Synthesis and Characterization of Pd-Ni Bimetallic Nanoparticles as Efficient Adsorbent for the Removal of Acid Orange 8 Present in Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Instruments Used
2.2. Instrumentation
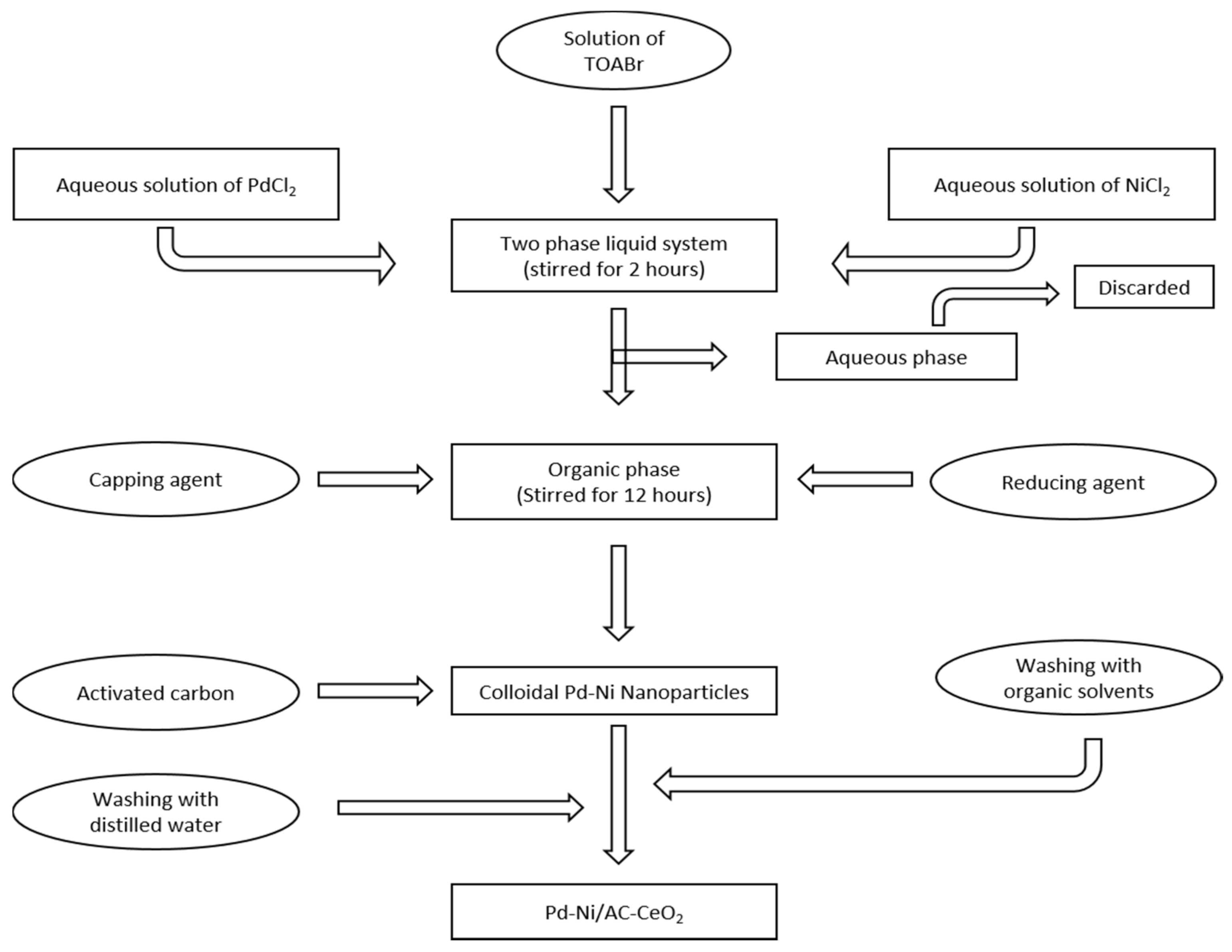
2.3. Synthesis of Pd-Ni Nanoparticles Supported on Activated Carbon and Cerium Oxide
2.4. Adsorption Experiments
3. Results and Discussion
3.1. Characterization
3.1.1. Scanning Electron Microscopy (SEM)
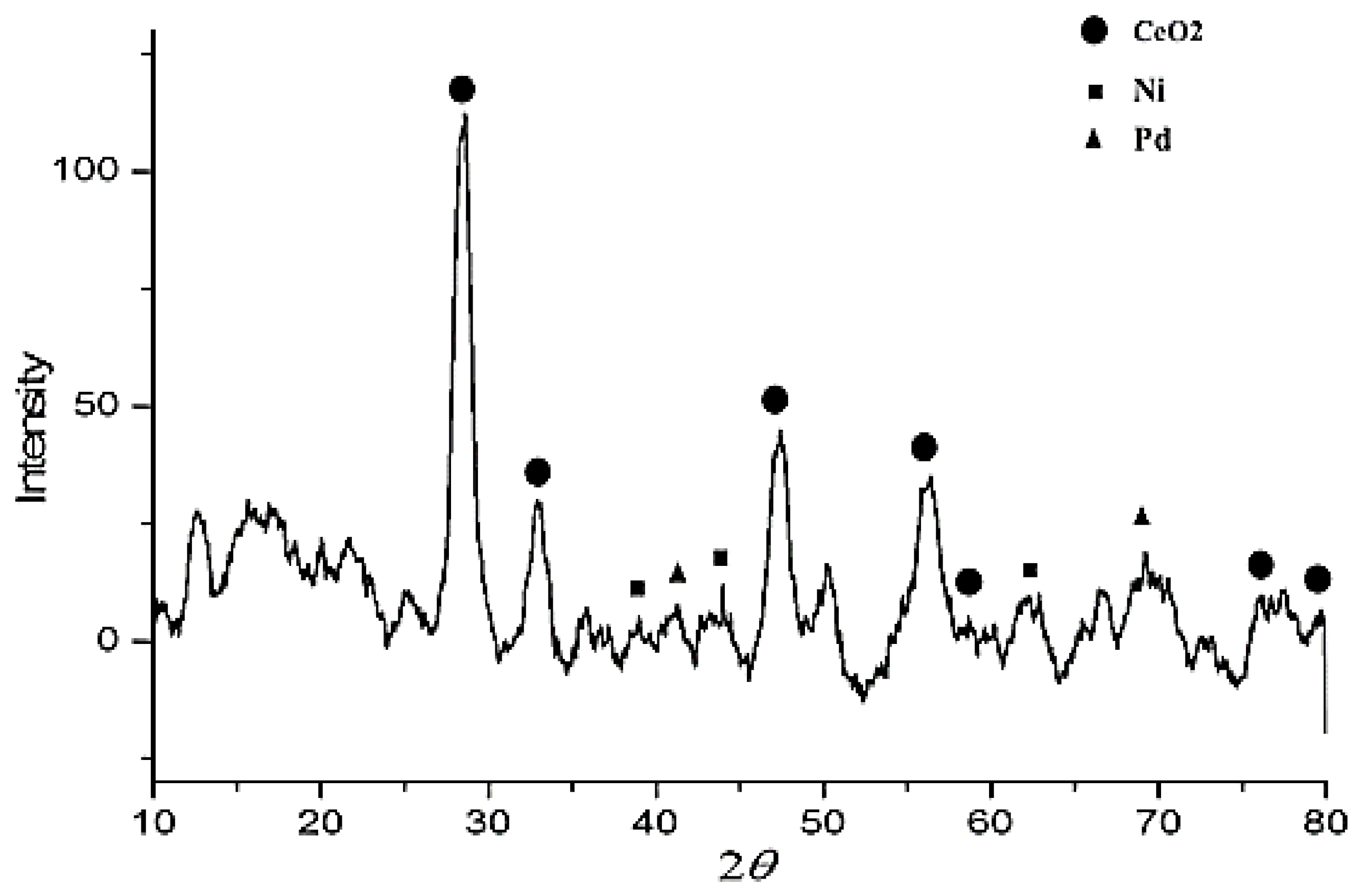
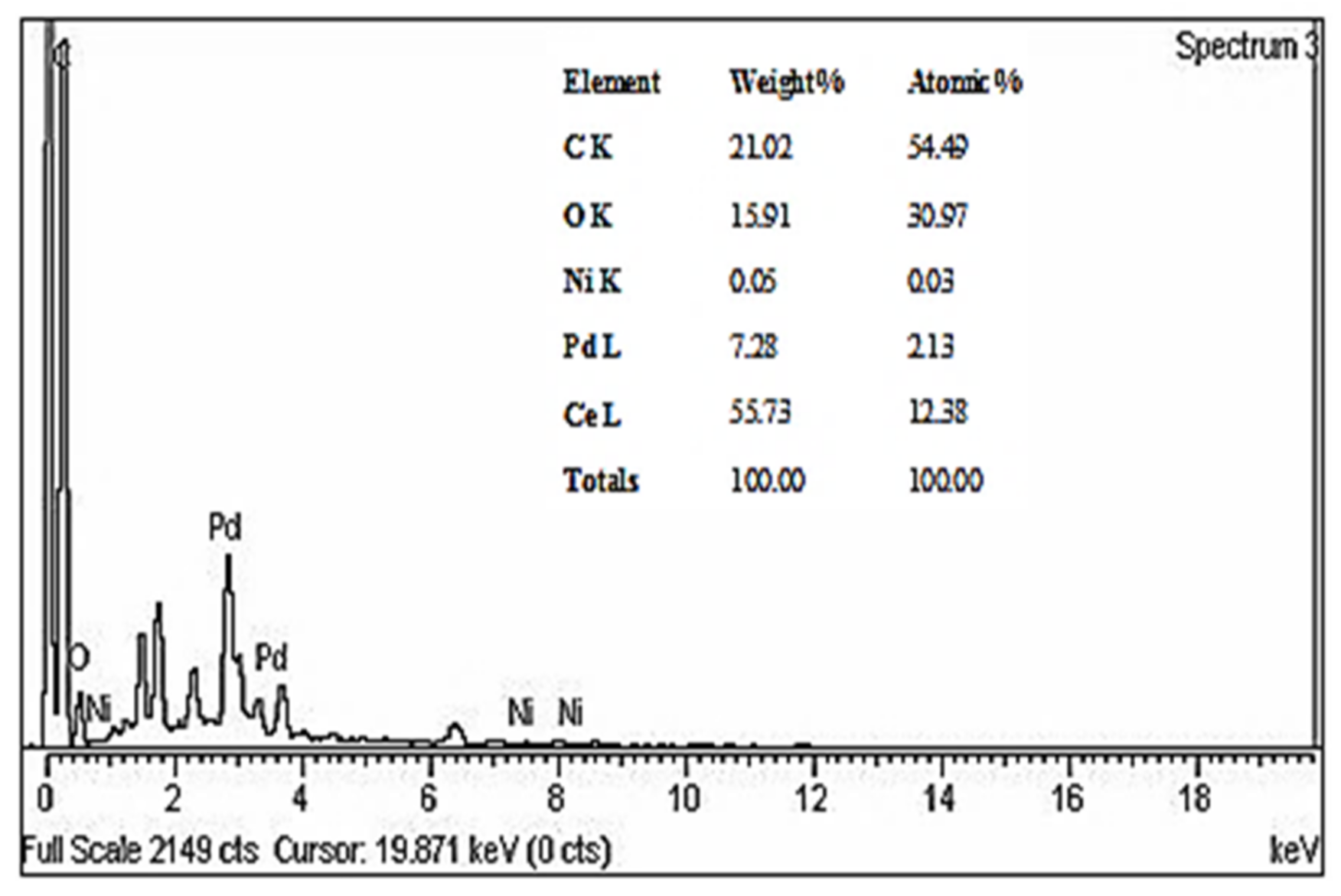
3.1.2. X-Ray Diffraction (XRD) and Energy-Dispersive X-Ray (EDX) Measures of Pd-Ni/AC-CeO2 Nanoparticles
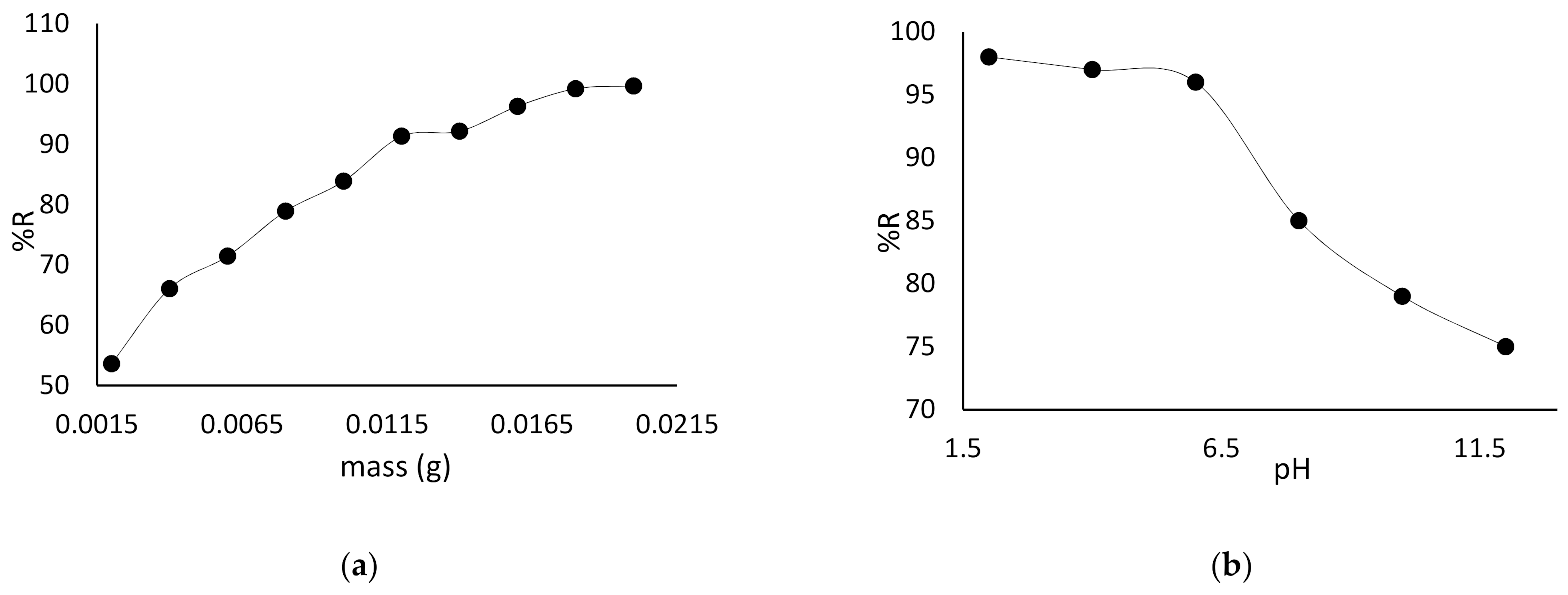
3.2. Effect of Adsorbent Dosage and pH
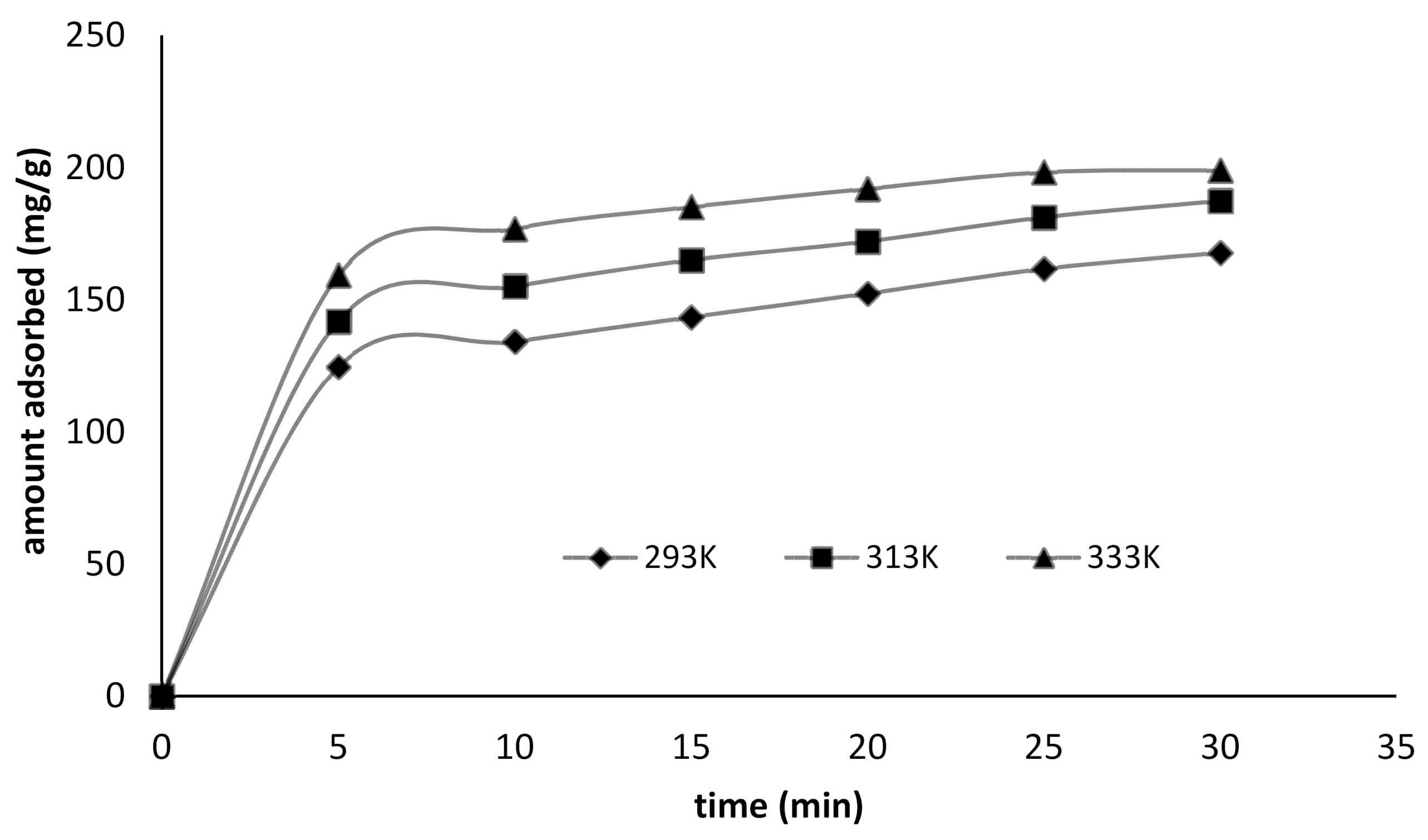
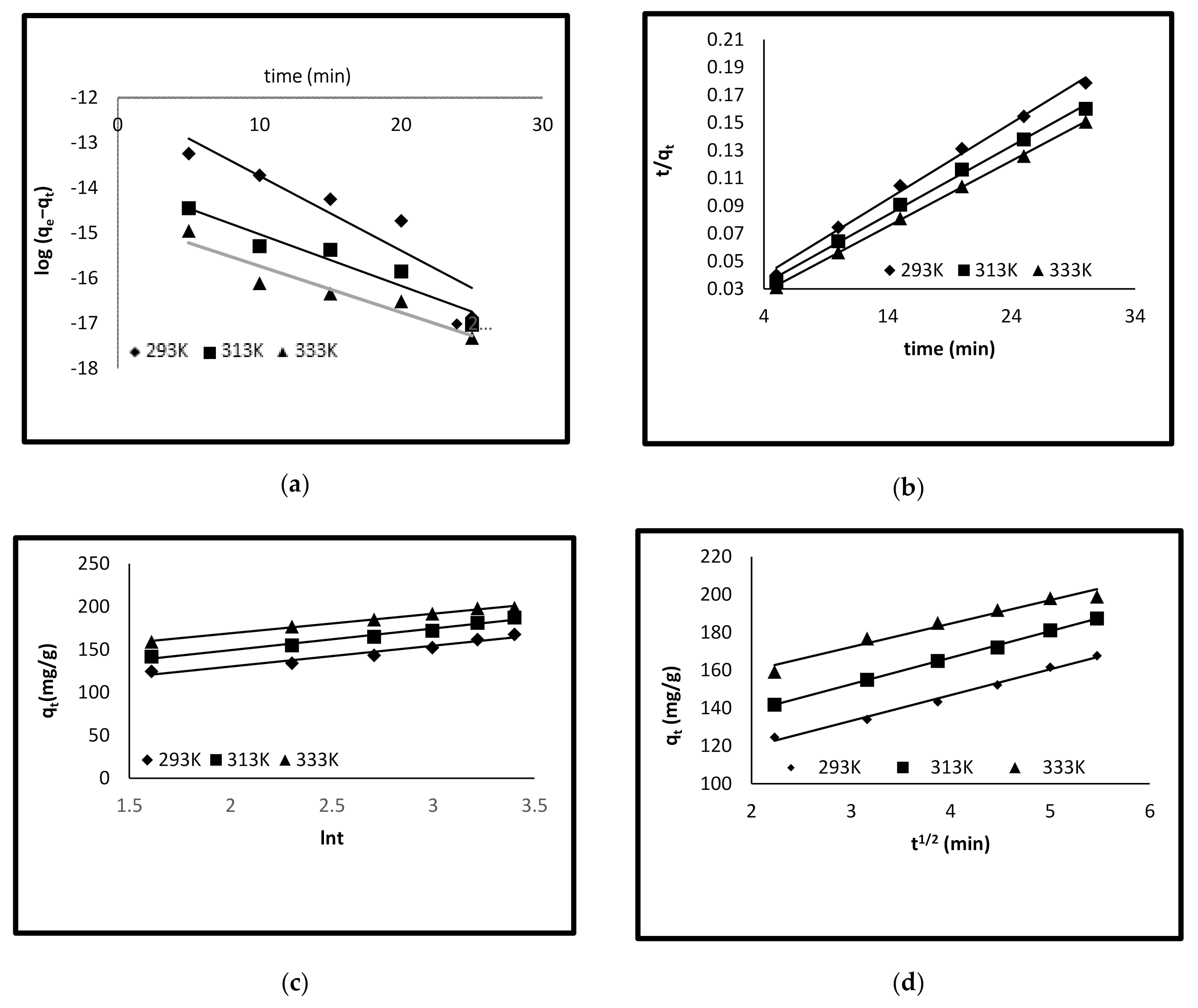
3.3. Adsorption Kinetics
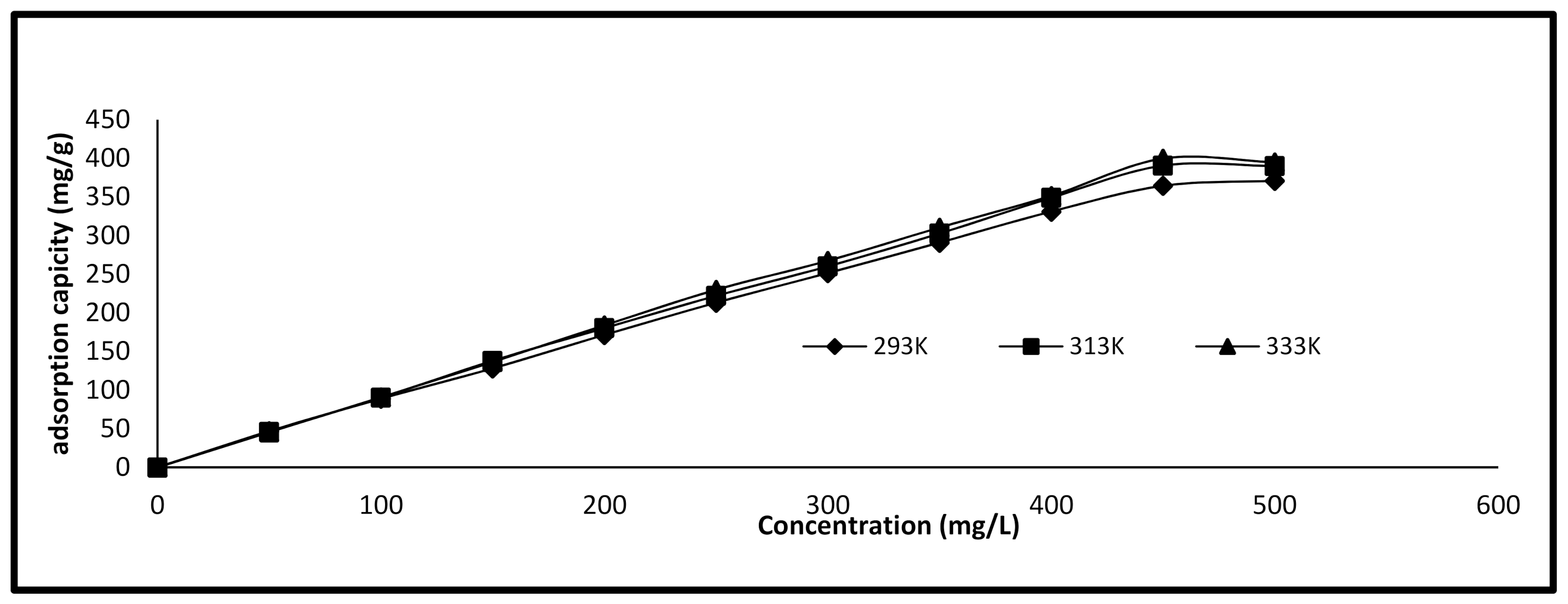
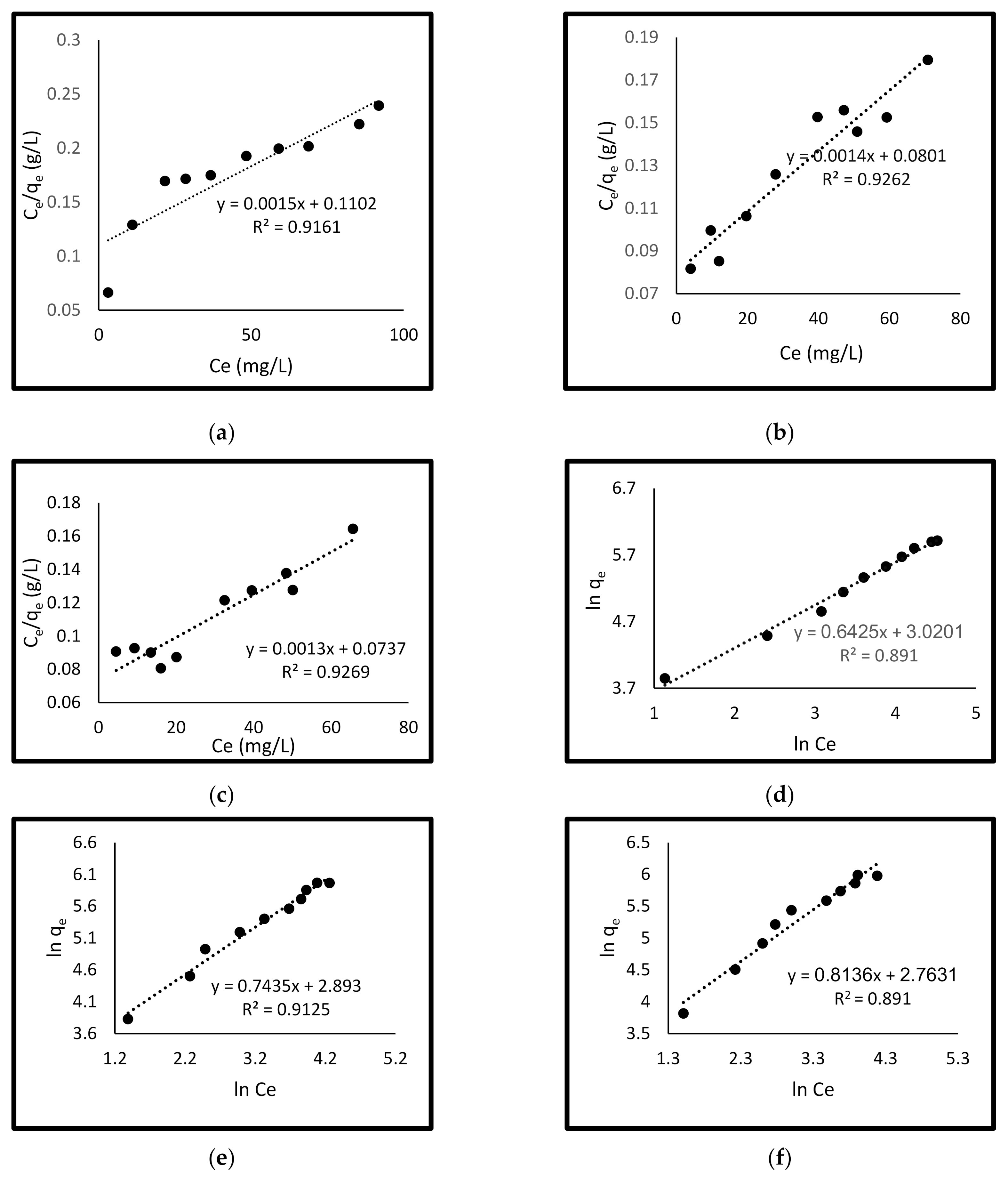
3.4. Isotherm Study
3.4.1. Langmuir Isotherm
3.4.2. Freundlich Isotherm Model
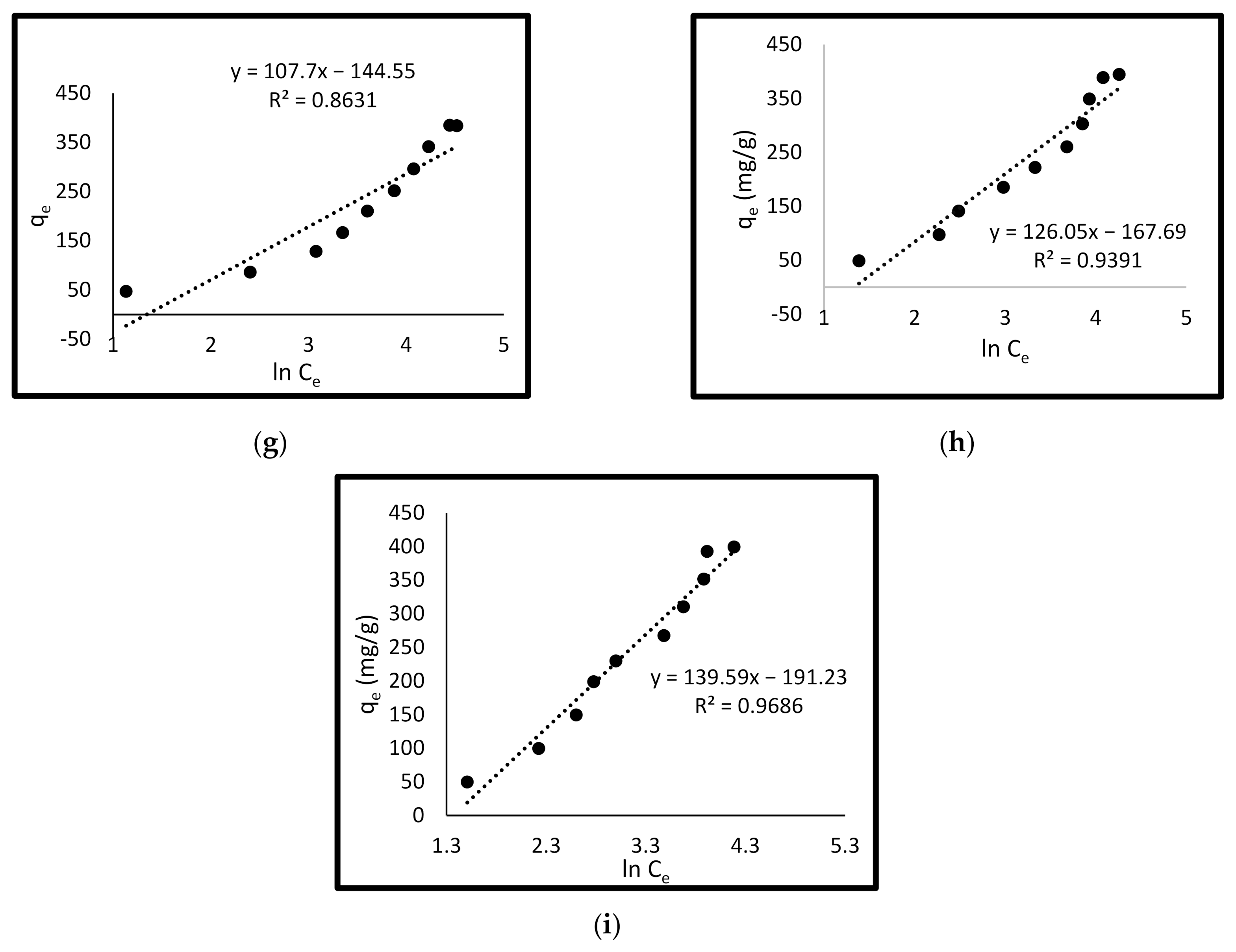
3.4.3. Temkin Isotherm Model
3.5. Thermodynamic Study
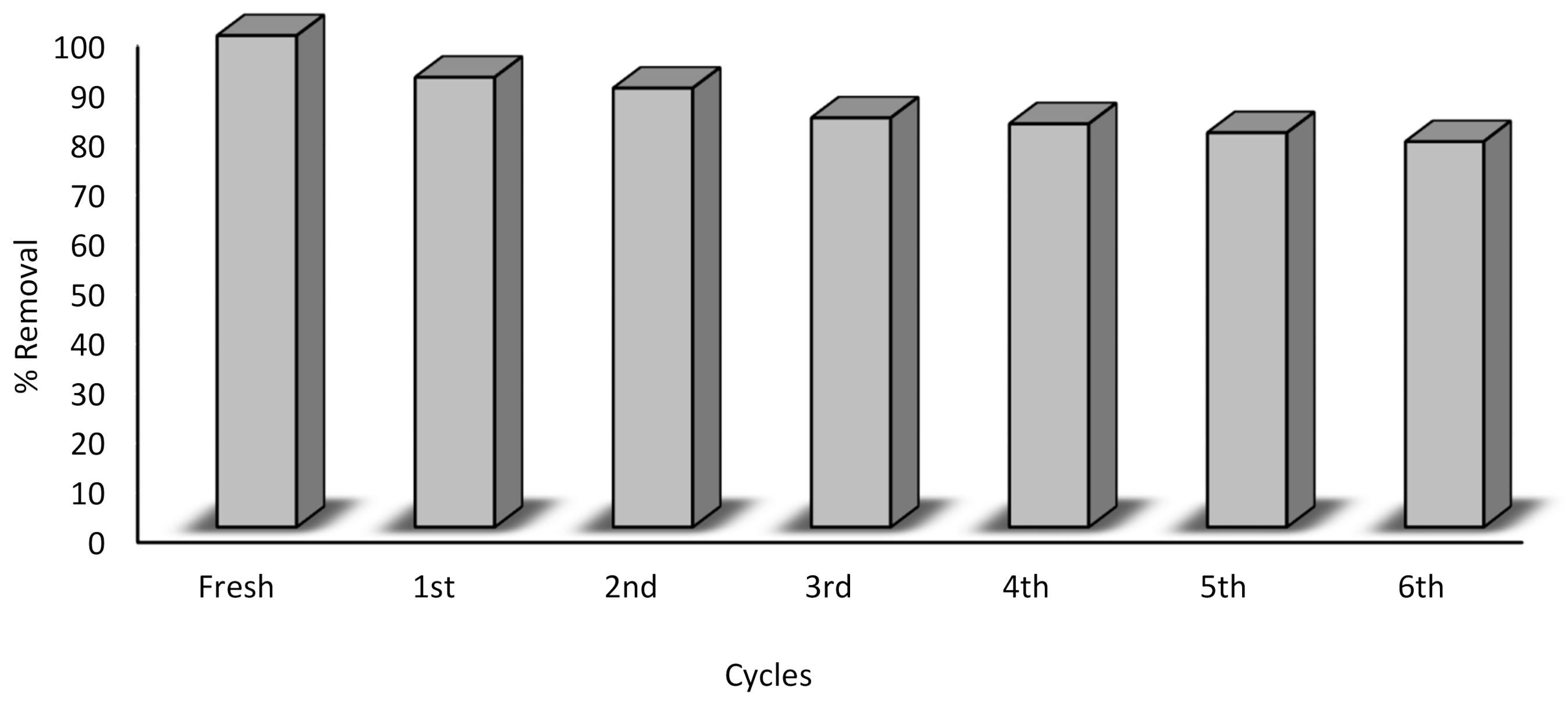
3.6. Regeneration of Pd-Ni/AC-CeO2
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ikram, M.; Zahoor, M.; Batiha, G.E.-S. Biodegradation and decolorization of textile dyes by bacterial strains: A biological approach for wastewater treatment. Z. Phys. Chem. 2020. [Google Scholar] [CrossRef]
- Khayam, S.M.U.; Zahoor, M.; Khan, E.; Shah, M. Reduction of keto group in drimarene blue by Aspergillus niger; a predominant reason for subsequent decolorization. Fresenius Environ. Bull. 2020, 29, 1397–1410. [Google Scholar]
- Ikram, M.; Zahoor, M.; Khan, E.; Khayam, S.M.U. Biodegradation of Novacron Turqueiose (Reactive Blue 21) by Pseudomonas aeruginosa. J. Chem. Soc. Pak. 2020, 42, 737–745. [Google Scholar]
- Sarkar, S.; Banerjee, A.; Halder, U.; Biswas, R.; Bandopadhyay, R. Degradation of Synthetic Azo Dyes of Textile Industry: A Sustainable Approach Using Microbial Enzymes. Water Conserv. Sci. Eng. 2017, 2, 121–131. [Google Scholar] [CrossRef] [Green Version]
- Wilson, M.; Kannangara, K.; Smith, G.; Simmons, M.; Raguse, B. Nanotechnology: Basic Science and Emerging Technologies; Chapman and Hall/CRC: London, UK, 2002. [Google Scholar]
- Kolhatkar, A.G.; Jamison, A.C.; Litvinov, D.; Willson, R.C.; Lee, T.R. Tuning the Magnetic Properties of Nanoparticles. Int. J. Mol. Sci. 2013, 14, 15977–16009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, D.; Xie, G.; Luo, J. Mechanical properties of nanoparticles: Basics and applications. J. Phys. D Appl. Phys. 2014, 47, 013001. [Google Scholar] [CrossRef] [Green Version]
- Sosa, I.O.; Noguez, C.; Barrera, R.G. Optical Properties of Metal Nanoparticles with Arbitrary Shapes. J. Phys. Chem. B 2003, 107, 6269–6275. [Google Scholar] [CrossRef] [Green Version]
- Schmid, G.; Simon, U. Gold nanoparticles: Assembly and electrical properties in 1–3 dimensions. Chem. Commun. 2005, 6, 697–710. [Google Scholar] [CrossRef]
- Delannoy, L.; El Hassan, N.; Musi, A.; Le To, N.N.; Krafft, J.-M.; Louis, C. Preparation of Supported Gold Nanoparticles by a Modified Incipient Wetness Impregnation Method. J. Phys. Chem. B 2006, 110, 22471–22478. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Cui, J.; Zheng, M.; Hu, C.; Tan, S.; Xiao, Y.; Yang, Q.; Liu, Y. One-step synthesis of amino-functionalized fluorescent carbon nanoparticles by hydrothermal carbonization of chi-tosan. Chem. Commun. 2012, 48, 380–382. [Google Scholar] [CrossRef]
- Makovec, D.; Košak, A.; Žnidaršič, A.; Drofenik, M. The synthesis of spinel–ferrite nanoparticles using precipitation in microemulsions for ferrofluid applications. J. Magn. Magn. Mater. 2005, 289, 32–35. [Google Scholar] [CrossRef]
- Chen, Q.; Rondinone, A.J.; Chakoumakos, B.C.; Zhang, Z.J. Synthesis of superparamagnetic MgFe2O4 nanoparticles by coprecipitation. J. Magn. Magn. Mater. 1999, 194, 1–7. [Google Scholar] [CrossRef]
- Acsente, T.; Negrea, R.F.; Nistor, L.C.; Logofatu, C.; Matei, E.; Birjega, R.; Grisolia, C.; Dinescu, G. Synthesis of flower-like tungsten nanoparticles by magnetron sputtering combined with gas aggregation. Eur. Phys. J. D 2015, 69, 161. [Google Scholar] [CrossRef]
- Swihart, M.T. Vapor-phase synthesis of nanoparticles. Curr. Opin. Colloid Interface Sci. 2003, 8, 127–133. [Google Scholar] [CrossRef]
- Sanvicens, N.; Marco, M.P. Multifunctional nanoparticles–properties and prospects for their use in human medicine. Trends Biotechnol. 2008, 26, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Kokura, S.; Handa, O.; Takagi, T.; Ishikawa, T.; Naito, Y.; Yoshikawa, T. Silver nanoparticles as a safe preservative for use in cosmetics. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 570–574. [Google Scholar] [CrossRef]
- Wei, Q. Surface Modification of Textiles; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Nawaz, H.R.; Solangi, B.A.; Zehra, B.; Nadeem, U. Preparation of nano zinc oxide and its application in leather as a retanning and antibacterial agent. Can. J. Sci. Ind. Res. 2011, 2, 164–170. [Google Scholar]
- Ogolo, N.A.; Olafuyi, O.A.; Onyekonwu, M.O. Enhanced Oil Recovery Using Nanoparticles. In Proceedings of the SPE Saudi Arabia Section Technical Symposium and Exhibition, Al-Khobar, Saudi Arabia, 8–11 April 2012. [Google Scholar] [CrossRef] [Green Version]
- Xia, Y.; Yang, H.; Campbell, C.T. Nanoparticles for Catalysis. Acc. Chem. Res. 2013, 46, 1671–1672. [Google Scholar] [CrossRef]
- Sankar, R.; Manikandan, P.; Malarvizhi, V.; Fathima, T.; Shivashangari, K.S.; Ravikumar, V. Green synthesis of colloidal copper oxide nanoparticles using Carica papaya and its application in photocatalytic dye degradation. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 121, 746–750. [Google Scholar] [CrossRef]
- Mak, S.-Y.; Chen, D.-H. Fast adsorption of methylene blue on polyacrylic acid-bound iron oxide magnetic nanoparticles. Dye. Pigment. 2004, 61, 93–98. [Google Scholar] [CrossRef]
- Saifuddin, N.; Nian, C.; Zhan, L.; Ning, K. Chitosan-silver Nanoparticles Composite as Point-of-use Drinking Water Filtration System for Household to Remove Pesticides in Water. Asian J. Biochem. 2011, 6, 142–159. [Google Scholar] [CrossRef] [Green Version]
- Couvreur, P.; Kante, B.; Roland, M.; Speiser, P. Adsorption of Antineoplastic Drugs to Polyalkylcyanoacrylate Nanoparticles and Their Release in Calf Serum. J. Pharm. Sci. 1979, 68, 1521–1524. [Google Scholar] [CrossRef]
- Rahmani, A.; Mousavi, H.Z.; Fazli, M. Effect of nanostructure alumina on adsorption of heavy metals. Desalination 2010, 253, 94–100. [Google Scholar] [CrossRef]
- Gautam, R.K.; Rawat, V.; Banerjee, S.; Sanroman, M.A.; Soni, S.; Singh, S.K.; Chattopadhyaya, M.C. Synthesis of bimetallic Fe–Zn nanoparticles and its application towards adsorptive removal of carcinogenic dye malachite green and Congo red in water. J. Mol. Liq. 2015, 212, 227–236. [Google Scholar] [CrossRef]
- Duran, C.; Ozdes, D.; Gundogdu, A.; Senturk, H.B. Kinetics and Isotherm Analysis of Basic Dyes Adsorption onto Almond Shell (Prunus dulcis) as a Low Cost Adsorbent. J. Chem. Eng. Data 2011, 56, 2136–2147. [Google Scholar] [CrossRef]
- Chen, K.-C.; Wu, J.-Y.; Huang, C.-C.; Liang, Y.-M.; Hwang, S.-C.J. Decolorization of azo dye using PVA-immobilized microorganisms. J. Biotechnol. 2003, 101, 241–252. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Naiya, T.; Mandal, S.; Das, S. Adsorption, kinetics and equilibrium studies on removal of Cr(VI) from aqueous solutions using different low-cost adsorbents. Chem. Eng. J. 2008, 137, 529–541. [Google Scholar] [CrossRef]
- Belhachemi, M.; Addoun, F. Comparative adsorption isotherms and modeling of methylene blue onto activated carbons. Appl. Water Sci. 2011, 1, 111–117. [Google Scholar] [CrossRef] [Green Version]
- Ghaedi, M.; Heidarpour, S.; Kokhdan, S.N.; Sahraie, R.; Daneshfar, A.; Brazesh, B. Comparison of silver and palladium nanoparticles loaded on activated carbon for efficient removal of Methylene blue: Kinetic and isotherm study of removal process. Powder Technol. 2012, 228, 18–25. [Google Scholar] [CrossRef]
- Asfaram, A.; Ghaedi, M.; Hajati, S.; Rezaeinejad, M.; Goudarzi, A.; Purkait, M.K. Rapid removal of Auramine-O and Methylene blue by ZnS: Cu nanoparticles loaded on activated carbon: A re-sponse surface methodology approach. J. Taiwan Inst. Chem. Eng. 2015, 53, 80–91. [Google Scholar] [CrossRef]
- Roosta, M.; Ghaedi, M.; Shokri, N.; Daneshfar, A.; Sahraei, R.; Asghari, A. Optimization of the combined ultrasonic assisted/adsorption method for the removal of malachite green by gold nanoparticles loaded on activated carbon: Experimental design. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 118, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Jia, Y.; Wu, X.; Wang, H. Removal of arsenic from water by supported nano zero-valent iron on activated carbon. J. Hazard. Mater. 2009, 172, 1591–1596. [Google Scholar] [CrossRef]
- Arabzadeh, S.; Ghaedi, M.; Ansari, A.; Taghizadeh, F.; Rajabi, M. Comparison of nickel oxide and palladium nanoparticle loaded on activated carbon for efficient removal of meth-ylene blue: Kinetic and isotherm studies of removal process. Human Exp. Toxicol. 2015, 34, 153–169. [Google Scholar] [CrossRef] [PubMed]
- da Silva, A.; Bion, N.; Epron, F.; Baraka, S.; Fonseca, F.; Rabelo-Neto, R.; Mattos, L.; Noronha, F. Effect of the type of ceria dopant on the performance of Ni/CeO2 SOFC anode for ethanol internal reforming. Appl. Catal. B Environ. 2017, 206, 626–641. [Google Scholar] [CrossRef]
- Chrzan, M.; Chlebda, D.; Jodłowski, P.; Salomon, E.; Kołodziej, A.; Gancarczyk, A.; Sitarz, M.; Łojewska, J. Towards methane combustion mechanism on metal oxides supported catalysts: Ceria supported palladium catalysts. Top. Catal. 2019, 62, 403–412. [Google Scholar] [CrossRef] [Green Version]
- Önal, Y.; Akmil-Başar, C.; Sarıcı-Özdemir, Ç. Investigation kinetics mechanisms of adsorption malachite green onto activated carbon. J. Hazard. Mater. 2007, 146, 194–203. [Google Scholar] [CrossRef]
- Crini, G. Kinetic and equilibrium studies on the removal of cationic dyes from aqueous solution by adsorption onto a cyclodextrin polymer. Dye. Pigment. 2008, 77, 415–426. [Google Scholar] [CrossRef]
- Cheung, W.; Szeto, Y.; McKay, G. Intraparticle diffusion processes during acid dye adsorption onto chitosan. Bioresour. Technol. 2007, 98, 2897–2904. [Google Scholar] [CrossRef]
- Largitte, L.; Pasquier, R. A review of the kinetics adsorption models and their application to the adsorption of lead by an activat-ed carbon. Chem. Eng. Res. Design 2016, 109, 495–504. [Google Scholar] [CrossRef]
- Qiu, H.; Lv, L.; Pan, B.-C.; Zhang, Q.-J.; Zhang, W.-M.; Zhang, Q.-X. Critical review in adsorption kinetic models. J. Zhejiang Univ. A 2009, 10, 716–724. [Google Scholar] [CrossRef]
- Saxena, M.; Sharma, N.; Saxena, R. Highly efficient and rapid removal of a toxic dye: Adsorption kinetics, isotherm, and mechanism studies on functionalized multiwalled carbon nanotubes. Surfaces Interfaces 2020, 21, 100639. [Google Scholar] [CrossRef]
- Langmuir, I. THE ADSORPTION OF GASES ON PLANE SURFACES OF GLASS, MICA AND PLATINUM. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef] [Green Version]
- Freundlich, H. Over the adsorption in solution. J. Phys. Chem. 1906, 57, 1100–1107. [Google Scholar]
- Munagapati, V.S.; Kim, D.-S. Equilibrium isotherms, kinetics, and thermodynamics studies for congo red adsorption using calcium alginate beads impregnated with nano-goethite. Ecotoxicol. Environ. Saf. 2017, 141, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Namasivayam, C.; Senthilkumar, S. Recycling of industrial solid waste for the removal of mercury (II) by adsorption process. Chemosphere 1997, 34, 357–375. [Google Scholar] [CrossRef]
- Aljeboree, A.M.; Baqir, S.J.; Alkaim, A.F. Experimental studies of Thermodynamics parameters: As a model Adsorption and Removal of Textile. J. Phys. Conf. Ser. 2020, 1664, 012099. [Google Scholar] [CrossRef]
- Tenno, T.; Rikmann, E.; Zekker, I.; Tenno, T. Modelling the solubility of sparingly soluble compounds depending on their particles size. Proc. Est. Acad. Sci. 2018, 67, 300–302. [Google Scholar] [CrossRef]
- Zekker, I.; Tenno, T.; Selberg, A.; Uiga, K. Dissolution Modeling and Experimental Measurement of CaS-H2O Binary System. Chin. J. Chem. 2011, 29, 2327–2336. [Google Scholar] [CrossRef]
- Tenno, T.; Rikmann, E.; Uiga, K.; Zekker, I.; Mashirin, A.; Tenno, T. A novel proton transfer model of the closed equilibrium system H2O–CO2–CaCO3–NHX. Proc. Est. Acad. Sci. 2018, 4017, 2. [Google Scholar] [CrossRef]
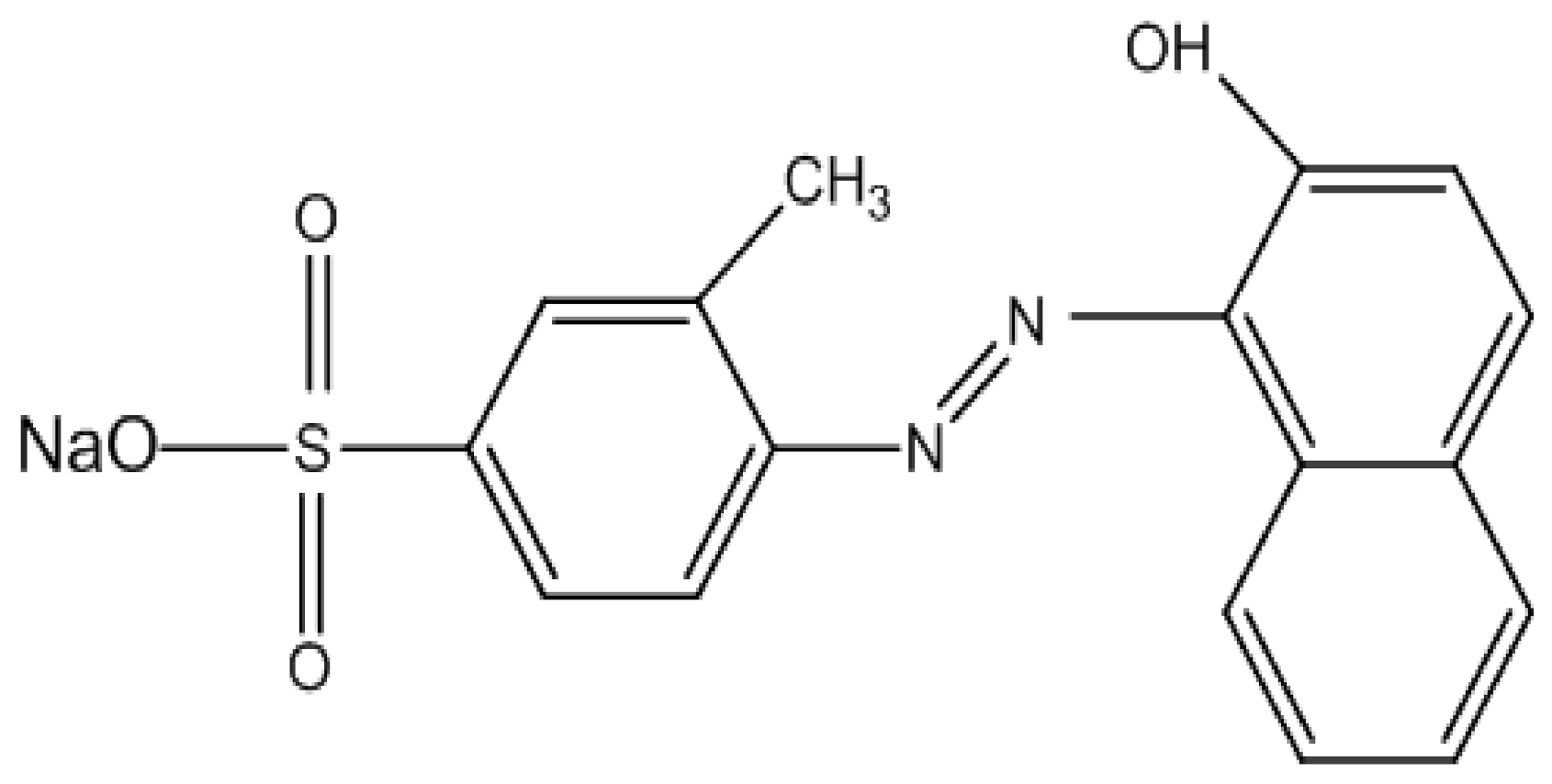


| Name of Dye | Acid Orange 8 |
|---|---|
| Molecular Formula | C17H13N2NaO4S |
| Molecular Weight | 364.4 g/mol |
| Λ max | 490 nm |
| Dye content | 65 % |
| Water solubility | Soluble in water 1 mg/mL |
| Solubility in other solvents | Acetone and ethanol, insoluble in other organic solvents |
| Refractive Index | n20D ~1.66 |
| Complexity | 566 |
| Manufacturing method | 4-Amino 3 methylebenzenesulfonic acid dia azo and Naphtalen-2-ol Coupling |
| Usage | wool, silk, cotton, vinegar, polyamide fiber, paper, leather dyeing |
| Parameters | 293 K | 313 K | 333 K |
|---|---|---|---|
| qe (mol/g) | 167.764 | 187.3706 | 198.9441 |
| %R | 83.87 | 93.68 | 99.47 |
| Pseudo-first order | |||
| qe (cal) (mg/g) | 82.22 | 82.451 | 99.174 |
| k1 (min−1) | −0.0937 | −0.0946 | −0.1473 |
| R2 | 0.9264 | 0.949 | 0.9478 |
| Pseudo-second order | |||
| qe (cal) (mg/g) | 181.81 | 200 | 212 |
| k2 (g/mg/min) | 0.0016 | 0.00181 | 0.00252 |
| R2 | 0.9942 | 0.9967 | 0.9996 |
| Elovich equation | |||
| α (mg/g min) | 691.83 | 1270.6 | 5152.3 |
| β (mg/g min) | 0.041 | 0.0397 | 0.044 |
| R2 | 0.9534 | 0.980 | 0.9933 |
| Intra-particle diffusion | |||
| kid (mg/g min −1/2) | 13.623 | 14.022 | 12.364 |
| C | 92.362 | 110.5 | 135.16 |
| R2 | 0.9918 | 0.9988 | 0.9634 |
| Parameter | 293 K | 313 K | 333 K |
|---|---|---|---|
| Langmuir isotherm model | |||
| Qm (mg/g) | 666.6 | 714 | 769 |
| Ka (g/mg) | 0.0136 | 0.0174 | 0.01763 |
| R2 | 0.9161 | 0.9262 | 0.9269 |
| RL | 0.128 | 0.103 | 0.101 |
| Freundlich isotherm model | |||
| 1/n | 0.6425 | 0.7435 | 0.8136 |
| Kf | 20.46 | 18.03 | 15.81 |
| R2 | 0.891 | 0.9125 | 0.891 |
| Temkin isotherm model | |||
| B1 | 107.7 | 126.05 | 139.59 |
| KT | 0.261 | 0.265 | 0.255 |
| R2 | 0.8631 | 0.9391 | 0.9686 |
| ΔG° kJ/mol | ΔH° kJ/mol | ΔS° kJ/ molK | ||
|---|---|---|---|---|
| 293 K | 313 K | 333 K | ||
| −18.7327 | −19.4870 | −20.584 | −7.593 | 0.0384 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umar, A.; Khan, M.S.; Alam, S.; Zekker, I.; Burlakovs, J.; dC Rubin, S.S.; Bhowmick, G.D.; Kallistova, A.; Pimenov, N.; Zahoor, M. Synthesis and Characterization of Pd-Ni Bimetallic Nanoparticles as Efficient Adsorbent for the Removal of Acid Orange 8 Present in Wastewater. Water 2021, 13, 1095. https://doi.org/10.3390/w13081095
Umar A, Khan MS, Alam S, Zekker I, Burlakovs J, dC Rubin SS, Bhowmick GD, Kallistova A, Pimenov N, Zahoor M. Synthesis and Characterization of Pd-Ni Bimetallic Nanoparticles as Efficient Adsorbent for the Removal of Acid Orange 8 Present in Wastewater. Water. 2021; 13(8):1095. https://doi.org/10.3390/w13081095
Chicago/Turabian StyleUmar, Ali, Muhammad Sufaid Khan, Sultan Alam, Ivar Zekker, Juris Burlakovs, Steven S. dC Rubin, Gourav Dhar Bhowmick, Anna Kallistova, Nikolai Pimenov, and Muhammad Zahoor. 2021. "Synthesis and Characterization of Pd-Ni Bimetallic Nanoparticles as Efficient Adsorbent for the Removal of Acid Orange 8 Present in Wastewater" Water 13, no. 8: 1095. https://doi.org/10.3390/w13081095







