Distribution and Geochemical Processes of Arsenic in Lake Qinghai Basin, China
Abstract
1. Introduction
2. Materials and Methods
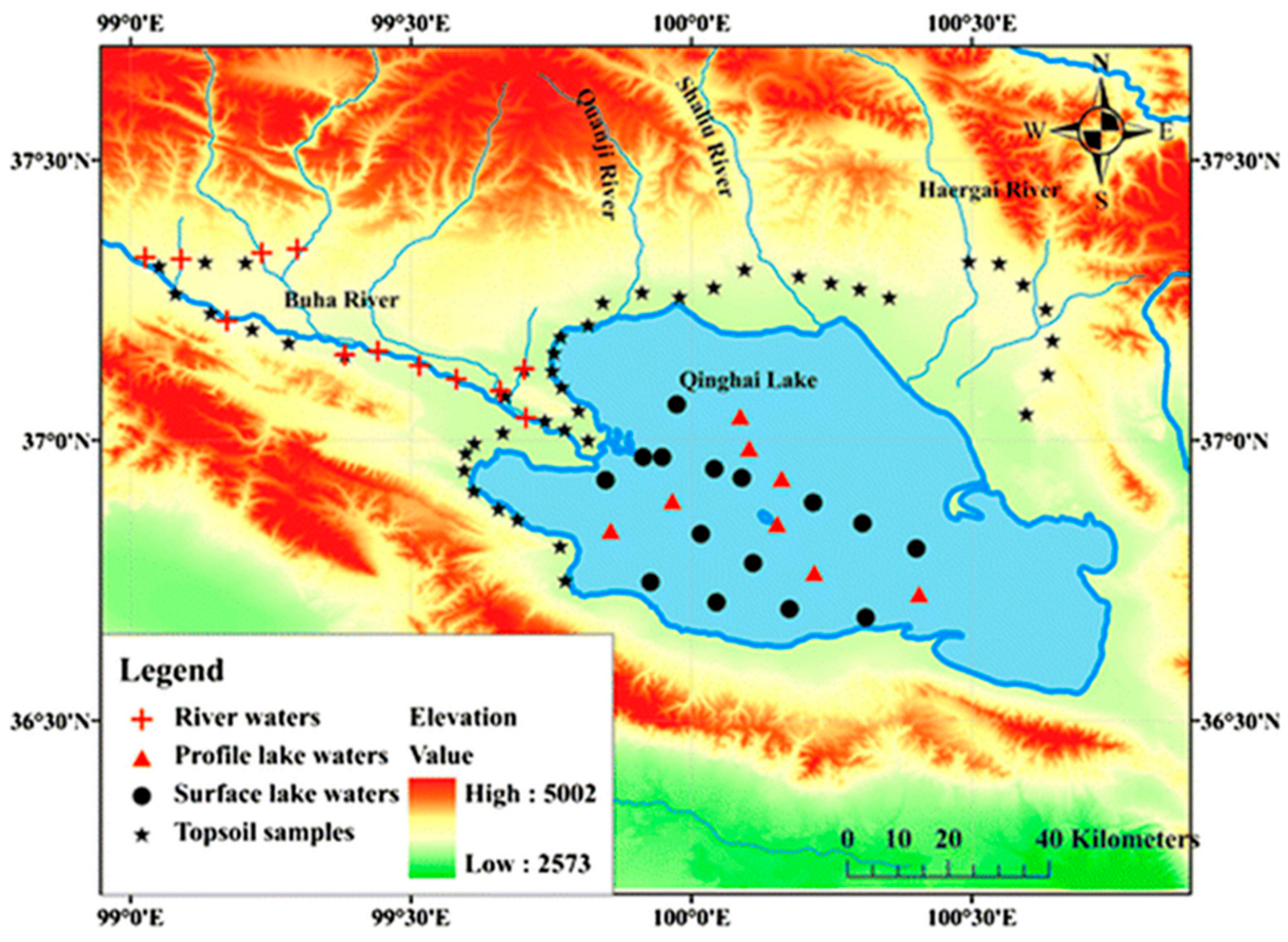
2.1. Study Area
2.2. Sample Collection
2.3. Sample Analysis
2.4. Quality Assurance and Control
3. Results and Discussion
3.1. Properties of the Water, Sediment, and Soil
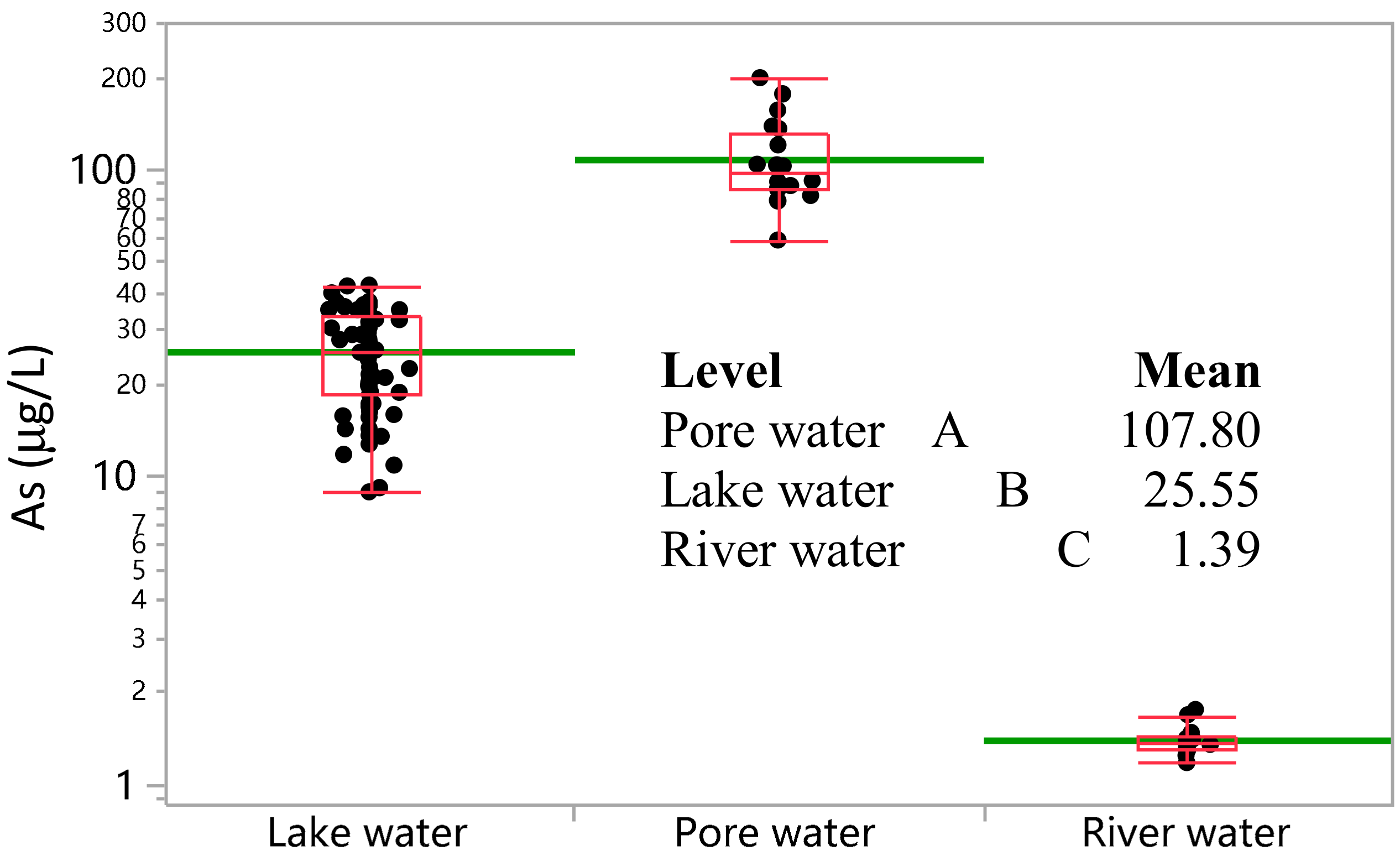
3.2. Concentration of As in the Water, Sediment, and Soil
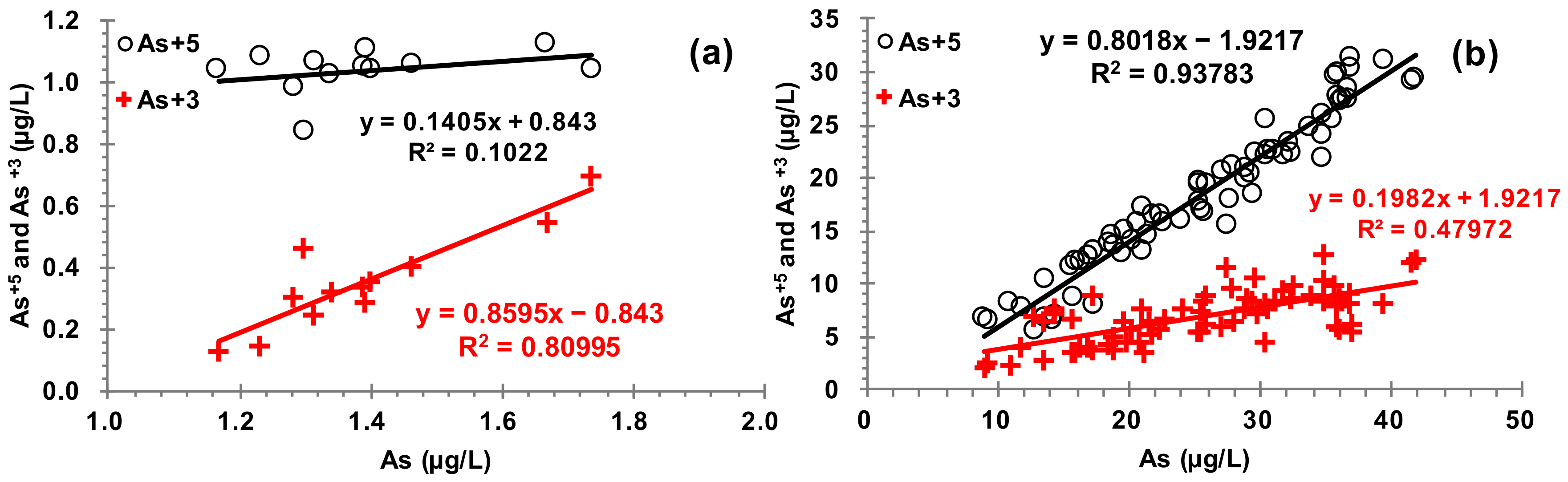
3.3. Relationships between As and Other Elements in the Water, Sediment, and Soil
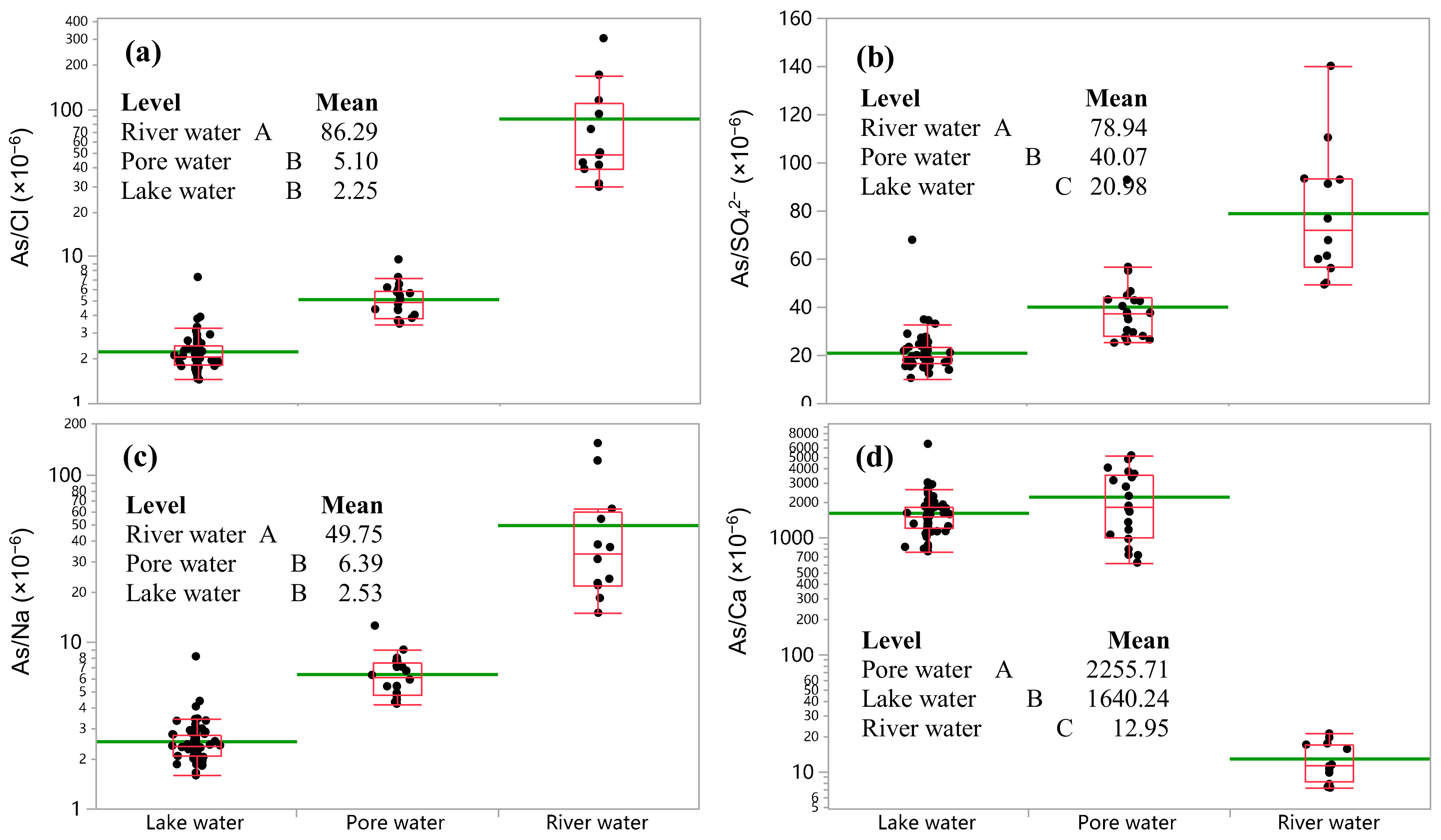
3.4. Ratio of As to Major or Reference Elements
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Camarero, L.; Rogora, M.; Mosello, R.; Anderson, N.J.; Barbieri, A.; Botev, I.; Kernan, M.; Kopáček, J.; Korhola, A.; Lotter, A.F.; et al. Regionalisation of chemical variability in European mountain lakes. Freshw. Biol. 2009, 54, 2452–2469. [Google Scholar] [CrossRef]
- Camarero, L.; Botev, I.; Muri, G.; Psenner, R.; Rose, N.; Stuchlik, E. Trace elements in alpine and arctic lake sediments as a record of diffuse atmospheric contamination across Europe. Freshw. Biol. 2009, 54, 2518–2532. [Google Scholar] [CrossRef]
- Battarbee, R.W.; Thompson, R.; Catalan, J.; Grytnes, J.; Birks, H. Climate variability and ecosystem dynamics of remote alpine and arctic lakes: The MOLAR project. J. Paleolimnol. 2002, 28, 1–6. [Google Scholar] [CrossRef]
- Jin, Z.; You, C.-F.; Yu, J. Toward a geochemical mass balance of major elements in Lake Qinghai, NE Tibetan Plateau: A significant role of atmospheric deposition. Appl. Geochem. 2009, 24, 1901–1907. [Google Scholar] [CrossRef]
- Xu, H.; Hou, Z.; An, Z.; Liu, X.; Dong, J. Major ion chemistry of waters in Lake Qinghai catchments, NE Qinghai-Tibet plateau, China. Quat. Int. 2010, 212, 35–43. [Google Scholar] [CrossRef]
- Li, X.; Liu, W.; Xu, L. Carbon isotopes in surface-sediment carbonates of modern Lake Qinghai (Qinghai–Tibet Plateau): Implications for lake evolution in arid areas. Chem. Geol. 2012, 300, 88–96. [Google Scholar] [CrossRef]
- Han, Y.M.; Wei, C.; Bandowe, B.A.M.; Wilcke, W.; Cao, J.J.; Xu, B.Q.; Gao, S.P.; Tie, X.X.; Li, G.H.; Jin, Z.D.; et al. Elemental Carbon and Polycyclic Aromatic Compounds in a 150-Year Sediment Core from Lake Qinghai, Tibetan Plateau, China: Influence of Regional and Local Sources and Transport Pathways. Environ. Sci. Technol. 2015, 49, 4176–4183. [Google Scholar] [CrossRef] [PubMed]
- Sha, Z.; Wang, Q.; Wang, J.; Du, J.; Hu, J.; Ma, Y.; Kong, F.; Wang, Z. Regional environmental change and human activity over the past hundred years recorded in the sedimentary record of Lake Qinghai, China. Environ. Sci. Pollut. Res. 2017, 24, 9662–9674. [Google Scholar] [CrossRef]
- Lin, C.; Xue, Y.; Zhang, X.; Renqin, D.; Zhou, X.; Cao, Y.; Guo, B.; Liu, X. Distribution of cadmium among multimedia in Lake Qinghai, China. Environ. Earth Sci. 2018, 77, 153. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, C.; Guo, B.; Cao, Y.; Lei, K.; Zhou, X.; Renqin, D. Distribution and geochemical processes of boron in the multimedia of Lake Qinghai, China. J. Great Lakes Res. 2018, 44, 1035–1042. [Google Scholar] [CrossRef]
- Xiong, X.; Zhang, K.; Chen, X.; Shi, H.; Luo, Z.; Wu, C. Sources and distribution of microplastics in China’s largest inland lake–Qinghai Lake. Environ. Pollut. 2018, 235, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Kabata-Pendias, A.; Mukherjee, A.B. Trace Elements from Soil to Human; Springer: Berlin, Germany, 2007; pp. 1–550. [Google Scholar]
- Alexakis, D. Multielement Contamination of Land in the Margin of Highways. Land 2021, 10, 230. [Google Scholar] [CrossRef]
- Gamvroula, D.; Alexakis, D.; Stamatis, G. Diagnosis of groundwater quality and assessment of contamination sources in the Megara basin (Attica, Greece). Arab. J. Geosci. 2012, 6, 2367–2381. [Google Scholar] [CrossRef]
- Alexakis, D. Human health risk assessment associated with Co, Cr, Mn, Ni and V contents in agricultural soils from a Mediterranean site. Arch. Argon. Soil Sci. 2015, 62, 359–373. [Google Scholar] [CrossRef]
- Caeiro, S.; Costa, M.; Ramos, T.; Fernandes, F.; Silveira, N.; Coimbra, A.; Medeiros, G.; Painho, M. Assessing heavy metal contamination in Sado Estuary sediment: An index analysis approach. Ecol. Indic. 2005, 5, 151–169. [Google Scholar] [CrossRef]
- Chang, J.-S.; Yoon, I.-H.; Kim, K.-W. Heavy metal and arsenic accumulating fern species as potential ecological indicators in As-contaminated abandoned mines. Ecol. Indic. 2009, 9, 1275–1279. [Google Scholar] [CrossRef]
- Kempter, H.; Krachler, M.; Shotyk, W.; Zaccone, C. Major and trace elements in Sphagnum moss from four southern German bogs, and comparison with available moss monitoring data. Ecol. Indic. 2017, 78, 19–25. [Google Scholar] [CrossRef]
- Schreck, E.; Sarret, G.; Oliva, P.; Calas, A.; Sobanska, S.; Guédron, S.; Barraza, F.; Point, D.; Huayta, C.; Couture, R.-M.; et al. Is Tillandsia capillaris an efficient bioindicator of atmospheric metal and metalloid deposition? Insights from five months of monitoring in an urban mining area. Ecol. Indic. 2016, 67, 227–237. [Google Scholar] [CrossRef]
- Tian, H.; Zhao, Y.; Megharaj, M.; He, W. Arsenate inhibition on kinetic characteristics of alkaline phosphatase as influenced by pH. Ecol. Indic. 2018, 85, 1101–1106. [Google Scholar] [CrossRef]
- Skoczko, I.; Szatylowicz, E. Removal of heavy metal ions by filtration on activated alumina-assisted magnetic field. Desalination Water Treat. 2018, 117, 345–352. [Google Scholar] [CrossRef]
- Szatyłowicz, E.; Skoczko, I. Studies on the Efficiency of Grundwater Treatment Process with Adsorption on Activated Alumina. J. Ecol. Eng. 2017, 18, 211–218. [Google Scholar] [CrossRef]
- Alexakis, D.; Gamvroula, D. Arsenic, Chromium, and Other Potentially Toxic Elements in the Rocks and Sediments of Oropos-Kalamos Basin, Attica, Greece. Appl. Environ. Soil Sci. 2014, 2014, 718534. [Google Scholar] [CrossRef][Green Version]
- Alexakis, D.E. Contaminated Land by Wildfire Effect on Ultramafic Soil and Associated Human Health and Ecological Risk. Land 2020, 9, 409. [Google Scholar] [CrossRef]
- Alexakis, D.E. Suburban areas in flames: Dispersion of potentially toxic elements from burned vegetation and buildings. Estimation of the associated ecological and human health risk. Environ. Res. 2020, 183, 109153. [Google Scholar] [CrossRef] [PubMed]
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behavior and distribution of arsenic in natural waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- Ryu, J.-H.; Gao, S.; Dahlgren, R.A.; Zierenberg, R.A. Arsenic distribution, speciation and solubility in shallow groundwater of Owens Dry Lake, California. Geochim. Cosmochim. Acta 2002, 66, 2981–2994. [Google Scholar] [CrossRef]
- Ramos, O.E.R.; Cáceres, L.F.; Muñoz, M.R.O.; Bhattacharya, P.; Quino, I.; Quintanilla, J.; Sracek, O.; Thunvik, R.; Bundschuh, J.; García, M.E. Sources and behavior of arsenic and trace elements in groundwater and surface water in the Poopó Lake Basin, Bolivian Altiplano. Environ. Earth Sci. 2011, 66, 793–807. [Google Scholar] [CrossRef]
- Ryu, J.-H.; Gao, S.; Tanji, K.K. Speciation and behavior of arsenic in evaporation basins, California, USA. Environ. Earth Sci. 2010, 61, 1599–1612. [Google Scholar] [CrossRef]
- Barbiero, L.; Furquím, S.A.C.; Valles, V.; Furian, S.; Sakamoto, A.; Rezende, F. Natural arsenic in groundwater and alkaline lakes at the upper Paraguay basin, Pantanal, Brazil. Elsevier Book Series Trace Metals and Other Contaminants in the Environment. Geoderma 2007, 9, 101–126. [Google Scholar]
- Gao, S.; Ryu, J.; Tanji, K.; Herbel, M. Arsenic speciation and accumulation in evapoconcentrating waters of agricultural evaporation basins. Chemosphere 2007, 67, 862–871. [Google Scholar] [CrossRef] [PubMed]
- Colman, S.M.; Yu, S.Y.; An, Z.; Shen, J.; Henderson, A.C.G. Late Cenozoic climate changes in China’s western interior: A review of research on Lake Qinghai and comparison with other records. Quat. Sci. Rev. 2007, 26, 2281–2300. [Google Scholar] [CrossRef]
- Wang, S.; Cao, X.; Lin, C.; Chen, X. Arsenic content and fractionation in the surface sediments of the Guangzhou section of the Pearl River in Southern China. J. Hazard. Mater. 2010, 183, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Lin, C.Y.; Cao, X.Z.; Zhong, X. Arsenic content, fractionation, and ecological risk in the surface sediments of lake. Int. J. Environ. Sci. Technol. 2011, 9, 31–40. [Google Scholar] [CrossRef][Green Version]
- Lin, C.; He, M.; Zhou, Y.; Guo, W.; Yang, Z. Distribution and contamination assessment of heavy metals in sediment of the Second Songhua River, China. Environ. Monit. Assess. 2007, 137, 329–342. [Google Scholar] [CrossRef]
- Tian, Y.; Yu, C.; Zha, X.; Wu, J.; Gao, X.; Feng, C.; Luo, K. Distribution and Potential Health Risks of Arsenic, Selenium, and Fluorine in Natural Waters in Tibet, China. Water 2016, 8, 568. [Google Scholar] [CrossRef]
- Lanzhou Institute of Geology, CAS. Report on Integrated Survey of Qinghai Lake; Science Press: Beijing, China, 1979; pp. 1–264. (In Chinese) [Google Scholar]
- Wedepohl, K.H. The Composition of Continental Crust. Geochim. Cosmochim. Acta 1995, 7, 1217–1232. [Google Scholar] [CrossRef]
- Wan, D.; Jin, Z.; Wang, Y. Geochemistry of eolian dust and its elemental contribution to Lake Qinghai sediment. Appl. Geochem. 2012, 27, 1546–1555. [Google Scholar] [CrossRef]
- Wang, P.; Cao, J.; Han, Y.; Jin, Z.; Wu, F.; Zhang, F. Elemental distribution in the topsoil of the Lake Qinghai catchment, NE Tibetan Plateau, and the implications for weathering in semi-arid areas. J. Geochem. Explor. 2015, 152, 1–9. [Google Scholar] [CrossRef]
- Che, F.-F.; Wang, D.-P.; Zhen, Z.; Yan, C.-Z.; Wang, Z.-S. Distributions of Arsenic Species in Different Eutrophic Waters of Lake Taihu and Their Relations to Environmental Factors. Environ. Sci. 2016, 37, 3340–3347, (In Chinese with English Abstract). [Google Scholar]
- Lu, C.J.; Guo, N.; Zhang, J.; Dong, J.; He, Z.F.; Yin, S.H. Speciation distribution characteristics and health risk assessment of arsenic and mercury concentrations in water of Dongping Lake. J. Environ. Health. 2017, 34, 423–426, (In Chinese with English Abstract). [Google Scholar]
- Romero, L.; Alonso, H.; Campano, P.; Fanfani, L.; Cidu, R.; Dadea, C.; Keegan, T.; Thornton, I.; Farago, M. Arsenic enrichment in waters and sediments of the Rio Loa (Second Region, Chile). Appl. Geochem. 2003, 18, 1399–1416. [Google Scholar] [CrossRef]
- Welch, A.H.; Lico, M.S. Factors controlling As and U in shallow ground water, southern Carson Desert, Nevada. Appl. Geochem. 1998, 13, 521–539. [Google Scholar] [CrossRef]
- Peterson, M.; Carpenter, R. Arsenic distributions in porewaters and sediments of Puget Sound, Lake Washington, the Washington coast and Saanich Inlet, BC. Geochim. Cosmochim. Acta 1986, 50, 353–369. [Google Scholar] [CrossRef]
- Azcue, J.M.; Nriagu, J.O. Impact of abandoned mine tailings on the arsenic concentrations in Moira Lake, Ontario. J. Geochem. Explor. 1995, 52, 81–89. [Google Scholar] [CrossRef]
- Cheng, H.; Hu, Y.; Luo, J.; Xu, B.; Zhao, J. Geochemical processes controlling fate and transport of arsenic in acid mine drainage (AMD) and natural systems. J. Hazard. Mater. 2009, 165, 13–26. [Google Scholar] [CrossRef]
- Liu, Y.; Mu, S.; Bao, A.; Zhang, D.; Pan, X. Effects of salinity and (an)ions on arsenic behavior in sediment of Bosten Lake, Northwest China. Environ. Earth Sci. 2014, 73, 4707–4716. [Google Scholar] [CrossRef]
- Kitts, H.; Millward, G.; Morris, A.; Ebdon, L. Arsenic Biogeochemistry in the Humber Estuary, UK. Estuar. Coast. Shelf Sci. 1994, 39, 157–172. [Google Scholar] [CrossRef]
- Guo, H.; Zhang, B.; Li, Y.; Berner, Z.; Tang, X.; Norra, S.; Stüben, D. Hydrogeological and biogeochemical constrains of arsenic mobilization in shallow aquifers from the Hetao basin, Inner Mongolia. Environ. Pollut. 2011, 159, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Kwon, H.-O.; Choi, S.-D.; Lee, J.-S.; Khim, J.S. Arsenic speciation in water, suspended particles, and coastal organisms from the Taehwa River Estuary of South Korea. Mar. Pollut. Bull. 2016, 108, 155–162. [Google Scholar] [CrossRef]
- Zhang, L.C.; Zhou, K.Z. Background values of trace elements in the source area of the Yangtze River. Sci. Total Environ. 1992, 125, 391–404. [Google Scholar]
- Zan, F.; Huo, S.; Zhang, J.; Zhang, L.; Xi, B.; Zhang, L. Arsenic fractionation and contamination assessment in sediments of thirteen lakes from the East Plain and Yungui Plateau Ecoregions, China. J. Environ. Sci. 2014, 26, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Shotyk, W.; Weiss, D.; Appleby, P.G.; Cheburkin, A.K.; Frei, R.; Gloor, M.; Kramers, J.D.; Reese, S.; Van Der Knaap, W.O. History of Atmospheric Lead Deposition Since 12,370 14C yr BP from a Peat Bog, Jura Mountains, Switzerland. Science 1998, 281, 1635–1640. [Google Scholar] [CrossRef] [PubMed]

| K | Na | Ca | Mg | S | Cl | Al | Mn | Fe | Sc | As | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Detection limits a | 0.05 | 0.84 | 0.2 | 0.15 | 0.18 | 0.61 | 2.99 | 0.29 | 0.09 | 0.17 | 0.01 |
| CV for the water (%) b | 2.98 | 1.02 | 1.3 | 1.51 | 1.15 | 4.97 | 2.2 | ||||
| CV for soil/sediment (%) c | 2.36 | 3.06 | 1.7 | 2.11 | 3.48 | 1.64 | 1.78 | 1.6 | 1.96 | 2.2 | |
| Relative errors (%) d | 5.1 | 1.8 | 4.4 | 3.5 | 0.2 | 0.5 | 7.7 | 2.3 | 3.4 | 2.2 |
| Unit | Mean | Median | SD | CV% | Max | Min | n | ||
|---|---|---|---|---|---|---|---|---|---|
| River | pH | 8.04 | 8.08 | 0.27 | 3.3 | 8.38 | 7.45 | 12 | |
| water | Alkalinity | mmol L−1 | 2.72 | 2.86 | 0.48 | 17.8 | 3.55 | 1.86 | 12 |
| K | mg L−1 | 2.00 | 1.81 | 1.00 | 50.0 | 3.93 | 0.69 | 12 | |
| Na | mg L−1 | 14.28 | 13.98 | 8.95 | 62.7 | 28.97 | 2.60 | 12 | |
| Ca | mg L−1 | 65.15 | 62.51 | 23.92 | 36.7 | 106.24 | 32.63 | 12 | |
| Mg | mg L−1 | 13.56 | 13.39 | 4.78 | 35.3 | 19.55 | 6.74 | 12 | |
| Cl− | mg L−1 | 12.42 | 12.53 | 7.29 | 58.7 | 25.49 | 2.03 | 12 | |
| SO42− | mg L−1 | 24.63 | 23.95 | 6.95 | 28.2 | 35.70 | 11.27 | 12 | |
| As | μg L−1 | 1.39 | 1.36 | 0.17 | 12.0 | 1.74 | 1.17 | 12 | |
| As+5 | μg L−1 | 1.04 | 1.05 | 0.07 | 7.1 | 1.13 | 0.84 | 12 | |
| As+3 | μg L−1 | 0.35 | 0.33 | 0.16 | 45.2 | 0.69 | 0.13 | 12 | |
| Lake water | pH | 9.23 | 9.22 | 0.08 | 0.9 | 9.44 | 9.12 | 69 | |
| Alkalinity | mmol L−1 | 22.87 | 22.50 | 3.02 | 13.2 | 29.69 | 15.23 | 69 | |
| K | mg L−1 | 162.29 | 159.95 | 60.96 | 37.6 | 297.90 | 42.57 | 69 | |
| Na | mg L−1 | 3310.80 | 3327.70 | 1330.22 | 40.2 | 6182.55 | 707.20 | 69 | |
| Ca | mg L−1 | 8.97 | 8.43 | 3.29 | 36.7 | 18.44 | 1.57 | 69 | |
| Mg | mg L−1 | 831.21 | 790.07 | 321.14 | 38.6 | 1637.94 | 109.34 | 69 | |
| Cl− | mg L−1 | 5772.26 | 5610.97 | 2358.29 | 40.9 | 11,030.03 | 1236.86 | 69 | |
| SO42− | mg L−1 | 1661.17 | 1560.45 | 638.60 | 38.4 | 3196.95 | 354.71 | 69 | |
| As | μg L−1 | 25.55 | 25.69 | 8.67 | 33.9 | 41.76 | 8.86 | 69 | |
| As+5 | μg L−1 | 18.56 | 17.98 | 7.18 | 38.7 | 31.43 | 5.70 | 69 | |
| As+3 | μg L−1 | 6.99 | 6.99 | 2.48 | 35.5 | 12.81 | 2.06 | 69 | |
| Pore water | K | mg L−1 | 321.47 | 347.47 | 65.06 | 20.2 | 432.54 | 206.16 | 20 |
| Na | mg L−1 | 5225.45 | 5800.23 | 878.68 | 16.8 | 6034.66 | 3429.23 | 20 | |
| Ca | mg L−1 | 34.34 | 29.13 | 19.19 | 55.9 | 90.38 | 11.90 | 20 | |
| Mg | mg L−1 | 1669.14 | 1778.50 | 291.42 | 17.5 | 2161.22 | 1137.85 | 20 | |
| Cl− | mg L−1 | 10,050.58 | 11,067.97 | 1673.77 | 16.7 | 11,522.44 | 6658.39 | 20 | |
| SO42− | mg L−1 | 3556.06 | 3698.14 | 731.15 | 20.6 | 5022.34 | 2251.91 | 20 | |
| As | μg L−1 | 107.80 | 96.63 | 35.33 | 32.8 | 197.50 | 58.50 | 20 | |
| Sediment | pH | 9.39 | 9.42 | 0.4 | 4.2 | 10.03 | 8.86 | 22 | |
| OM | % | 10.25 | 10.16 | 2.39 | 23.3 | 15.59 | 3.03 | 22 | |
| Carbonate | % | 33.76 | 34.15 | 6.67 | 19.8 | 49.88 | 16.37 | 22 | |
| Mn | mg kg−1 | 490.03 | 509.45 | 69.38 | 14.2 | 589.27 | 253.49 | 22 | |
| Al2O3 | % | 8.96 | 9.03 | 1.04 | 11.6 | 11.29 | 5.67 | 22 | |
| Fe2O3 | % | 3.25 | 3.37 | 0.56 | 17.2 | 4.19 | 1.43 | 22 | |
| MgO | % | 4.56 | 3.61 | 2.57 | 56.4 | 14.59 | 2.66 | 22 | |
| CaO | % | 18.27 | 18.41 | 2.90 | 15.9 | 22.86 | 11.65 | 22 | |
| Na2O | % | 1.64 | 1.61 | 0.24 | 14.4 | 2.52 | 1.36 | 22 | |
| K2O | % | 1.91 | 1.93 | 0.25 | 13.1 | 2.42 | 1.23 | 22 | |
| As | mg kg−1 | 16.75 | 16.20 | 2.76 | 16.5 | 21.93 | 12.40 | 22 | |
| Soil | pH | 8.83 | 8.76 | 0.55 | 6.3 | 10.39 | 7.95 | 45 | |
| OM | % | 7.48 | 7.97 | 3.33 | 44.5 | 13.24 | 1.21 | 45 | |
| Mn | mg kg−1 | 584.09 | 596.68 | 87.77 | 15.0 | 752.97 | 394.15 | 45 | |
| Al2O3 | % | 10.90 | 10.89 | 1.25 | 11.5 | 13.51 | 7.77 | 45 | |
| Fe2O3 | % | 4.20 | 4.28 | 0.60 | 14.3 | 5.32 | 2.86 | 45 | |
| MgO | % | 2.21 | 2.12 | 0.52 | 23.7 | 4.76 | 1.50 | 45 | |
| CaO | % | 8.26 | 8.32 | 2.74 | 33.2 | 13.55 | 2.15 | 45 | |
| Na2O | % | 1.74 | 1.73 | 0.15 | 8.5 | 2.08 | 1.42 | 45 | |
| K2O | % | 2.15 | 2.13 | 0.20 | 9.4 | 2.54 | 1.61 | 45 | |
| As | mg kg−1 | 13.14 | 13.65 | 2.41 | 18.3 | 17.19 | 7.25 | 45 |
| As | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| River Water, n = 12 | Lake Water, n = 69 | Pore Water, n = 20 | Sediment, n = 22 | Soil, n = 45 | ||||||
| R2 | p | R2 | p | R2 | p | R2 | p | R2 | p | |
| V | 0.15 | 0.63 | 0.96 | <0.0001 | 0.23 | 0.37 | −0.02 | 0.94 | 0.87 | <0.0001 |
| Sc | −0.23 | 0.47 | 0.61 | <0.0001 | −0.30 | 0.22 | 0.04 | 0.86 | 0.89 | <0.0001 |
| B | 0.77 | 0.00 | 0.93 | <0.0001 | 0.58 | 0.01 | 0.27 | 0.22 | 0.31 | 0.04 |
| P | 0.39 | 0.21 | 0.84 | <0.0001 | −0.14 | 0.57 | −0.32 | 0.14 | 0.49 | 0.00 |
| S | −0.05 | 0.88 | 0.81 | <0.0001 | 0.16 | 0.52 | −0.66 | 0.00 | 0.29 | 0.05 |
| Cl | 0.52 | 0.08 | 0.87 | <0.0001 | 0.41 | 0.09 | ||||
| Mn | 0.50 | 0.10 | 0.53 | <0.0001 | −0.04 | 0.88 | 0.09 | 0.68 | 0.81 | <0.0001 |
| Al | −0.45 | 0.14 | −0.13 | 0.29 | 0.24 | 0.36 | 0.26 | 0.24 | 0.87 | <0.0001 |
| Fe | 0.46 | 0.13 | 0.59 | <0.0001 | 0.10 | 0.69 | 0.03 | 0.88 | 0.87 | <0.0001 |
| Mg | 0.44 | 0.15 | 0.74 | <0.0001 | 0.46 | 0.06 | 0.26 | 0.25 | 0.46 | 0.00 |
| Ca | 0.16 | 0.62 | 0.68 | <0.0001 | −0.37 | 0.17 | −0.58 | 0.00 | 0.07 | 0.65 |
| Na | 0.59 | 0.05 | 0.88 | <0.0001 | 0.35 | 0.15 | 0.21 | 0.36 | −0.45 | 0.00 |
| K | 0.60 | 0.04 | 0.85 | <0.0001 | 0.45 | 0.06 | 0.07 | 0.75 | 0.80 | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, Y.; Lin, C.; Zhang, X. Distribution and Geochemical Processes of Arsenic in Lake Qinghai Basin, China. Water 2021, 13, 1091. https://doi.org/10.3390/w13081091
Cao Y, Lin C, Zhang X. Distribution and Geochemical Processes of Arsenic in Lake Qinghai Basin, China. Water. 2021; 13(8):1091. https://doi.org/10.3390/w13081091
Chicago/Turabian StyleCao, Yuanxin, Chunye Lin, and Xuan Zhang. 2021. "Distribution and Geochemical Processes of Arsenic in Lake Qinghai Basin, China" Water 13, no. 8: 1091. https://doi.org/10.3390/w13081091
APA StyleCao, Y., Lin, C., & Zhang, X. (2021). Distribution and Geochemical Processes of Arsenic in Lake Qinghai Basin, China. Water, 13(8), 1091. https://doi.org/10.3390/w13081091





