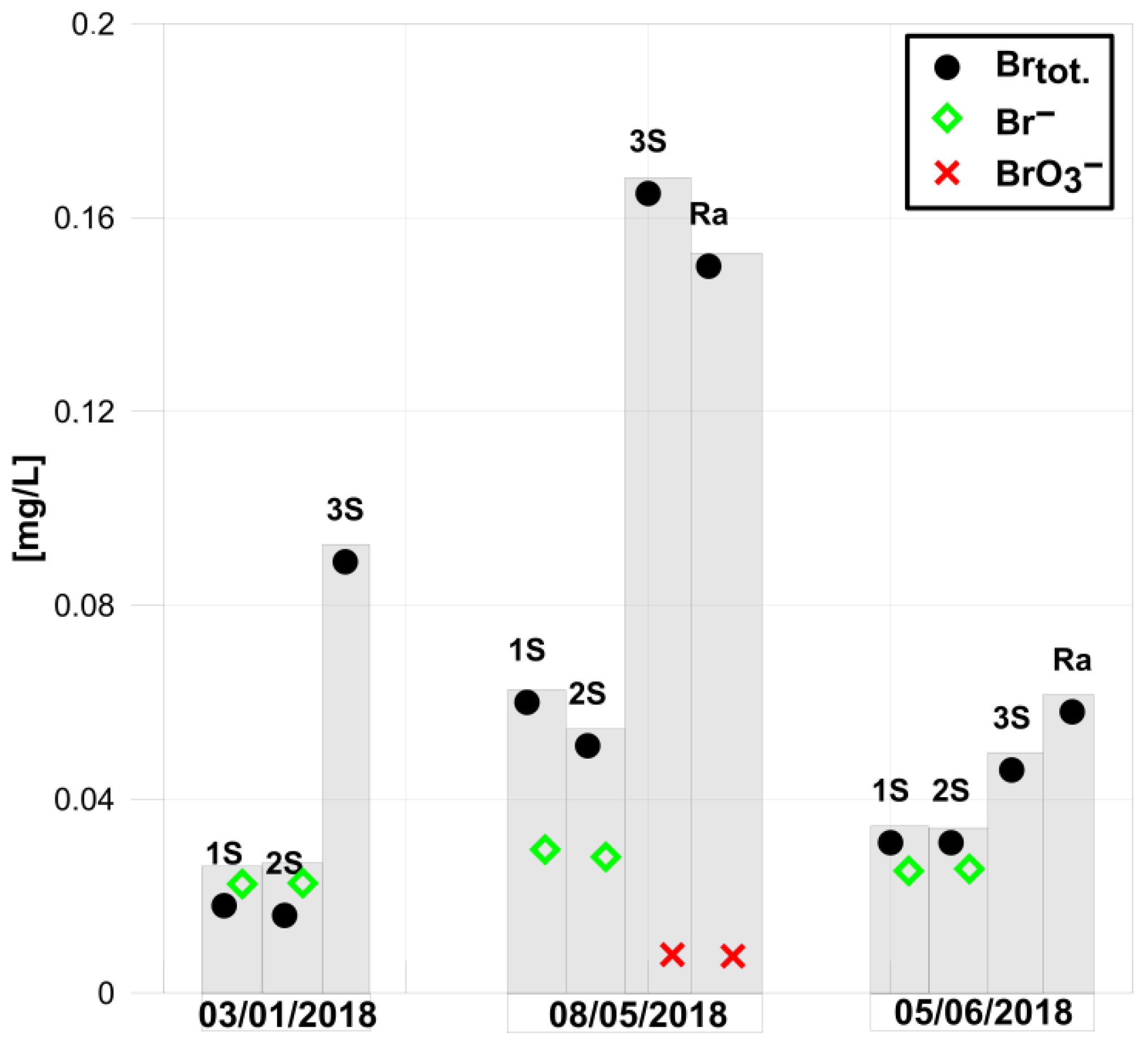
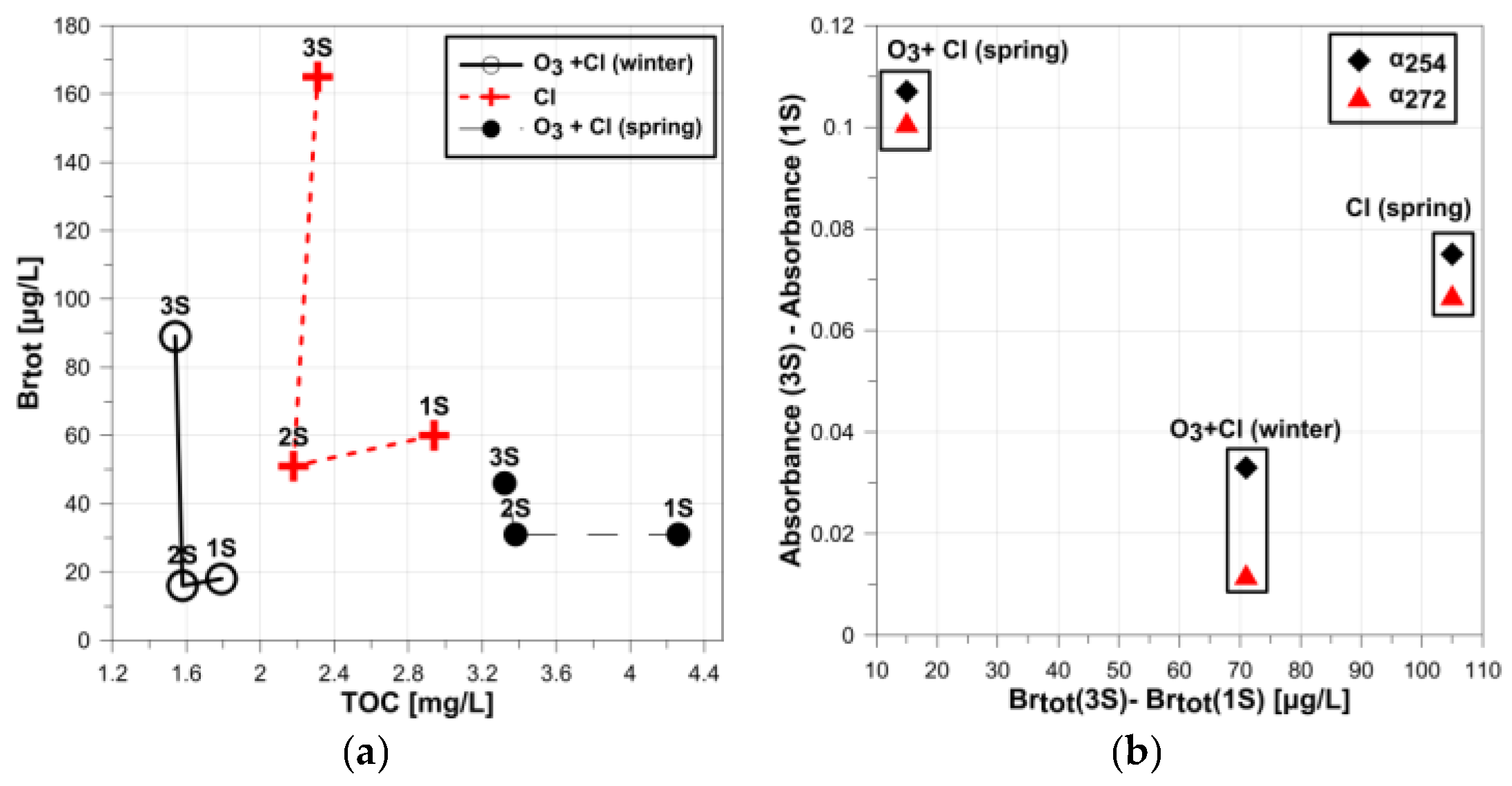
- Free bromine and no bromates were found in water samples after disinfection (3S) and in water samples collected at users’ homes (Ra);
- Elevated total bromine contents in relation to samples 1S and 2S were detected in samples after disinfection (3S and Ra) (Figure 5);
- In raw water samples (1S) and in water collected before disinfection (2S), bromine ions and total bromine contents were at similar levels, which indicates that bromine will occur in the form of ions in water.
- Less space needed for the assembly of the system;
- The possibility of moving to another place in the event of the necessity to use it on another section of the water supply network;
- Short investment time;
- Lower investment costs;
- Ease of system operation, process management and control;
- The ability to precisely dose ozone to water.
4. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tsitsifli, S.; Kanakoudis, V. Disinfection Impacts to Drinking Water Safety—A Review. Proceedings 2018, 2, 603. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Parveen, T.; Umar, K.; Mohamad Ibrahim, M.N. Role of nanomaterials in the treatment of wastewater: A review. Water 2020, 12, 495. [Google Scholar] [CrossRef]
- Setty, K.; Loret, J.; Courtois, S.; Hammer, C.; Hartemann, P.; Lafforgue, M.; Bartram, J. Faster and safer: Research priorities in water and health. Int. J. Hyg. Environ. Health 2019, 222, 539–606. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, A.A.; Ahmad, H.; Parveen, T.; Ahmad, A.; Oves, M.; Ismail, I.M.; Qari, H.A.; Umar, K.; Ibrahim, M.N.M. Recent advances in metal decorated nanomaterials and their various biological applications: A review. Front. Chem. 2020, 8, 341. [Google Scholar] [CrossRef] [PubMed]
- Tsitsifli, S.; Kanakoudis, V. Determining hazards’ prevention critical control points in water supply systems. Environ. Sci. Proc. 2020, 2, 53. [Google Scholar] [CrossRef]
- Tsitsifli, S.; Kanakoudis, V. Assessing the impact of DMAs and the use of boosters on chlorination in a water distribution network in Greece. Water 2021, 13, 2141. [Google Scholar] [CrossRef]
- Tsitsifli, S.; Kanakoudis, V. Identification of Suitable Locations in a Small Water Supply Network for the Placement of Water Quality Sensors Based on Different Criteria under Demand-Driven Conditions. Water 2022, 14, 2504. [Google Scholar] [CrossRef]
- Biń, A. Application of ozonation and advanced oxidation processes for drinking water and wastewater treatment in Poland. Rocz. Ochr. Sr. 1999, 1, 7–25. Available online: https://ros.edu.pl/images/roczniki/archive/pp_1999_001.pdf (accessed on 10 March 2022).
- Trussell, R.R. Water treatment the past 30 years. J. Am. Water Works Ass. 2006, 98, 100–108. [Google Scholar] [CrossRef]
- Lim, S.; Shi, J.L.; von Gunten, U.; McCurry, D.L. Ozonation of organic compounds in water and wastewater: A critical review. Water Res. 2022, 213, 118053. [Google Scholar] [CrossRef] [PubMed]
- Anselme, C.; Suddet, I.H.; Mallevialle, J. Effects of ozonation on tastes and odors. J. Am. Water Works Ass. 1988, 80, 45–51. [Google Scholar] [CrossRef]
- Gao, J.; Proulx, F.; Rodriguez, F.M.J. Effects of ozonation on halogenated acetaldehydes and trihalomethanes formation: Strategy of process control for a full-scale plant. J. Water. Process. Eng. 2020, 35, 101205. [Google Scholar] [CrossRef]
- Richardson, S.D.; Plewa, M.; Wagner, E.; Schoeny, R.; De Marini, D. Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: A review and roadmap for research. Mutat. Res. 2007, 636, 178–242. [Google Scholar] [CrossRef] [PubMed]
- Hebert, A.; Forestier, D.; Lenes, D.; Benanou, D.; Jacob, S.; Arfi, C.; Lambolez, L.Y.; Levi, Y. Innovative method for prioritizing emerging disinfection by-products (DBPs) in drinking water on the basis of their potential impact on public health. Water Res. 2010, 44, 3147–3165. [Google Scholar] [CrossRef]
- Tugulea, A.M.; Aranda-Rodriguez, R.; Bérube, D.; Giddings, M.; Lemieux, F.; Hnatiw, J.; Dabeka, L.; Breton, F. The influence of precursors and treatment process on the formation of Iodo-THMs in Canadian drinking water. Water Res. 2018, 130, 215–223. [Google Scholar] [CrossRef]
- IARC International Agency for Research on Cancer. Agents Classified by the IARC Monographs, V.1–132 at 2022. Available online: https://monographs.iarc.who.int/agents-classified-by-the-iarc/ (accessed on 7 April 2022).
- Regulation on the Quality of Water Intended for Human Consumption. Journal of Laws, 2017 Item 2294. Available online: https://www.ecolex:details/legislation/regulation-on-the-quality-of-water-intended-for-human-consumption-lex-faoc182571/ (accessed on 5 May 2022).
- Chowdhury, S.; Champagne, P.; McLellan, J. Models for predicting disinfection byproduct (DBP) formation in drinking waters: A chronological review. Sci. Total Environ. 2009, 407, 4189–4206. [Google Scholar] [CrossRef] [PubMed]
- Chaukura, C.; Marais, S.S.; Moyo, W.; Mbali, N.; Thakalekoala, L.C.; Ingwani, T.; Mamba, B.B.; Jarvis, P.; Nkambule, T.T.I. Contemporary issues on the occurrence and removal of disinfection byproducts in drinking water—A review. J. Environ. Chem. Eng. 2020, 8, 103659. [Google Scholar] [CrossRef]
- Khan, F.; Zuthi, M.F.R.; Hossain, M.D.; Bhuiyan, M.N.I. Prediction of trihalomethanes in water supply of Chattogram city by empirical models and cancer risk through multi-pathway exposure. J. Water Process Eng. 2021, 42, 102165. [Google Scholar] [CrossRef]
- Stefán, D.; Balogh, J.; Záray, G.; Vargha, M. Comparison of disinfection by-product formation and distribution during breakpoint chlorination and chlorine-based disinfection in drinking water. Water 2022, 14, 1372. [Google Scholar] [CrossRef]
- Von Gunten, U. Ozonation of drinking water: Part II, Disinfection and by-product formation in presence of bromide iodide or chloride. Water Res. 2003, 37, 1469–1487. [Google Scholar] [CrossRef]
- Von Gunten, U. Oxidation processes in water treatment: Are we on track? Environ. Sci. Technol. 2018, 52, 5062–5075. [Google Scholar] [CrossRef]
- Wang, Y.; Man, T.; Zhang, R.; Yan, X.; Wang, A.; Zhang, M.; Wang, P.; Ren, L.; Yu, J.; Li, C. Effects of organic matter, ammonia, bromide, and hydrogen peroxide on bromate formation during water ozonation. Chemosphere 2021, 285, 131352. [Google Scholar] [CrossRef] [PubMed]
- Biń, A.; Możaryn, W. Bromate problem—Current state of research and advances in water treatment technologies. Ochr. Sr. 1999, 21, 49–56. Available online: http://www.os.not.pl/docs/czasopismo/1999/Bin_3-1999.pdf (accessed on 10 March 2022).
- Jarvis, P.; Parsons, S.A.; Smith, R. Modeling bromate formation during ozonation. Ozone-Sci. Eng. 2007, 29, 429–442. [Google Scholar] [CrossRef]
- Olsińska, U.; Kuś, K. Ozonation by-products in bromide-containing water. Ochr. Sr. 1997, 19, 33–38. Available online: http://www.os.not.pl/docs/czasopismo/1997/Olsinska_3-1997.pdf (accessed on 10 March 2022).
- Welté, B.; Montiel, A. Evolution of bromates and organobrominated compounds in two drinking water treatment plants. Ozone Sci. Eng. 2001, 23, 229–238. [Google Scholar] [CrossRef]
- Sorlini, S.; Collivignarelli, C. Trihalomethanes formation during chemical oxidation with chlorine, chlorine dioxide and ozone of ten Italian natural waters. Desalination 2005, 176, 103–111. [Google Scholar] [CrossRef]
- Uyak, V.; Toroz, I.; Meric, S. Monitoring and modeling, of trihalomethanes (THMs) for a water treatment plant in Istanbul. Desalination. 2005, 176, 91–101. [Google Scholar] [CrossRef]
- Wert, E.C.; Rosario-Ortiz, F.L. Effect of ozonation on trihalomethane and haloacetic acid formation and speciation in a full-scale distribution system. Ozone Sci. Eng. 2011, 33, 14–22. [Google Scholar] [CrossRef]
- Włodyga-Bergier, A.; Bergier, T. Impact of change in water disinfection method on chlorination by-products occurrence in Raba water distribution system in Krakow. Ochr. Sr. 2015, 37, 19–23. Available online: http://www.os.not.pl/docs/czasopismo/2015/3-2015/Wlodyka_3-2015.pdf (accessed on 10 March 2022).
- Nguyen, P.D.; Lea, T.D.Q.; Nguyen, N.H.; Tranc, K.T.; Nguyen, M.T.; Huynh, K.A. Reducing disinfection byproduct precursors and chlorine consuming substances by a special integration of biofiltration and ozonation: A pilot study. J. Water Process Eng. 2020, 37, 101419. [Google Scholar] [CrossRef]
- Bernstein, A.; Studny, R.; Shyntychea, V.; Kurtzman, D.; Ganot, Y.; Katz, Y.; Asfaw, B.A.; Sakaguchi-Söder, K.; Schüth, C.; Siebner, H. Low trihalomethane formation during managed aquifer recharge with chlorinated desalinated water. Water 2020, 12, 711. [Google Scholar] [CrossRef]
- Zin, N.N.; Kazama, S.; Takizawa, S. Network model analysis of residual chlorine to reduce disinfection byproducts in water supply systems in Yangon city, Myanmar. Water 2021, 13, 2921. [Google Scholar] [CrossRef]
- Aranda-Rodriguez, R.; Lemieux, F.; Jin, Z.; Hnatiw, J.; Tugulea, A.M. (Yet more) challenges for water treatment plants: Potential contribution of hypochlorite solutions to bromate, chlorate, chlorite and perchlorate in drinking water. J. Water Supply Res. Technol. 2017, 66, 621–631. [Google Scholar] [CrossRef]
- Chowdhury, S. Occurrences and changes of disinfection by-products in small water supply systems. Environ. Monit. Assess. 2018, 190, 1–19. [Google Scholar] [CrossRef]
- Ali, S.I.; Arnold, M.; Liesner, F.; Fesselet, J.F. Characterization of disinfection by-products levels at an emergency surface water treatment plant in a Refugee settlement in northern Uganda. Water 2019, 11, 647. [Google Scholar] [CrossRef]
- Sharma, N.; Mohapatra, S.; Padhye, L.P.; Mukherji, S. Role of precursors in the formation of trihalomethanes during chlorination of drinking water and wastewater effluents from a metropolitan region in western India. J. Water Process Eng. 2021, 40, 101928. [Google Scholar] [CrossRef]
- Nobukawa, T.; Sanukida, S. The genotoxicity of by-products by chlorination and ozonation of the river water in the presence of bromide ions. Water Sci. Technol. 2000, 42, 259–264. [Google Scholar] [CrossRef]
- Myllykangas, T.; Nissinen, T.K.; Mäki-Paakkanen, J.; Hirvonen, A.; Vartiainen, T. Bromide affecting drinking water mutagenicity. Chemosphere 2003, 53, 745–756. [Google Scholar] [CrossRef]
- Zbieć, E.; Dojlido, J. By-products of water disinfection. Ochr. Sr. 1999, 21, 37–44. Available online: http://www.os.not.pl/docs/czasopismo/1999/Zbiec_3-1999.pdf (accessed on 10 March 2022).
- Amjad, H.; Imran, H.; Rehman, M.S.U.; Awan, M.A.; Ghaffar, S.; Khan, Z. Cancer and non-cancer risk assessment of trihalomethanes in urban drinking water supplies in Pakistan. Ecotoxicol. Environ. Safe 2013, 91, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Tsitsifli, S.; Kanakoudis, V. Developing THMs’ Predictive models in two water supply systems in Greece. Water 2020, 12, 1422. [Google Scholar] [CrossRef]
- Tsitsifli, S.; Kanakoudis, V. Total and Specific THMs’ Prediction Models in Drinking Water Pipe Networks. Environ. Sci. Proc. 2020, 2, 55. [Google Scholar] [CrossRef]
- Von Gunten, U. Ozonation of drinking water: Part I, Oxidation kinetics and product formation. Water Res. 2003, 37, 1443–1467. [Google Scholar] [CrossRef]
- Weinberg, H.S.; Delcomyn, C.A.; Unnam, V. Bromate in chlorinated drinking waters: Occurrence and implications for future regulation. Environ. Sci. Technol. 2003, 37, 3104–3110. [Google Scholar] [CrossRef]
- Bouland, S.; Duguet, J.P.; Montiel, A. Evaluation of bromate ions level introduced by sodium hypochlorite during post-disinfection of drinking water. Environ. Technol. 2005, 26, 121–125. [Google Scholar] [CrossRef]
- Asami, M.; Kosaka, K.; Kunikane, S. Bromate, chlorate, chlorite and perchlorate in sodium hypochlorite solution used in water supply. J. Water Supply Res. Technol. 2009, 58, 107–115. [Google Scholar] [CrossRef]
- Garcia-Villanova, R.J.; Leite, M.V.O.D.; Hierro, J.M.H.; De Castro Alfageme, S.; Hernández, C.G. Occurrence of bromate, chlorite and chlorate in drinking waters disinfected with hypochlorite reagents. Tracing their origins. Sci. Total Environ. 2010, 408, 2616–2620. [Google Scholar] [CrossRef]
- Dąbrowska, A. Possible reasons for elevated aldehyde levels in surface waters. Ochr. Sr. 2013, 33, 13–16. [Google Scholar]
- Formaldehyde in Drinking-Water, Geneva: World Health Organization (WHO). 2005. Available online: https://www.who.int/water_sanitation_health/dwq/chemicals/formaldehyde.pdf.p.6 (accessed on 31 March 2022).
- WHO. Guidelines for Drinking Water Quality, 4th ed.; Incorporating The First Addendum; World Health Organization: Geneva, Switzerland, 2017; ISBN 978-92-4-154995-0. [Google Scholar]
- Water Treatment Plan Skawina. Available online: http://zwik,skawina,pl/produkcja_wody,opis_technologii (accessed on 10 March 2022).
- EPP—Environmental Protection Program for the Skawina Commune for the Years 2014–2017 with a Perspective for 2018–2021. 2014. Available online: https://bip,malopolska,pl/pobierz/1074497.html (accessed on 31 March 2022).
- Regulation on Requirements for Surface Waters Used for Human Consumption. Journal of Laws 2019; Item 1747. Available online: https://www.ecolex:details/legislation/regulation-on-requirements-for-surface-waters-used-for-human-consumption-lex-faoc192325/ (accessed on 5 May 2022).
- Chowaniec, J.; Witek, K. Hydrogeological Map of Poland, 1:50,000, Myślenice (996), with Commentary; PIG: Warsaw, Poland, 1997. Available online: https://geolog.pgi.gov.pl/ (accessed on 10 March 2022).
- The Quality of Potable Water. 2017–2019 The Chief Sanitary Inspector. Available online: https://www.gov.pl/web/gis/raport-o-jakosci-wody-przeznaczonej-do-spozycia-przez-ludzi-za-lata-2017–2019 (accessed on 10 March 2022).
- Witczak, S.; Kania, J.; Kmiecik, E. Guidebook on Selected Physical and Chemical Indicators of Groundwater Contamination and Methods of Their Determination; Institute of Environmental Protection: Warsaw, Poland, 2013; p. 717. ISBN 9788361227137. [Google Scholar]
- Rice, A.; Baird, E.W.; Eaton, R.B. Apha 2017 Standard Methods for Examination of Water and Wastewater; American Public Health Association; American Water Works Association; Water Environment Federation: Washington, DC, USA, 2017; p. 1360. [Google Scholar]
- Sanepid: Database for Małopolska Region; Sanitary-Facility, WSSE: Kraków, Poland, [unpublished].
- Mołczan, M.; Szlachta, M.; Karpińska, A.; Biłyk, A. Water quality assessment in terms of specific UV absorbance. Ochr. Sr. 2006, 28, 11–16. Available online: http://www.os.not.pl/docs/czasopismo/2006/Molczan%20Szlachta_4-2006.pdf (accessed on 10 March 2022).
- Nowacka, A.; Włodarczyk-Makuła, M. Monitoring of polycyclic aromatic hydrocarbons in water during preparation processes. Polycycl. Aromat. Compd. 2013, 33, 430–450. [Google Scholar] [CrossRef]
- Wittmer, A.; Heisele, A.; Mc Ardell, C.S.; Böhler, M.; Longree, P.; Siegrist, H. Decreased UV absorbance as an indicator of micropollutant removal efficiency in wastewater treated with ozone. Water Sci. Technol. 2015, 71, 980–985. [Google Scholar] [CrossRef]
- Marais, S.S.; Ncube, E.J.; Msagati, T.A.M.; Mamba, B.B.; Nkambule, T.T.I. Assessment of trihalomethane (THM) precursors using specific ultraviolet absorbance (SUVA) and molecular size distribution (MSD). J. Water Process. Eng. 2019, 27, 143–151. [Google Scholar] [CrossRef]
- Musteret, C.P.; Morosanu, I.; Ciobanu, R.; Plavan, O.; Gherghel, A.; Al-Refai, M.; Roman, I.; Teodosiu, C. Assessment of coagulation–flocculation process efficiency for the natural organic matter removal in drinking water treatment. Water 2021, 13, 3073. [Google Scholar] [CrossRef]
- Beauchamp, N.; Laflamme, O.; Simard, S.; Dorea, C.; Pelletier, G.; Bouchard, C.; Rodriguez, M. Relationships between DBP concentrations and differential UV absorbance in full-scale condition. Water Res. 2018, 131, 110–121. [Google Scholar] [CrossRef]
- Papageorgiou, A.; Voutsa, D.; Papadakis, N. Occurrence and fate of ozonation by-products at a full-scale drinking water treatment plant. Sci. Total Environ. 2014, 481, 392–400. [Google Scholar] [CrossRef]
- Samadi, M.T.; Azarian, G.; Seifipour, F.; Huang, C.; Yang, X.; Poormohammadi, A. The formation of aldehydes and ketones ozonation by-products and their variation through general water treatment plant in Hamadan. Iran. Glob. Nest J. 2015, 17, 682–691. [Google Scholar]
- Zhong, X.; Cui, C.; Yu, S. Seasonal evaluation of disinfection by-products throughout two full-scale drinking water treatment plants. Chemosphere 2017, 179, 290–297. [Google Scholar] [CrossRef]
- Wu, Y.; Wu, C.D.; Zhang, Z.L.; Naluswata, F.; Yuan, B.J.; Liang, J.L. Behaviour of ozonation by-products during advanced drinking water treatment with pearl river water. Environ. Eng. Manag. J. 2018, 17, 1035–1041. [Google Scholar] [CrossRef]
- Dąbrowska, A.; Kasprzyk-Hordern, B.; Nawrocki, J. Aldehydes formation during water disinfection by ozonation and chlorination process. Glob. Nest J. 2005, 7, 61–71. [Google Scholar]
- Neal, C.; Neal, M.; Hughes, S.; Wickham, H.; Hill, L.; Harman, S. Bromine and bromide in rainfall, cloud, stream and groundwater in the Plynlimon area of mid-Wales. Hydrol. Earth Syst. Sci. 2007, 11, 301–312. [Google Scholar] [CrossRef]
- Sollars, C.J.; Peters, C.J.; Perry, R. Bromide in urban runoff—Water quality considerations. In Effects of Waste Disposal on Groundwater and Surface Water; Proceedings of the Exeter Symposium; International Association of Hydrological Sciences London: London, UK, 1982; pp. 101–112. [Google Scholar]
- Aitkenhead-Peterson, J.A.; Nahar, N.; Harclerode, C.L.; Stanley, N.C. Effect of urbanization on surface water chemistry in south-central Texas. Urban Ecosyst. 2011, 14, 195–210. [Google Scholar] [CrossRef]
- Heeb, M.B.; Criquet, J.; Zimmermann-Steffens, S.G.; von Gunten, U. Oxidative treatment of bromide-containing waters: Formation of bromine and its reactions with inorganic and organic compounds—A critical review. Water Res. 2014, 48, 15–42. [Google Scholar] [CrossRef]

| Sample 1S | Sample 2S | Sample 3S | |
|---|---|---|---|
| pH | 7.01 | 7.61 | 7.4 |
| EC [mS/cm] | 0.452 | 0.473 | 0.478 |
| Absorbance (α = 254) | 0.071 | 0.043 | 0.038 |
| Absorbance α = 272) | 0.038 | 0.032 | 0.026 |
| Absorbance (α = 436) | 0.008 | 0.007 | 0.001 |
| Formaldehyde [μg/L] | <6 | <6 | <6 |
| Brtotal [μg/L] | 18 | 16 | 89 |
| Br− [μg/L] | 22.5 | 22.7 | <20 |
| BrO3− [μg/L] | <2 | <2 | <2 |
| TOC [mg/L] | 1.79 | 1.58 | 1.54 |
| CHCl3 [μg/L] | 6.5 | ||
| CHBrCl2 [μg/L] | 2.5 | ||
| CHBr2Cl [μg/L] | <2 | ||
| CHBr3 [μg/L] | <2 | ||
| ΣTHM [μg/L] | 9.0 |
| Sample 1S | Sample 2S | Sample 3S | Ra (Radziszów) | Parametric Value * | Standard Analytical Methods | ||
|---|---|---|---|---|---|---|---|
| pH | 7.16 | 7.18 | 7.19 | 7.28 | 6.5–9.5 | PN-EN ISO 10523:2012 | |
| EC [mS/cm] | 0.532 | 0.544 | 0.547 | 0.544 | 2.5 | PN-EN 27888:1999 | |
| Absorbance α = 254 | 0.104 | 0.042 | 0.029 | 0.030 | PN-C-04572,1984 | ||
| Absorbance α = 272 | 0.093 | 0.034 | 0.026 | 0.026 | |||
| Absorbance α = 436 | 0.016 | 0.000 | - | - | |||
| Na+ [mg/L] | 23.40 | 26.43 | 27.07 | 200 | PN-ISO 9964–2 1994 | ||
| K+ [mg/L] | 4.96 | 4.996 | 4.784 | - | |||
| Ca2+ [mg/L] | 60.12 | 76.15 | 80.16 | PN-ISO 6058,1999 | |||
| Mg2+ [mg/L] | 19.46 | 12.04 | 13.13 | 7–125 | PN-ISO 6059,1999 | ||
| Fe2+ [mg/L] | 0.56 | <0.2 | 0.2 | PN-ISO 6332,2001 | |||
| Cl− [mg/L] | 49.64 | 48.87 | 42.55 | 250 | PN-ISO 9297,1994 | ||
| HCO3− [mg/L] | 27.46 | 30.51 | 27.46 | PN-EN ISO 9963–1, 2001 | |||
| NH4+ [mg/L] | 0.19 | 0.22 | 0.20 | 0.5 | PN-EN ISO 14911:2002 | ||
| NO3− [mg/L] | 4.6 | 4.8 | 4.9 | 50 | PN EN ISO 10304–1:2009 | ||
| NO2− [mg/L] | <0.01 | <0.01 | <0.01 | 0.5 | |||
| TOC [mg/L] | 2.94 | 2.18 | 2.31 | PN-EN 1484, 1999 | |||
| DOC [mg/L] | 2.37 | 1.88 | 2.01 | ||||
| Formaldehyde [μg/L] | 13 | 10 | 11 | PB-W-11 | |||
| Brtotal [μg/L] | 60 | 51 | 165 | 150 | PN-EN ISO 17294 | ||
| Br− [μg/L] | 29.6 | 28.1 | <20 | <20 | PN-EN ISO 10304–1:2009 | ||
| BrO3− [μg/L] | Lab.1 | <5 | <5 | <5 | <5 | 10 | DIN EN ISO 15061 |
| Lab.2 | 7.9 | 6.6 | |||||
| CHCl3 [μg/L] | 12.6 | 16.7 | 30 | PN-EN ISO 10301:2002 | |||
| CHBrCl2 [μg/L] | 2.8 | 6.0 | 15 | ||||
| CHBr2Cl [μg/L] | <2 | <2 | |||||
| CHBr3 [μg/L] | <2 | <2 | |||||
| ΣTHM [μg/L] | 15.4 | 22.7 | 100 | ||||
| Sample 1S | Sample 2S | Sample 3S | Ra (Radziszów) | ||
|---|---|---|---|---|---|
| pH | 7.28 | 7.25 | 7.27 | 7.30 | |
| EC [mS/cm] | 0.515 | 0.513 | 0.520 | 0.554 | |
| Absorbance (α = 254) | 0.172 | 0.080 | 0.065 | 0.042 | |
| Absorbance α = 272) | 0.149 | 0.065 | 0.048 | 0.030 | |
| Absorbance (α = 436) | 0.036 | 0.006 | 0.001 | 0.001 | |
| Na+ [mg/L] | 25.51 | 29.66 | 30.98 | ||
| K+ [mg/L] | 5.65 | 5.55 | 5.62 | ||
| Ca2+ [mg/L] | 58.52 | 54.78 | 62.79 | ||
| Mg2+ [mg/L] | 8.76 | 8.92 | 4.86 | ||
| Fe2+ [mg/L] | 0.75 | <0.2 | |||
| Cl− [mg/L] | 46.09 | 56.73 | 63.82 | ||
| HCO3− [mg/L] | 24.41 | 27.46 | 25.93 | ||
| Ca2+− [mg/L] | 58.52 | 54.78 | 62.79 | ||
| Mg2+− [mg/L] | 8.76 | 8.92 | 4.86 | ||
| NH4+ [mg/L] | 0.35 | 0.063 ± 0.011 | <0.015 | - | |
| NO3− [mg/L] | 3.6 | 4.2 ± 0.5 | 4.2 ± 0.5 | - | |
| NO2− [mg/L] | 0.21 | - | - | - | |
| TOC [mg/L] | 4.26 | 3.38 | 3.32 | - | |
| DOC [mg/L] | 3.71 | 3.34 | 3.07 | - | |
| Formaldehyde [μg/L] | 11 | <6 | 14 | - | |
| Brtotal [μg/L] | 31 | 31 | 46 | 58 | |
| Br− [μg/L] | 25.2 | 25.6 | <20 | <20 | |
| BrO3− [μg/L] | Lab.1 | <2 | <2 | ||
| Lab.2 | <5 | <5 | |||
| CHCl3 [μg/L] | Lab.1 | 18.4 | 17.5 | ||
| Lab.2 | 16.2 | 14.6 | |||
| CHBrCl2 [μg/L] | Lab.1 | 2.6 | 6.2 | ||
| Lab.2 | 2.36 | 5.13 | |||
| CHBr2Cl [μg/L] | Lab.1 | <2 | <2 | ||
| Lab.2 | <1 | 1.31 | |||
| CHBr3 [μg/L] | Lab.1 | <2 | <2 | ||
| Lab.2 | <1 | <1 | |||
| ΣTHM [μg/L] | Lab.1 | 21.1 | 23.7 | ||
| Lab.2 | 18.2 | 21.1 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Winid, B.; Muszański, R.; Wilkosz, J. Modification of Disinfection Process at a Local Water Treatment Plant—Skawina (Poland). Water 2022, 14, 2924. https://doi.org/10.3390/w14182924
Winid B, Muszański R, Wilkosz J. Modification of Disinfection Process at a Local Water Treatment Plant—Skawina (Poland). Water. 2022; 14(18):2924. https://doi.org/10.3390/w14182924
Chicago/Turabian StyleWinid, Bogumiła, Robert Muszański, and Jan Wilkosz. 2022. "Modification of Disinfection Process at a Local Water Treatment Plant—Skawina (Poland)" Water 14, no. 18: 2924. https://doi.org/10.3390/w14182924
APA StyleWinid, B., Muszański, R., & Wilkosz, J. (2022). Modification of Disinfection Process at a Local Water Treatment Plant—Skawina (Poland). Water, 14(18), 2924. https://doi.org/10.3390/w14182924






