Skeletal Growth Rates in Porites lutea Corals from Pulau Tinggi, Malaysia
Abstract
1. Introduction
2. Materials and Methods
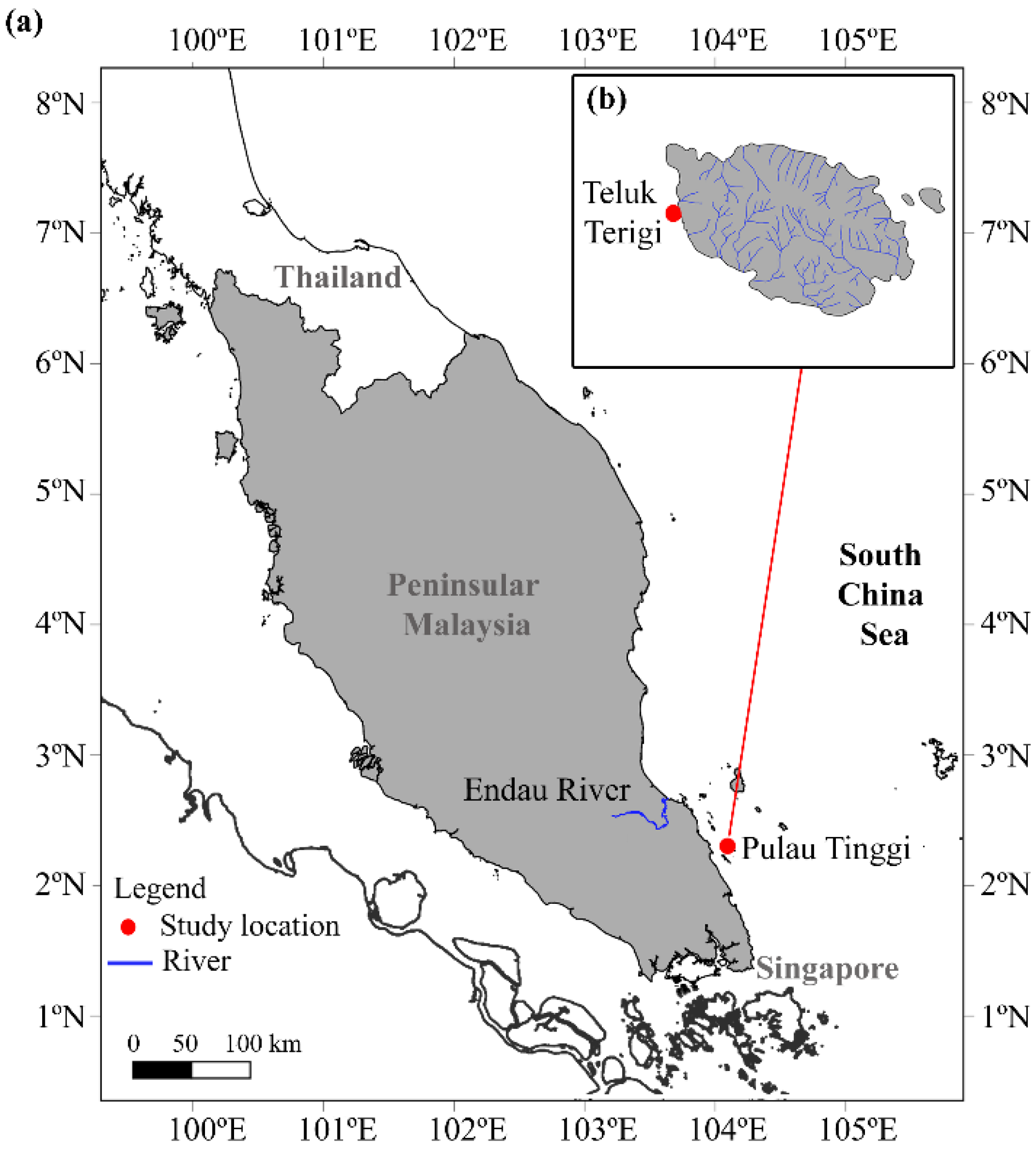
2.1. Study Area
2.2. Coral Sampling and Processing
2.3. Sclerochronology
2.4. Sea Temperature Data
2.5. Statistical Analysis
3. Results
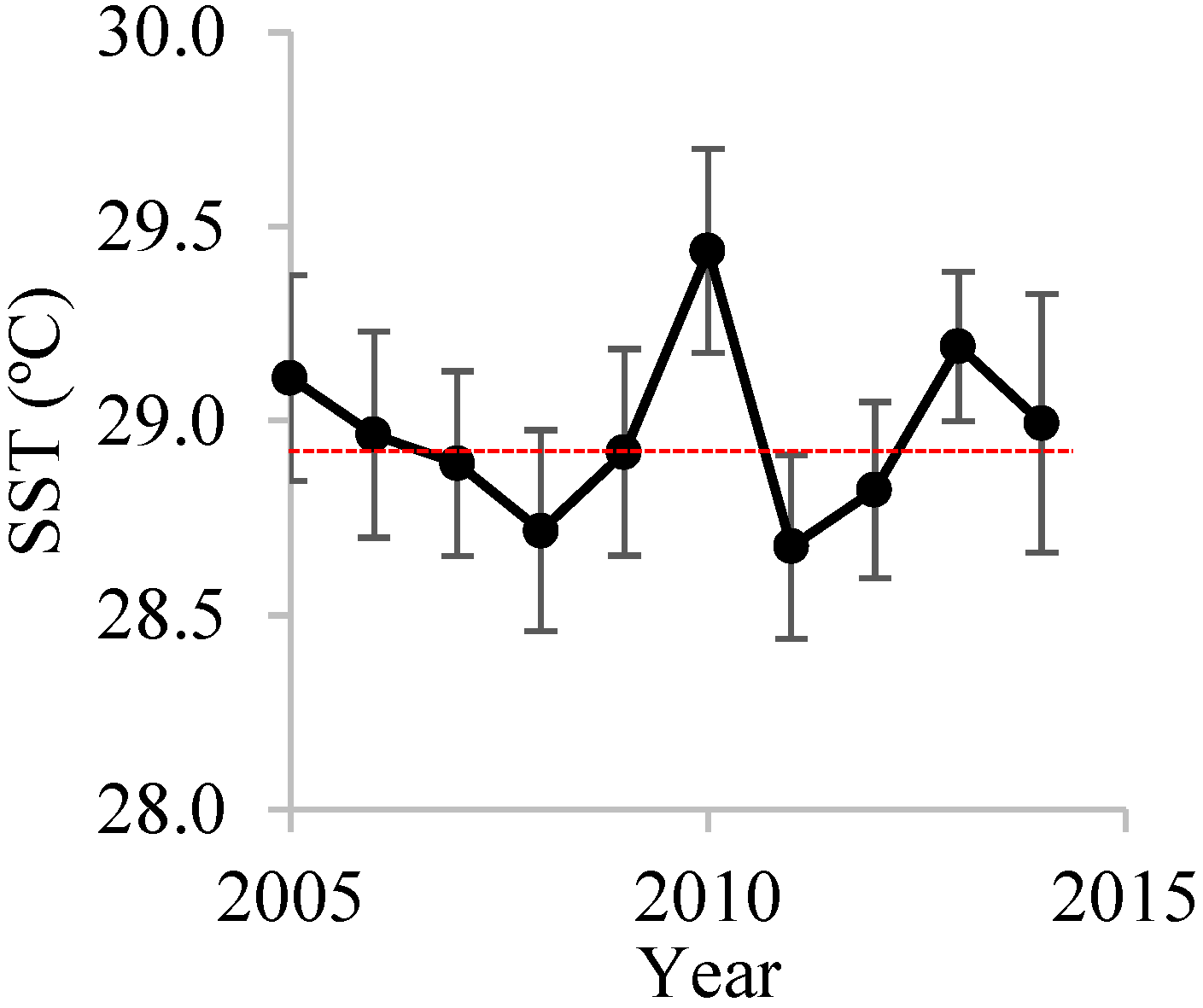
3.1. Inter-Annual Variations in SST
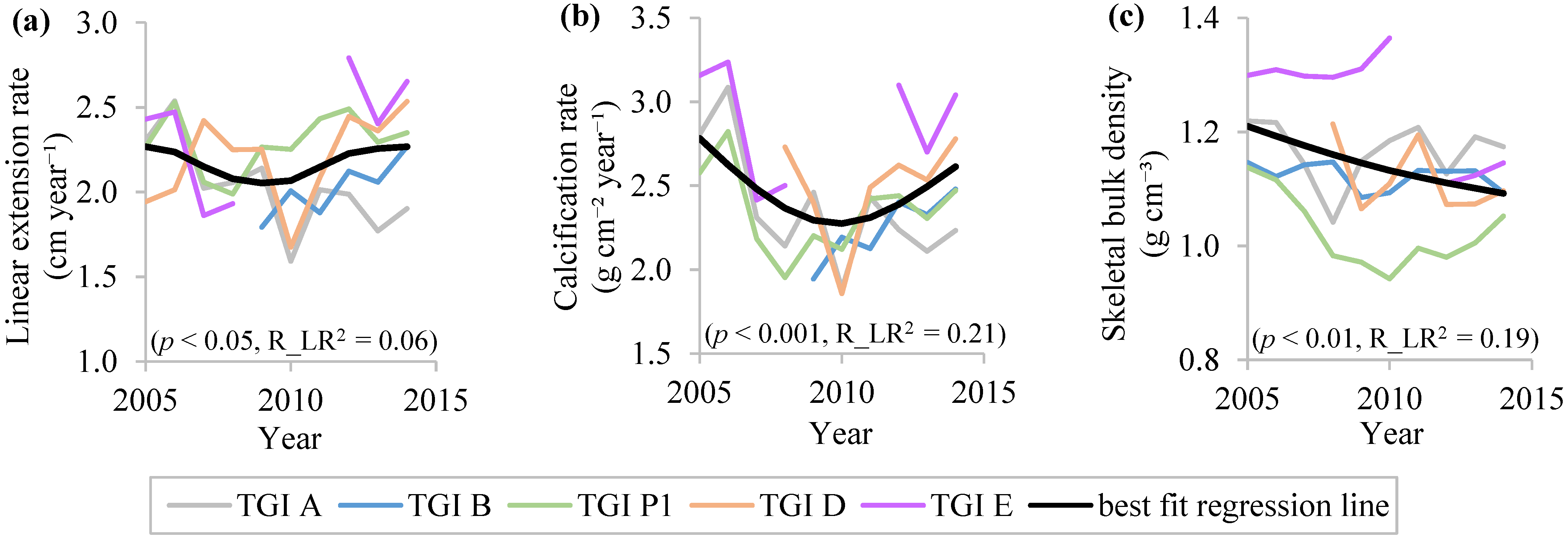
3.2. Inter-Annual Variations in Coral Skeletal Growth Rates
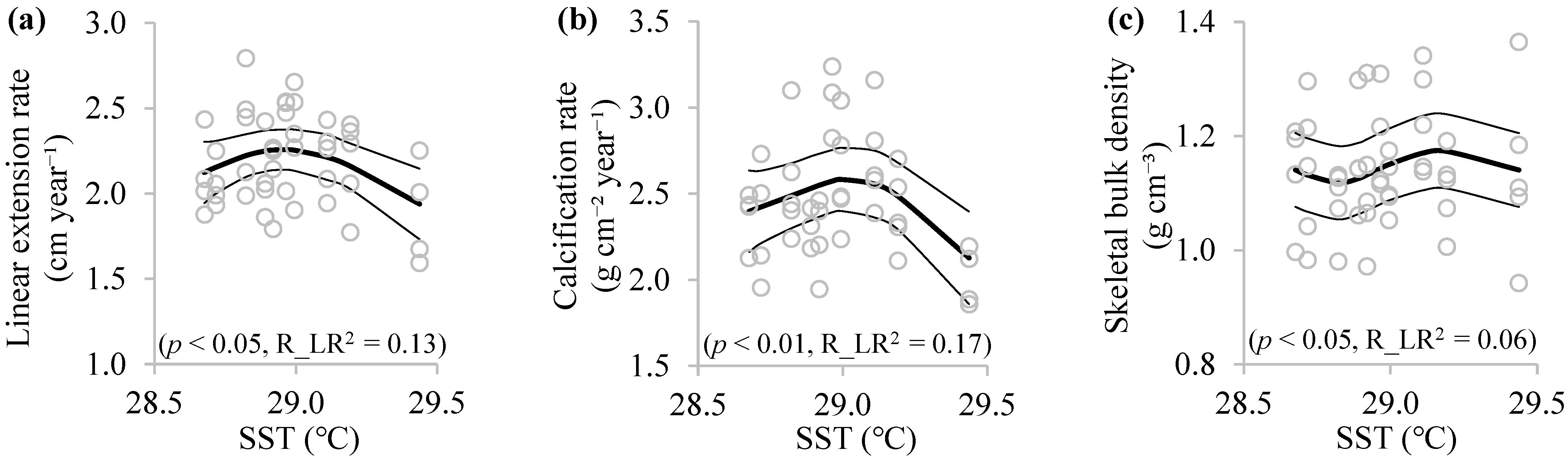
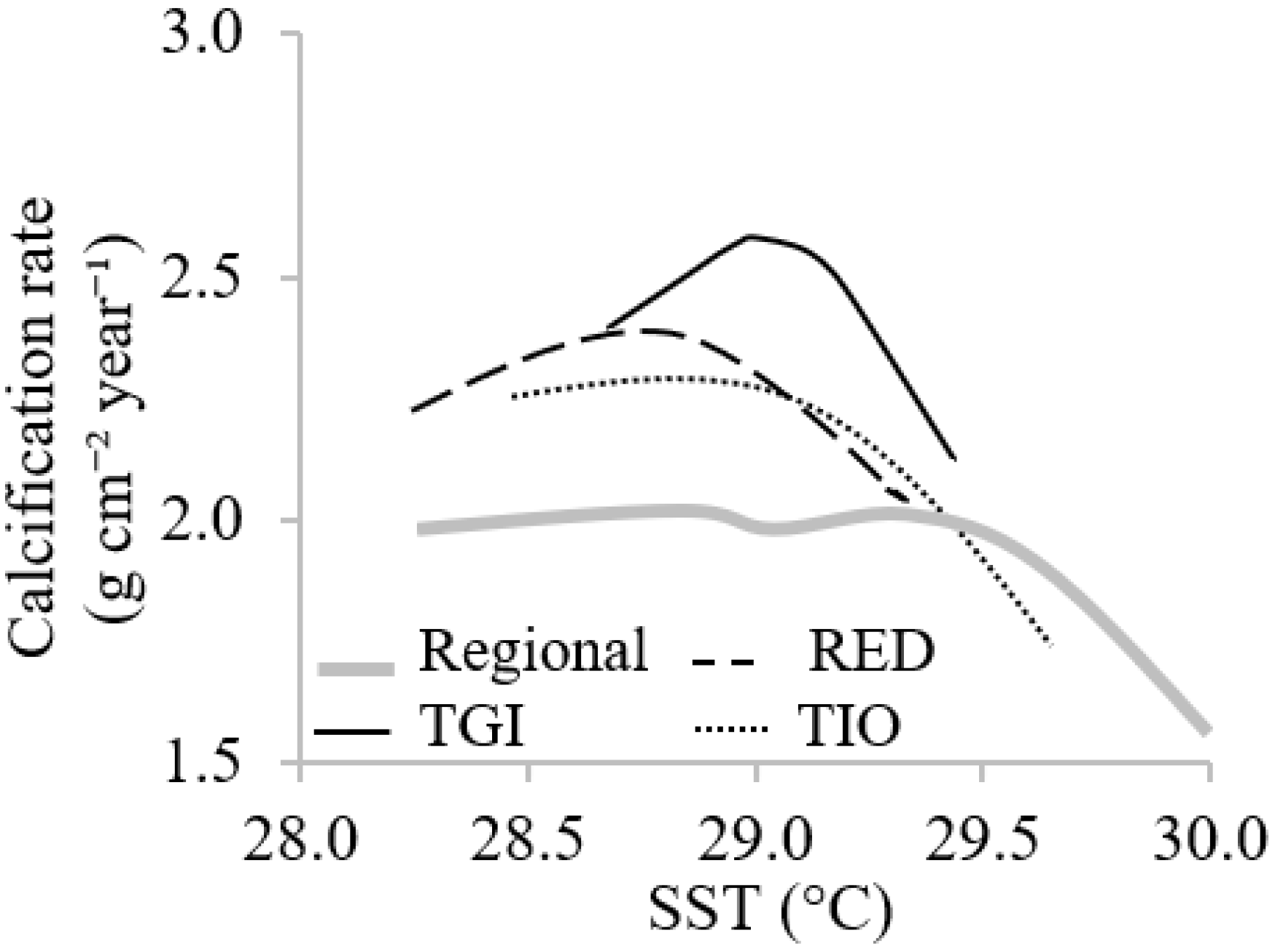
3.3. Relationships between Coral Skeletal Growth and SST
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Helmle, K.P.; Dodge, R.E. Sclerochronology. In Encyclopedia of Modern Coral Reefs, 1st ed.; Hopley, D., Ed.; Springer: Dordrecht, The Netherlands, 2011; pp. 958–966. [Google Scholar]
- Knutson, D.W.; Buddemeier, R.W.; Smith, S.V. Coral chronometers: Seasonal growth bands in reef corals. Science 1972, 177, 270–272. [Google Scholar] [CrossRef]
- Wild, F.J.; Jones, A.C.; Tudhope, A.W. Investigation of luminescent banding in solid coral: The contribution of phosphorescence. Coral Reefs 2000, 19, 132–140. [Google Scholar] [CrossRef][Green Version]
- Hudson, J.H.; Shinn, E.A.; Halley, R.B.; Lidz, B. Sclerochronology: A tool for interpreting past environments. Geology 1976, 4, 361–364. [Google Scholar] [CrossRef]
- Klein, R.; Loya, Y.; Gvirtzman, G.; Isdale, P.J.; Susic, M. Seasonal rainfall in the Sinai Desert during the late Quaternary inferred from fluorescent bands in fossil corals. Nature 1990, 345, 145–147. [Google Scholar] [CrossRef]
- Scoffin, T.P.; Tudhope, A.W.; Brown, B.E.; Chansang, H.; Cheeney, R.F. Patterns and possible environmental controls of skeletogenesis of Porites lutea, south Thailand. Coral Reefs 1992, 11, 1–11. [Google Scholar] [CrossRef]
- Lough, J.M.; Barnes, D.J. Several centuries of variation in skeletal extension, density and calcification in massive Porites colonies from the Great Barrier Reef: A proxy for seawater temperature and a background of variability against which to identify unnatural change. J. Exp. Mar. Biol. Ecol. 1997, 211, 29–67. [Google Scholar] [CrossRef]
- Lough, J.M.; Barnes, D.J.; McAllister, F.A. Luminescent lines in corals from the Great Barrier Reef provide spatial and temporal records of reefs affected by land runoff. Coral Reefs 2002, 21, 333–343. [Google Scholar] [CrossRef]
- Carricart-Ganivet, J.P.; Lough, J.M.; Barnes, D.J. Growth and luminescence characteristics in skeletons of massive Porites from a depth gradient in the central Great Barrier Reef. J. Exp. Mar. Biol. Ecol. 2007, 351, 27–36. [Google Scholar] [CrossRef]
- Tanzil, J.T.I.; Brown, B.E.; Dunne, R.P.; Lee, J.N.; Kaandorp, A.; Todd, P.A. Regional decline in growth rates of massive Porites coral in Southeast Asia. Glob. Chang. Biol. 2013, 19, 3011–3023. [Google Scholar] [CrossRef]
- Tanzil, J.T.I.; Lee, J.N.; Brown, B.E.; Quax, R.; Kaandorp, J.A.; Lough, J.M.; Todd, P.A. Luminescence and density banding patterns in massive Porites corals around the Thai-Malay Peninsula, Southeast Asia. Limnol. Oceanogr. 2016, 61, 2003–2026. [Google Scholar] [CrossRef]
- Mohamed, K.N.; May, M.S.Y.; Zainuddin, N. Water quality assessment of marine park islands in Johor, Malaysia. BESM 2015, 3, 19–27. [Google Scholar]
- Department of Irrigation and Drainage (Ministry of Environment and Water, Kuala Lumpur, Malaysia). Data Stream Flow 2004–2014 for Endau River: Station 2533474; Department of Irrigation and Drainage: Kuala Lumpur, Malaysia, 2019; Unpublished data. [Google Scholar]
- Department of Marine Park Malaysia. Pulau Tinggi and Pulau Sibu Marine Park Management Plan; Department of Marine Park Malaysia: Putrajaya, Malaysia, 2013; p. 4. [Google Scholar]
- Yanagi, T.; Sachoemar, S.; Takao, T.; Fujiwara, S. Seasonal variation of stratification in the Gulf of Thailand. J. Oceanogr. 2001, 57, 461–470. [Google Scholar] [CrossRef]
- Yendra, R.; Anofrizen, Z.W.Z.W.; Jemain, A.A.; Fudholi, A. Spatial analysis of storm behavior in Peninsular Malaysia during monsoon seasons. Int. J. Appl. Eng. Res. 2017, 12, 2559–2566. [Google Scholar]
- Lee, J.N.; Mohamed, C.A.R. Accumulation of settling particles in some coral reef areas of Peninsular Malaysia. Sains Malays. 2011, 40, 549–554. [Google Scholar]
- Reef Check Malaysia. Status of Coral Reefs in Malaysia 2019; Reef Check Malaysia: Kuala Lumpur, Malaysia, 2019; pp. 26–27. [Google Scholar]
- Grove, C.A.; Rodriguez-Ramirez, A.; Merschel, G.; Tjallingii, R.; Zinke, J.; Macia, A.; Brummer, G.J.A. Chapter 23 UV-spectral luminescence scanning: Technical updates and calibration developments. In Micro-XRF Studies of Sediment Cores: Applications of a Non-Destructive Tool for the Environmental Sciences; Croudace, I.W., Rothwell, R.G., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 563–581. [Google Scholar] [CrossRef]
- Grove, C.A.; Nagtegaal, R.; Zinke, J.; Scheufen, T.; Koster, B.; Kasper, S.; McCulloch, M.T.; Bergh, G.V.D.; Brummer, G.J.A. River runoff reconstructions from novel spectral luminescence scanning of massive coral skeletons. Coral Reefs 2010, 29, 579–591. [Google Scholar] [CrossRef]
- Kaushal, N.; Yang, L.; Tanzil, J.T.I.; Lee, J.N.; Goodkin, N.F.; Martin, P. Sub-annual fluorescence measurements of coral skeleton: Relationship between skeletal luminescence and terrestrial humic-like substances. Coral Reefs 2020, 39, 1257–1272. [Google Scholar] [CrossRef]
- Carricart-Ganivet, J.P.; Barnes, D.J. Densitometry from digitized images of X-radiographs: Methodology for measurement of coral skeletal density. J. Exp. Mar. Biol. Ecol. 2007, 344, 67–72. [Google Scholar] [CrossRef]
- Dodge, R.E.; Brass, G.W. Skeletal extension, density and calcification of the reef coral, Montastrea annularis: St. Croix, U.S. Virgin Islands. Bull. Mar. Sci. 1984, 34, 288–307. [Google Scholar]
- Reynolds, R.W.; Rayner, N.A.; Smith, T.M.; Stokes, D.C.; Wang, W. An improved in situ and satellite SST analysis for climate. J. Clim. 2002, 15, 1609–1625. [Google Scholar] [CrossRef]
- Cooper, T.F.; O’Leary, R.A.; Lough, J.M. Supporting online material for growth of Western Australian corals in the Anthropocene. Science 2012, 335, 593–596. [Google Scholar] [CrossRef]
- Wood, S.N. Generalized Additive Models: An Introduction with R, 2nd ed.; Chapman and Hall: Boca Raton, FL, USA, 2017. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. nlme: Linear and Nonlinear Mixed Effects Models; R Package Version 3.1-140; R Foundation: Vienna, Austria, 2019. [Google Scholar]
- Bartoń, K. MuMIn: Multi-Model Inference; R Package Version 1.43.17; R Foundation: Vienna, Austria, 2020. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation: Vienna, Austria, 2019. [Google Scholar]
- Elizalde-Rendon, E.M.; Horta-Puga, G.; Gonzalez-Diaz, P.; Carricart-Ganivet, J.P. Growth characteristics of the reef-building coral Porites astreoides under different environment conditions in the Western Atlantic. Coral Reefs 2010, 29, 607–614. [Google Scholar] [CrossRef]
- Shi, Q.; Yu, K.F.; Chen, T.R.; Zhang, H.L.; Zhao, M.X.; Yan, H.Q. Two centuries-long records of skeletal calcification in massive Porites colonies from Meiji Reef in the southern South China Sea and its responses to atmospheric CO2 and seawater temperature. Sci. China Earth Sci. 2012, 55, 1–12. [Google Scholar] [CrossRef]
- Guest, J.R.; Baird, A.H.; Maynard, J.A.; Muttaqin, E.; Edwards, A.J.; Campbell, S.J.; Yewdall, K.; Affendi, Y.A.; Chou, L.M. Contrasting patterns of coral bleaching susceptibility in 2010 suggest an adaptive response to thermal stress. PLoS ONE 2012, 7, e33353. [Google Scholar] [CrossRef]
- Tun, K.; Chou, L.K.; Low, J.; Yeemin, T.; Phongsuwan, N.; Setiasih, N.; Wilson, J.; Amri, A.Y.; Adzis, K.A.A.; Lane, D.; et al. A regional overview on the 2010 coral bleaching event in Southeast Asia. In Status of Coral Reefs in East Asian Sea Region: 2010; Japan Wildlife Research Center, Ed.; Ministry of Environment: Tokyo, Japan, 2010; pp. 7–28. [Google Scholar]
- Tan, C.H.; Heron, S.F. First observed severe mass bleaching in Malaysia, greater coral triangle. Galaxea J. Coral Reef Stud. 2011, 13, 27–28. [Google Scholar] [CrossRef]
- Carilli, J.E.; Norris, R.D.; Black, B.A.; Walsh, S.M.; McField, M. Local stressors reduce coral resilience to bleaching. PLoS ONE 2009, 4, e6324. [Google Scholar] [CrossRef] [PubMed]
- Cantin, N.E.; Lough, J.M. Surviving coral bleaching events: Porites growth anomalies on the Great Barrier Reef. PLoS ONE 2014, 9, e88720. [Google Scholar] [CrossRef]
- D’Olivo, J.P.; Georgiou, L.; Falter, J.; DeCarlo, T.M.; Irigoien, X.; Voolstra, C.R.; Roder, C.; Trotter, J.; McCulloch, M.T. Long-term impacts if the 1997–1998 bleaching event on the growth and resilience of massive Porites corals from the Central Red Sea. Geochem. Geophys. Geosyst. 2019, 20, 2936–2954. [Google Scholar] [CrossRef]
- Mohamed, C.A.R.; Hamid, S.A.; Zulkifli, F.A. Marine Biodiversity Expedition Report 2012: Southern East Coast of Peninsular Malaysia—Tinggi Islands Archipelago Volume 5; Department of Marine Park Malaysia: Putrajaya, Malaysia, 2013. [Google Scholar]
- Lough, J.M.; Barnes, D.J. Environmental controls on growth of the massive coral Porites. J. Exp. Mar. Biol. Ecol. 2000, 245, 225–243. [Google Scholar] [CrossRef]
- Carricart-Ganivet, J.P. Annual density banding in massive coral skeletons: Result of growth strategies to inhabit reefs with high microborers’ activity? Mar. Biol. 2007, 153, 1–5. [Google Scholar] [CrossRef]
- Carricart-Ganivet, J.P. Sea surface temperature and the growth of the West Atlantic reef-building coral Montastraea annularis. J. Exp. Mar. Biol. Ecol. 2004, 302, 249–260. [Google Scholar] [CrossRef]
- Cooper, T.F.; De’ath, G.; Fabricius, K.E.; Lough, J.M. Declining coral calcification in massive Porites in two nearshore regions of the northern Great Barrier Reef. Glob. Chang. Biol. 2008, 14, 529–538. [Google Scholar] [CrossRef]
- Cantin, N.E.; Cohen, A.L.; Karnauskas, K.B.; Tarrant, A.M.; McCorkle, D.C. Ocean warming slows coral growth in the central Red Sea. Science 2010, 329, 322–325. [Google Scholar] [CrossRef] [PubMed]
| Reef Location | Years | Linear Extension Rate (cm year−1) | Calcification Rate (g cm−2 year−1) | Skeletal Bulk Density (g cm−3) |
|---|---|---|---|---|
| Pulau Tinggi [this study] | 2005–2014 | 2.18 ± 0.04 | 2.46 ± 0.05 | 1.14 ± 0.01 |
| Pulau Tioman [10] | 1980–2010 | 1.72 ± 0.30 | 2.24 ± 0.23 | 1.31 ± 0.13 |
| Pulau Redang [10] | 1980–2010 | 1.97 ± 0.22 | 2.32 ± 0.22 | 1.19 ± 0.06 |
| Singapore [10] | 1980–2010 | 1.66 ± 0.45 | 1.71 ± 0.33 | 1.13 ± 0.28 |
| Phuket, Thailand [10] | 1980–2010 | 2.08 ± 0.38 | 2.29 ± 0.29 | 1.12 ± 0.14 |
| Great Barrier Reef [7] | 1934–1982 | 1.48 ± 0.32 | 1.72 ± 0.36 | 1.17 ± 0.10 |
| Western Atlantic [30] | 1995–2006 | 0.37 ± 0.65 | 0.55 ± 0.12 | 1.49 ± 0.16 |
| Nansha Island [31] | 1716–2005 | 0.91 ± 0.26 | 1.30 ± 0.35 | 1.45 ± 0.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ong, C.K.; Lee, J.N.; Tanzil, J.T.I. Skeletal Growth Rates in Porites lutea Corals from Pulau Tinggi, Malaysia. Water 2022, 14, 38. https://doi.org/10.3390/w14010038
Ong CK, Lee JN, Tanzil JTI. Skeletal Growth Rates in Porites lutea Corals from Pulau Tinggi, Malaysia. Water. 2022; 14(1):38. https://doi.org/10.3390/w14010038
Chicago/Turabian StyleOng, Chai Kee, Jen Nie Lee, and Jani Thuaibah Isa Tanzil. 2022. "Skeletal Growth Rates in Porites lutea Corals from Pulau Tinggi, Malaysia" Water 14, no. 1: 38. https://doi.org/10.3390/w14010038
APA StyleOng, C. K., Lee, J. N., & Tanzil, J. T. I. (2022). Skeletal Growth Rates in Porites lutea Corals from Pulau Tinggi, Malaysia. Water, 14(1), 38. https://doi.org/10.3390/w14010038







