Major, Trace and Rare Earth Element Distribution in Water, Suspended Particulate Matter and Stream Sediments of the Ob River Mouth
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sample Collection
2.3. Sample Analysis
2.4. Statistical Treatment
3. Results
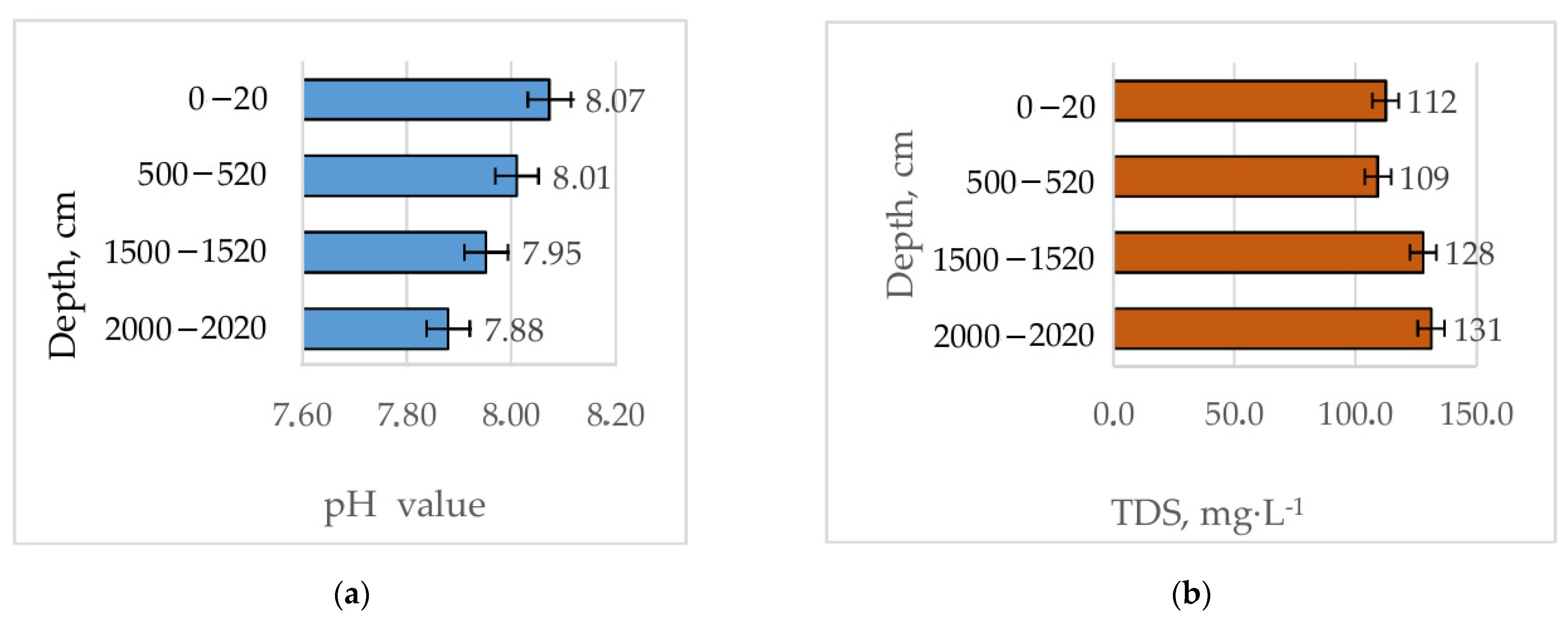
3.1. Flow Rate and Physicochemical Parameters
3.2. Elemental Composition
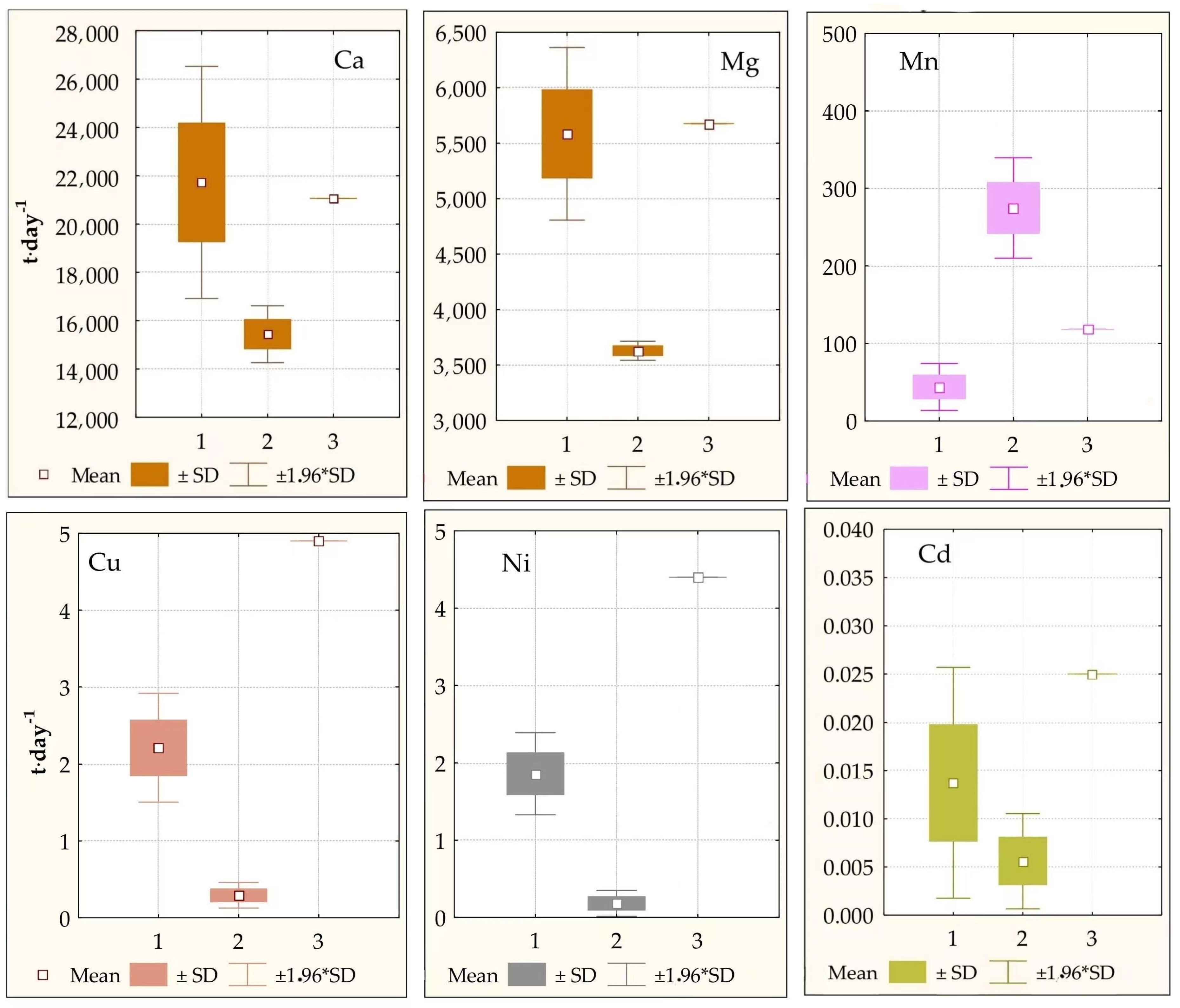
3.3. Fluxes of Elements
3.4. Seasonal Dynamics
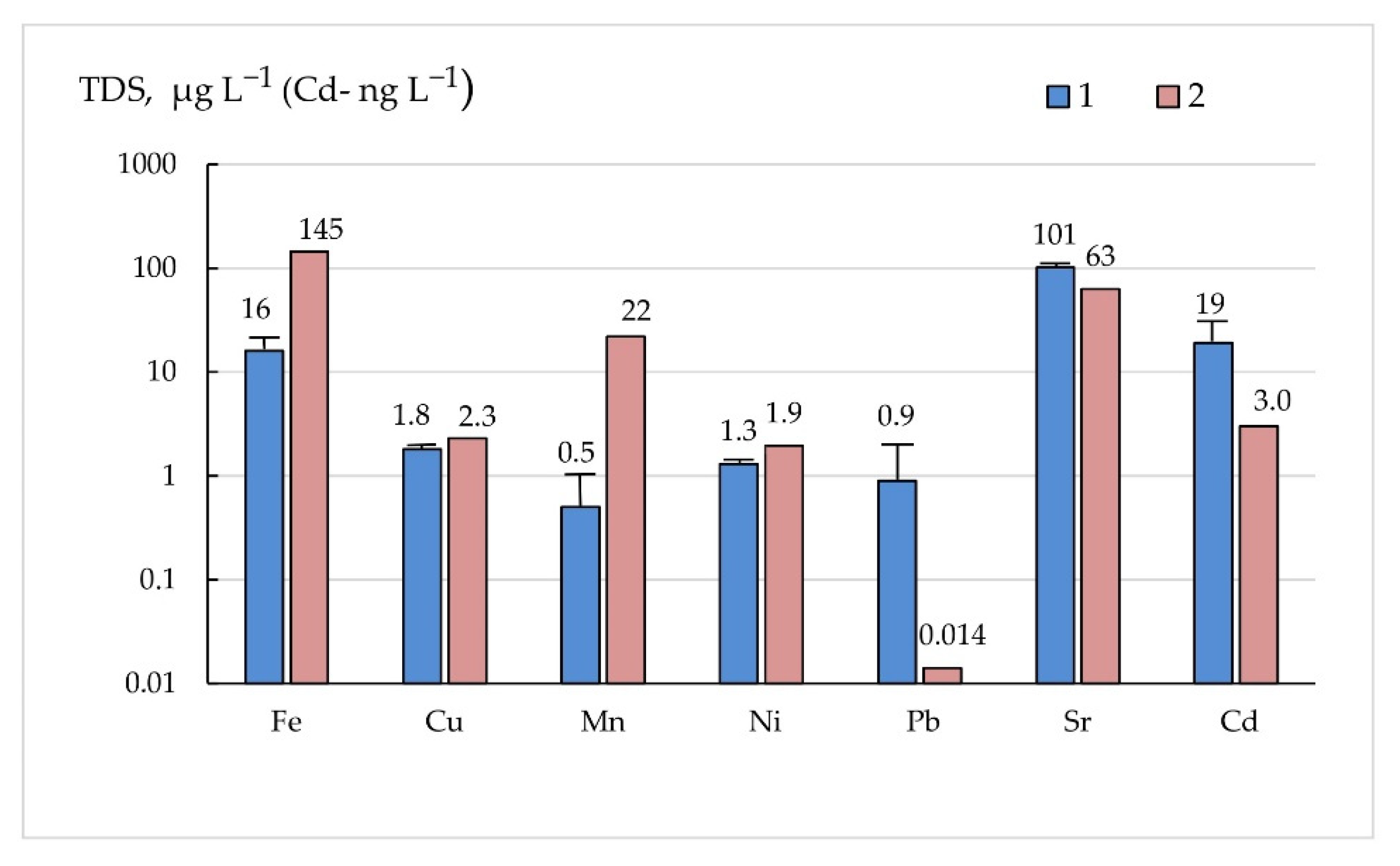
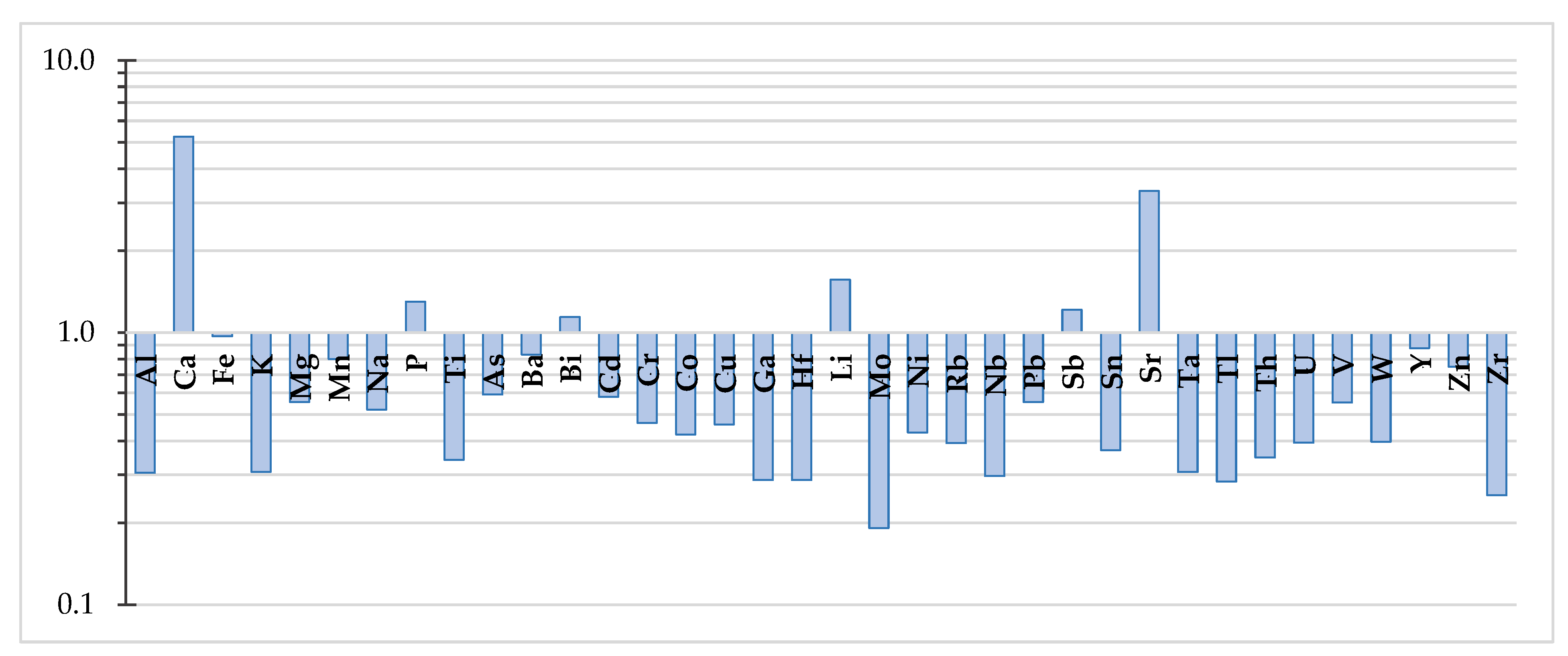
3.5. Partitioning of Elements
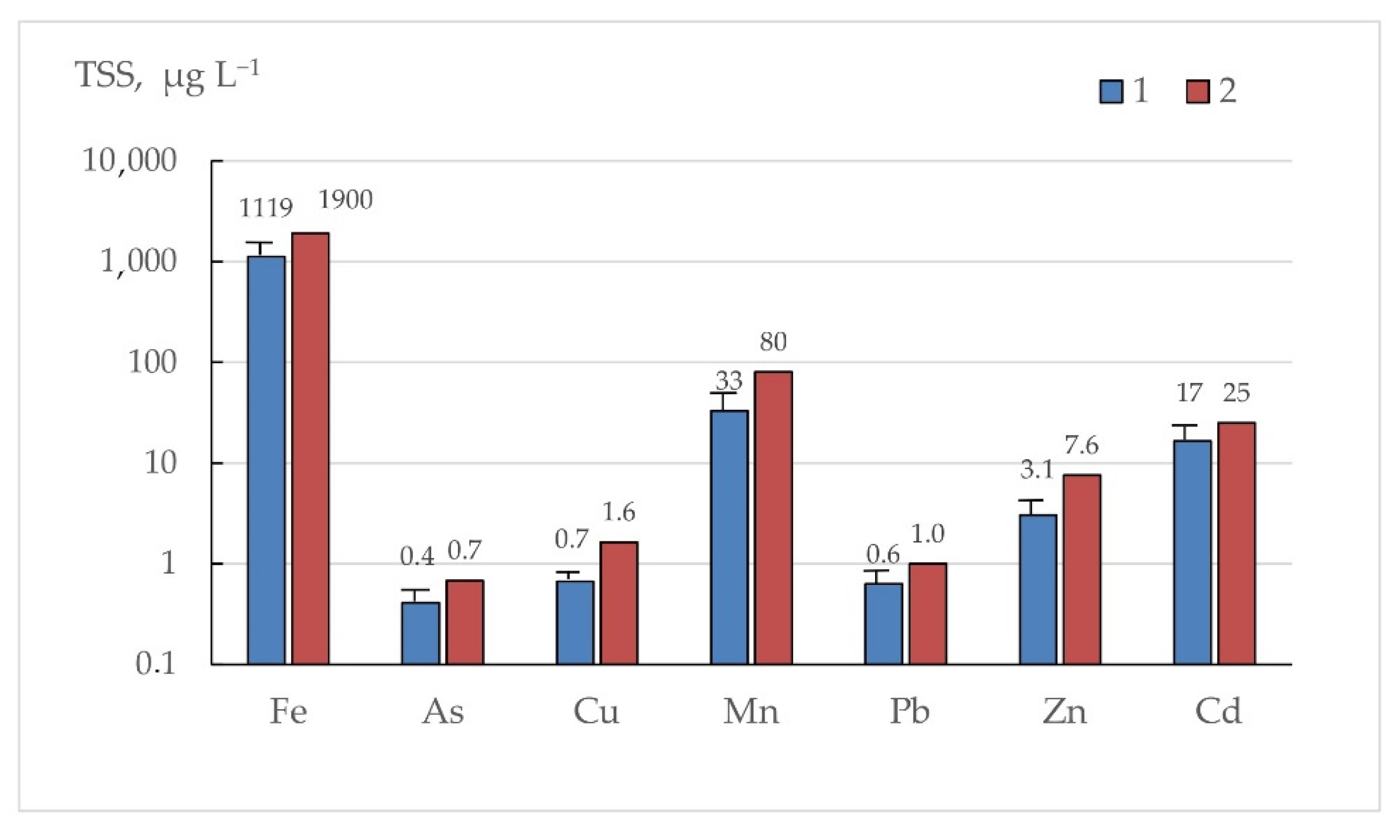
3.6. Influence of Suspended Matter on Bottom Sediment Composition
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Gordeev, V.V. Pollution of the Arctic. Reg. Environ. Change 2002, 3, 88–98. [Google Scholar] [CrossRef]
- Ilina, S.M.; Lapitskiy, S.A.; Alekhin, Y.V.; Viers, J.; Benedetti, M.; Pokrovsky, O.S. Speciation, Size Fractionation and Transport of Trace Elements in the Continuum Soil Water–Mire–Humic Lake–River–Large Oligotrophic Lake of a Subarctic Watershed. Aquat. Geochem. 2016, 22, 65–95. [Google Scholar] [CrossRef]
- Savenko, A.V.; Pokrovsky, O.S. Distribution of Dissolved Matter in the Yenisei Estuary and Adjacent Kara Sea Areas and Its Inter-Annual Variability. Geochem. Int. 2019, 57, 1201–1212. [Google Scholar] [CrossRef]
- Malkova, G.V.; Pavlov, A.V.; Skachkov, Y.B. Assessment of Permafrost Stability Under Contemporary Climatic Changes. Kriosf. Zemli 2011, 15, 29–32. (In Russian) [Google Scholar]
- Pavlov, A.V.; Malkova, G.V. Dynamic of Permafrost Zone of Russia Under Changing Climate Conditions. Kriosf. Zemli 2010, 5, 44–51. (In Russian) [Google Scholar]
- Romanovsky, V.E.; Drozdov, D.S.; Oberman, N.G.; Malkova, G.V.; Kholodov, A.L.; Marchenko, S.S.; Moskalenko, N.G.; Sergeev, D.O.; Ukraintseva, N.G.; Abramov, A.A.; et al. Thermal State of Permafrost in Russia. Permafr. Periglac. Process. 2010, 21, 136–155. [Google Scholar] [CrossRef]
- Shmakin, A.B. Climatic Characteristics of Snow Cover Over North Eurasia the Last Decades. Ice Snow. 2010, 1, 43–57. [Google Scholar]
- Vinokurov, Y.I.; Tsymbaley, Y.M.; Zinchenko, G.S.; Stoyasheva, N.V. General Characteristics of the Ob basin. In The Current State of Water Resources and the Functioning of the Water Management Complex of the Ob and Irtysh Basin; Vinokurov, Y.I., Puzanov, A.V., Bezmaternykh, D.M., Eds.; Publishing House SB RAS: Novosibirsk, Russia, 2012; pp. 9–11. (In Russian) [Google Scholar]
- Moiseenko, T.I.; Gashkina, N.A.; Dinu, M.I.; Kremleva, T.A.; Khoroshavin, V.Y. Aquatic Geochemistry of Small Lakes: Effects of Environment Changes. Geochem. Int. 2013, 51, 1031–1148. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Shirokova, L.S.; Kirpotin, S.N.; Audry, S.; Viers, J.; Dupre, B. Effect of Permafrost Thawing on Organic Carbon and Trace Element Colloidal Speciation in The Thermokarst Lakes of Western Siberia. Biogeosciences 2011, 8, 565–583. [Google Scholar] [CrossRef] [Green Version]
- Patton, A.I.; Rathburn, S.L.; Capps, D.M. Landslide Response to Climate Change in Permafrost Regions. Geomorphology 2019, 340, 116–128. [Google Scholar] [CrossRef]
- Ji, X.; Abakumov, E.; Tomashunas, V.; Polyakov, V.; Kouzov, S. Geochemical Pollution of Trace Metals in Permafrost-Affected Soil in the Russian Arctic Marginal Environment. Environ. Geochem. Health 2020, 42, 4407–4429. [Google Scholar] [CrossRef] [PubMed]
- Nikanorov, A.M.; Smirnov, M.P.; Klimenko, O.A. Long-Term Trends in Total and Anthropogenic Discharge of Organic and Biogenic Substances by Russian Rivers into the Arctic and Pacific Seas. Water. Resour. 2010, 37, 361–371. [Google Scholar] [CrossRef]
- Krickov, I.V.; Lim, A.G.; Manasypov, R.M.; Loiko, S.V.; Vorobyev, S.N.; Shevchenko, V.P.; Dara, O.M.; Gordeev, V.V.; Pokrovsky, O.S. Major and Trace Elements in Suspended Matter of Western Siberian Rivers: First Assessment Across Permafrost Zones and Landscape Parameters of Watersheds. Geochim. Cosmochim. Acta 2020, 269, 429–450. [Google Scholar] [CrossRef]
- Frey, K.E.; Siegel, D.I.; Smith, L.C. Geochemistry of West Siberian Streams and Their Potential Response to Permafrost Degradation. Water Resour. Res. 2007, 43. [Google Scholar] [CrossRef] [Green Version]
- Magritskiy, D.V.; Chalov, S.R.; Agafonova, S.A. Hydrological Regime of The Lower Ob in Modern Hydroclimatic Conditions and Under the Influence of Large-Scale Water Management. Sci. Bull. Yamalo-Nenets Auton. Okrug. 2019, 1, 106–115. (In Russian) [Google Scholar] [CrossRef]
- Neff, J.C.; Finlay, J.C.; Zimov, S.A.; Davydov, S.P.; Carrasco, J.J.; Schuur, E.A.G.; Davydova, A.I. Seasonal Changes in the Age and Structure of Dissolved Organic Carbon in Siberian Rivers and Streams. Geophys. Res. Lett. 2006, 33, 1–5. [Google Scholar] [CrossRef]
- Magritsky, D.V.; Frolova, N.L.; Evstigneev, V.M.; Povalishnikova, E.S.; Kireeva, M.B.; Pakhomova, O.M. Long-Term Changes of River Water Inflow into the Seas of the Russian Arctic Sector. Polarforschung 2017, 87, 177–194. [Google Scholar] [CrossRef]
- Gordeev, V.V.; Kochenkova, A.I.; Lokhov, A.S.; Yakovlev, A.E.; Belorukov, S.K.; Fedulov, V.Y. Seasonal and Inter Annual Variations between Concentrations and Fluxes of Dissolved and Particulate Organic Carbon, Iron and Manganese from the Northern Dvina River to the White Sea. Oceanology 2021, 61, 34–47. [Google Scholar] [CrossRef]
- Dai, M.H.; Martin, J.M. First data on the trace metal concentration level and behaviour in two major Arctic River estuarine systems Ob and Yenisey and in the adjacent Kara Sea. Earth Planet. Sci. Lett. 1995, 131, 127–141. [Google Scholar] [CrossRef]
- Galimov, E.M.; Kodina, L.A.; Stepanets, O.V.; Korobeinik, G.S. Biogeochemistry of the Russian Arctic. Kara Sea: Research Results under the SIRRO Project, 1995–2003. Geochem. Int. 2006, 44, 1053–1104. [Google Scholar] [CrossRef]
- Gordeev, V.V.; Beeskow, B.; Rachold, V. Geochemistry of the Ob and Yenisey estuaries: A comparative study. Ber. Polarforsch. Meeresforsch (Rep. Polar Mar. Res.) 2007, 565, 235. [Google Scholar]
- Demina, L.L.; Gordeev, V.V.; Galkin, S.V.; Kravchishina, M.D.; Aleksankina, S.P. The Biogeochemistry of Some Heavy Metals and Metalloids in the Ob River Estuary–Kara Sea Section. Oceanology 2010, 50, 729–742. [Google Scholar] [CrossRef]
- Savenko, A.V.; Savenko, V.S.; Pokrovsky, O.S. New Data on the Concentrations of Dissolved Trace Elements in Waters of Russian Arctic Rivers. Dokl. Earth Sci. 2020, 491, 257–263. [Google Scholar] [CrossRef]
- Huser, B.l.; Köhler, S.; Wilander, A.; Johansson, K.; Fölster, J. Temporal and Spatial Trends for Trace Metals in Streams and Rivers Across Sweden (1996–2009). Biogeosciences 2011, 8, 1813–1823. [Google Scholar] [CrossRef] [Green Version]
- Shiklomanov, I.A.; Shiklomanov, A.I.; Lammers, R.B.; Peterson, B.J.; Vorosmarty, C.J. The Dynamics of River Water Inflow to the Arctic Ocean. In The Freshwater Budget of the Arctic Ocean. NATO Science Series (Series 2. Environment Security); Lewis, E.L., Jones, E.P., Lemke, P., Prowse, T.D., Wadhams, P., Eds.; Springer: Dordrecht, The Netherlands, 2000; Volume 70. [Google Scholar] [CrossRef]
- Maslov, A.V.; Shevchenko, V.P. REE–Th Systematics of the Suspended Particulate Matter and Bottom Sediments from the Mouth Zones of the World Rivers of Different Categories/Classes and Some Large Russian Arctic Rivers. Geochem. Int. 2019, 57, 56–73. [Google Scholar] [CrossRef]
- Peregon, A.; Maksyutov, S.; Yamagata, Y. An Image-Based Inventory of the Spatial Structure of West Siberian wetlands. Environ. Res. Lett. 2009, 4, 045014. [Google Scholar] [CrossRef]
- Stepanova, V.A.; Pokrovsky, O.S.; Viers, J.; Mironycheva-Tokareva, N.P.; Kosykh, N.P.; Vishnyakova, E.K. Elemental Composition of Peat Profiles in Western Siberia: Effect of the Micro-Landscape, Latitude Position and Permafrost Coverage. Appl. Geochem. 2015, 53, 53–70. [Google Scholar] [CrossRef]
- Savichev, O.G. Geochemical Parameters of Bog Waters in the Taiga Zone of the Western Siberia. Izv. Ross. Akad. Nauk Seriya Geogr. 2015, 4, 47–57. (In Russian) [Google Scholar] [CrossRef] [Green Version]
- Revenga, C.; Murray, S.; Abramovitz, J.; Hammond, A. Watersheds of the World: Ecological Value and Vulnerability; World Resources Institute: Washington, DC, USA, 1998; p. 172. [Google Scholar]
- Yang, D.; Ye, B.; Shiklomanov, F. Discharge Characteristics and Changes Over the Ob River Watershed in Siberia. J. Hydrometeorol. 2004, 5, 595–610. [Google Scholar] [CrossRef] [Green Version]
- Shvartsev, S.I.; Savichev, O.G.; Vertman, G.O. Ecologo-Geochemical Conditions of Waters in The Middle Ob River Basin. Water Resour. 1996, 22, 723–731. (In Russian) [Google Scholar]
- Stoyashcheva, N.V.; Rybkina, I.D. Water Resources of the Ob-Irtysh River Basin and Their Use. Water Resour. 2014, 41, 1–7. [Google Scholar] [CrossRef]
- Kalinin, V.M.; Khoroshavin, V.Y.; Gerter, O.V.; Znamenshchikov, A.N.; Maltsev, A.G.; Pislegin, D.V. Practical Application of Automated Hydrological Complex for Environmental Monitoring in the Oil Fields. Tyumen State Univ. Her. 2011, 12, 150–159. (In Russian) [Google Scholar]
- Shiklomanov, A.I.; Holmes, R.M.; McClelland, J.W.; Tank, S.E.; Spencer, R.G.M. Arctic Great Rivers Observatory. Discharge Dataset, Version 20211118. 2021. Available online: https://www.arcticrivers.org/ (accessed on 8 February 2022).
- Carling, G.T.; Diaz, X.; Ponce, M.; Perez, L.; Nasimba, L.; Pazmino, E.; Rudd, A.; Merugu, S.; Fernandez, D.P.; Gale, B.K.; et al. Particulate and Dissolved Trace Element Concentrations in Three Southern Ecuador Rivers Impacted by Artisanal Gold Mining. Water Air Soil Pollut. 2013, 224, 1415. [Google Scholar] [CrossRef]
- Babushkin, A.G.; Moskovchenko, D.V.; Pikunov, S.V. Hydrochemical Monitoring of the Surface Waters of the Khanty-Mansiisk Autonomous Okrug—Yugra. Nauka Publ: Novosib. Russ. 2007, 152. (In Russian) [Google Scholar]
- Papina, T.S.; Galakhov, V.P.; Tsymbaley, Y.M. Comprehensive Assessment of Water-Resource and Water-Ecological Potential. In The Current State of Water Resources and the Functioning of the Water Management Complex of the Ob and Irtysh Basin; Vinokurov, Y.I., Puzanov, A.V., Bezmaternykh, D.M., Eds.; Publishing House SB RAS: Novosibirsk, Russia, 2012; pp. 61–92. (In Russian) [Google Scholar]
- Xiao, Y.H.; Räike, A.; Hartikainen, H.; Vähätalo, A.V. Iron as a Source of Color in River Waters. Sci. Total Environ. 2015, 536, 914–923. [Google Scholar] [CrossRef]
- Shiklomanov, I.A.; Skakalsky, B.C. Studying Water, Sediment and Contaminant Runoff of Siberian Rivers: Modern Status and Prospects. In Proceedings of the Workshop on Arctic Contamination, Anchorage, AK, USA, 2–7 May 1993; pp. 295–306. [Google Scholar]
- Demina, L.L. Quantification of the Role of Organisms in the Geochemical Migration of Trace Metals in the Ocean. Geochem. Int. 2015, 53, 224–240. [Google Scholar] [CrossRef]
- Papina, T.S. Ecological–Analytical Studies of Heavy Metals in Water Ecosystems of Ob River Basin. Ph.D. Thesis, RUDN University, Moscow, Russia, 2004. (In Russian). [Google Scholar]
- Papina, T.S.; Eirikh, A.N.; Serykh, T.G.; Dryupina, E.Y. Space and Time Regularities in the Distribution of Dissolved and Suspended Manganese Forms in Novosibirsk Reservoir Water. Water Resour. 2017, 44, 276–283. [Google Scholar] [CrossRef]
- Dinu, M.I.; Moiseyenko, T.I.; Kremleva, T.A. Effect of Humic Substance Complex Formation on Metal Migration Forms in the Natural Waters of The Northern Taiga and Forest-Steppe Zones in The Tyumen Region. Tyumen State Univ. Her. 2012, 12, 63–70. (In Russian) [Google Scholar]
- Konovalov, G.S.; Ivanova, A.A.; Kolesnikov, T.H. Dissolved and Particulate Forms of Trace Elements in The Main Rivers of the USSR. In Geochemistry of Sedimentary Rocks and Ores; Strakhov, N.M., Ed.; Nauka Publishing: Moscow, Russia, 1968; Volume 5, pp. 72–87. (In Russian) [Google Scholar]
- Gaillardet, J.; Viers, J.; Dupré, B. Trace elements in River Waters. In Treatise on Geochemistry; Drever, J.I., Holland, H.D., Turekian, K.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; pp. 225–272. [Google Scholar]
- Vinogradova, E.L. Variability of the Nutrient Runoff of the Rivers of The Arctic Basin. Oceanology 2008, 48, 483–492. [Google Scholar] [CrossRef]
- Wedepohl, K.H. Handbook of Geochemistry. v. II/2. Phosphorus (15); Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1978. [Google Scholar]
- Gordeev, V.V.; Rachold, V.; Vlasova, I.E. Geochemical Behaviour of Major and Trace Elements in Suspended Particulate Material of the Irtysh River, the Main Tributary of the Ob River, Siberia. J. Appl. Geochem. 2004, 19, 593–610. [Google Scholar] [CrossRef] [Green Version]
- Kasimov, N.S. Landscape Ecogeochemistry; IP Filimonov MV Publishing: Moscow, Russia, 2013; p. 208. (In Russian) [Google Scholar]
- Martin, J.M.; Meybeck, M. Elemental Mass-Balance of Material Carried by Major World Rivers. Mar. Chem. 1979, 7, 173–206. [Google Scholar] [CrossRef]
- Saad, M.A.H.; Hassan, E.M. Heavy Metals in the Rosetta Estuary of the Nile and the Adjoining Mediterranean Waters: Evidence of Removal of Dissolved Heavy Metals from Waters as A Result of Possible Binding to Suspended Matter. Hydrobiologia 2002, 469, 131–147. [Google Scholar] [CrossRef]
- Lisitsyn, A.P. Marginal Filter of Oceans. Oceanologiya 1994, 34, 735–747. (In Russian) [Google Scholar]
- Gubar'kov, A.A.; Leibman, M.O. Bead-Shaped Channel Forms as Evidence of Paragenesis of Cryogenic and Hydrological Processes in the Small-River Valleys of Central Yamal.46. Kriosf. Zemli 2010, 14, 41–49. (In Russian) [Google Scholar]
- Opekunova, M.G.; Opekunov, A.Y.; Kukushkin, S.Y.; Ganul, A.G. Background Contents of Heavy Metals in Soils and Bottom Sediments in the North of Western Siberia. Eurasian Soil Sci. 2019, 52, 380–395. [Google Scholar] [CrossRef]
- Semenova, L.A.; Aleksyuk, V.A. Zooplankton of the lower Ob River. Bull. Ecol. For. Landsc. Stud. 2010, 10, 156–169. (In Russian) [Google Scholar]
- Viers, J.; Dupre, B.; Gaillardet, J. Chemical Composition of Suspended Sediments in World Rivers: New Insights from A New Database. Sci. Total Environ. 2009, 407, 853–868. [Google Scholar] [CrossRef]
- Kruopiene, J. Distribution of Heavy Metals in Sediments of the Nemunas River (Lithuania). Pol. J. Environ. Stud. 2007, 16, 715–722. [Google Scholar] [CrossRef]
- Martincic, D.; Kwokal, Z.; Branica, M. Distribution of Zinc, Lead, Cadmium and Copper Between Different Size Fractions of Sediments I. The Limski Kanal (North Adriatic Sea). Sci. Total Environ. 1990, 95, 201–215. [Google Scholar] [CrossRef]
- Moskovchenko, D.V.; Romanenko, E.A. Elemental Composition of Soils of The Pur-Taz Interfluve. Dokuchaev Soil Bull. 2020, 103, 51–84. (In Russian) [Google Scholar] [CrossRef]
- Song, H.; Liu, J.; Yin, P.; Zhang, Y. Distribution, Enrichment and Source of Heavy Metals in Rizhao Offshore Area, Southeast Shandong Province. Mar. Pollut. Bull. 2017, 119, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yin, P.; Chen, X.; Cao, K. Distribution, Enrichment and Transport of Trace Metals in Sediments from the Dagu River Estuary in the Jiaozhou Bay, Qingdao, China. Minerals 2019, 9, 545. [Google Scholar] [CrossRef] [Green Version]
- Sutherland, R. Bed Sediment-Associated Trace Metals in An Urban Stream, Oahu, Hawaii. Environ. Geol. 2000, 39, 611–627. [Google Scholar] [CrossRef]
- Moskovchenko, D.V. Biogeochemical Properties of the High Bogs in Western Siberia. Geogr. Nat. Resour. 2006, 1, 63–70. (In Russian) [Google Scholar]
- Gonzalez, A.G.; Pokrovsky, O.S. Metal Adsorption on Mosses: Towards A Universal Adsorption Model. J. Colloid. Interface Sci. 2014, 415, 169–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Index | August 2020 (n = 60) April 2021 (n = 30) | |
|---|---|---|
| pH | 8.01 (7.52–8.87) | 6.97 (6.90–7.00) |
| TDS, mg L−1 | 116.0 (50–307) | 190.7 (186–194) |
| TSM, mg L−1 | 21 (13–34) | NM |
| Color index | 87.6 (65–93) | 29.9 (28.8–30.7) |
| EC, µS | 151 (143–158) | 251 (242–253) |
| DO mg O2 L−1 | 8.7(7.8–9.3) | 6.2 (4.2–9.9) |
| Element | August 2020 (n = 60) | March–April 2021 (n = 30) | ||||
|---|---|---|---|---|---|---|
| Mean | SD | Fluxes, t·day−1 | Mean | SD | Fluxes, t·day−1 | |
| Macroelements, µg L−1 | ||||||
| Ca | 19,141 | 1108 | 21,995 | 35,672 | 295 | 15,318 |
| Fe | 762 | 159 | 876 | 3030 | 431.5 | 1301 |
| K | 920 | 45.4 | 1057 | 1194 | 29.1 | 513 |
| Mg | 4859 | 348 | 5584 | 8386 | 76.2 | 3601 |
| Na | 6831 | 468 | 7850 | 11,921 | 253 | 5119 |
| P | 75 | 12.6 | 86 | 102 | 14.0 | 44 |
| Si | 2811 | 255 | 3230 | 7840 | 849 | 3367 |
| Trace metals and metalloids, µg L−1 | ||||||
| As | 1.58 | 0.19 | 1.8 | 1.42 | 0.07 | 0.61 |
| Ba | 22.2 | 3.53 | 25.5 | 34.2 | 0.95 | 14.7 |
| Be | 0.0156 | 0.0063 | 0.018 | 0.0017 | 0.0040 | 0.0007 |
| Bi | 0.0054 | 0.0017 | 0.006 | 0.0014 | 0.0003 | 0.0006 |
| Cd | 0.012 | 0.0057 | 0.013 | 0.013 | 0.0049 | 0.0056 |
| Co | 0.17 | 0.07 | 0.20 | 0.85 | 0.06 | 0.36 |
| Cr | <DL | - | - | 0.11 | 0.58 | 0.05 |
| Cu | 1.94 | 0.28 | 2.2 | 0.68 | 0.15 | 0.29 |
| Li | 2.6 | 0.17 | 3.0 | 3.9 | 0.039 | 1.7 |
| Mn | 37.9 | 13.4 | 43.6 | 645 | 54.4 | 277 |
| Mo | 0.37 | 0.028 | 0.43 | 0.39 | 0.019 | 0.17 |
| Ni | 1.62 | 0.24 | 1.9 | 0.39 | 0.31 | 0.17 |
| Rb | 0.77 | 0.049 | 0.88 | 0.82 | 0.02 | 0.35 |
| Pb | 0.39 | 0.09 | 0.45 | 0.15 | 0.06 | 0.06 |
| Sb | 0.094 | 0.027 | 0.11 | 0.061 | 0.018 | 0.03 |
| Sr | 114 | 6.12 | 131 | 206 | 1.83 | 88.5 |
| V | 1.58 | 0.25 | 1.8 | 0.67 | 0.05 | 0.29 |
| Zn | 2.25 | 0.93 | 2.6 | 1.29 | 0.51 | 0.55 |
| Rare earth elements, ng L−1 | ||||||
| Ce | 644.9 | 299.3 | 0.74 | 263.5 | 21.4 | 0.11 |
| Dy | 65.4 | 15.5 | 0.08 | 28.2 | 2.1 | 0.012 |
| Er | 34.9 | 7.9 | 0.040 | 16.2 | 1.0 | 0.007 |
| Eu | 17.3 | 4.4 | 0.020 | 6.4 | 0.6 | 0.003 |
| Gd | 80.9 | 19.6 | 0.093 | 34.0 | 2.3 | 0.015 |
| Ho | 12.8 | 2.9 | 0.015 | 5.7 | 0.4 | 0.002 |
| La | 346.7 | 155.1 | 0.40 | 118.0 | 9.4 | 0.051 |
| Nd | 368.8 | 93.8 | 0.42 | 136.9 | 9.8 | 0.059 |
| Pr | 85.6 | 21.8 | 0.10 | 30.6 | 2.0 | 0.013 |
| Sm | 77.8 | 19.4 | 0.09 | 30.5 | 1.7 | 0.013 |
| Tb | 11.8 | 2.8 | 0.014 | 4.9 | 0.4 | 0.002 |
| Tm | 4.6 | 1.0 | 0.005 | 2.3 | 0.2 | 0.001 |
| Y | 334.5 | 73.6 | 0.38 | 145.8 | 8.3 | 0.063 |
| Yb | 29.4 | 6.5 | 0.034 | 15.6 | 1.1 | 0.007 |
| Element | TSM | TDM | WA RDM | AEA RDM | Ratio TSM/(TSM + TDM) | ||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||||
| Macroelements, µg L−1 | |||||||
| Al | 586 | 375 | 13.3 | 8.9 | 32 | 78 | 98 |
| Ca | 3020 | 1741 | 15,332 | 2553 | 14,600 | - | 16 |
| Fe | 1119 | 380 | 16.0 | 12.3 | 66 | 271 | 99 |
| K | 115 | 73 | 930 | 37 | 1350 | - | 11 |
| Mg | 152 | 78 | 4887 | 91 | 3800 | - | 3 |
| Na | 79.0 | 38.9 | 7096 | 203 | 5100 | - | 1 |
| P | 51.1 | 11.1 | 22.8 | 9.2 | 9 | 12 | 69 |
| Si | 299 | 168 | 2510 | 85 | 4070 | 2540 | 11 |
| Ti | 31.6 | 18.8 | <DL | - | 0.489 | 0.0012 | ND |
| Trace metals and metalloids, µg L−1 | |||||||
| As | 0.41 | 0.12 | 1.1 | 0.06 | 0.62 | 0.77 | 27 |
| Ba | 9.2 | 3.89 | 13.4 | 2.3 | 23 | 14.4 | 41 |
| Co | 0.20 | 0.11 | <DL | - | 0.148 | 0.073 | ND |
| Cr | 1.21 | 0.44 | <DL | - | 0.7 | 0.47 | ND |
| Cu | 0.67 | 0.14 | 1.8 | 0.21 | 1.48 | 1.29 | 27 |
| Ga | 0.11 | 0.07 | <DL | - | 0.03 | 0.025 | ND |
| Li | 0.30 | 0.18 | 2.69 | 0.11 | 1.84 | 2.92 | 10 |
| Mn | 32.6 | 15.3 | 0.5 | 0.38 | 34 | 18.7 | 98 |
| Nb | 0.09 | 0.06 | <DL | - | 0.017 | - | ND |
| Ni | 0.66 | 0.29 | 1.3 | 0.14 | 0.801 | 0.89 | 34 |
| Pb | 0.63 | 0.25 | 0.9 | 1.1 | 0.079 | 0.118 | 34 |
| Rb | 0.67 | 0.42 | 0.75 | 0.19 | 1.63 | 1.14 | 47 |
| Sr | 13.7 | 7.6 | 101.3 | 10.7 | 60 | 149 | 12 |
| V | 0.39 | 0.14 | 0.8 | 0.1 | 0.71 | 0.82 | 33 |
| Zn | 3.05 | 1.25 | <DL | - | 0.6 | 4.2 | ND |
| Zr | 0.86 | 0.50 | 0.06 | 0.02 | 0.039 | 0.176 | 93 |
| ng L−1 | |||||||
| Cd | 16.5 | 7.0 | 19.0 | 11.3 | 80 | 15 | 46 |
| Hg | 0.9 | 0.3 | <DL | - | - | - | ND |
| Mo | 11.0 | 2.4 | 394 | 18 | 420 | 270 | 3 |
| Sb | 45.0 | 34.0 | 193 | 53 | 70 | 72 | 19 |
| Th | 88.3 | 46.8 | 1.8 | 0.3 | 41 | 26 | 98 |
| U | 25.9 | 10.0 | 277 | 33 | 372 | 145 | 9 |
| W | 16.3 | 7.7 | 59 | 147 | 100 | 12 | 22 |
| Rare earth elements, ng L−1 | |||||||
| Ce | 803.5 | 368.5 | 13.3 | 8.6 | 262 | 328 | 98 |
| Dy | 69.1 | 26.5 | 2.4 | 1.1 | 30 | 33 | 97 |
| Er | 38.7 | 14.5 | 2.3 | 1.0 | 20 | 19 | 94 |
| Gd | 84.4 | 32.6 | 1.9 | 0.8 | 40 | 44 | 98 |
| La | 417.7 | 173.8 | 8.3 | 5.4 | 120 | 177 | 98 |
| Nd | 410.0 | 166.0 | 6.7 | 4.6 | 152 | 213 | 98 |
| Pr | 97.8 | 40.4 | 1.8 | 0.9 | 28 | 50 | 98 |
| Sc | 120 | 70 | <DL | - | 1200 | - | ND |
| Tb | 11.9 | 4.5 | 1.1 | 0.4 | 5.5 | 6 | 92 |
| Ho | 13.5 | 5.2 | 0.9 | 0.3 | 7 | 6.3 | 94 |
| Sm | 89.1 | 35.6 | 1.8 | 0.8 | 36 | 44 | 98 |
| Yb | 34.0 | 13.8 | 1.8 | 0.8 | 17 | 12 | 95 |
| Elements | Suspended Solids | Bottom Sediments | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | EF | WARSM | AEA RSM | Mean | SD | EF | |
| Macroelements, % | ||||||||
| Al | 2.70 | 0.95 | 1.0 | 8.7 | 5.9 | 6.09 | 1.03 | 1.0 |
| Ca | 13.6 | 6.0 | 16.1 | 2.59 | 1.28 | 0.97 | 0.11 | 0.5 |
| Fe | 5.62 | 1.92 | 4.3 | 5.8 | 8.0 | 3.18 | 0.44 | 1.1 |
| K | 0.52 | 0.17 | 0.7 | 1.69 | 1.34 | 1.74 | 0.05 | 1.0 |
| Mg | 0.70 | 0.09 | 1.4 | 1.26 | 0.93 | 0.69 | 0.16 | 0.6 |
| Mn | 0.16 | 0.05 | 6.2 | 0.2 | 0.22 | 0.15 | 0.10 | 2.5 |
| Na | 0.37 | 0.09 | 0.5 | 0.71 | 0.63 | 1.07 | 0.15 | 0.6 |
| P | 0.26 | 0.09 | 12.1 | 0.2 | 0.41 | 0.074 | 0.020 | 1.5 |
| Ti | 0.15 | 0.05 | 1.2 | 0.44 | 0.46 | 0.43 | 0.09 | 1.4 |
| Trace metals and metalloids, µg g−1 | ||||||||
| As | 21.5 | 10.7 | 13.5 | 36.3 | 36 | 6.95 | 4.00 | 1.9 |
| Bi | 0.97 | 1.93 | 18.4 | 0.85 | - | 0.14 | 0.03 | 1.1 |
| Cd | 0.90 | 0.64 | 30.3 | 1.55 | 0.55 | 0.16 | 0.07 | 2.4 |
| Cr | 60.6 | 19.9 | 2.0 | 130 | 108 | 60.5 | 11.0 | 0.9 |
| Co | 9.5 | 3.55 | 1.7 | 22.5 | 24 | 20.4 | 9.47 | 1.6 |
| Cu | 34.9 | 13.3 | 3.8 | 75.9 | 46 | 16.5 | 3.18 | 0.8 |
| Ga | 5.2 | 1.97 | 0.9 | 18.1 | - | 11.9 | 1.67 | 0.9 |
| Hf | 1.16 | 0.35 | 0.66 | 4.04 | - | 2.33 | 0.46 | 0.6 |
| Hg | NM | NM | - | - | 0.05 | 0.07 | 0.01 | 1.8 |
| Li | 13.3 | 3.4 | 1.9 | 8.5 | - | 20.3 | 3.9 | 1.3 |
| Ni | 32.0 | 11.0 | 2.1 | 74.5 | 52 | 39.2 | 8.33 | 1.1 |
| Rb | 30.8 | 10.6 | 1.1 | 78.5 | - | 70.2 | 5.87 | 1.1 |
| Nb | 4.01 | 1.45 | 1.0 | 13.5 | - | 8.55 | 1.25 | 1.0 |
| Pb | 33.9 | 23.2 | 6.0 | 61.1 | 34 | 14.8 | 1.6 | 1.2 |
| Sb | 2.66 | 3.41 | 20.1 | 2.19 | - | 0.63 | 0.04 | 2.1 |
| Sr | 620 | 236 | 5.9 | 187 | 145 | 170 | 13 | 0.7 |
| Ta | 0.39 | 0.39 | 1.3 | 1.27 | - | 0.62 | 0.09 | 0.9 |
| Tl | 0.15 | 0.05 | 0.51 | 0.53 | - | 0.31 | 0.02 | 0.5 |
| Th | 4.20 | 1.37 | 1.21 | 12.1 | - | 6.82 | 1.05 | 0.9 |
| U | 1.30 | 0.55 | 1.5 | 3.3 | - | 1.57 | 0.22 | 0.8 |
| V | 71.4 | 26.0 | 2.2 | 129 | 134 | 89.5 | 13.8 | 1.2 |
| Zn | 156 | 82.3 | 7.0 | 208 | 263 | 59.0 | 11.4 | 1.2 |
| Zr | 40.4 | 14.0 | 0.6 | 160 | 118 | 90.1 | 17.0 | 0.6 |
| Rare earth elements, µg g−1 | ||||||||
| Ce | 38.9 | 12.3 | 1.9 | 73.6 | 32 | 60.3 | 6.8 | 1.3 |
| Eu | 1.00 | 0.31 | 3.0 | 1.29 | 0.86 | 1.03 | 0.08 | 1.4 |
| Gd | 4.18 | 1.40 | 3.2 | 5.25 | 3.3 | 4.03 | 0.31 | 1.3 |
| La | 20.5 | 6.6 | 2.0 | 37.4 | 21 | 26.7 | 2.3 | 1.2 |
| Lu | 0.24 | 0.08 | 2.3 | 0.35 | 0.29 | 0.27 | 0.02 | 1.2 |
| Nd | 20.2 | 6.6 | 2.3 | 32.2 | 9.5 | 24.1 | 1.9 | 1.2 |
| Pr | 4.80 | 1.54 | 2.0 | 7.95 | - | 6.21 | 0.53 | 1.2 |
| Sc | 5.45 | 1.86 | 1.2 | 18.2 | - | 10.2 | 1.85 | 1.0 |
| Sm | 4.40 | 1.47 | 2.8 | 6.12 | 1.7 | 4.82 | 0.36 | 1.4 |
| Y | 19.2 | 6.12 | 2.8 | 21.9 | - | 18.3 | 1.40 | 1.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soromotin, A.; Moskovchenko, D.; Khoroshavin, V.; Prikhodko, N.; Puzanov, A.; Kirillov, V.; Koveshnikov, M.; Krylova, E.; Krasnenko, A.; Pechkin, A. Major, Trace and Rare Earth Element Distribution in Water, Suspended Particulate Matter and Stream Sediments of the Ob River Mouth. Water 2022, 14, 2442. https://doi.org/10.3390/w14152442
Soromotin A, Moskovchenko D, Khoroshavin V, Prikhodko N, Puzanov A, Kirillov V, Koveshnikov M, Krylova E, Krasnenko A, Pechkin A. Major, Trace and Rare Earth Element Distribution in Water, Suspended Particulate Matter and Stream Sediments of the Ob River Mouth. Water. 2022; 14(15):2442. https://doi.org/10.3390/w14152442
Chicago/Turabian StyleSoromotin, Andrei, Dmitriy Moskovchenko, Vitaliy Khoroshavin, Nikolay Prikhodko, Alexander Puzanov, Vladimir Kirillov, Mikhail Koveshnikov, Eugenia Krylova, Aleksander Krasnenko, and Aleksander Pechkin. 2022. "Major, Trace and Rare Earth Element Distribution in Water, Suspended Particulate Matter and Stream Sediments of the Ob River Mouth" Water 14, no. 15: 2442. https://doi.org/10.3390/w14152442






