Nanoplastic-Induced Nanostructural, Nanomechanical, and Antioxidant Response of Marine Diatom Cylindrotheca closterium
Abstract
:1. Introduction
2. Materials and Methods
2.1. Diatom Culture
2.2. Polystyrene Nanoparticles
2.3. Exposure Experiment
2.4. Growth Inhibition Determination
2.5. Reactive Oxygen Species (ROS) Determination
2.6. Measurement of Enzyme Activities and Malondialdehyde (MDA) Content
2.6.1. Protein Extraction
2.6.2. Enzymatic Activity Assays
2.6.3. Malondialdehyde Content
2.7. Atomic Force Microscopy Working in Force Spectroscopy Mode
2.8. Sample Preparation for Force Spectroscopy Measurements
2.9. Atomic Force Microscopy Imaging
2.10. Sample Preparation for AFM Imaging
3. Results
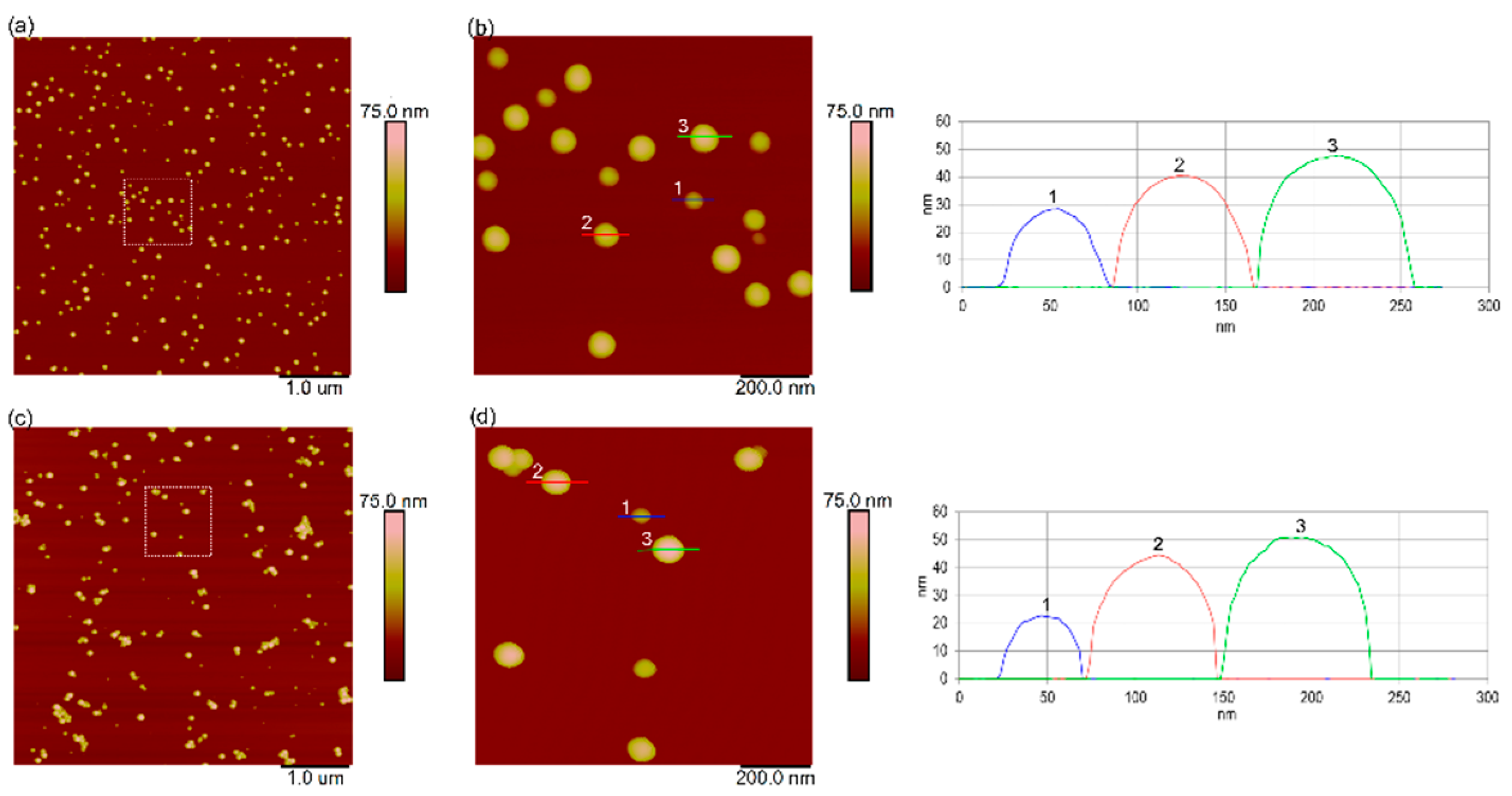
3.1. Characterization of Nanoplastics in Ultrapure Water and Natural Seawater
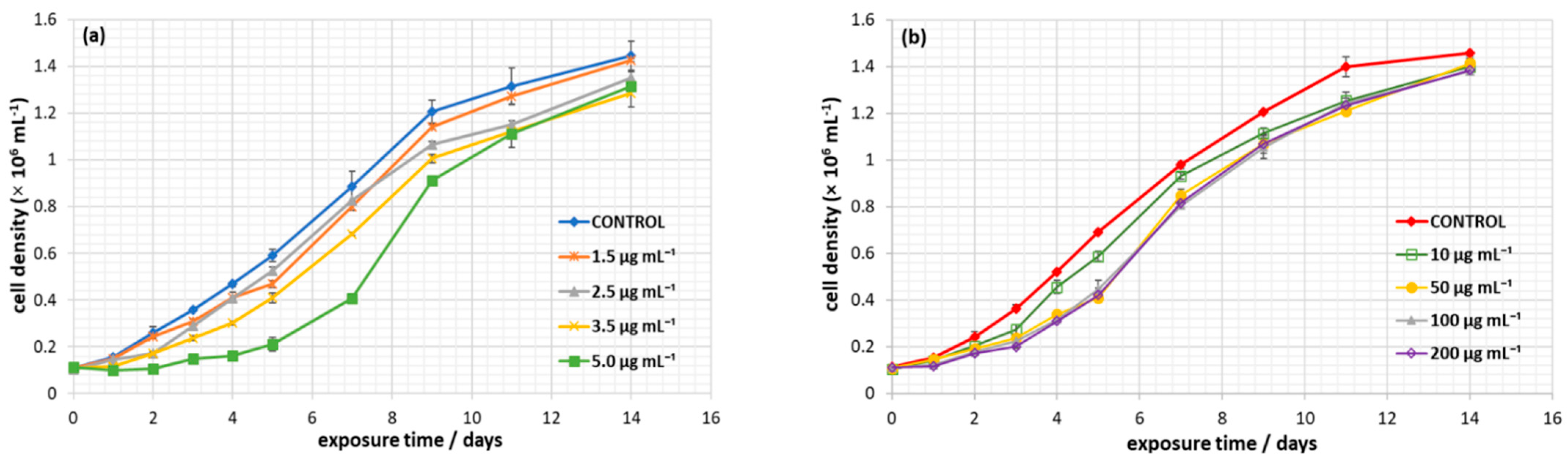
3.2. Effect of Nanoplastics on C. closterium Growth
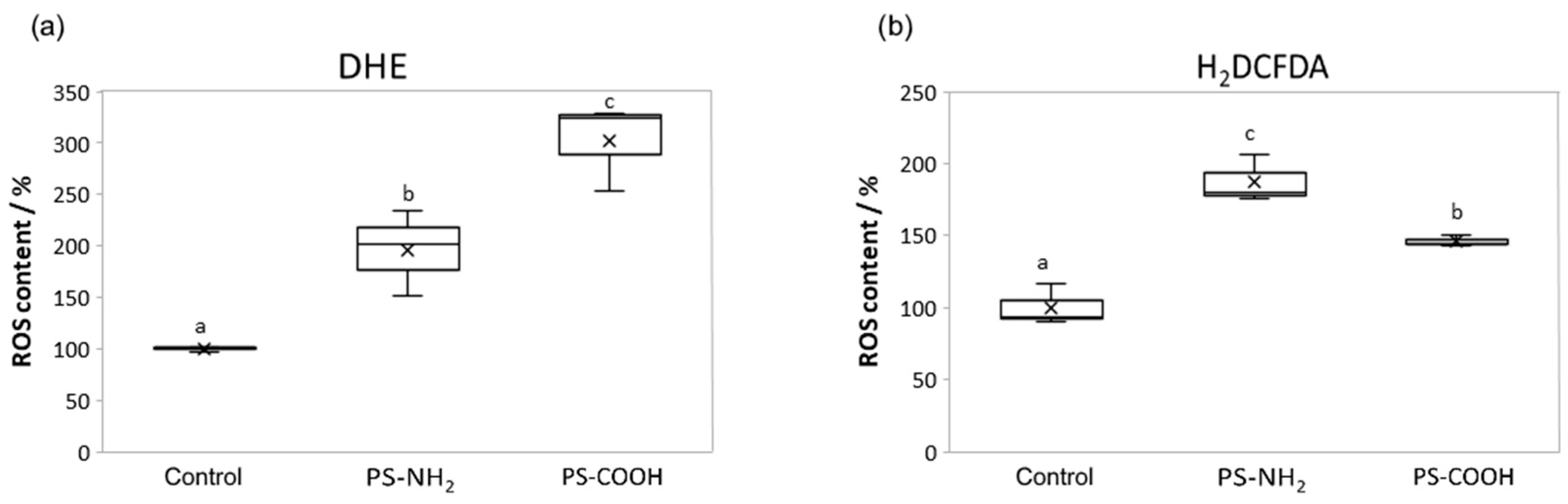
3.3. In Situ ROS Formation
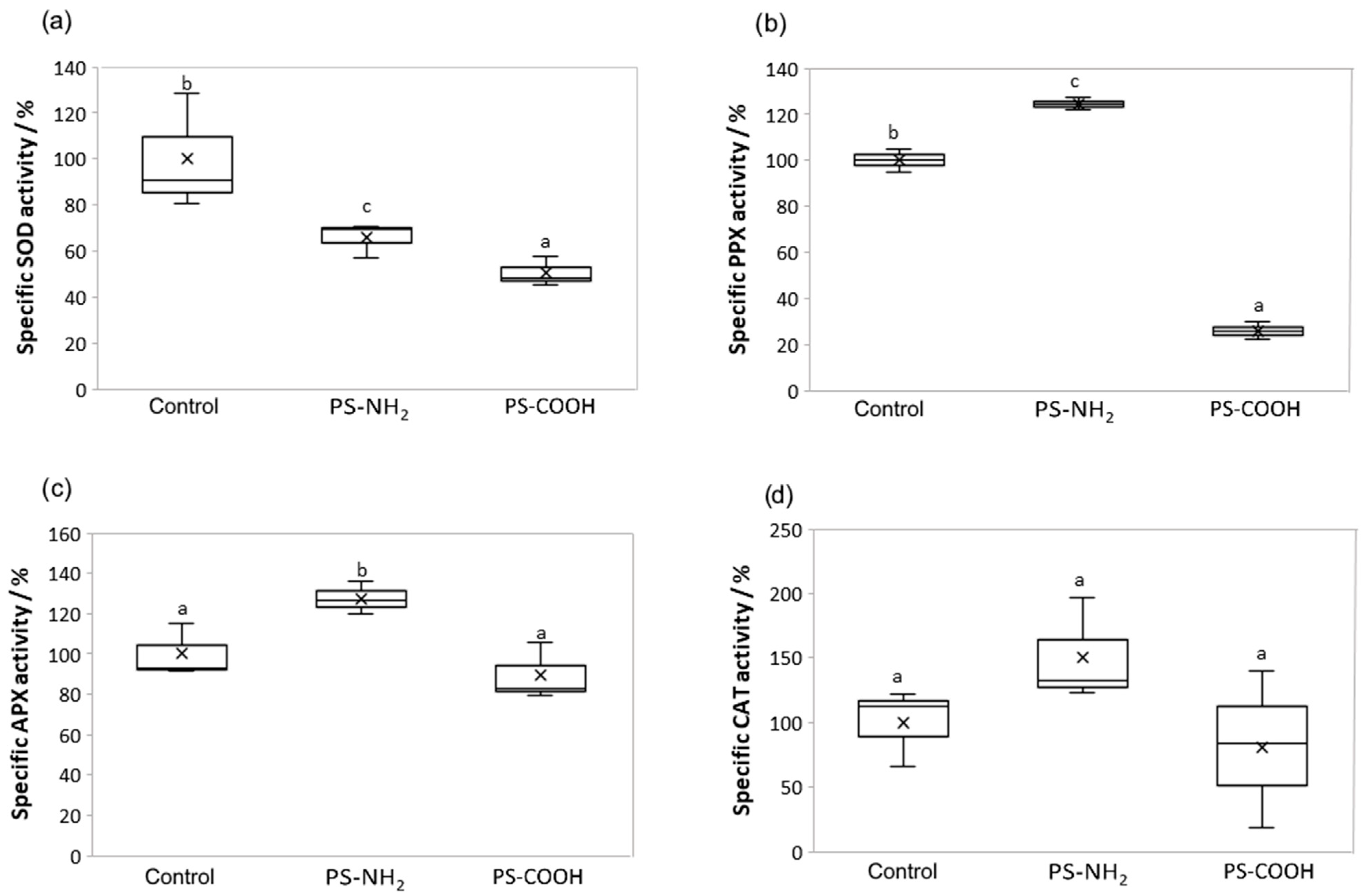
3.4. Effect on Antioxidant Enzyme Activity
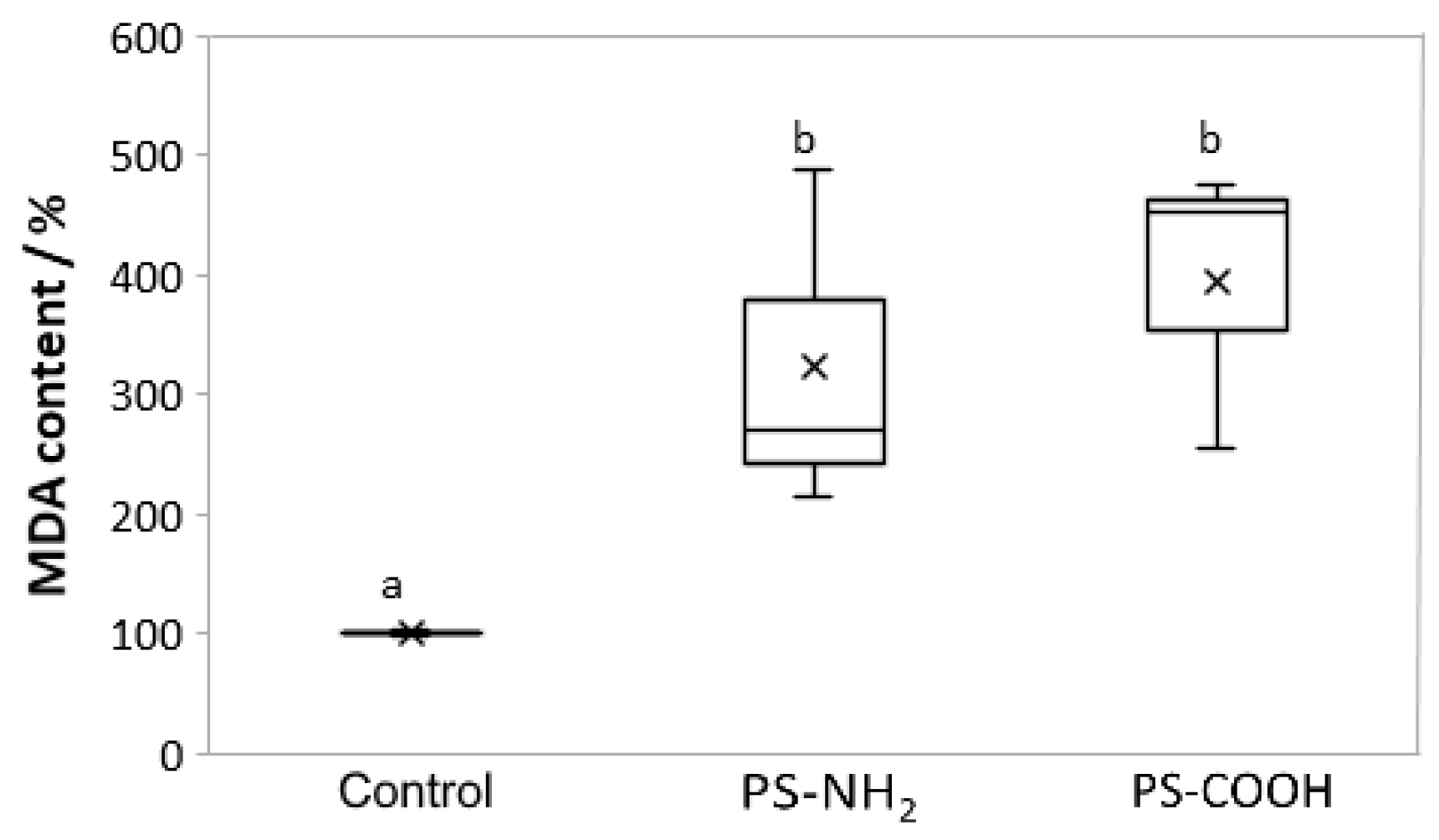
3.5. Effect on Lipid Perodixation
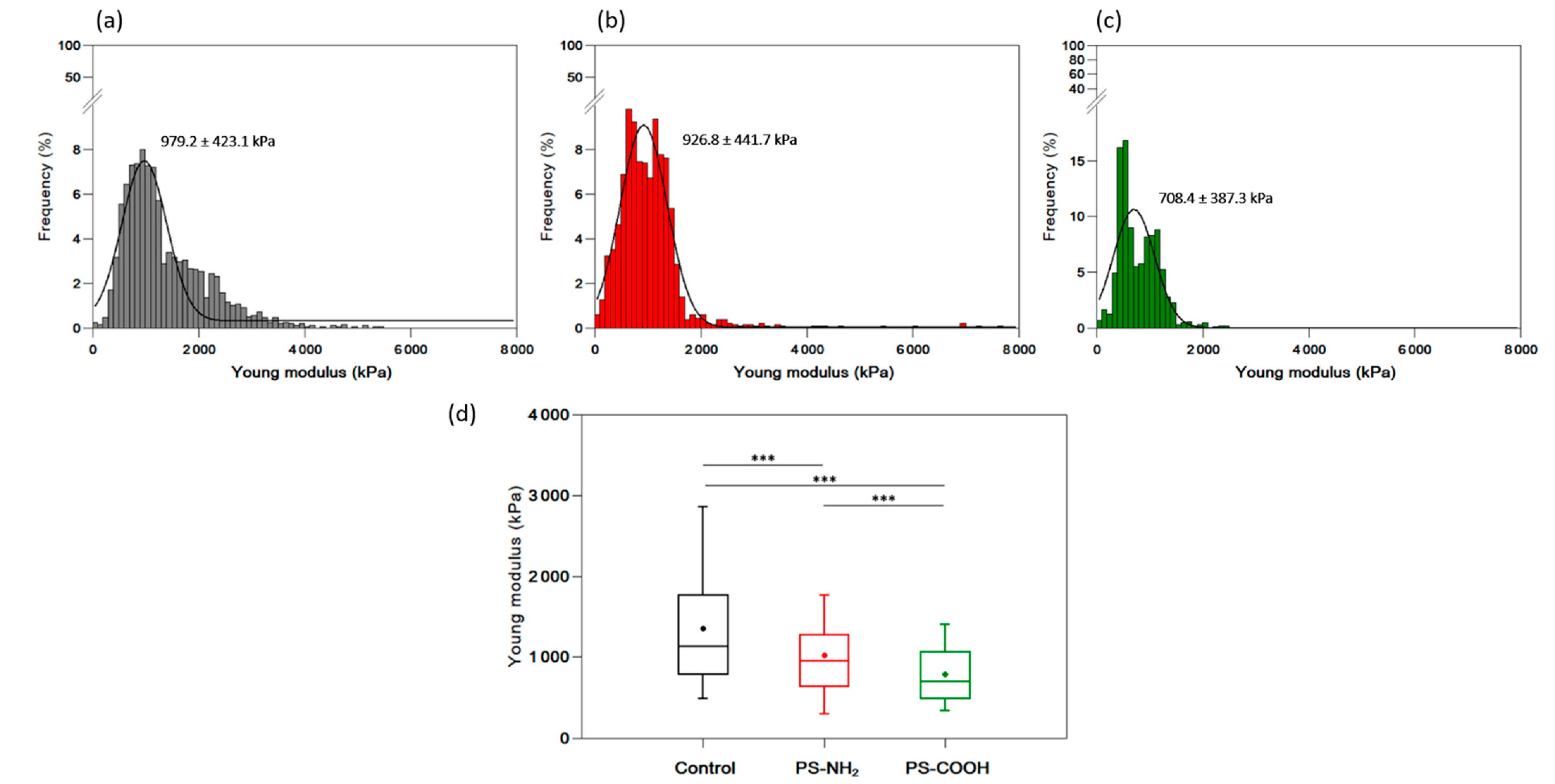
3.6. Effect of Nanoplastics on the Nanomechanical Properties of C. closterium
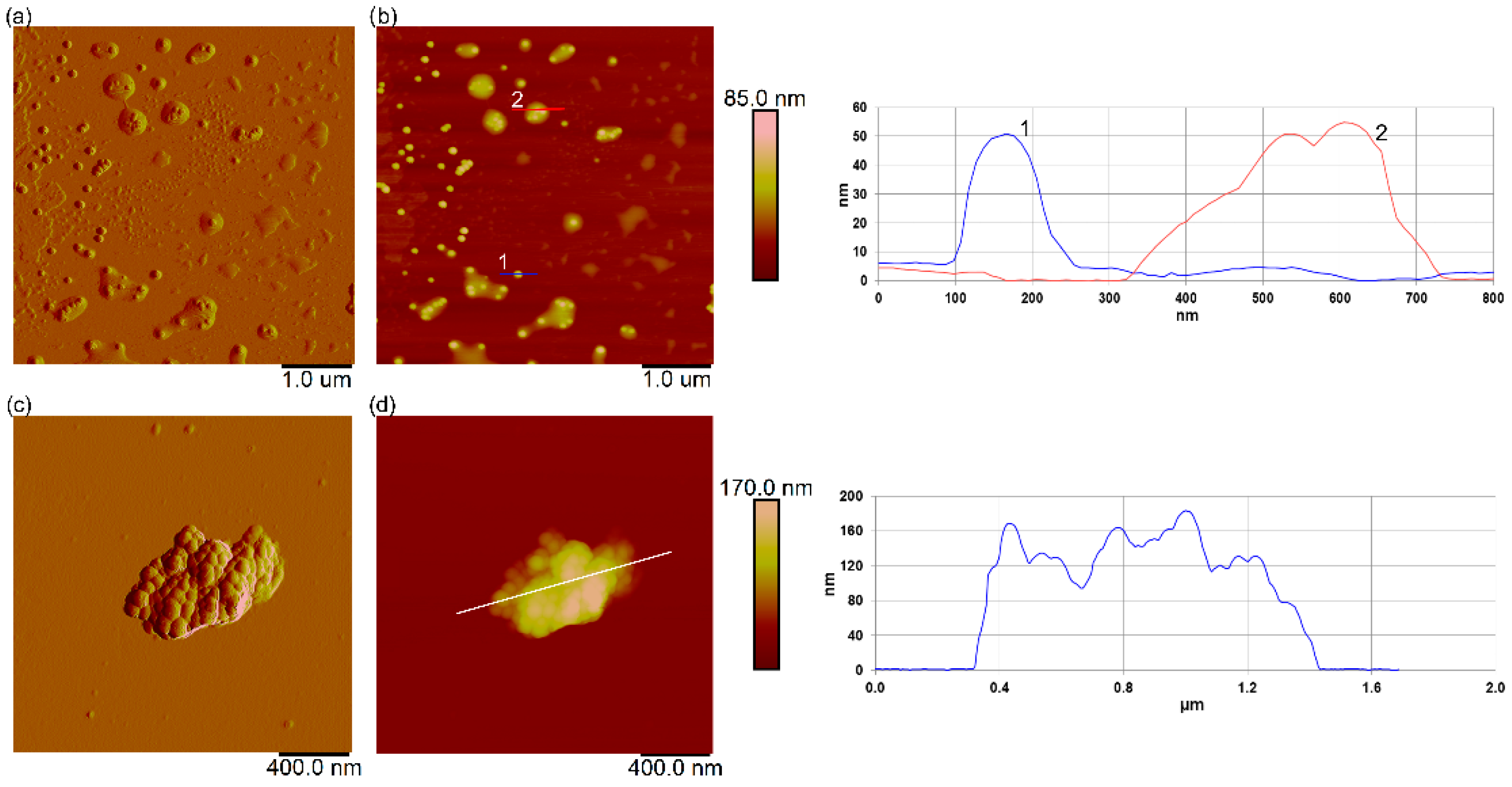
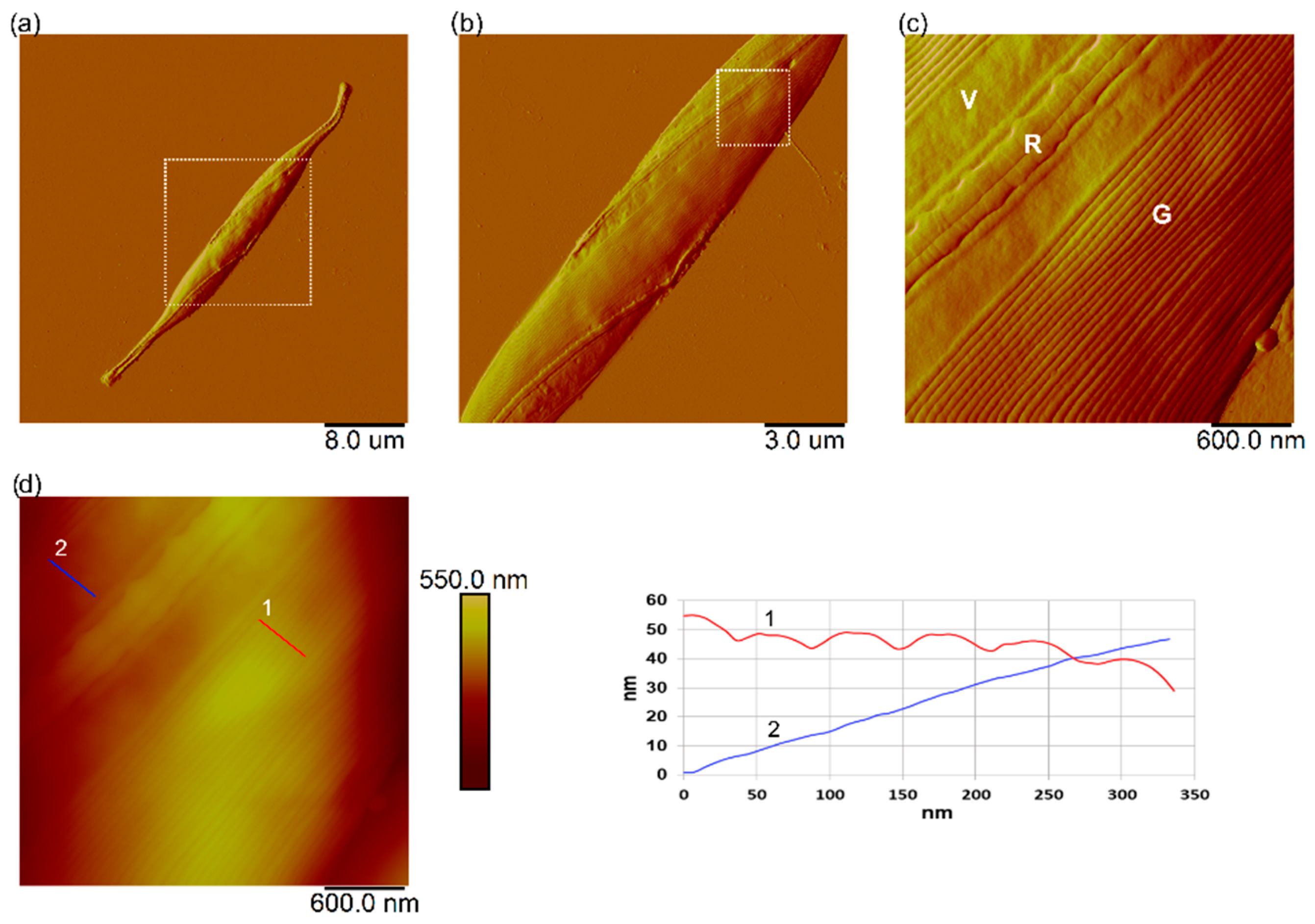
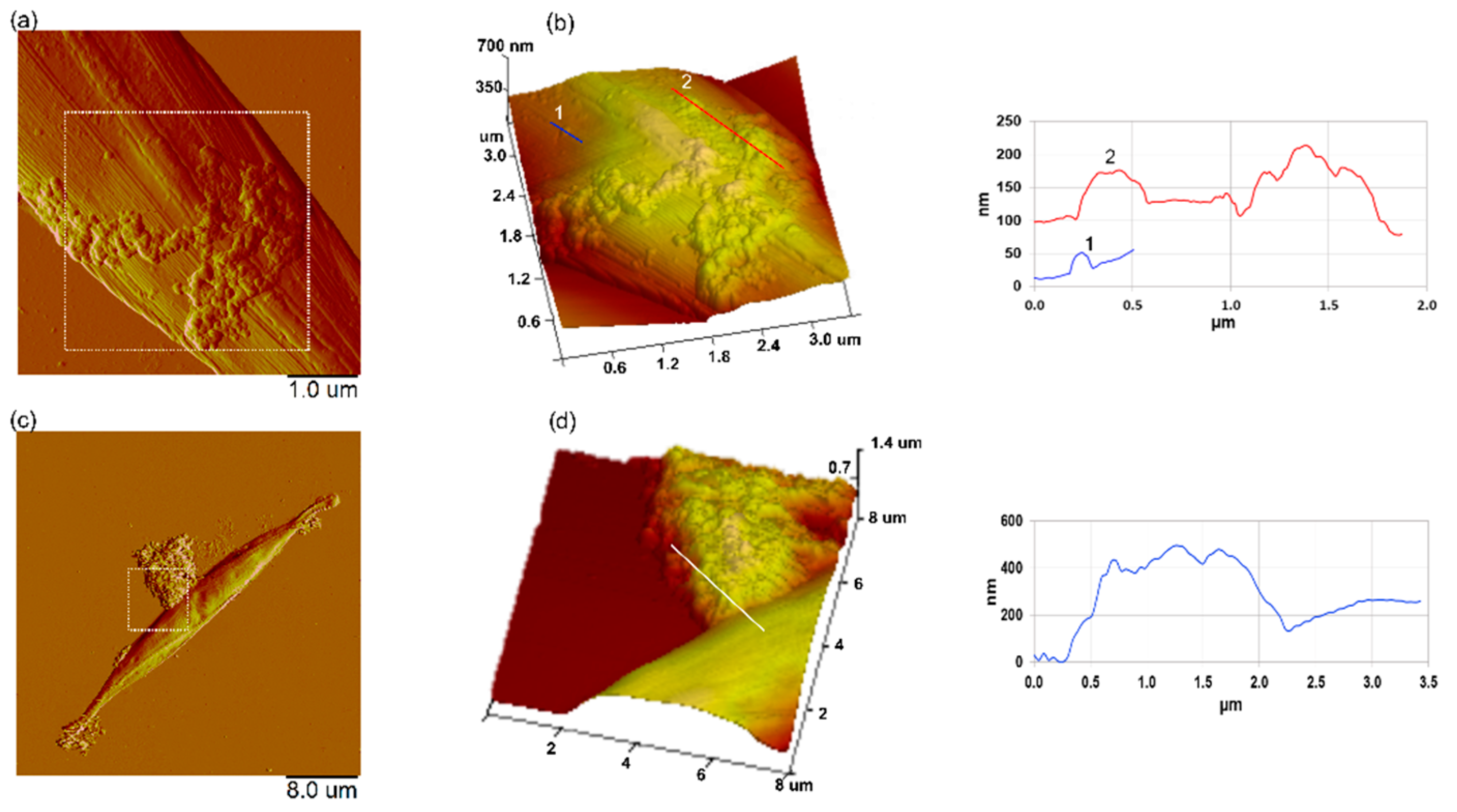
3.7. Interaction of Nanoplastics with C. closterium Cells and Released Extracellular Polymers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFM | Atomic force microscopy |
| APX | Ascorbate peroxidase |
| CAT | Catalase |
| DLS | Dynamic light scattering |
| DOC | Dissolved organic carbon |
| dh | Hydrodynamic diameter |
| DHE | Dihydroethidium |
| EDTA | Ethylenediaminetetraacetic acid |
| ELS | Electrophoretic light scattering |
| EC50 | Effective concentration of substance resulting in a 50% reduction in growth |
| EPS | Extracellular polymers |
| FSW | Filtered seawater |
| H2DCFDA | 2′,7′-dichlorofluorescein diacetate |
| I | Inhibition |
| MDA | Malondialdehyde |
| NBT | Nitroblue tetrazolium |
| NPs | Nanoplastics |
| PDMS | Polydimethylsiloxane |
| PEI | Polyethylenimine |
| PMMA | Poly(methyl methacrylate) |
| PPX | Pyrogallol peroxidase |
| PS | Polystyrene |
| PS NPs | Polystyrene nanoplastics |
| PS-COOH | Carboxyl-modified polystyrene |
| PS-NH2 | Amine-modified polystyrene |
| ROS | Reactive oxygen species |
| SDS | Sodium dodecyl sulfate |
| SOD | Superoxide dismutase |
| UPW | Ultrapure water |
| Ym | Young modulus |
| γ | Mass concentration |
| ζ-potential | Zeta potential |
| µ | Specific growth rate |
References
- Jambeck, J.R.; Andrady, A.; Geyer, R.; Narayan, R.; Perryman, M.; Siegler, T.; Wilcox, C.; Lavender Law, K. Plastic waste inputs from land into the ocean. Mar. Pollut. 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Rochman, C.M. Microplastics research—From sink to source in freshwater systems. Science 2018, 360, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Leslie, H.A.; van Velzen, M.J.M.; Brandsma, S.H.; Vethaak, A.D.; Garcia-Vallejo, J.J.; Lamoree, M.H. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 2022, 163, 107199. [Google Scholar] [CrossRef] [PubMed]
- Jahnke, A.; Arp, H.P.H.; Escher, B.I.; Gewert, B.; Gorokhova, E.; Kühnel, D.; Ogonowski, M.; Potthoff, A.; Rummel, C.; Schmitt-Jansen, M.; et al. Reducing uncertainty and confronting ignorance about the possible impacts of weathering plastic in the marine environment. Environ. Sci. Technol. Lett. 2017, 4, 85–90. [Google Scholar] [CrossRef]
- Prata, J.C.; da Costa, J.P.; Lopes, I.; Duarte, A.C.; Rocha-Santos, T. Effects of microplastics on microalgae populations: A critical review. Sci. Total Environ. 2019, 665, 400–405. [Google Scholar] [CrossRef]
- Gigault, J.; ter Halle, A.; Baudrimont, M.; Pascal, P.Y.; Gauffre, F.; Phi, T.L.; Hadri, H.E.; Grassl, B.; Reynaud, S. Current opinion: What is a nanoplastic? Environ. Pollut. 2018, 235, 1030–1034. [Google Scholar] [CrossRef]
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.G.; McGonigle, D.; Russell, A.E. Lost at sea: Where is all the plastic? Science 2004, 304, 838. [Google Scholar] [CrossRef]
- Kik, K.; Bukowska, B.; Sicińska, P. Polystyrene nanoparticles: Sources, occurrence in the environment, distribution in tissues, accumulation and toxicity to various organisms. Environ. Pollut. 2020, 262, 114297. [Google Scholar] [CrossRef]
- Laganà, P.; Caruso, G.; Corsi, I.; Bergami, E.; Venuti, V.; Majolino, D.; La Ferla, R.; Azzaro, M.; Cappello, S. Do plastics serve as a possible vector for the spread of antibiotic resistance? First insights from bacteria associated to a polystyrene piece from King George Island (Antarctica). Int. J. Hyg. Environ. Health 2019, 222, 89–100. [Google Scholar] [CrossRef]
- Lithner, D.; Larsson, A.; Dave, G. Environmental and health hazard ranking and assessment of plastic polymers based on chemical composition. Sci. Total Environ. 2011, 409, 3309–3324. [Google Scholar] [CrossRef]
- Syranidou, E.; Karkanorachaki, K.; Amorotti, F.; Franchini, M.; Repouskou, E.; Kaliva, M.; Vamvakaki, M.; Kolvenbach, B.; Fava, F.; Corvini, P.F.-X.; et al. Biodegradation of weathered polystyrene films in seawater microcosms. Sci. Rep. 2017, 7, 17991. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Chu, W.; Kok, Y.; Lee, C. Distribution of microplastics and nanoplastics in aquatic ecosystems and their impacts on aquatic organisms, with emphasis on microalgae. In Reviews of Environmental Contamination and Toxicology; de Voogt, P., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; Volume 246, pp. 133–158. [Google Scholar]
- Gonçalves, J.M.; Bebianno, M.J. Nanoplastics impact on marine biota: A review. Environ. Pollut. 2021, 273, 116426. [Google Scholar] [CrossRef] [PubMed]
- Zaki, M.R.M.; Aris, A.Z. An overview of the effects of nanoplastics on marine organisms. Sci. Total Environ. 2022, 831, 154757. [Google Scholar] [CrossRef] [PubMed]
- Bitton, G.; Dutka, B.J. Introduction and review of microbial and biochemical toxicity screening procedures. In Toxicity Test Using Microorganisms, 1st ed.; Bitton, G., Dutka, B.J., Eds.; CRC Press: New York, NY, USA, 1986; pp. 1–8. [Google Scholar]
- Armbrust, E.V. The life of diatoms in the world’s oceans. Nature 2009, 459, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Falkowski, P.; Katz, M.E.; Knoll, A.H.; Quigg, A.; Raven, J.A.; Schofield, O.; Taylor, F.J.R. The evolution of modern eukaryotic phytoplankton. Science 2004, 305, 354–360. [Google Scholar] [CrossRef]
- Granum, E.; Raven, J.A.; Leegood, R.C. How do marine diatoms fix 10 billion tonnes of inorganic carbon per year? Can. J. Bot. 2005, 83, 898–908. [Google Scholar] [CrossRef]
- De Stefano, L.; Rotiroti, L.; De Stefano, M.; Lamberti, A.; Lettieri, S.; Setaro, A.; Maddalena, P. Marine diatoms as optical biosensors. Biosens. Bioelectron. 2009, 24, 1580–1584. [Google Scholar] [CrossRef]
- Rimet, F. Recent views on river pollution and diatoms. Hydrobiologia 2012, 683, 1–24. [Google Scholar] [CrossRef]
- Tudesque, L.; Grenouillet, G.; Gevrey, M.; Khazraie, K.; Brosse, S. Influence of small-scale gold mining on French Guiana streams: Are diatom assemblages valid disturbance sensors? Ecol. Indic. 2012, 14, 100–106. [Google Scholar] [CrossRef]
- Sjollema, S.B.; Redondo-Hasselerharm, P.; Leslie, H.A.; Kraak, M.H.S.; Vethaak, A.D. Do plastic particles affect microalgal photosynthesis and growth? Aquat. Toxicol. 2016, 170, 259–261. [Google Scholar] [CrossRef]
- Bergami, E.; Pugnalini, S.; Vannuccini, M.L.; Manfra, L.; Faleri, C.; Savorelli, F.; Dawson, K.A.; Corsi, I. Long-term toxicity of surface-charged polystyrene nanoplastics to marine planktonic species Dunaliella tertiolecta and Artemia franciscana. Aquat. Toxicol. 2017, 189, 159–169. [Google Scholar] [CrossRef] [PubMed]
- González-Fernández, C.; Le Grand, F.; Bideau, A.; Huvet, A.; Paul-Pont, I.; Soudant, P. Nanoplastics exposure modulate lipid and pigment compositions in diatoms. Environ. Pollut. 2020, 262, 114274. [Google Scholar] [CrossRef] [PubMed]
- Khoshnamvand, M.; Hanachi, P.; Ashtiani, S.; Walker, T.R. Toxic effects of polystyrene nanoplastics on microalgae Chlorella vulgaris: Changes in biomass, photosynthetic pigments and morphology. Chemosphere 2021, 280, 130725. [Google Scholar] [CrossRef]
- Hazeem, L.J.; Yesilay, G.; Bououdina, M.; Perna, S.; Cetin, D.; Suludere, Z.; Barras, A.; Boukherroub, R. Investigation of the toxic effects of different polystyrene micro-and nanoplastics on microalgae Chlorella vulgaris by analysis of cell viability, pigment content, oxidative stress and ultrastructural changes. Mar. Pollut. Bull. 2020, 156, 111278. [Google Scholar] [CrossRef] [PubMed]
- Sendra, M.; Staffieri, E.; Yeste, P.M.; Moreno-Garrido, I.; Gatica, J.M.; Corsi, I.; Julián Blasco, J. Are the primary characteristics of polystyrene nanoplastics responsible for toxicity and ad/absorption in the marine diatom Phaeodactylum tricornutum? Environ. Pollut. 2019, 249, 610–619. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.C.; Liu, F.F.; Huang, T.Y.; Fan, J.L.; Gao, Z.Y.; Liu, G.Z. Effects of nanoplastics on the dinoflagellate Amphidinium Carterae Hulburt from the perspectives of algal growth, oxidative stress and hemolysin production. Nanomaterials 2021, 11, 2471. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Tan, L.; Zhu, X.; Huang, W.; Wang, J. Size-dependent oxidative stress effect of nano/micro-scaled polystyrene on Karenia mikimotoi. Mar. Pollut. Bull. 2020, 154, 111074. [Google Scholar] [CrossRef]
- Venâncio, C.; Ferreira, I.; Martins, M.A.; Soares, A.M.V.M.; Lopes, I.; Oliveira, M. The effects of nanoplastics on marine plankton: A case study with polymethylmethacrylate. Ecotoxicol. Environ. Saf. 2019, 184, 109632. [Google Scholar] [CrossRef]
- Bellingeri, A.; Bergami, E.; Grassi, G.; Faleri, C.; Redondo-Hasselerharm, P.; Koelmans, A.A.; Corsi, I. Combined effects of nanoplastics and copper on the freshwater alga Raphidocelis subcapitata. Aquat. Toxicol. 2019, 210, 179–187. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Lin, S.; Turner, J.P.; Ke, P.C. Physical adsorption of charged plastic nanoparticles affects algal photosynthesis. J. Phys. Chem. C 2010, 114, 16556–16561. [Google Scholar] [CrossRef]
- Chae, Y.; Kim, D.; Kim, S.W.; An, Y.J. Trophic transfer and individual impact of nano-sized polystyrene in a four-species freshwater food chain. Sci. Rep. 2018, 8, 284. [Google Scholar] [CrossRef] [PubMed]
- Nolte, T.M.; Hartmann, N.B.; Kleijn, J.M.; Garnæs, J.; van de Meent, D.; Hendriks, A.J.; Baun, A. The toxicity of plastic nanoparticles to green algae as influenced by surface modification, medium hardness and cellular adsorption. Aquat. Toxicol. 2017, 183, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Gomes, T.; Almeida, A.C.; Georgantzopoulou, A. Characterization of cell responses in Rhodomonas baltica exposed to PMMA nanoplastics. Sci. Total Environ. 2020, 726, 138547. [Google Scholar] [CrossRef] [PubMed]
- Guillard, R.R.L. Culture of phytoplankton for feeding marine invertebrates. In Culture of Marine Invertebrate Animals, Proceedings of the 1st Conference on Culture of Marine Invertebrate Animals Greenport, New York, NY, USA, October 1972; Smith, W.L., Chanley, M.H., Eds.; Springer: Boston, MA, USA, 1975; pp. 29–60. [Google Scholar]
- Al-Sid-Cheikh, M.; Rowland, S.J.; Stevenson, K.; Rouleau, C.; Henry, T.B.; Thompson, R.C. Uptake, whole-body distribution, and depuration of nanoplastics by the scallop Pecten maximus at environmentally realistic concentrations. Environ. Sci. Technol. 2018, 52, 14480–14486. [Google Scholar] [CrossRef]
- Organisation for Economic Cooperation and Development. OECD Test No. 201. Freshwater algae and cyanobacteria, growth inhibition test. In OECD Guidelines for the Testing of Chemicals, Section 2. Effects on Biotic Systems; Organisation for Economic Cooperation and Development: Paris, France, 2011. [Google Scholar]
- Cvjetko, P.; Milošić, A.; Domijan, A.M.; Vinković, I.V.; Tolić, S.; Štefanić, P.P.; Letofsky-Papst, I.; Tkalec, M.; Balen, B. Toxicity of silver ions and differently coated silver nanoparticles in Allium cepa roots. Ecotoxicol. Environ. Saf. 2017, 137, 18–28. [Google Scholar] [CrossRef]
- Hong, Y.; Hu, H.Y.; Xie, X.; Sakoda, A.; Sagehashi, M.; Li, F.M. Gramine-induced growth inhibition, oxidative damage and antioxidant responses in freshwater cyanobacterium Microcystis aeruginosa. Aquat. Toxicol. 2009, 91, 262–269. [Google Scholar] [CrossRef]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assay and an assay applicable to PAGE. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts. I. Kinetics and stechiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Hertz, H. Über die Berührung fester elastischer Körper. Gruyter 2009, 92, 156–171. [Google Scholar] [CrossRef]
- Hutter, J.L.; Bechhoefer, J. Calibration of atomic-force microscope tips. Rev. Sci. Instrum. 1993, 64, 1868–1873. [Google Scholar] [CrossRef]
- Balnois, E.; Wilkinson, K.J. Sample preparation techniques for the observation of environmental biopolymers by atomic force microscopy. Colloid. Surf. A 2022, 207, 229–242. [Google Scholar] [CrossRef]
- Mišić Radić, T.; Svetličić, V.; Žutić, V.; Boulgaropoulos, B. Seawater at the nanoscale: Marine gel imaged by atomic force microscopy. J. Mol. Recognit. 2011, 24, 397–405. [Google Scholar] [CrossRef]
- Mišić Radić, T.; Čačković, A.; Penezić, A.; Dautović, J.; Lončar, J.; Omanović, D.; Juraić, K.; Ljubešić, Z. Physiological and morphological response of marine diatom Cylindrotheca closterium (Bacillariophyceae) exposed to cadmium. Eur. J. Phycol. 2021, 56, 24–36. [Google Scholar] [CrossRef]
- Tallec, K.; Blard, O.; González-Fernández, C.; Brotons, G.; Berchel, M.; Soudant, P.; Huvet, A.; Paul-Pont, I. Surface functionalization determines behavior of nanoplastic solutions in model aquatic environments. Chemosphere 2019, 225, 639–646. [Google Scholar] [CrossRef]
- El Badawy, A.M.; Luxton, T.P.; Silva, R.G.; Scheckel, K.G.; Suidan, M.T.; Tolaymat, T.M. Impact of environmental conditions (pH, ionic strength, and electrolyte type) on the surface charge and aggregation of silver nanoparticles suspensions. Environ. Sci. Technol. 2010, 44, 1260–1266. [Google Scholar] [CrossRef]
- Yu, S.J.; Shen, M.H.; Li, S.S.; Fu, Y.J.; Zhang, D.; Liu, H.Y.; Liu, J.F. Aggregation kinetics of different surface-modified polystyrene nanoparticles in monovalent and divalent electrolytes. Environ. Pollut. 2019, 255, 113302. [Google Scholar] [CrossRef]
- Saavedra, J.; Stoll, S.; Slaveykova, V.I. Influence of nanoplastic surface charge on eco-corona formation, aggregation and toxicity to freshwater zooplankton. Environ. Pollut. 2019, 252, 715–722. [Google Scholar] [CrossRef]
- Summers, S.; Gutierrez, T.; Henry, T. Agglomeration of nano- and microplastic particles in seawater by autochthonous and de novo-produced sources of exopolymeric substances. Mar. Pollut. Bull. 2018, 130, 258–267. [Google Scholar] [CrossRef]
- Lin, S.; Mortimer, M.; Chen, R.; Kakinen, A.; Riviere, J.E.; Davis, T.P.; Ding, F.; Ke, P.C. NanoEHS beyond toxicity–focusing on biocorona. Environ. Sci. Nano 2017, 4, 1433–1454. [Google Scholar] [CrossRef] [PubMed]
- Besseling, E.; Wang, B.; Lürling, M.; Koelmans, A.A. Nanoplastic affects growth of S. obliquus and reproduction of D. magna. Environ. Sci. Technol. 2014, 48, 12336–12343. [Google Scholar] [CrossRef] [PubMed]
- Nugnes, R.; Lavorgna, M.; Orlo, E.; Russo, C.; Isidori, M. Toxic impact of polystyrene microplastic particles in freshwater organisms. Chemosphere 2022, 299, 134373. [Google Scholar] [CrossRef] [PubMed]
- Biba, R.; Košpić, K.; Komazec, B.; Markulin, D.; Cvjetko, P.; Pavoković, D.; Peharec Štefanić, P.; Tkalec, M.; Balen, B. Surface coating-modulated phytotoxic responses of silver nanoparticles in plants and freshwater green algae. Nanomaterials 2022, 12, 24. [Google Scholar] [CrossRef]
- Liu, Z.; Huang, Y.; Jiao, Y.; Chen, Q.; Wu, D.; Yu, P.; Li, Y.; Cai, M.; Zhao, Y. Polystyrene nanoplastic induces ROS production and affects the MAPK-HIF-1/NFkB-mediated antioxidant system in Daphnia pulex. Aquat Toxicol. 2020, 220, 105420. [Google Scholar] [CrossRef]
- Demir, I.; Blockx, J.; Dague, E.; Guiraud, P.; Thielemans, W.; Muylaert, K.; Formosa-Dague, C. Nanoscale evidence unravels microalgae flocculation mechanism induced by chitosan. ACS Appl. Bio. Mater. 2020, 3, 8446–8459. [Google Scholar] [CrossRef]
- Formosa-Dague, C.; Gernigon, V.; Castelain, M.; Daboussi, F.; Guiraud, P. Towards a better understanding of the flocculation/flotation mechanism of the marine microalgae Phaeodactylum tricornutum under increased pH using atomic force microscopy. Algal Res. 2018, 33, 369–378. [Google Scholar] [CrossRef]
- Francius, G.; Tesson, B.; Dague, E.; Martin-Jézéquel, V.; Dufrêne, Y.F. Nanostructure and nanomechanics of live Phaeodactylum tricornutum morphotypes. Environ. Microbiol. 2018, 10, 1344–1356. [Google Scholar] [CrossRef]
- Ma, J.; Zhou, B.; Duan, D.; Pan, K. Salinity-dependent nanostructures and composition of cell surface and its relation to Cd toxicity in an estuarine diatom. Chemosphere 2019, 215, 807–814. [Google Scholar] [CrossRef]
- Novosel, N.; Mišić Radić, T.; Levak Zorinc, M.; Zemla, J.; Lekka, M.; Vrana, I.; Gašparović, B.; Horvat, L.; Kasum, D.; Legović, T.; et al. Salinity induced chemical, mechanical and behavioral changes in marine microalgae. J. Appl. Phycol. 2022, 34, 1293–1309. [Google Scholar] [CrossRef]
- Novosel, N.; Mišić Radić, T.; Zemla, J.; Lekka, M.; Čačković, A.; Kasum, D.; Legović, T.; Žutinić, P.; Gligora Udovič, M.; Ivošević DeNardis, N. Temperature-induced response in algal cell surface properties and behaviour: An experimental approach. J. Appl. Phycol. 2022, 34, 243–259. [Google Scholar] [CrossRef]
- Vrana, I.; Bakija Alempijević, S.; Novosel, N.; Ivošević DeNardis, N.; Žigon, D.; Ogrinc, N.; Gašparović, B. Hyposalinity induces significant polar lipid remodeling in the marine microalga Dunaliella tertiolecta (Chlorophyceae). J. Appl. Phycol. 2022, 34, 1457–1470. [Google Scholar] [CrossRef]
- Yap, B.H.J.; Crawford, S.A.; Dagastine, R.R.; Scales, P.J.; Martin, G.J.O. Nitrogen deprivation of microalgae: Effect on cell size, cell wall thickness, cell strength, and resistance to mechanical disruption. J. Ind. Microbiol. Biotechnol. 2016, 43, 1671–1680. [Google Scholar] [CrossRef]
- Ivošević DeNardis, N.; Pečar Ilić, J.; Ružić, I.; Novosel, N.; Mišić Radić, T.; Weber, A.; Kasum, D.; Pavlinska, Z.; Balogh, R.K.; Hajdu, B.; et al. Algal cell response to laboratory-induced cadmium stress: A multimethod approach. Eur. Biophys. J. 2019, 48, 231–248. [Google Scholar] [CrossRef]
- Underwood, G.J.C.; Boulcott, M.; Raines, C.A.; Waldron, K. Environmental effects on exopolymer production by marine benthic diatoms: Dynamics, changes in composition, and pathways of production. J. Phycol. 2004, 40, 293–304. [Google Scholar] [CrossRef]
- Underwood, G.J.C.; Paterson, D.M. The importance of extracellular carbohydrate production by marine epipelic diatoms. Adv. Bot. Res. 2003, 40, 184–240. [Google Scholar] [CrossRef]
- Pletikapić, G.; Mišić Radić, T.; Hozić Zimmermann, A.; Svetličić, V.; Pfannkuchen, M.; Marić, D.; Godrijan, J.; Žutić, V. AFM imaging of extracellular polymer release by marine diatom Cylindrotheca closterium (Ehrenberg) Reiman & J.C. Lewin. J. Mol. Recognit. 2011, 24, 436–445. [Google Scholar] [CrossRef]
- Urbani, R.; Magaletti, E.; Sist, P.; Cicero, A.M. Extracellular carbohydrates released by the marine diatoms Cylindrotheca closterium, Thalassiosira pseudonana and Skeletonema costatum: Effect of P-depletion and growth status. Sci. Total. Environ. 2005, 353, 300–306. [Google Scholar] [CrossRef]
- Mishra, A.; Kavita, K.; Jha, B. Characterization of extracellular polymeric substances produced by micro-algae Dunaliella salina. Carbohydr. Polym. 2011, 83, 852–857. [Google Scholar] [CrossRef]
- Pletikapić, G.; Vinković Vrček, I.; Žutić, V.; Svetličić, V. Atomic force microscopy characterization of silver nanoparticles interactions with marine diatom cells and extracellular polymeric substance. J. Mol. Recognit. 2012, 25, 309–317. [Google Scholar] [CrossRef]

| Nanoplastics | γ(NP)/µg mL−1 | Specific Growth Rates µ/d−1 | Growth Inhibitions I/% | ||||||
|---|---|---|---|---|---|---|---|---|---|
| D3 | D4 | D9 | D14 | D3 | D4 | D9 | D14 | ||
| PS-NH2 | 0 | 0.40 | 0.36 | 0.27 | 0.18 | - | - | - | - |
| 1.5 | 0.35 | 0.33 | 0.26 | 0.18 | 12.5 | 8.3 | 3.7 | 0 | |
| 2.5 | 0.34 | 0.34 | 0.26 | 0.18 | 15.0 | 5.5 | 3.7 | 0 | |
| 3.5 | 0.26 | 0.25 | 0.25 | 0.18 | 35.0 | 30.5 | 7.4 | 0 | |
| 5.0 | 0.09 | 0.09 | 0.23 | 0.17 | 77.5 | 75.0 | 14.8 | 5.6 | |
| PS-COOH | 0 | 0.39 | 0.38 | 0.26 | 0.18 | - | - | - | - |
| 10 | 0.33 | 0.37 | 0.26 | 0.18 | 15.4 | 5.3 | 0 | 0 | |
| 50 | 0.26 | 0.28 | 0.25 | 0.18 | 33.3 | 26.3 | 3.8 | 0 | |
| 100 | 0.24 | 0.26 | 0.25 | 0.18 | 38.5 | 31.6 | 3.8 | 0 | |
| 200 | 0.19 | 0.25 | 0.25 | 0.18 | 51.3 | 34.2 | 3.8 | 0 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mišić Radić, T.; Vukosav, P.; Komazec, B.; Formosa-Dague, C.; Domazet Jurašin, D.; Peharec Štefanić, P.; Čačković, A.; Juraić, K.; Ivošević DeNardis, N. Nanoplastic-Induced Nanostructural, Nanomechanical, and Antioxidant Response of Marine Diatom Cylindrotheca closterium. Water 2022, 14, 2163. https://doi.org/10.3390/w14142163
Mišić Radić T, Vukosav P, Komazec B, Formosa-Dague C, Domazet Jurašin D, Peharec Štefanić P, Čačković A, Juraić K, Ivošević DeNardis N. Nanoplastic-Induced Nanostructural, Nanomechanical, and Antioxidant Response of Marine Diatom Cylindrotheca closterium. Water. 2022; 14(14):2163. https://doi.org/10.3390/w14142163
Chicago/Turabian StyleMišić Radić, Tea, Petra Vukosav, Bruno Komazec, Cécile Formosa-Dague, Darija Domazet Jurašin, Petra Peharec Štefanić, Andrea Čačković, Krunoslav Juraić, and Nadica Ivošević DeNardis. 2022. "Nanoplastic-Induced Nanostructural, Nanomechanical, and Antioxidant Response of Marine Diatom Cylindrotheca closterium" Water 14, no. 14: 2163. https://doi.org/10.3390/w14142163
APA StyleMišić Radić, T., Vukosav, P., Komazec, B., Formosa-Dague, C., Domazet Jurašin, D., Peharec Štefanić, P., Čačković, A., Juraić, K., & Ivošević DeNardis, N. (2022). Nanoplastic-Induced Nanostructural, Nanomechanical, and Antioxidant Response of Marine Diatom Cylindrotheca closterium. Water, 14(14), 2163. https://doi.org/10.3390/w14142163










