Does Biological Activated Carbon Filtration Make Chlor(am)inated Drinking Water Safer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Experimental Procedures
2.2.1. Water Samples
2.2.2. Chlor(am)ine Disinfection By-Products Formation Potential Tests
2.3. DBPs Measurement
2.4. Analytical Methods
3. Results
3.1. Water Characteristics
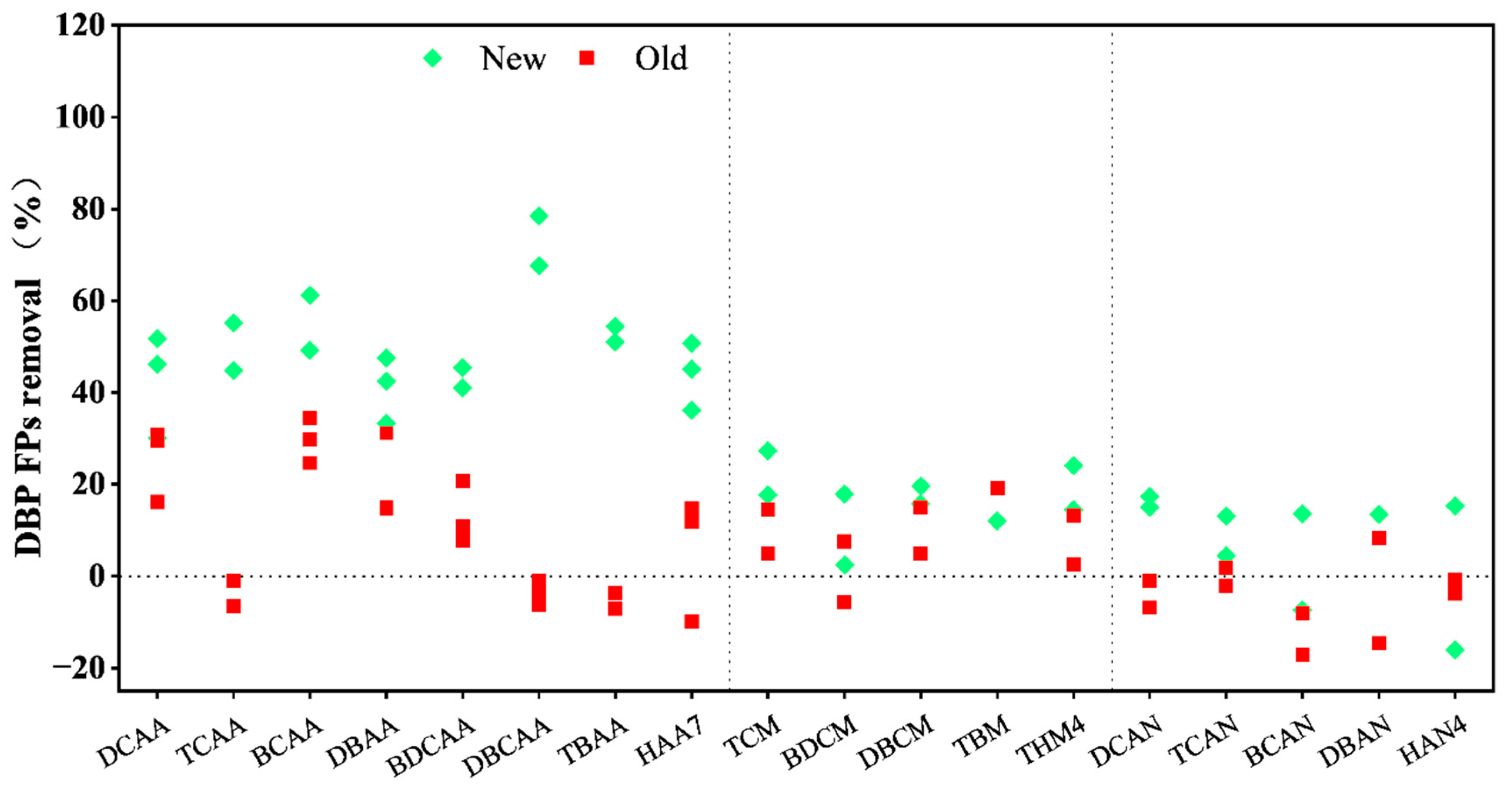
3.2. The Formation of Chlorine Disinfection By-Products
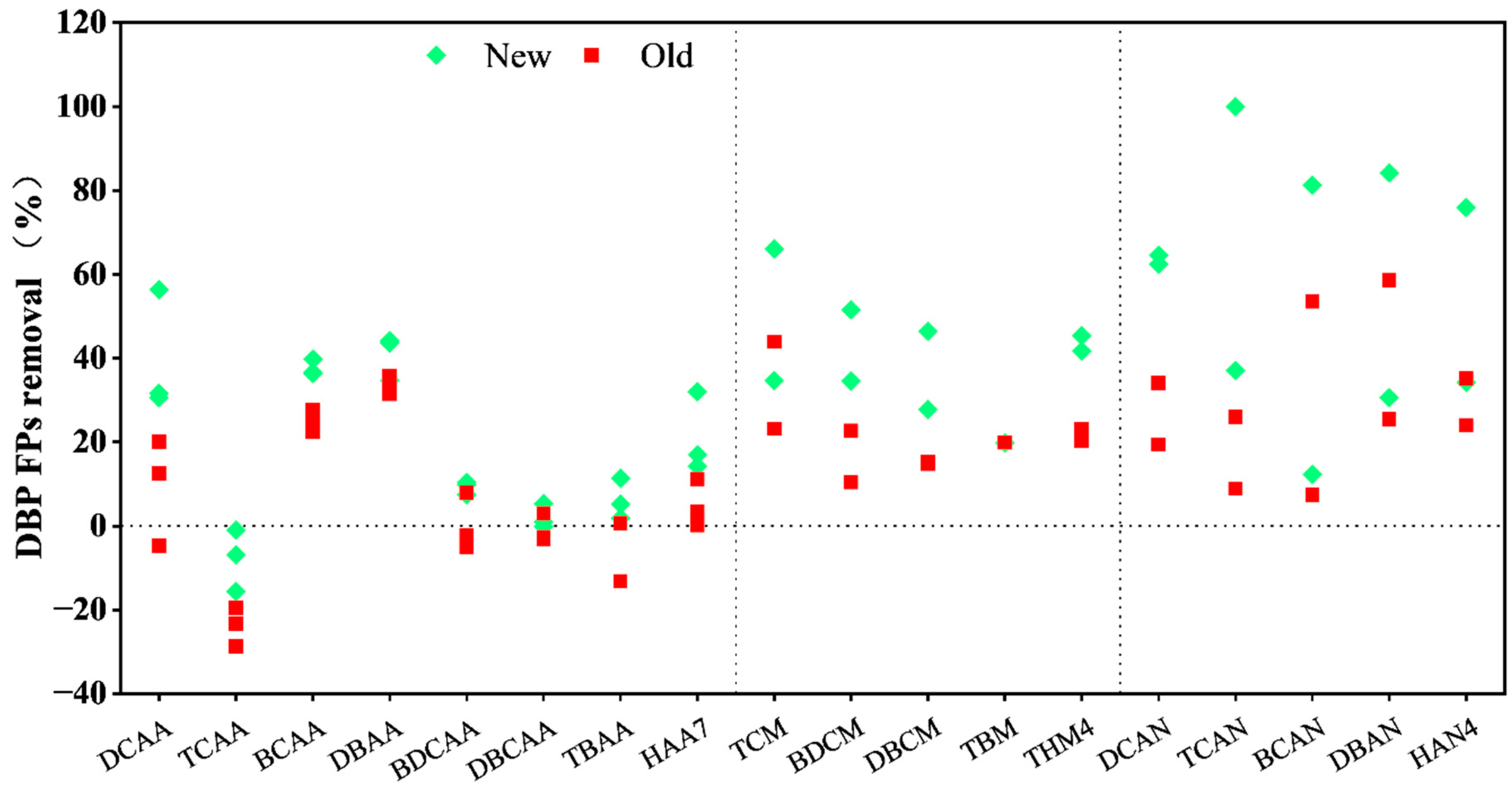
3.3. The Formation of Chloramine Disinfection By-Products
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bull, R.J. Health effects of drinking water disinfectants and disinfectant by-products. Environ. Sci. Technol. 1982, 16, 554A–559A. [Google Scholar] [CrossRef] [PubMed]
- Calderon, R.L. The epidemiology of chemical contaminants of drinking water. Food Chem. Toxicol. 2000, 38, S13–S20. [Google Scholar] [CrossRef]
- Zhang, X.; Echigo, S.; Minear, R.A.; Plewa, M.J. Characterization and Comparison of Disinfection by-Products of Four Major Disinfectants; ACS Symposium Series; Oxford University Press: Oxford, UK, 2000; pp. 299–314. [Google Scholar]
- Richardson, S.D.; Plewa, M.J.; Wagner, E.D.; Schoeny, R.; DeMarini, D.M. Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: A review and roadmap for research. Mutat. Res. 2007, 636, 178–242. [Google Scholar] [CrossRef]
- Plewa, M.J.; Wagner, E.D.; Jazwierska, P.; Richardson, S.D.; Chen, P.H.; McKague, A.B. Halonitromethane Drinking Water Disinfection Byproducts: Chemical Characterization and Mammalian Cell Cytotoxicity and Genotoxicity. Environ. Sci. Technol. 2004, 38, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Chu, W.; Gao, N.; Yin, D.; Krasner, S.W. Formation and speciation of nine haloacetamides, an emerging class of nitrogenous DBPs, during chlorination or chloramination. J. Hazard. Mater. 2013, 260, 806–812. [Google Scholar] [CrossRef]
- Cuthbertson, A.A.; Kimura, S.Y.; Liberatore, H.K.; Knappe, D.R.; Stanford, B.; Summers, R.S.; Dickenson, E.R.; Maness, J.C.; Glover, C.; Selbes, M.; et al. GAC to BAC: Does it make chloraminated drinking water safer? Water Res. 2020, 172, 115432. [Google Scholar] [CrossRef] [PubMed]
- Krasner, S.W.; Lee, T.C.F.; Westerhoff, P.; Fischer, N.; Hanigan, D.; Karanfil, T.; Beita-Sandi, W.; Taylor-Edmonds, L.; Andrews, R.C. Granular Activated Carbon Treatment May Result in Higher Predicted Genotoxicity in the Presence of Bromide. Environ. Sci. Technol. 2016, 50, 9583–9591. [Google Scholar] [CrossRef]
- Tugulea, A.M.; Aranda-Rodriguez, R.; Bérubé, D.; Giddings, M.; Lemieux, F.; Hnatiw, J.; Dabeka, L.; Breton, F. The influence of precursors and treatment process on the formation of Iodo-THMs in Canadian drinking water. Water Res. 2018, 130, 215–223. [Google Scholar] [CrossRef]
- Dong, H.; Qjang, Z.; Richardson, S.D. Formation of Iodinated Disinfection Byproducts (I-DBPs) in Drinking Water: Emerging Concerns and Current Issues. Acc. Chem. Res. 2019, 52, 896–905. [Google Scholar] [CrossRef]
- Liu, L.; Xing, X.; Hu, C.; Wang, H. O3-BAC-Cl2: A multi-barrier process controlling the regrowth of opportunistic waterborne pathogens in drinking water distribution systems. J. Environ. Sci. 2019, 76, 142–153. [Google Scholar] [CrossRef]
- Arnold, M.; Batista, J.; Dickenson, E.; Gerrity, D. Use of ozone-biofiltration for bulk organic removal and disinfection byproduct mitigation in potable reuse applications. Chemosphere 2018, 202, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, P.; Zhu, W.; Han, W.; Zhang, Z. Comparison of O3-BAC, UV/O3-BAC and TiO2/UV/O3-BAC processes for removing organic pollutants in secondary effluents. J. Photochem. Photobiol. A Chem. 2005, 171, 145–151. [Google Scholar] [CrossRef]
- Zhao, X.; Chen, J.; Guo, M.; Li, C.; Hou, N.; Bai, S. Constructed Wetlands Treating Synthetic Wastewater in Response to Day-Night Alterations: Performance and Mechanisms. Chem. Eng. J. 2022, 446, 137460. [Google Scholar]
- Zheng, J.; Lin, T.; Chen, W. Removal of the Precursors of N-Nitrosodiethylamine (Ndea), an Emerging Disinfection Byproduct, in Drinking Water Treatment Process and Its Toxicity to Adult Zebrafish (Danio Rerio). Chemosphere 2018, 191, 1028–1037. [Google Scholar]
- Di Tommaso, C.; Taylor-Edmonds, L.; Andrews, S.A.; Andrews, R.C. The Contribution of Biofilm to Nitrogenous Disinfection by-Product Formation in Full-Scale Cyclically-Operated Drinking Water Biofilters. Water Res. 2019, 155, 403–409. [Google Scholar]
- Li, C.; Wang, D.; Xu, X.; Xu, M.; Wang, Z.; Xiao, R. Tracking changes in composition and amount of dissolved organic matter throughout drinking water treatment plants by comprehensive two-dimensional gas chromatography–quadrupole mass spectrometry. Sci. Total Environ. 2017, 609, 123–131. [Google Scholar] [CrossRef]
- Chu, W.; Li, C.; Gao, N.; Templeton, M.R.; Zhang, Y. Terminating pre-ozonation prior to biological activated carbon filtration results in increased formation of nitrogenous disinfection by-products upon subsequent chlorination. Chemosphere 2015, 121, 33–38. [Google Scholar] [CrossRef]
- Wang, S.; Lin, T.; Chen, W.; Chen, H. Optimization of the Precursor Removal of Dichloroacetonitrile (Dcan), an Emerging Nitrogenous Disinfection by-Product, in an up-Flow Bac Filter. Chemosphere 2017, 189, 309–318. [Google Scholar]
- Xing, X.; Li, T.; Bi, Z.; Qi, P.; Li, Z.; Wang, H.; Lyu, L.; Gao, Y.; Hu, C. Efficient Removal of Disinfection by-Products Precursors and Inhibition of Bacterial Detachment by Strong Interaction of Eps with Coconut Shell Activated Carbon in Ozone/Biofiltration. J. Hazard. Mater. 2020, 392, 122077. [Google Scholar]
- Hong, S.; Tang, X.; Wu, N.; Chen, H. Leakage of Soluble Microbial Products from Biological Activated Carbon Filtration in Drinking Water Treatment Plants and Its Influence on Health Risks. Chemosphere 2018, 202, 626–636. [Google Scholar] [CrossRef]
- Zheng, J.; Lin, T.; Chen, W.; Tao, H.; Tan, Y.; Ma, B. Removal of Precursors of Typical Nitrogenous Disinfection Byproducts in Ozonation Integrated with Biological Activated Carbon (O3/Bac). Chemosphere 2018, 209, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.; Laurent, P.; Kihn, A.; Prévost, M.; Servais, P. Impact of Temperature on Nitrification in Biological Activated Carbon (Bac) Filters Used for Drinking Water Treatment. Water Res. 2001, 35, 2923–2934. [Google Scholar]
- Dong, B.; Qin, X.; Li, T.; Du, J.; Wang, J.; Jiang, F.; Zhang, X. Variation of Water Quality of Biological Activated Carbon for a Long Times Operation. Water Wastewater Eng. 2021, 47, 16–23.962E. [Google Scholar]
- Gerrity, D.; Arnold, M.; Dickenson, E.; Moser, D.; Sackett, J.D.; Wert, E.C. Microbial Community Characterization of Ozone-Biofiltration Systems in Drinking Water and Potable Reuse Applications. Water Res. 2018, 135, 207–219. [Google Scholar]
- Hu, S.; Shi, L.; Da, Y.; Hu, K. Tracking Analysis of Activated Carbon Filter in Water Treatment Plant. Water Wastewater Eng. 2019, 45, 13–17. [Google Scholar]
- Krasner, S.W.; Weinberg, H.S.; Richardson, S.D.; Pastor, S.J.; Chinn, R.; Sclimenti, M.J.; Onstad, G.D.; Thruston, A.D., Jr. Occurrence of a New Generation of Disinfection Byproducts. Environ. Sci. Technol. 2006, 40, 7175–7185. [Google Scholar] [CrossRef] [PubMed]
- Chu, W.H.; Gao, N.Y.; Deng, Y.; Templeton, M.R.; Yin, D.Q. Formation of nitrogenous disinfection by-products from pre-chloramination. Chemosphere 2011, 85, 1187–1191. [Google Scholar] [CrossRef] [PubMed]
- Mitch, W.A.; Sedlak, D.L. Formation of N-nitrosodimethylamine (NDMA) from dimethylamine during chlorination. Environ. Sci. Technol. 2002, 36, 588–595. [Google Scholar] [CrossRef]
- Zhang, A.; Chu, W.; Bond, T.; Wang, F.; Pan, Y.; Tong, J.; Zhu, H. Interference from haloacetamides during the determination of haloacetic acids using gas chromatography. J. Hromatography A 2020, 1612, 460652. [Google Scholar] [CrossRef]
- Fu, J.; Lee, W.N.; Coleman, C.; Meyer, M.; Carter, J.; Nowack, K.; Huang, C.H. Pilot investigation of two-stage biofiltration for removal of natural organic matter in drinking water treatment. Chemosphere 2017, 166, 311–322. [Google Scholar] [CrossRef]
- Fu, J.; Huang, C.H.; Dang, C.; Wang, Q. A review on treatment of disinfection byproduct precursors by biological activated carbon process. Chin. Chem. Lett. 2022, 33, 4495–4504. [Google Scholar] [CrossRef]
- Fu, J.; Lee, W.N.; Coleman, C.; Nowack, K.; Carter, J.; Huang, C.H. Removal of disinfection byproduct (DBP) precursors in water by two-stage biofiltration treatment. Water Res. 2017, 123, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Gui, X.; Zhang, H.; Ji, B.; Ma, J.; Xu, M.; Li, Y.; Yan, M. Study on the Control of Dichloroacetonitrile Generation by Two-Point Influent Activated Carbon-Quartz Sand Biofilter. Membranes 2022, 12, 137. [Google Scholar] [CrossRef]
- Liao, X.; Chen, C.; Zhang, J.; Dai, Y.; Zhang, X.; Xie, S. Operational performance, biomass and microbial community structure: Impacts of backwashing on drinking water biofilter. Environ. Sci. Pollut. Res. 2015, 22, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Lin, T.; Jiang, F.; Dong, J.; Chen, W.; Zhou, D. The shadow of dichloroacetonitrile (DCAN), a typical nitrogenous disinfection by-product (N-DBP), in the waterworks and its backwash water reuse. Chemosphere 2017, 181, 569–578. [Google Scholar] [CrossRef] [PubMed]
| GAC Bed Depth (m) | GAC Particle Size (mm) | Loading Density (g/dm3) | Empty Bed Contact Time (min) | Backwash Time (min) |
|---|---|---|---|---|
| 2.1 | 0.88 | 488 | 20–30 | 7.0 |
| GC/ECD: | Clarus 680, PerkinElmer, USA | ||
| Columns: | Elite-5, 30 m × 0.25 mm ID, 0.25 μm film thickness | ||
| Carrier gas: | Nitrogen, constant flow at 3 mL per minute | ||
| Injection volume: | 1 μL | ||
| Temperature programmes: | |||
| Vaporizing chamber: | 200 °C | ||
| for THM4 | for HAA7 | for HAN4 | |
| GC column | Initial temperature at 37 °C for 3 min, then 10 °C per minute to 80 °C and hold for 2 min and finally 20 °C per minute to 220 °C and hold for 1 min. | Initial temperature at 40 °C for 7 min, then 2.5 °C per minute to 65 °C and 5 °C per minute to 85 °C and hold for 1 min finally 20 °C per minute to 210 °C and hold for 5 min. | Initial temperature at 30 °C for 10 min, then 17 °C per minute to 72 °C and hold for 1 min and finally 40 °C per minute to 200 °C and hold for 2 min. |
| Detector: | 300 °C | ||
| Sampling Time | Water Sample | DOC (mg/L) | SUVA254 | TDN (mg/L) |
|---|---|---|---|---|
| January | Influent | 4.02 ± 0.09 | 0.82 | 1.49 ± 0.01 |
| Effluent of new BAC | 3.01 ± 0.15 | 0.86 | 1.53 ± 0.04 | |
| Effluent of old BAC | 4.09 ± 0.21 | 0.68 | 1.50 ± 0.02 | |
| October | Influent | 3.81 ± 0.26 | 0.93 | 1.36 ± 0.06 |
| Effluent of new BAC | 3.35 ± 0.24 | 0.67 | 1.62 ± 0.18 | |
| Effluent of old BAC | 3.51 ± 0.26 | 0.96 | 1.37 ± 0.06 |
| DBP Category | Sampling Time | DBP Species | Influent Concentration (μg/L) | Effluent Concentration in New BAC (μg/L) | Effluent Concentration in Old BAC (μg/L) |
|---|---|---|---|---|---|
| THMs | Jan. | TCM | 60.81 ± 0.40 | 50.06 ± 2.52 | 57.80 ± 1.45 |
| BDCM | 18.37 ± 2.68 | 17.92 ± 1.18 | 19.41 ± 0.27 | ||
| DBCM | 4.73 ± 0.43 | 3.80 ± 0.17 | 4.50 ± 0.07 | ||
| TBM | Not Detected | Not Detected | Not Detected | ||
| Oct. | TCM | 22.71 ± 1.08 | 16.51 ± 0.65 | 19.40 ± 0.70 | |
| BDCM | 6.81 ± 0.25 | 5.59 ± 0.01 | 6.29 ± 0.49 | ||
| DBCM | 3.33 ± 0.31 | 2.87 ± 0.10 | 2.83 ± 0.14 | ||
| TBM | 0.34 ± 0.04 | 0.30 ± 0.01 | 0.27 ± 0.02 | ||
| Total average | 58.55 ± 35.86 | 48.50 ± 32.94 | 55.26 ± 37.40 | ||
| HANs | Jan. | DCAN | 64.25 ± 2.51 | 53.09 ± 2.78 | 64.94 ± 8.65 |
| TCAN | 7.82 ± 0.19 | 7.46 ± 0.18 | 7.97 ± 0.31 | ||
| BCAN | 7.35 ± 0.66 | 6.35 ± 0.21 | 8.61 ± 0.56 | ||
| DBAN | 15.49 ± 0.70 | 13.41 ± 0.36 | 14.21 ± 0.79 | ||
| Oct. | DCAN | 1.18 * | 1.54 ± 0.11 | 1.93 ± 0.17 | |
| TCAN | 19.41 ± 0.01 | 16.82 ± 1.57 | 19.06 ± 0.98 | ||
| BCAN | 6.68 ± 0.13 | 7.18 ± 0.36 | 7.22 ± 0.60 | ||
| DBAN | 6.90 * | 14.82 ± 0.43 | 7.91 ± 0.89 | ||
| Total average | 62.86 ± 42.50 | 60.36 ± 28.23 | 65.93 ± 42.14 | ||
| HAAs | Jan. | DCAA | 128.76 ± 8.10 | 73.20 ± 9.57 | 95.57 ± 4.62 |
| TCAA | 73.83 ± 6.50 | 38.53 ± 3.93 | 73.55 ± 2.16 | ||
| BCAA | 36.02 ± 1.79 | 16.57 ± 2.88 | 25.29 ± 0.64 | ||
| DBAA | 23.24 ± 1.16 | 14.96 ± 3.44 | 19.94 ± 0.83 | ||
| BDCAA | 28.30 ± 0.25 | 16.69 ± 3.89 | 21.40 ± 1.08 | ||
| DBCAA | 71.36 ± 2.17 | 19.54 ± 5.58 | 73.98 ± 1.31 | ||
| TBAA | 60.31 ± 1.87 | 29.02 ± 1.44 | 64.20 ± 1.44 | ||
| Total | 421.84 ± 18.49 | 208.55 ± 46.13 | 373.95 ± 4.23 |
| DBP Category | Sampling Time | DBP Species | Influent Concentration (μg/L) | Effluent Concentration in Newew BAC (μg/L) | Effluent Concentration in Od BAC (μg/L) | |
|---|---|---|---|---|---|---|
| THMs | Jan. | TCM | 3.02 ± 0.51 | 1.97 ± 0.10 | 2.31 ± 0.17 | |
| BDCM | 4.65 ± 0.24 | 2.25 ± 0.09 | 3.60 ± 0.11 | |||
| DBCM | 3.65 ± 0.09 | 1.95 ± 0.02 | 3.11 ± 0.04 | |||
| TBM | Not Detected | Not Detected | Not Detected | |||
| Oct. | TCM | 1.50 ± 0.01 | 0.51 ± 0.27 | 0.84 ± 0.05 | ||
| BDCM | 1.48 ± 0.03 | 0.96 ± 0.06 | 1.33 ± 0.03 | |||
| DBCM | 1.54 ± 0.01 | 1.12 ± 0.03 | 1.31 ± 0.05 | |||
| TBM | 0.22 ± 0.01 | 0.18 ± 0.003 | 0.17 ± 0.002 | |||
| Total average | 8.03 ± 4.65 | 4.47 ± 2.41 | 6.34 ± 3.80 | |||
| HANs | Jan. | DCAN | 34.37 ± 3.18 | 12.17 ± 1.11 | 27.69 ± 6.49 | |
| TCAN | 6.62 ± 0.50 | Not Detected | 6.03 ± 0.41 | |||
| BCAN | 23.24 ± 1.81 | 4.36 ± 0.53 | 10.79 ± 1.68 | |||
| DBAN | 12.46 ± 1.31 | 1.97 ± 0.41 | 5.17 ± 0.82 | |||
| Oct. | DCAN | 0.63 * | 0.24 * | 0.41 * | ||
| TCAN | 52.32 ± 2.06 | 32.90 ± 3.83 | 38.71 ± 0.21 | |||
| BCAN | 7.18 ± 0.19 | 6.30 ± 0.50 | 6.65 ± 0.45 | |||
| DBAN | 4.21 ± 0.92 | 2.92 ± 0.04 | 3.14 ± 0.10 | |||
| Total average | 70.51 ± 8.74 | 30.43 ± 16.87 | 49.30 ± 0.55 | |||
| HAAs | Jan. | DCAA | 24.48 ± 2.73 | 20.01 ± 0.15 | 27.70 ± 1.06 | |
| TCAA | 12.27 ± 0.63 | 13.21 ± 0.38 | 15.18 ± 0.37 | |||
| BCAA | 9.61 ± 0.32 | 5.99 ± 0.03 | 7.21 ± 0.14 | |||
| DBAA | 4.72 ± 0.16 | 2.79 ± 0.25 | 3.14 ± 0.01 | |||
| BDCAA | 8.48 ± 0.18 | 7.70 ± 0.13 | 8.46 ± 0.41 | |||
| DBCAA | 9.77 ± 0.25 | 9.57 ± 0.07 | 9.86 ± 0.13 | |||
| TBAA | 5.41 ± 0.48 | 5.06 ± 0.20 | 5.28 ± 0.45 | |||
| Total | 77.73 ± 2.81 | 64.33 ± 5.41 | 73.84 ± 1.77 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, J.; Wang, F.; Zhang, L.; Hu, Y.; He, C. Does Biological Activated Carbon Filtration Make Chlor(am)inated Drinking Water Safer. Water 2022, 14, 2640. https://doi.org/10.3390/w14172640
Pan J, Wang F, Zhang L, Hu Y, He C. Does Biological Activated Carbon Filtration Make Chlor(am)inated Drinking Water Safer. Water. 2022; 14(17):2640. https://doi.org/10.3390/w14172640
Chicago/Turabian StylePan, Jiazheng, Feifei Wang, Lu Zhang, Yulin Hu, and Chiquan He. 2022. "Does Biological Activated Carbon Filtration Make Chlor(am)inated Drinking Water Safer" Water 14, no. 17: 2640. https://doi.org/10.3390/w14172640









