Characterization of South African Brewery Wastewater: Oxidation-Reduction Potential Variation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Brewery Wastewater Sample
2.2. Analytical Technique
2.3. Statistical Analysis
3. Results and Discussion
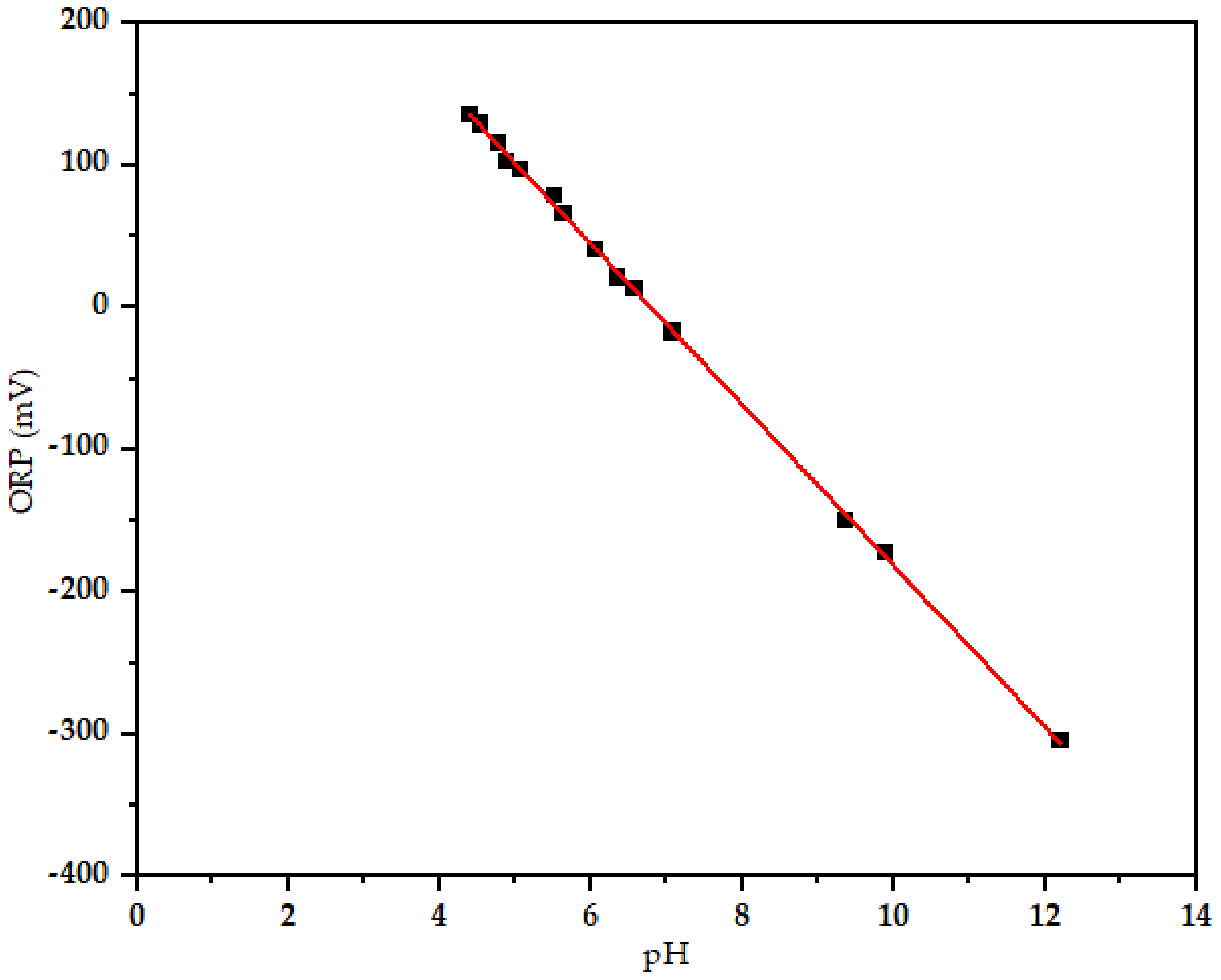
3.1. Oxidation Reduction Potential (ORP) versus pH
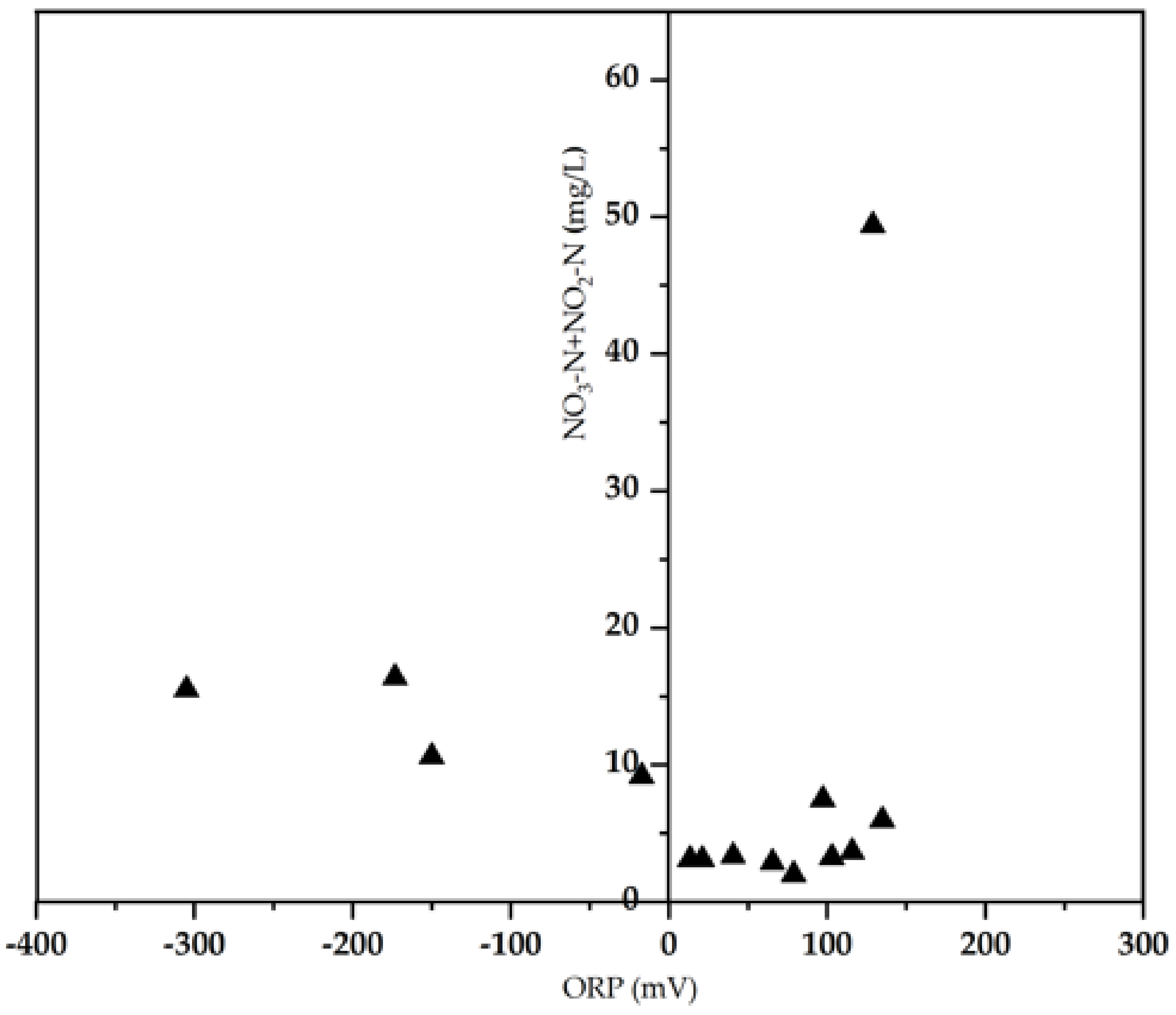
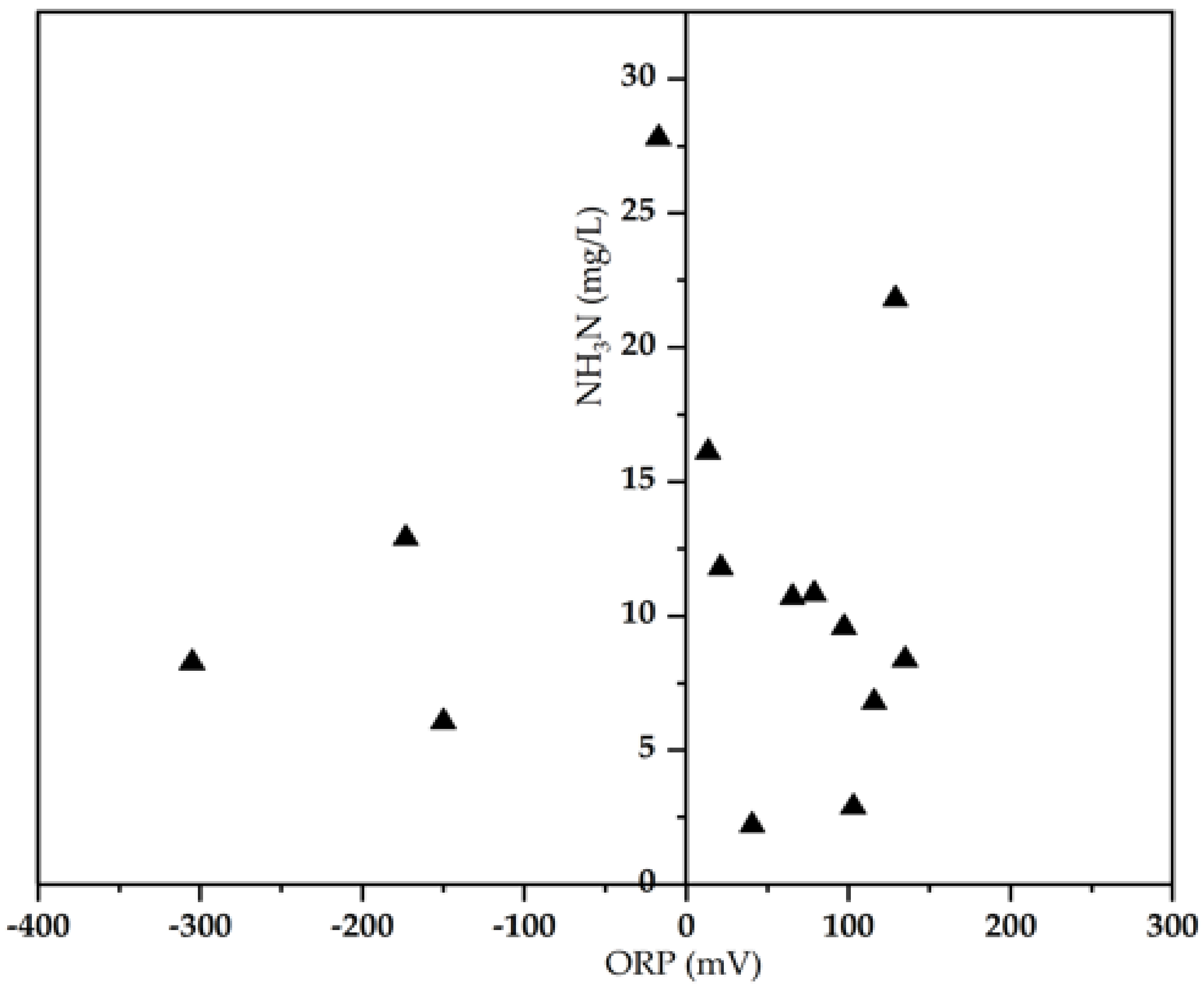
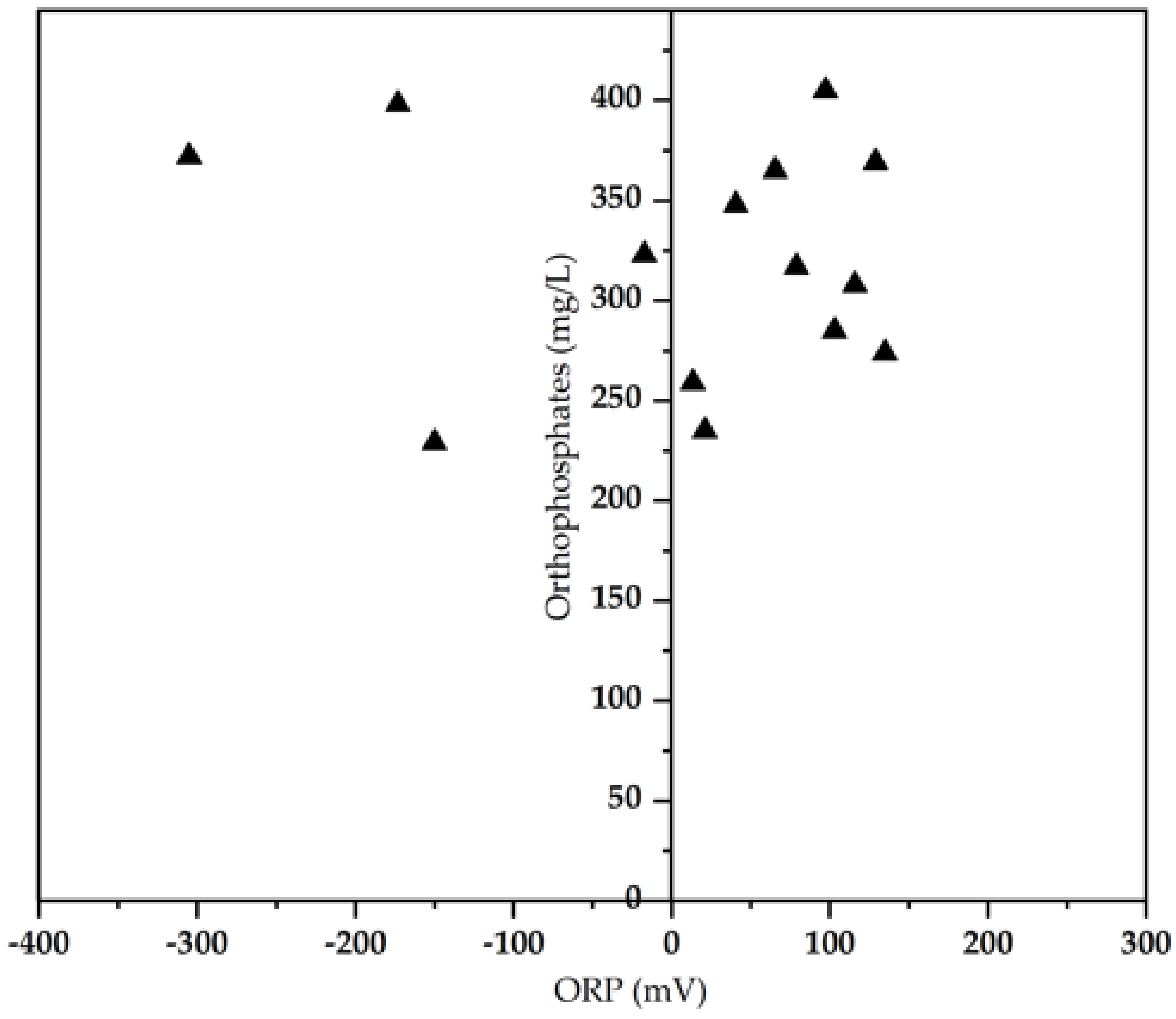
3.2. Biological Pollutants Composition versus ORP
3.3. Brewery Wastewater Composition
3.4. Orthophosphate Material Balance
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simate, G.S. The treatment of brewery wastewater for reuse by integration of coagulation/flocculation and sedimentation with carbon nanotubes ‘sandwitched’ in a granular filter bed. J. Ind. Eng. Chem. 2015, 21, 1277–1285. [Google Scholar] [CrossRef]
- Chen, H.; Chang, S.; Guo, Q.; Hong, Y.; Wu, P. Brewery wastewater treatment using an anaerobic membrane bioreactor. Biochem. J. 2016, 105, 321–331. [Google Scholar] [CrossRef]
- Enitan, A.M.; Adeyemo, J.; Kumari, S.; Swalaha, F.M.; Bux, F. Characterization of Brewery Wastewater Composition. Int. J. Environ. Eng. 2015, 9, 1073–1076. [Google Scholar] [CrossRef]
- Tetteh, E.K.; Asante-Sackey, D.; Armah, E.K.; Rathilal, S. Tapping wastewater resource: Why and how? In Handbook of Biofuels; Sahay, S., Ed.; Academic Press: Bhopal, India, 2022; pp. 125–146. [Google Scholar]
- Kumar, J.A.; Sathish, S.; Krithiga, T.; Praveenkumar, T.R.; Lokesh, S.; Prabu, D.; Renita, A.A.; Prakash, P.; Rajasimman, M. A comprehensive review on bio-hydrogen production from brewery industrial wastewater and its treatment methodologies. Fuel 2022, 319, 123–594. [Google Scholar]
- Simpson, W.J.; Hammond, J.R.M. The response of brewing yeasts to acid washing. J. Inst. Brew. 1989, 95, 347–354. [Google Scholar] [CrossRef]
- Corsino, S.F.; di Biase, A.; Devlin, T.R.; Munz, G.; Torregrossa, M.; Oleszkiewicz, J.A. Effects of extended famine conditions on aerobic granular sludge stability in the treatment of brewery wastewater. Bioresour. Technol. 2017, 226, 150–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, F.; Huang, Z.; Miao, H.; Ren, H.; Zhao, M.; Ruan, W. Identical full-scale biogas-lift reactors (BLRs) with anaerobic granular sludge and residual activated sludge for brewery wastewater treating and kinetic modeling. Res. J. Environ. Sci. 2013, 25, 2031–2040. [Google Scholar]
- Goldammer, T. The Brewers’s Handbook; Keen Vision Publishing: Huntsville, AL, USA, 1999. [Google Scholar]
- Simate, G.S.; Cluett, J.; Iyuke, S.E.; Musapatika, E.T.; Ndlovu, S.; Walubita, L.F.; Alvarez, A.E. The treatment of brewery wastewater for reuse: State of the art. Desalination 2011, 273, 235–247. [Google Scholar] [CrossRef]
- Atalay, S.; Ersöz, G. Novel Catalysts in Advanced Oxidation of Organic Pollutants; Springer International Publishing: New York, NY, USA, 2016; pp. 23–34. [Google Scholar]
- Li, B.; Bishop, P.L. Oxidation-Reduction Potential Chnages in Aeration Tanks and Microprofiles of Activated Sludge Floc in Medium and Low-Strength Wastewaters. Water Environ. Res. 2004, 76, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Charpentier, J.; Godart, G.; Martin, G.; Mogno, Y. Oxidation-Reduction Potential (ORP) regulation as a way to optimize aeration and C, N and P removal: Experimental basis and various full-scale examples. Water Sci. Technol. 1989, 21, 1209–1223. [Google Scholar] [CrossRef]
- Copeland, A.; Lytle, D.A. Measuring the oxidation-reduction potential of important oxidants in drinking water. J. Am. Water Works Assoc. 2014, 106, E10–E20. [Google Scholar] [CrossRef]
- Environmental Yellow Springs Instruments. Available online: http://www.ysi.com/media/pdfs/A567-ORP-Management-in-Wastewater-as-an-Indicator-of-Process-Efficiency.pdf (accessed on 1 May 2022).
- Chen, K.C.; Chen, C.Y.; Peng, J.W.; Houng, J.Y. Real-time control of an immobilized-cell reactor for wastewater treatment using ORP. Water Res. 2002, 36, 230–238. [Google Scholar] [CrossRef]
- Nghiem, L.D.; Manassa, P.; Dawson, M.; Fitzgerald, S.K. Oxidation reduction potential as a parameter to regulate micro-oxygen injection into anaerobic digester for reducing hydrogen sulphide concentration in biogas. Bioresour. Technol. 2014, 173, 443–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, R.F.; Chen, H.W.; Cheng, W.P.; Lin, T.Y.J.; Huang, C.L. Monitoring of ORP, pH and DO in heterogeneous Fenton oxidation using nZVI as a catalyst for the treatment of azo-dye textile wastewater. J. Taiwan Inst. Chem. Eng. 2014, 45, 947–954. [Google Scholar] [CrossRef]
- Wang, M.; Loccozia, J.; Sun, L.; Lin, C.; Lin, Z. Inorganic-modified semiconductor TiO2 nantotube arrays for photocatalysis. Enegry Environ. Sci. 2014, 7, 2182–2202. [Google Scholar]
- Sambaza, S.S.; Maity, A.; Pillay, K. Polyaniline-coated TiO2 nanorods for photocatalytic degradation of bisphenol A in water. ACS Omega 2020, 5, 29642–29656. [Google Scholar] [CrossRef] [PubMed]
- APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Kent, R.; Johnson, T.D.; Rosen, M.R. Status and trends of orthophosphate concentrations in groundwater used for public supply in California. Environ. Monit. Assess. 2020, 192, 550. [Google Scholar] [CrossRef] [PubMed]

| Biological Reaction | ORP Range (mV) |
|---|---|
| Nitrification | +100 to +350 |
| BOD degradation | +50 to +250 |
| Biological phosphorus removal | +25 to 250 |
| Denitrification | −50 to +50 |
| Biological phosphorus release | −100 to −250 |
| Parameter | Mean ± SD | Range |
|---|---|---|
| Temperature (°C) | 31 ± 3.7 | 25.3–37 |
| pH | 6.5 ± 2.4 | 4.4–12.2 |
| ORP (mV) | 13.7 ± 1.4 | −305–135 |
| Conductivity (µS/cm) | 2718 ± 10.20 | 1893–6017 |
| Turbidity (NTU) | 570 ± 16.4 | 303–1039 |
| TCOD (mg/L) | 7687 ± 20.30 | 3447–11,813 |
| SCOD (mg/L) | 6323 ± 15.42 | 2287–8627 |
| PCOD (mg/L) | 1454 ± 91.7 | 127–3693 |
| PO43− (mg/L) | 343 ± 6.4 | 229–424 |
| NH3-N (mg/L) | 12.2 ± 7.5 | 2.21–27.8 |
| TKN (mg/L) | 29.3 ± 2.6 | 6.24–94.7 |
| Total Nitrogen (mg/L) | 38.6 ± 2.9 | 13.7–106 |
| NO3-N + NO2-N (mg/L) | 10 ± 1.1 | 2.87–49.4 |
| Total Organic Nitrogen (mg/L) | 8.92 ± 11.1 | 0–39.1 |
| Total Inorganic Nitrogen (mg/L) | 34.4 ± 2.2 | 7.78–93 |
| Total solids (mg/L) | 5951 ± 33.9 | 2942–14,981 |
| Total Dissolved Solids (mg/L) | 4121 ± 15.03 | 2198–7400 |
| Fixed VS (mg/L) | 2327 ± 11.2 | 825–4975 |
| Volatile Suspended Solids (mg/L) | 1799 ± 57.1 | 1043–2572 |
| Parameter | Present Study | [17] | [18] |
|---|---|---|---|
| Temperature (°C) | 25.3–37 | 24–30.5 | 25–35 |
| pH | 4.4–12.2 | 4.6–7.3 | 3.3–6.3 |
| ORP (mV) | −305–135 | --- | --- |
| Conductivity (µS/cm) | 1893–6017 | 1.044–1.622 | --- |
| Turbidity (NTU) | 303–1039 | --- | --- |
| TCOD (mg/L) | 3447–11,813 | 1096–8926 | 8240–20,000 |
| SCOD (mg/L) | 2287–8627 | 1178–5847 | --- |
| PCOD (mg/L) | 127–3693 | --- | --- |
| PO43− (mg/L) | 229–424 | 7.51–74.1 | 16–124 |
| NH3-N (mg/L) | 2.21–27.8 | 0.48–13.1 | --- |
| TKN (mg/L) | 6.24–94.7 | --- | --- |
| Total Nitrogen (mg/L) | 13.7–106 | --- | 0.0196–0.0336 |
| NO3-N + NO2-N (mg/L) | 2.87–49.4 | 1.14–11.6 | --- |
| Total Organic Nitrogen (mg/L) | 0–39.1 | 0–5.36 | --- |
| Total Inorganic Nitrogen (mg/L) | 7.78–93 | --- | --- |
| Total Organic Nitrogen (mg/L) | 2942–14,981 | 1289–12,248 | 5100–8750 |
| Total Inorganic Nitrogen (mg/L) | 2198–7400 | --- | 2020–5940 |
| Total solids (mg/L) | 825–4975 | --- | --- |
| Total Dissolved Solids (mg/L) | 1043–2572 | 804–1278 | --- |
| Batch No | SBR Influent (g/d) | SBR Consumption (g/d) | SBR Effluent (g/d) | SBR Removal Efficiency (%) |
|---|---|---|---|---|
| 1 | 0.275 | 0.091 | 0.184 | 33 |
| 2 | 0.317 | 0.130 | 0.187 | 41 |
| 3 | 0.235 | 0.169 | 0.066 | 72 |
| 4 | 0.229 | 0.169 | 0.060 | 74 |
| 5 | 0.285 | 0.225 | 0.055 | 79 |
| 6 | 0.274 | 0.219 | 0.073 | 80 |
| 7 | 0.348 | 0.275 | 0.247 | 79 |
| 8 | 0.398 | 0.151 | 0.148 | 38 |
| 9 | 0.308 | 0.160 | 0.066 | 52 |
| 10 | 0.365 | 0.299 | 0.093 | 82 |
| 11 | 0.405 | 0.312 | 0.054 | 77 |
| 12 | 0.259 | 0.205 | 0.061 | 79 |
| 13 | 0.323 | 0.262 | 0.089 | 81 |
| 14 | 0.372 | 0.283 | 0.187 | 76 |
| 15 | 0.424 | 0.237 | 0.133 | 56 |
| 16 | 0.369 | 0.236 | 0.118 | 64 |
| 17 | 0.423 | 0.305 | 0.118 | 72 |
| 18 | 0.396 | 0.317 | 0.079 | 80 |
| 19 | 0.392 | 0.306 | 0.086 | 78 |
| 20 | 0.403 | 0.326 | 0.077 | 81 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khumalo, S.M.; Bakare, B.F.; Rathilal, S.; Tetteh, E.K. Characterization of South African Brewery Wastewater: Oxidation-Reduction Potential Variation. Water 2022, 14, 1604. https://doi.org/10.3390/w14101604
Khumalo SM, Bakare BF, Rathilal S, Tetteh EK. Characterization of South African Brewery Wastewater: Oxidation-Reduction Potential Variation. Water. 2022; 14(10):1604. https://doi.org/10.3390/w14101604
Chicago/Turabian StyleKhumalo, Siphesihle Mangena, Babatunde Femi Bakare, Sudesh Rathilal, and Emmanuel Kweinor Tetteh. 2022. "Characterization of South African Brewery Wastewater: Oxidation-Reduction Potential Variation" Water 14, no. 10: 1604. https://doi.org/10.3390/w14101604
APA StyleKhumalo, S. M., Bakare, B. F., Rathilal, S., & Tetteh, E. K. (2022). Characterization of South African Brewery Wastewater: Oxidation-Reduction Potential Variation. Water, 14(10), 1604. https://doi.org/10.3390/w14101604









