Cyanobacterial Bloom Phenology in Green Bay Using MERIS Satellite Data and Comparisons with Western Lake Erie and Saginaw Bay
Abstract
:1. Introduction
2. Materials and Methods
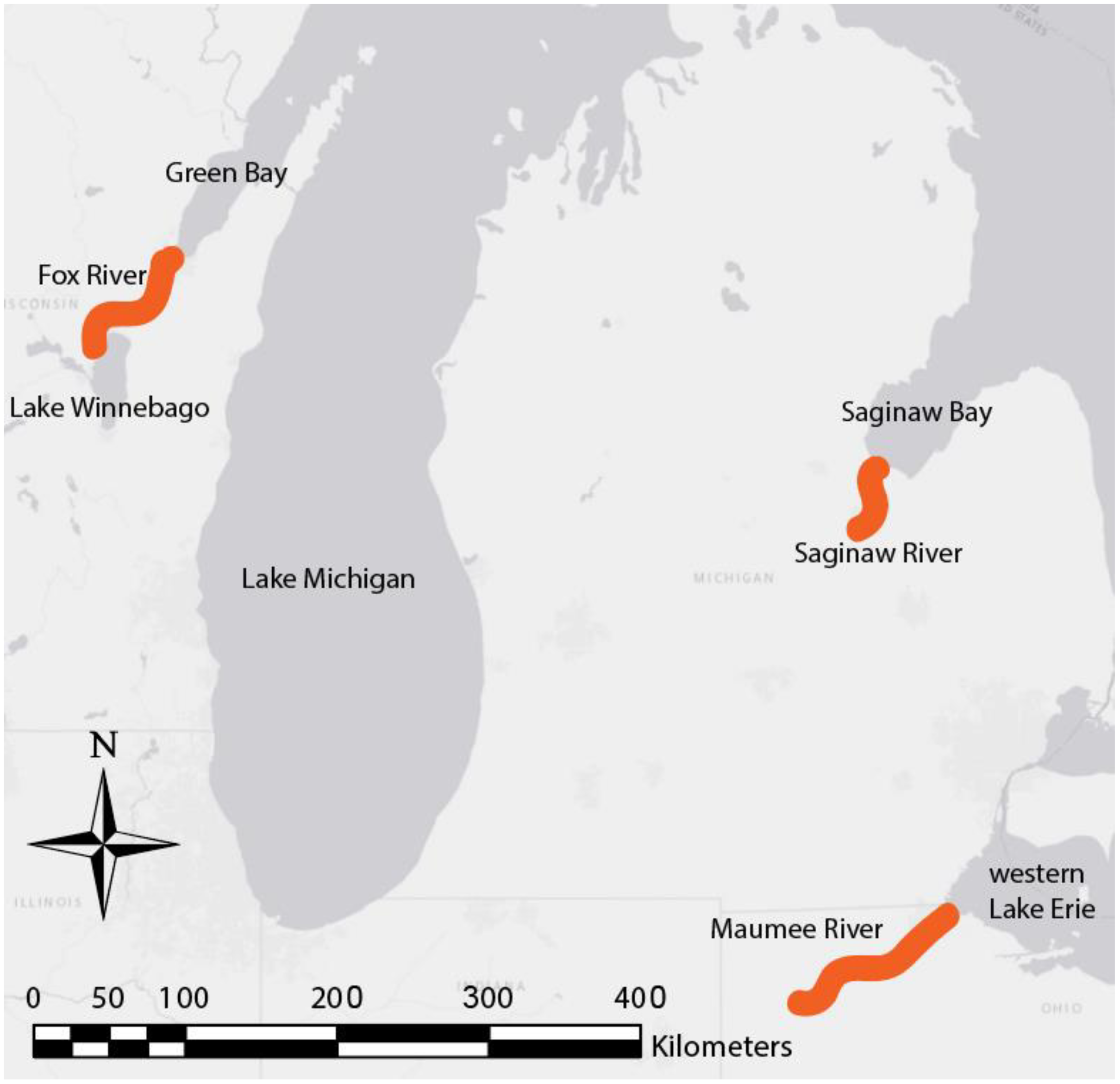
2.1. Study Site
2.2. Satellite Imagery
2.3. Environmental Data
2.3.1. Biovolume Enumeration
2.3.2. Quantifying Cyanobacterial from Other Planktonic Groups and Algorithmic Validation
2.4. Climatology
2.5. Model Building in Green Bay
2.6. River Discharge
2.7. Other Environmental Data from NASA Giovanni
2.7.1. Water Temperature
2.7.2. Wind Speed
2.7.3. Gelbstoff and Detrital Absorption
2.7.4. Latent Heat Flux
2.8. Statistical Comparisons between Basins
2.8.1. Differences in Green Bay Relative to Western Lake Erie and Saginaw Bay
2.8.2. High Bloom Years vs. Low Bloom Years
3. Results
3.1. Estimating Cyanobacteria Using CIcyano
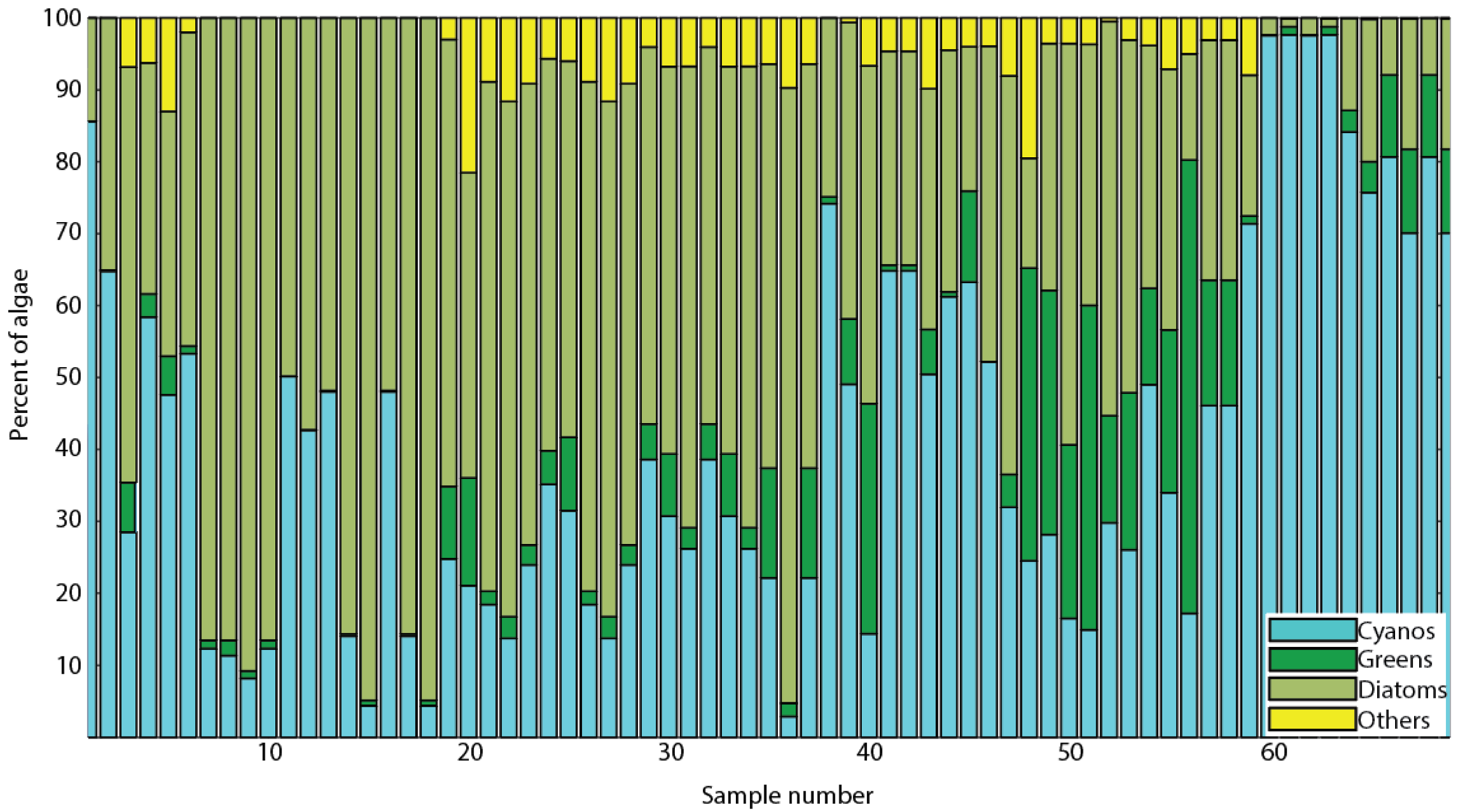
3.2. Algal Diversity in Green Bay
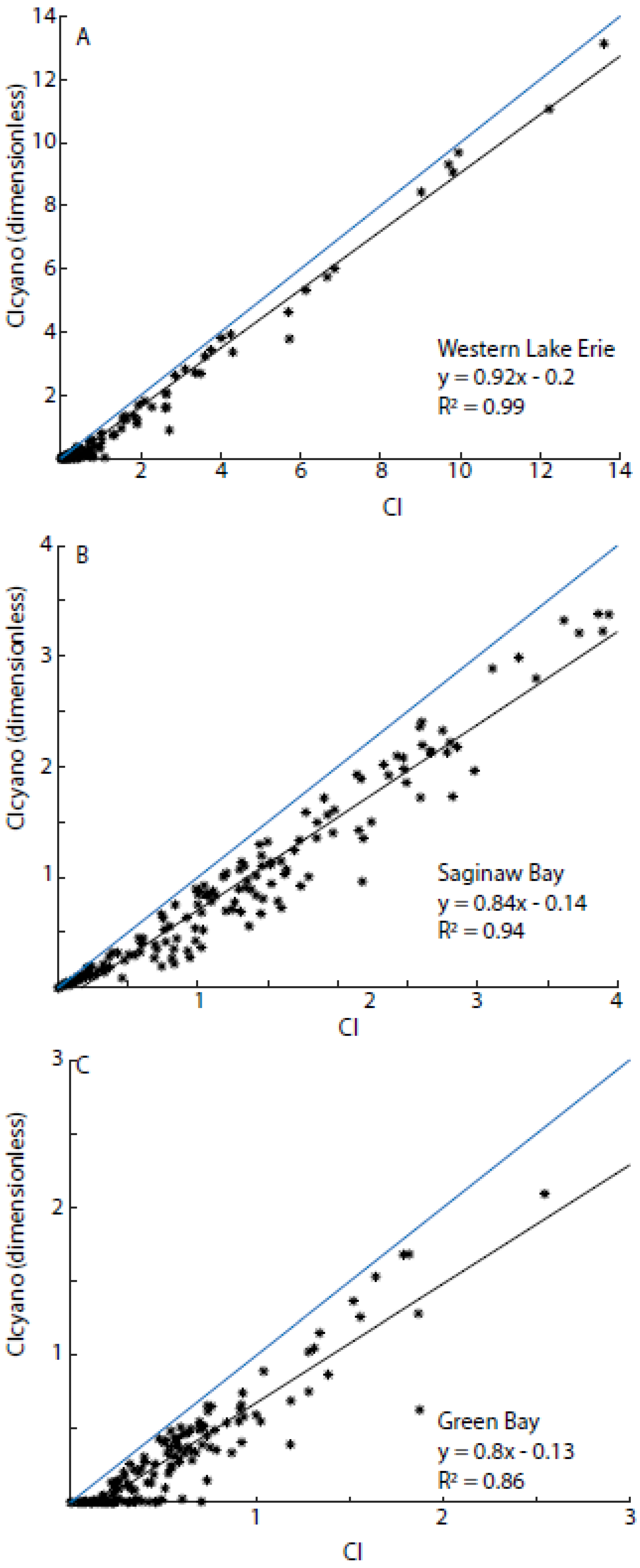
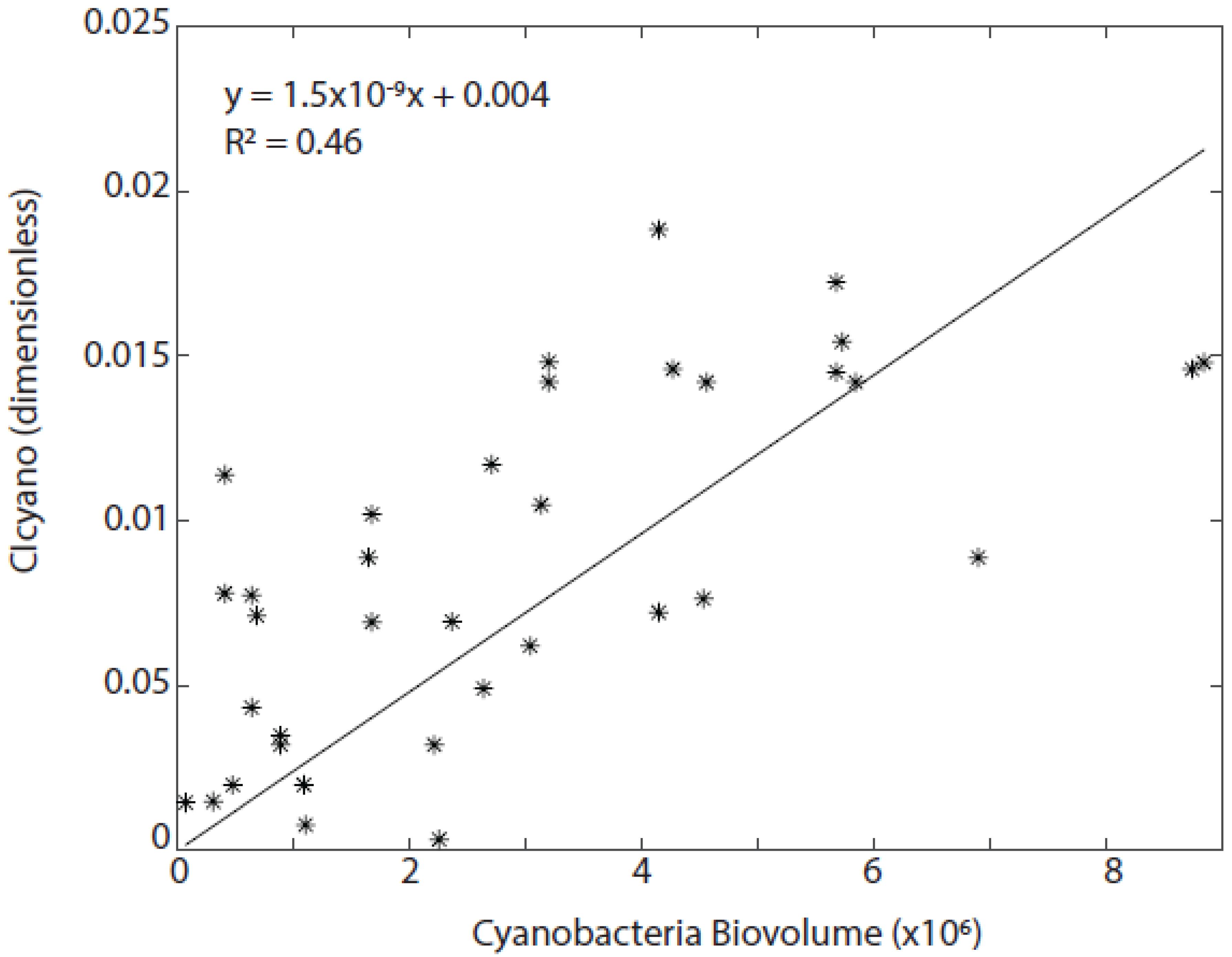
3.3. Algorithmic Validation
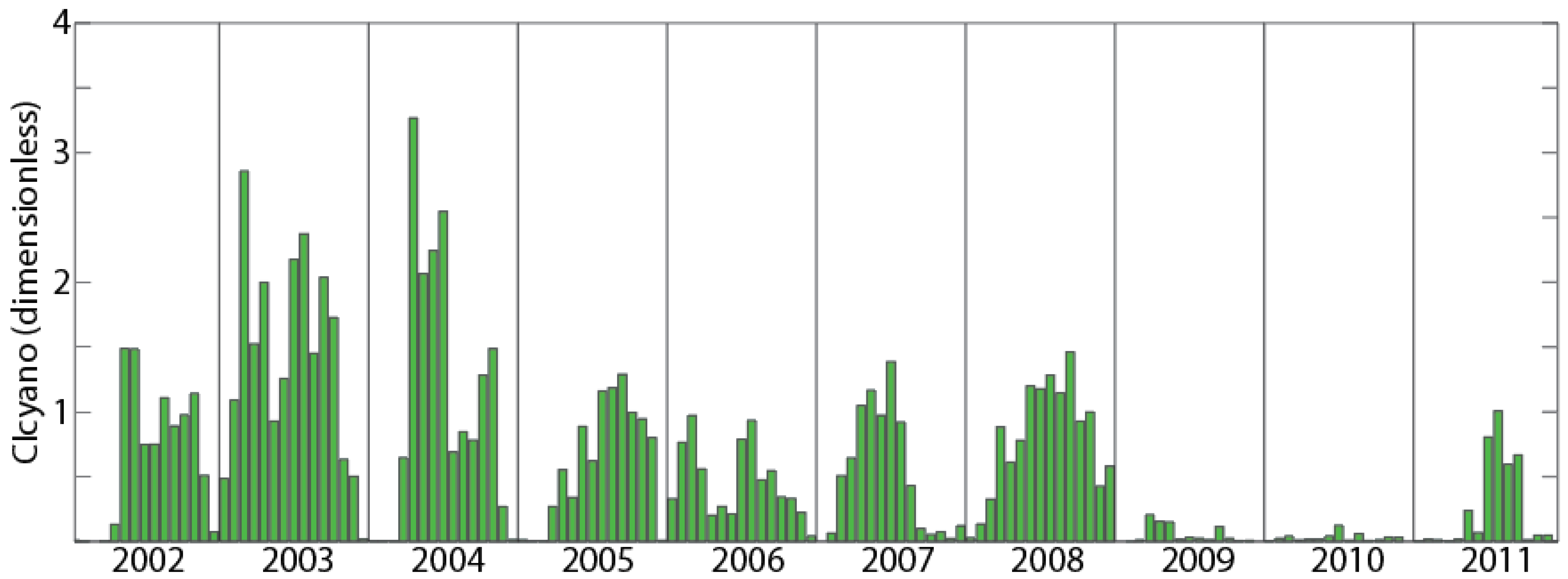
3.4. Climatological Analysis
3.5. Model Building
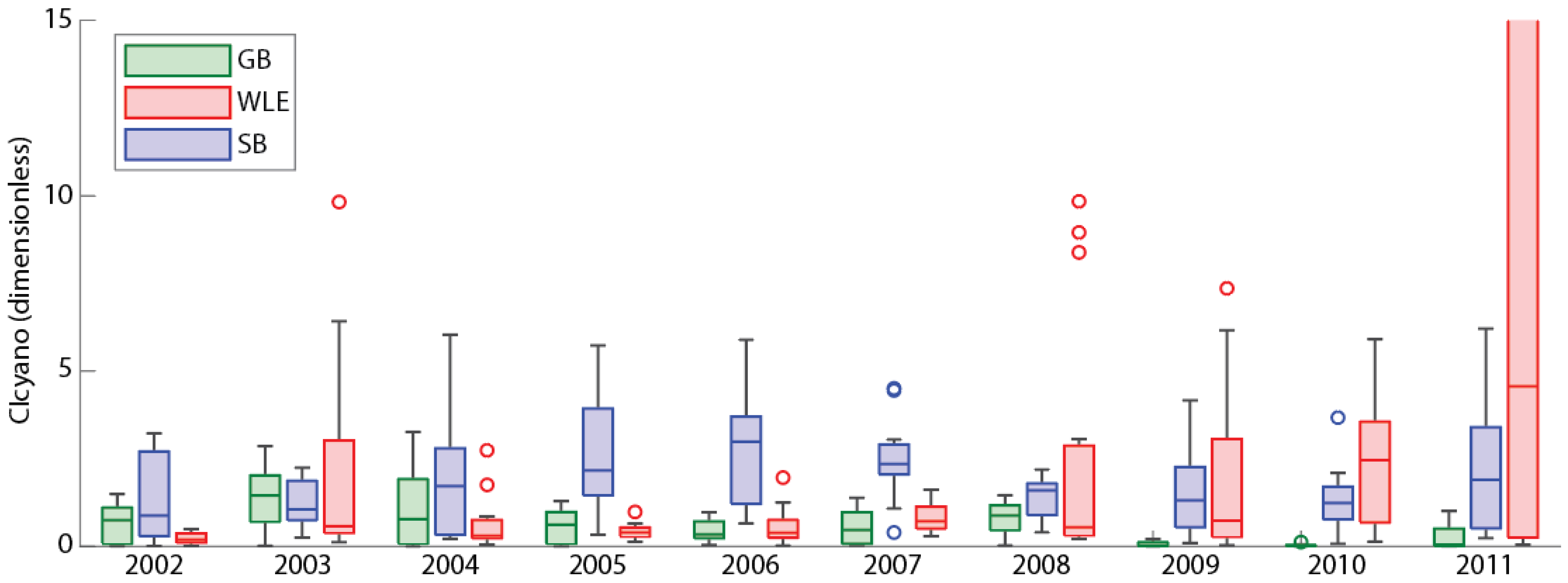
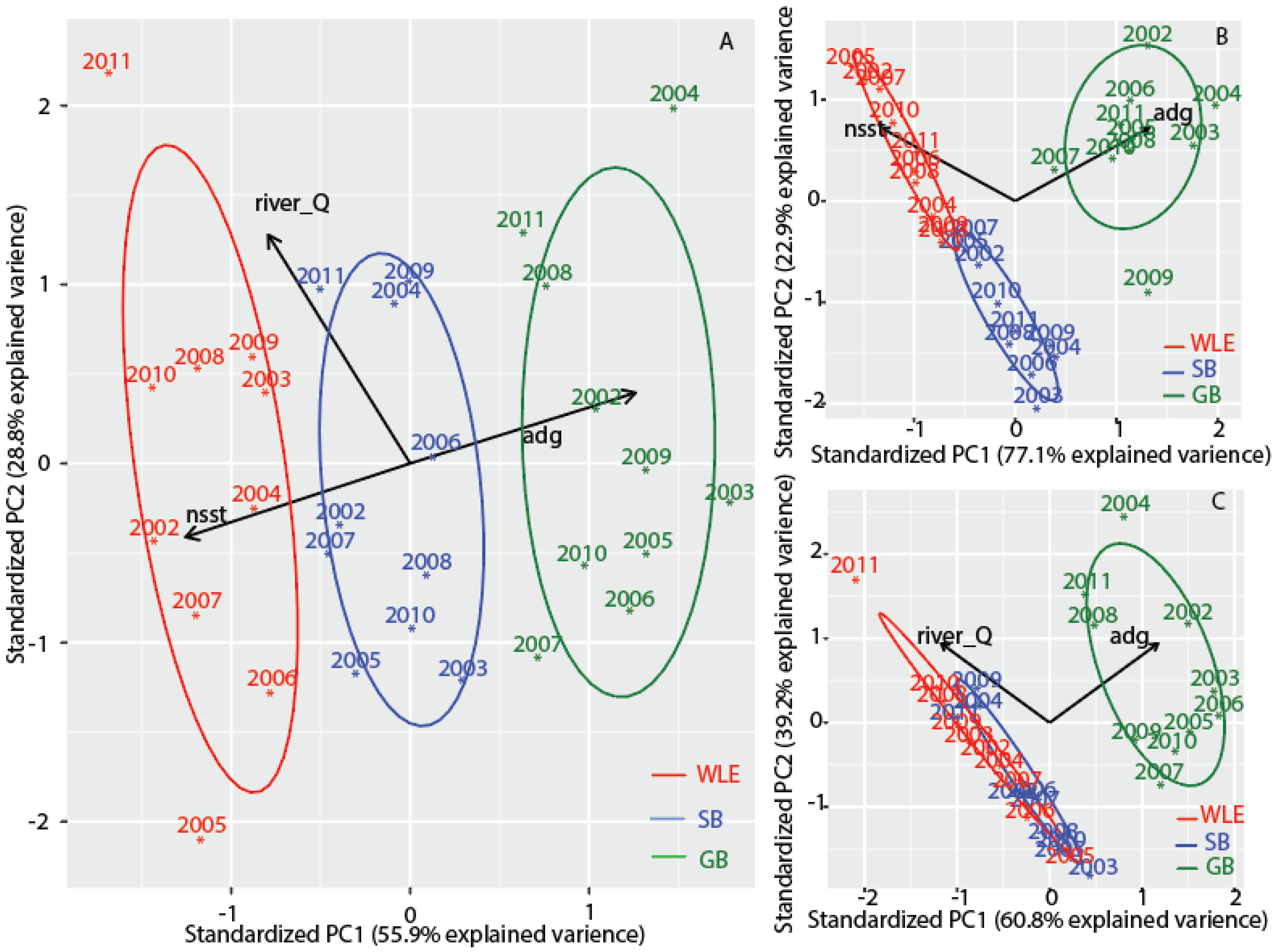
3.6. Comparisons with Western Lake Erie and Saginaw Bay
3.7. Separating High Bloom Years from Low Bloom Years
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Klump, J.V.; Bratton, J.; Fermanich, K.; Forsythe, P.; Harris, H.J.; Howe, R.W.; Kaster, J.L. Green bay, lake Michigan: A proving ground for great lakes restoration. J. Great Lakes Res. 2018, 44, 825–828. [Google Scholar] [CrossRef]
- Klump, J.V.; Fitzgerald, S.A.; Waplesa, J.T. Benthic biogeochemical cycling, nutrient stoichiometry, and carbon and nitrogen mass balances in a eutrophic freshwater bay. Limnol. Oceanogr. 2009, 54, 692–712. [Google Scholar] [CrossRef]
- Harris, H.J.; Wenger, R.B.; Sager, P.E.; Klump, J.V. The Green Bay saga: Environmental change, scientific investigation, and watershed management. J. Great Lakes Res. 2018, 44, 829–836. [Google Scholar] [CrossRef]
- Ditton, R.B.; Goodale, T.L. Water quality perception and the recreational uses of Green Bay, Lake Michigan. Water Resour. Res. 1973, 9, 569–579. [Google Scholar] [CrossRef]
- Sayers, M.; Fahnenstiel, G.L.; Shuchman, R.A.; Whitley, M. Cyanobacteria blooms in three eutrophic basins of the Great Lakes: A comparative analysis using satellite remote sensing. Int. J. Remote Sens. 2016, 37, 4148–4171. [Google Scholar] [CrossRef]
- De Stasio, B.T.; Schrimpf, M.B.; Cornwell, B.H. Phytoplankton communities in Green Bay, Lake Michigan after invasion by dreissenid mussels: Increased dominance by cyanobacteria. Diversity 2014, 6, 681–704. [Google Scholar] [CrossRef]
- Arnott, D.L.; Vanni, M.J. Nitrogen and phosphorus recycling by the zebra mussel (Dreissena polymorpha) in the western basin of Lake Erie. Can. J. Fish. Aquat. Sci. 1996, 53, 646–659. [Google Scholar] [CrossRef]
- Conroy, J.D.; Edwards, W.J.; Pontius, R.A.; Kane, D.D.; Zhang, H.; Shea, J.F.; Richey, J.N.; Culver, D.A. Soluble nitrogen and phosphorus excretion of exotic freshwater mussels (Dreissena spp.): Potential impacts for nutrient remineralisation in western Lake Erie. Freshw. Biol. 2005, 50, 1146–1162. [Google Scholar] [CrossRef]
- Chen, L.; Giesy, J.P.; Adamovsky, O.; Svirčev, Z.; Meriluoto, J.; Codd, G.A.; Mijovic, B.; Shi, T.; Tuo, X.; Li, S.-C. Challenges of using blooms of Microcystis spp. in animal feeds: A comprehensive review of nutritional, toxicological and microbial health evaluation. Sci. Total Environ. 2021, 764, 142319. [Google Scholar] [CrossRef]
- Carmichael, W.W. A status report of planktonic cyanobacteria (blue-green algae) and their toxins. U. S. Environ. Prot. Agency 1992, 600, 32–33. [Google Scholar]
- Chorus, I.; Bartram, J. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Steffen, M.M.; Davis, T.W.; McKay, R.M.L.; Bullerjahn, G.S.; Krausfeldt, L.E.; Stough, J.M.A.; Neitzey, M.L.; Gilbert, N.E.; Boyer, G.L.; Johengen, T.H.; et al. Ecophysiological Examination of the Lake Erie Microcystis Bloom in 2014: Linkages between Biology and the Water Supply Shutdown of Toledo, OH. Environ. Sci. Technol. 2017, 51, 6745–6755. [Google Scholar] [CrossRef]
- Backer, L.C. Cyanobacterial harmful algal blooms (CyanoHABs): Developing a public health response. Lake Reserv. Manag. 2002, 18, 20–31. [Google Scholar] [CrossRef]
- Dodds, W.K.; Bouska, W.W.; Eitzmann, J.L.; Pilger, T.J.; Pitts, K.L.; Riley, A.J.; Schloesser, J.T.; Thornbrugh, D.J. Eutrophication of US Freshwaters: Analysis of Potential Economic Damages; ACS Publications: Washington, DC, USA, 2009. [Google Scholar]
- Ibelings, B.W.; Vonk, M.; Los, H.F.; van der Molen, D.T.; Mooij, W.M. Fuzzy modeling of cyanobacterial surface waterblooms: Validation with NOAA-AVHRR satellite images. Ecol. Appl. 2003, 13, 1456–1472. [Google Scholar] [CrossRef]
- Brooks, B.W.; Lazorchak, J.M.; Howard, M.D.; Johnson, M.V.V.; Morton, S.L.; Perkins, D.A.; Reavie, E.D.; Scott, G.I.; Smith, S.A.; Steevens, J.A. Are harmful algal blooms becoming the greatest inland water quality threat to public health and aquatic ecosystems? Environ. Toxicol. Chem. 2016, 35, 6–13. [Google Scholar] [CrossRef]
- Bartlett, S.L.; Brunner, S.L.; Klump, J.V.; Houghton, E.M.; Miller, T.R. Spatial analysis of toxic or otherwise bioactive cyanobacterial peptides in Green Bay, Lake Michigan. J. Great Lakes Res. 2018, 44, 924–933. [Google Scholar] [CrossRef]
- Kraft, M.E. Sustainability and water quality: Policy evolution in Wisconsin’s Fox-Wolf River basin. Public Work. Manag. Policy 2006, 10, 202–213. [Google Scholar] [CrossRef]
- Dolan, D.M.; Chapra, S.C. Great Lakes total phosphorus revisited: 1. Loading analysis and update (1994–2008). J. Great Lakes Res. 2012, 38, 730–740. [Google Scholar] [CrossRef]
- Wynne, T.T.; Stumpf, R.P.; Litaker, R.W.; Hood, R.R. Cyanobacterial bloom phenology in Saginaw Bay from MODIS and a comparative look with western Lake Erie. Harmful Algae 2021, 103, 101999. [Google Scholar] [CrossRef]
- Stumpf, R.P.; Wynne, T.T.; Baker, D.B.; Fahnenstiel, G.L. Interannual Variability of Cyanobacterial Blooms in Lake Erie. PLoS ONE 2012, 7, e42444. [Google Scholar] [CrossRef]
- Gons, H.J.; Auer, M.T.; Effler, S.W. MERIS satellite chlorophyll mapping of oligotrophic and eutrophic waters in the Laurentian Great Lakes. Remote Sens. Environ. 2008, 112, 4098–4106. [Google Scholar]
- Wynne, T.T.; Stumpf, R.P. Spatial and temporal patterns in the seasonal distribution of toxic cyanobacteria in western lake erie from 2002–2014. Toxins 2015, 7, 1649–1663. [Google Scholar] [CrossRef] [PubMed]
- Vincent, R.K.; Qin, X.; McKay, R.M.L.; Miner, J.; Czajkowski, K.; Savino, J.; Bridgeman, T. Phycocyanin detection from LANDSAT TM data for mapping cyanobacterial blooms in Lake Erie. Remote Sens. Environ. 2004, 89, 381–392. [Google Scholar] [CrossRef]
- Wynne, T.T.; Stumpf, R.P.; Tomlinson, M.C.; Dyble, J. Characterizing a cyanobacterial bloom in western Lake Erie using satellite imagery and meteorological data. Limnol. Oceanogr. 2010, 55, 2025–2036. [Google Scholar] [CrossRef]
- Wynne, T.T.; Stumpf, R.P.; Tomlinson, M.C.; Warner, R.A.; Tester, P.A.; Dyble, J.; Fahnenstiel, G.L. Relating spectral shape to cyanobacterial blooms in the Laurentian Great Lakes. Int. J. Remote Sens. 2008, 29, 3665–3672. [Google Scholar] [CrossRef]
- Seppälä, J.; Ylöstalo, P.; Kaitala, S.; Hällfors, S.; Raateoja, M.; Maunula, P. Ship-of-opportunity based phycocyanin fluorescence monitoring of the filamentous cyanobacteria bloom dynamics in the Baltic Sea. Estuar. Coast. Shelf Sci. 2007, 73, 489–500. [Google Scholar] [CrossRef]
- Michalak, A.M.; Anderson, E.J.; Beletsky, D.; Boland, S.; Bosch, N.S.; Bridgeman, T.B.; Chaffin, J.D.; Cho, K.; Confesor, R.; Daloglu, I.; et al. Record-setting algal bloom in Lake Erie caused by agricultural and meteorological trends consistent with expected future conditions. Proc. Natl. Acad. Sci. USA 2013, 110, 6448–6452. [Google Scholar] [CrossRef]
- Lunetta, R.S.; Schaeffer, B.A.; Stumpf, R.P.; Keith, D.; Jacobs, S.A.; Murphy, M.S. Evaluation of cyanobacteria cell count detection derived from MERIS imagery across the eastern USA. Remote Sens. Environ. 2015, 157, 24–34. [Google Scholar] [CrossRef]
- Simis, S.G.H.; Peters, S.W.M.; Gons, H.J. Optical changes associated with cyanobacterial bloom termination by viral lysis. J. Plankton Res. 2005, 27, 937–949. [Google Scholar] [CrossRef]
- Matthews, M.W.; Odermatt, D. Improved algorithm for routine monitoring of cyanobacteria and eutrophication in inland and near-coastal waters. Remote Sens. Environ. 2015, 156, 374–382. [Google Scholar] [CrossRef]
- Wynne, T.T.; Meredith, A.; Briggs, T.; Litaker, W.; Stumpf, R.P. Harmful Algal Bloom Forecasting Branch Ocean Color Satellite Imagery Processing Guidelines; NOAA Technical Memorandum NOS NCCOS; NOAA: Washington, DA, USA, 2018; p. 296. [Google Scholar] [CrossRef]
- Stumpf, R.P.; Johnson, L.T.; Wynne, T.T.; Baker, D.B. Forecasting annual cyanobacterial bloom biomass to inform management decisions in Lake Erie. J. Great Lakes Res. 2016, 42, 1174–1183. [Google Scholar] [CrossRef]
- Hunter, P.; Tyler, A.; Willby, N.; Gilvear, D. The spatial dynamics of vertical migration by Microcystis aeruginosa in a eutrophic shallow lake: A case study using high spatial resolution time-series airborne remote sensing. Limnol. Oceanogr. 2008, 53, 2391–2406. [Google Scholar] [CrossRef]
- Medrano, E.A.; Uittenbogaard, R.; Pires, L.D.; Van De Wiel, B.; Clercx, H. Coupling hydrodynamics and buoyancy regulation in Microcystis aeruginosa for its vertical distribution in lakes. Ecol. Model. 2013, 248, 41–56. [Google Scholar] [CrossRef]
- Brookes, J.D.; Ganf, G.G.; Green, D.; Whittington, J. The influence of light and nutrients on buoyancy, filament aggregation and flotation of Anabaena circinalis. J. Plankton Res. 1999, 21, 327–341. [Google Scholar] [CrossRef]
- Wynne, T.T.; Stumpf, R.P.; Briggs, T.O. Comparing MODIS and MERIS spectral shapes for cyanobacterial bloom detection. Int. J. Remote Sens. 2013, 34, 6668–6678. [Google Scholar] [CrossRef]
- Pope, R.M.; Fry, E.S. Absorption spectrum (380–700 nm) of pure water. II. Integrating cavity measurements. Appl. Opt. 1997, 36, 8710–8723. [Google Scholar] [CrossRef]
- Fahnenstiel, G.; Millie, D.; Dyble, J.; Litaker, R.; Tester, P.; McCormick, M.; Rediske, R.; Klarer, D. Microcystin concentrations and cell quotas in Saginaw Bay, Lake Huron. Aquat. Ecosyst. Health Manag. 2008, 11, 190–195. [Google Scholar] [CrossRef]
- Wilson, A.E.; Wilson, W.A.; Hay, M.E. Intraspecific variation in growth and morphology of the bloom-forming cyanobacterium Microcystis aeruginosa. Appl. Environ. Microbiol. 2006, 72, 7386–7389. [Google Scholar] [CrossRef]
- De Stasio, B.T.; Beranek, A.E.; Schrimpf, M.B. Zooplankton-phytoplankton interactions in Green Bay, Lake Michigan: Lower food web responses to biological invasions. J. Great Lakes Res. 2018, 44, 910–923. [Google Scholar] [CrossRef]
- Wetzel, R.G.; Likens, G.E. Inorganic nutrients: Nitrogen, phosphorus, and other nutrients. In Limnological Analyses; Springer: Berlin/Heidelberg, Germany, 1991; pp. 81–105. [Google Scholar]
- Kutser, T. Passive optical remote sensing of cyanobacteria and other intense phytoplankton blooms in coastal and inland waters. Int. J. Remote Sens. 2009, 30, 4401–4425. [Google Scholar] [CrossRef]
- Hawkins, P.R.; Holliday, J.; Kathuria, A.; Bowling, L. Change in cyanobacterial biovolume due to preservation by Lugol’s Iodine. Harmful Algae 2005, 4, 1033–1043. [Google Scholar] [CrossRef]
- NASA. Giovanni. Available online: https://giovanni.gsfc.nasa.gov/giovanni/ (accessed on 12 July 2022).
- Dolan, D.M.; Yui, A.K.; Geist, R.D. Evaluation of river load estimation methods for total phosphorus. J. Great Lakes Res. 1981, 7, 207–214. [Google Scholar] [CrossRef]
- Baker, D.; Confesor, R.; Ewing, D.; Johnson, L.; Kramer, J.; Merryfield, B. Phosphorus loading to Lake Erie from the Maumee, Sandusky and Cuyahoga rivers: The importance of bioavailability. J. Great Lakes Res. 2014, 40, 502–517. [Google Scholar] [CrossRef]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Huisman, J. Blooms like it hot. Science 2008, 320, 57–58. [Google Scholar] [CrossRef]
- Huisman, J.; Sharples, J.; Stroom, J.M.; Visser, P.M.; Kardinaal, W.E.A.; Verspagen, J.M.; Sommeijer, B. Changes in turbulent mixing shift competition for light between phytoplankton species. Ecology 2004, 85, 2960–2970. [Google Scholar] [CrossRef]
- Liu, M.; Ma, J.; Kang, L.; Wei, Y.; He, Q.; Hu, X.; Li, H. Strong turbulence benefits toxic and colonial cyanobacteria in water: A potential way of climate change impact on the expansion of Harmful Algal Blooms. Sci. Total Environ. 2019, 670, 613–622. [Google Scholar] [CrossRef]
- Neilan, B.A.; Pearson, L.A.; Muenchhoff, J.; Moffitt, M.C.; Dittmann, E. Environmental conditions that influence toxin biosynthesis in cyanobacteria. Environ. Microbiol. 2013, 15, 1239–1253. [Google Scholar] [CrossRef]
- Blough, N. Photochemical processes. In Encyclopedia of Ocean Sciences; Steele, J., Thorpe, S., Turekian, K., Eds.; Academic Press: London, UK, 2001; pp. 2162–2172. [Google Scholar]
- Lee, Z.; Carder, K.; Peacock, T.; Davis, C.; Mueller, J. Method to derive ocean absorption coefficients from remote-sensing reflectance. Appl. Opt. 1996, 35, 453–462. [Google Scholar] [CrossRef]
- Kirk, J. Yellow substance (gelbstoff) and its contribution to the attenuation of photosynthetically active radiation in some inland and coastal south-eastern Australian waters. Mar. Freshw. Res. 1976, 27, 61–71. [Google Scholar] [CrossRef]
- Kuczynska, P.; Jemiola-Rzeminska, M.; Strzalka, K. Photosynthetic pigments in diatoms. Mar. Drugs 2015, 13, 5847–5881. [Google Scholar] [CrossRef]
- Yu, L. Sea surface exchanges of momentum, heat, and freshwater determined by satellite remote sensing. Encycl. Ocean Sci. 2009, 2, 202–211. [Google Scholar]
- Jones, I.; George, G.; Reynolds, C. Quantifying effects of phytoplankton on the heat budgets of two large limnetic enclosures. Freshw. Biol. 2005, 50, 1239–1247. [Google Scholar] [CrossRef]
- Serra, T.; Vidal, J.; Casamitjana, X.; Soler, M.; Colomer, J. The role of surface vertical mixing in phytoplankton distribution in a stratified reservoir. Limnol. Oceanogr. 2007, 52, 620–634. [Google Scholar] [CrossRef]
- Tsydenov, B.O. Simulating phytoplankton growth during the spring thermal bar in a deep lake. J. Mar. Syst. 2019, 195, 38–49. [Google Scholar] [CrossRef]
- Tsydenov, B. Effects of Heat Fluxes on the Phytoplankton Distribution in a Freshwater Lake. Atmos. Ocean. Opt. 2021, 34, 603–610. [Google Scholar] [CrossRef]
- Dokulil, M.T.; Teubner, K. Cyanobacterial dominance in lakes. Hydrobiologia 2000, 438, 1–12. [Google Scholar] [CrossRef]
- PCA-Biplot. Available online: http://agroninfotech.blogspot.com/2020/06/biplot-for-principal-component-analysis.html (accessed on 1 January 2022).
- R-Blogger. Available online: https://www.r-bloggers.com/2013/11/computing-and-visualizing-pca-in-r/ (accessed on 1 January 2022).
- Stow, C.A.; Dyble, J.; Kashian, D.R.; Johengen, T.H.; Winslow, K.P.; Peacor, S.D.; Francoeur, S.N.; Burtner, A.M.; Palladino, D.; Morehead, N. Phosphorus targets and eutrophication objectives in Saginaw Bay: A 35 year assessment. J. Great Lakes Res. 2014, 40, 4–10. [Google Scholar] [CrossRef]
- Lanerolle, L.W.J.; Stumpf, R.P.; Wynne, T.T.; Patchen, R.C. A One-Dimensional Numerical Vertical Mixing Model with Application to Western Lake Erie; NOAA Technical Memorandum: Silver Spring, MD, USA, 2011. [Google Scholar]
- LaBuhn, S.; Klump, J.V. Estimating summertime epilimnetic primary production via in situ monitoring in an eutrophic freshwater embayment, Green Bay, Lake Michigan. J. Great Lakes Res. 2016, 42, 1026–1035. [Google Scholar] [CrossRef]
- Fahnenstiel, G.L.; Bridgeman, T.B.; Lang, G.A.; McCormick, M.J.; Nalepa, T.F. Phytoplankton productivity in Saginaw Bay, Lake Huron: Effects of zebra mussel (Dreissena polymorpha) colonization. J. Great Lakes Res. 1995, 21, 464–475. [Google Scholar] [CrossRef]
- Davis, T.W.; Koch, F.; Marcoval, M.A.; Wilhelm, S.W.; Gobler, C.J. Mesozooplankton and microzooplankton grazing during cyanobacterial blooms in the western basin of Lake Erie. Harmful Algae 2012, 15, 26–35. [Google Scholar] [CrossRef]
- Kahru, M.; Leppanen, J.-M.; Rud, O. Cyanobacterial blooms cause heating of the sea surface. Mar. Ecol. Prog. Ser. 1993, 101, 1–7. [Google Scholar] [CrossRef]
- Houser, J.N. Water color affects the stratification, surface temperature, heat content, and mean epilimnetic irradiance of small lakes. Can. J. Fish. Aquat. Sci. 2006, 63, 2447–2455. [Google Scholar] [CrossRef]
- Bowling, L. Heat contents, thermal stabilities and Birgean wind work in dystrophic Tasmanian lakes and reservoirs. Mar. Freshw. Res. 1990, 41, 429–441. [Google Scholar] [CrossRef]
- Margalef, R. Life-forms of phytoplankton as survival alternatives in an unstable environment. Oceanol. Acta 1978, 1, 493–509. [Google Scholar]
- Margalef, R. The food web in the pelagic environment. Helgol. Wiss. Meeresunters. 1967, 15, 548–559. [Google Scholar] [CrossRef] [Green Version]
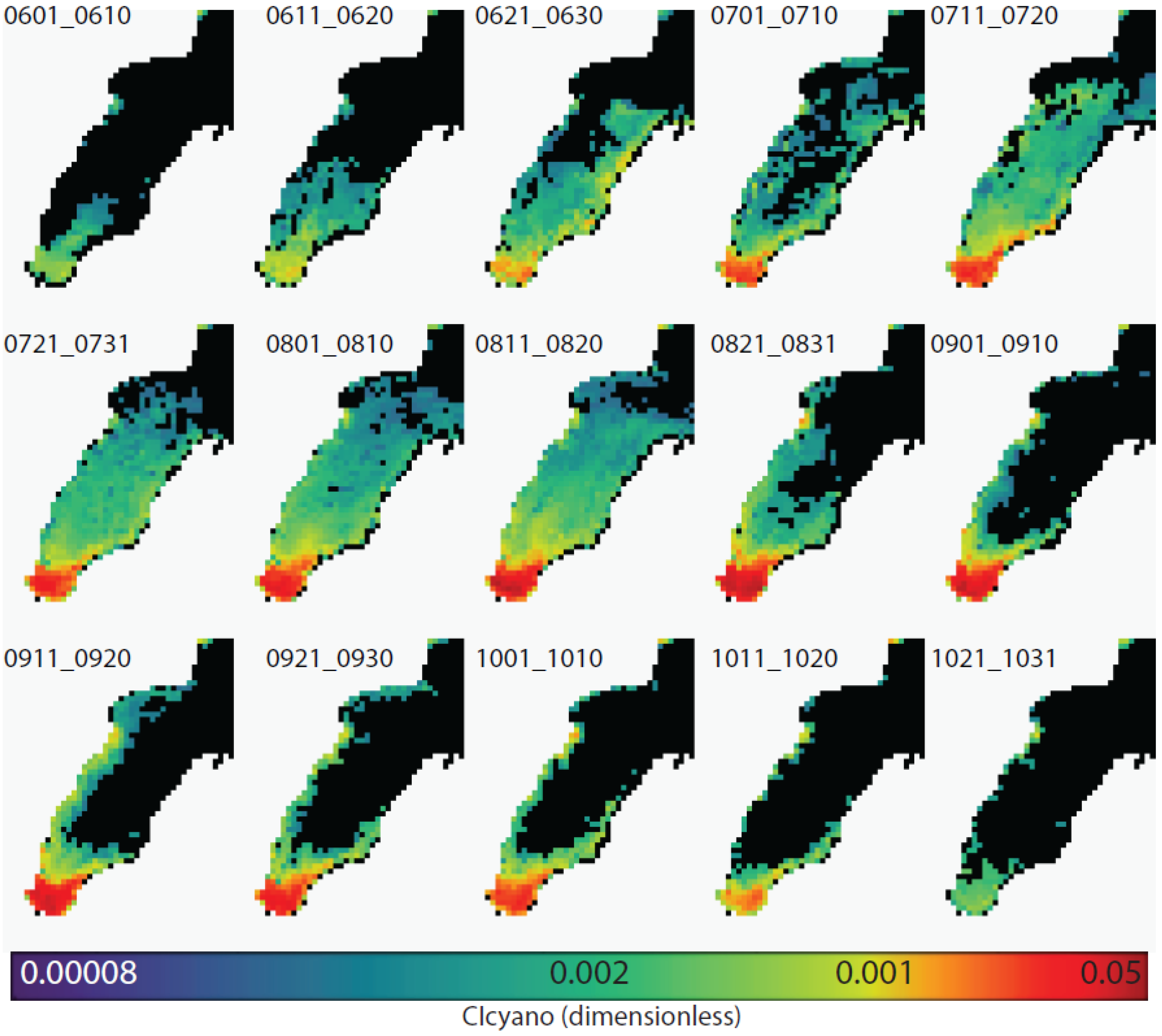
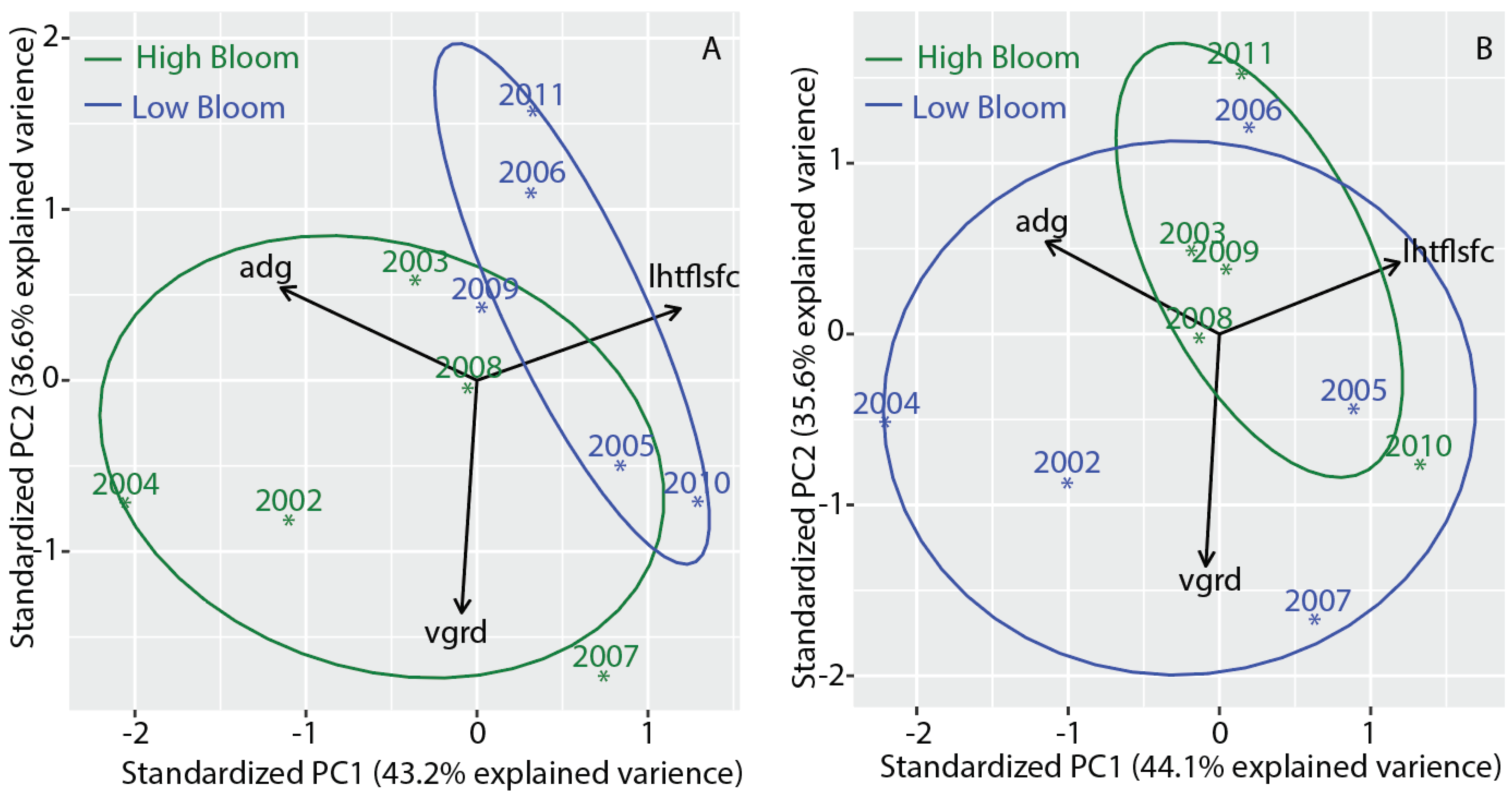

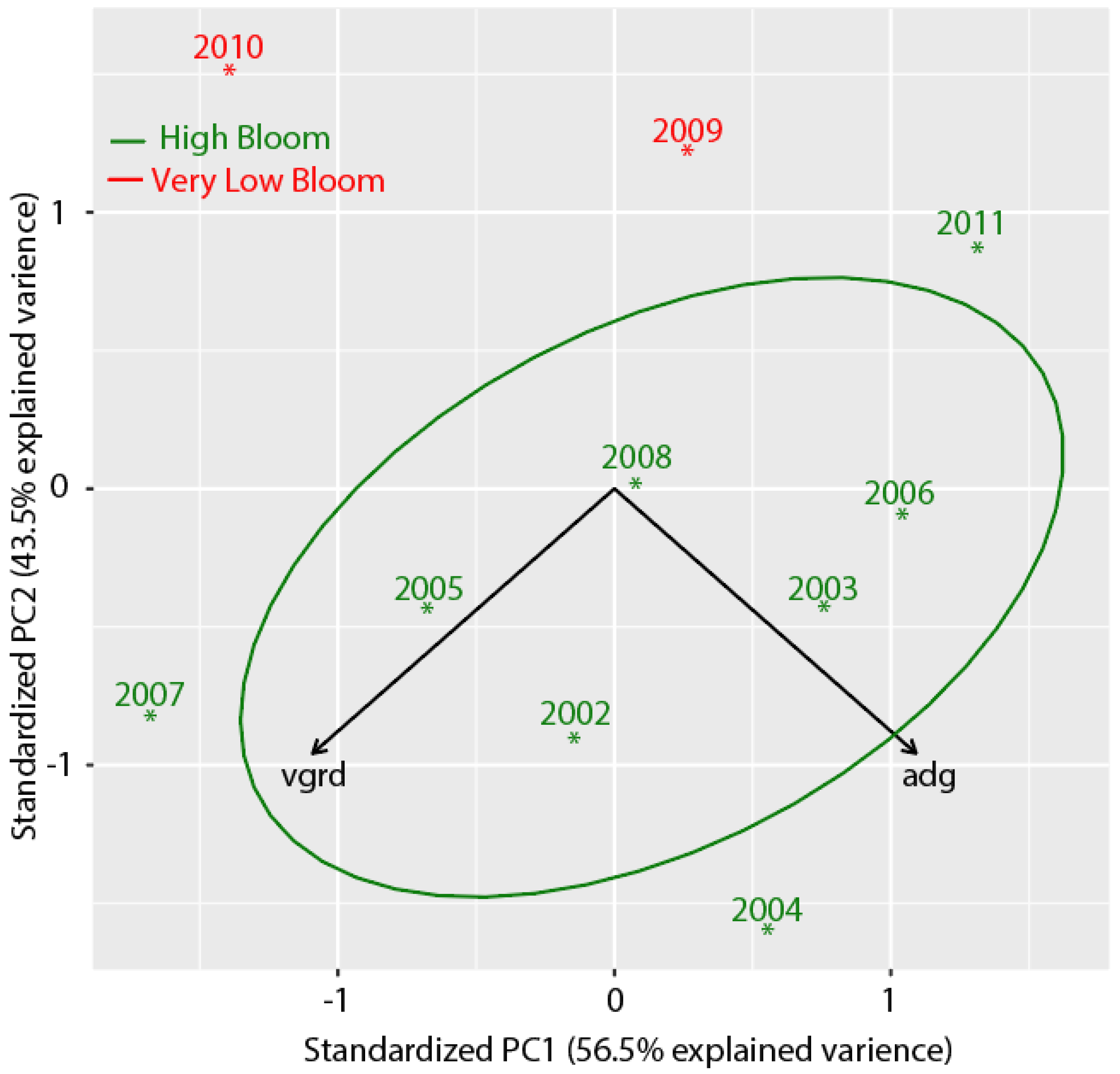
| Composite Number | Start Date | End Date | Mean Date |
|---|---|---|---|
| 1 | June 1 | June 10 | June 5 |
| 2 | June 11 | June 20 | June 15 |
| 3 | June 21 | June 30 | June 20 |
| 4 | July 1 | July 10 | July 5 |
| 5 | July 11 | July 20 | July 15 |
| 6 | July 21 | July 31 | July 25 |
| 7 | August 1 | August 10 | August 5 |
| 8 | August 11 | August 20 | August 15 |
| 9 | August 21 | August 31 | August 25 |
| 10 | September 1 | September 10 | September 5 |
| 11 | September 11 | September 20 | September 15 |
| 12 | September 21 | September 30 | September 25 |
| 13 | October 1 | October 10 | October 5 |
| 14 | October 11 | October 20 | October 15 |
| 15 | October 21 | October 31 | October 25 |
| Year | Green Bay | Saginaw Bay | Western Lake Erie |
|---|---|---|---|
| Mean ± SD | 6.7 ± 2.8 (July 21–31) | 9 ± 2.4 (August 11–20) | 10.3 ± 1.2 (September 1–10) |
| Median | 6.7 (August 1–10) | 9 (August 21–31) | 10 (September 1–10) |
| Mode | 8 (June 21 –30) | 12 (August 21–31) | 10 (September 1–10) |
| Year | Green Bay | Saginaw Bay | Western Lake Erie |
|---|---|---|---|
| Mean ± SD | 5.7 ± 2.2 (July 11–20) | 4 ± 1.5 (July 1–10) | 7.3 ± 2.1 (August 1–10) |
| Median | 5.7 (July 11–20) | 4 (July 1–10) | 8 (August 11–20) |
| Mode | 5 (July 11 –20) | 4 (July 1–10) | 8 (August 11–20) |
| Region | Max Annual CI | Year | Min Annual CI | Year | Variability (Max Annual CI/Min Annual CI) |
|---|---|---|---|---|---|
| Green Bay | 3.27 | 2004 | 0.12 | 2010 | 27.25 |
| Western Lake Erie | 37.9 | 2011 | 0.5 | 2002 | 75.8 |
| Saginaw Bay | 9.2 | 2008 | 2.2 | 2003 | 4.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wynne, T.T.; Stumpf, R.P.; Pokrzywinski, K.L.; Litaker, R.W.; De Stasio, B.T.; Hood, R.R. Cyanobacterial Bloom Phenology in Green Bay Using MERIS Satellite Data and Comparisons with Western Lake Erie and Saginaw Bay. Water 2022, 14, 2636. https://doi.org/10.3390/w14172636
Wynne TT, Stumpf RP, Pokrzywinski KL, Litaker RW, De Stasio BT, Hood RR. Cyanobacterial Bloom Phenology in Green Bay Using MERIS Satellite Data and Comparisons with Western Lake Erie and Saginaw Bay. Water. 2022; 14(17):2636. https://doi.org/10.3390/w14172636
Chicago/Turabian StyleWynne, Timothy T., Richard P. Stumpf, Kaytee L. Pokrzywinski, R. Wayne Litaker, Bart T. De Stasio, and Raleigh R. Hood. 2022. "Cyanobacterial Bloom Phenology in Green Bay Using MERIS Satellite Data and Comparisons with Western Lake Erie and Saginaw Bay" Water 14, no. 17: 2636. https://doi.org/10.3390/w14172636






