Long-Term Oxbow Lake Trophic State under Agricultural Best Management Practices
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site Description
2.2. In-Situ Water Measurements, Sampling, and Analysis
2.3. Trophic State Data Analysis
3. Results
3.1. General Water Quality and Trophic State
3.2. Temporal Changes in Trophic State
3.3. Management Practices and Trophic State
4. Discussion
4.1. Lake Trophic Changes and Stable State
4.2. Prospects for Further Improvements of Trophic State
4.3. Future Research Focus
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jeppesen, E.; Søndergaard, M.; Lauridsen, T.L.; Kronvang, B.; Beklioglu, M.; Lammens, E.; Jensen, H.S.; Köhler, J.; Ventalä, M.; Tarvainen, M.; et al. Danish and other European experiences in managing shallow lakes. Lake Reserv. Manag. 2007, 23, 439–451. [Google Scholar] [CrossRef][Green Version]
- Renwick, W.H.; Vanni, M.J.; Zhang, Q.; Patton, J. Water quality trends and changing agricultural practices in a Midwest U.S. watershed, 1994–2006. J. Environ. Qual. 2008, 37, 1862–1874. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, Q.; Liu, G. Effects of watershed land use and lake morphometry on the trophic state of Chinese lakes: Implications for eutrophic control. Clean Soil Air Water 2011, 39, 35–42. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, Q.; Liu, G. Influences of watershed landscape composition and configuration on lake-water quality in the Yangtze River basin of China. Hydrol. Process. 2012, 26, 570–578. [Google Scholar] [CrossRef]
- US Department of Commerce, United States Census Bureau. U.S. and World Population Clock. Available online: https://www.census.gov/popclock/ (accessed on 21 February 2018).
- Cooke, G.D. History of eutrophic lake rehabilitation in North America with arguments for including social sciences in the paradigm. Lake Reserv. Manag. 2007, 23, 323–329. [Google Scholar] [CrossRef]
- Carlson, R.E. The trophic state concept: A lake management perspective. Lake Reserv. Manag. 1984, 1, 427–430. [Google Scholar] [CrossRef]
- Moss, B.; Beklioglu, M.; Carvalho, L.; Kilinc, S.; McGowan, S.; Stephen, D. Vertically-challenged limnology; contrasts between deep and shallow lakes. Hydrobiologia 1997, 342/343, 257–267. [Google Scholar] [CrossRef]
- Carpenter, S.R.; Ludwig, D.; Brock, W.A. Management of eutrophication for lakes subject to potentially irreversible change. Ecol. Appl. 1999, 9, 751–771. [Google Scholar] [CrossRef]
- Søndergaard, M.; Jeppesen, E.; Lauridsen, T.L.; Skov, C.; van Nes, E.H.; Roijackers, R.; Lammans, E.; Portielje, R. Lake restoration: Successes, failures and long-term effects. J. Appl. Ecol. 2007, 44, 1095–1105. [Google Scholar] [CrossRef]
- Justus, B. Water quality of least-impaired lakes in eastern and southern Arkansas. Environ. Monit. Assess. 2010, 168, 363–383. [Google Scholar] [CrossRef]
- Knowlton, M.F.; Jones, J.R. Temporal variation and assessment of trophic state indicators in Missouri reservoirs: Implication for lake monitoring and management. Lake Reserv. Manag. 2006, 22, 261–271. [Google Scholar] [CrossRef]
- Dodds, W.K. Determining ecoregional reference conditions for nutrients, Secchi depth and chlorophyll a in Kansas lakes and reservoirs. Lake Reserv. Manag. 2006, 22, 151–159. [Google Scholar] [CrossRef]
- Jones, J.R.; Obrecht, D.V.; Perkins, B.D.; Knowlton, M.F.; Thorpe, A.P.; Watanabe, S.; Bacon, R.R. Nutrients, seston, and transparency of Missouri reservoirs and oxbow lakes: An analysis of regional limnology. Lake Reserv. Manag. 2008, 24, 155–180. [Google Scholar] [CrossRef]
- Thomatou, A.A.; Triantafyllidou, M.; Chalkia, E.; Kehayias, G.; Konstantinou, I.; Zacharias, I. Land use changes do not rapidly change the trophic state of a deep lake. Amvrakia Lake, Greece. J. Environ. Prot. 2013, 4, 426–434. [Google Scholar] [CrossRef][Green Version]
- Poor, N.D. Effect of lake management efforts on the trophic state of a subtropical shallow lake in Lakeland, Florida, USA. Water Air Soil Pollut. 2010, 207, 333–347. [Google Scholar] [CrossRef]
- Havens, K.; Ji, G. Inferences about seston composition and phytoplankton limiting factors during recovery of a large shallow lake from hurricane impacts. Inland Waters 2017, 7, 236–247. [Google Scholar] [CrossRef]
- Meals, D.W.; Dressing, S.A.; Davenport, T.E. Lag time in water quality response to best management practices: A review. J. Environ. Qual. 2010, 39, 85–96. [Google Scholar] [CrossRef]
- Tuppad, P.; Santhi, C.; Srinivasan, R. Assessing BMP effectiveness: Multiprocedure analysis of observed water quality data. Environ. Monit. Assess. 2010, 170, 315–329. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, X.; Zhang, M.; Dahlgren, R.A.; Eitzel, M. A review of vegetated buffers and a meta-analysis of their mitigation efficacy in reducing nonpoint source pollution. J. Environ. Qual. 2010, 39, 76–84. [Google Scholar] [CrossRef]
- Payne, F.E.; Bjork, T.M. The effectiveness of BMPs and sediment control structures and their relationship to in-lake water quality. Lake Reserv. Manag. 1984, 1, 82–86. [Google Scholar] [CrossRef][Green Version]
- Makarewicz, J.C.; Lewis, T.W.; Bosch, I.; Noll, M.R.; Herendeen, N.; Simon, R.D.; Zollweg, J.; Vodacek, A. The impact of agricultural best management practices on downstream systems: Soil loss and nutrient chemistry and flux to Conesus Lake, New York, USA. J. Great Lakes Res. 2009, 35, 23–36. [Google Scholar] [CrossRef]
- Lizotte, R.E.; Yasarer, L.M.W.; Locke, M.A.; Bingner, R.L.; Knight, S.S. Lake nutrient responses to integrated conservation best management practices in an agricultural watershed. J. Environ. Qual. 2017, 46, 330–338. [Google Scholar] [CrossRef]
- Locke, M.; Knight, S.; Smith, S.; Cullum, R.; Zablotowicz, R.; Yuan, Y.; Bingner, R.L. Environmental quality research in the Beasley Lake watershed, 1995 to 2007: Succession from conventional to conservation practices. J. Soil Water Conserv. 2008, 63, 430–442. [Google Scholar] [CrossRef]
- Tomer, M.D.; Sadler, E.J.; Lizotte, R.E.; Bryant, R.B.; Potter, T.L.; Moore, M.T.; Veith, T.L.; Baffaut, C.; Locke, M.A.; Walbridge, M.R. A decade of conservation effects assessment research by the USDA Agricultural Research Service: Progress overview and future outlook. J. Soil Water Conserv. 2014, 69, 365–373. [Google Scholar] [CrossRef]
- Lizotte, R.E.; Knight, S.S.; Locke, M.A.; Bingner, R.L. Influence of integrated watershed-scale agricultural conservation practices on lake water quality. J. Soil Water Conserv. 2014, 69, 160–170. [Google Scholar] [CrossRef]
- Lizotte, R.E.; Locke, M.A.; Bingner, R.L.; Steinriede, R.W.; Smith, S. Effectiveness of integrated best management practices on mitigation of atrazine and metolachlor in an agricultural lake watershed. Bull. Environ. Contam. Toxicol. 2017, 98, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Cullum, R.F.; Locke, M.A.; Knight, S.S. Effects of conservation reserve program on runoff and lake water quality in an oxbow lake watershed. J. Int. Appl. Sci. 2010, 5, 318–328. [Google Scholar]
- Locke, M.A.; Weaver, M.A.; Zablotowicz, R.M.; Steinriede, R.W.; Bryson, C.T.; Cullum, R.F. Constructed wetlands as a component of the agricultural landscape: Mitigation of herbicides in simulated runoff from upland drainage areas. Chemosphere 2011, 83, 1532–1538. [Google Scholar] [CrossRef] [PubMed]
- Lizotte, R.; Locke, M. Assessment of runoff water quality for an integrated best-management practice system in an agricultural watershed. J. Soil Water Conserv. 2018, 73, 247–256. [Google Scholar] [CrossRef]
- Wetzel, R.G.; Likens, G.E. Limnological Analyses, 3rd ed.; Springer: New York, NY, USA, 2000; p. 429. [Google Scholar]
- Eaton, A.D.; Clesceri, L.S.; Rice, E.W.; Greenburg, A.E. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2005; p. 1231. [Google Scholar]
- Carlson, R.E. A trophic state index for lakes. Limnol. Oceanogr. 1977, 22, 361–369. [Google Scholar] [CrossRef]
- Carlson, R.E. Expanding the Trophic State Concept to Identify Non-Nutrient Limited Lakes and Reservoirs, Proceedings of the National Conference on Enhancing the States’ Lake Management Programs; Monitoring and Lake Impact Assessment: Chicago, IL, USA, 1992; pp. 59–71. [Google Scholar]
- Kratzer, C.; Brezonik, P. A Carlson-type trophic state index for nitrogen in Florida lakes. Wat. Res. Bull. 1981, 17, 713–715. [Google Scholar] [CrossRef]
- Lake and Reservoir Classification System; US Environmental Protection Agency: Washington, DC, USA, 1979. Available online: https://nepis.epa.gov/Exe/ZyNET.exe/9101QXT0.TXT?ZyActionD=ZyDocument&Client=EPA&Index=1976+Thru+1980&Docs=&Query=&Time=&EndTime=&SearchMethod=1&TocRestrict=n&Toc=&TocEntry=&QField=&QFieldYear=&QFieldMonth=&QFieldDay=&IntQFieldOp=0&ExtQFieldOp=0&XmlQuery=&File=D%3A%5Czyfiles%5CIndex%20Data%5C76thru80%5CTxt%5C00000034%5C9101QXT0.txt&User=ANONYMOUS&Password=anonymous&SortMethod=h%7C-&MaximumDocuments=1&FuzzyDegree=0&ImageQuality=r75g8/r75g8/x150y150g16/i425&Display=hpfr&DefSeekPage=x&SearchBack=ZyActionL&Back=ZyActionS&BackDesc=Results%20page&MaximumPages=1&ZyEntry=1&SeekPage=x&ZyPURL (accessed on 21 February 2018).
- Carlson, R.E.; Havens, K.E. Simple graphical methods for the interpretation of relationships between trophic state variables. Lake Reserv. Manag. 2005, 21, 107–118. [Google Scholar] [CrossRef]
- Helsel, D.R. Advantages of nonparametric procedures for analysis of water quality data. Hydrol. Sci. J. 1987, 32, 179–190. [Google Scholar] [CrossRef]
- Yu, Y.-S.; Zhou, S.; Whittemore, D. Non-parametric trend analysis of water quality data of rivers in Kansas. J. Hydrol. 1993, 150, 61–80. [Google Scholar] [CrossRef]
- Haggard, B.E.; Scott, J.T.; Longing, S.D. Sestonic chlorophyll-a shows hierarchical structure and thresholds with nutrients across the Red River Basin, USA. J. Environ. Qual. 2013, 42, 437–445. [Google Scholar] [CrossRef]
- Stevenson, R.J.; Bennett, B.J.; Jordan, D.N.; French, R.D. Phosphorus regulates stream injury by filamentous green algae, DO, and pH with thresholds in responses. Hydrobiologia 2012, 695, 25–42. [Google Scholar] [CrossRef]
- Taylor, R. Interpretation of the correlation coefficient: A basic review. J. Diagn. Med. Sonogr. 1990, 6, 35–39. [Google Scholar] [CrossRef]
- Havens, K.E. Particulate light attenuation in a large subtropical lake. Can. J. Fish. Aquat. Sci. 1995, 52, 1803–1811. [Google Scholar] [CrossRef]
- Havens, K.E. Secondary nitrogen limitation in a subtropical lake impacted by non-point source agricultural pollution. Environ. Pollut. 1995, 89, 241–246. [Google Scholar] [CrossRef]
- Knight, S.; Cullum, R.; Lizotte, R. The limnology of a Mississippi River Alluvial Plain oxbow lake following application of conservation practices. JWARP 2015, 7, 707–714. [Google Scholar] [CrossRef][Green Version]
- Scheffer, M. Ecology of Shallow Lakes; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004; p. 357. [Google Scholar]
- Havens, K.E.; James, R.T.; East, T.L.; Smith, V.H. N:P ratios, light limitation, and cyanobacterial dominance in a subtropical lake impacted by non-point source nutrient pollution. Environ. Pollut. 2003, 122, 379–390. [Google Scholar] [CrossRef]
- Hawkins, P.R.; Griffiths, D.J. Light attenuation in a small tropical reservoir (Solomon Dam, North Queensland): Seasonal changes and the effects of artificial aeration. Aust. J. Mar. Freshwat. Res. 1986, 37, 199–208. [Google Scholar] [CrossRef]
- Oliver, R.L.; Ganf, G.G. The optical properties of a turbid reservoir and its phytoplankton in relation to photosynthesis and growth (Mount Bold Reservoir, South Australia). J. Plankton Res. 1988, 10, 1155–1177. [Google Scholar] [CrossRef]
- Paul, W.J.; Hamilton, D.P.; Ostrovsky, I.; Miller, S.D.; Zhang, A.; Muraoka, K. Catchment land use and trophic state impacts on phytoplankton composition: A case study from the Roturua lakes’ district, New Zealand. Hydrobiologia 2012, 698, 133–146. [Google Scholar] [CrossRef]
- Scheffer, M.; Rinaldi, S.; Gragnani, A.; Mur, L.R.; van Nes, E.H. On the dominance of filamentous cyanobacteria in shallow, turbid lakes. Ecology 1997, 78, 272–282. [Google Scholar] [CrossRef]
- Scheffer, M.; van Nes, E.H. Shallow lakes theory revisited: Various alternative regimes driven by climate, nutrients, depth and lake size. Hydrobiologia 2007, 584, 455–466. [Google Scholar] [CrossRef]
- Janse, J.H.; De Senerpont Domis, L.N.; Scheffer, M.; Lijklema, L.; van Liere, L.; Klinge, M.; Mooij, W.M. Critical phosphorus loading of different types of shallow lakes and the consequences for management estimated with the ecosystem model PCLake. Limnologica 2008, 38, 203–219. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Meerhoff, M.; Lauridsen, T.L.; Jensen, J.P. Shallow lake restoration—Some recent findings and challenges ahead. Hydrobiologia 2007, 584, 239–252. [Google Scholar] [CrossRef]
- Zaccara, S.; Canziani, A.; Roella, V.; Crosa, G. A northern Italian shallow lake as a case study for eutrophication control. Limnologica 2007, 8, 155–160. [Google Scholar] [CrossRef]
- Paerl, H.W.; Scott, J.T.; McCarthy, M.J.; Newell, S.E.; Gardner, W.S.; Havens, K.E.; Hoffman, D.K.; Wilhelm, S.W.; Wurtsbaugh, W.A. It takes two to tango: When and where dual nutrient (N & P) reductions are needed to protect lakes and downstream ecosystems. Environ. Sci. Technol. 2016, 50, 10805–10813. [Google Scholar] [CrossRef]
- Taylor, J.M.; (US Department of Agriculture-Agricultural Research Service, Oxford, MS, USA). Personal communication, 2016.
- Ullah, S.; Breitenbeck, G.A.; Faulkner, S.P. Denitrification and N2O emission from forested and cultivated alluvial clay soil. Biogeochemistry 2005, 73, 499–513. [Google Scholar] [CrossRef]
- Ullah, S.; Faulkner, S.P. Denitrification potential of different land-use types in an agricultural watershed, lower Mississippi valley. Ecol. Eng. 2006, 28, 131–140. [Google Scholar] [CrossRef]
- Søndergaard, M.; Bjerring, R.; Jeppesen, E. Persistent internal phosphorus loading during summer in a shallow eutrophic lake. Hydrobiologia 2013, 710, 95–107. [Google Scholar] [CrossRef]
- Gołdyn, R.; Podsiadłowski, S.; Dondajewska, R.; Kozak, A. The sustainable restoration of lakes—towards the challenges of the Water Framework Directive. Ecohydrol. Hydrobiol. 2014, 14, 68–74. [Google Scholar] [CrossRef]
- Dondajewska, R.; Kozak, A.; Kowalczewska-Madura, K.; Budzyńska, A.; Gołdyn, R.; Podsiadłowski, S.; Tomkowiak, A. The response of a shallow hypertrophic lake to innovative restoration measures-Uzarzewskie Lake case study. Ecol. Eng. 2018, 121, 72–82. [Google Scholar] [CrossRef]
- Kowalczewska-Madura, K.; Dondajewska, R.; Gołdyn, R.; Rosińska, J.; Podsiadłowski, S. Internal phosphorus loading as the response to complete and then limited sustainable restoration of a shallow lake. Annales Limnol. Int. J. Lim. 2019, 55, 4. [Google Scholar] [CrossRef]
- Yasarer, L.M.W.; Bingner, R.L.; Garbrecht, J.D.; Locke, M.A.; Lizotte, R.E.; Momm, H.G.; Busteed, P.R. Climate change impacts on runoff, sediment, and nutrient loads in an agricultural watershed in the lower Mississippi River basin. Appl. Eng. Agric. 2017, 33, 379–392. [Google Scholar] [CrossRef]
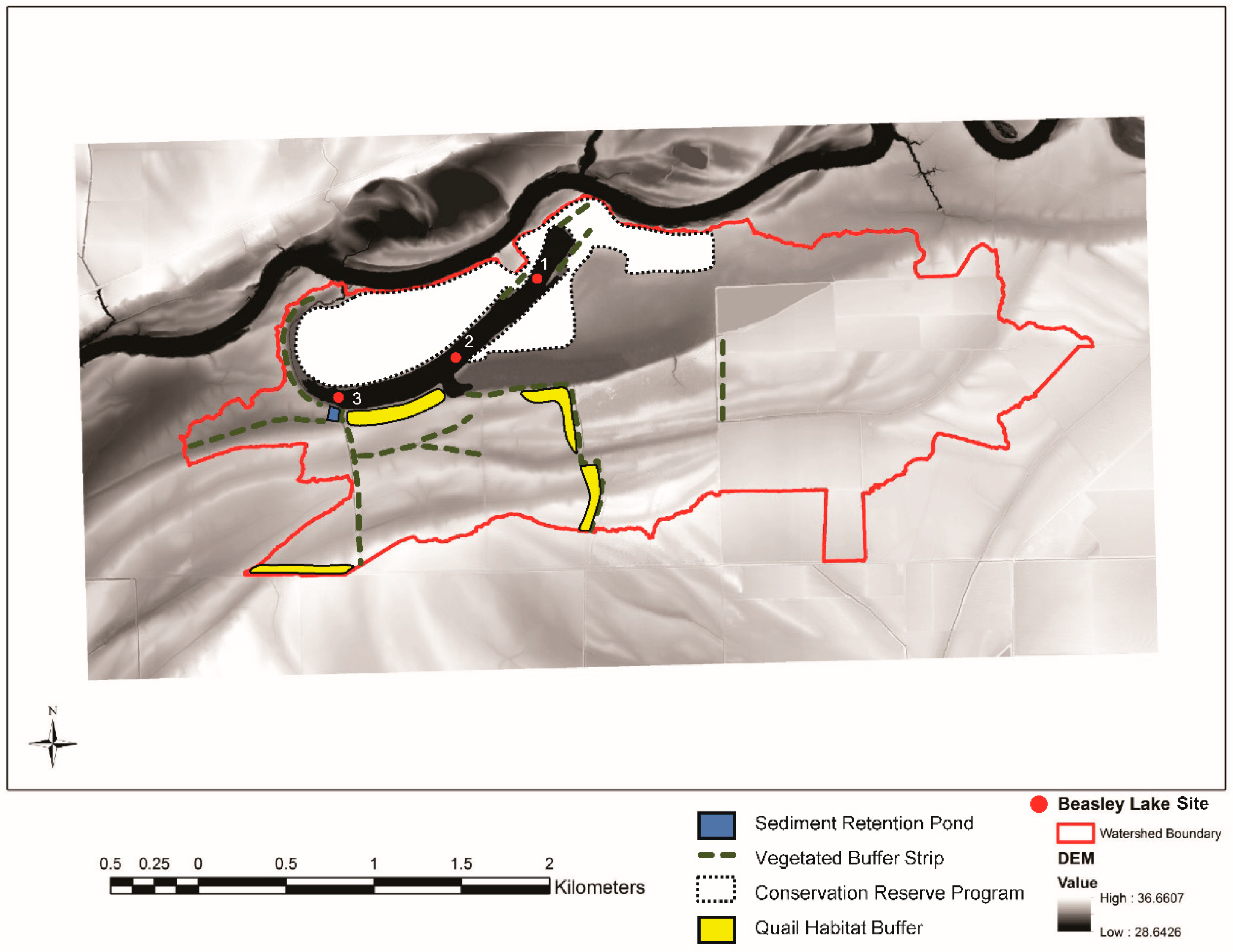
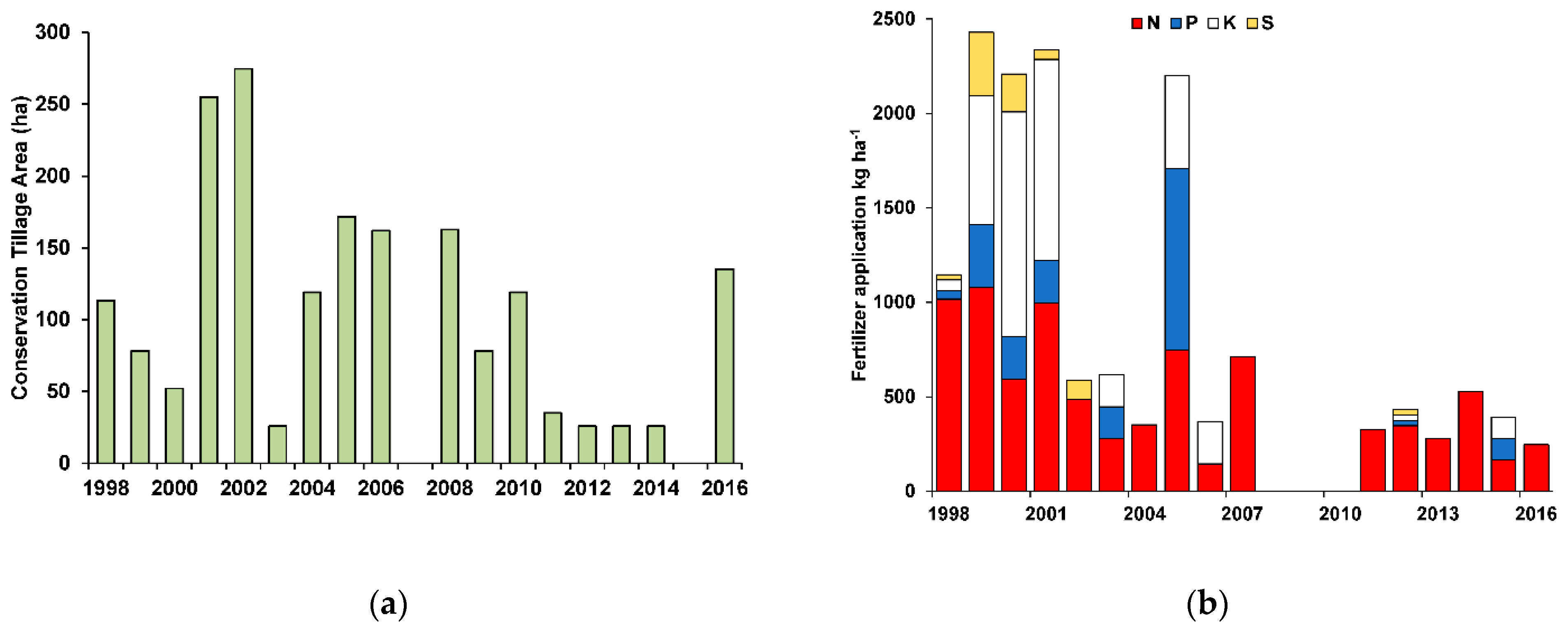
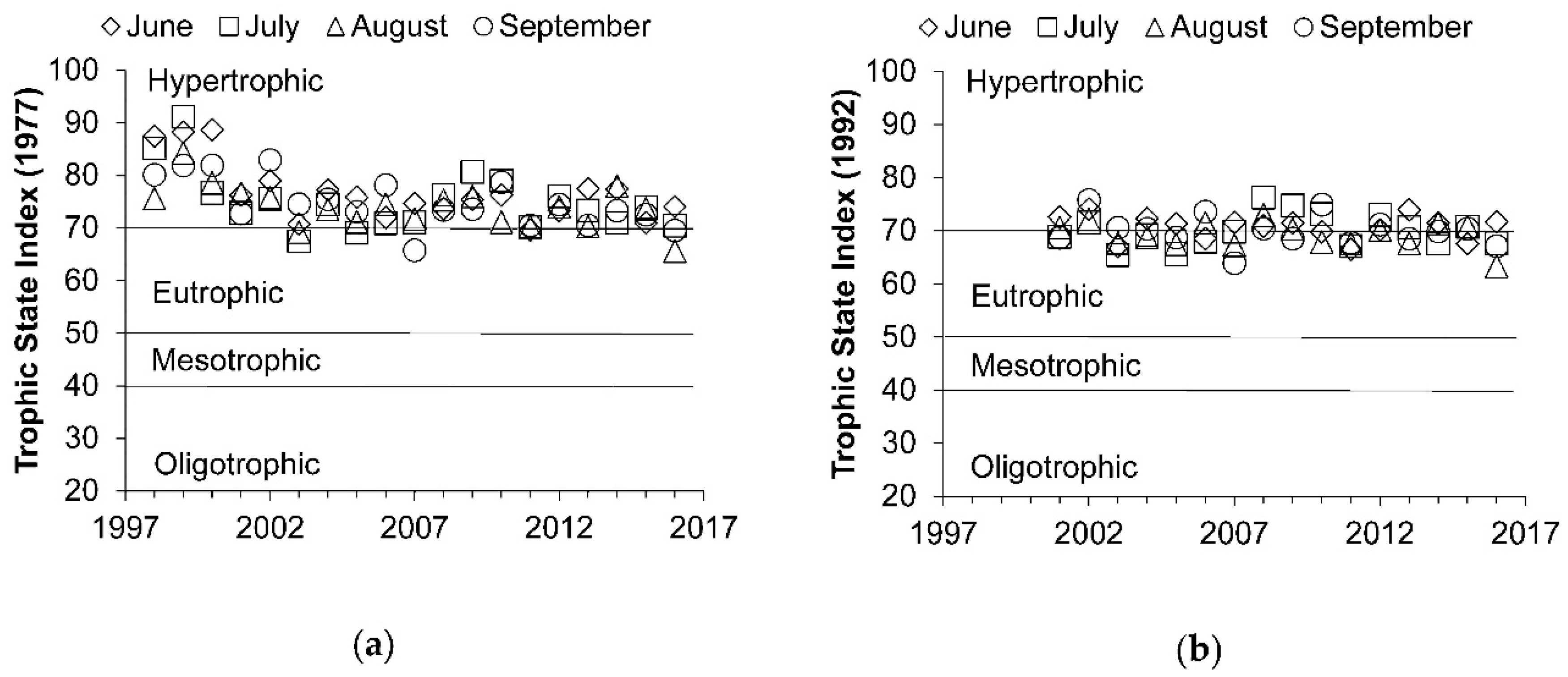

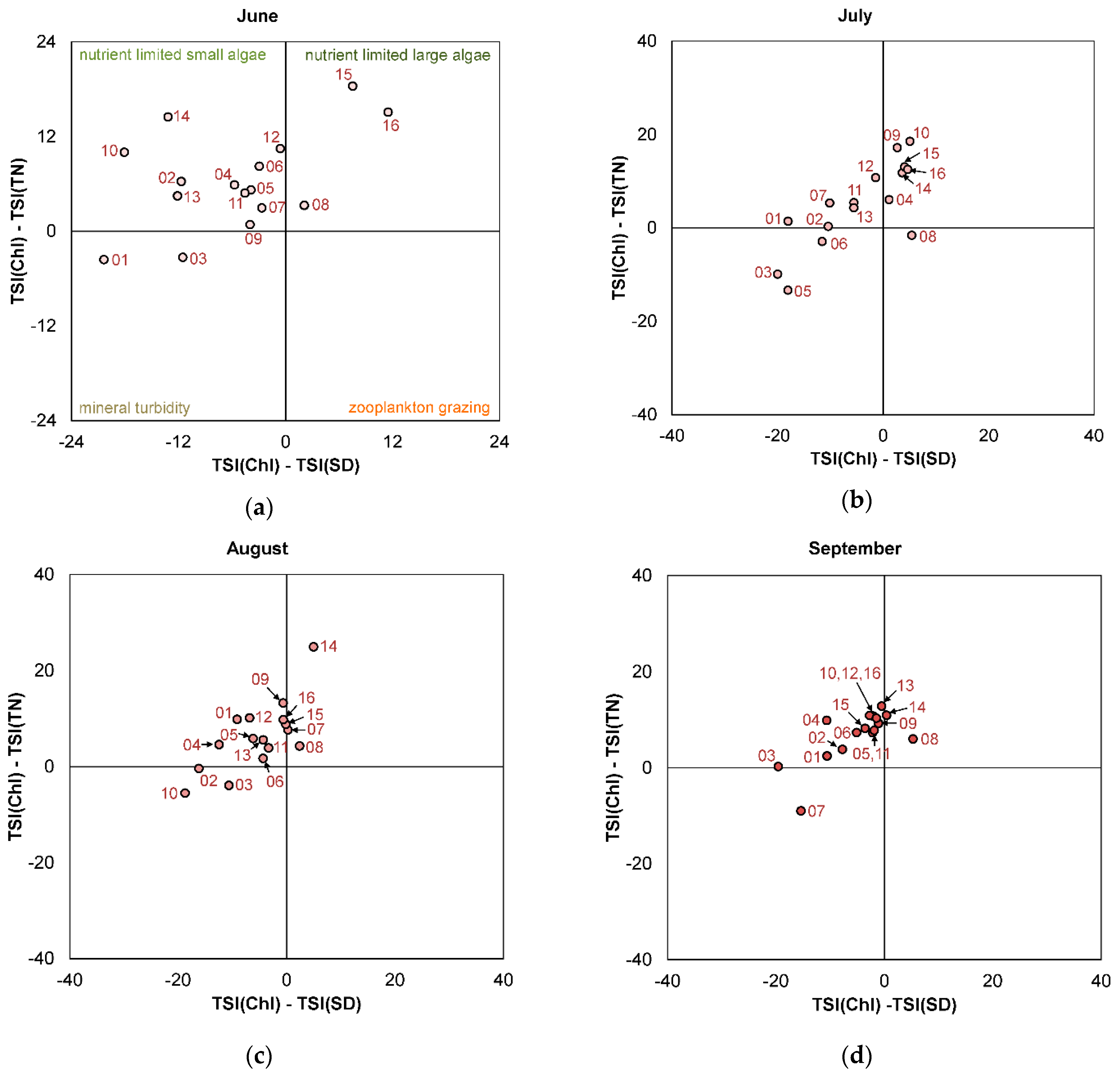
| Parameter | Abbreviation | Units | Value |
|---|---|---|---|
| Area | A | m2 | 251,202 |
| Volume | V | m3 | 360,917 |
| Maximum length | Lmax | m | 1140 |
| Maximum width | Lamax | m | 95 |
| Maximum depth | Zmax | m | 2.9 |
| Average depth | Z | m | 2.4 |
| Perimeter | M | m | 4400 |
| Shoreline development index | DL | unitless | 2.22 |
| Minimum hydraulic retention time | HRTmin | days | 5.6 |
| Average hydraulic retention time | HRT | days | 87 |
| Maximum flow rate | Qmax | m3 s−1 | 2.02 |
| Average flow rate | Q | m3 s−1 | 0.0376 |
| Variable | June | July | August | September |
|---|---|---|---|---|
| Median (Range) | Median (Range) | Median (Range) | Median (Range) | |
| SD (m) | 0.42 (0.04–0.93) | 0.47 (0.05–1.07) | 0.45 (0.13–0.77) | 0.36 (0.06–0.90) |
| TP (mg L−1) | 0.49 (0.06–3.65) | 0.37 (0.06–1.72) | 0.33 (0.05–0.85) | 0.38 (0.07–0.95) |
| TN (mg L−1) | 1.46 (0.67–2.18) | 1.49 (0.82–7.02) | 1.28 (0.17–2.99) | 1.47 (0.79–3.32) |
| Chl (μg L−1) | 33 (0–483) | 35 (0–147) | 31 (0–204) | 37 (0–257) |
| TSI(SD) | 72 (61–106) | 71 (59–103) | 72 (64–89) | 75 (62–101) |
| TSI(TP) | 93 (64–122) | 89 (63–112) | 88 (61–101) | 90 (66–103) |
| TSI(TN) | 60 (49–66) | 60 (52–83) | 58 (29–70) | 60 (51–72) |
| TSI(Chl) | 65 (0–91) | 65 (0–80) | 64 (0–83) | 66 (0–85) |
| Index | n | τ | n | τ | n | τ | n | τ |
|---|---|---|---|---|---|---|---|---|
| June | July | August | September | |||||
| TSI(SD) | 46 | −0.3911 | 43 | −0.450 | 40 | −0.422 | 44 | −0.383 |
| TSI(TP) | 46 | −0.577 | 43 | −0.602 | 41 | −0.457 | 43 | −0.548 |
| TSI(TN) | 34 | −0.014 | 31 | 0.166 | 31 | 0.163 | 32 | 0.133 |
| TSI(Chl) | 46 | 0.255 | 43 | 0.340 | 41 | 0.482 | 43 | 0.466 |
| TSI 1977 | 46 | −0.364 | 43 | −0.384 | 40 | −0.313 | 43 | −0.483 |
| TSI 1992 | 34 | −0.125 | 31 | −0.002 | 31 | −0.176 | 32 | −0.165 |
| TSI(Chl)–TSI(SD) | 46 | 0.563 | 43 | 0.543 | 40 | 0.512 | 43 | 0.516 |
| TSI(Chl)–TSI(TP) | 46 | 0.501 | 43 | 0.667 | 41 | 0.588 | 43 | 0.599 |
| TSI(Chl)–TSI(TN) | 34 | 0.316 | 31 | 0.295 | 31 | 0.231 | 32 | 0.319 |
| TSI | Node | Mean ± SD | Change Point | PRE | Improvement |
|---|---|---|---|---|---|
| TSI(SD) June | 1 | 78.5 ± 13.7 | 2004 VBS 8.2 ha | 0.822 | 0.822 |
| TSI(SD) July | 1 | 74.8 ± 10.7 | 2004 VBS 8.2 ha | 0.690 | 0.690 |
| 2 | 90.1 ± 9.9 | 1999 CT 78.4 ha | 0.858 | 0.168 | |
| TSI(SD) August | 1 | 73.1 ± 6.4 | 2004 VBS 8.2 ha | 0.641 | 0.641 |
| 3 | 70.4 ± 3.7 | 2003 VBS 8.8 ha | 0.697 | 0.056 | |
| TSI(SD) September | 1 | 76.0 ± 9.2 | 2004 VBS 8.2 ha | 0.676 | 0.676 |
| TSI(TP) June | 1 | 93.6 ± 11.7 | 2002 VBS 8.2 ha | 0.412 | 0.412 |
| 3 | 83.0 ± 9.4 | 2009 CT 78.4 ha | 0.549 | 0.137 | |
| TSI(TP) July | 1 | 88.2 ± 10.6 | 2006 QB 9 ha | 0.412 | 0.412 |
| 2 | 95.3 ± 7.5 | 2000 CT 52.1 ha | 0.479 | 0.067 | |
| 3 | 81.8 ± 8.7 | 2010 SP 1 ha | 0.543 | 0.065 | |
| TSI(TP) August | 1 | 85.5 ± 8.8 | 2010 SP 1 ha | 0.485 | 0.485 |
| 2 | 89.7 ± 5.2 | 2000 CT 52.1 ha | 0.584 | 0.099 | |
| 3 | 76.3 ± 8.5 | 2016 CT 135.4 ha | 0.676 | 0.092 | |
| 4 | 78.6 ± 7.3 | 2004 CT 119 ha | 0.740 | 0.064 | |
| TSI(TP) September | 1 | 87.1 ± 9.6 | 2010 SP 1 ha | 0.681 | 0.681 |
| TSI (Chl) June | 1 | 62.8 ± 12.5 | 2006 QB 9 ha | 0.064 | 0.064 |
| TSI (Chl) July | 1 | 63.4 ± 12.1 | 2006 QB 9 ha | 0.214 | 0.214 |
| 2 | 57.6 ± 11.8 | 2003 CRP 87.3 ha | 0.273 | 0.058 | |
| TSI (Chl) August | 1 | 61.7 ± 12.9 | 2006 QB 9 ha | 0.133 | 0.133 |
| 2 | 57.0 ± 15.1 | 1998 CT 113 ha | 0.194 | 0.061 | |
| 5 | 53.8 ± 17.2 | 2001 CT 254.7 ha | 0.245 | 0.050 | |
| TSI (Chl) September | 1 | 64.4 ± 10.9 | 2010 SP 1 ha | 0.194 | 0.194 |
| 2 | 61.3 ± 11.5 | 2012 CT 25.7 ha | 0.412 | 0.218 | |
| TSI 1977 June | 1 | 78 ± 8 | 2004 VBS 8.2 ha | 0.575 | 0.575 |
| TSI 1977 July | 1 | 75 ± 7 | 2004 VBS 8.2 ha | 0.359 | 0.359 |
| 2 | 83 ± 7 | 2009 CT 78.4 ha | 0.463 | 0.103 | |
| TSI 1977 August | 1 | 74 ± 5 | 2003 CRP 87.3 ha | 0.141 | 0.141 |
| 2 | 76 ± 5 | 1998 CT 113 ha | 0.216 | 0.075 | |
| TSI 1977 September | 1 | 76 ± 6 | 2003 CRP 87.3 ha | 0.367 | 0.367 |
| 2 | 80 ± 4 | 2001 VBS 9.1 ha | 0.462 | 0.094 | |
| 3 | 73 ± 3 | 2012 CT 25.7 ha | 0.557 | 0.096 | |
| 6 | 68 ± 8 | 2010 SP 1 ha | 0.639 | 0.082 | |
| TSI(Chl)–TSI(SD) June | 1 | −15.7 ± 20.4 | 2004 VBS 8.2 ha | 0.544 | 0.544 |
| 2 | −40.3 ± 21.4 | 1998 CT 113 ha | 0.635 | 0.091 | |
| TSI(Chl)–TSI(SD) July | 1 | −10.9 ± 16.4 | 2006 QB 9 ha | 0.407 | 0.407 |
| 2 | −22.1 ± 15.8 | 2010 CT 119 ha | 0.520 | 0.112 | |
| 4 | −28.7 ± 12.6 | 2009 CT 78.4 ha | 0.580 | 0.060 | |
| TSI(Chl)–TSI(SD) August | 1 | −11.4 ± 16.0 | 2006 QB 9 ha | 0.287 | 0.287 |
| 2 | −20.2 ± 17.2 | 2002 VBS 8.2 ha | 0.360 | 0.073 | |
| 4 | −27.1 ± 18.4 | 1998 CT 113 ha | 0.457 | 0.097 | |
| TSI(Chl)–TSI(SD) September | 1 | −11.9 ± 14.2 | 2004 VBS 8.2 ha | 0.379 | 0.379 |
| 3 | −6.8 ± 12.3 | 2011 CTR 35 ha | 0.431 | 0.052 | |
| 4 | −12.1 ± 17.7 | 2010 SP 1 ha | 0.566 | 0.135 | |
| TSI(Chl)–TSI(TP) June | 1 | −30.8 ± 19.1 | 2002 VBS 8.2 ha | 0.280 | 0.280 |
| 3 | −24.7 ± 14.3 | 1999 CT 78.4 ha | 0.358 | 0.078 | |
| 4 | −17.2 ± 13.3 | 2006 QB 9 ha | 0.422 | 0.063 | |
| TSI(Chl)–TSI(TP) July | 1 | −24.8 ± 16.8 | 2006 QB 9 ha | 0.547 | 0.547 |
| 3 | −13.1 ± 11.4 | 2010 SP 1 ha | 0.613 | 0.067 | |
| TSI(Chl)–TSI(TP) August | 1 | −24.1 ± 16.8 | 2006 QB 9 ha | 0.284 | 0.284 |
| 3 | −15.2 ± 13.5 | 2009 CT 78.4 ha | 0.351 | 0.067 | |
| 5 | −21.2 ± 15.5 | 2016 CT 135.4 ha | 0.410 | 0.059 | |
| 7 | −13.9 ± 12.0 | 2010 SP 1 ha | 0.471 | 0.061 | |
| TSI(Chl)–TSI(TP) September | 1 | −22.7 ± 16.4 | 2010 SP 1 ha | 0.602 | 0.602 |
| TSI(Chl)–TSI(TN) June | 1 | 5.6 ± 7.9 | 2010 SP 1 ha | 0.103 | 0.103 |
| TSI(Chl)–TSI(TN) July | 1 | 4.3 ± 13.2 | 2006 QB 9 ha | 0.206 | 0.206 |
| 2 | −5.2 ± 16.6 | 2002 VBS 8.8 ha | 0.324 | 0.118 | |
| 3 | 8.1 ± 9.4 | 2006 CT 161.9 ha | 0.384 | 0.061 | |
| TSI(Chl)–TSI(TN) September | 1 | 5.2 ± 11.2 | 2012 CT 25.7 ha | 0.147 | 0.147 |
| 2 | −6.1 ± 25.2 | 2010 SP 1 ha | 0.358 | 0.212 | |
| 3 | 6.8 ± 6.2 | 2006 QB 9 ha | 0.429 | 0.071 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lizotte, R.E., Jr.; Yasarer, L.M.W.; Bingner, R.L.; Locke, M.A.; Knight, S.S. Long-Term Oxbow Lake Trophic State under Agricultural Best Management Practices. Water 2021, 13, 1123. https://doi.org/10.3390/w13081123
Lizotte RE Jr., Yasarer LMW, Bingner RL, Locke MA, Knight SS. Long-Term Oxbow Lake Trophic State under Agricultural Best Management Practices. Water. 2021; 13(8):1123. https://doi.org/10.3390/w13081123
Chicago/Turabian StyleLizotte, Richard E., Jr., Lindsey M. W. Yasarer, Ronald L. Bingner, Martin A. Locke, and Scott S. Knight. 2021. "Long-Term Oxbow Lake Trophic State under Agricultural Best Management Practices" Water 13, no. 8: 1123. https://doi.org/10.3390/w13081123
APA StyleLizotte, R. E., Jr., Yasarer, L. M. W., Bingner, R. L., Locke, M. A., & Knight, S. S. (2021). Long-Term Oxbow Lake Trophic State under Agricultural Best Management Practices. Water, 13(8), 1123. https://doi.org/10.3390/w13081123








