Impact of a Dinophysis acuminata Bloom on the Copepod Acartia clausi: First Indications
Abstract
:1. Introduction
2. Materials and Methods
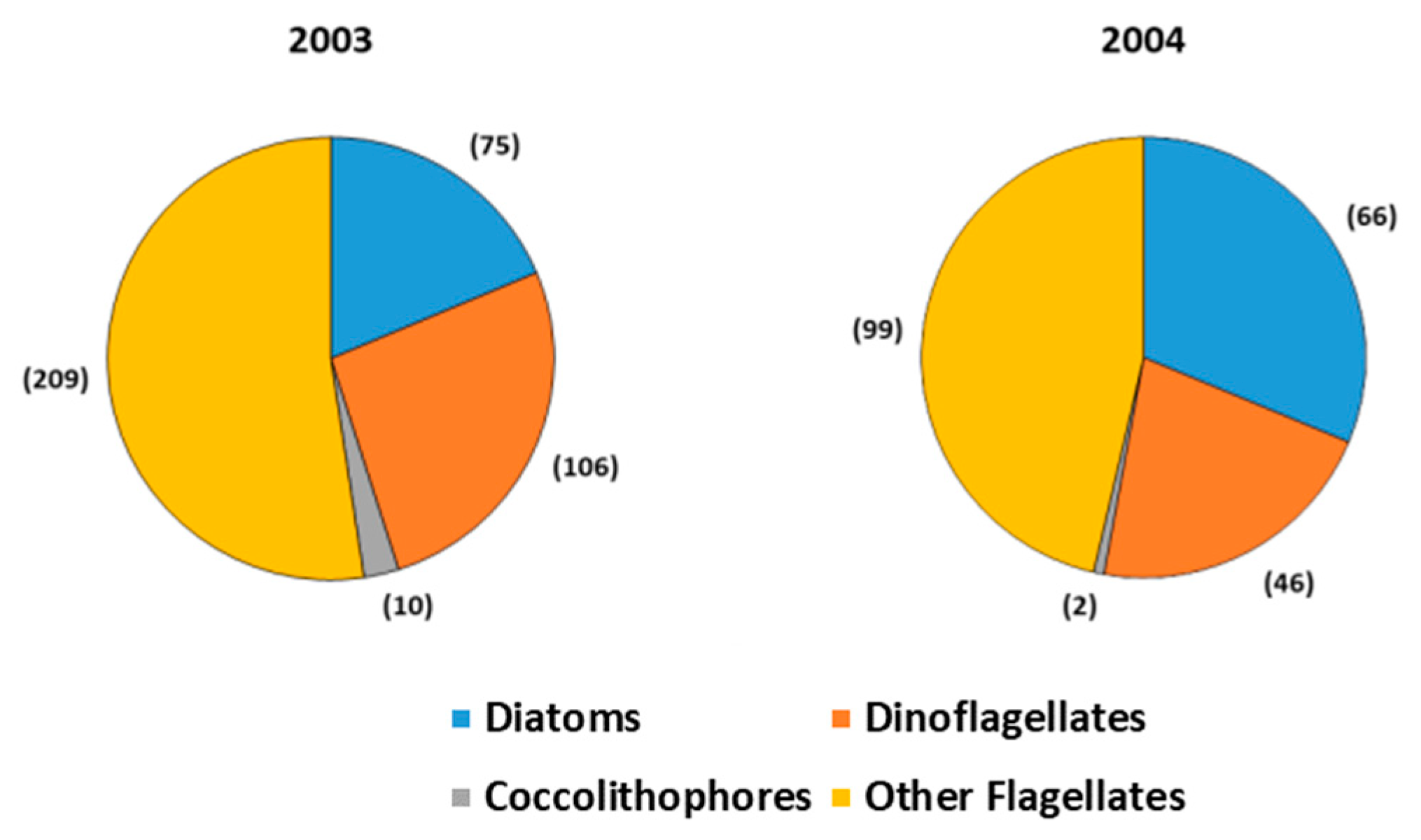
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Karageorgis, A.P.; Skourtos, M.S.; Kapsimalis, V.; Kontogianni, A.D.; Skoulikidis, N.T.; Pagou, K.; Nikolaidis, N.P.; Drakopoulou, P.; Zanou, B.; Karamanos, H.; et al. An integrated approach to watershed management within the DPSIR framework: Axios River catchment and Thermaikos Gulf. Reg. Environ. Chang. 2005, 5, 138–160. [Google Scholar] [CrossRef]
- Hayashi, T.; Shimizu, Y.; White, A.W. Toxin profile of herbivorous zooplankton during a Gonyaulax bloom in the Bay of Fundy. Bull. Jap. Soc. Sci. Fish. 1982, 48, 1673. [Google Scholar] [CrossRef] [Green Version]
- Turner, J.T.; Doucette, G.J.; Powell, C.L.; Kulis, D.M.; Keafer, B.A.; Anderson, D.M. Accumulation of red tide toxins in larger size fractions of zooplankton assemblages from Massachusetts Bay, USA. Mar. Ecol. Prog. Ser. 2000, 203, 95–107. [Google Scholar] [CrossRef] [Green Version]
- Frangopulos, M.; Guisande, C.; Maneiro, I.; Riveiro, I.; Franco, J. Short-term and long-term effects of the toxic dinoflagellate Alexandrium minutum on the copepod Acartia clausi. Mar. Ecol. Prog. Ser. 2000, 203, 161–169. [Google Scholar] [CrossRef] [Green Version]
- Maneiro, I.; Frangopulos, M.; Guisande, C.; Fernandez, M.; Reguera, B.; Riveiro, I. Zooplankton as a potential vector of diarrhetic shellfish poisoning toxins through the food web. Mar. Ecol. Prog. Ser. 2000, 201, 155–163. [Google Scholar] [CrossRef] [Green Version]
- Riser, C.W.; Jansen, S.; Bathmann, U.; Wassmann, P. Grazing of Calanus helgolandicus on Dinophysis norvegica during bloom conditions in the North Sea: Evidence from investigations of faecal pellets. Mar. Ecol. Prog. Ser. 2003, 256, 301–304. [Google Scholar] [CrossRef]
- Turner, J.T. Zooplankton fecal pellets, marine snow and sinking phytoplankton blooms. Aquat. Microb. Ecol. 2002, 27, 57–102. [Google Scholar] [CrossRef] [Green Version]
- Frangoulis, C.; Christou, E.D.; Hecq, J.H. Comparison of marine copepod outfluxes: Nature, rate, fate and role in the carbon, and nitrogen cycles. Adv. Mar. Biol. 2005, 47, 251–307. [Google Scholar] [CrossRef]
- Teegarden, G.J.; Campbell, R.G.; Durbin, E.G. Zooplankton feeding behavior and particle selection in natural plankton assemblages containing toxic Alexandrium spp. Mar. Ecol. Prog. Ser. 2001, 218, 213–226. [Google Scholar] [CrossRef] [Green Version]
- Turner, J.T.; Tester, P.A. Toxic marine phytoplankton, zooplankton grazers, and pelagic food webs. Limnol. Oceanogr. 2002, 47, 1430–1437. [Google Scholar] [CrossRef] [Green Version]
- Ives, J.D. Possible mechanisms underlying copepod grazing responses to levels of toxicity in red tide dinoflagellates. J. Exp. Mar. Biol. Ecol. 1987, 112, 131–145. [Google Scholar] [CrossRef]
- Uye, S.; Takamatsu, K. Feeding interactions between planktonic copepods and red tide flagellates from Japanese coastal waters. Mar. Ecol. Prog. Ser. 1990, 59, 97–107. [Google Scholar] [CrossRef]
- Teegarden, G.J. Copepod grazing selection and particle discrimination on the basis of PSP toxin content. Mar. Ecol. Prog. Ser. 1999, 181, 163–176. [Google Scholar] [CrossRef]
- Colin, S.P.; Dam, H.G. Effects of the toxic dinoflagellate Alexandrium fundyense on the copepod Acartia hudsonica: A test of the mechanisms that reduce ingestion rates. Mar. Ecol. Prog. Ser. 2003, 248, 55–65. [Google Scholar] [CrossRef]
- Ianora, A.; Poulet, S.A. Egg viability in the copepod Temora stylifera. Limnol. Oceanogr. 1993, 38, 1615–1626. [Google Scholar] [CrossRef]
- Laabir, M.; Poulet, S.A.; Ianora, A. Measuring production and viability of eggs in Calanus helgolandicus. J. Plankton Res. 1995, 17, 1125–1142. [Google Scholar] [CrossRef]
- Uye, S. Induction of reproductive failure in the planktonic copepod Calanus pacificus by diatoms. Mar. Ecol. Prog. Ser. 1996, 133, 89–97. [Google Scholar] [CrossRef]
- Ban, S.; Burns, C.; Castel, J.; Chaudron, Y.; Christou, E.D.; Escribano, R.; Umani, S.F.; Gasparini, S.; Ruiz, F.G.; Hoffmeyer, M.; et al. The paradox of diatom-copepod interactions. Mar. Ecol. Prog. Ser. 1997, 157, 287–293. [Google Scholar] [CrossRef]
- Jansen, S.; Riser, C.W.; Wassmann, P.; Bathmann, U. Copepod feeding behaviour and egg production during a dinoflagellate bloom in the North Sea. Harmful Algae 2006, 5, 102–112. [Google Scholar] [CrossRef] [Green Version]
- Delgado, M.; Garces, E.; Camp, J. Growth and behaviour of Dinophysis sacculus from NW Mediterranean. In Harmful and Toxic Algal Blooms; Yasumoto, T., Oshima, Y., Fukuyo, Y., Eds.; Intergovernmental Oceanographic Commission of UNE: Paris, France, 1996; pp. 261–264. [Google Scholar]
- Lee, J.S.; Igarashi, T.; Fraga, S.; Dahl, E.; Hovgaard, P.; Yasumoto, Y. Determination of diarrhetic shellfish toxins in various dinoflagellate species. J. Appl. Phycol. 1989, 1, 147–152. [Google Scholar] [CrossRef]
- Morlaix, M.; Lassus, P. Nitrogen and Phosphorus Effects Upon Division Rate and Toxicity of Prorocentrum lima (Ehrenberg) Dodge. Cryptogam. Algol. 1992, 13, 187–195. [Google Scholar]
- Sohet, K.; Pereira, A.; Braekman, J.C.; Houvenaghel, G. Growth and toxicity of Prorocentrum lima (Ehrenberg) Dodge in different culture media: Effect of humic acids and organic phosphorus. In Harmful Marine Algal Blooms; Lassus, P., Arzul, G., Erard-Le Denn, E., Gentien, P., Marcaillou-Le Baut, C., Eds.; Lavoisier/Intercept: Paris, France, 1995; pp. 669–674. [Google Scholar]
- Pan, Y.; Cembella, A.D.; Quilliam, M.A. Cell cycle and toxin production in the benthic dinoflagellate Prorocentrum lima. Mar. Biol. 1999, 134, 541–549. [Google Scholar] [CrossRef]
- Carlsson, P.; Granéli, E.; Finenko, G.; Maestrini, S. Copepod grazing on a phytoplankton community containing the toxic dinoflagellate Dinophysis acuminata. J. Plankton Res. 1995, 17, 1925–1938. [Google Scholar] [CrossRef]
- Maneiro, I.; Guisande, C.; Frangopulos, M.; Riveiro, I. Importance of copepod faecal pellets to the fate of the DSP toxins produced by Dinophysis spp. Harmful Algae 2002, 1, 333–341. [Google Scholar] [CrossRef]
- Turner, J.T.; Anderson, D.M. Zooplankton grazing during dinoflagellate blooms in Cape Cod embayment, with observations of predation upon tintinnids by copepods. Mar. Ecol. 1983, 4, 359–374. [Google Scholar] [CrossRef]
- Koukaras, K.; Nikolaidis, G. Dinophysis blooms in Greek coastal waters (Thermaikos Gulf, NW Aegean Sea). J. Plankton Res. 2004, 26, 445–457. [Google Scholar] [CrossRef] [Green Version]
- Pagou, K. Eutrophication in Hellenic coastal areas. In State of the Hellenic Marine Environment; Papathanassiou, E., Zenetos, A., Eds.; HCMR, Institute of Oceanography: Athens, Greece, 2005; pp. 311–317. [Google Scholar]
- Holm-Hansen, O.; Lorenzen, C.J.; Holmes, R.W.; Strickland, J.D.H. Fluorometric determination of chlorophyll. ICES J. Mar. Sci. 1965, 30, 3–15. [Google Scholar] [CrossRef]
- Utermohl, H. Zur vervollkommnung der quantitativen Phytoplankton Methodik. Mitt. Int. Ver. Theor. Angew. Limnol. 1958, 9, 1–38. [Google Scholar] [CrossRef]
- Granéli, E.; Anderson, D.M.; Carlsson, P.; Maestrini, S.Y. Light and dark carbon uptake by Dinophysis species in comparison to other photosynthetic and heterotrophic dinoflagellates. Aquat. Microb. Ecol. 1997, 13, 177–186. [Google Scholar] [CrossRef]
- Richardson, K.; Heath, M.R.; Pedersen, S.M. Studies of a larval herring (Clupea harengus L.) patch in the Buchan area. III. Phytoplankton distribution and primary productivity in relation to hydrographic features. Dana 1986, 6, 25–35. [Google Scholar]
- Booth, B.C.; Lewin, J.; Postel, J.R. Temporal variation in the structure of autotrophic and heterotrophic communities in the Subarctic Pacific. Prog. Oceanogr. 1993, 32, 57–99. [Google Scholar] [CrossRef]
- Zhou, M.; Li, J.; Luckas, B.; Yu, R.; Yan, T.; Hummert, C.; Kastrup, S. A recent shellfish toxin investigation in China. Mar. Pollut. Bull. 1999, 39, 331–334. [Google Scholar] [CrossRef]
- Nikolaidis, G.; Koukaras, K.; Aligizaki, K.; Heracleous, A.; Kalopesa, E.; Moschandreou, K.; Tsolaki, E.; Mantoudis, A. Harmful microalgal episodes in Greek coastal waters. J. Biol. Res. 2005, 3, 77–85. [Google Scholar]
- Siokou-Frangou, I.; Papathanassiou, E. Differentiation of zooplankton populations in a polluted area. Mar. Ecol. Prog. Ser. 1991, 76, 41–51. [Google Scholar] [CrossRef]
- Siokou-Frangou, I.; Shinganova, T.; Christou, E.D.; Kamburska, L.; Gubanova, A.; Konsulov, A.; Musaeva, E.; Skryabin, V.; Khoroshilov, V. Mesozooplankton communities in the Aegean and Black Seas: A comparative study. Mar. Biol. 2004, 144, 1111–1126. [Google Scholar] [CrossRef]
- Burkart, C.A.; Kleppel, G.S. A new incubation system for the measurement of copepod egg production and egg hatching success in the field. J. Exp. Mar. Biol. Ecol. 1998, 221, 89–97. [Google Scholar] [CrossRef]
- Calliari, D.; Tiselius, P. Feeding and reproduction in a small calanoid copepod: Acartia clausi can compensate quality with quantity. Mar. Ecol. Prog. Ser. 2005, 298, 241–250. [Google Scholar] [CrossRef] [Green Version]
- Uriarte, L.; Cotano, U.; Villate, F. Effects of estuarine conditions and organic enrichment on the fecundity and hatching success of Acartia clausi in contrasting systems. J. Exp. Mar. Biol. Ecol. 2005, 320, 105–122. [Google Scholar] [CrossRef]
- Wiadnyana, N.N.; Rassoulzadegan, F. Selective feeding of Acartia clausi and Centropages typicus on microzooplankton. Mar. Ecol. Prog. Ser. 1989, 53, 37–45. [Google Scholar] [CrossRef]
- Guisande, C.; Frangopulos, M.; Carotenuto, Y.; Maneiro, I.; Riveiro, I.; Vergara, A.R. Fate of paralytic shellfish poisoning toxins ingested by the copepod Acartia clausi. Mar. Ecol. Prog. Ser. 2002, 240, 105–115. [Google Scholar] [CrossRef]
- Kozlowsky-Suzuki, B.; Carlsson, P.; Rühl, A.; Granéli, E. Food selectivity and grazing impact on toxic Dinophysis spp. by copepods feeding on natural plankton assemblages. Harmful Algae 2006, 5, 57–68. [Google Scholar] [CrossRef]
- Varkitzi, I.; Pagou, K.; Granéli, E.; Hatzianestis, I.; Krasakopoulou, E.; Pavlidou, A. Spatio-temporal distribution of Dinophysis spp. in relation to organic matter availability and other parameters in Thermaikos Gulf, Greece (Eastern Mediterranean). In Proceedings of the 14th International Conference on Harmful Algae, Crete, Greece, 1–5 November 2010; pp. 51–53. [Google Scholar]
- Siokou-Frangou, I.; Zervoudaki, S.; Zoulias, T. Study of mesozooplankton in the Thermaikos Gulf. In Monitoring of the Marine Environment Quality of Thessaloniki Gulf (Thermaikos Gulf); Final Technical Report; NCMR: Athens, Greece, 2003. [Google Scholar]


| Period (March) | 2003 | 2004 |
|---|---|---|
| Chl-a (μg L−1) | 4.1–5.3 | 0.2–2.7 |
| Total phytoplankton density (×105 cells L−1) | 19.0–46.0 | 9.9–28.8 |
| Range of D. ac. density (cells L−1) | <100 | 600 to 10,700 |
| D. ac.% in total phytoplankton (% cells) | <0.01 | <0.5 |
| D. ac.% in total phytoplankton (% carbon) | <0.06 | 1.7–18 |
| OA in D. acuminata cells (pg cell−1) | 3.7–8.6 | 4.4–14.0 |
| Period (March) | 2003 | 2004 |
|---|---|---|
| faecal pellet production (pellets ind−1 d−1) | 6.8 ± 2.6 (n = 9) | 8.6 ± 2.8 (n = 3) |
| egg production rate (eggs ind−1 d−1) | 15.8 ± 5.4 (n = 7) | 47.6 ± 9.0 (n = 4) |
| egg hatching success (% d−1) | Not measured | 69.1 ± 3.7 (n = 5) |
| D. acuminata cells per faecal pellet | 0.05 ± 0.2 (n = 30) | 0.11 ± 0.4 (n = 53) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frangoulis, C.; Christou, E.D.; Varkitzi, I.; Zervoudaki, S.; Maneiro, I.; Svensen, C.; Pagou, K.; Assimakopoulou, G.; Hatzianestis, I.; Granéli, E. Impact of a Dinophysis acuminata Bloom on the Copepod Acartia clausi: First Indications. Water 2022, 14, 2204. https://doi.org/10.3390/w14142204
Frangoulis C, Christou ED, Varkitzi I, Zervoudaki S, Maneiro I, Svensen C, Pagou K, Assimakopoulou G, Hatzianestis I, Granéli E. Impact of a Dinophysis acuminata Bloom on the Copepod Acartia clausi: First Indications. Water. 2022; 14(14):2204. https://doi.org/10.3390/w14142204
Chicago/Turabian StyleFrangoulis, Constantin, Epaminondas D. Christou, Ioanna Varkitzi, Soultana Zervoudaki, Isabel Maneiro, Camilla Svensen, Kalliopi Pagou, Georgia Assimakopoulou, Ioannis Hatzianestis, and Edna Granéli. 2022. "Impact of a Dinophysis acuminata Bloom on the Copepod Acartia clausi: First Indications" Water 14, no. 14: 2204. https://doi.org/10.3390/w14142204






