Arsenite to Arsenate Oxidation and Water Disinfection via Solar Heterogeneous Photocatalysis: A Kinetic and Statistical Approach
Abstract
:1. Introduction
2. Materials and Methods
2.1. Location and Operation Timeframe for Experiments
2.2. Photocatalytic Reactors
2.3. Experimental Conditions
2.4. Kinetic Analysis
2.5. Fluence Analysis
2.6. Collector Area per Order Determination
2.7. Experimental Design and Statistical Analysis
3. Results and Discussion
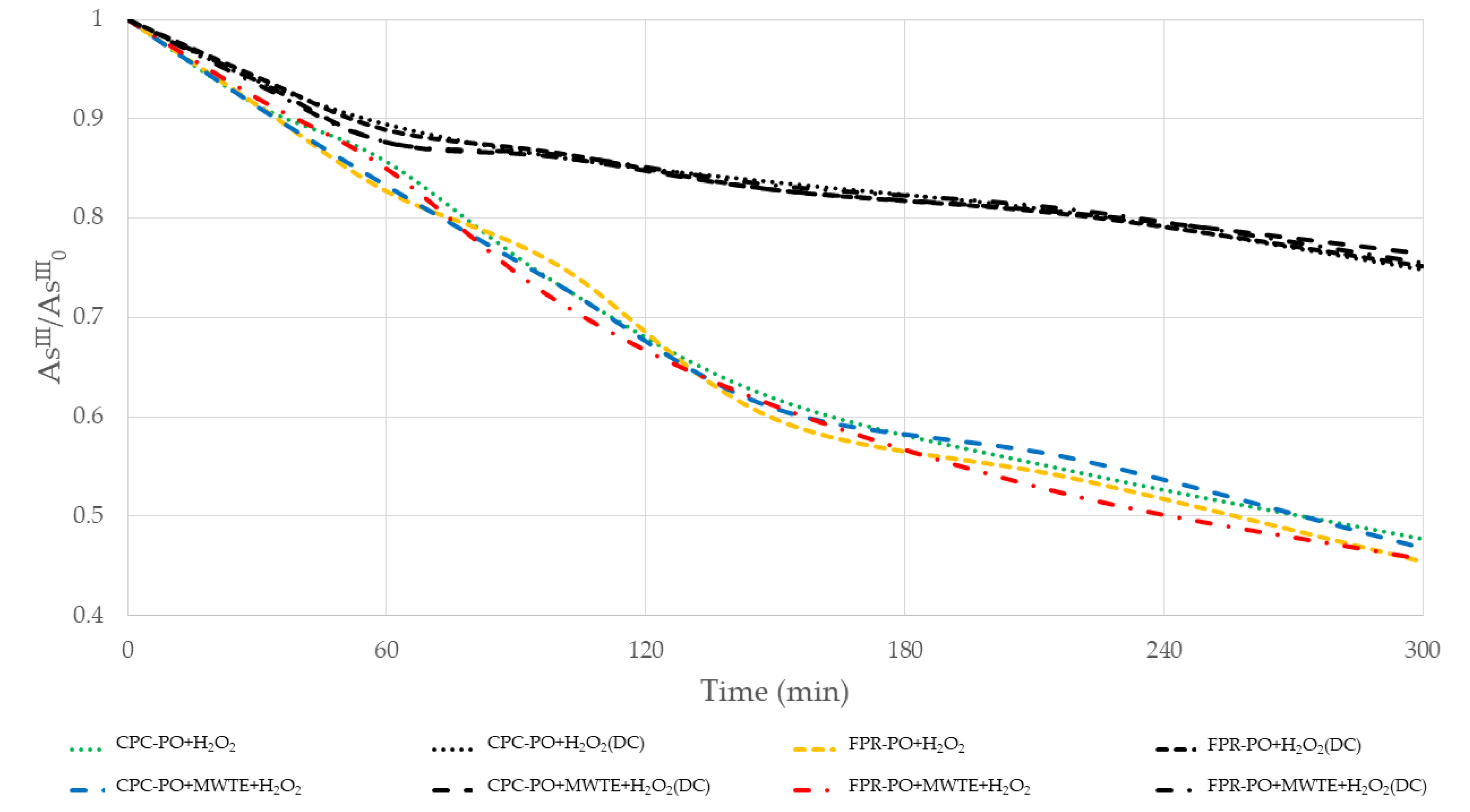
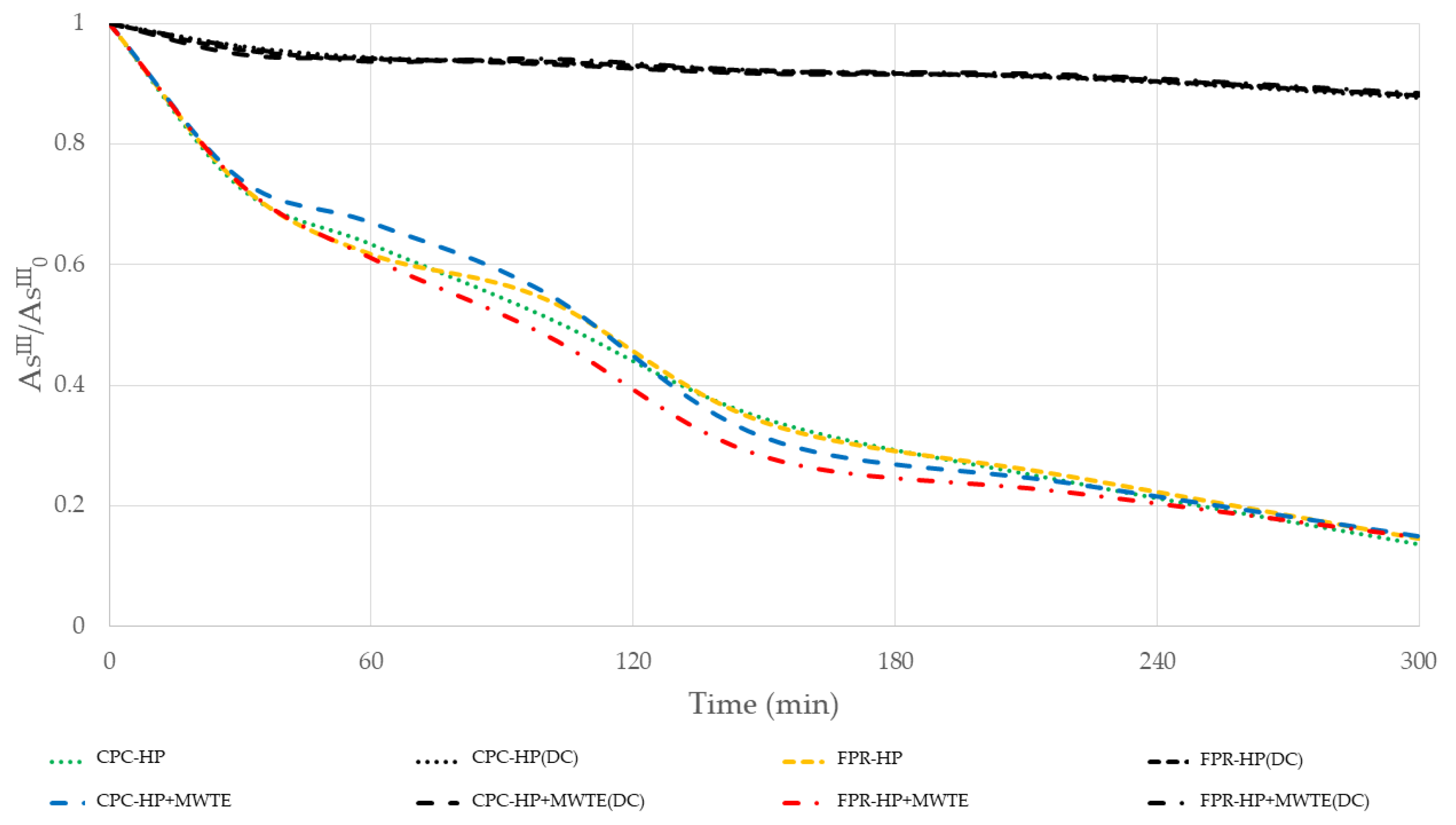
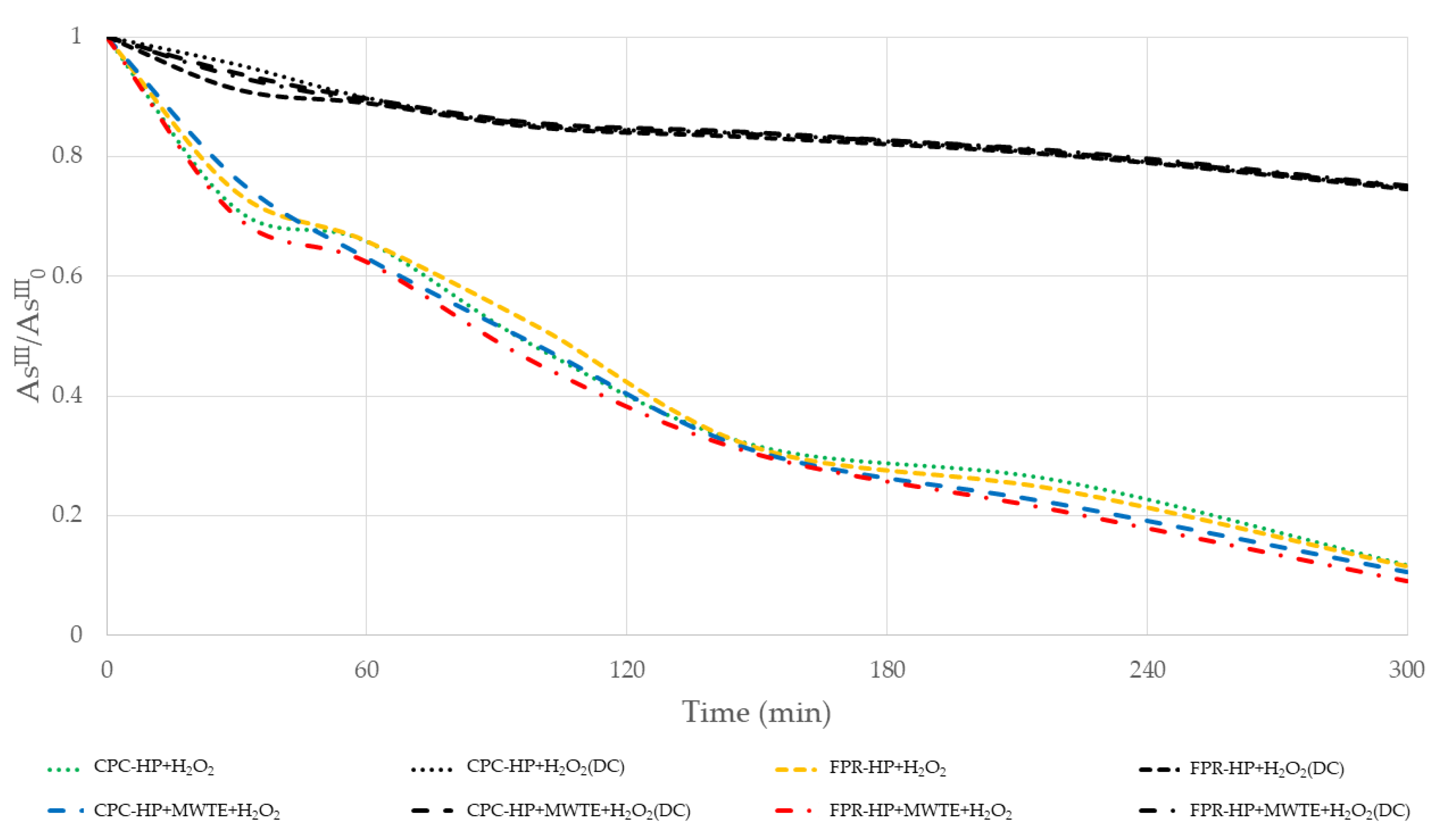
3.1. Arsenic Oxidation
3.2. Arsenic Removal
3.3. Coliform Disinfection
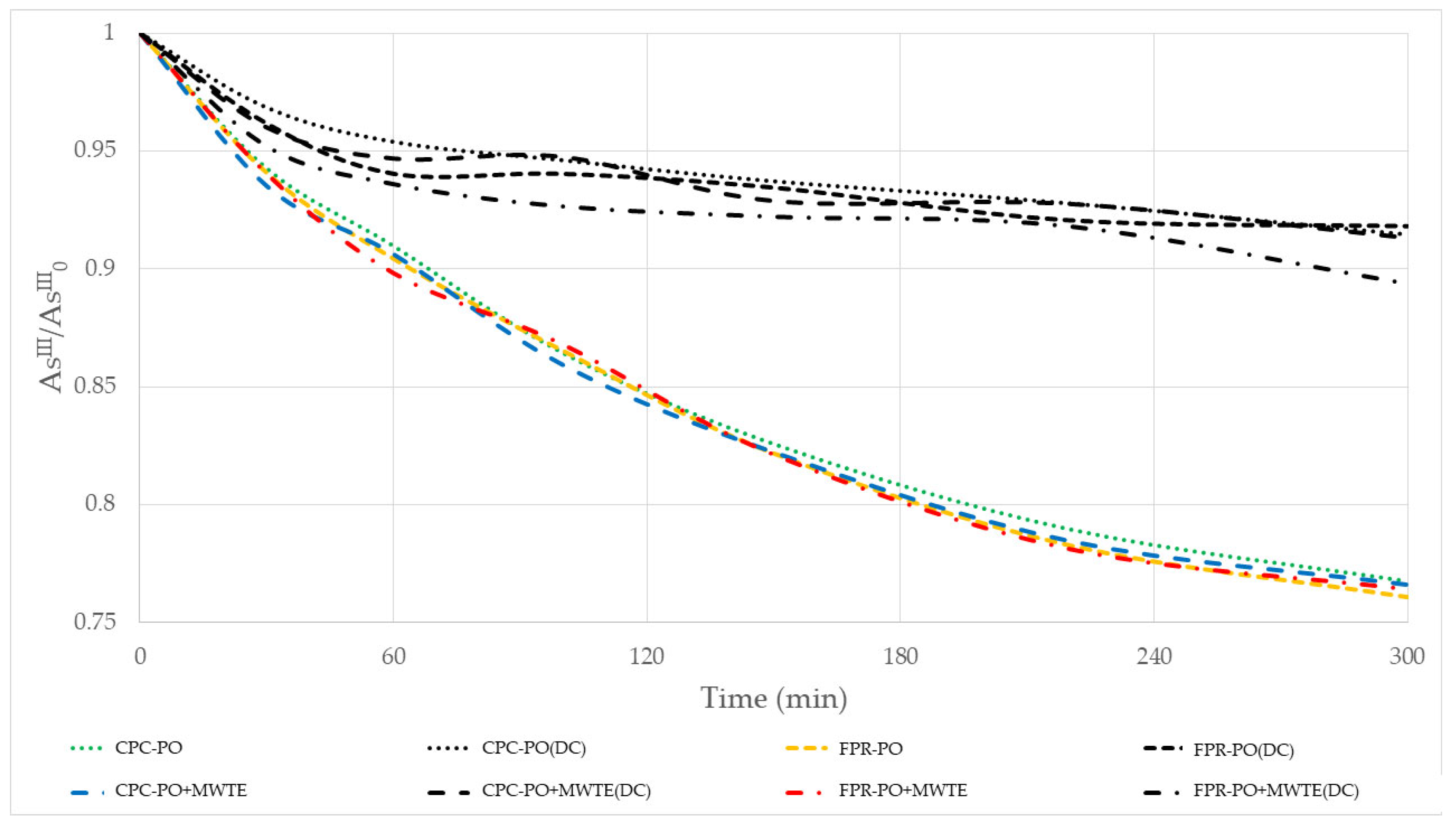
3.4. Kinetic Analysis
3.5. Fluence Analysis
3.6. Collector Area per Order
3.7. ANOVA Results
3.8. Perspectives and Outlook
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mandal, B. Arsenic Round the World: A Review. Talanta 2002, 58, 201–235. [Google Scholar] [CrossRef]
- Urseler, N.; Bachetti, R.; Morgante, V.; Agostini, E.; Morgante, C. Groundwater Quality and Vulnerability in Farms from Agricultural-Dairy Basin of the Argentine Pampas. Environ. Sci. Pollut. Res. 2022. [Google Scholar] [CrossRef]
- Jain, N.; Chandramani, S. Arsenic Poisoning- An Overview. Indian J. Med. Spec. 2018, 9, 143–145. [Google Scholar] [CrossRef]
- Flora, S.J.S. Arsenic and Dichlorvos: Possible Interaction between Two Environmental Contaminants. J. Trace Elem. Med. Biol. 2016, 35, 43–60. [Google Scholar] [CrossRef]
- Bjørklund, G.; Oliinyk, P.; Lysiuk, R.; Rahaman, M.S.; Antonyak, H.; Lozynska, I.; Lenchyk, L.; Peana, M. Arsenic Intoxication: General Aspects and Chelating Agents. Arch. Toxicol. 2020, 94, 1879–1897. [Google Scholar] [CrossRef]
- Anand, V.; Kaur, J.; Srivastava, S.; Bist, V.; Singh, P.; Srivastava, S. Arsenotrophy: A Pragmatic Approach for Arsenic Bioremediation. J. Environ. Chem. Eng. 2022, 10, 107528. [Google Scholar] [CrossRef]
- Yu, S.; Li, L.-H.; Lee, C.-H.; Jeyakannu, P.; Wang, J.-J.; Hong, C.-H. Arsenic Leads to Autophagy of Keratinocytes by Increasing Aquaporin 3 Expression. Sci. Rep. 2021, 11, 17523. [Google Scholar] [CrossRef]
- García-Rosales, G.; Longoria-Gándara, L.C.; Cruz-Cruz, G.J.; Olayo-González, M.G.; Mejía-Cuero, R.; Pérez, P.Á. Fe-TiOx Nanoparticles on Pineapple Peel: Synthesis, Characterization and As(V) Sorption. Environ. Nanotechnol. Monit. Manag. 2018, 9, 112–121. [Google Scholar] [CrossRef]
- Khan, M.I.; Ahmad, M.F.; Ahmad, I.; Ashfaq, F.; Wahab, S.; Alsayegh, A.A.; Kumar, S.; Hakeem, K.R. Arsenic Exposure through Dietary Intake and Associated Health Hazards in the Middle East. Nutrients 2022, 14, 2136. [Google Scholar] [CrossRef]
- Guglielmi, G. Arsenic in Drinking Water Threatens up to 60 Million in Pakistan. Science 2017, 14, 2136. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Fatoki, J.O.; Badmus, J.A. Arsenic as an Environmental and Human Health Antagonist: A Review of Its Toxicity and Disease Initiation. J. Hazard. Mater. Adv. 2022, 5, 100052. [Google Scholar] [CrossRef]
- Raju, N.J. Arsenic in the Geo-Environment: A Review of Sources, Geochemical Processes, Toxicity and Removal Technologies. Environ. Res. 2022, 203, 111782. [Google Scholar] [CrossRef]
- Taviani, E.; Pedro, O. Impact of the Aquatic Pathobiome in Low-Income and Middle-Income Countries (LMICs) Quest for Safe Water and Sanitation Practices. Curr. Opin. Biotechnol. 2022, 73, 220–224. [Google Scholar] [CrossRef]
- Hanif, Z.; Tariq, M.Z.; Khan, Z.A.; La, M.; Choi, D.; Park, S.J. Polypyrrole-Coated Nanocellulose for Solar Steam Generation: A Multi-Surface Photothermal Ink with Antibacterial and Antifouling Properties. Carbohydr. Polym. 2022, 292, 119701. [Google Scholar] [CrossRef]
- Javaid, M.; Qasim, H.; Zia, H.Z.; Bashir, M.A.; Syeda Amber Hameed, A.Q.; Samiullah, K.; Hashem, M.; Morsy, K.; Dajem, S.B.; Muhammad, T.; et al. Bacteriological Composition of Groundwater and Its Role in Human Health. J. King Saud Univ. Sci. 2022, 34, 102128. [Google Scholar] [CrossRef]
- Adelodun, B.; Ajibade, F.O.; Ighalo, J.O.; Odey, G.; Ibrahim, R.G.; Kareem, K.Y.; Bakare, H.O.; Tiamiyu, A.O.; Ajibade, T.F.; Abdulkadir, T.S.; et al. Assessment of Socioeconomic Inequality Based on Virus-Contaminated Water Usage in Developing Countries: A Review. Environ. Res. 2021, 192, 110309. [Google Scholar] [CrossRef]
- Ahmad, A.; Bhattacharya, P. Arsenic in Drinking Water: Is 10 Μg/L a Safe Limit? Curr. Pollut. Rep. 2019, 5, 1–3. [Google Scholar] [CrossRef] [Green Version]
- Wen, X.; Chen, F.; Lin, Y.; Zhu, H.; Yuan, F.; Kuang, D.; Jia, Z.; Yuan, Z. Microbial Indicators and Their Use for Monitoring Drinking Water Quality—A Review. Sustainability 2020, 12, 2249. [Google Scholar] [CrossRef] [Green Version]
- Valdiviezo Gonzales, L.G.; García Ávila, F.F.; Cabello Torres, R.J.; Castañeda Olivera, C.A.; Alfaro Paredes, E.A. Scientometric Study of Drinking Water Treatments Technologies: Present and Future Challenges. Cogent Eng. 2021, 8, 1929046. [Google Scholar] [CrossRef]
- Sundar, K.P.; Kanmani, S. Progression of Photocatalytic Reactors and It’s Comparison: A Review. Chem. Eng. Res. Des. 2020, 154, 135–150. [Google Scholar] [CrossRef]
- Jabbar, Z.H.; Esmail Ebrahim, S. Recent Advances in Nano-Semiconductors Photocatalysis for Degrading Organic Contaminants and Microbial Disinfection in Wastewater: A Comprehensive Review. Environ. Nanotechnol. Monit. Manag. 2022, 17, 100666. [Google Scholar] [CrossRef]
- Sibhatu, A.K.; Weldegebrieal, G.K.; Sagadevan, S.; Tran, N.N.; Hessel, V. Photocatalytic Activity of CuO Nanoparticles for Organic and Inorganic Pollutants Removal in Wastewater Remediation. Chemosphere 2022, 300, 134623. [Google Scholar] [CrossRef]
- Wang, H.; Li, X.; Zhao, X.; Li, C.; Song, X.; Zhang, P.; Huo, P.; Li, X. A Review on Heterogeneous Photocatalysis for Environmental Remediation: From Semiconductors to Modification Strategies. Chin. J. Catal. 2022, 43, 178–214. [Google Scholar] [CrossRef]
- Xie, L.; Hao, J.G.; Chen, H.Q.; Li, Z.X.; Ge, S.Y.; Mi, Y.; Yang, K.; Lu, K.Q. Recent Advances of Nickel Hydroxide-Based Cocatalysts in Heterogeneous Photocatalysis. Catal. Commun. 2022, 162, 106371. [Google Scholar] [CrossRef]
- Karim, A.V.; Krishnan, S.; Shriwastav, A. An Overview of Heterogeneous Photocatalysis for the Degradation of Organic Compounds: A Special Emphasis on Photocorrosion and Reusability. J. Indian Chem. Soc. 2022, 99, 100480. [Google Scholar] [CrossRef]
- Sharma, A.; Ahmad, J.; Flora, S.J.S. Application of Advanced Oxidation Processes and Toxicity Assessment of Transformation Products. Environ. Res. 2018, 167, 223–233. [Google Scholar] [CrossRef]
- Marinho, B.A.; Cristóvão, R.O.; Boaventura, R.A.R.; Vilar, V.J.P. As(III) and Cr(VI) Oxyanion Removal from Water by Advanced Oxidation/Reduction Processes—A Review. Environ. Sci. Pollut. Res. 2019, 26, 2203–2227. [Google Scholar] [CrossRef]
- García, F.E.; Litter, M.I.; Sora, I.N. Assessment of the Arsenic Removal From Water Using Lanthanum Ferrite. ChemistryOpen 2021, 10, 790–797. [Google Scholar] [CrossRef]
- Deng, Y.; Li, Y.; Li, X.; Sun, Y.; Ma, J.; Lei, M.; Weng, L. Influence of Calcium and Phosphate on PH Dependency of Arsenite and Arsenate Adsorption to Goethite. Chemosphere 2018, 199, 617–624. [Google Scholar] [CrossRef]
- Hosseini, F.; Assadi, A.A.; Nguzen-Tri, P.; Ali, I.; Rtimi, S. Titanium-Based Photocatalytic Coatings for Bacterial Disinfection: The Shift from Suspended Powders to Catalytic Interfaces. Surf. Interfaces 2022, 32, 102078. [Google Scholar] [CrossRef]
- John, D.; Jose, J.; Bhat, S.G.; Achari, V.S. Integration of Heterogeneous Photocatalysis and Persulfate Based Oxidation Using TiO2-Reduced Graphene Oxide for Water Decontamination and Disinfection. Heliyon 2021, 7, e07451. [Google Scholar] [CrossRef] [PubMed]
- Byrne, J.; Dunlop, P.; Hamilton, J.; Fernández-Ibáñez, P.; Polo-López, I.; Sharma, P.; Vennard, A. A Review of Heterogeneous Photocatalysis for Water and Surface Disinfection. Molecules 2015, 20, 5574–5615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Y.; Liu, Q.; Liu, C.; Zhai, Y.; Xie, M.; Huang, L.; Xu, H.; Li, H.; Jing, J. Visible-Light-Driven Ag/AgBr/ZnFe2O4 Composites with Excellent Photocatalytic Activity for E. coli Disinfection and Organic Pollutant Degradation. J. Colloid Interface Sci. 2018, 512, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Serrà, A.; Philippe, L.; Perreault, F.; Garcia-Segura, S. Photocatalytic Treatment of Natural Waters. Reality or Hype? The Case of Cyanotoxins Remediation. Water Res. 2021, 188, 116543. [Google Scholar] [CrossRef] [PubMed]
- Antonopoulou, M.; Kosma, C.; Albanis, T.; Konstantinou, I. An Overview of Homogeneous and Heterogeneous Photocatalysis Applications for the Removal of Pharmaceutical Compounds from Real or Synthetic Hospital Wastewaters under Lab or Pilot Scale. Sci. Total Environ. 2021, 765, 144163. [Google Scholar] [CrossRef]
- Espíndola, J.C.; Vilar, V.J.P. Innovative Light-Driven Chemical/Catalytic Reactors towards Contaminants of Emerging Concern Mitigation: A Review. Chem. Eng. J. 2020, 394, 124865. [Google Scholar] [CrossRef]
- Chen, L.; Tang, J.; Song, L.-N.; Chen, P.; He, J.; Au, C.-T.; Yin, S.-F. Heterogeneous Photocatalysis for Selective Oxidation of Alcohols and Hydrocarbons. Appl. Catal. B Environ. 2019, 242, 379–388. [Google Scholar] [CrossRef]
- Binjhade, R.; Mondal, R.; Mondal, S. Continuous Photocatalytic Reactor: Critical Review on the Design and Performance. J. Environ. Chem. Eng. 2022, 10, 107746. [Google Scholar] [CrossRef]
- Zaruma-Arias, P.E.; Núñez-Núñez, C.M.; González-Burciaga, L.A.; Proal-Nájera, J.B. Solar Heterogenous Photocatalytic Degradation of Methylthionine Chloride on a Flat Plate Reactor: Effect of PH and H2O2 Addition. Catalysts 2022, 12, 132. [Google Scholar] [CrossRef]
- Silerio-Vázquez, F.; Alarcón-Herrera, M.T.; Proal-Nájera, J.B. Solar Heterogeneous Photocatalytic Degradation of Phenol on TiO2/Quartz and TiO2/Calcite: A Statistical and Kinetic Approach on Comparative Efficiencies towards a TiO2/Glass System. Environ. Sci. Pollut. Res. 2022, 29, 42319–42330. [Google Scholar] [CrossRef]
- Núñez-Núñez, C.M.; Osorio-Revilla, G.I.; Villanueva-Fierro, I.; Antileo, C.; Proal-Nájera, J.B. Solar Fecal Coliform Disinfection in a Wastewater Treatment Plant by Oxidation Processes: Kinetic Analysis as a Function of Solar Radiation. Water 2020, 12, 639. [Google Scholar] [CrossRef] [Green Version]
- González-Burciaga, L.A.; Núñez-Núñez, C.M.; Morones-Esquivel, M.M.; Avila-Santos, M.; Lemus-Santana, A.; Proal-Nájera, J.B. Characterization and Comparative Performance of TiO2 Photocatalysts on 6-Mercaptopurine Degradation by Solar Heterogeneous Photocatalysis. Catalysts 2020, 10, 118. [Google Scholar] [CrossRef] [Green Version]
- Silerio-Vázquez, F.d.J.; Núñez-Núñez, C.M.; Alarcón-Herrera, M.T.; Proal-Nájera, J.B. Comparative Efficiencies for Phenol Degradation on Solar Heterogeneous Photocatalytic Reactors: Flat Plate and Compound Parabolic Collector. Catalysts 2022, 12, 575. [Google Scholar] [CrossRef]
- Leiva-Aravena, E.; Vera, M.A.; Nerenberg, R.; Leiva, E.D.; Vargas, I.T. Biofilm Formation of Ancylobacter Sp. TS-1 on Different Granular Materials and Its Ability for Chemolithoautotrophic As(III)-Oxidation at High Concentrations. J. Hazard. Mater. 2022, 421, 126733. [Google Scholar] [CrossRef] [PubMed]
- Vieira, B.R.C.; Pintor, A.M.A.; Boaventura, R.A.R.; Botelho, C.M.S.; Santos, S.C.R. Arsenic Removal from Water Using Iron-Coated Seaweeds. J. Environ. Manag. 2017, 192, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Laky, D.; Licskó, I. Arsenic Removal by Ferric-Chloride Coagulation—Effect of Phosphate, Bicarbonate and Silicate. Water Sci. Technol. 2011, 64, 1046–1055. [Google Scholar] [CrossRef]
- Cravo, A.; Barbosa, A.B.; Correia, C.; Matos, A.; Caetano, S.; Lima, M.J.; Jacob, J. Unravelling the Effects of Treated Wastewater Discharges on the Water Quality in a Coastal Lagoon System (Ria Formosa, South Portugal): Relevance of Hydrodynamic Conditions. Mar. Pollut. Bull. 2022, 174, 113296. [Google Scholar] [CrossRef]
- Luvhimbi, N.; Tshitangano, T.G.; Mabunda, J.T.; Olaniyi, F.C.; Edokpayi, J.N. Water Quality Assessment and Evaluation of Human Health Risk of Drinking Water from Source to Point of Use at Thulamela Municipality, Limpopo Province. Sci. Rep. 2022, 12, 6059. [Google Scholar] [CrossRef]
- Hile, T.D.; Dunbar, S.G.; Sinclair, R.G. Microbial Contamination of Drinking Water from Vending Machines of Eastern Coachella Valley. Water Supply 2021, 21, 1618–1628. [Google Scholar] [CrossRef]
- Horváth, E.; Gabathuler, J.; Bourdiec, G.; Vidal-Revel, E.; Benthem Muñiz, M.; Gaal, M.; Grandjean, D.; Breider, F.; Rossi, L.; Sienkiewicz, A.; et al. Solar Water Purification with Photocatalytic Nanocomposite Filter Based on TiO2 Nanowires and Carbon Nanotubes. npj Clean Water 2022, 5, 10. [Google Scholar] [CrossRef]
- García, A.; Rosales, M.; Thomas, M.; Golemme, G. Arsenic Photocatalytic Oxidation over TiO2-Loaded SBA-15. J. Environ. Chem. Eng. 2021, 9, 106443. [Google Scholar] [CrossRef]
- Wei, Z.; Fang, Y.; Wang, Z.; Liu, Y.; Wu, Y.; Liang, K.; Yan, J.; Pan, Z.; Hu, G. PH Effects of the Arsenite Photocatalytic Oxidation Reaction on Different Anatase TiO2 Facets. Chemosphere 2019, 225, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Kanth, N.; Xu, W.; Prasad, U.; Ravichandran, D.; Kannan, A.M.; Song, K. PMMA-TiO2 Fibers for the Photocatalytic Degradation of Water Pollutants. Nanomaterials 2020, 10, 1279. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, Z.; Dong, Z.; Wu, Y.; Zhu, X.; Cheng, Z.; Liu, Y.; Wang, Y.; Zheng, Z.; Cao, X.; et al. CuS/TiO2 Nanotube Arrays Heterojunction for the Photoreduction of Uranium (VI). J. Solid State Chem. 2021, 303, 122499. [Google Scholar] [CrossRef]
- Kuhn, H.; Försterling, H.-D.; Waldeck, D.H. Chemical Kinetics. In Principles of Physical Chemistry; John Wiley & Sons, LTD: West Sussex, UK, 2000; pp. 735–794. ISBN 978-0-470-08964-4. [Google Scholar]
- Giménez, J.; Curcó, D.; Queral, M. Photocatalytic Treatment of Phenol and 2,4-Dichlorophenol in a Solar Plant in the Way to Scaling-Up. Catal. Today 1999, 54, 229–243. [Google Scholar] [CrossRef]
- Gutiérrez-Alfaro, S.; Acevedo, A.; Rodríguez, J.; Carpio, E.A.; Manzano, M.A. Solar Photocatalytic Water Disinfection of Escherichia coli, Enterococcus spp. and Clostridium perfringens Using Different Low-Cost Devices. J. Chem. Technol. Biotechnol. 2016, 91, 2026–2037. [Google Scholar] [CrossRef]
- Mac Mahon, J.; Pillai, S.C.; Kelly, J.M.; Gill, L.W. Solar Photocatalytic Disinfection of E. Coli and Bacteriophages MS2, ΦX174 and PR772 Using TiO2, ZnO and Ruthenium Based Complexes in a Continuous Flow System. J. Photochem. Photobiol. B Biol. 2017, 170, 79–90. [Google Scholar] [CrossRef]
- Pereira, J.H.O.S.; Vilar, V.J.P.; Borges, M.T.; González, O.; Esplugas, S.; Boaventura, R.A.R. Photocatalytic Degradation of Oxytetracycline Using TiO2 under Natural and Simulated Solar Radiation. Sol. Energy 2011, 85, 2732–2740. [Google Scholar] [CrossRef]
- Rincón, A.-G.; Pulgarin, C. Field Solar E. Coli Inactivation in the Absence and Presence of TiO2: Is UV Solar Dose an Appropriate Parameter for Standardization of Water Solar Disinfection? Sol. Energy 2004, 77, 635–648. [Google Scholar] [CrossRef]
- Bolton, J.R.; Bircher, K.G.; Tumas, W.; Tolman, C.A. Figures-of-Merit for the Technical Development and Application of Advanced Oxidation Technologies for Both Electric- and Solar-Driven Systems (IUPAC Technical Report). Pure Appl. Chem. 2001, 73, 627–637. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef] [Green Version]
- Manjiao, C.; Zhengfu, Z.; Xinjun, H.; Jianping, T.; Jingsong, W.; Rundong, W.; Xian, Z.; Xinjun, Z.; PeiLun, S.; Dianwen, L. Oxidation Mechanism of the Arsenopyrite Surface by Oxygen with and without Water: Experimental and Theoretical Analysis. Appl. Surf. Sci. 2022, 573, 151574. [Google Scholar] [CrossRef]
- Heiba, H.F.; Bullen, J.C.; Kafizas, A.; Petit, C.; Skinner, S.J.; Weiss, D. The Determination of Oxidation Rates and Quantum Yields during the Photocatalytic Oxidation of As(III) over TiO2. J. Photochem. Photobiol. A Chem. 2022, 424, 113628. [Google Scholar] [CrossRef]
- Su, J.; Lyu, T.; Cooper, M.; Mortimer, R.J.G.; Pan, G. Efficient Arsenic Removal by a Bifunctional Heterogeneous Catalyst through Simultaneous Hydrogen Peroxide (H2O2) Catalytic Oxidation and Adsorption. J. Clean. Prod. 2021, 325, 129329. [Google Scholar] [CrossRef]
- Wang, X.; Chen, J.; Bu, Z.; Wang, H.; Wang, W.; Li, W.; Sun, T. Accelerated C-Face Polishing of Silicon Carbide by Alkaline Polishing Slurries with Fe3O4 Catalysts. J. Environ. Chem. Eng. 2021, 9, 106863. [Google Scholar] [CrossRef]
- Hong, J.; Liu, L.; Ning, Z.; Liu, C.; Qiu, G. Synergistic Oxidation of Dissolved As(III) and Arsenopyrite in the Presence of Oxygen: Formation and Function of Reactive Oxygen Species. Water Res. 2021, 202, 117416. [Google Scholar] [CrossRef]
- Song, J.; Yan, L.; Duan, J.; Jing, C. TiO2 Crystal Facet-Dependent Antimony Adsorption and Photocatalytic Oxidation. J. Colloid Interface Sci. 2017, 496, 522–530. [Google Scholar] [CrossRef]
- Ning, R.Y. Arsenic Removal by Reverse Osmosis. Desalination 2002, 143, 237–241. [Google Scholar] [CrossRef]
- Litter, M.I. Last Advances on TiO2-Photocatalytic Removal of Chromium, Uranium and Arsenic. Curr. Opin. Green Sustain. Chem. 2017, 6, 150–158. [Google Scholar] [CrossRef]
- Silerio-Vázquez, F.; Proal Nájera, J.B.; Bundschuh, J.; Alarcon-Herrera, M.T. Photocatalysis for Arsenic Removal from Water: Considerations for Solar Photocatalytic Reactors. Environ. Sci. Pollut. Res. 2021. [Google Scholar] [CrossRef]
- Meng, F.; Zhang, S.; Zeng, Y.; Zhang, M.; Zou, H.; Zhong, Q.; Li, Y. Promotional Effect of Surface Fluorine on TiO2: Catalytic Conversion of O3 and H2O2 into ·OH and ·O2− Radicals for High-Efficiency NO Oxidation. Chem. Eng. J. 2021, 424, 130358. [Google Scholar] [CrossRef]
- Naniwa, S.; Yamamoto, A.; Yoshida, H. Visible Light-Induced Minisci Reaction through Photoexcitation of Surface Ti-Peroxo Species. Catal. Sci. Technol. 2021, 11, 3376–3384. [Google Scholar] [CrossRef]
- Shan, C.; Liu, Y.; Huang, Y.; Pan, B. Non-Radical Pathway Dominated Catalytic Oxidation of As(III) with Stoichiometric H2O2 over Nanoceria. Environ. Int. 2019, 124, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Liu, Y.; Liu, F.; Li, F.; Shen, C.; Wu, Z. Extremely Efficient Electro-Fenton-like Sb(III) Detoxification Using Nanoscale Ti-Ce Binary Oxide: An Effective Design to Boost Catalytic Activity via Non-Radical Pathway. Chin. Chem. Lett. 2021, 32, 2519–2523. [Google Scholar] [CrossRef]
- Ghanbari, F.; Giannakis, S.; Lin, K.-Y.A.; Wu, J.; Madihi-Bidgoli, S. Acetaminophen Degradation by a Synergistic Peracetic Acid/UVC-LED/Fe(II) Advanced Oxidation Process: Kinetic Assessment, Process Feasibility and Mechanistic Considerations. Chemosphere 2021, 263, 128119. [Google Scholar] [CrossRef]
- Zhang, X.; Dong, Q.; Wang, Y.; Zhu, Z.; Guo, Z.; Li, J.; Lv, Y.; Chow, Y.T.; Wang, X.; Zhu, L.; et al. Water-Stable Metal–Organic Framework (UiO-66) Supported on Zirconia Nanofibers Membrane for the Dynamic Removal of Tetracycline and Arsenic from Water. Appl. Surf. Sci. 2022, 596, 153559. [Google Scholar] [CrossRef]
- Lou, T.; Song, S.; Gao, X.; Qian, W.; Chen, X.; Li, Q. Sub-20-Nm Anatase TiO2 Anchored on Hollow Carbon Spheres for Enhanced Photocatalytic Degradation of Reactive Red 195. J. Colloid Interface Sci. 2022, 617, 663–672. [Google Scholar] [CrossRef]
- Dudek, S.; Kołodyńska, D. Arsenic(V) Removal on the Lanthanum-Modified Ion Exchanger with Quaternary Ammonium Groups Based on Iron Oxide. J. Mol. Liq. 2022, 347, 117985. [Google Scholar] [CrossRef]
- Chi, Z.; Zhu, Y.; Liu, W.; Huang, H.; Li, H. Selective Removal of As(III) Using Magnetic Graphene Oxide Ion-Imprinted Polymer in Porous Media: Potential Effect of External Magnetic Field. J. Environ. Chem. Eng. 2021, 9, 105671. [Google Scholar] [CrossRef]
- Zeng, H.; Zhai, L.; Qiao, T.; Yu, Y.; Zhang, J.; Li, D. Efficient Removal of As(V) from Aqueous Media by Magnetic Nanoparticles Prepared with Iron-Containing Water Treatment Residuals. Sci. Rep. 2020, 10, 9335. [Google Scholar] [CrossRef]
- Weerasundara, L.; Ok, Y.-S.; Bundschuh, J. Selective Removal of Arsenic in Water: A Critical Review. Environ. Pollut. 2021, 268, 115668. [Google Scholar] [CrossRef]
- Hug, S.J.; Leupin, O. Iron-Catalyzed Oxidation of Arsenic (III) by Oxygen and by Hydrogen Peroxide: pH-Dependent Formation of Oxidants in the Fenton Reaction. Environ. Sci. Technol. 2003, 37, 2734–2742. [Google Scholar] [CrossRef]
- García-Gil, Á.; Feng, L.; Moreno-SanSegundo, J.; Giannakis, S.; Pulgarín, C.; Marugán, J. Mechanistic Modelling of Solar Disinfection (SODIS) Kinetics of Escherichia coli, Enhanced with H2O2—Part 2: Shine on You, Crazy Peroxide. Chem. Eng. J. 2022, 439, 135783. [Google Scholar] [CrossRef]
- Cowie, B.E.; Porley, V.; Robertson, N. Solar Disinfection (SODIS) Provides a Much Underexploited Opportunity for Researchers in Photocatalytic Water Treatment (PWT). ACS Catal. 2020, 10, 11779–11782. [Google Scholar] [CrossRef]
- Oturan, M.A.; Aaron, J.-J.J. Advanced Oxidation Processes in Water/Wastewater Treatment: Principles and Applications. A Review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2577–2641. [Google Scholar] [CrossRef]
- Amyot, M.; Bélanger, D.; Simon, D.F.; Chételat, J.; Palmer, M.; Ariya, P. Photooxidation of Arsenic in Pristine and Mine-Impacted Canadian Subarctic Freshwater Systems. J. Hazard. Mater. Adv. 2021, 2, 100006. [Google Scholar] [CrossRef]
- Saleh, S.; Mohammadnejad, S.; Khorgooei, H.; Otadi, M. Photooxidation/Adsorption of Arsenic (III) in Aqueous Solution over Bentonite/Chitosan/TiO2 Heterostructured Catalyst. Chemosphere 2021, 280, 130583. [Google Scholar] [CrossRef]
- Shen, J.; Yu, H.; Shu, Y.; Ma, M.; Chen, H. A Robust ROS Generation Strategy for Enhanced Chemodynamic/Photodynamic Therapy via H2O2/O2 Self-Supply and Ca2+ Overloading. Adv. Funct. Mater. 2021, 31, 2106106. [Google Scholar] [CrossRef]
- Huang, L.; Liu, S.; Li, X.; Peng, X.; Liu, D. Controllable High-Efficiency Transformation of H2O2 to Reactive Oxygen Species via Electroactivation of Ti-Peroxo Complexes. Sep. Purif. Technol. 2022, 289, 120747. [Google Scholar] [CrossRef]
- Pretali, L.; Fasani, E.; Sturini, M. Current Advances on the Photocatalytic Degradation of Fluoroquinolones: Photoreaction Mechanism and Environmental Application. Photochem. Photobiol. Sci. 2022, 21, 899–912. [Google Scholar] [CrossRef]
- Abdel-Maksoud, Y.K.; Imam, E.; Ramadan, A.R. TiO2 Water-Bell Photoreactor for Wastewater Treatment. Sol. Energy 2018, 170, 323–335. [Google Scholar] [CrossRef]
- Ding, Y.; Zhou, W.; Gao, J.; Sun, F.; Zhao, G. H2O2 Electrogeneration from O2 Electroreduction by N-Doped Carbon Materials: A Mini-Review on Preparation Methods, Selectivity of N Sites, and Prospects. Adv. Mater. Interfaces 2021, 8, 2002091. [Google Scholar] [CrossRef]
- Dharma, H.N.C.; Jaafar, J.; Widiastuti, N.; Matsuyama, H.; Rajabsadeh, S.; Othman, M.H.D.; Rahman, M.A.; Jafri, N.N.M.; Suhaimin, N.S.; Nasir, A.M.; et al. A Review of Titanium Dioxide (TiO2)-Based Photocatalyst for Oilfield-Produced Water Treatment. Membranes 2022, 12, 345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Mo, Y. A Scalable Light-Diffusing Photochemical Reactor for Continuous Processing of Photoredox Reactions. Chem. Eng. J. 2022, 435, 134889. [Google Scholar] [CrossRef]
- Kanakaraju, D.; anak Kutiang, F.D.; Lim, Y.C.; Goh, P.S. Recent Progress of Ag/TiO2 Photocatalyst for Wastewater Treatment: Doping, Co-Doping, and Green Materials Functionalization. Appl. Mater. Today 2022, 27, 101500. [Google Scholar] [CrossRef]
- Nur, A.S.M.; Sultana, M.; Mondal, A.; Islam, S.; Robel, F.N.; Islam, A.; Sumi, M.S.A. A Review on the Development of Elemental and Codoped TiO2 Photocatalysts for Enhanced Dye Degradation under UV–Vis Irradiation. J. Water Process Eng. 2022, 47, 102728. [Google Scholar] [CrossRef]
- Laky, D. Predictive model for drinking water treatment technology design – the efficiency of arsenic removal by in-situ formed ferric-hydroxide. Period. Polytech. Civ. Eng. 2010, 54, 45. [Google Scholar] [CrossRef]
- Tshukudu, T.; Zheng, H.; Yang, J. Optimization of Coagulation with PFS-PDADMAC Composite Coagulants Using the Response Surface Methodology Experimental Design Technique. Water Environ. Res. 2013, 85, 456–465. [Google Scholar] [CrossRef]
- Corral Bobadilla, M.; Lorza, R.; Escribano García, E.; Somovilla Gómez, F.; Vergara González, E. Coagulation: Determination of Key Operating Parameters by Multi-Response Surface Methodology Using Desirability Functions. Water 2019, 11, 398. [Google Scholar] [CrossRef] [Green Version]
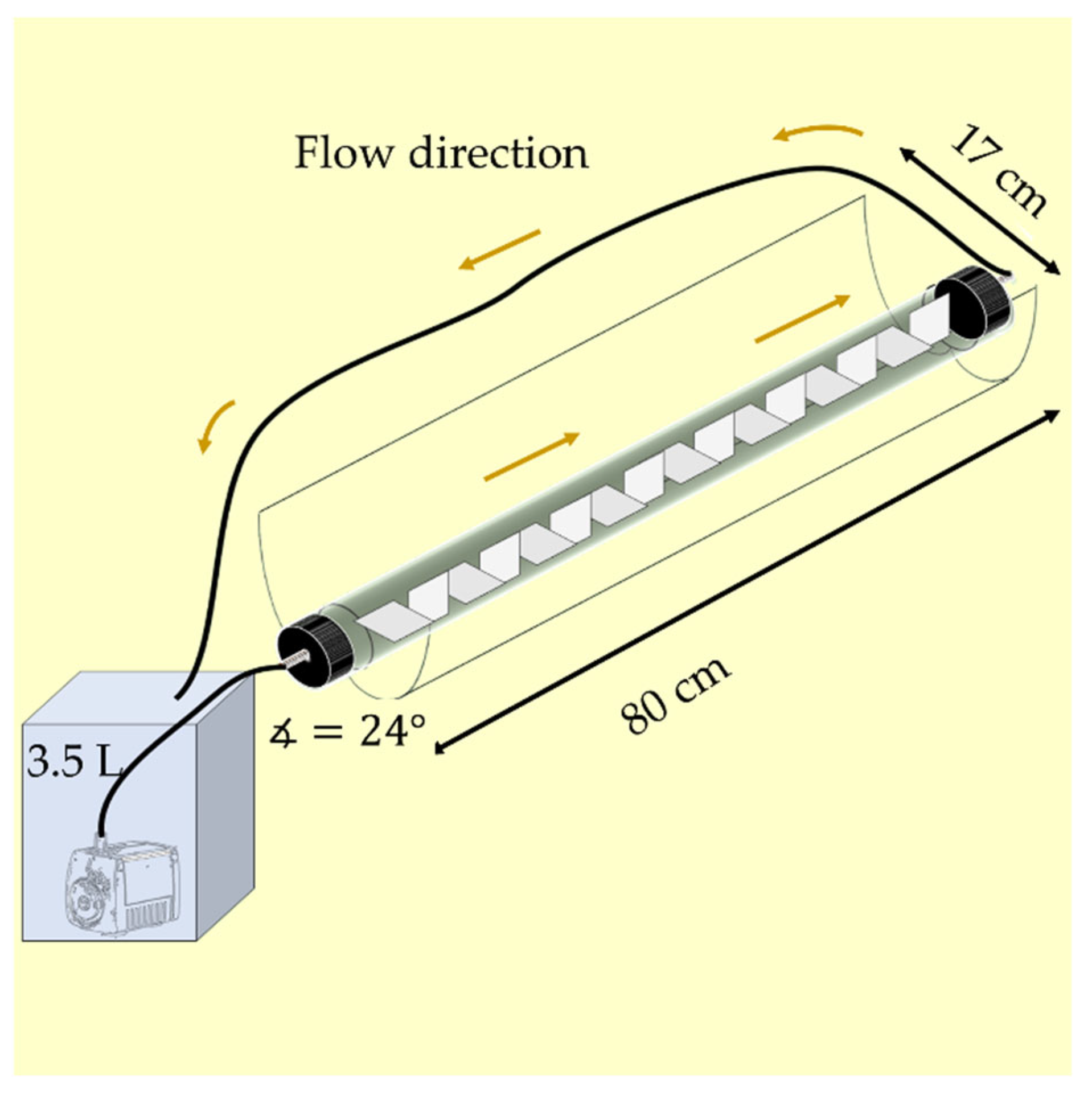
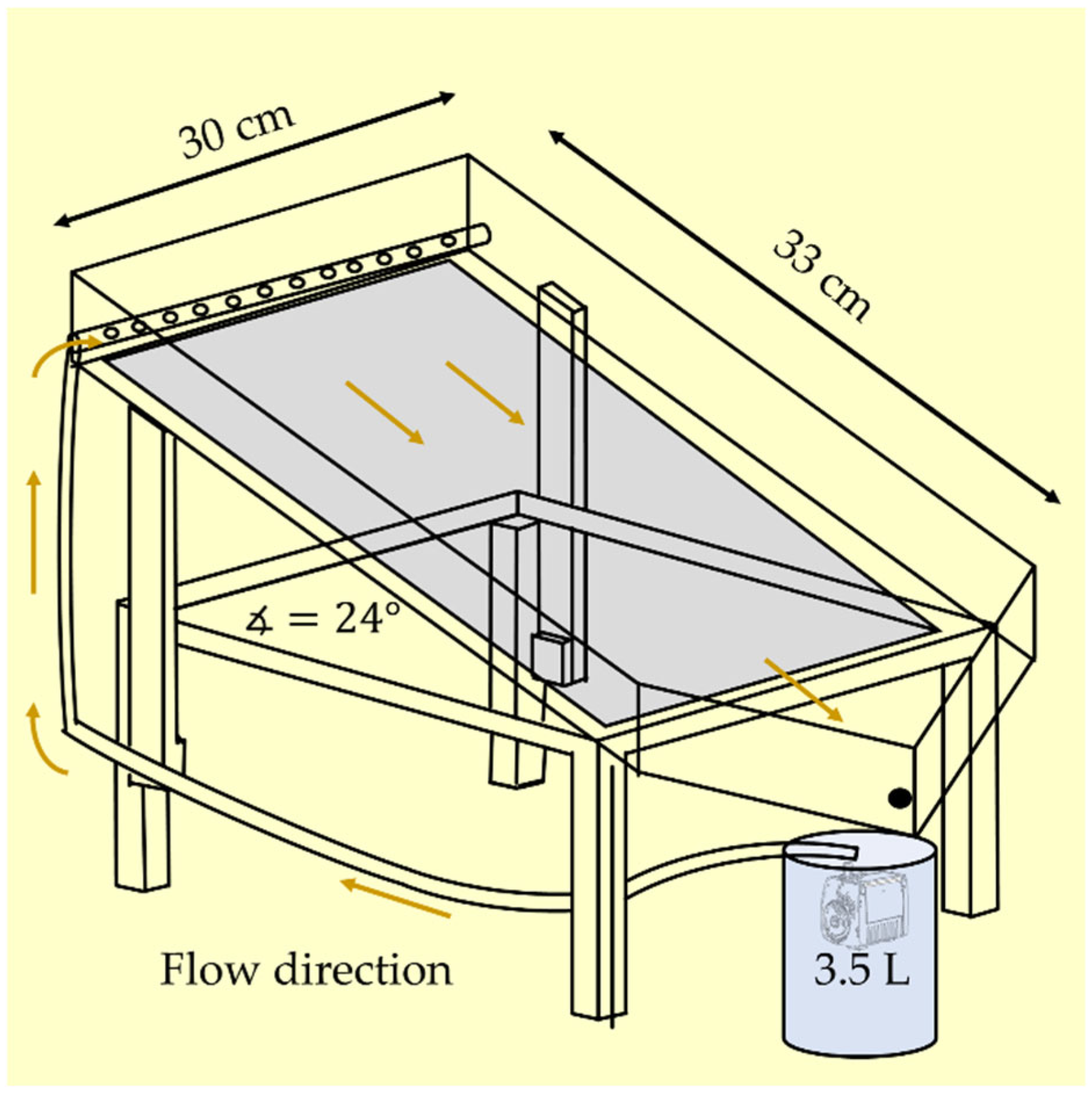
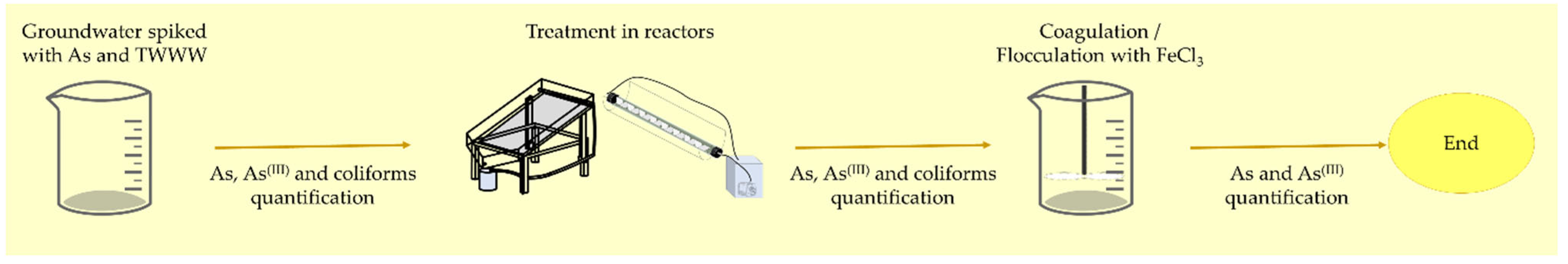
| Reactor | Illuminated Net Area (m2) | Photocatalyst Covered Area (m2) | Volumetric Flow (m3/h) |
|---|---|---|---|
| FPR | 0.10 a | 0.10 a | 0.18 |
| CPC | 1.40 b | 0.07 c | 1.50 |
| pH | 8.52 |
|---|---|
| Electrical Conductivity | 548.25 μS/cm |
| Major ions (mg/L) | |
| Na+ | 4.38 mg/L |
| K+ | 53.10 mg/L |
| Ca+2 | 9.87 mg/L |
| Mg+2 | 60.55 mg/L |
| F− | 1.54 mg/L |
| NO3- | 38.35 mg/L |
| NO2− | 1.83 mg/L |
| Cl− | 26.39 mg/L |
| HCO3− | 148.50 mg/L |
| SO4−2 | 59.75 mg/L |
| Arsenic (µg/L) | |
| AsIII | 46.06 |
| AsV | 5.46 |
| AOP | H2O2 | Reactor | MWTE Spike | Irradiation |
|---|---|---|---|---|
| Photooxidation | 0 mM | CPC | 0 mL | No irradiation (dark control) |
| Heterogeneous photocatalysis | 1 mM | FPR | 10 mL | Solar UV |
| Treatment | As (µg/L) | As Removed (%) | Treatment | As (µg/L) | As Removed (%) |
|---|---|---|---|---|---|
| CPC–PO | 23.4 ± 2.9 | 93.0 ± 0.9% | CPC–HP | 8.9 ± 2.2 | 98.1 ± 0.6% |
| CPC–PO(DC) | 25.1 ± 1.4 | 92.3 ± 0.4% | CPC–HP(DC) | 22.7 ± 0.3 | 93.5 ± 0.1% |
| FPR–PO | 20.72 ± 0.8 | 93.9 ± 0.3% | FPR–HP | 8.2 ± 1.3 | 93.3 ± 0.4% |
| FPR–PO(DC) | 24.3 ± 1.1 | 92.8 ± 0.3% | FPR–HP(DC) | 16.2 ± 1.4 | 92.5 ± 1.1% |
| CPC–PO + MWTE | 23.9 ± 0.8 | 93.3 ± 0.2% | CPC–HP + MWTE | 9.2 ± 1.4 | 97.2 ± 0.4% |
| CPC–PO + MWTE(DC) | 24.9 ± 2.1 | 92.5 ± 0.6% | CPC–HP + MWTE(DC) | 21.1 ± 1.2 | 94 ± 0.4% |
| FPR–PO + MWTE | 22.3 ± 2.5 | 92.8 ± 0.7% | FPR–HP + MWTE | 8.3 ± 0.9 | 97.5 ± 0.3% |
| FPR–PO + MWTE(DC) | 24.9 ± 2.1 | 92.6 ± 0.6% | FPR–HP + MWTE(DC) | 23.3 ± 0.1 | 93.3 ± 0.1% |
| CPC–PO + H2O2 | 16.5 ± 1.7 | 95.8 ± 0.5% | CPC–HP + H2O2 | 5.3 ± 1.1 | 98.7 ± 0.3% |
| CPC–PO + H2O2(DC) | 18.7 ± 0.5 | 94.5 ± 0.2% | CPC–HP + H2O2(DC) | 10.2 ± 1.2 | 96.8 ± 0.4% |
| FPR–PO + H2O2 | 16.5 ± 0.3 | 95.3 ± 0.1% | FPR–HP + H2O2 | 5.1 ± 0.1 | 98.6 ± 0.0% |
| FPR–PO + H2O2(DC) | 19.5 ± 0.7 | 94.2 ± 0.2% | FPR–HP + H2O2(DC) | 10.2 ± 2.7 | 96.6 ± 0.8% |
| CPC–PO + MWTE + H2O2 | 16.9 ± 0.8 | 94.9 ± 0.2% | CPC–HP + MWTE + H2O2 | 1.6 ± 0.5 | 99.7 ± 0.1% |
| CPC–PO + MWTE+ H2O2(DC) | 18.7 ± 0.5 | 94.5 ± 0.2% | CPC–HP + MWTE + H2O2(DC) | 11.8 ± 0.8 | 94.9 ± 0.2% |
| FPR–PO + MWTE + H2O2 | 16.2 ± 0.2 | 95.5 ± 0.2% | FPR–HP + MWTE + H2O2 | 1.7 ± 0.2 | 99.5 ± 0.1% |
| FPR–PO + MWTE + H2O2(DC) | 18.7 ± 0.5 | 94.5 ± 0.2% | FPR–HP + MWTE + H2O2(DC) | 11.5 ± 1.5 | 94.8 ± 0.4% |
| Treatment | 0 min (MPN/100 mL) | 300 min (MPN/100 mL) | Treatment | 0 min (MPN/100 mL) | 300 min (MPN/100 mL) |
|---|---|---|---|---|---|
| CPC–PO + MWTE | >2419 | N.D.a | CPC–HP + MWTE | >2419 | N.D. |
| CPC–PO + MWTE (DC) | >2419 | >2419 | CPC–HP + MWTE (DC) | >2419 | >2419 |
| FPR–PO + MWTE | >2419 | N.D. | FPR–HP + MWTE | >2419 | N.D. |
| FPR–PO + MWTE (DC) | >2419 | >2419 | FPR–HP + MWTE (DC) | >2419 | >2419 |
| CPC–PO + MWTE + H2O2 | >2419 | N.D. | CPC–HP + MWTE + H2O2 | >2419 | N.D. |
| CPC–PO + MWTE + H2O2 (DC) | >2419 | 574–727 | CPC–HP + MWTE + H2O2 (DC) | >2419 | 658–755 |
| FPR–PO + MWTE + H2O2 | >2419 | N.D. | FPR–HP + MWTE + H2O2 | >2419 | N.D. |
| FPR–PO + MWTE + H2O2 (DC) | >2419 | 629–686 | FPR–HP + MWTE + H2O2 (DC) | >2419 | 613–689 |
| Treatment | kpo (×10−3 min−1) | Treatment | kpo (×10−3 min−1) | Treatment | KphC (×10−3 min−1) | Treatment | KphC (×10−3 min−1) |
|---|---|---|---|---|---|---|---|
| CPC–PO | 0.8 | CPC–PO + H2O2 | 2.6 | CPC–HP | 6.2 | CPC–HP + H2O2 | 6.8 |
| CPC–PO (DC) | 0.2 | CPC–PO + H2O2 (DC) | 0.8 | CPC–HP (DC) | 0.3 | CPC–HP + H2O2 (DC) | 0.8 |
| FPR–PO | 0.8 | FPR–PO + H2O2 | 2.6 | FPR–HP | 6.0 | FPR–HP + H2O2 | 6.7 |
| FPR–PO (DC) | 0.2 | FPR–PO + H2O2 (DC) | 0.8 | FPR–HP (DC) | 0.3 | FPR–HP + H2O2 (DC) | 0.8 |
| Treatment | QUV (kJ L−1) | KUV (×10−3 kJ−1 L) | Treatment | QUV (kJ L−1) | KUV (×10−3 kJ−1 L) |
|---|---|---|---|---|---|
| CPC–PO | 354.99 | 0.7 | FPR–PO | 251.03 | 1.0 |
| CPC–PO + H2O2 | 339.45 | 2.2 | FPR–PO + H2O2 | 240.04 | 3.1 |
| CPC–HP | 366.79 | 5.1 | FPR–HP | 259.38 | 7.0 |
| CPC–HP + H2O2 | 361.57 | 5.6 | FPR–HP + H2O2 | 255.69 | 7.9 |
| Treatment | ACO (m2·m−3-Order) | Treatment | ACO (m2·m−3-Order) | Efficiency (ε) |
|---|---|---|---|---|
| CPC–PO | 83.3 | FPR–PO | 121.7 | 31.5 |
| CPC–PO + H2O2 | 28.4 | FPR–PO + H2O2 | 39.2 | 27.4 |
| CPC–HP | 11.3 | FPR–HP | 15.3 | 26.4 |
| CPC–HP + H2O2 | 10.2 | FPR–HP + H2O2 | 15.0 | 32.0 |
| Source | DF | Sum of Squares (×104) | Mean Square (×104) | F-Value | p-Value Probr > F |
|---|---|---|---|---|---|
| Model | 6 | 4.7 | 0.7 | 49.61 | <0.0001 |
| AOP | 1 | 1.3 | 1.3 | 88.53 | <0.0001 |
| H2O2 addition | 1 | 0.2 | 0.2 | 13.96 | 0.0003 |
| MWTE spike | 1 | 0.0 | 0.0 | 0.01 | 0.9355 |
| Reactor employed | 1 | 0.0 | 0.0 | 0.01 | 0.9410 |
| Irradiation exposure | 1 | 3.0 | 3.0 | 195.13 | <0.0001 |
| QUV | 1 | 0.0 | 0.0 | 0.00 | 0.9662 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silerio-Vázquez, F.d.J.; Núñez-Núñez, C.M.; Proal-Nájera, J.B.; Alarcón-Herrera, M.T. Arsenite to Arsenate Oxidation and Water Disinfection via Solar Heterogeneous Photocatalysis: A Kinetic and Statistical Approach. Water 2022, 14, 2450. https://doi.org/10.3390/w14152450
Silerio-Vázquez FdJ, Núñez-Núñez CM, Proal-Nájera JB, Alarcón-Herrera MT. Arsenite to Arsenate Oxidation and Water Disinfection via Solar Heterogeneous Photocatalysis: A Kinetic and Statistical Approach. Water. 2022; 14(15):2450. https://doi.org/10.3390/w14152450
Chicago/Turabian StyleSilerio-Vázquez, Felipe de J., Cynthia M. Núñez-Núñez, José B. Proal-Nájera, and María T. Alarcón-Herrera. 2022. "Arsenite to Arsenate Oxidation and Water Disinfection via Solar Heterogeneous Photocatalysis: A Kinetic and Statistical Approach" Water 14, no. 15: 2450. https://doi.org/10.3390/w14152450
APA StyleSilerio-Vázquez, F. d. J., Núñez-Núñez, C. M., Proal-Nájera, J. B., & Alarcón-Herrera, M. T. (2022). Arsenite to Arsenate Oxidation and Water Disinfection via Solar Heterogeneous Photocatalysis: A Kinetic and Statistical Approach. Water, 14(15), 2450. https://doi.org/10.3390/w14152450






