Application of Central Composite Design for Optimization of Adsorption of Chromium(VI) by Spirulina platensis Algae Biomass
Abstract
:1. Introduction
2. Materials and Methods
2.1. Algae Biomass
2.2. Chromium Solution
2.3. Batch Adsorption Tests
2.4. Determination of Cr
2.5. Central Composite Desing
3. Results
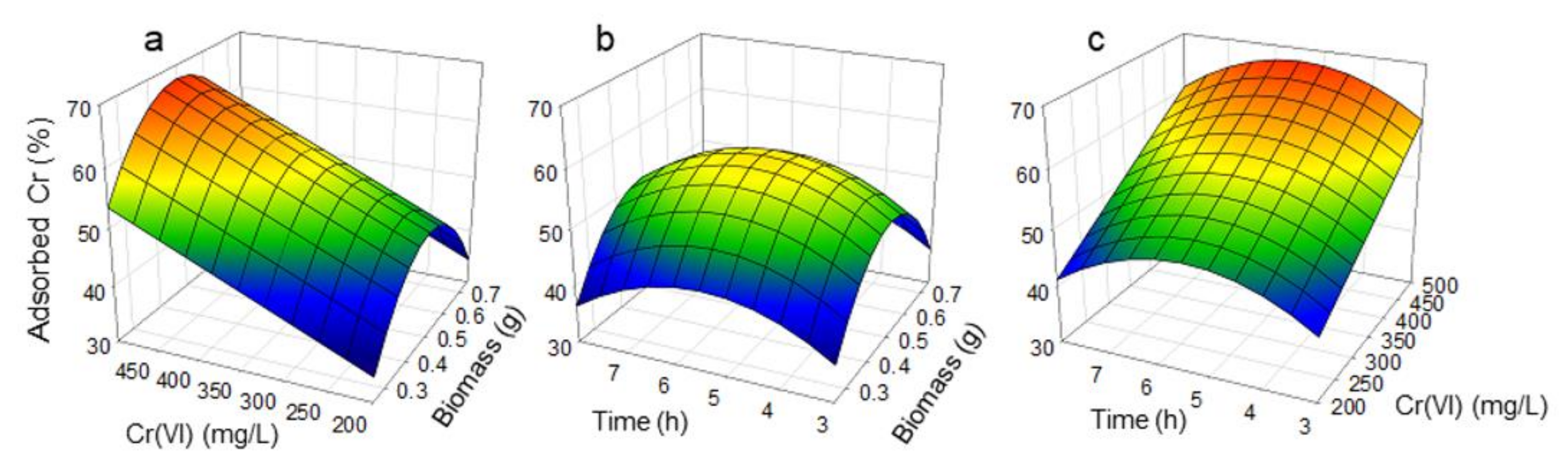
3.1. Percentage of Adsorbed Chromium
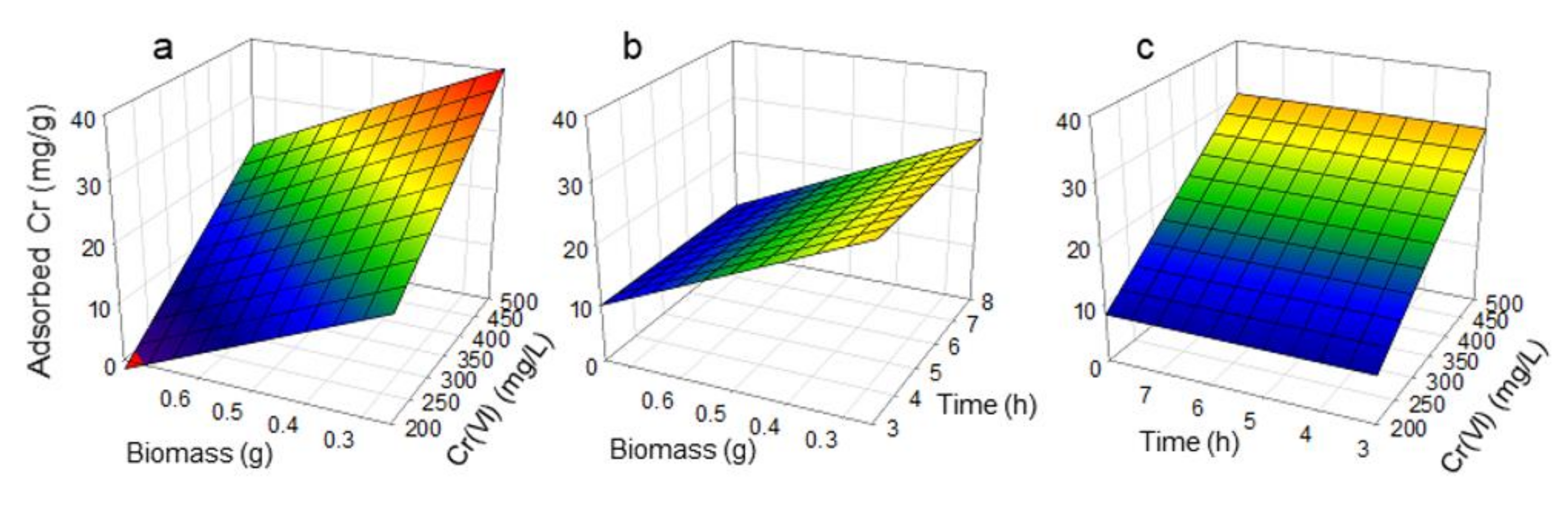
3.2. Sorption Capacity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Costa, M.; Klein, C. Toxicity and carcinogenicity of chromium compounds in humans. Crit. Rev. Toxicol. 2006, 36, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Barnhart, J. Occurrences, uses, and properties of chromium. Regul. Toxicol. Pharmacol. 1997, 26, S3–S7. [Google Scholar] [CrossRef]
- Caussy, D.; Gochfeld, M.; Gurzau, E.; Neagu, C.; Ruedel, H. Lessons from case studies of metals: Investigating exposure, bioavailability, and risk. Ecotoxicol. Environ. Saf. 2003, 56, 45–51. [Google Scholar] [CrossRef]
- Botsou, F.; Koutsopoulou, E.; Andrioti, A.; Dassenakis, M.; Scoullos, M.; Karageorgis, A.P. Chromium speciation, mobility, and Cr (VI) retention–release processes in ultramafic rocks and Fe–Ni lateritic deposits of Greece. Environ. Geochem. Health 2022, 44, 2815–2834. [Google Scholar] [CrossRef] [PubMed]
- Owlad, M.; Aroua, M.K.; Daud, W.A.W.; Baroutian, S. Removal of hexavalent chromium-contaminated water and wastewater: A review. Water Air Soil Pollut. 2009, 200, 59–77. [Google Scholar] [CrossRef]
- Peng, H.; Guo, J. Removal of chromium from wastewater by membrane filtration, chemical precipitation, ion exchange, adsorption electrocoagulation, electrochemical reduction, electrodialysis, electrodeionization, photocatalysis and nanotechnology: A review. Environ. Chem. Lett. 2020, 18, 2055–2068. [Google Scholar] [CrossRef]
- Zhao, J.; Boada, R.; Cibin, G.; Palet, C. Enhancement of selective adsorption of Cr species via modification of pine biomass. Sci. Total Environ. 2021, 756, 143816. [Google Scholar] [CrossRef]
- Gasemloo, S.; Khosravi, M.; Sohrabi, M.R.; Dastmalchi, S.; Gharbani, P. Response surface methodology (RSM) modeling to improve removal of Cr (VI) ions from tannery wastewater using sulfated carboxymethyl cellulose nanofilter. J. Clean. Prod. 2019, 208, 736–742. [Google Scholar] [CrossRef]
- Khan, M.I.; Su, J.; Guo, L. Development of triethanolamine functionalized-anion exchange membrane for adsorptive removal of methyl orange from aqueous solution. Desal. Water Treat 2021, 209, 342–352. [Google Scholar] [CrossRef]
- Zhao, Y.; Kang, D.; Chen, Z.; Zhan, J.; Wu, X. Removal of chromium using electrochemical approaches: A. Int. J. Electrochem. Sci. 2018, 13, 1250–1259. [Google Scholar] [CrossRef]
- El-Kammah, M.; Elkhatib, E.; Gouveia, S.; Cameselle, C.; Aboukila, E. Cost-effective ecofriendly nanoparticles for rapid and efficient indigo carmine dye removal from wastewater: Adsorption equilibrium, kinetics and mechanism. Environ. Technol. Innov. 2022, 28, 102595. [Google Scholar] [CrossRef]
- El-Kammah, M.; Elkhatib, E.; Gouveia, S.; Cameselle, C.; Aboukila, E. Enhanced removal of Thiamethoxam from wastewater using waste-derived nanoparticles: Adsorption performance and mechanisms. Environ. Technol. Innov. 2022, 28, 102713. [Google Scholar] [CrossRef]
- El-Kammah, M.; Elkhatib, E.; Gouveia, S.; Cameselle, C.; Aboukila, E. Enhanced removal of Indigo Carmine dye from textile effluent using green cost-efficient nanomaterial: Adsorption, kinetics, thermodynamics and mechanisms. Sustain. Chem. Pharm. 2022, 29, 100753. [Google Scholar] [CrossRef]
- Saravanan, A.; Kumar, P.S.; Yashwanthraj, M. Sequestration of toxic Cr (VI) ions from industrial wastewater using waste biomass: A review. Desal. Water Treat 2017, 68, 245–266. [Google Scholar]
- Ugwu, E.I.; Agunwamba, J.C. A review on the applicability of activated carbon derived from plant biomass in adsorption of chromium, copper, and zinc from industrial wastewater. Environ. Monit. Assess. 2020, 192, 1–12. [Google Scholar] [CrossRef]
- Abbas, M.; Nadeem, R.; Zafar, M.N.; Arshad, M. Biosorption of chromium (III) and chromium (VI) by untreated and pretreated Cassia fistula biomass from aqueous solutions. Water Air Soil Pollut. 2008, 191, 139–148. [Google Scholar] [CrossRef]
- Znad, H.; Awual, M.R.; Martini, S. The utilization of algae and seaweed biomass for bioremediation of heavy metal-contaminated wastewater. Molecules 2022, 27, 1275. [Google Scholar] [CrossRef]
- Spennati, E.; Mirizadeh, S.; Casazza, A.A.; Solisio, C.; Converti, A. Chlorella vulgaris and Arthrospira platensis growth in a continuous membrane photobioreactor using industrial winery wastewater. Algal Res. 2021, 60, 102519. [Google Scholar] [CrossRef]
- Almomani, F.; Bhosale, R.R. Bio-sorption of toxic metals from industrial wastewater by algae strains Spirulina platensis and Chlorella vulgaris: Application of isotherm, kinetic models and process optimization. Sci. Total Environ. 2021, 755, 142654. [Google Scholar] [CrossRef]
- De Oliveira, M.; Monteiro, M.; Robbs, P.; Leite, S. Growth and chemical composition of Spirulina maxima and Spirulina platensis biomass at different temperatures. Aquac. Int. 1999, 7, 261–275. [Google Scholar] [CrossRef]
- Wuang, S.C.; Khin, M.C.; Chua, P.Q.D.; Luo, Y.D. Use of Spirulina biomass produced from treatment of aquaculture wastewater as agricultural fertilizers. Algal Res. 2016, 15, 59–64. [Google Scholar] [CrossRef]
- Baird, R.B.; Rice, C.E.W.; Eaton, A.D. Standard Methods for the Examination of Water and Wastewater, 23rd ed; Water Environment Federation: Alexandria, VA, USA; American Public Health Association: Washington, DC, USA; American Water Works Association: Denver, CO, USA, 2017; ISBN 9780875532875. [Google Scholar]
- Leardi, R. Experimental design in chemistry: A tutorial. Anal. Chim. Acta 2009, 652, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, H. Biosorption of chromium by using Spirulina sp. Arab. J. Chem. 2016, 9, 846–853. [Google Scholar] [CrossRef]
- Nithya, K.; Sathish, A.; Pradeep, K.; Baalaji, S.K. Algal biomass waste residues of Spirulina platensis for chromium adsorption and modeling studies. J. Environ. Chem. Eng. 2019, 7, 103273. [Google Scholar] [CrossRef]
- Finocchio, E.; Lodi, A.; Solisio, C.; Converti, A. Chromium (VI) removal by methylated biomass of Spirulina platensis: The effect of methylation process. Chem. Eng. J. 2010, 156, 264–269. [Google Scholar] [CrossRef]
- Gokhale, S.V.; Jyoti, K.K.; Lele, S.S. Kinetic and equilibrium modeling of chromium (VI) biosorption on fresh and spent Spirulina platensis/Chlorella vulgaris biomass. Bioresour. Technol. 2008, 99, 3600–3608. [Google Scholar] [CrossRef]
- Lodi, A.; Soletto, D.; Solisio, C.; Converti, A. Chromium (III) removal by Spirulina platensis biomass. Chem. Eng. J. 2008, 136, 151–155. [Google Scholar] [CrossRef]
- Balaji, S.; Kalaivani, T.; Rajasekaran, C.; Shalini, M.; Siva, R.; Singh, R.K.; Akthar, M.A. Arthrospira (Spirulina) species as bioadsorbents for lead, chromium, and cadmium–a comparative study. CLEAN–Soil Air Water 2014, 42, 1790–1797. [Google Scholar] [CrossRef]
- Jobby, R.; Jha, P.; Yadav, A.K.; Desai, N. Biosorption and biotransformation of hexavalent chromium [Cr (VI)]: A comprehensive review. Chemosphere 2018, 207, 255–266. [Google Scholar] [CrossRef]
- Fernández, P.M.; Viñarta, S.C.; Bernal, A.R.; Cruz, E.L.; Figueroa, L.I. Bioremediation strategies for chromium removal: Current research, scale-up approach and future perspectives. Chemosphere 2018, 208, 139–148. [Google Scholar] [CrossRef]
| Component | Composition |
|---|---|
| Phycocyanin | 140 g/kg |
| Chlorophyll | 6.1–10 g/kg |
| Carotenoid | 3.7 g/kg |
| β-carotene | 1.5–1.9 g/kg |
| Protein | 55–70% |
| Moisture | 4–7% |
| Ashes * | 6–13% |
| Variable | Factor | Factor Level and Variable Value | Units | ||||
|---|---|---|---|---|---|---|---|
| −α | −1 | 0 | 1 | +α | |||
| Algal biomass | A | 0.0795 | 0.25 | 0.5 | 0.75 | 0.9205 | g |
| Cr(VI) | B | 97.7 | 200 | 350 | 500 | 602.3 | mg/L |
| Time | C | 1.295 | 3 | 5.5 | 8 | 9.705 | h |
| Test | Factor A | Factor B | Factor C | Biomass | Cr(VI) | Time | pH | EC * |
|---|---|---|---|---|---|---|---|---|
| (g) | (mg/L) | (h) | (mS/cm) | |||||
| 1 | −1 | −1 | −1 | 0.25 | 200 | 3 | 5.94 | 0.96 |
| 2 | 1 | −1 | −1 | 0.75 | 200 | 3 | 6.88 | 1.7 |
| 3 | −1 | 1 | −1 | 0.25 | 500 | 3 | 5.71 | 1.64 |
| 4 | 1 | 1 | −1 | 0.75 | 500 | 3 | 6.41 | 2.35 |
| 5 | −1 | −1 | 1 | 0.25 | 200 | 8 | 6.31 | 1 |
| 6 | 1 | −1 | 1 | 0.75 | 200 | 8 | 6.06 | 0.97 |
| 7 | −1 | 1 | 1 | 0.25 | 500 | 8 | 5.34 | 1.39 |
| 8 | 1 | 1 | 1 | 0.75 | 500 | 8 | 5.61 | 1.55 |
| 9 | −1.682 | 0 | 0 | 0.0795 | 350 | 5.5 | 5.57 | 0.98 |
| 10 | 1.682 | 0 | 0 | 0.9205 | 350 | 5.5 | 5.67 | 1.03 |
| 11 | 0 | −1.682 | 0 | 0.5 | 97.7 | 5.5 | 6.74 | 1.05 |
| 12 | 0 | 1.682 | 0 | 0.5 | 602.3 | 5.5 | 5.81 | 1.59 |
| 13 | 0 | 0 | −1.682 | 0.5 | 350 | 1.295 | 6.21 | 1.33 |
| 14 | 0 | 0 | 1.682 | 0.5 | 350 | 9.705 | 6.08 | 1.5 |
| 15 | 0 | 0 | 0 | 0.5 | 350 | 5.5 | 5.89 | 1.16 |
| 16 | 0 | 0 | 0 | 0.5 | 350 | 5.5 | 5.89 | 1.16 |
| 17 | 0 | 0 | 0 | 0.5 | 350 | 5.5 | 5.89 | 1.16 |
| 18 | 0 | 0 | 0 | 0.5 | 350 | 5.5 | 5.89 | 1.16 |
| 19 | 0 | 0 | 0 | 0.5 | 350 | 5.5 | 5.89 | 1.16 |
| 20 | 0 | 0 | 0 | 0.5 | 350 | 5.5 | 5.89 | 1.16 |
| Test | Factor A | Factor B | Factor C | Residual Cr (mg/L) | Adsorbed Cr (%) | Adsorbed Cr q (mg/g)) |
|---|---|---|---|---|---|---|
| 1 | −1 | −1 | −1 | 175.70 | 12.15 | 17.8 |
| 2 | 1 | −1 | −1 | 152.74 | 23.63 | 9.6 |
| 3 | −1 | 1 | −1 | 285.15 | 42.97 | 40.4 |
| 4 | 1 | 1 | −1 | 228.75 | 54.25 | 21.0 |
| 5 | −1 | −1 | 1 | 130.58 | 34.71 | 17.8 |
| 6 | 1 | −1 | 1 | 128.02 | 35.99 | 10.6 |
| 7 | −1 | 1 | 1 | 288.35 | 42.33 | 40.4 |
| 8 | 1 | 1 | 1 | 282.95 | 43.41 | 21.0 |
| 9 | −1.682 | 0 | 0 | 306.11 | 12.54 | 35.7 |
| 10 | 1.682 | 0 | 0 | 269.14 | 23.10 | 3.1 |
| 11 | 0 | −1.682 | 0 | 57.36 | 41.29 | 2.4 |
| 12 | 0 | 1.682 | 0 | 159.90 | 73.45 | 38.4 |
| 13 | 0 | 0 | −1.682 | 237.35 | 32.19 | 19.4 |
| 14 | 0 | 0 | 1.682 | 202.85 | 42.04 | 20.3 |
| 15 | 0 | 0 | 0 | 146.23 | 58.22 | 18.6 |
| 16 | 0 | 0 | 0 | 147.01 | 57.12 | 18.3 |
| 17 | 0 | 0 | 0 | 149.54 | 55.46 | 19.5 |
| 18 | 0 | 0 | 0 | 144.67 | 59.34 | 20.8 |
| 19 | 0 | 0 | 0 | 145.23 | 59.02 | 19.7 |
| 20 | 0 | 0 | 0 | 146.88 | 58.55 | 19.5 |
| Source | Sum of Squares | Degrees of Freedom | Mean Square | F Value | p-Value Prob > F | |
|---|---|---|---|---|---|---|
| Model | 5333.44 | 9 | 592.60 | 2.88 | 0.0575 | significant |
| A-Biomass | 134.7 | 1 | 134.70 | 0.65 | 0.4374 | |
| B-Cr(VI) | 1248.99 | 1 | 1248.99 | 6.07 | 0.0335 | |
| C-Time | 117.31 | 1 | 117.31 | 0.57 | 0.4677 | |
| AB | 0.020 | 1 | 0.020 | 9.717 × 105 | 0.9923 | |
| AC | 52.02 | 1 | 52.02 | 0.25 | 0.6260 | |
| BC | 269.12 | 1 | 269.12 | 1.31 | 0.2795 | |
| A2 | 2939.57 | 1 | 2939.57 | 14.28 | 0.0036 | |
| B2 | 1.29 | 1 | 1.29 | 6.259 × 10−3 | 0.9385 | |
| C2 | 801.68 | 1 | 801.68 | 3.89 | 0.0767 | |
| Residual | 2058.32 | 10 | 205.83 | |||
| Lack of Fit | 2058.32 | 5 | 411.86 | |||
| Pure Error | 0.000 | 5 | 0.000 | |||
| Cor Total | 7391.76 | 19 |
| Source | Sum of Squares | Degrees of Freedom | Mean Square | F Value | p-Value Prob > F | |
|---|---|---|---|---|---|---|
| Model | 3034.25 | 3 | 1011.42 | 11.60 | 0.0003 | significant |
| A-Biomass | 1284.43 | 1 | 1284.43 | 14.73 | 0.0015 | |
| B-Cr(VI) | 1746.29 | 1 | 1746.29 | 20.03 | 0.0004 | |
| C-Time | 3.53 | 1 | 3.53 | 0.040 | 0.8431 | |
| Residual | 1394.85 | 16 | 87.18 | |||
| Lack of Fit | 1394.85 | 11 | 126.80 | |||
| Pure Error | 0.000 | 5 | 0.000 | |||
| Cor Total | 4429.10 | 19 |
| Biomass | Contaminant | Adsorption Conditions | Adsorption Capacity (q) | Ref. |
|---|---|---|---|---|
| Spirulina sp. Raw biomass | Cr(VI) | pH 5 1 h | 90.91 mg/g | [24] |
| Spirulina platensis. Waste from the biodiesel extraction | Cr(VI) | pH 1 60 °C. 2 h | 45.5–60 mg/g | [25] |
| Spirulina platensis. Methylated biomass | Cr(VI) | pH 7–8 | 7.4–16.7 mg/g | [26] |
| Spirulina platensis. Fresh biomass | Cr(VI) | pH 1.5 25 °C | 188 mg/g | [27] |
| Spirulina platensis. Spent biomass after the extraction of β-carotene | Cr(VI) | pH 1.5 25 °C | 212 mg/g | [28] |
| Spirulina platensis. Dry biomass | Cr(III) | pH 6 20 °C | 30–36 mg/g | [29] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urréjola-Madriñán, S.; Paz-Armada, I.; Cameselle, C.; Gouveia, S. Application of Central Composite Design for Optimization of Adsorption of Chromium(VI) by Spirulina platensis Algae Biomass. Water 2022, 14, 2539. https://doi.org/10.3390/w14162539
Urréjola-Madriñán S, Paz-Armada I, Cameselle C, Gouveia S. Application of Central Composite Design for Optimization of Adsorption of Chromium(VI) by Spirulina platensis Algae Biomass. Water. 2022; 14(16):2539. https://doi.org/10.3390/w14162539
Chicago/Turabian StyleUrréjola-Madriñán, Santiago, Iñaki Paz-Armada, Claudio Cameselle, and Susana Gouveia. 2022. "Application of Central Composite Design for Optimization of Adsorption of Chromium(VI) by Spirulina platensis Algae Biomass" Water 14, no. 16: 2539. https://doi.org/10.3390/w14162539
APA StyleUrréjola-Madriñán, S., Paz-Armada, I., Cameselle, C., & Gouveia, S. (2022). Application of Central Composite Design for Optimization of Adsorption of Chromium(VI) by Spirulina platensis Algae Biomass. Water, 14(16), 2539. https://doi.org/10.3390/w14162539






