Mediterranean and Black Sea Monstrilloid Copepods (Copepoda: Monstrilloida): Rediscovering the Diversity of Transient Zooplankters
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Species Diversity of the Monstrilloida
3.2. Mediterranean-Black Sea Diversity of the Monstrilloida
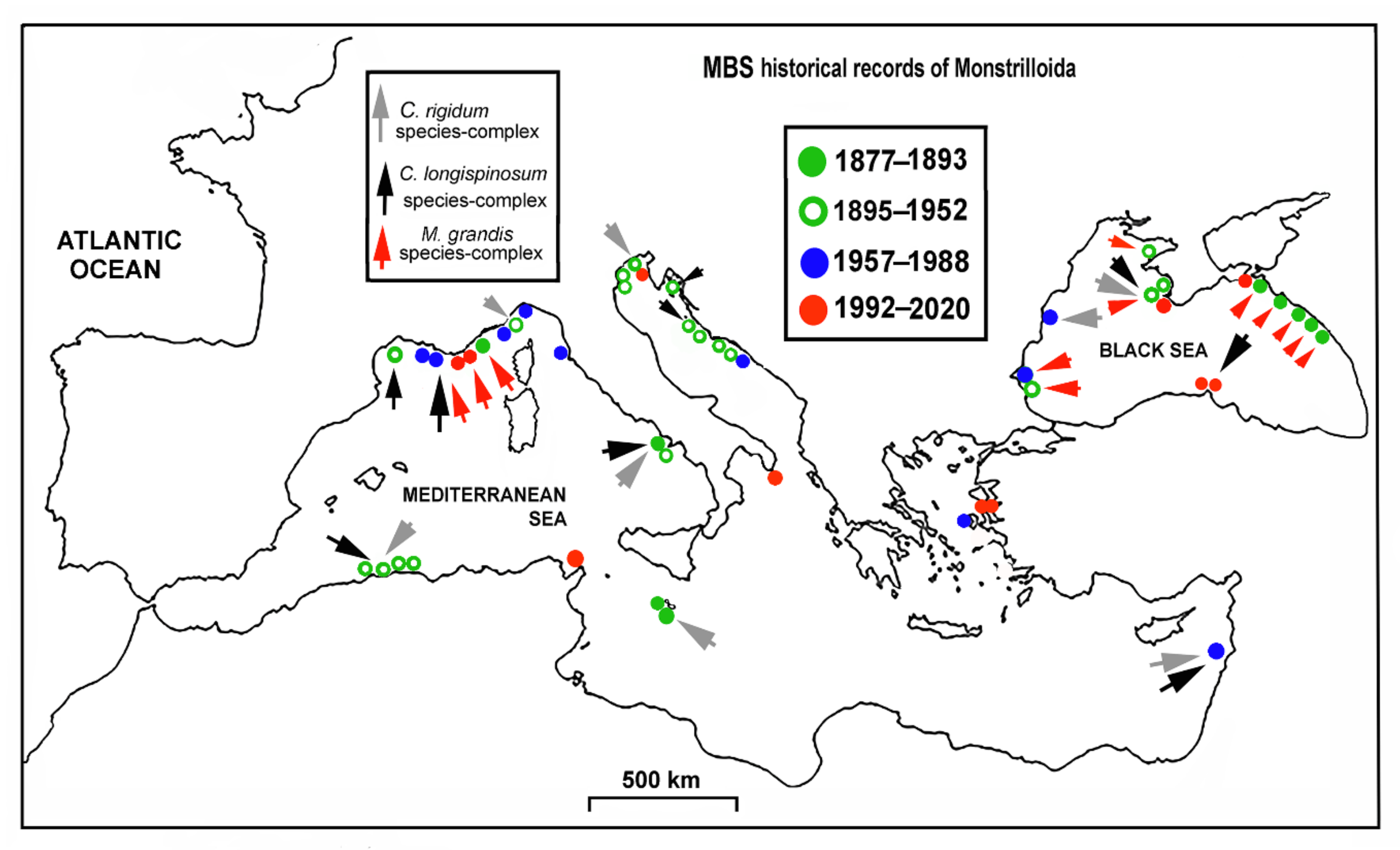
3.3. MBS Monstrilloid Diversity, Historical Account
3.4. Analysis of Selected Species-Groups
3.4.1. Cymbasoma longispinosum Species-Group
3.4.2. Cymbasoma rigidum Species-Group
3.4.3. Monstrilla grandis Species-Group
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khodami, S.; McArthur, J.V.; Blanco-Bercial, L.; Martinez Arbizu, P. Molecular phylogeny and revision of copepod orders (Crustacea: Copepoda). Sci. Rep. 2017, 7, 9164. [Google Scholar] [CrossRef]
- Boxshall, G.A.; Halsey, S.H. An Introduction to Copepod Diversity; The Ray Society: London, UK, 2004; Part II; pp. 422–966. [Google Scholar]
- Belmonte, G. Calanoida (Crustacea: Copepoda) of the Italian fauna: A review. Eur. Zool. J. 2018, 85, 273–289. [Google Scholar] [CrossRef]
- Razouls, C. Diversité et répartition géographique chez les copépodes pélagiques. 2. Platycopioida, Misophrioida, Mormonilloida, Cyclopoida, Poecilostomatoida, Siphonostomatoida, Harpacticoida, Monstrilloida. Ann. Inst. Océanogr. N. Sér. 1996, 72, 1–149. [Google Scholar]
- Razouls, C.; Desreumaux, N.; Kouwenberg, J.; de Bovée, F. Biodiversity of Marine Planktonic Copepods (Morphology, Geographical Distribution and Biological Data); Sorbonne Univ.-CNRS: Paris, France, 2005–2021; Available online: http://copepodes.obs-banyuls.fr/en (accessed on 15 February 2021).
- Suárez-Morales, E. An aggregation of monstrilloid copepods in a western Caribbean reef area: Ecological and conceptual implications. Crustaceana 2001, 74, 689–696. [Google Scholar] [CrossRef]
- Suárez-Morales, E. Diversity of the Monstrilloida (Crustacea: Copepoda). PLoS ONE 2011, 6, e22915. [Google Scholar] [CrossRef] [PubMed]
- Suárez-Morales, E. Crustacea Orden Monstrilloida Revista IDE@ (Ibero-Diversidad Entomológica @ccesible). Rev. IDE@ (Ibero-Diversidad Entomológica @Ccesible). SEA Soc. Ent. Arag. 2015, 20, 1–12. [Google Scholar]
- Walter, T.C.; Boxshall, G.A. World of Copepods Database. Monstrilloida. Accessed 2021-02-25 through: World Register of Marine Species 2021. Available online: http://marinespecies.org/aphia.php?p=taxdetails&id=1106 (accessed on 11 February 2021).
- Grygier, M.J.; Ohtsuka, S. A new genus of monstrilloid copepods (Crustacea) with anteriorly pointing ovigerous spines and related adaptations for subthoracic brooding. Zool. J. Linn. Soc. 2008, 152, 459–506. [Google Scholar] [CrossRef]
- Suárez-Morales, E. A new genus of the Monstrilloida (Copepoda) with large rostral process and metasomal spines, and redescription of Monstrilla spinosa Park, 1967. Crustaceana 2019, 92, 1099–1112. [Google Scholar] [CrossRef]
- Jeon, D.; Lee, W.; Soh, H.Y. New genus and two new species of monstrilloid copepods (Copepoda: Monstrillidae): Integrating morphological, molecular, phylogenetic, and ecological evidence. J. Crustac. Biol. 2018, 38, 45–65. [Google Scholar] [CrossRef]
- Jeon, D.; Lim, D.; Lee, W.; Soh, H.Y. First use of molecular evidence to match sexes in the Monstrilloida (Crustacea: Copepoda), and taxonomic implications of the newly recognized and described, partly Maemonstrilla-like females of Monstrillopsis longilobata Lee, Kim & Chang, 2016. Peer J. 2018, 6, e4938. [Google Scholar]
- Grygier, M.J. Annotated chronological bibliography of Monstrilloida (Crustacea: Copepoda). Galaxea 1995, 12, 1–82. [Google Scholar]
- Suárez-Morales, E.; McKinnon, A.D. The Australian Monstrilloida (Crustacea: Copepoda) I. Monstrillopsis Sars, Maemonstrilla Grygier & Ohtsuka, and Australomonstrillopsis gen. nov. Zootaxa 2014, 3779, 301–340. [Google Scholar] [PubMed]
- Suárez-Morales, E.; McKinnon, A.D. The Australian Monstrilloida (Crustacea: Copepoda) II. Cymbasoma Thompson, 1888. Zootaxa Monogr. 2016, 4102, 1–129. [Google Scholar] [CrossRef]
- Chang, C.Y. Two new records of monstrilloid copepods (Crustacea) from Korea. Anim. Syst. Evol. Div. 2014, 30, 206–214. [Google Scholar] [CrossRef]
- Lee, J.; Chang, C.Y. A new species of Monstrilla Dana, 1849 (Copepoda: Monstrilloida: Monstrillidae) from Korea, including a key to species from the northwest Pacific. Zootaxa 2016, 4174, 396–409. [Google Scholar] [CrossRef]
- Lee, J.; Kim, D.; Chang, C.Y. Two new species of the genus Monstrillopsis Sars, 1921 (Copepoda: Monstrilloida: Monstrillidae) from South Korea. Zootaxa 2016, 4174, 410–423. [Google Scholar] [CrossRef]
- Jeon, D.; Lee, W.; Soh, H.Y.; Eyun, S.-I. A new species of Monstrillopsis Sars, 1921 (Copepoda: Monstrilloida) with an unusually reduced urosome. Diversity 2020, 19, 9. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Riccardi, N. Redescription and first record of Cymbasoma tenue (Isaac, 1975) (Copepoda: Monstrilloida) in the Mediterranean Sea. Proc. Biol. Soc. Wash. 1997, 110, 99–106. [Google Scholar]
- Suárez-Morales, E. Redescription of the male of Cymbasoma tumorifrons (Isaac, 1975) from the Mediterranean Sea (Copepoda: Monstrilloida). Arthrop. Sel. 1999, 8, 67–71. [Google Scholar]
- Suárez-Morales, E. Taxonomic report on some monstrilloids (Copepoda, Monstrilloida) from Toulon Bay, France. Bull. Inst. Royal Sci. Nat. Belg. Biol. 2000, 70, 107–118. [Google Scholar]
- Suárez-Morales, E. A new species of Cymbasoma (Copepoda: Monstrilloida) from the Mediterranean Sea with remarks on the female of C. tumorifrons (Isaac). Mitt. Mus. Naturk. Berlin Zool. Reihe 2002, 78, 87–96. [Google Scholar] [CrossRef]
- Suárez-Morales, E. Validation and redescription of Cymbasoma germanicum (Timm) (Crustacea: Copepoda: Monstrilloida) from Helgoland with comments on C. rigidum Thompson. Helgol. Mar. Res. 2006, 60, 171–179. [Google Scholar] [CrossRef]
- Suárez-Morales, E. On the taxonomic status of Monstrilla leucopis Sars (Crustacea: Copepoda: Monstrilloida) from Norway, with comments on the male of M. longiz Giesbrecht. Zootaxa 2010, 2510, 55–67. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Bello-Smith, A.; Palma, S. A revision of the genus Monstrillopsis Sars (Crustacea: Copepoda: Monstrilloida) with description of a new species from Chile. Zool. Anz. 2006, 245, 95–107. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Goruppi, A.; de Olazabal, A.; Tirelli, V. Monstrilloids (Crustacea: Copepoda) from the Mediterranean Sea (Northern Adriatic Sea), with a description of six new species. J. Nat. Hist. 2017, 51, 1795–1834. [Google Scholar] [CrossRef]
- Rose, M. Copépodes pélagiques. Faune Fr. 1933, 26, 1–374. [Google Scholar]
- Rose, M.; Vaissière, R. Catalogue préliminaire des Copépodes de l’Afrique du Nord. Bull. Soc. Hist. Nat. Afrique Nord 1952, 43, 113–136. [Google Scholar]
- Trégouboff, G.; Rose, M. Manuel de Planctonologie Méditerranéenne; Centre National de la Recherche Scientifique: Paris, France, 1957; Volumes I–II, pp. 1–590.
- Grygier, M.J. Identity of Thaumatoessa (Thaumaleus) typica Krøyer, the first described monstrilloid copepod. Sarsia 1993, 78, 235–242. [Google Scholar] [CrossRef]
- Grygier, M.J. Nomenclature, redescription and new record of Cymbasoma morii Sekiguchi, 1982 (Monstrilloida). Hydrobiologia 1994, 292, 23–29. [Google Scholar] [CrossRef]
- Dolgopolskaya, M.A. K faune Monstrillidae Chernogo morya. Trud. Zool. Inst. Akad. Nauk SSSR. 1948, 7, 177–183. (In Russian) [Google Scholar]
- Suárez-Morales, E.; Üstün, F. Report on some monstrilloids (Crustacea: Copepoda: Monstrilloida) from Turkey, with description of two new species. Cah. Biol. Mar. 2018, 59, 547–562. [Google Scholar]
- Razouls, C.; Durand, J. Inventaire des copépodes planctoniques Méditerranéens. Vie Milieu 1991, 41, 73–77. [Google Scholar]
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Lasram, F.B.R.; Aguzzi, J.; Ballesteros, E.; Bianchi, C.N.; Corbera, J.; Dailianis, T.; et al. The biodiversity of the Mediterranean Sea: Estimates, patterns, and threats. PLoS ONE 2010, 5, e11842. [Google Scholar] [CrossRef] [PubMed]
- Gabrié, J.; Marbà, N.; Duarte, C.M. Mediterranean seagrass vulnerable to regional climate warming. Nat. Clim. Chang. 2012, 2, 821–824. [Google Scholar] [CrossRef]
- Kriczagin, N.A. Account of an excursion to the NW coast of the Black Sea, carried out as a mission of the Kiev Society of Naturalists in the summer of 1874. Zap. Obshch. Estestvoispyt. 1877, 5, 1–56. (In Russian) [Google Scholar]
- Melville, R.V. Monstrilla intermedia Kriczagin, 1877 (Copepoda) suppressed. Bull. Zool. Nomencl. 1981, 38, 107–109. [Google Scholar]
- Isaac, M.J. Copepoda, Suborder: Monstrilloida. Fich. Ident. Zooplankt. 1975, 144–145, 1–10. [Google Scholar]
- Thompson, I.C. Second report on the Copepoda of Liverpool Bay. Proc. Liverpool Biol. Soc. 1888, 2, 63–71, pls. I-II. [Google Scholar]
- Thompson, I.C. Copepoda of Madeira and the Canary Islands, with descriptions of new genera and species. J. Linn. Soc. London Zool. 1888, 20, 145–156. [Google Scholar] [CrossRef]
- Bourne, G.C. Notes on the genus Monstrilla Dana. Quart. J. Microscop. Sci. 1890, 30, 565–578. [Google Scholar]
- Thompson, I.C. Monstrilla and the Cymbasomatidae. Proc. Trans. Liverpool Biol. Soc. 1890, 4, 115–124. [Google Scholar]
- Giesbrecht, W. Systematik und Faunistik der pelagischen Copepoden des Golfes von Neapel und der angrenzenden Meeres-Abschnitte. Fauna Flora Golfes Neapel Monogr. 1893, 19, 1–831, 54 plates. [Google Scholar]
- Karavayev, V. Material towards the fauna of copepods (Copepoda) of the Black Sea. Zap. Kiyevsk. Obshch. Estestvoispyt. 1895, 14, 117–174. (In Russian) [Google Scholar]
- Porumb, F.I. Contributii la cunoasterea familiei Monstrillidae din dreptul litoralului rominesc al Marii Negre. Com. Acad. Republ. Pop. Rom. 1961, 11, 1223–1231. [Google Scholar]
- Graeffe, E. Uebersicht der Fauna des Golfes von Triest nebst Notizen über Vorkommen, Lebenweise, Erscheinungs- und Laichzeit der einzelen Arten. V. Crustacea. Arb. Zool. Inst. Univ. Wien Zool. Stat. Triest 1900, 13, 33–80. [Google Scholar]
- Car, L. Planktonproben aus dem Adriatischen Meere und einigen süssen und brakischen Gewässern Dalmatiens. Zool. Anz. 1902, 25, 601–605. [Google Scholar]
- Grandori, R. I Copepodi. In Richerche sul plancton dell Laguna Veneta; Carazzi, D., Grandori, R., Eds.; University of Padova: Padova, Italy, 1912; pp. 1–40. [Google Scholar]
- Grandori, R. Studi biologici sul Plancton della Laguna Veneta e dell’alto Adriatico. Atti Acad. Sci. Ven. Trent. Istr. 1914, 3, 149–171. [Google Scholar]
- Pesta, O. Die Planctoncopepoden der Adria. Zool. Jahrb. 1920, 43, 471–659. [Google Scholar]
- Lo Bianco, S. Le pesche aissali eseguire de FA Krupp col yacht Puritan nelle adiacensze di Capri ed in altre localitá del Mediterraneo. Mitt. Zool. Stat. Neapel 1903, 16, 109–128. [Google Scholar]
- Chichkoff, G. Contribution a l’étude de la faune de la Mer Noire. Animaux récoltés sur les cotes bulgares. Arch. Zool. Expér. Gén. 1912, 10, 29–31. [Google Scholar]
- Rose, M. Notes faunistiques sur les Copépodes pélagiques des côtes de France. Bull. Soc. Zool. France 1926, 51, 134–138. [Google Scholar]
- Rose, M. Le plancton de la baie d’Alger pendant le mois de décembre (Suite). Bull. Soc. Hist. Nat. Afrique Nord 1926, 17, 44–48. [Google Scholar]
- Rose, M. Observations préliminaires sur le plankton de la région d’Alger. Bull. Trav. Sta. Aquicult. Pêche Castiglione 1927, 1, 129–164. [Google Scholar]
- Rose, M. Recherches preliminaires sur le Plankton de profundeur de la Baie d’Alger. Bull. Trav. Sta. Aquicult. Pêche Castiglione 1934, 1, 43–81. [Google Scholar]
- Rose, M. Recherches complémentaires sur les Copépodes Pélagiques de la Baie d’Alger. Bull. Soc. Hist. Nat. Afrique Nord 1934, 25, 145–148. [Google Scholar]
- Hure, J.; Kršinić, F. Planktonic copepods of the Adriatic Sea. Spatial and temporal distribution. Natur. Croat. 1998, 7, 1–135. [Google Scholar]
- Mužinić, S. Ekološka Ispitivanja na Jadranskoj Srdeli (Clupea pilchardus Walb.); Štamparija Dragomira Popovića: Belgrade, Yugoslavia, 1936; pp. 1–109. [Google Scholar]
- Gamulin, T. Qualitative and quantitative research of planktonic copepods in the eastern coastal waters of the central Adriatic over the years 1936/37. Yugosl. Acad. Sci. Arts 1939, 22, 97–180, (not seen). [Google Scholar]
- Mathias, P.; Euzet, L. Sur les Copépodes planctoniques de l’Étang de Thau. Bull. Soc. Hist. Nat. Toulouse 1951, 86, 184–188. [Google Scholar]
- Rusev, B.; Dimov, I. Qualitative and quantitative studies of the zooplankton of Varna Bay. Nauch.–Izsled. Inst. Ribarst. Rib. Promishl. 1957, 1, 79–109. [Google Scholar]
- Gaudy, R. Biologie des Copépodes pélagiques de Golfe de Marseille. Rec. Trav. Stat. Mar. d’Endoume 1962, 27, 93–184. [Google Scholar]
- Djordjevic, M. Observation sur les Copépodes pélagiques en rade de Villefranche de mars à aoȗt 1962. Comm. Int. Explor. Scient. Mer Méd. Monaco. Rapp. Procés-Verb. Réun. 1963, 17, 575–580. [Google Scholar]
- Buljan, M.; Hure, J.; Pucher-Petković, T. Hydrographic and productivity conditions in the Bay of Mali Ston. Report on the work made during the dam construction of the Trebišnjica Hydroelectric Power Plant. Acta Adriat. 1973, 15, 3–60. [Google Scholar]
- Basso, M.P.; Cevasco, M.G.; Della Croce, N.; Picone, P. Caratteristiche ecologiche e popolamento zoplanctonico in ambienti portuali del Mar Ligure a Alto Tirreno. Univ. Genova Catt. Idrobiol. Pescicolt. Fac. Sci. Fis. Mat. Natur. Rap. Tec. 1980, 12, 2–48. [Google Scholar]
- Lakkis, S. On the presence of some rare copepods in the Levantine Basin. Crustaceana Suppl. 1984, 7, 286–304. [Google Scholar]
- Citarella, G. Les Copépodes des eaux portuaires de Marseille (Méditerranée nord-occidentale). Syllogeus 1986, 58, 276–282. [Google Scholar]
- Isaac, M.J. Studies on Planktonic Arthropods. Ph.D. Thesis, University College of Swansea, University of Wales, Swansea, UK, 1974. [Google Scholar]
- Grygier, M.J.; Suárez-Morales, E. Recognition and partial solution of nomenclatural issues involving copepods of the family Monstrillidae (Crustacea: Copepoda: Monstrilloida). Zootaxa 2018, 4486, 497–509. [Google Scholar] [CrossRef]
- Razouls, C. Bilan Actuel des Copépodes Planctoniques Marins et des eaux Saumâtres. Corrections et Complements; Laboratoire Arago: Banyuls-sur-Mer, France, 1991; pp. 1–240. [Google Scholar]
- Annabi-Trabelsi, M.N.; Daly-Yahia, M.S.; Romdhane, A.; Ben Maïz, N. Seasonal variability of planktonic copepods in Tunis North Lagoon (Tunisia, North Africa). Cah. Biol. Mar. 2005, 46, 325–333. [Google Scholar]
- Onorato, M.; Belmonte, G. Submarine caves of the Salento Peninsula: Faunal aspects. Thal. Salent. 2017, 39, 47–72. [Google Scholar]
- Zaremba, N.B. Zooplankton of the south-eastern part of the Kerch Peninsula in the summer and autumn seasons of 2015. Trudy YUgNIRO 2017, 54, 77–81, (In Russian with English summary). [Google Scholar]
- Galagovets, E.A.; Prusova, I.Y. First observations of tumor-like anomalies in platonic copepods in the Crimean coastal waters. J. Siber. Fed. Univ. Biol. 2016, 4, 441–451. [Google Scholar] [CrossRef]
- Pulat, İ.; Özel, İ. The plankton fauna of the northern lagoonary system of Izmir Bay. EU J. Fish. Aquat. Sci. 2003, 20, 399–403. [Google Scholar]
- Gurkan, S.; Sever, T.M.; Taskavak, E. The biometric analysis of pipefish species from Çamalti Lagoon (İzmir Bay, Aegean Sea). EU J. Fish. Aquat. Sci. 2008, 25, 53–56. [Google Scholar]
- Üstün, F.; Terbiyik Kurt, T.; Suárez-Morales, E. A new species of Cymbasoma (Copepoda, Monstrilloida) from the northern coast of Turkey (Black Sea) with comments on the C. longispinosum species-group. Crustaceana 2014, 87, 1393–1410. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Üstün, F. Validation of Cymbasoma turcorum and Monstrillopsis pontoeuxinensis, two monstrilloids from Turkey described by Suárez-Morales & Üstün in 2018 (Crustacea: Copepoda: Monstrilloida). Zootaxa 2020, 4779, 149–150. [Google Scholar]
- Vives, F.; Shmeleva, A.A. Crustacea, Copépodos Marinos II. In Non Calanoida. Fauna Ibérica; CSIC: Madrid, Spain, 2010; Volume 33, pp. 1–486. [Google Scholar]
- Suárez-Morales, E.; Carrillo, A.; Morales-Ramírez, A. Report on some monstrilloids (Crustacea: Copepoda) from a reef area off the Caribbean coast of Costa Rica, Central America with description of two new species. J. Nat. Hist. 2013, 47, 619–638. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Ivanenko, V.N. Two new species of Monstrillopsis Sars (Copepoda: Monstrilloida) from the White Sea and Norway, with comments on M. dubia Scott. Arctic 2004, 57, 37–46. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Dias, C.O.; Costa-Bonecker, S.L. Discovery of the female Cymbasoma rochai Suárez-Morales & Dias, 2001 (Copepoda: Monstrilloida: Monstrillidae), the first Brazilian member of the C. longispinosum species-group. Crustaceana 2020, 93, 1091–1101. [Google Scholar]
- Suárez-Morales, E.; Vásquez-Yeomans, L.; Santoya, L. A new species of the Cymbasoma longispinosum species-group (Copepoda, Monstrilloida, Monstrillidae) from Belize, western Caribbean Sea. Rev. Mex. Biodiv. 2020, 91, 1–11. [Google Scholar] [CrossRef]
- Giesbrecht, W. Elenco dei Copepodi pelagici raccolti dal tenente di vascello Gaetano Chierchia durante el viaggio della R. Corvetta “Vettor Pisani” negli anni 1882-1885, e dal tenente di vascello Francesco Orsini nel Mar Rosso, nel 1884. Rend. Sed. R. Acad. Lincei 1891, 7, 474–481. [Google Scholar]
- Dias, C.O.; Bonecker, S.L.C. Study of Monstrilloida distribution (Crustacea, Copepoda) in the Southwest Atlantic. Pan-Amer. J. Aquat. Sci. 2007, 2, 270–278. [Google Scholar]
- Bonk, T.V.; Sushkevich, N.S.; Lozovoy, A.P. The first foundation the Monstrilloida (Copepoda) species in Okhotsk Sea near Kamchatka shore. In Conservation of biodiversity of Kamchatka and Coastal Waters: Materials of the XX International Scientific Conference, 150th Anniversary of V.L. Komarov’s Birthday; Tokranov, A.M., Ed.; Kamchatpress: Petropavlovsk-Kamchatski, Russia, 2019; pp. 157–159. (In Russian) [Google Scholar]
- Ramírez, F.C. Nuevas localidades para Monstrilla grandis Giesbrecht 1892 y Monstrilla helgolandica Claus 1863 (Copepoda, Monstrilloida) hallados en aguas de la plataforma Argentina. Physis Buenos Aires 1971, 30, 377–383. [Google Scholar]
| Phase 1: 1877–1893 | Phase 2: 1895–1952 | Phase 3: 1957–1986 | Phase 4: 1992–2020 (Current Checklist) |
|---|---|---|---|
| Cymbasoma claparedii C. herdmani | Cymbasoma claparedii | Cymbasoma longispinosum | Cymbasoma clauderazoulsi Suárez-Morales, Goruppi, de Olazabal and Tirelli, 2017 |
| C. longispinosum | C. longispinosum | C. rigidum | C. cf. longispinosum (Bourne, 1890) |
| C. reticulatum | C. rigidum | C. tenue | C. mediterraneum Suárez-Morales, Goruppi, de Olazabal and Tirelli, 2017 |
| C. rigidum | C. thompsonii | C. thompsonii | C. nicolettae Suárez-Morales, 2002 C. pseudobidentatum Suárez-Morales, Goruppi, de Olazabal and Tirelli |
| Monstrilla gracilicauda | Monstrilla gracilicauda | Monstrilla grandis | C. cf. rigidum Thompson, 1888 |
| M. intermedia (purported synonym of M. grandis; suppressed by ICZN) | M. grandis (original source of record unclear) | M. leucopis (likely erroneous) | C. reticulatum (Giesbrecht, 1893) |
| M. longissima (nomen nudum) | M. helgolandica | M. longicornis | C. sinopense Üstün, Terbiyik and Suárez-Morales, 1914 |
| M. longiremis | M. longicornis (original source of record unclear) | M. longiremis | C. specchii Suárez-Morales, Goruppi, de Olazabal and Tirelli, 2017 |
| M. pontica (insufficiently known) | M. longiremis | M. tumorifrons (nomen nudum; later made available) | C. tenue (Isaac, 1975) C. tergestinum Suárez-Morales, Goruppi, de Olazabal and Tirelli |
| M. ostroumowi (now in Cymbasoma; possibly a synonym of C. rigidum) | Monstrillopsis angustipes (nomen nudum) | C. tumorifrons Suárez-Morales, 1999 C. turcorum Suárez-Morales and Üstün, 2018 | |
| M. serricornis (supposed junior synonym of M. helgolandica; original source of record unclear) | Monstrilla ghirardellii Suárez-Morales, Goruppi, de Olazabal and Tirelli, 2017 | ||
| Monstrillopsis dubia (species complex; original source of record unclear) | M. grandis Giesbrecht, 1891 | ||
| Monstrillopsis zernowi (probably a Monstrilla) | M. longicornis Thompson, 1890 M. longiremis Giesbrecht, 1893 | ||
| Monstrillopsis pontoeuxinensis Suárez-Morales and Üstün, 2018 | |||
| Monstrillopsis zernowi Dolgopolskaya, 1948 | |||
| Caromiobenella cf. helgolandica (Claus, 1863) | |||
| Ca. pygmaea (Suárez-Morales, 2000) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suárez-Morales, E.; Grygier, M.J. Mediterranean and Black Sea Monstrilloid Copepods (Copepoda: Monstrilloida): Rediscovering the Diversity of Transient Zooplankters. Water 2021, 13, 1036. https://doi.org/10.3390/w13081036
Suárez-Morales E, Grygier MJ. Mediterranean and Black Sea Monstrilloid Copepods (Copepoda: Monstrilloida): Rediscovering the Diversity of Transient Zooplankters. Water. 2021; 13(8):1036. https://doi.org/10.3390/w13081036
Chicago/Turabian StyleSuárez-Morales, Eduardo, and Mark J. Grygier. 2021. "Mediterranean and Black Sea Monstrilloid Copepods (Copepoda: Monstrilloida): Rediscovering the Diversity of Transient Zooplankters" Water 13, no. 8: 1036. https://doi.org/10.3390/w13081036
APA StyleSuárez-Morales, E., & Grygier, M. J. (2021). Mediterranean and Black Sea Monstrilloid Copepods (Copepoda: Monstrilloida): Rediscovering the Diversity of Transient Zooplankters. Water, 13(8), 1036. https://doi.org/10.3390/w13081036





