Single and Combined Toxicity Effects of Zinc Oxide Nanoparticles: Uptake and Accumulation in Marine Microalgae, Toxicity Mechanisms, and Their Fate in the Marine Environment
Abstract
:1. Introduction
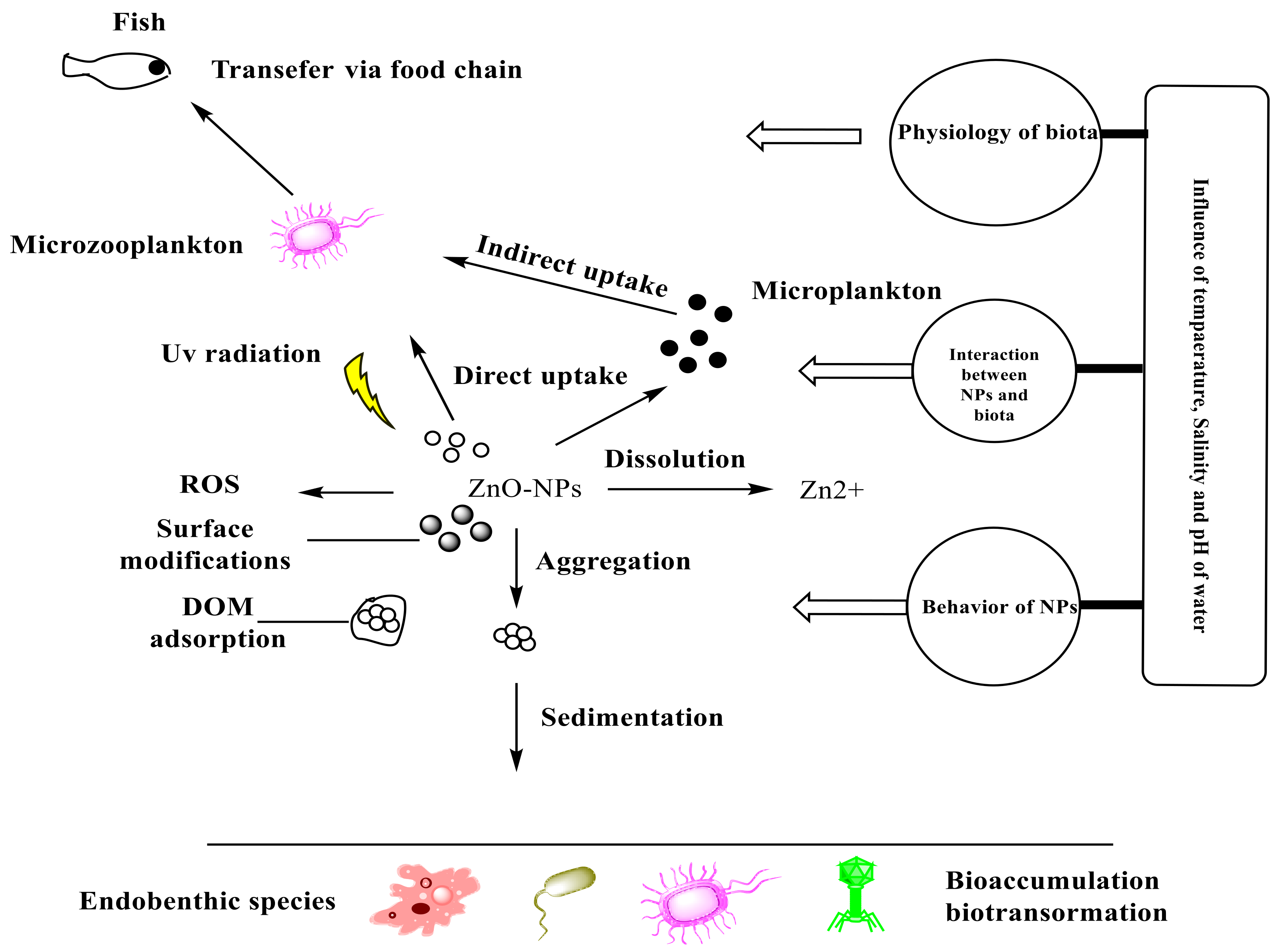
2. Fate and Behavior of ZnO-NPs in the Marine Environment
2.1. Existence and Persistence
2.2. Aggregation
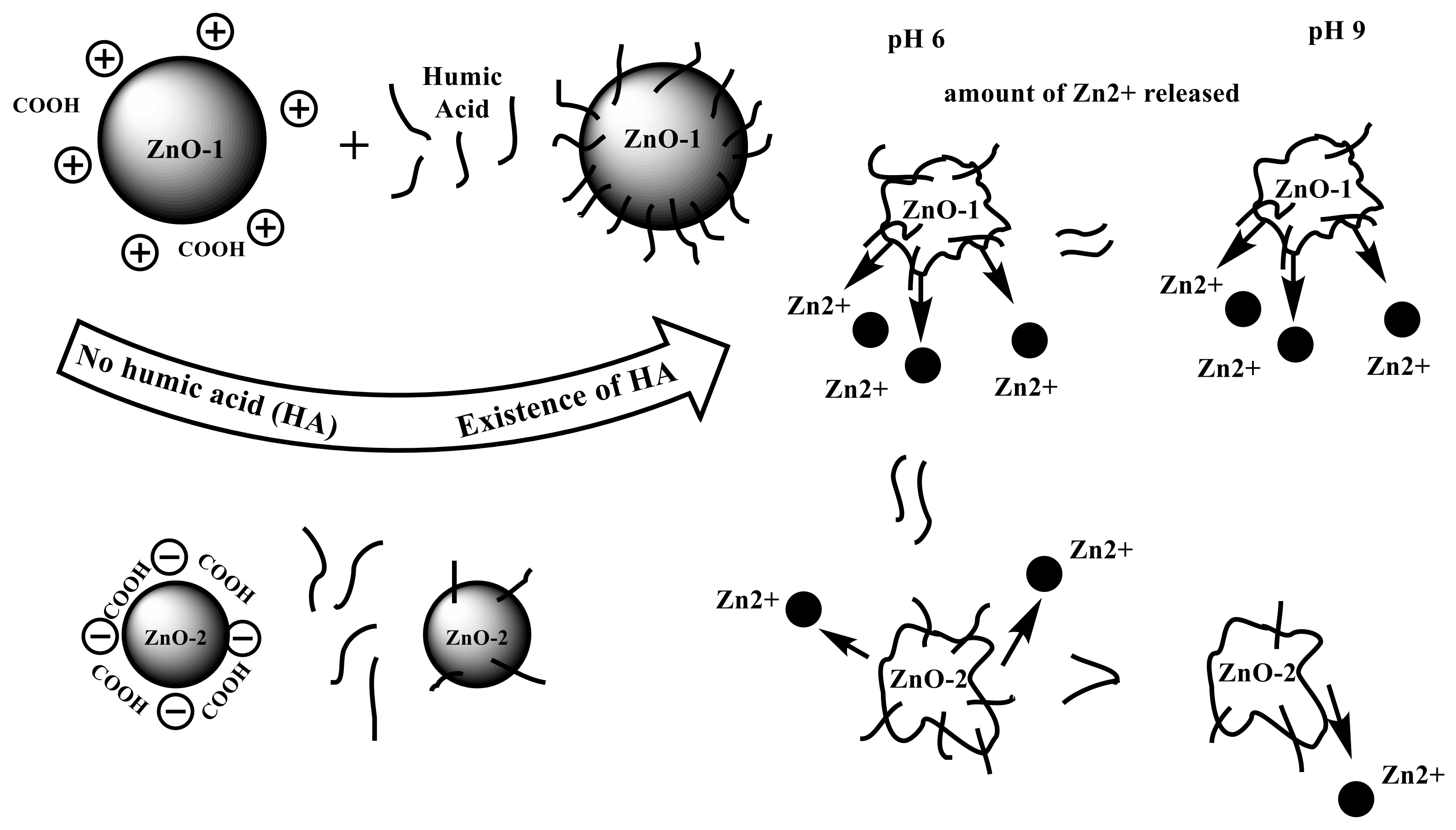
2.3. Adsorption
2.4. Transformation
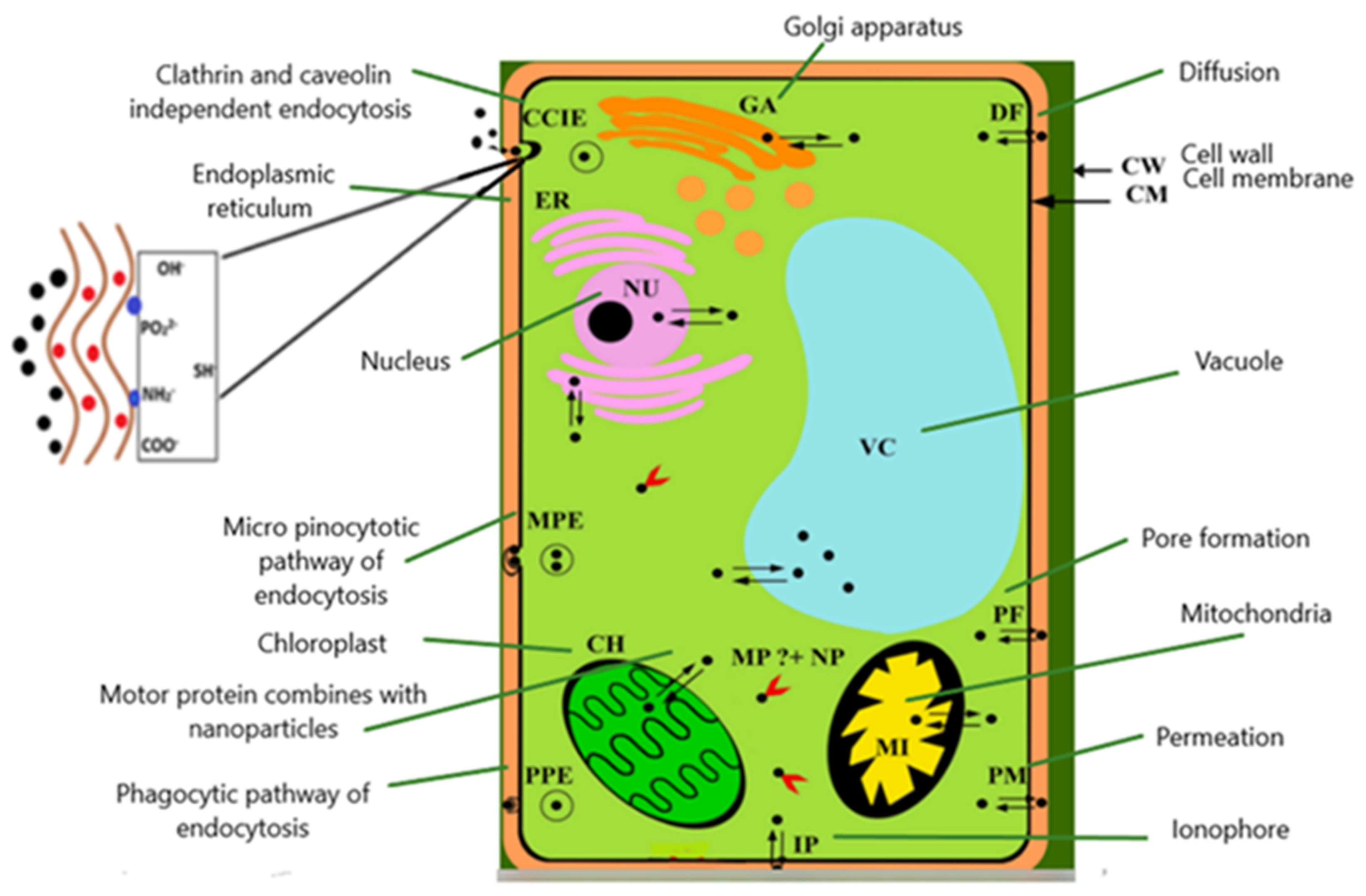
3. Uptake and Accumulation of NPs in Microalgae
4. Single and Combined Effects of ZnO-NPs on Marine Microalgae
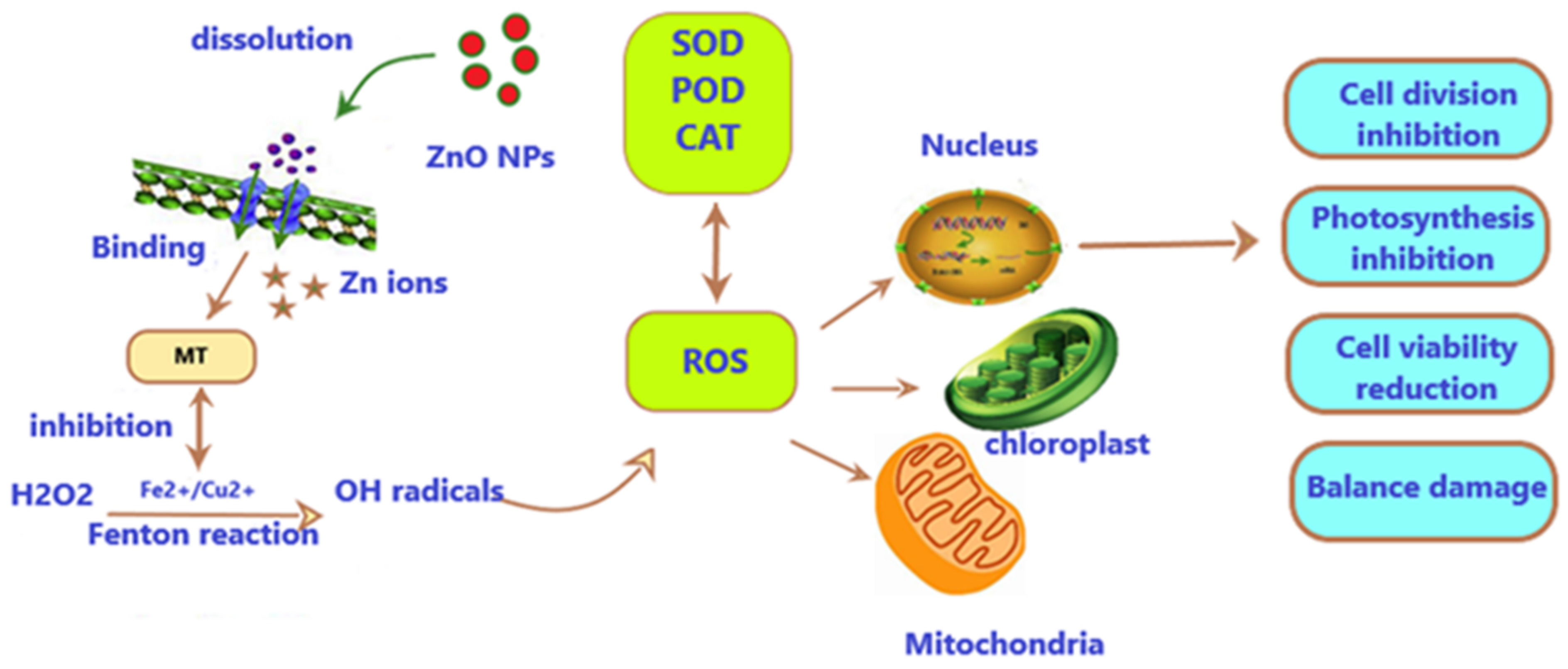
4.1. Toxicity Mechanisms of ZnO-NPs
4.2. Single Effect of ZnO-NPs
| Size of ZnO-NPs | Microalgae Species | Growth Conditions (h) | Endpoint (s) | Toxic Effect | Reference |
|---|---|---|---|---|---|
| “20 nm” | “Thalassiosira pseudonana” | “48” | “Growth rate”, | “IC50 = 0.39 µM” The dissolution of ZnO influenced its toxicity and the dissolution rate of ZnO-NPs was accelerated in seawater. | [69] |
| “20 nm” | “Skeletonema costatum” | “96” | “Growth inhibition” | “IC50 = 2.36 mg/L” | [76] |
| “Thalassiosira pseudonana” | IC50 = 4.56 mg/L | ||||
| “20–30 nm” | “Thalassiosira pseudonana” | “96” | “Growth rate” | ZnO-NPs At concentrations between 1 and 1000 µg L−1 reduced the growth rate of all four species | [68] |
| “Skeletonema marinoi” | |||||
| “Dunaliella tertiolecta” | |||||
| “Isochrysis galbana” | |||||
| “6.3–15.7 nm (sphere); 242–862 nm (rod)” | “Thalassiosira pseudonana” | “100” | “Growth inhibition test” | NPs at all concentrations stopped the growth of T. pseudonana and C. gracilis, whereas P. tricornutum was the least sensitive | [71] |
| “Thalassiosira pseudonana” | |||||
| “Phaeodactylum tricornatum” | Rod shaped ZnO with one-dimensional structures were more toxic than spheres NPs | ||||
| “100 nm 200 nm” | “Dunaliella tertiolecta” | “96” | “Growth inhibition” | EC50 = 1.94 mg/L (100 nm) EC50 = 3.57 mg/L (200 nm). The higher toxicity of nano ZnO is related to its physiochemical properties. ZnO-NPs significantly affected the growth rate of D. Tertiolecta. | [77] |
| “25nm” | “Phaeodactylum tricornutum” “Alexandrium minutum” “Tetraselmis suecica” | “Long term” | “Growth inhibition” | “The growth of microalgae was inhibited at concentration more than 10 mg/L. Decrease in chlorophyll fluorescence and high ROS” | [97] |
| “40–48 nm” | “Chlorella vulgaris” | “72” | “Cell viability” “LDH” “Oxidative stress” “Lipid peroxidation” “FTIR analysis” | “The toxicity was concentration and time dependent” | [87] |
| “<100 nm” | “96” | “Growth rate” | Marine species are more sensitive to ZnO-NPs compared to freshwater algae used in the same study. | [89] | |
| “Dunaliella Tertiolecta” | “96-h IC50: 1.5 mg/L (f/2) 96-h IC50: 1.33 mg/L (salt ⅓N BG-11)” | ||||
| “Tetraselmis suecica” | “96-h IC50: 2.1 mg/L (f/2) 96-h IC50: 2.57 mg/L (salt ⅓N BG-11)” | ||||
| “100 nm” | “Dunaliella Tertiolecta” | “72” | “Cell viability” “ROS” “COMET assay” | “EC50: 2.0 mg/L NOEC: 0.1 mg/L” “Significant increase of ROS at ZnO > 10 mg/L at 72-h” “ZnO-NPs was found to be genotoxic at 5 mg/L” | [84] |
| “50 nm” | “Skeletonema Costatum” | “96” | “Growth inhibition” “Lipid peroxidation” | “96-h EC50: 3.6 (50 nm)” “96-h EC50: 5.5 mg/L (5.5 (>100nm)” “High MDA level with the nano-ZnO” | [88] |
| “50 nm” | “Picochlorum sp.” | “32 days” | “Growth inhibition” “Chlorophyll a” | “The toxicity of NPs reduced due to aggregation and sedimentation of NPs after long incubation time” | [98] |
| “100 nm” “200 nm” “ZnSO4” | “Tetraselmis suecica” | “72” | “Growth inhibition” | “72-h EC50 (ZnO 100 nm): 3.91 mg/L 72-h EC50 (ZnO 200 nm): 7.12 mg/L 72-h EC50 (ZnSO4: 5.61 mg/L” | [81] |
| “Phaeodactylum triconutum” | “72-h EC50 (ZnO 100 nm): 1.09 mg/L 72-h EC50 (ZnO 200 nm): 3.47 mg/L 72-h EC50 (ZnSO4: 3.22 mg/L” P. triconatum showed a higher sensitivity than T. suecica | ||||
| “<100 nm” | “Dunaliella Tertiolecta” | “72” | “Growth inhibition” | Marine algae were more sensitive than bacteria and crustaceans used in the same study. “72-h EC50: 2.2 mg/L” | [82] |
| “<100 nm” | “Phaeodactylum tricornutum” | “72” | “Growth inhibition” | “72-h EC50: 2.5–5 mg/L” | [99] |
| “Isochrysis galbana” | “72-h EC50: 5 mg/L” | ||||
| “Tetraselmis suecica” | “72-h EC50: 2.5–5 mg/L” | ||||
| “30, 90 and 200 nm” | “Chlorella sp.” | “96” | “Chlorophyll a” “Oxidative stress (SOD and CAT) 1” “Lipid peroxidation (MDA) 2” | “96-h lowest chlorophyll a concentration was measured in zinc ions” “Highest oxidative stress was detected in cells exposed to 30 nm ZnO-NPs and Zn ions” “Highest MDA measured with 30 nm ZnO-NPs followed by Zn ions.” | [90] |
| “ZnO nano” “ZnO-Bulk” | “Platymonas subcordiformis” | “24” | “Motility of microalgae as a major endpoint in addition to other growth parameters” | “Significant reduction in motility parameters when microalgae exposed to ZnO-NPs”. “No negative effect recorded with bulk ZnO” | [93] |
4.3. Combined Toxicity Effect of ZnO with Other Materials
5. Critical Evaluation of the Existing Literature on the Single and Combined Effects of ZnO-NPs on Marine Microalgae
6. Research, Knowledge Gap and Recommendations:
- Toxic mechanisms of ZnO-NPs at the molecular level in marine organisms at different trophic levels should be examined using advanced techniques including transcriptomic, proteomic, and metabolomic methods to discriminate the diverse modes of toxicity of ZnO-NPs when used separately or combined with other materials.
- Research should be performed on the toxicity of ZnO-NPs under environmentally relevant UV radiation because of its photoactive properties. The assessments should be applied using different marine organisms at different trophic levels. ROS released due to oxidative stress imposed by the exposure to ZnO-NPs should also be investigated.
- Acute and chronic toxicity studies are needed to better understand the effects of ZnO-NPs on various marine organisms at various stages of development.
- Trophic transfer of ZnO-NPs should be examined for the accumulation, cellular localization, or biotransformation of NPs in a well-defined marine food chain.
- There have been few investigations of the combined effect of ZnO-NPs and other contaminants. It should be addressed properly in a variety of environmental conditions, including temperature, pH, UV radiation, and the presence of dissolved organic material. The data could be used to improve water quality criteria and thereby conserve marine life by limiting the usage and release of ZnO-NPs.
- Evaluating surface modification of ZnO-NPs is required. Methods should be developed to maintain the initial chemical features of ZnO-NPs while decreasing their biological toxicity.
- There is to date a substantial lack of characterization of ZnO-NPs and theirs exposure system in toxicity studies. The effects of ZnO-NPs depend on exposure conditions, for instance, water chemistry and irradiation. It is essential to have a detailed characterization of the exposure conditions to effectively explain the toxicity data.
- Certain international regulations and policies should be applied for the production and use of NPs. Furthermore, wastes containing NPs should be well treated before disposal. Public awareness programs should alert people to take extra care in the use and disposal of NPs.
- Cost-effective methods for the elimination and retrieval of NPs from waste before disposal should be established to retrieve and reuse the original forms of the NPs.
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Nguyen, M.K.; Moon, J.Y.; Lee, Y.C. Microalgal ecotoxicity of nanoparticles: An updated review. Ecotoxic. Environ. Saf. 2020, 201, 110781. [Google Scholar] [CrossRef] [PubMed]
- Shevlin, D.; O’Brien, N.; Cummins, E. Silver engineered nanoparticles in freshwater systems-likely fate and behavior through natural attenuation processes. Sci. Total. Environ. 2018, 621, 1033–1046. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, Applications and Toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Adochite, C.; Andronic, L. Aquatic Toxicity of Photocatalyst Nanoparticles to Green Microalgae Chlorella vulgaris. Water 2021, 13, 77. [Google Scholar] [CrossRef]
- Ma, S.; Zhou, K.; Yang, K.; Lin, D. Hetero agglomeration of oxide nanoparticles with algal cells: Effects of particle type, ionic strength and pH. Environ. Sci. Technol. 2015, 49, 932–939. [Google Scholar] [CrossRef]
- Li, S.; Ma, H.; Wallis, L.K.; Etterson, M.A.; Riley, B.; Hoff, D.J.; Diamond, S.A. Impact of natural organic matter on particle behavior and phototoxicity of titanium dioxide nanoparticles. Sci. Total. Environ. 2016, 542, 324–333. [Google Scholar] [CrossRef]
- Nolte, T.M.; Hartmann, N.B.; Kleijn, J.M.; Garnæs, J.; van de Meent, D.; Jan Hendriks, A.; Baun, A. The toxicity of plastic nanoparticles on green algae as influenced by surface modification, medium hardness and cellular adsorption. Aquat. Toxicol. 2017, 183, 11–20. [Google Scholar] [CrossRef]
- Sangchay, W. The Self-cleaning and Photocatalytic Properties of TiO2 Doped with SnO2, Thin Films Preparation by Sol-gel Method. Energy Procedia 2016, 89, 170–176. [Google Scholar] [CrossRef]
- Ali, I.; Alharbi, O.M.L.; Tkachev, A.; Galunin, E.; Burakov, A.; Grachev, V.A. Water treatment by new-generation graphene materials: Hope for bright future. Environ. Sci. Pollut. Res. 2018, 25, 7315–7329. [Google Scholar] [CrossRef]
- Corma, A.; Atienzar, P.; García, H.; Chane-Ching, J.-Y. Hierarchically mesostructured doped CeO2 with potential for solar-cell use. Nat. Mater. 2004, 3, 394–397. [Google Scholar] [CrossRef]
- Piccinno, F.; Gottschalk, F.; Seeger, S.; Nowack, B. Industrial production quantities and uses of ten engineered nanomaterials in Europe and the world. J. Nanopart. Res. 2012, 14, 1109. [Google Scholar] [CrossRef]
- Handy, R.D.; Owen, R.; Valsamijones, E. The ecotoxicology of nanoparticles and nanomaterials: Current status, knowledge gaps, challenges, and future needs. Ecotoxicology 2008, 17, 315–325. [Google Scholar] [CrossRef]
- Rogers, F.; Arnott, P.; Zielinska, B.; Sagebiel, J.; Kelly, K.E.; Wagner, D.; Lighty, J.S.; Sarofim, A.F. Real-Time Measurements of Jet Aircraft Engine Exhaust. J. Air Waste Manag. Assoc. 2005, 55, 583–593. [Google Scholar] [CrossRef]
- Seames, W.S.; Fernandez, A.; Wendt, J.O.L. A study of fine particulate emissions from combustion of treated pulverized municipal sewage sludge. Environ. Sci. Technol. 2002, 36, 2772–2776. [Google Scholar] [CrossRef]
- Mahana, A.; Guliy, O.I.; Mehta, S.K. Accumulation and cellular toxicity of engineered metallic nanoparticle in freshwater microalgae: Current status and future challenges. Ecotoxicol. Environ. Saf. 2021, 208, 111662. [Google Scholar] [CrossRef]
- Junam, Y.; Lead, J.R. Manufactured nanoparticles: An overview of their chemistry, interactions and potential environmental implications. Sci. Total Environ. 2008, 400, 396–414. [Google Scholar] [CrossRef]
- Trojanowski, R.; Fthenakis, V. Nanoparticle emissions from residential wood combustion: A critical literature review, characterization, and recommendations. Renew. Sustain. Energy Rev. 2019, 103, 515–528. [Google Scholar] [CrossRef]
- Chen, C.; Huang, W. Aggregation kinetics of diesel soot nanoparticles in wet environments. Environ. Sci. Technol. 2017, 51, 2077–2086. [Google Scholar] [CrossRef]
- Somasundaran, P.; Fang, X.; Ponnurangam, S.; Li, B. Nanoparticles: Characteristics, mechanisms, and modulation of biotoxicity. KONA Power Part. J. 2010, 28, 38–49. [Google Scholar] [CrossRef]
- Lu, P.J.; Huang, S.C.; Chen, Y.P.; Chiueh, L.C.; Shih, D.Y.C. Analysis of titanium dioxide and zinc oxide nanoparticles in cosmetics. J. Food Drug Anal. 2015, 23, 587–594. [Google Scholar] [CrossRef] [Green Version]
- Bairi, V.G.; Lim, J.-H.; Fong, A.; Linder, S.W. Size characterization of metal oxide nanoparticles in commercial sunscreen products. J. Nanopart. Res. 2017, 19, 256. [Google Scholar] [CrossRef]
- Fatehah, M.O.; Aziz, H.A.; Stoll, S. Nanoparticle Properties, behavior, Fate in Aquatic Systems and Characterization Methods. J. Colloid Sci. Biotechnol. 2014, 3, 111–1140. [Google Scholar] [CrossRef]
- McIntyre, R.A. Common nanomaterials and their use in real world applications. Sci. Prog. 2012, 95, 1–22. [Google Scholar] [CrossRef]
- Smijs, T.G.; Pavel, S. Titanium dioxide and zinc oxide nanoparticles in sunscreens: Focus on their safety and effectiveness. Nanotechnol. Sci. Appl. 2011, 4, 95–112. [Google Scholar] [CrossRef]
- Mirzaei, H.; Darroudi, M. Zinc oxide nanoparticles: Biological synthesis and biomedical applications. Ceram. Int. 2017, 43, 907–914. [Google Scholar] [CrossRef]
- Ma, H.; Williams, P.L.; Diamond, S.A. Ecotoxicity of manufactured ZnO nanoparticles—A review. Environ. Pollut. 2013, 172, 76–85. [Google Scholar] [CrossRef]
- Gottschalk, F.; Sonderer, T.; Scholz, R.W.; Nowack, B. Modeled Environmental Concentrations of Engineered Nanomaterials (TiO2, ZnO, Ag, CNT, Fullerenes) for Different Regions. Environ. Sci. Technol. 2009, 43, 9216–9222. [Google Scholar] [CrossRef]
- Mehta, S.K.; Gaur, P. Use of Algae for Removing Heavy Metal Ions From Wastewater: Progress and Prospect. Crit. Rev. Biotechnol. 2005, 25, 113–152. [Google Scholar] [CrossRef]
- Abdel-Raouf, N.; Al-Homaidan, A.A.; Ibraheem, I.B.M. Microalgae and wastewater treatment. Saudi J. Biol. Sci. 2012, 19, 257–275. [Google Scholar] [CrossRef]
- Nowack, B. The occurrence, behavior, and effects of engineered nanomaterials in the environment. In Advances in Nanotechnology and the Environment; Kim, J., Ed.; Pan Stanford: Singapore, 2012; pp. 197–207. [Google Scholar]
- Boxall, A.B.A.; Chaudhry, Q.; Sinclair, C.; Jones, A.D.; Aitken, R.; Jefferson, B.; Watts, C. Current and Future Predicted Environmental Exposure to Engineered Nanoparticles; Central Science Laboratory, Department of the Environment and Rural Affairs: London, UK, 2007; 89p.
- Gottschalk, F.; Sun, T.Y.; Nowack, B. Environmental concentrations of engineered nanomaterials: Review of modeling and analytical studies. Environ. Pollut. 2013, 181, 287–300. [Google Scholar] [CrossRef]
- Gottschalk, F.; Sonderer, T.; Scholz, R.W.; Nowack, B. Possibility and limitations of modeling environmental exposure to engineered nanomaterials by probabilistic material flow analysis. Environ. Toxicol. Chem. 2010, 29, 1036–1048. [Google Scholar] [CrossRef]
- Tiede, K.; Hassellöv, M.; Breitbarth, E.; Chaudhry, Q.; Boxall, A.B.A. Considerations for environmental fate and ecotoxicity testing to support environmental risk assessments for engineered nanoparticles. J. Chromatogr. A 2009, 1216, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Fairbairn, E.A.; Keller, A.A.; Madler, L.; Zhou, D.; Pokhrel, S. Metal oxide nanoparticles in seawater: Linking physiochemical characteristics with biological response in sea urchin development. J. Hazard. Mater. 2011, 192, 1565–1571. [Google Scholar] [CrossRef] [PubMed]
- Bian, S.W.; Mudunkotuwa, I.A.; Rupasinghe, T.; Grassian, V.H. Aggregation and dissolution of 4nm ZnO nanoparticles in aqueous environments: Influence of pH, ionic strength, size, and adsorption of humic acid. Langmuir 2011, 27, 6059–6068. [Google Scholar] [CrossRef]
- Tang, E.; Cheng, G.; Ma, X.; Pang, X.; Zhao, Q. Surface modification of zinc oxide nanoparticle by PMAA and its dispersion in aqueous system. Appl. Surf. Sci. 2006, 252, 5227–5232. [Google Scholar] [CrossRef]
- Lowry, G.V.; Wiesner, M.R. Environmental considerations: Occurrences, fate, and characterization of nanoparticles in the environment. In Nanotoxicology: Characterization, Dosing and Health Effects; Monteiro-Riviere, N.A., Lang Tran, C., Eds.; Informa Healthcare: New York, NY, USA, 2007; pp. 369–389. [Google Scholar]
- Morales, J.; Manso, J.A.; Cid, A.; Mejuto, J.C. Degradation of carbofuran and carbofuran derivatives in presence of humic substances under basic conditions. Chemosphere 2012, 89, 1267–1271. [Google Scholar] [CrossRef]
- Thio, B.J.R.; Zhou, D.; Keller, A.A. Influence of natural organic matter on the aggregation and deposition of titanium dioxide nanoparticles. J. Hazard. Mater. 2011, 189, 556–563. [Google Scholar] [CrossRef]
- Litvin, V.A.; Galagan, R.L.; Minaev, B.F. Kinetic and mechanism formation of silver nanoparticles coated by synthetic humic substances. Colloids Surf. A Physicochem. Eng. Asp. 2012, 414, 234–243. [Google Scholar] [CrossRef]
- Sutton, R.; Sposito, G. Molecular structure in soil humic substances: The new view. Environ. Sci. Technol. 2005, 39, 9009–9015. [Google Scholar] [CrossRef]
- Ojwang’, L.M.; Cook, R.L. Environmental conditions that influence the ability of humic acids to induce permeability in model bio membranes. Environ. Sci. Technol. 2013, 47, 8280–8287. [Google Scholar]
- Tenzer, S.; Doctor, D.; Kuharev, J.; Musyanovych, A.; Fetz, V.; Hecht, R.; Schlenk, F.; Fischer, D.; Kiouptsi, K.; Reindhardt, C.; et al. Rapid formation of plasma protein corona critically affects nanoparticle pathophysiology. Nat. Nanotechnol. 2013, 8, 772–781. [Google Scholar] [CrossRef]
- Nowak, B.; Bucheli, T.D. Occurrence, behavior and effects of nanoparticles in the environment. Environ. Pollut. 2007, 150, 5–22. [Google Scholar] [CrossRef]
- Efthimiou, I.; Georgiou, Y.; Vlastos, D.; Dailianis, S.; Deligiannakis, Y. Assessing the cyto-genotoxic potential of model zinc oxide nanoparticles in the presence of humic-acid-like-poly condensate (HALP) and the leaonardite HA (LHA). Sci. Total Environ. 2020, 721, 137625. [Google Scholar] [CrossRef]
- Yunga, M.M.N.; Mouneyracb, C.; Leung, K.M.Y. Ecotoxicity of Zinc Oxide Nanoparticles in the Marine Environment. In Encyclopedia of Nanotechnology; Bhushan, B., Ed.; Springer: Dordrecht, The Netherlands, 2014; pp. 1–17. [Google Scholar]
- Han, Y.; Kim, D.; Hwang, G.; Lee, B.; Eom, I.; Kim, P.J.; Tong, M.; Kim, H. Aggregation and dissolution of ZnO nanoparticles synthesized by different methods: Influence of ionic strength and humic acid. Colloids Surf. A 2014, 451, 7–15. [Google Scholar] [CrossRef]
- Wang, X.; Lu, J.; Xu, M.; Xing, B. Sorption of pyrene by regular and nanoscaled metal oxide particles: Influence of adsorbed organic matter. Environ. Sci. Technol. 2008, 42, 7267–7272. [Google Scholar] [CrossRef]
- Lipovsky, A.; Tzitrinovich, Z.; Friedmann, H.; Applerot, G.; Gedanken, A.; Lubart, R. EPR study of visible light-induced ROS generation by nanoparticles of ZnO. J. Phys. Chem C 2009, 113, 15997–16001. [Google Scholar] [CrossRef]
- Estevez, J.M.; Kasuli, L.; Fernandez, P.V.; Dupree, P.; Ciancia, M. Chemical in situ characterization of macromolecular components of the complex cell walls from the coenocytic green alga Codium fragile. Glycobiology 2009, 18, 250–259. [Google Scholar]
- Shankar, P.D.; Shobana, S.; Karuppusamy, I.; Pugazhendhi, A.; Ramkumar, V.S.; Arvindnarayan, S.; Kumar, G. A review on the biosynthesis of metallic nanoparticles (gold and silver) using bio-components of microalgae: Formation mechanism and applications. Enzyme Microb. Technol. 2016, 95, 28–44. [Google Scholar] [CrossRef]
- Ting, Y.P.; Prince, I.G.; Lawson, F. Uptake of cadmium and zinc by the alga Chlorella vulgaris: II. Multi-ion situation. Biotechnol. Bioeng. 1991, 37, 445–455. [Google Scholar] [CrossRef]
- Mehta, S.K.; Gaur, J.P. Heavy-metal-induced proline accumulation and its role in ameliorating metal toxicity in Chlorella vulgaris. New Phytol. 1999, 143, 253–259. [Google Scholar] [CrossRef]
- Crist, R.H.; Martin, J.R.; Guptill, P.W.; Eslinger, J.M.; Crist, D.R. Interaction of Metal Ions with Acid Sites of Bio sorbents Peat Moss and Vaucheria and Model Substances Alginic and Humic Acids. Environ. Sci. Technol. 1990, 24, 337–342. [Google Scholar] [CrossRef]
- Crist, R.H.; Martin, J.R.; Carr, D.; Watson, J.R.; Clarke, H.J.; Crist, D.R. Interaction of metals and protons with algae. Ion-exchange vs adsorption models and a reassessment of Scatchard plots; ion-exchange rates and equilibria compared with calcium alginate. Environ. Sci. Technol. 1994, 28, 1859–1866. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, N.P.; Hurst, G.B.; Wang, W.; Foster, C.M.; Nallathamby, P.D.; Retterer, S.T. Dynamic development of the protein corona on silica nanoparticles: Composition and role in toxicity. Nanoscale 2013, 5, 6372–6380. [Google Scholar] [CrossRef] [PubMed]
- Izak-Nau, E.; Voetz, M.; Eiden, S.; Duschl, A.; Puntes, V.F. Altered characteristics of silica nanoparticles in bovine and human serum: The importance of nanomaterial characterization prior to its toxicological evaluation. Part. Fibre Toxicol. 2013, 10, 56. [Google Scholar] [CrossRef]
- Kim, S.T.; Saha, K.; Kim, C.; Rotello, V.M. The role of surface functionality in determining nanoparticle cytotoxicity. Acc. Chem. Res. 2013, 46, 681–691. [Google Scholar] [CrossRef]
- Ehrenberg, M.S.; Friedman, A.E.; Finkelstein, J.N.; Oberdörster, G.; Mcgrath, J.L. The influence of protein adsorption on nanoparticle association with cultured endothelial cells. Biomaterials 2009, 30, 603–610. [Google Scholar] [CrossRef]
- Verma, A.; Stellacci, F. Effect of surface properties on nanoparticle-cell interactions. Small 2010, 6, 12–21. [Google Scholar] [CrossRef]
- Djearamane, S.; Wong, L.S.; Mooi, L.Y.; Lee, P.F. Cytotoxic effects of zinc oxide nanoparticles on cyanobacterium Spirulina (Arthrospira) platensis. PeerJ 2018, 6, e4682. [Google Scholar] [CrossRef]
- Gunawan, C.; Sirimanoonphan, A.; Teoh, W.Y.; Marquis, C.P.; Amal, R. Submicron and nano formulations of titanium dioxide and zinc oxide stimulate unique cellular toxicological responses in the green microalga Chlamydomonas reinhardtii. J. Hazard. Mater. 2013, 260, 984–992. [Google Scholar] [CrossRef]
- Hu, C.; Liu, Y.; Li, X.; Li, M. Biochemical responses of duckweed (Spirodela polyrhiza) to zinc oxide nanoparticles. Arch. Environ. Contam. Toxicol. 2013, 64, 643–651. [Google Scholar] [CrossRef]
- Shen, C.; James, S.A.; De Jonge, M.D.; Turney, T.W.; Wright, P.F.A.; Feltis, B.N. Relating cytotoxicity, zinc ions, and reactive oxygen in ZnO nanoparticle-exposed human immune cells. Toxicol. Sci. 2013, 136, 120–130. [Google Scholar] [CrossRef]
- Hou, J.; Wu, Y.; Li, X.; Wei, B.; Li, S.; Wang, X. Toxic effects of different types of zinc oxide nanoparticles on algae, plants, invertebrates, vertebrates and microorganisms. Chemosphere 2018, 193, 852–860. [Google Scholar] [CrossRef]
- Colvin, R.A.; Fontaine, C.P.; Laskowski, M.; Thomas, D. Zn2+ transporters and Zn2+ homeostasis in neurons. Eur. J. Pharmacol. 2003, 479, 171–185. [Google Scholar] [CrossRef]
- Miller, R.J.; Lenihan, H.S.; Muller, E.B.; Tseng, N.; Hanna, S.K.; Keller, A.A. Impact of metal oxide nanoparticles on marine phytoplankton. Environ. Sci. Technol. 2010, 44, 7329–7334. [Google Scholar] [CrossRef]
- Miao, A.J.; Zhang, X.Y.; Luo, Z.; Chen, C.S.; Chin, W.C.; Santschi, P.H.; Quigg, A. Zinc oxide engineered nanoparticles: Dissolution and toxicity to marine phytoplankton. Environ. Toxicol. Chem. 2010, 29, 2814–2822. [Google Scholar] [CrossRef]
- Wong, S.W.Y.; Leung, P.T.Y.; Djurišić, A.B.; Leung, K.M.Y. Toxicities of nano zinc oxide to five marine organisms: Influences of aggregate size and ion solubility. Anal. Bioanal. Chem. 2010, 396, 609–618. [Google Scholar] [CrossRef]
- Peng, X.; Palma, S.; Fisher, N.S.; Wong, S.S. Effect of morphology of ZnO nanostructures on their toxicity to marine algae. Aquat. Toxicol. 2011, 102, 186–196. [Google Scholar] [CrossRef]
- Eisler, R. Zinc Hazards to Fish, Wildlife, and Invertebrates: A Synoptic Review; No. 26; U.S. Department of the Interior, Fish and Wildlife Service: Washington, DC, USA, 1993; p. 106.
- Brand, L.E.; Sunda, W.G.; Guillard, R.R.L. Limitation of Marine Phytoplankton Reproductive Rates by Zinc, Manganese, and Iron. Limnol. Oceanogr. 1983, 28, 1182–1198. [Google Scholar] [CrossRef]
- Franklin, N.M.; Rogers, N.J.; Apte, S.C.; Batley, G.E.; Gadd, G.E.; Casey, P.S. Comparative toxicity of nanoparticulate ZnO, bulk ZnO, and ZnCl2 to a freshwater microalga (Pseudokirchneriella subcapitata): The importance of particle solubility. Environ. Sci. Technol. 2007, 41, 8484–8490. [Google Scholar] [CrossRef]
- Aruoja, V.; Kahru, A.; Dubourguier, H.C. Toxicity of ZnO, TiO2 and CuO nanoparticles to microalgae Pseudokirchneriella subcapitata. Toxicol. Lett. 2008, 180, S220. [Google Scholar] [CrossRef]
- Xia, T.; Kovochich, M.; Liong, M.; Madler, L.; Gilbert, B.; Shi, H.; Yeh, J.; Zink, J.I.; Nel, A.E. Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS Nano 2008, 2, 2124–2134. [Google Scholar] [CrossRef]
- Manzo, S.; Miglietta, M.L.; Rametta, G.; Buono, S.; Di Francia, G. Toxic effects of ZnO nanoparticles towards marine algae Dunaliella tertiolecta. Sci. Total Environ. 2013, 445, 371–376. [Google Scholar] [CrossRef]
- Maness, P.C.; Huang, Z.; Smolinski, S.; Jacoby, W.; Blake, D.; Wolfrum, E. Photosterilization and photominerization of microbial cells with titanium dioxide. Photochem. Photobiol. 1999, 69, 64s–65s. [Google Scholar]
- Navarro, E.; Piccapietra, F.; Wagner, B.; Marconi, F.; Kaegi, R.; Odzak, N.; Sigg, L.; Behra, R. Toxicity of silver nanoparticles to Chlamydomonas reinhardtii. Environ. Sci. Technol. 2008, 42, 8959–8964. [Google Scholar] [CrossRef]
- Magdolenova, Z.; Collins, A.; Kumar, A.; Dhawan, A.; Stone, V.; Dusinska, M. Mechanisms of genotoxicity. A review of in vitro and in vivo studies with engineered nanoparticles. Nanotoxicology 2014, 8, 233–278. [Google Scholar] [CrossRef]
- Li, J.; Schiavo, S.; Rametta, G.; Miglietta, M.L.; Ferrara, V.L.; Wu, C.; Manzo, S. Comparative toxicity of nano ZnO and bulk ZnO towards marine algae Tetraselmis suecica and Phaeodactylum tricornutum. Environ. Sci. Pollut. Res. 2017, 24, 6543–6553. [Google Scholar] [CrossRef]
- Schiavo, S.; Oliviero, M.; Li, J.; Manzo, S. Testing ZnO nanoparticle ecotoxicity: Linking time variable exposure to effect on different marine model organisms. Environ. Sci. Pollut. Res. 2018, 25, 4871–4880. [Google Scholar] [CrossRef]
- Gong, N.; Shao, K.; Feng, W.; Lin, Z.; Liang, C.; Sun, Y. Biotoxicity of nickel oxide nanoparticles and bio-remediation by microalgae Chlorella vulgaris. Chemosphere 2011, 83, 510–516. [Google Scholar] [CrossRef]
- Schiavo, S.; Oliviero, M.; Miglietta, M.; Rametta, G.; Manzo, S. Genotoxic and cytotoxic effects of ZnO nanoparticles for Dunaliella tertiolecta and comparison with SiO2 and TiO2 effects at population growth inhibition levels. Sci. Total Environ. 2016, 550, 619–627. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, X.; Lao, Y.; Lv, X.; Tao, Y.; Huang, B.; Wange, J.; Zhou, J.; Cai, Z. TiO2 nanoparticles in the marine environment: Physical effects responsible for the toxicity on algae Phaeodactylum tricornutum. Sci. Total Environ. 2016, 565, 818–826. [Google Scholar] [CrossRef]
- Ji, J.; Long, Z.; Lin, D. Toxicity of oxide nanoparticles to the green algae Chlorella sp. Chem. Eng. J. 2011, 170, 525–530. [Google Scholar] [CrossRef]
- Suman, T.Y.; Rajasree, S.R.R.; Kirubagaran, R. Evaluation of zinc oxide nanoparticles toxicity on marine algae Chlorella vulgaris through flow cytometric, cytotoxicity and oxidative stress analysis. Ecotoxicol. Environ. Saf. 2015, 113, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, J.; Tan, L.; Chen, X. Toxic effects of nano-ZnO on marine microalgae Skeletonema costatum: Attention to the accumulation of intracellular Zn. Aquat. Toxicol. 2016, 178, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Aravantinou, A.F.; Tsarpali, V.; Dailianis, S.; Manariotis, I.D. Effect of cultivation media on the toxicity of ZnO nanoparticles to freshwater and marine microalgae. Ecotoxicol. Environ. Saf. 2015, 114, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Wei, M.; Wang, Y. Toxic effects of different particle size ZnO NPs on marine microalgae Chlorella sp. IOP Conf. Ser. Earth Environ. Sci. 2021, 770, 012022. [Google Scholar] [CrossRef]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of Nanoparticle Toxicity on Their Physical and Chemical Properties. Nanoscale Res. Lett. 2018, 13, 44. [Google Scholar] [CrossRef]
- Dobretsov, S.; Sathe, P.; Bora, T.; Barry, M.; Myint, M.T.Z.; Al Abri, M. Toxicity of Different Zinc Oxide Nanomaterials at 3 Trophic Levels: Implications for Development of Low-Toxicity Antifouling Agents. Environ. Toxicol. Chem. 2020, 39, 1343–1354. [Google Scholar] [CrossRef]
- Du, X.; Zhou, W.; Zhang, W.; Sun, S.; Han, Y.; Tang, Y.; Shi, W.; Liu, G. Toxicities of three metal oxide nanoparticles to a marine microalga: Impacts on the motility and potential affecting mechanisms. Environ. Pollut. 2021, 290, 118027. [Google Scholar] [CrossRef]
- Lin, D.; Xing, B. Root Uptake and Phytotoxicity of ZnO Nanoparticles. Environ. Sci. Technol. 2008, 42, 5580–5585. [Google Scholar] [CrossRef]
- Chevion, M.A. site-specific mechanism for free radical induced biological damage: The essential role of redox-active transition metals. Free Radic. Biol. Med. 1988, 5, 27–37. [Google Scholar] [CrossRef]
- Guo, D.; Bi, H.; Liu, B.; Wu, Q.; Wang, D.; Cui, Y. Reactive oxygen species induced cytotoxic effects of zinc oxide nanoparticles in rat retinal ganglion cells. Toxicol. Vitro. 2013, 27, 731–738. [Google Scholar] [CrossRef]
- Castro-Bugallo, A.; González-Fernández, Á.; Guisande, C.; Barreiro, A. Comparative Responses to Metal Oxide Nanoparticles in Marine Phytoplankton. Arch. Environ. Contam. Toxicol. 2014, 67, 483–493. [Google Scholar] [CrossRef]
- Hazeem, L.J.; Bououdina, M.; Rashdan, S.; Brunet, L.; Slomianny, C.; Boukherroub, R. Cumulative effect of zinc oxide and titanium oxide nanoparticles on growth and chlorophyll a content of Picochlorum sp. Environ. Sci. Pollut. Res. 2016, 23, 2821–2830. [Google Scholar] [CrossRef]
- Broccoli, A.; Anselmi, S.; Cavallo, A.; Ferrari, V.; Prevedelli, D.; Pastorino, P.; Renzi, M. Ecotoxicological effects of new generation pollutants (nanoparticles, amoxicillin and white musk) on freshwater and marine phytoplankton species. Chemosphere 2021, 279, 130623. [Google Scholar] [CrossRef]
- Schiavo, S.; Oliviero, M.; Philippe, A.; Sonia Manzo, S. Nanoparticles based sunscreens provoke adverse effects on marine microalgae Dunaliella tertiolecta. Environ. Sci. Nano 2018, 5, 3011–3022. [Google Scholar] [CrossRef]
- Gunasekarana, D.; Chandrasekarana, N.; Jenkinsb, D.; Mukherjeea. Plain polystyrene microplastics reduce the toxic effects of ZnO particles on marine microalgae Dunaliella salina. J. Environ. Chem. Eng. 2020, 8, 104250. [Google Scholar] [CrossRef]
- Baek, S.; Joo, S.H.; Su, C.; Toborek, M. Toxicity of ZnO/TiO2-conjugated carbon-based nanohybrids on the coastal marine alga Thalassiosira pseudonana. Environ. Toxicol. 2020, 35, 87–96. [Google Scholar] [CrossRef]
- Li, J.; Mao, S.; Ye, Y.; Lü, J.; Jing, F.; Guo, Y.; Liu, H.; Wang, P.; Ma, W.; Qi, P.; et al. Micro-polyethylene particles reduce the toxicity of nano zinc oxide in marine microalgae by adsorption. Environ. Pollut. 2021, 290, 118042. [Google Scholar] [CrossRef]
- Zhu, X.; Tan, L.; Zhao, T.; Huang, W.; Guo, X.; Wang, J.; Wang, J. Alone and combined toxicity of ZnO nanoparticles and graphene quantum dots on microalgae Gymnodinium. Environ. Sci. Pollut. Res. 2022, 29, 47310–47322. [Google Scholar] [CrossRef]

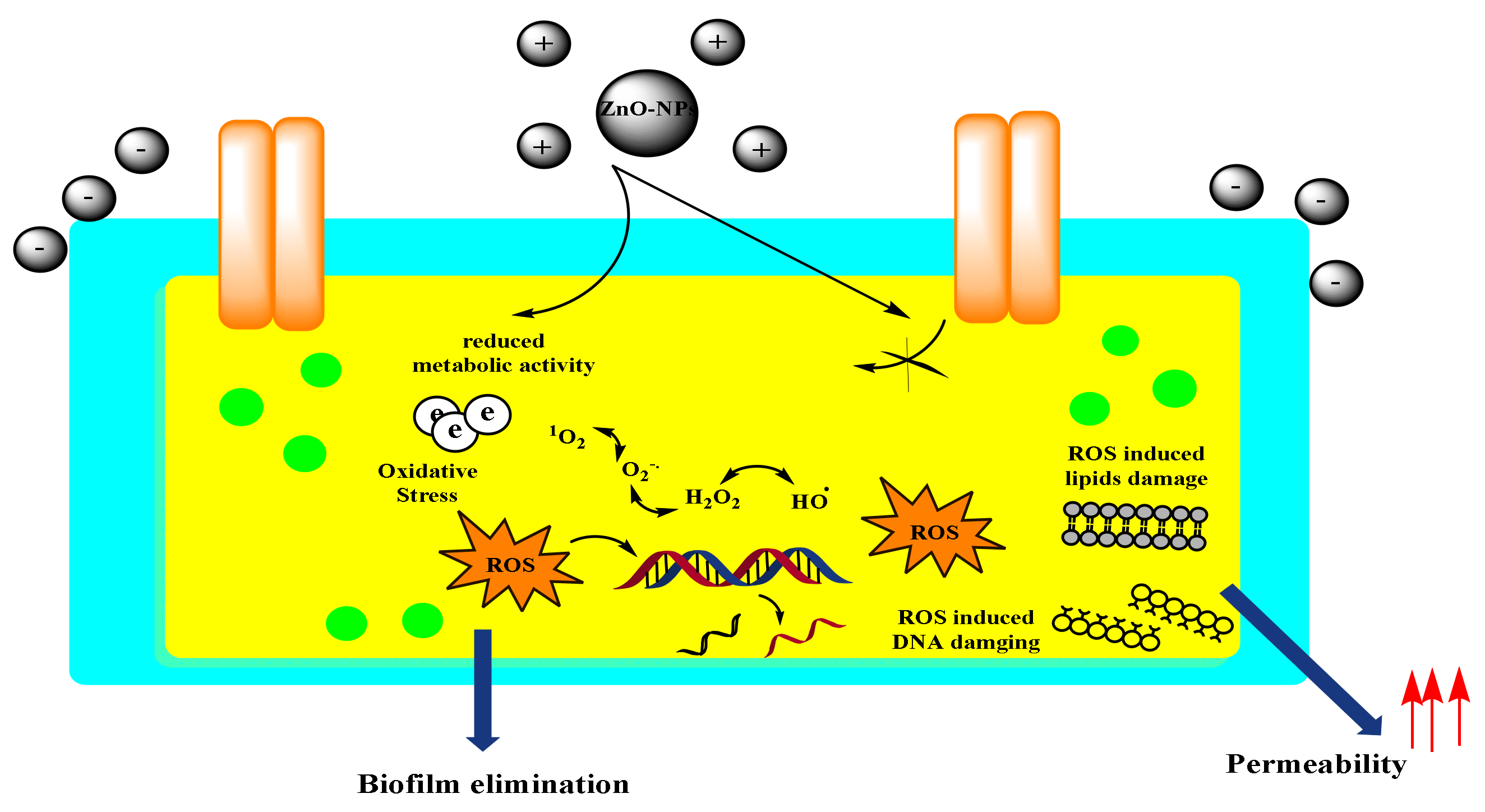
| Compartment | Predicted Environmental Concentration Mode (Q0.15, Q0.85) (µg/L, µg/m3 or µg/Kg) | ||
|---|---|---|---|
| Europe | U.S. | Switzerland | |
| Surface water | 0.010 (0.008, 0.055) | 0.001 (0.001, 0.003) | 0.013 (0.011, 0.058) |
| STP effluent | 0.432 (0.340, 1.42) | 0.3 (0.22, 0.74) | 0.441 (0.343, 1.32) |
| STP sludge | 17.1 (13.6, 57.0) | 23.2 (17.4, 57.7) | 21.4 (16.8, 64.7) |
| Sediment | 2.9 (2.65, 51.7) | 0.51 (0.49, 8.36) | 3.33 (3.30, 56.0) |
| Soil | 0.093 (0.085, 0.661) | 0.050 (0.041, 0.274) | 0.032 (0.026, 0.127) |
| Sludge treated soil | 3.25 (2.98, 23.1) | 1.99 (1.62, 10.9) | na |
| Air | <0.0005 | <0.0005 | <0.0005 |
| Tested Materials | NP Size and Characteristics | Concentration | Microalgae Species | Growth Conditions | Toxic Effect | References |
|---|---|---|---|---|---|---|
| ZnO and TiO2 | “ZnO (50 nm) TiO2 (21 nm; Anatase and rutile phases)” | “10 mg/L” | “Picochlorumsp.” | “5 weeks incubation experiment” | “No significant difference between control and treated cells” | [98] |
| ZnO and TiO2 | - | - | “Dunaliella tertiolecta” | - | “ROS and DNA damage was evidenced in algae cells exposed to sunscreen containing both ZnO and TiO2” | [100] |
| ZnO and polystyrene (PS) microplastic | “ZnO < 50 nm ZnO-Bulk > 100 nm” | “ZnO ((1.22, 12.28 and 122.88 μM) Microplastic (1 mg/L)” | “Dunaliella salina” | “72 h” | “The harmful effects of bulk and nano-sized ZnO particles were considerably reduced in the presence of plain PS microplastics” | [101] |
| ZnO-GO ZnO-CNTs | “GO (1–5 μm diameter, and 0.8–1.2 nm thickness)” “CNT (6–13 nm [O.D.] × 2.5−20 μm [L]” | “50 and 100 mg/L” | “Thalassiosira pseudonana” | “72 h” | “ZnO-GO was the most inhibitory material. The primary mechanism identified was the generation of ROS” | [102] |
| ZnO-NPs Polyethylene Microplastic (PEMP) | “ZnO (20–30 nm)” “150 µm” | “ZnO (2.7 mg/L)” “PEMP (1g/L)” | “Tetraselmis helgolandica var. tsingtaoensis” | “96 h” | “The inhibitory effect of ZnO reduced by 14.4% in the presence of PEMP” | [103] |
| ZnO-NPs Graphene Quantum Dots (GQDs) | “1–20 mg/L” | “Gymnodinium sp. | “The inhibitory effect was decreased compared to the single GQDs and showed antagonistic effect” | [104] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hazeem, L. Single and Combined Toxicity Effects of Zinc Oxide Nanoparticles: Uptake and Accumulation in Marine Microalgae, Toxicity Mechanisms, and Their Fate in the Marine Environment. Water 2022, 14, 2669. https://doi.org/10.3390/w14172669
Hazeem L. Single and Combined Toxicity Effects of Zinc Oxide Nanoparticles: Uptake and Accumulation in Marine Microalgae, Toxicity Mechanisms, and Their Fate in the Marine Environment. Water. 2022; 14(17):2669. https://doi.org/10.3390/w14172669
Chicago/Turabian StyleHazeem, Layla. 2022. "Single and Combined Toxicity Effects of Zinc Oxide Nanoparticles: Uptake and Accumulation in Marine Microalgae, Toxicity Mechanisms, and Their Fate in the Marine Environment" Water 14, no. 17: 2669. https://doi.org/10.3390/w14172669






