Application of Extraction and Determination Based on Deep Eutectic Solvents in Different Types of Environmental Samples
Abstract
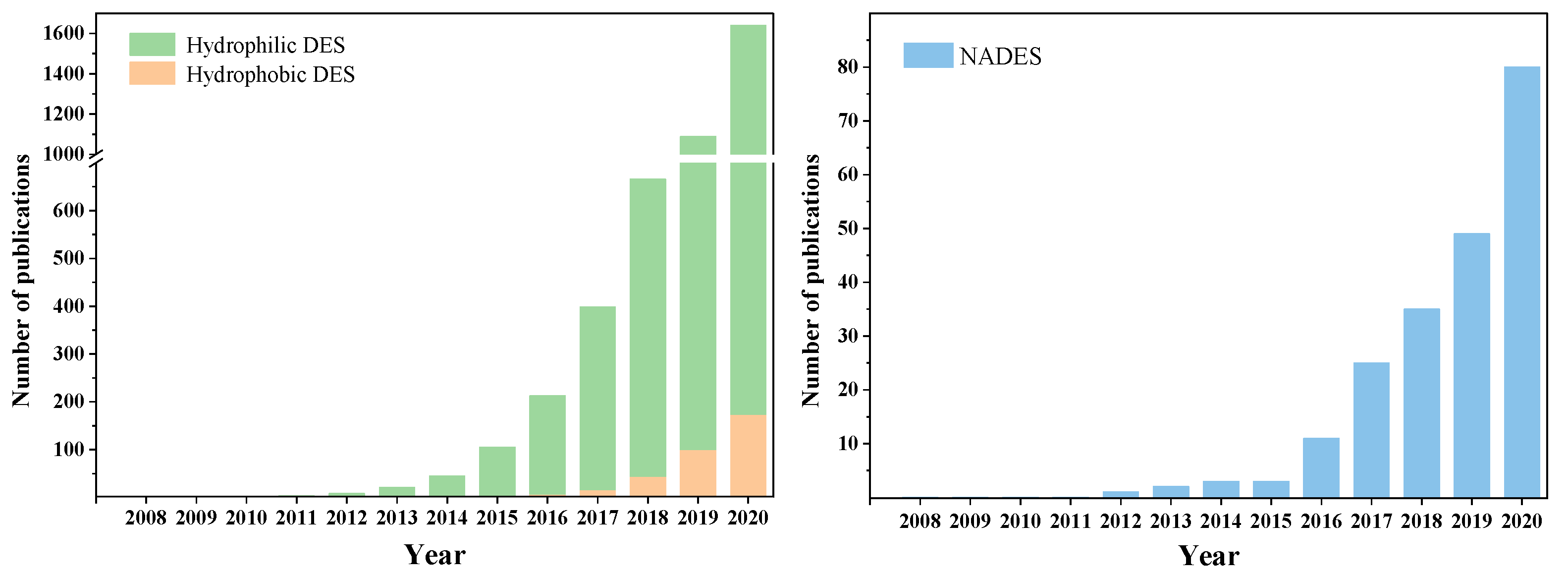
:1. Introduction
2. Deep Eutectic Solvents
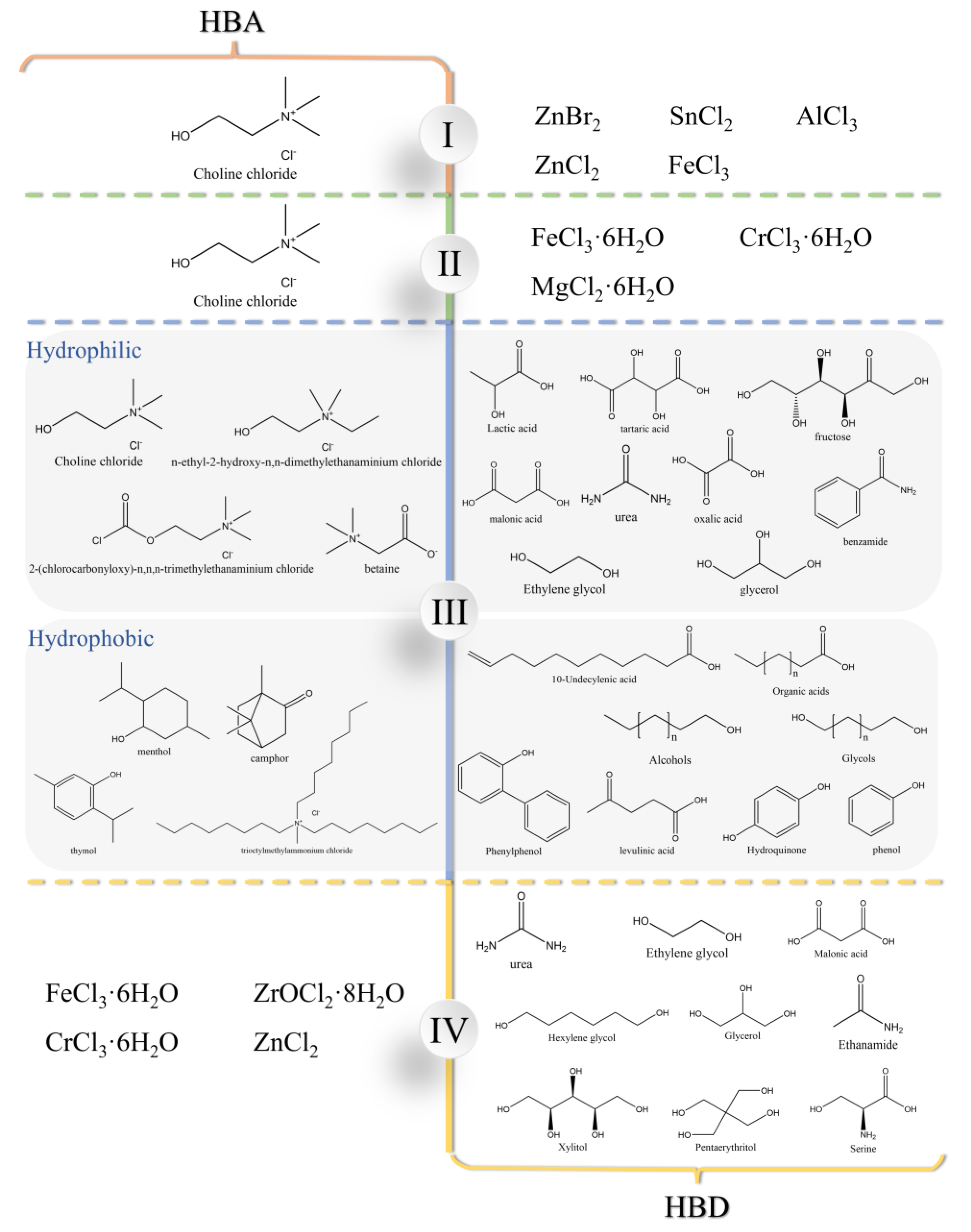
2.1. Classification of DESs
2.2. Hydrophilic and Hydrophobic DESs
2.3. Toxicity of DES
3. Application Forms in Environmental Analysis
| Techniques | DES | Sample | Other Features | Analytes | Instrumental Analysis | LOD (ug/L,g) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| HBA | HBD | Molar Ratio | Volume | Type | Volume | |||||
| SLLE [52] | N8881-Cl | Octanol/octanoic acid | 1:2:3 | 2 mL | Plant leaves | 0.2 g | Two DES phases were involved | Flavonoids Terpene trilactones Procyanidin Polyprenyl acetates | HPLC-UV | |
| LLE [53] | Menthol | Dodecanoic acid | 2:1 | Water | Lower alcohols Ethanol 1-Propanol 1-Butanol | NMR | ||||
| LLE [54] | Dodecanoic acid | Octanoic acid Nonanoic acid Decanoic acid | 1:3 1:3 1:2 | 2 mL | Water | 2 mL | Bisphenol A | UV–vis | ||
| DLLME [55] | N8881-Cl | Oleic acid | 1:2 | 20 uL | Water and biological samples | 5 mL | Vortex assisted | Nitrite | HPLC-UV | 0.2 |
| DLLME [56] | Quaternary ammonium salt | DL-menthol | Aqueous samples | Air assisted | Benzophenone | HPLC-UV | ||||
| DLLME [57] | ChCl | Triethylamine | 1:1 | Biological and environmental samples | 20 mL | Air assisted Volume of DES/triethylamine (TEA) (1:1) is 100 uL. | Heavy Metals | FAAS | 0.31–0.99 | |
| DLLME [58] | ChCl | Phenol | 1:3 | 450 uL | Lake water | 10 mL | Ultrasound assisted | Chromium (III/VI) | FAAS | 5.5 |
| DLLME [59] | ChCl | Phenol | 1:3 | 1000 uL | Soil, sediment, and water | 25 mL | Ultrasound assisted | Arsenic | ETAAS | 0.01 |
| DLLME [60] | N4444-Cl | Decanoic acid | 1:2 | 200 uL | Liver samples | 10 mL | Ultrasound assisted DES (ChCl-lactic acid) is digestion solution | Copper | MS-FAAS | 4.00 |
| HS-SDME [61] | N4444-Br | Dodecanol | 1:2 | 1.5 uL | Plant samples | 50 mg | Terpenes | GC-MS | 0.87–86.40 | |
| HF-LPME [62] | ChCl | Phenylethanol | 1:4 | 40 uL | Human plasma urine and pharmaceutical wastewater | 10 mL | Three-phase (liquid–liquid–liquid) microextraction | Antiarrhythmic agents Propranolol Carvedilol Verapamil Amlodipine | HPLC-UV | |
3.1. DLLME
3.2. SDME
3.3. HF-LPME
4. Applications in Environmental Matrix
4.1. Extraction from Aqueous Samples
4.2. Extraction from Air Samples and Soil/Sediment Samples
4.3. Extraction from Organism Samples
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Abbreviations | |
| AA | Air assisted |
| BTRs | Benzotriazole derivatives |
| BTs | Benzothiazole derivatives |
| ChCl | Choline chloride |
| DES | Deep eutectic solvent |
| DLLME | Dispersive liquid–liquid microextraction |
| DNA | Deoxyribonucleic acid |
| [EMIM][OAc] | 1-Ethyl-3-methyl imidazolium acetate |
| HBA | Hydrogen bond acceptor |
| HBD | Hydrogen bond donor |
| HDESs | Hydrophobic deep eutectic solvent |
| HF-LPME | Fiber-based liquid-phase microextraction |
| HPLC | High-performance liquid chromatography |
| HS-SDME | Headspace single-drop microextraction |
| LLE | Liquid-liquid extraction |
| LPME | Liquid-phase microextraction |
| MA | Microwave assisted |
| MTPB | Methyltriphenylphosphonium bromide |
| NADES | Nature deep eutectic solvent |
| PAHs | Polycyclic aromatic hydrocarbons |
| SAs | Sulfonamides |
| SDME | Single-drop microextraction |
| SLM | Supported liquid membrane |
| SPE | Solid-phase extraction |
| SPME | Solid-phase microextraction |
| TBAB | Tetrabutylammonium bromide |
| THF | Tetrahydrofuran |
| UA | Ultrasound-assisted |
| VA-LLME | Vortex-assisted liquid–liquid microextraction |
| Nomenclature | |
| -- | Dipole–dipole forces |
| ρ | Density |
| -- | Ion–dipole forces |
| mp | Melting point |
| S | Solubility |
| -- | Polarity |
| ƞ | Viscosity |
| -- | van der Waals (dispersion) forces |
| -- | π–π forces |
References
- Acosta-Rodríguez, I.; Rodríguez-Pérez, A.; Pacheco-Castillo, N.; Enríquez-Domínguez, E.; Cárdenas-González, J.; Martínez-Juárez, V.-M. Removal of Cobalt (II) from Waters Contaminated by the Biomass of Eichhornia crassipes. Water 2021, 13, 1725. [Google Scholar] [CrossRef]
- Maurya, P.K.; Ali, S.A.; Alharbi, R.S.; Yadav, K.K.; Alfaisal, F.M.; Ahmad, A.; Ditthakit, P.; Prasad, S.; Jung, Y.-K.; Jeon, B.-H. Impacts of Land Use Change on Water Quality Index in the Upper Ganges River near Haridwar, Uttarakhand: A GIS-Based Analysis. Water 2021, 13, 3572. [Google Scholar] [CrossRef]
- Rahmawati, R.R.; Putro, A.H.S.; Lee, J.L. Analysis of Long-Term Shoreline Observations in the Vicinity of Coastal Structures: A Case Study of South Bali Beaches. Water 2021, 13, 3527. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 39, 70–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, B.; Zhang, H.; Row, K.H. Application of deep eutectic solvents in the extraction and separation of target compounds from various samples. J. Sep. Sci. 2015, 38, 1053–1064. [Google Scholar] [CrossRef]
- Shishov, A.; Bulatov, A.; Locatelli, M.; Carradori, S.; Andruch, V. Application of deep eutectic solvents in analytical chemistry. A review. Microchem. J. 2017, 135, 33–38. [Google Scholar] [CrossRef]
- Ndizeye, N.; Suriyanarayanan, S.; Nicholls, I.A. Polymer synthesis in non-ionic deep eutectic solvents. Polym. Chem. 2019, 10, 5289–5295. [Google Scholar] [CrossRef] [Green Version]
- Nkuku, C.A.; LeSuer, R.J. Electrochemistry in Deep Eutectic Solvents. J. Phys. Chem. B 2007, 111, 13271–13277. [Google Scholar] [CrossRef]
- Santana-Mayor, Á.; Rodríguez-Ramos, R.; Herrera-Herrera, A.V.; Socas-Rodríguez, B.; Rodríguez-Delgado, M. Ángel Deep eutectic solvents. The new generation of green solvents in analytical chemistry. TrAC Trends Anal. Chem. 2021, 134, 116108. [Google Scholar] [CrossRef]
- Gomez, F.J.V.; Espino, M.; Fernández, M.A.; Silva, M.F. A Greener Approach to Prepare Natural Deep Eutectic Solvents. Chem. 2018, 3, 6122–6125. [Google Scholar] [CrossRef]
- Faraji, M. Novel hydrophobic deep eutectic solvent for vortex assisted dispersive liquid-liquid micro-extraction of two auxins in water and fruit juice samples and determination by high performance liquid chromatography. Microchem. J. 2019, 150, 104130. [Google Scholar] [CrossRef]
- Makoś, P.; Słupek, E.; Gębicki, J. Hydrophobic deep eutectic solvents in microextraction techniques–A review. Microchem. J. 2020, 152, 104384. [Google Scholar] [CrossRef]
- Tang, W.; An, Y.; Row, K.H. Emerging applications of (micro) extraction phase from hydrophilic to hydrophobic deep eutectic solvents: Opportunities and trends. TrAC Trends Anal. Chem. 2021, 136, 116187. [Google Scholar] [CrossRef]
- Musarurwa, H.; Tavengwa, N.T. Deep eutectic solvent-based dispersive liquid-liquid micro-extraction of pesticides in food samples. Food Chem. 2021, 342, 127943. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.; Sun, D.; Yang, S.; Liu, H.; Chen, L. Strategies of dispersive liquid-liquid microextraction for coastal zone environmental pollutant determination. J. Chromatogr. A 2021, 1658, 462615. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Hu, X.; Wang, J.; Cheng, H.; Chen, L.; Qi, Z. Overview of acidic deep eutectic solvents on synthesis, properties and applications. Green Energy Environ. 2020, 5, 8–21. [Google Scholar] [CrossRef]
- Choi, S.-A.; Lee, J.-S.; Oh, Y.-K.; Jeong, M.-J.; Kim, S.W.; Park, J.-Y. Lipid extraction from Chlorella vulgaris by molten-salt/ionic-liquid mixtures. Algal Res. 2014, 3, 44–48. [Google Scholar] [CrossRef]
- Abbott, A.P.; Frisch, G.; Hartley, J.; Ryder, K.S. Processing of metals and metal oxides using ionic liquids. Green Chem. 2011, 13, 471–481. [Google Scholar] [CrossRef]
- Abbott, A.P.; Barron, J.C.; Ryder, K.; Wilson, D. Eutectic-Based Ionic Liquids with Metal-Containing Anions and Cations. Chem. Eur. J. 2007, 13, 6495–6501. [Google Scholar] [CrossRef]
- Abbott, A.P.; Al-Barzinjy, A.A.; Abbott, P.D.; Frisch, G.; Harris, R.C.; Hartley, J.; Ryder, K.S. Speciation, physical and electrolytic properties of eutectic mixtures based on CrCl3·6H2O and urea. Phys. Chem. Chem. Phys. 2014, 16, 9047–9055. [Google Scholar] [CrossRef] [Green Version]
- Shahabi, D.; Tavakol, H. One-pot synthesis of quinoline derivatives using choline chloride/tin (II) chloride deep eutectic solvent as a green catalyst. J. Mol. Liq. 2016, 220, 324–328. [Google Scholar] [CrossRef]
- Liu, F.; Xue, Z.; Zhao, X.; Mou, H.; He, J.; Mu, T. Catalytic deep eutectic solvents for highly efficient conversion of cellulose to gluconic acid with gluconic acid self-precipitation separation. Chem. Commun. 2018, 54, 6140–6143. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Zhao, J.; Duan, H.; Guan, Y.; Zhao, L. Green and efficient extraction of four bioactive flavonoids from Pollen Typhae by ultrasound-assisted deep eutectic solvents extraction. J. Pharm. Biomed. Anal. 2018, 161, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.-S.; Zhong, Z.-F.; Bian, G.-L.; Cheng, X.-J.; Li, D.-Q. Ultra-rapid, enhanced and eco-friendly extraction of four main flavonoids from the seeds of Oroxylum indicum by deep eutectic solvents combined with tissue-smashing extraction. Food Chem. 2020, 319, 126555. [Google Scholar] [CrossRef] [PubMed]
- El Kantar, S.; Rajha, H.N.; Boussetta, N.; Vorobiev, E.; Maroun, R.G.; Louka, N. Green extraction of polyphenols from grapefruit peels using high voltage electrical discharges, deep eutectic solvents and aqueous glycerol. Food Chem. 2019, 295, 165–171. [Google Scholar] [CrossRef]
- Ali, M.C.; Chen, J.; Zhang, H.; Li, Z.; Zhao, L.; Qiu, H. Effective extraction of flavonoids from Lycium barbarum L. fruits by deep eutectic solvents-based ultrasound-assisted extraction. Talanta 2019, 203, 16–22. [Google Scholar] [CrossRef]
- Ozturk, B.; Parkinson, C.; Gonzalez-Miquel, M. Extraction of polyphenolic antioxidants from orange peel waste using deep eutectic solvents. Sep. Purif. Technol. 2018, 206, 1–13. [Google Scholar] [CrossRef]
- Nie, J.; Chen, D.; Lu, Y. Deep Eutectic Solvents Based Ultrasonic Extraction of Polysaccharides from Edible Brown Seaweed Sargassum horneri. J. Mar. Sci. Eng. 2020, 8, 440. [Google Scholar] [CrossRef]
- Gao, C.; Cai, C.; Liu, J.; Wang, Y.; Chen, Y.; Wang, L.; Tan, Z. Extraction and preliminary purification of polysaccharides from Camellia oleifera Abel. seed cake using a thermoseparating aqueous two-phase system based on EOPO copolymer and deep eutectic solvents. Food Chem. 2020, 313, 126164. [Google Scholar] [CrossRef]
- Shafie, M.H.; Yusof, R.; Gan, C.-Y. Deep eutectic solvents (DES) mediated extraction of pectin from Averrhoa bilimbi: Optimization and characterization studies. Carbohydr. Polym. 2019, 216, 303–311. [Google Scholar] [CrossRef]
- van Osch, D.J.; Zubeir, L.F.; Bruinhorst, A.V.D.; Rocha, M.A.; Kroon, M.C. Hydrophobic deep eutectic solvents as water-immiscible extractants. Green Chem. 2015, 17, 4518–4521. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, B.D.; Florindo, C.; Iff, L.C.; Coelho, M.A.Z.; Marrucho, I. Menthol-based Eutectic Mixtures: Hydrophobic Low Viscosity Solvents. ACS Sustain. Chem. Eng. 2015, 3, 2469–2477. [Google Scholar] [CrossRef]
- Martins, M.A.R.; Crespo, E.A.; Pontes, P.V.A.; Silva, L.P.; Bülow, M.; Maximo, G.J.; Batista, E.A.C.; Held, C.; Pinho, S.P.; Coutinho, J.A.P. Tunable Hydrophobic Eutectic Solvents Based on Terpenes and Monocarboxylic Acids. ACS Sustain. Chem. Eng. 2018, 6, 8836–8846. [Google Scholar] [CrossRef]
- Makoś, P.; Przyjazny, A.; Boczkaj, G. Hydrophobic deep eutectic solvents as “green” extraction media for polycyclic aromatic hydrocarbons in aqueous samples. J. Chromatogr. A 2018, 1570, 28–37. [Google Scholar] [CrossRef]
- van Osch, D.J.G.P.; Parmentier, D.; Dietz, C.H.J.T.; Bruinhorst, A.V.D.; Tuinier, R.; Kroon, M.C. Removal of alkali and transition metal ions from water with hydrophobic deep eutectic solvents. Chem. Commun. 2016, 52, 11987–11990. [Google Scholar] [CrossRef] [Green Version]
- Cao, J.; Yang, M.; Cao, F.; Wang, J.; Su, E. Well-Designed Hydrophobic Deep Eutectic Solvents As Green and Efficient Media for the Extraction of Artemisinin from Artemisia annua Leaves. ACS Sustain. Chem. Eng. 2017, 5, 3270–3278. [Google Scholar] [CrossRef]
- Křížek, T.; Bursová, M.; Horsley, R.; Kuchař, M.; Tuma, P.; Čabala, R.; Hložek, T. Menthol-based hydrophobic deep eutectic solvents: Towards greener and efficient extraction of phytocannabinoids. J. Clean. Prod. 2018, 193, 391–396. [Google Scholar] [CrossRef]
- Paul, N.; Naik, P.K.; Ribeiro, B.D.; Pattader, P.S.G.; Marrucho, I.M.; Banerjee, T. Molecular Dynamics Insights and Water Stability of Hydrophobic Deep Eutectic Solvents Aided Extraction of Nitenpyram from an Aqueous Environment. J. Phys. Chem. B 2020, 124, 7405–7420. [Google Scholar] [CrossRef]
- Florindo, C.; Lima, F.; Branco, L.C.; Marrucho, I.M. Hydrophobic Deep Eutectic Solvents: A Circular Approach to Purify Water Contaminated with Ciprofloxacin. ACS Sustain. Chem. Eng. 2019, 7, 14739–14746. [Google Scholar] [CrossRef]
- Shishov, A.; Chislov, M.; Nechaeva, D.; Moskvin, L.; Bulatov, A. A new approach for microextraction of non-steroidal anti-inflammatory drugs from human urine samples based on in-situ deep eutectic mixture formation. J. Mol. Liq. 2018, 272, 738–745. [Google Scholar] [CrossRef]
- Florindo, C.; Monteiro, N.V.; Ribeiro, B.D.; Branco, L.; Marrucho, I. Hydrophobic deep eutectic solvents for purification of water contaminated with Bisphenol-A. J. Mol. Liq. 2020, 297, 111841. [Google Scholar] [CrossRef]
- Bystrzanowska, M.; Tobiszewski, M. Assessment and design of greener deep eutectic solvents—A multicriteria decision analysis. J. Mol. Liq. 2021, 321, 114878. [Google Scholar] [CrossRef]
- Hayyan, M.; Hashim, M.A.; Hayyan, A.; Al-Saadi, M.A.; AlNashef, I.M.; Mirghani, M.E.; Saheed, O.K. Are deep eutectic solvents benign or toxic? Chemosphere 2013, 90, 2193–2195. [Google Scholar] [CrossRef]
- Radošević, K.; Zeleznjak, J.; Bubalo, M.C.; Redovniković, I.R.; Slivac, I.; Srček, V.G. Comparative in vitro study of cholinium-based ionic liquids and deep eutectic solvents toward fish cell line. Ecotoxicol. Environ. Saf. 2016, 131, 30–36. [Google Scholar] [CrossRef]
- Wen, Q.; Chen, J.-X.; Tang, Y.-L.; Wang, J.; Yang, Z. Assessing the toxicity and biodegradability of deep eutectic solvents. Chemosphere 2015, 132, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Torregrosa-Crespo, J.; Marset, X.; Guillena, G.; Ramón, D.J.; Martínez-Espinosa, R.M. New guidelines for testing “Deep eutectic solvents” toxicity and their effects on the environment and living beings. Sci. Total Environ. 2020, 704, 135382. [Google Scholar] [CrossRef] [PubMed]
- Macário, I.; Oliveira, H.; Menezes, A.C.; Ventura, S.; Pereira, J.L.; Gonçalves, A.M.M.; Coutinho, J.; Gonçalves, F.J.M. Cytotoxicity profiling of deep eutectic solvents to human skin cells. Sci. Rep. 2019, 9, 3932. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macário, I.P.; Jesus, F.; Pereira, J.L.; Ventura, S.P.; Gonçalves, A.M.; Coutinho, J.A.; Gonçalves, F.J. Unraveling the ecotoxicity of deep eutectic solvents using the mixture toxicity theory. Chemosphere 2018, 212, 890–897. [Google Scholar] [CrossRef]
- Li, K.; Jin, Y.; Jung, D.; Park, K.; Kim, H.; Lee, J. In situ formation of thymol-based hydrophobic deep eutectic solvents: Application to antibiotics analysis in surface water based on liquid-liquid microextraction followed by liquid chromatography. J. Chromatogr. A 2020, 1614, 460730. [Google Scholar] [CrossRef] [PubMed]
- Ge, D.; Wang, Y.; Jiang, Q.; Dai, E. A Deep Eutectic Solvent as an Extraction Solvent to Separate and Preconcentrate Parabens in Water Samples Using in situ Liquid-Liquid Microextraction. J. Braz. Chem. Soc. 2019, 30, 1203–1210. [Google Scholar] [CrossRef]
- Zarei, A.R.; Nedaei, M.; Ghorbanian, S.A. Ferrofluid of magnetic clay and menthol based deep eutectic solvent: Application in directly suspended droplet microextraction for enrichment of some emerging contaminant explosives in water and soil samples. J. Chromatogr. A 2018, 1553, 32–42. [Google Scholar] [CrossRef]
- Cao, J.; Chen, L.; Li, M.; Cao, F.; Zhao, L.; Su, E. Two-phase systems developed with hydrophilic and hydrophobic deep eutectic solvents for simultaneously extracting various bioactive compounds with different polarities. Green Chem. 2018, 20, 1879–1886. [Google Scholar] [CrossRef]
- Verma, R.; Banerjee, T. Liquid–Liquid Extraction of Lower Alcohols Using Menthol-Based Hydrophobic Deep Eutectic Solvent: Experiments and COSMO-SAC Predictions. Ind. Eng. Chem. Res. 2018, 57, 3371–3381. [Google Scholar] [CrossRef]
- Florindo, C.; Romero, L.; Rintoul, I.; Branco, L.C.; Marrucho, I.M. From Phase Change Materials to Green Solvents: Hydrophobic Low Viscous Fatty Acid–Based Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2018, 6, 3888–3895. [Google Scholar] [CrossRef]
- Zhang, K.; Li, S.; Liu, C.; Wang, Q.; Wang, Y.; Fan, J. A hydrophobic deep eutectic solvent-based vortex-assisted dispersive liquid-liquid microextraction combined with HPLC for the determination of nitrite in water and biological samples. J. Sep. Sci. 2019, 42, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Ge, D.; Zhang, Y.; Dai, Y.; Yang, S. Air-assisted dispersive liquid-liquid microextraction based on a new hydrophobic deep eutectic solvent for the preconcentration of benzophenone-type UV filters from aqueous samples. J. Sep. Sci. 2018, 41, 1635–1643. [Google Scholar] [CrossRef]
- Ezoddin, M.; Lamei, N.; Siami, F.; Abdi, K.; Karimi, M.A. Deep Eutectic Solvent Based Air Assisted Ligandless Emulsification Liquid–Liquid Microextraction for Preconcentration of Some Heavy Metals in Biological and Environmental Samples. Bull. Environ. Contam. Toxicol. 2018, 101, 814–819. [Google Scholar] [CrossRef]
- Yilmaz, E.; Soylak, M. Ultrasound assisted-deep eutectic solvent based on emulsification liquid phase microextraction combined with microsample injection flame atomic absorption spectrometry for valence speciation of chromium(III/VI) in environmental samples. Talanta 2016, 160, 680–685. [Google Scholar] [CrossRef]
- Zounr, R.A.; Tuzen, M.; Khuhawar, M.Y. Ultrasound assisted deep eutectic solvent based on dispersive liquid liquid microextraction of arsenic speciation in water and environmental samples by electrothermal atomic absorption spectrometry. J. Mol. Liq. 2017, 242, 441–446. [Google Scholar] [CrossRef]
- Kanberoglu, G.S.; Yilmaz, E.; Soylak, M. Usage of deep eutectic solvents for the digestion and ultrasound-assisted liquid phase microextraction of copper in liver samples. J. Iran. Chem. Soc. 2018, 15, 2307–2314. [Google Scholar] [CrossRef]
- Triaux, Z.; Petitjean, H.; Marchioni, E.; Boltoeva, M.; Marcic, C. Deep eutectic solvent–based headspace single-drop microextraction for the quantification of terpenes in spices. Anal. Bioanal. Chem. 2020, 412, 933–948. [Google Scholar] [CrossRef] [PubMed]
- Rajabi, M.; Ghassab, N.; Hemmati, M.; Asghari, A. Highly effective and safe intermediate based on deep eutectic medium for carrier less-three phase hollow fiber microextraction of antiarrhythmic agents in complex matrices. J. Chromatogr. B 2019, 1104, 196–204. [Google Scholar] [CrossRef]
- Wu, B.; Guo, Z.; Li, X.; Huang, X.; Teng, C.; Chen, Z.; Jing, X.; Zhao, W. Analysis of pyrethroids in cereals by HPLC with a deep eutectic solvent-based dispersive liquid–liquid microextraction with solidification of floating organic droplets. Anal. Methods 2021, 13, 636–641. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhu, T. Emulsification liquid–liquid microextraction based on deep eutectic solvents: An extraction method for the determination of sulfonamides in water samples. Anal. Methods 2017, 9, 4747–4753. [Google Scholar] [CrossRef]
- Werner, J. Novel deep eutectic solvent-based ultrasounds-assisted dispersive liquid-liquid microextraction with solidification of the aqueous phase for HPLC-UV determination of aromatic amines in environmental samples. Microchem. J. 2020, 153, 104405. [Google Scholar] [CrossRef]
- El-Deen, A.K.; Shimizu, K. Deep eutectic solvent as a novel disperser in dispersive liquid-liquid microextraction based on solidification of floating organic droplet (DLLME-SFOD) for preconcentration of steroids in water samples: Assessment of the method deleterious impact on the environment using Analytical Eco-Scale and Green Analytical Procedure Index. Microchem. J. 2019, 149, 103988. [Google Scholar] [CrossRef]
- Wang, H.; Xu, Q.; Jiao, J.; Wu, H. A solidified floating organic drop-dispersive liquid–liquid microextraction based on in situ formed fatty acid-based deep eutectic solvents for the extraction of benzophenone-UV filters from water samples. New J. Chem. 2021, 45, 14082–14090. [Google Scholar] [CrossRef]
- Lamei, N.; Ezoddin, M.; Abdi, K. Air assisted emulsification liquid-liquid microextraction based on deep eutectic solvent for preconcentration of methadone in water and biological samples. Talanta 2017, 165, 176–181. [Google Scholar] [CrossRef]
- Kailasa, S.K.; Koduru, J.R.; Park, T.J.; Singhal, R.K.; Wu, H.-F. Applications of single-drop microextraction in analytical chemistry: A review. Trends Environ. Anal. Chem. 2021, 29, e00113. [Google Scholar] [CrossRef]
- Farooq, M.Q.; Zeger, V.R.; Anderson, J.L. Comparing the extraction performance of cyclodextrin-containing supramolecular deep eutectic solvents versus conventional deep eutectic solvents by headspace single drop microextraction. J. Chromatogr. A 2021, 1658, 462588. [Google Scholar] [CrossRef]
- Yousefi, S.M.; Shemirani, F.; Ghorbanian, S.A.; Ali, S. Enhanced headspace single drop microextraction method using deep eutectic solvent based magnetic bucky gels: Application to the determination of volatile aromatic hydrocarbons in water and urine samples. J. Sep. Sci. 2017, 41, 966–974. [Google Scholar] [CrossRef]
- Abbasi-Ahd, A.; Shokoufi, N.; Kargosha, K. Headspace single-drop microextraction coupled to microchip-photothermal lens microscopy for highly sensitive determination of captopril in human serum and pharmaceuticals. Microchim. Acta 2017, 184, 2403–2409. [Google Scholar] [CrossRef]
- Pedersen-Bjergaard, S.; Rasmussen, K.E. Liquid−Liquid−Liquid Microextraction for Sample Preparation of Biological Fluids Prior to Capillary Electrophoresis. Anal. Chem. 1999, 71, 2650–2656. [Google Scholar] [CrossRef]
- Khataei, M.M.; Yamini, Y.; Nazaripour, A.; Karimi, M. Novel generation of deep eutectic solvent as an acceptor phase in three-phase hollow fiber liquid phase microextraction for extraction and preconcentration of steroidal hormones from biological fluids. Talanta 2018, 178, 473–480. [Google Scholar] [CrossRef]
- Hansen, F.A.; Santigosa-Murillo, E.; Ramos-Payán, M.; Muñoz, M.; Øiestad, E.L.; Pedersen-Bjergaard, S. Electromembrane extraction using deep eutectic solvents as the liquid membrane. Anal. Chim. Acta 2021, 1143, 109–116. [Google Scholar] [CrossRef]
- Chen, Z.; Reznicek, W.D.; Wan, C. Deep eutectic solvent pretreatment enabling full utilization of switchgrass. Bioresour. Technol. 2018, 263, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Feng, F.; Jiang, J.; Qiao, Y.; Wu, T.; Voglmeir, J.; Chen, Z.-G. Green and efficient extraction of rutin from tartary buckwheat hull by using natural deep eutectic solvents. Food Chem. 2017, 221, 1400–1405. [Google Scholar] [CrossRef]
- Dai, Y.; Rozema, E.; Verpoorte, R.; Choi, Y.H. Application of natural deep eutectic solvents to the extraction of anthocyanins from Catharanthus roseus with high extractability and stability replacing conventional organic solvents. J. Chromatogr. A 2016, 1434, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Matong, J.M.; Nyaba, L.; Nomngongo, P.N. Determination of As, Cr, Mo, Sb, Se and V in agricultural soil samples by inductively coupled plasma optical emission spectrometry after simple and rapid solvent extraction using choline chloride-oxalic acid deep eutectic solvent. Ecotoxicol. Environ. Saf. 2017, 135, 152–157. [Google Scholar] [CrossRef]
- Bağda, E.; Altundağ, H.; Tüzen, M.; Soylak, M. A Novel Selective Deep Eutectic Solvent Extraction Method for Versatile Determination of Copper in Sediment Samples by ICP-OES. Bull. Environ. Contam. Toxicol. 2017, 99, 264–269. [Google Scholar] [CrossRef]
- Rajabi, M.; Ghassab, N.; Hemmati, M.; Asghari, A. Emulsification microextraction of amphetamine and methamphetamine in complex matrices using an up-to-date generation of eco-friendly and relatively hydrophobic deep eutectic solvent. J. Chromatogr. A 2018, 1576, 1–9. [Google Scholar] [CrossRef]
- Florindo, C.; Branco, L.; Marrucho, I. Development of hydrophobic deep eutectic solvents for extraction of pesticides from aqueous environments. Fluid Phase Equilibria 2017, 448, 135–142. [Google Scholar] [CrossRef]
- Nedaei, M.; Zarei, A.R.; Ghorbanian, S.A. Miniaturized matrix solid-phase dispersion based on deep eutectic solvent and carbon nitride associated with high-performance liquid chromatography: A new feasibility for extraction and determination of trace nitrotoluene pollutants in soil samples. J. Chromatogr. A 2019, 1601, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Kachangoon, R.; Vichapong, J.; Santaladchaiyakit, Y.; Burakham, R.; Srijaranai, S. An Eco-Friendly Hydrophobic Deep Eutectic Solvent-Based Dispersive Liquid–Liquid Microextraction for the Determination of Neonicotinoid Insecticide Residues in Water, Soil and Egg Yolk Samples. Molecules 2020, 25, 2785. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Liu, C.; Li, S.; Fan, J. A hydrophobic deep eutectic solvent based vortex-assisted liquid-liquid microextraction for the determination of formaldehyde from biological and indoor air samples by high performance liquid chromatography. J. Chromatogr. A 2019, 1589, 39–46. [Google Scholar] [CrossRef]
- Ruggeri, S.; Poletti, F.; Zanardi, C.; Pigani, L.; Zanfrognini, B.; Corsi, E.; Dossi, N.; Salomäki, M.; Kivelä, H.; Lukkari, J.; et al. Chemical and electrochemical properties of a hydrophobic deep eutectic solvent. Electrochimica Acta 2019, 295, 124–129. [Google Scholar] [CrossRef]
- Yousefi, S.M.; Shemirani, F.; Ghorbanian, S.A. Hydrophobic Deep Eutectic Solvents in Developing Microextraction Methods Based on Solidification of Floating Drop: Application to the Trace HPLC/FLD Determination of PAHs. Chromatographia 2018, 81, 1201–1211. [Google Scholar] [CrossRef]
- Tereshatov, E.E.; Boltoeva, M.Y.; Folden, C.M. First evidence of metal transfer into hydrophobic deep eutectic and low-transition-temperature mixtures: Indium extraction from hydrochloric and oxalic acids. Green Chem. 2016, 18, 4616–4622. [Google Scholar] [CrossRef]
- Schaeffer, N.; Martins, M.A.R.; Neves, C.M.S.S.; Pinho, S.P.; Coutinho, J.A.P. Sustainable hydrophobic terpene-based eutectic solvents for the extraction and separation of metals. Chem. Commun. 2018, 54, 8104–8107. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.-C.; Ao, Y.-T.; Ding, W.-H. Determination of microcystins in water samples by deep eutectic solvent-based vortex-assisted liquid–liquid microextraction coupled with ultrahigh-performance liquid chromatography-high resolution mass spectrometry. RSC Adv. 2019, 9, 38669–38676. [Google Scholar] [CrossRef] [Green Version]
- Ao, Y.-T.; Chen, Y.-C.; Ding, W.-H. Deep eutectic solvent-based ultrasound-assisted emulsification microextraction for the rapid determination of benzotriazole and benzothiazole derivatives in surface water samples. J. Hazard. Mater. 2021, 401, 123383. [Google Scholar] [CrossRef] [PubMed]
- Shishov, A.; Pochivalov, A.; Nugbienyo, L.; Andruch, V.; Bulatov, A. Deep eutectic solvents are not only effective extractants. TrAC Trends Anal. Chem. 2020, 129, 115956. [Google Scholar] [CrossRef]
- Habibi, E.; Ghanemi, K.; Fallah-Mehrjardi, M.; Dadolahi-Sohrab, A. A novel digestion method based on a choline chloride–oxalic acid deep eutectic solvent for determining Cu, Fe, and Zn in fish samples. Anal. Chim. Acta 2013, 762, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Ghanemi, K.; Navidi, M.-A.; Fallah-Mehrjardi, M.; Dadolahi-Sohrab, A. Ultra-fast microwave-assisted digestion in choline chloride–oxalic acid deep eutectic solvent for determining Cu, Fe, Ni and Zn in marine biological samples. Anal. Methods 2014, 6, 1774–1781. [Google Scholar] [CrossRef]
- Santana, A.P.; Andrade, D.F.; Vargas, J.A.M.; Amaral, C.; de Oliveira, A.P.; Gonzalez, M.H. Natural deep eutectic solvents for sample preparation prior to elemental analysis by plasma-based techniques. Talanta 2019, 199, 361–369. [Google Scholar] [CrossRef]
- Helalat–Nezhad, Z.; Ghanemi, K.; Fallah–Mehrjardi, M. Dissolution of biological samples in deep eutectic solvents: An approach for extraction of polycyclic aromatic hydrocarbons followed by liquid chromatography-fluorescence detection. J. Chromatogr. A 2015, 1394, 46–53. [Google Scholar] [CrossRef]
- Rastbood, S.; Hadjmohammadi, M.R.; Majidi, S.M. Development of a magnetic dispersive micro-solid-phase extraction method based on a deep eutectic solvent as a carrier for the rapid determination of meloxicam in biological samples. Anal. Methods 2020, 12, 2331–2337. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents providing enhanced stability of natural colorants from safflower (Carthamus tinctorius). Food Chem. 2014, 159, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Jiang, Y.; Liu, Q.; Tian, Y.; Liu, Y.; Xu, X.; Kang, M. Catchment versus Riparian Buffers: Which Land Use Spatial Scales Have the Greatest Ability to Explain Water Quality Changes in a Typical Temperate Watershed? Water 2021, 13, 1758. [Google Scholar] [CrossRef]
- Vorobyev, S.; Kolesnichenko, Y.; Korets, M.; Pokrovsky, O. Testing Landscape, Climate and Lithology Impact on Carbon, Major and Trace Elements of the Lena River and Its Tributaries during a Spring Flood Period. Water 2021, 13, 2093. [Google Scholar] [CrossRef]
- Cunha, S.C.; Fernandes, J.O. Extraction techniques with deep eutectic solvents. TrAC Trends Anal. Chem. 2018, 105, 225–239. [Google Scholar] [CrossRef]
- Liang, X.; Zhu, Y.; Qi, B.; Li, S.; Luo, J.; Wan, Y. Structure-property-performance relationships of lactic acid-based deep eutectic solvents with different hydrogen bond acceptors for corn stover pretreatment. Bioresour. Technol. 2021, 336, 125312. [Google Scholar] [CrossRef] [PubMed]
| Sample Matrix | Analytes | DES Composition (Mole Ratio) | Method of Extraction | Instrumental Analysis | LODs |
|---|---|---|---|---|---|
| Hydrophilic deep eutectic solvents | |||||
| Waters (tap, lake, waste) [58] | Cr (III/VI) | ChCl/phenol (1:3) | UALME | FAAS | 5.5 ug/L |
| Switchgrass [76] | Cellulose-rich pulp, lignin, and xylose-rich liquor. | ChCl/glycerol (1:2) | HPLC-RID, NMR, ATR-FTIR, XRD | ||
| Tartary buckwheat Hulls [77] | Flavonoid (rutin) | ChCl/glycerol (1:1) | UAME | HPLC-UV | |
| Flower petals [78] | Anthocyanins | Lactic acid/glucose 1,2-propanediol/ChCl | UAE | HPLC-DAD | |
| Soil samples [79] | As, Cr, Mo, Sb, Se and V | ChCl/oxalic acid | UAE | ICP-OES | 0.009–0.1 ug/g |
| Sediment samples [80] | Cu | ChCl/oxalic acid (1.5:1) | SLE | ICP-OES | 1.2 ug/L |
| Hydrophobic deep eutectic solvents | |||||
| Wastewater and human plasma [81] | Amphetamine-type stimulants | ChCl/phenylethanol (1:4) | AA-EME | HPLC-UV | 2.0–5.0 ng/mL |
| Surface water [49] | Fluoroquinolones | Thymol/Heptanoic acid (2:1) | In situ LPME | HPLC-UV | 3 ng/mL |
| Wastewater [82] | Neonicotinoids | DL-menthol/organic acids | LLE | UV–vis | |
| River water [56] | BP, BP-1, BP-3, BP-6, 4OH-BP | DL-menthol/decanoic acid (1:1) | Air-assisted DLLME | HPLC-DAD | 0.05–0.2 ng/mL |
| Water and soil samples [51] | Explosives | DL-menthol/decanoic acid (1:2) | Ferrofluid-based LPME | HPLC-UV | 0.22–0.91, 0.01–0.04 mg/mL |
| Soil samples [83] | Nitrotoluene | Borneol/menthol | MSPD | HPLC-UV | 0.12–0.33 ug/g |
| Water, soil, egg yolk samples [84] | Insecticide | N4444-Br/decanoic acid | DLLME | HPLC-UV | 0.001–0.003 ug/mL |
| Water, and biological samples [55] | Nitrite | N8851-Cl/oleic acid (1:2) | Vortex-assisted DLLME | HPLC-UV | 0.2 ng/mL |
| Biological and indoor air sample [85] | Formaldehyde | N8851-Cl/4-cyanophenol (1:1) | VA-LLME | HPLC-DAD | 0.2 ng/mL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Li, N.; Jiang, S.; Chen, X. Application of Extraction and Determination Based on Deep Eutectic Solvents in Different Types of Environmental Samples. Water 2022, 14, 46. https://doi.org/10.3390/w14010046
Wang Y, Li N, Jiang S, Chen X. Application of Extraction and Determination Based on Deep Eutectic Solvents in Different Types of Environmental Samples. Water. 2022; 14(1):46. https://doi.org/10.3390/w14010046
Chicago/Turabian StyleWang, Yonghua, Na Li, Shengnan Jiang, and Xi Chen. 2022. "Application of Extraction and Determination Based on Deep Eutectic Solvents in Different Types of Environmental Samples" Water 14, no. 1: 46. https://doi.org/10.3390/w14010046
APA StyleWang, Y., Li, N., Jiang, S., & Chen, X. (2022). Application of Extraction and Determination Based on Deep Eutectic Solvents in Different Types of Environmental Samples. Water, 14(1), 46. https://doi.org/10.3390/w14010046






