Environmental Risk Assessment of Silver Nanoparticles in Aquatic Ecosystems Using Fuzzy Logic
Abstract
:1. Introduction
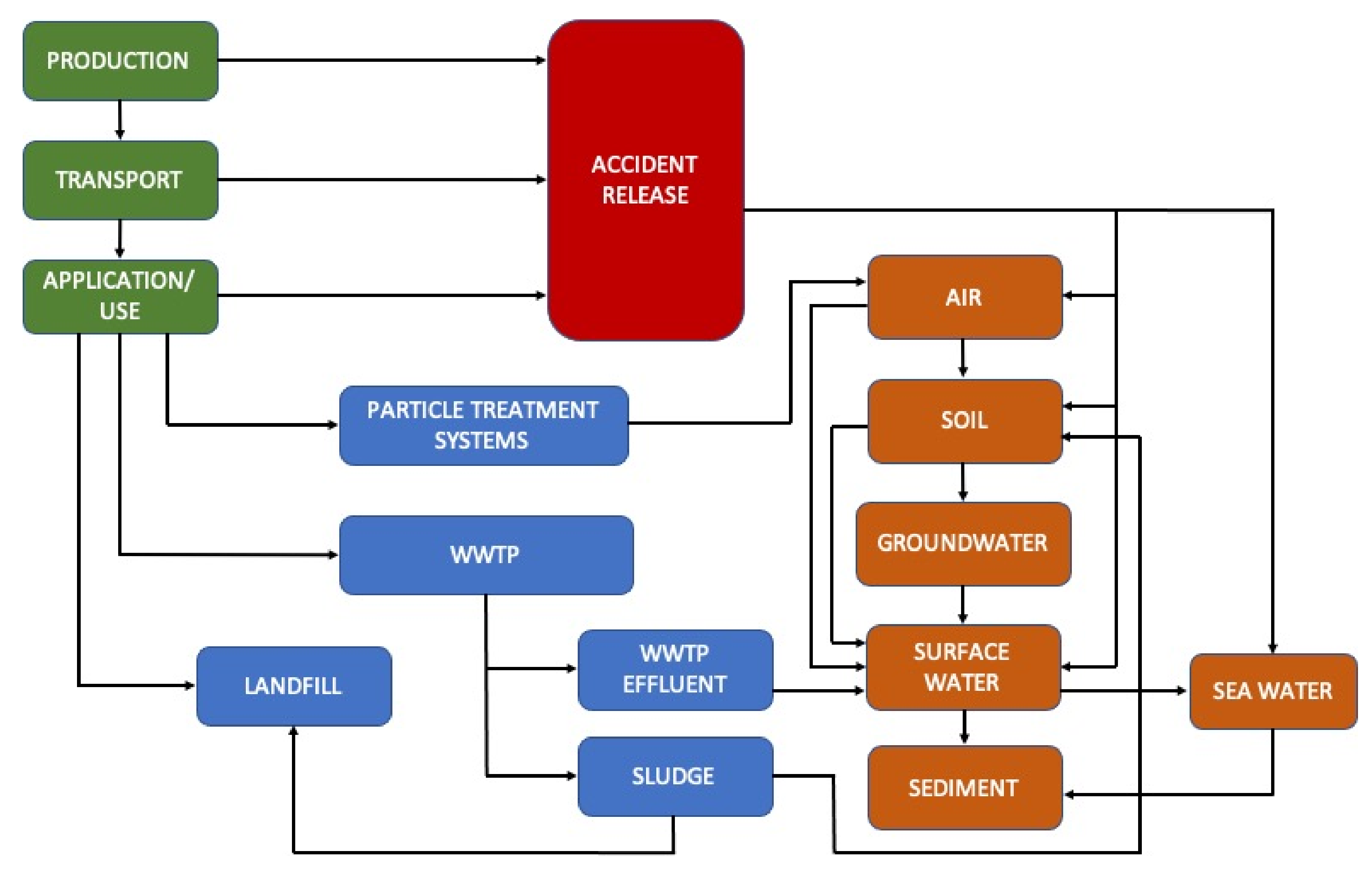
2. Materials and Methods
2.1. Fuzzy Logic Model
- ➢
- Identification of the system variables/inputs (e.g., pollutant concentration, toxicity, coating);
- ➢
- Fuzzification by establishing fuzzy sets (e.g., high, medium, low) as well as membership functions and ranges for each variable;
- ➢
- Use of a Fuzzy Inference Process by establishing fuzzy propositions or rules used to connect the inputs of the problem with the output;
- ➢
- Defuzzification to obtain the final output: risk assessment.
2.2. Case Studies
2.3. Sensitivity Analysis
3. Results and Discussion
3.1. Montcada WWTP Effluents Case Study
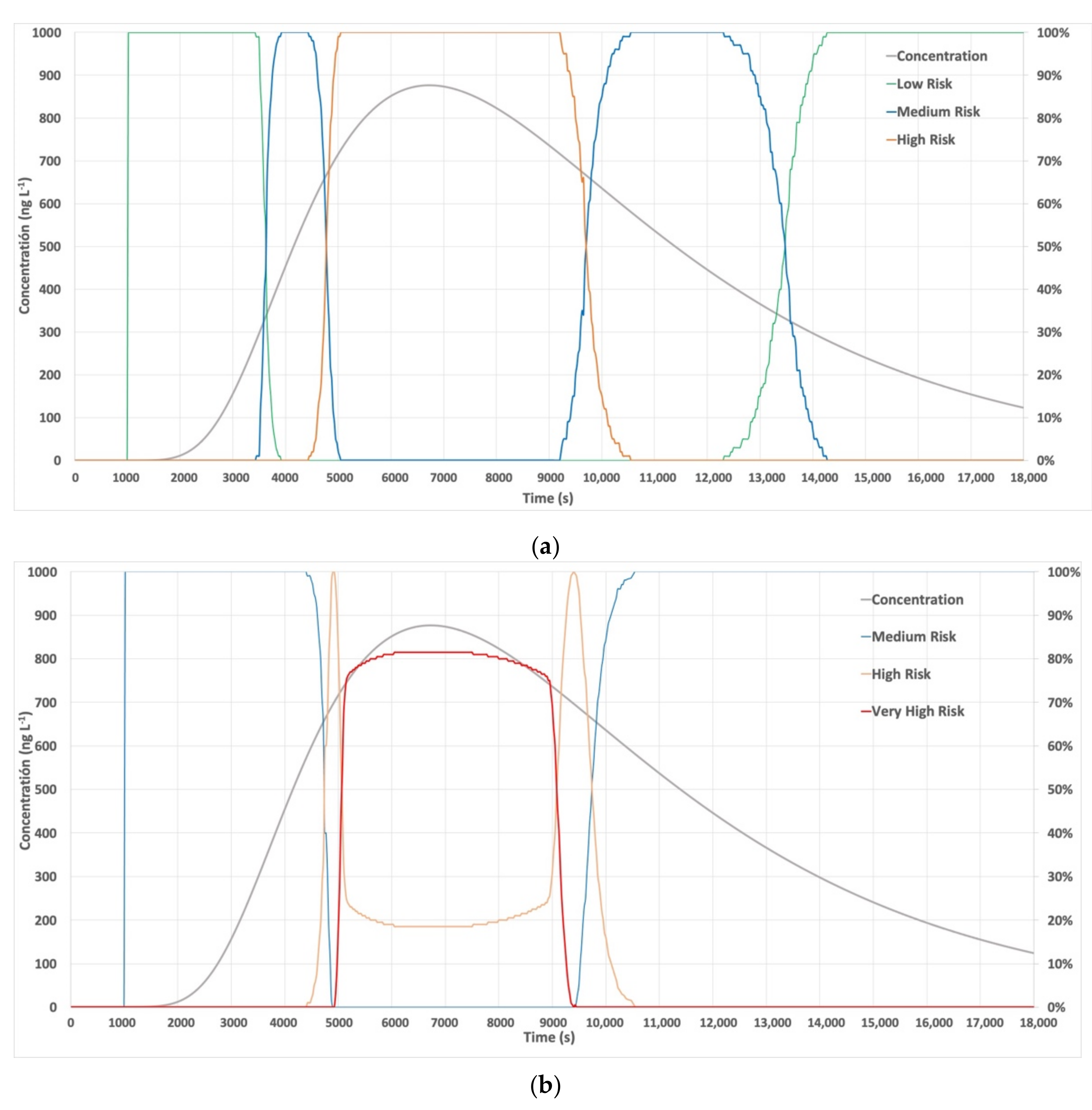
3.2. Accidental Release Case Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Keat, C.L.; Aziz, A.; Eid, A.M.; Elmarzugi, N.A. Biosynthesis of nanoparticles and silver nanoparticles. Bioresour. Bioprocess. 2015, 2, 47. [Google Scholar] [CrossRef] [Green Version]
- Lowry, G.V.; Gregory, K.B.; Apte, S.C.; Lead, J.R. Transformations of nanomaterials in the environment. Environ. Sci. Technol. 2012, 46, 6893–6899. [Google Scholar] [CrossRef]
- Janković, N.Z.; Plata, D.L. Engineered nanomaterials in the context of global element cycles. Environ. Sci. Nano 2019, 6, 2697–2711. [Google Scholar] [CrossRef] [Green Version]
- Pulit-Prociak, J.; Banach, M. Silver nanoparticles—A material of the future...? Open Chem. 2016, 14, 76–91. [Google Scholar] [CrossRef]
- Sohn, E.K.; Johari, S.A.; Kim, T.G.; Kim, J.K.; Kim, E.; Lee, J.H.; Chung, Y.S.; Yu, I.J. Aquatic toxicity comparison of silver nanoparticles and silver nanowires. Biomed. Res. Int. 2015, 2015, 893049. [Google Scholar] [CrossRef] [PubMed]
- Cui, R.; Chae, Y.; An, Y.J. Dimension-dependent toxicity of silver nanomaterials on the cladocerans Daphnia magna and Daphnia galeata. Chemosphere 2017, 185, 205–212. [Google Scholar] [CrossRef]
- Silva, T.; Pokhrel, L.R.; Dubey, B.; Tolaymat, T.M.; Maier, K.J.; Liu, X. Particle size, surface charge and concentration dependent ecotoxicity of three organo-coated silver nanoparticles: Comparison between general linear model-predicted and observed toxicity. Sci. Tot Environ. 2014, 468–469, 968–976. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Zhou, Y.; Wang, C.; Li, S.; Wang, X. Toxic Effects and Molecular Mechanism of Different Types of Silver Nanoparticles to the Aquatic Crustacean Daphnia magna. Environ. Sci. Technol. 2017, 51, 12868–12878. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Ma, B.; Pan, T.; Chen, S.; Sun, J. Silver-Nanoparticle-Colored Cotton Fabrics with Tunable Colors and Durable Antibacterial and Self-Healing Superhydrophobic Properties. Adv. Funct. Mater. 2016, 26, 569–576. [Google Scholar] [CrossRef]
- Gottschalk, F.; Nowack, B. The release of engineered nanomaterials to the environment. J. Environ. Monit. 2011, 13, 1145–1155. [Google Scholar] [CrossRef]
- Nowack, B.; Ranville, J.F.; Diamond, S.; Gallego-Urrea, J.A.; Metcalfe, C.; Rose, J.; Horne, N.; Koelmans, A.A.; Klaine, S.J. Potential scenarios for nanomaterial release and subsequent alteration in the environment. Environ. Toxicol. Chem. 2012, 31, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Lassen, C.; Kjoelholt, J.; Christensen, F.; Nowack, B. Modeling flows and concentrations of nine engineered nanomaterials in the Danish environment. Int. J. Environ. Res. Public Health 2015, 12, 5581–5602. [Google Scholar] [CrossRef] [Green Version]
- Sanchís, J.; Jiménez-Lamana, J.; Abad, E.; Szpunar, J.; Farré, M. Occurrence of Cerium-, Titanium-, and Silver-Bearing Nanoparticles in the Besòs and Ebro Rivers. Environ. Sci. Technol. 2020, 54, 3969–3978. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Sonderer, T.; Scholz, R.W.; Nowack, B. Modeled environmental concentrations of engineered nanomaterials (TiO2, ZnO, Ag, CNT, fullerenes) for different regions. Environ. Sci. Technol. 2009, 43, 9216–9222. [Google Scholar] [CrossRef]
- Johnson, A.; Cisowska, I.; Jürgens, M.; Keller, V.; Lawlor, A.; Williams, R. Exposure Assessment for Engineered Silver Nanoparticles throughout the Rivers of England and Wales (CB0433); UK Centre For Ecology & Hydrology: Lancaster, UK, 2011. [Google Scholar]
- Maurer-Jones, M.A.; Gunsolus, I.L.; Murphy, C.J.; Haynes, C.L. Toxicity of engineered nanoparticles in the environment. Anal. Chem. 2013, 85, 3036–3049. [Google Scholar] [CrossRef] [Green Version]
- Johnson, A.C.; Jürgens, M.D.; Lawlor, A.J.; Cisowska, I.; Williams, R.J. Particulate and colloidal silver in sewage effluent and sludge discharged from British wastewater treatment plants. Chemosphere 2014, 112, 49–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Stoiber, M.; Wimmer, A.; Xu, Z.; Lindenblatt, C.; Helmreich, B.; Schuster, M. To What Extent Can Full-Scale Wastewater Treatment Plant Effluent Influence the Occurrence of Silver-Based Nanoparticles in Surface Waters? Environ. Sci. Technol. 2016, 50, 6327–6333. [Google Scholar] [CrossRef] [PubMed]
- Bäuerlein, P.S.; Emke, E.; Tromp, P.; Hofman, J.A.M.H.; Carboni, A.; Schooneman, F.; de Voogt, P.; van Wezel, A.P. Is there evidence for man-made nanoparticles in the Dutch environment? Sci. Total Environ. 2017, 576, 273–283. [Google Scholar] [CrossRef]
- Markus, A.A.; Krystek, P.; Tromp, P.C.; Parsons, J.R.; Roex, E.W.M.; de Voogt, P.; Laane, R.W.P.M. Determination of metal-based nanoparticles in the river Dommel in the Netherlands via ultrafiltration, HR-ICP-MS and SEM. Sci. Total Environ. 2018, 631–632, 485–495. [Google Scholar] [CrossRef]
- Peters, R.J.B.; van Bemmel, G.; Milani, N.B.L.; den Hertog, G.C.T.; Undas, A.K.; van der Lee, M.; Bouwmeester, H. Detection of nanoparticles in Dutch surface waters. Sci. Total Environ. 2018, 621, 210–218. [Google Scholar] [CrossRef]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of Nanoparticle Toxicity on Their Physical and Chemical Properties. Nanoscale Res. Lett. 2018, 13, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Comandella, D.; Gottardo, S.; Rio-Echevarria, I.M.; Rauscher, H. Quality of physicochemical data on nanomaterials: An assessment of data completeness and variability. Nanoscale 2020, 12, 4695–4708. [Google Scholar] [CrossRef] [Green Version]
- Akter, M.; Sikder, M.T.; Rahman, M.M.; Ullah, A.K.M.A.; Hossain, K.F.B.; Banik, S.; Hosokawa, T.; Saito, T.; Kurasaki, M. A systematic review on silver nanoparticles-induced cytotoxicity: Physicochemical properties and perspectives. J. Adv. Res. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Tortella, G.R.; Rubilar, O.; Durán, N.; Diez, M.C.; Martínez, M.; Parada, J.; Seabra, A.B. Silver nanoparticles: Toxicity in model organisms as an overview of its hazard for human health and the environment. J. Hazard. Mater. 2020, 390, 121974. [Google Scholar] [CrossRef] [PubMed]
- Krysanov, E.Y.; Pavlov, D.S.; Demidova, T.B.; Dgebuadze, Y.Y. Effect of nanoparticles on aquatic organisms. Biol. Bull. 2010, 37, 406–412. [Google Scholar] [CrossRef]
- Liu, H.; Wang, X.; Wu, Y.; Hou, J.; Zhang, S.; Zhou, N.; Wang, X. Toxicity responses of different organs of zebrafish (Danio rerio) to silver nanoparticles with different particle sizes and surface coatings. Environ. Pollut. 2019, 246, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Boyle, D.; Goss, G.G. Effects of silver nanoparticles in early life-stage zebrafish are associated with particle dissolution and the toxicity of soluble silver. NanoImpact 2018, 12, 1–8. [Google Scholar] [CrossRef]
- Sorensen, S.N.; Baun, A. Controlling silver nanoparticle exposure in algal toxicity testing-A matter of timing. Nanotoxicology 2015, 9, 201–209. [Google Scholar] [CrossRef] [Green Version]
- Kleiven, M.; Macken, A.; Oughton, D.H. Growth inhibition in Raphidocelis subcapita—Evidence of nanospecific toxicity of silver nanoparticles. Chemosphere 2019, 221, 785–792. [Google Scholar] [CrossRef]
- Sakka, Y.; Skjolding, L.M.; Mackevica, A.; Filser, J.; Baun, A. Behavior and chronic toxicity of two differently stabilized silver nanoparticles to Daphnia magna. Aquat. Toxicol. 2016, 177, 526–535. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Dumitrescu, E.; Kumar, A.; Austin, D.; Goia, D.; Wallace, K.N.; Andreescu, S. Differential lethal and sublethal effects in embryonic zebrafish exposed to different sizes of silver nanoparticles. Environ. Pollut. 2019, 248, 627–634. [Google Scholar] [CrossRef]
- Abramenko, N.B.; Demidova, T.B.; Abkhalimov, E.V.; Ershov, B.G.; Krysanov, E.Y.; Kustov, L.M. Ecotoxicity of different-shaped silver nanoparticles: Case of zebrafish embryos. J. Hazard. Mater. 2018, 347, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.H.; An, Y.J. Size- and shape-dependent toxicity of silver nanomaterials in green alga Chlorococcum infusionum. Ecotoxicol. Environ. Saf. 2019, 168, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Malysheva, A.; Voelcker, N.; Holm, P.E.; Lombi, E. Unraveling the complex behavior of AgNPs driving NP-cell interactions and toxicity to algal cells. Environ. Sci. Technol. 2016, 50, 12455–12463. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, J.; Ginebreda, A.; Guillén, D.; Martínez, E.; Barceló, D.; Moragas, L.; Robusté, J.; Darbra, R.M. Fuzzy logic based risk assessment of effluents from waste-water treatment plants. Sci. Total Environ. 2012, 439, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Seguí, X.; Pujolasus, E.; Betrò, S.; Àgueda, A.; Casal, J.; Ocampo-Duque, W.; Rudolph, I.; Barra, R.; Páez, M.; Barón, E.; et al. Fuzzy model for risk assessment of persistent organic pollutants in aquatic ecosystems. Environ. Pollut. 2013, 178, 23–32. [Google Scholar] [CrossRef]
- Topuz, E.; van Gestel, C.A.M. An approach for environmental risk assessment of engineered nanomaterials using Analytical Hierarchy Process (AHP) and fuzzy inference rules. Environ. Int. 2016, 92–93, 334–347. [Google Scholar] [CrossRef] [PubMed]
- Ocampo-Duque, W.; Ferré-Huguet, N.; Domingo, J.L.; Schuhmacher, M. Assessing water quality in rivers with fuzzy inference systems: A case study. Environ. Int. 2006, 32, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Zou, Z.; An, Y. Water quality assessment in Qu River based on fuzzy water pollution index method. J. Environ. Sci. 2016, 50, 87–92. [Google Scholar] [CrossRef]
- Tashayo, B.; Alimohammadi, A.; Sharif, M. A hybrid fuzzy inference system based on dispersion model for quantitative environmental health impact assessment of urban transportation planning. Sustainability 2017, 9, 134. [Google Scholar] [CrossRef] [Green Version]
- De Paula, D.L.M.; de Lima, A.C.; de Vinagre, M.V.; Pontes, A.N. Sanitation on fluvial passenger ships in amazônia: An analysis of risks to the environment and to health through fuzzy logic. Eng. Sanit. Ambient. 2019, 24, 283–294. [Google Scholar] [CrossRef]
- Jahangoshai Rezaee, M.; Yousefi, S.; Eshkevari, M.; Valipour, M.; Saberi, M. Risk analysis of health, safety and environment in chemical industry integrating linguistic FMEA, fuzzy inference system and fuzzy DEA. Stoch. Environ. Res. Risk Assess. 2020, 34, 201–218. [Google Scholar] [CrossRef]
- Betrò, S. Environmental Risk Assessment of Polybrominated Diphenyl Ether (PBDE) and Hexabromocyclododecane (HBCD) in Ebro River Basin. Master’s Thesis, Universitat Politecnica de Catalunya, Barcelona, Spain, February 2011. [Google Scholar]
- González, R.J.; Darbra, R.M.; Arnaldos, J. Using fuzzy logic to introduce the human factor in the failure frequency estimation of storage vessels in chemical plants. Chem. Eng. Trans. 2013, 32, 193–198. [Google Scholar] [CrossRef]
- Yoo-iam, M.; Chaichana, R.; Satapanajaru, T. Toxicity, bioaccumulation and biomagnification of silver nanoparticles in green algae (Chlorella sp.), water flea (Moina macrocopa), blood worm (Chironomus spp.) and silver barb (Barbonymus gonionotus). Chem. Speciat. Bioavailab. 2014, 26, 257–265. [Google Scholar] [CrossRef]
- Tolaymat, T.M.; El Badawy, A.M.; Genaidy, A.; Scheckel, K.G.; Luxton, T.P.; Suidan, M. An evidence-based environmental perspective of manufactured silver nanoparticle in syntheses and applications: A systematic review and critical appraisal of peer-reviewed scientific papers. Sci. Total Environ. 2010, 408, 999–1006. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jurašin, D.D.; Ćurlin, M.; Capjak, I.; Crnković, T.; Lovrić, M.; Babič, M.; Horák, D.; Vrček, I.V.; Gajović, S. Surface coating affects behavior of metallic nanoparticles in a biological environment. Beilstein J. Nanotechnol. 2016, 7, 246–262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huertas, E.; Folch, M.; Salgot, M.; Gonzalvo, I.; Passarell, C. Constructed wetlands effluent for streamflow augmentation in the Besòs River (Spain). Desalination 2006, 188, 141–147. [Google Scholar] [CrossRef]
- Catalunya Parks The Besòs River Park. Available online: https://parcs.diba.cat/es/web/fluvial/el-parc-fluvial (accessed on 14 October 2021).
- Plasmachem Silver Nanoparticles Additional Product Information. Available online: https://shop.plasmachem.com/index.php?controller=attachment&id_attachment=37 (accessed on 17 September 2021).
- Rajaee, T. Wavelet and neuro-fuzzy conjunction approach for suspended sediment prediction. Clean Soil Air Water 2010, 38, 275–286. [Google Scholar] [CrossRef]
- Rajaee, T.; Mirbagheri, S.A.; Nourani, V.; Alikhani, A. Prediction of daily suspended sediment load using wavelet and neuro-fuzzy combined model. Int. J. Environ. Sci. Technol. 2010, 7, 93–110. [Google Scholar] [CrossRef] [Green Version]
- Stolze, L.; Volpin, F. Modeling of the Elk river spill 2014. Environ. Sci. Pollut. Res. 2015, 22, 7980–7985. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Fu, H.; Skøtt, K.; Yang, M. Modeling the spill in the Songhua River after the explosion in the petrochemical plant in Jilin. Environ. Sci. Pollut. Res. 2008, 15, 178–181. [Google Scholar] [CrossRef] [Green Version]
- Rivord, J.; Saito, L.; Miller, G.; Stoddard, S.S. Modeling Contaminant Spills in the Truckee River in the Western United States. J. Water Resour. Plan. Manag. 2014, 140, 343–354. [Google Scholar] [CrossRef]
- Sethi, R.; Di Molfetta, A. Analytical Solutions of the Differential Equation of Mass Transport for Reactive Solutes. In Groundwater Engineering; Springer: Cham, Switzerland, 2019; pp. 239–247. [Google Scholar] [CrossRef]
- Fischer, H.B. Discussion of ‘Simple method for predicting dispersion in stream’. J. Environ. Eng. Div. 1975, 101, 453–455. [Google Scholar] [CrossRef]
- Agencia Catalana de l’Aigua Estacions Depuradores d’aigua Residual. Available online: http://aca.gencat.cat/web/.content/20_Aigua/02_infraestructures/05_estacions_depuradores_daigues_residuals/LlistatEDAR_Servei.xlsx (accessed on 17 September 2021).
- Agencia Catalana de l’Aigua. Consulta de les Dades de Control de la Qualitat i la Quantitat de l’aigua al medi. Available online: http://aca-web.gencat.cat/sdim21/seleccioXarxes.do;jsessionid=4B6442745A6C3695855A89488EBD1B3F (accessed on 17 September 2021).
- Agencia Catalana de l’Aigua (ACA). El agua en Tiempo Real. Available online: http://aca-web.gencat.cat/aetr/vishid (accessed on 17 September 2021).
- USEPA. Risk Assessment Guidance for Superfund (RAGS): Process for Conducting Probabilistic Risk Assessment Volume 3A; USEPA: Washington, DC, USA, 2001. [Google Scholar]
- Ferraro, D.O. Fuzzy knowledge-based model for soil condition assessment in Argentinean cropping systems. Environ. Model. Softw. 2009, 24, 359–370. [Google Scholar] [CrossRef]


| Data Type | Concentration (ng L−1) | Location | Reference |
|---|---|---|---|
| WWTP Effluents | |||
| Estimated | 32.9–11 | Europe | [14] |
| Estimated | 16.4–74.7 | EE. UU | [14] |
| Estimated | 29.8–127 | Switzerland | [14] |
| Estimated | <0.5–12.7 | United Kingdom | [15] |
| Estimated | 16.4–17000 | Global | [16] |
| Estimated | 0.012–59 | Denmark | [12] |
| Measured | 2.7–12.7 | United Kingdom | [17] |
| Measured | 0.7–11 | Germany | [18] |
| Measured | 1000–1700 | Netherlands | [19] |
| Measured | 13 | Netherlands | [20] |
| Measured | 0–7.2 * | Spain | [13] |
| Surface Waters | |||
| Estimated | 0.588–2.16 | Europe | [14] |
| Estimated | 0.088–0.428 | EE. UU | [14] |
| Estimated | 0.555–2.63 | Switzerland | [14] |
| Estimated | 0.088–10,000 | Global | [16] |
| Estimated | 0–0.044 | Denmark | [12] |
| Measured | 2–8.6 | Isar River, Germany | [18] |
| Measured | 0.5–1.3 | Chiemsee Lake, Germany | [18] |
| Measured | 0.3–6.6 | Meuse River, Netherlands | [21] |
| Measured | 0.3–2.5 | IJssel River, Netherlands | [21] |
| Measured | 30 | Dommel River, Netherlands | [20] |
| Measured | 0.4–0.9 * | Besòs River, Spain | [13] |
| Measured | 0.4–0.7 * | Ebro River, Spain | [13] |
| Organism | Shape * | Size ** (nm) | Coating Type *** | End Point | Concentration μg/L | Conclusions | References |
|---|---|---|---|---|---|---|---|
| Daphnia magna | Sph | 11 | BPEI | EC50 Mortality 48 h | 0.41 | Regarding coating type, toxicity levels were as follows: BPEI > Citrate > PVP. BPEIs caused significantly higher daphnid mortality, whereas PVP-AgNPs resulted in the least toxicity. | [32] |
| Cit | 2.88 | ||||||
| PVP | 4.79 | ||||||
| Daphnia magna | Sph | 40 | Cit | EC50 Mortality 24 h | 8.9 | Citrate-coated AgNPs were more toxic than PVP-coated AgNPs. Additionally, the smaller their size the higher their toxicity. | [8] |
| 110 | 17.43 | ||||||
| 40 | PVP | 24.97 | |||||
| 110 | 38.35 | ||||||
| Daphnia magna | Sph | 56.6 | PVP | EC50 Mortality 48 h | 44.83 | NWs have the lowest toxicity. PL, with the smallest size, exhibited the highest degree of toxicity compared to other shapes. | [6] |
| NWs | 41.3 L = 10,000 | 256.2 | |||||
| 42.1 L = 20,000 | 247.1 | ||||||
| PL | 30 | 27.92 | |||||
| Oryzias latipes | Sph | 35 | N/A | LC50 Mortality 72 h | 1800 | Sph were estimated to be more than twice as toxic as NWs. | [5] |
| NWs | L = 7400 | 4180 | |||||
| Danio rerio | Sph | 20 | Cit | LC50 Mortality 96 h | 200 | Citrate-AgNPs were more toxic than PVP-AgNPs, and 20-nm AgNPs were more toxic than 100-nm AgNPs. | [27] |
| 100 | 400 | ||||||
| 20 | PVP | 400 | |||||
| 100 | 800 | ||||||
| Danio rerio | Sph | 10.1 | Cit and PVP | LC50 Mortality 96 h | 41.5 | PL induced higher toxicity than spheres, even at larger particle sizes. | [33] |
| PL | 33.8 | 16.9 | |||||
| Pseudokirchneriella subcapitata | Sph | 30 | Cit | EC50 Inhibition 48 h | 310 | Smaller AgNPs were the most toxic. | [29] |
| 15 | 75 | ||||||
| 30 | EC50 Assimilation 2 h | 710 | |||||
| 15 | 150 | ||||||
| Chlorococcum infusionum | Sph | 57 | PVP | EC50 Mortality 72 h | 100 | Regarding shape, toxicity level was as follows: Plates > Wires > Sph. | [34] |
| NWs | 42 L = 21,000 | 45 | |||||
| PL | 40 | 21 | |||||
| Pseudokirchneriella subcapitata | Sph | 10 | Cit | EC50 Inhibition 48 h | 23.13 | Citrate-coated AgNPs were more toxic than those coated with BPEI, with the exception of the 10 nm BPEI AgNPs, which showed similar toxicity to the 10 nm Citrate AgNPs. | [35] |
| 30 | 38.28 | ||||||
| 70 | 118.1 | ||||||
| 10 | BPEI | 22.92 | |||||
| 30 | 67.10 | ||||||
| 70 | 307.4 |
| Variables | Fuzzy Set | Ranges | MF Types |
|---|---|---|---|
| Shape * | Wires | 0–5 | Z Shape |
| Spheres | 2.5–7.5 | Pi Shape | |
| Plates | 5–10 | S Shape | |
| Size ** | Very Small | 5–30 nm | Z Shape |
| Small | 15–45 nm | Pi Shape | |
| Medium | 35–75 nm | Pi Shape | |
| Big | 65–95 nm | Pi Shape | |
| Very big | 80–450 nm | Z Shape | |
| Coating * | PVP | 0–5 | Z Shape |
| Citrate | 2.5–7.5 | Pi Shape | |
| BPEI | 5–10 | S Shape | |
| Toxicity * | Low | 0–0.5 | Z Shape |
| Medium | 0.2–0.8 | Pi Shape | |
| High | 0.5–1 | S Shape | |
| Concentration | Low | 0–500 ng L−1 | Z Shape |
| Medium | 250–750 ng L−1 | Pi Shape | |
| High | 500–1000 *** ng L−1 | S Shape | |
| Risk * | Very Low | 0–0.25 | Z Shape |
| Low | 0–0.5 | Pi Shape | |
| Medium | 0.25–0.75 | Pi Shape | |
| High | 0.5–1 | Pi Shape | |
| Very High | 0.75–1 | Z Shape |
| Variables | WWTP Effluents | Accidental Spill | Section | |
|---|---|---|---|---|
| Analytical Model (see Supplementary Materials) | ||||
| Source | (L) | N/A | 200 | S2.1 Supplementary material |
| t0 (s) | Continuous | 1000 ** | ||
| (m3 s−1) | 0.6–0.84 * | 0.0002 | ||
| M (kg) | N/A | 0.02 | ||
| (ng L−1) | 3.8 | 108 | ||
| River | (m3 s−1) | 2–52 * | 2.473 | S2.2 Supplementary material |
| h (m) | N/A | 0.14 | ||
| W (m) | N/A | 29 | ||
| Section (m2) | N/A | 4.1 | ||
| (m2 s−1) | N/A | 402 | ||
| v (m s−1) | N/A | 0.61 | ||
| x (m) | N/A | 5000 ** | ||
| Fuzzy Logic | ||||
| AgNPs | Size (nm) | 16 | 10 | 2 Article |
| Shape (spheres, wires, plate) | Spheres–Citrate; Plates–BPEI;Wires–PVP | Spheres–Citrate;Plates–BPEI; | ||
| Coating (PVP, citrate, BPEI) | ||||
| WWTP Particle Types | |||
|---|---|---|---|
| Variable | Spheres Citrate | Plates BPEI | Wires PVP |
| Size (nm) | 16 | 16 | 16 |
| Shape * | 5 | 10 | 0 |
| Coating * | 5 | 10 | 0 |
| Toxicity * | 0.515 | 0.855 | 0.145 |
| Concentration in the river (ng L−1) | 0–3.7 ** | 0–3.7 ** | 0–3.7 ** |
| Risk (fuzzy value) * | 0.25 | 0.5 | 0.07 |
| Risk (fuzzy qualitative value) | 100% Low | 100% Medium | 83% Very low–17% Low |
| Accidental Spill Particle Types | ||
|---|---|---|
| Variables | Spheres Citrate | Plates BPEI |
| Size (nm) | 10 | 10 |
| Shape * | 5 | 10 |
| Coating * | 5 | 10 |
| Toxicity * | 0.515 | 0.855 |
| Concentration in the river (ng L−1) | 0–1000 ** | 0–1000 ** |
| Risk (fuzzy value) * | 0.76 | 0.93 |
| Maximum Risk (fuzzy qualitative value) | 100% High | 81.5% Very High– 18.5% High |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramirez, R.; Martí, V.; Darbra, R.M. Environmental Risk Assessment of Silver Nanoparticles in Aquatic Ecosystems Using Fuzzy Logic. Water 2022, 14, 1885. https://doi.org/10.3390/w14121885
Ramirez R, Martí V, Darbra RM. Environmental Risk Assessment of Silver Nanoparticles in Aquatic Ecosystems Using Fuzzy Logic. Water. 2022; 14(12):1885. https://doi.org/10.3390/w14121885
Chicago/Turabian StyleRamirez, Rosember, Vicenç Martí, and Rosa Mari Darbra. 2022. "Environmental Risk Assessment of Silver Nanoparticles in Aquatic Ecosystems Using Fuzzy Logic" Water 14, no. 12: 1885. https://doi.org/10.3390/w14121885
APA StyleRamirez, R., Martí, V., & Darbra, R. M. (2022). Environmental Risk Assessment of Silver Nanoparticles in Aquatic Ecosystems Using Fuzzy Logic. Water, 14(12), 1885. https://doi.org/10.3390/w14121885







