Integration of Constructed Floodplain Ponds into Nature-Like Fish Passes Supports Fish Diversity in a Heavily Modified Water Body
Abstract
:1. Introduction
2. Materials and Methods
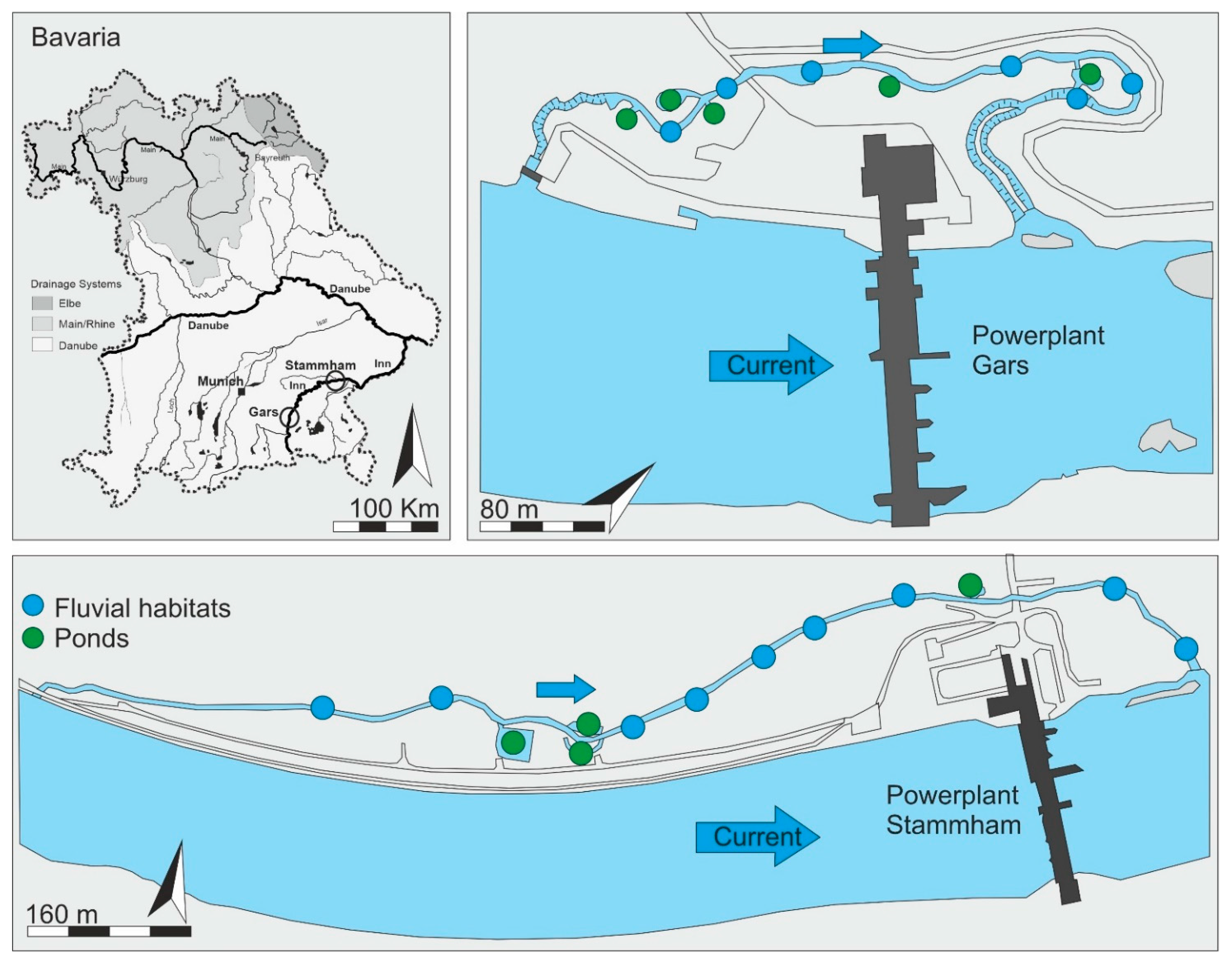
2.1. Study Area
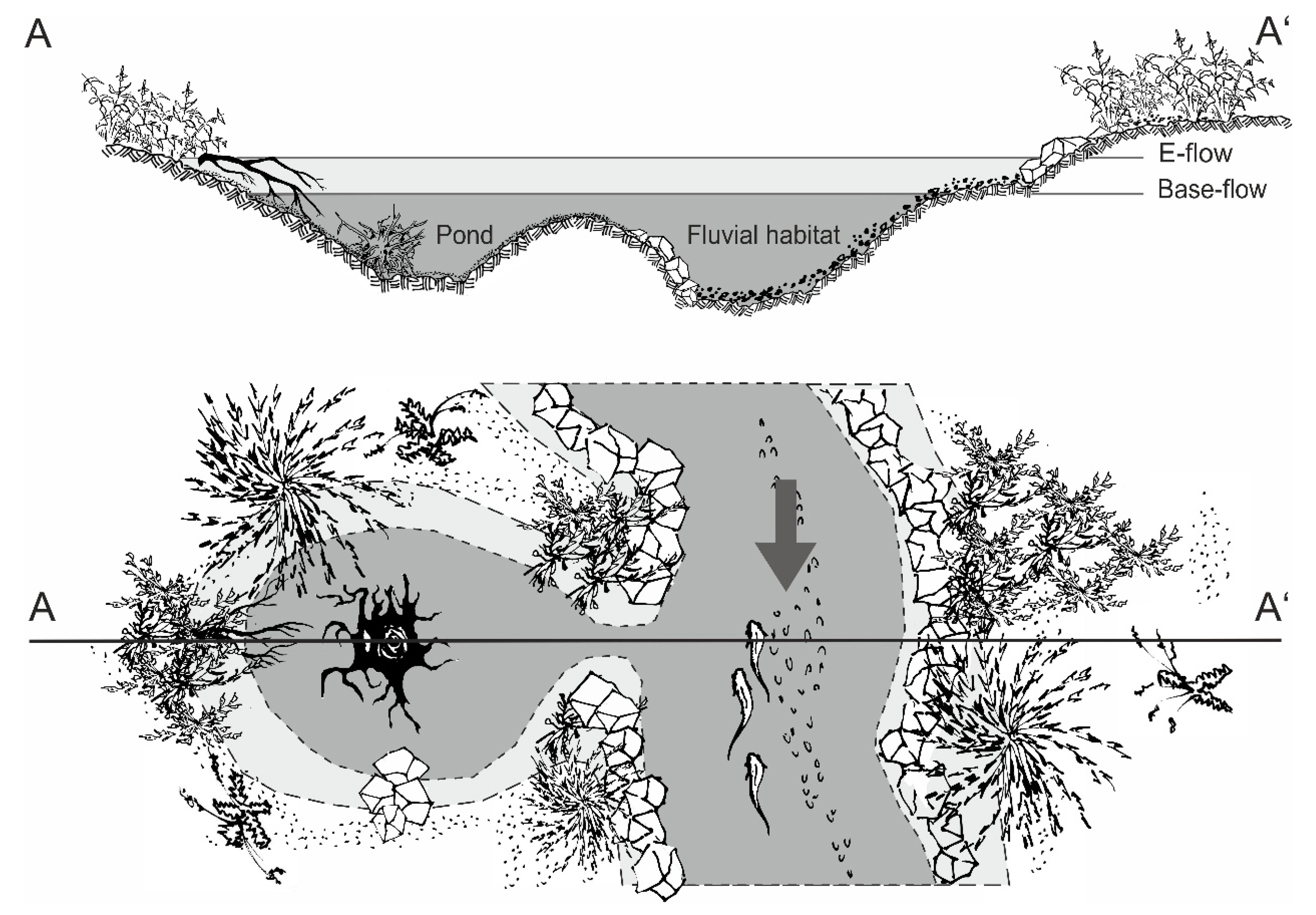
2.2. Characterization of the Fish Passes
2.3. Fish Sampling
2.4. Physicochemical Habitat Characteristics and Vegetation
2.5. Data Analysis
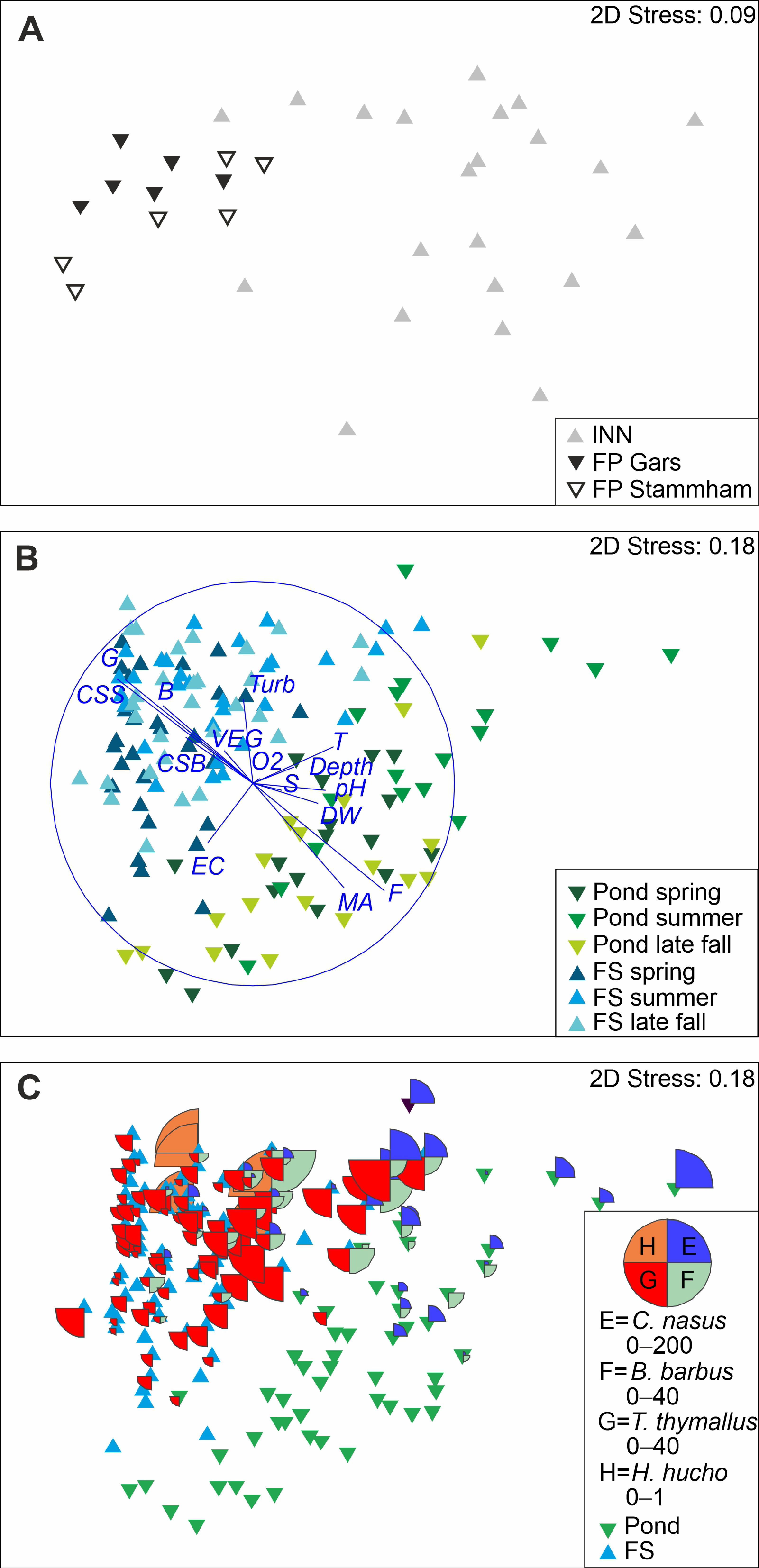
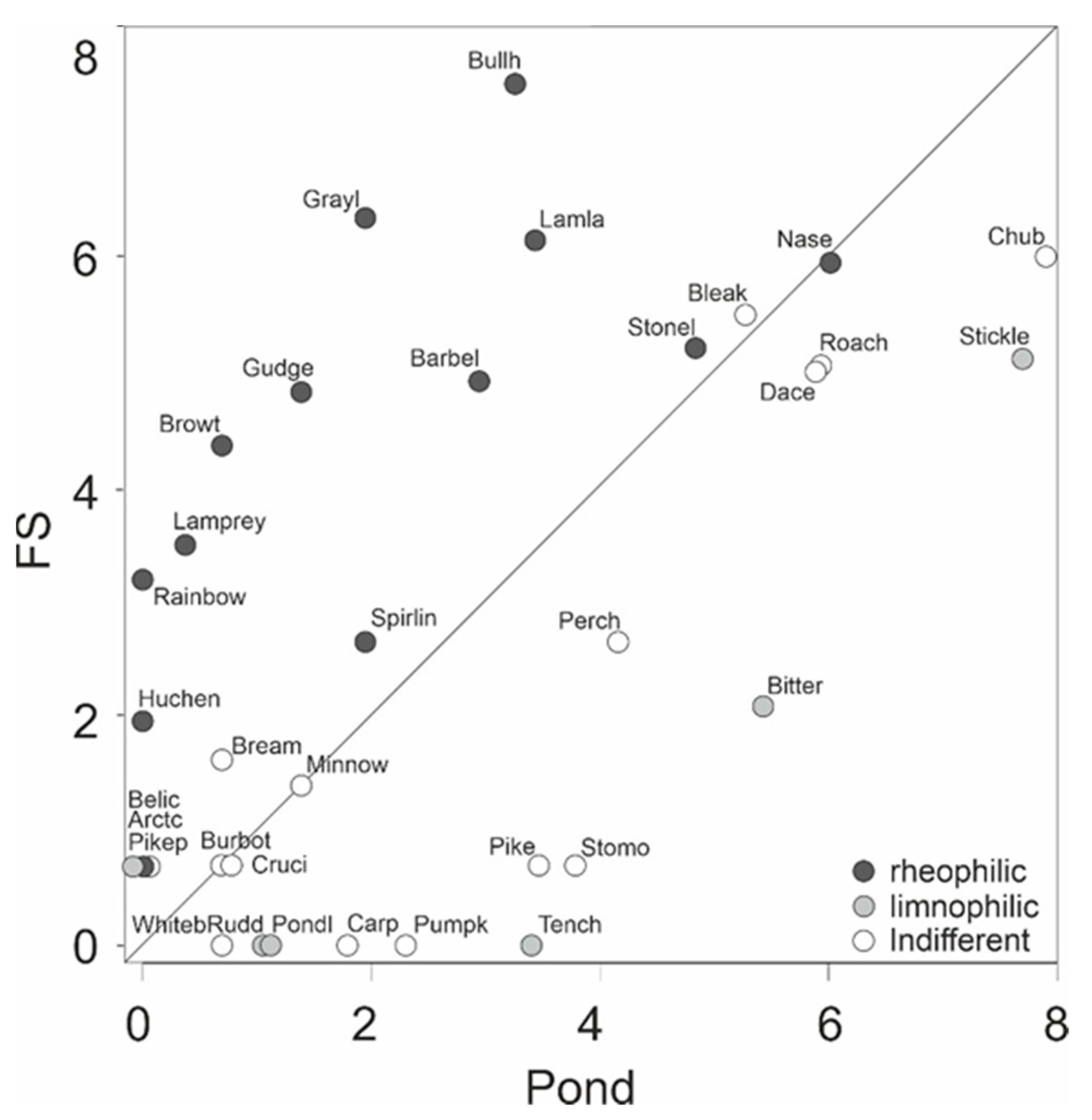
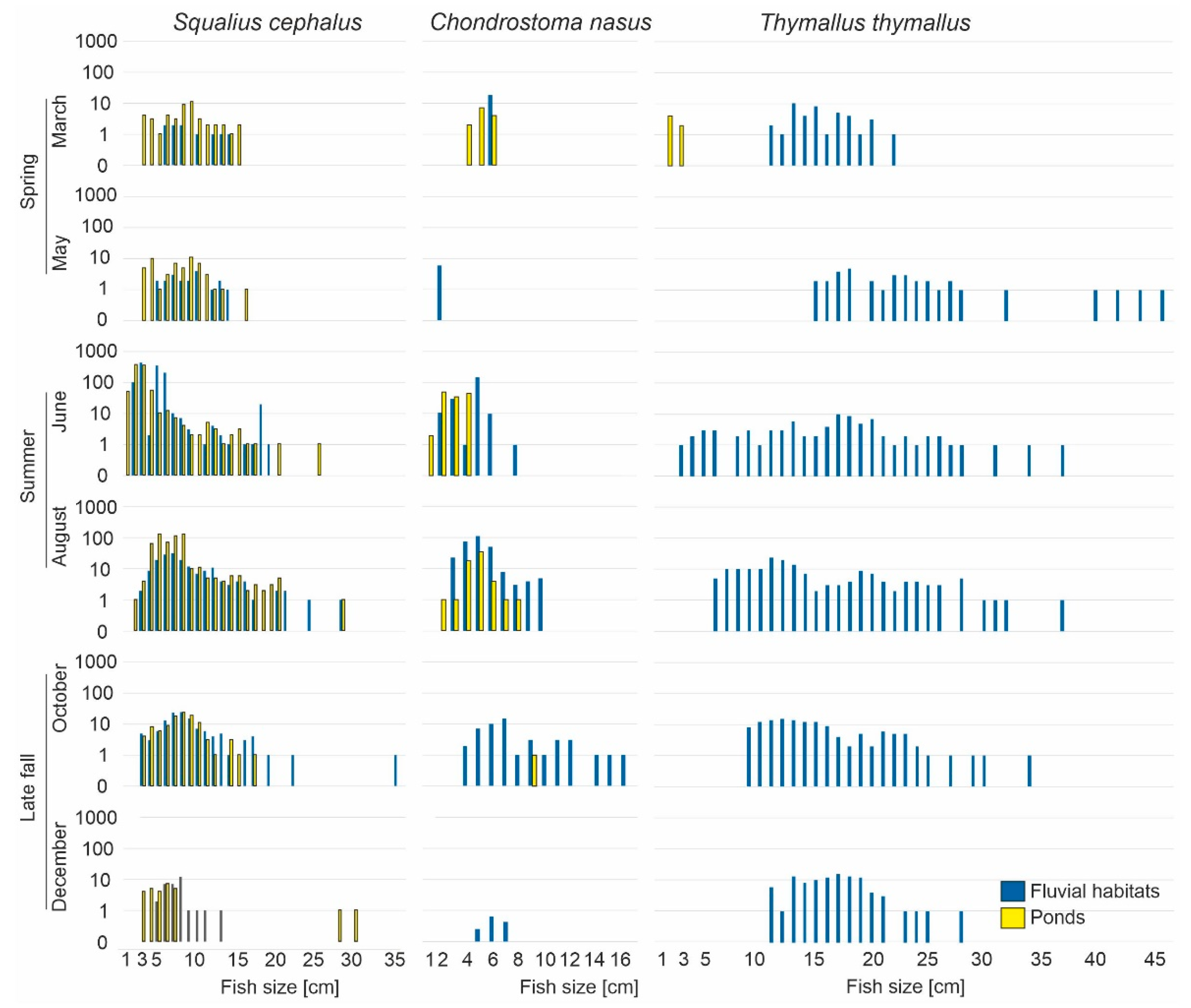
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dynesius, M.; Nilsson, C. Fragmentation and Flow Regulation of River Systems in The northern Third of the World. Science 1994, 266, 753–762. [Google Scholar] [CrossRef]
- Bierschenk, A.M.; Mueller, M.; Pander, J.; Geist, J. Impact of Catchment Land Use on Fish Community Composition in the Headwater Areas of Elbe, Danube and Main. Sci. Total. Environ. 2019, 652, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M.; Bierschenk, A.M.; Bierschenk, B.M.; Pander, J.; Geist, J. Effects of Multiple Stressors on the Distribution of Fish Communities in 203 Headwater Streams of Rhine, Elbe and Danube. Sci. Total. Environ. 2020, 703, 134523. [Google Scholar] [CrossRef] [PubMed]
- Larinier, M.; Marmulla, G. Fish Passes: Types, Principles and Geographical Distribution-an Overview. In Proceedings of the Second International Symposium on the Management of Large Rivers for Fisheries; Food and Agriculture Organization: Rome, Italy, 2004; Volume 2, pp. 183–206. Available online: http://www.fao.org/3/ad526e/ad526e0g.htm (accessed on 6 April 2021).
- Silva, A.T.; Lucas, M.C.; Castro-Santos, T.; Katopodis, C.; Baumgartner, L.J.; Thiem, J.D.; Aarestrup, K.; Pompeu, P.S.; O’Brien, G.C.; Braun, D.C.; et al. The Future of Fish Passage Science, Engineering, and Practice. Fish Fish. 2018, 19, 340–362. [Google Scholar] [CrossRef] [Green Version]
- Venus, T.E.; Smialek, N.; Pander, J.; Harby, A.; Geist, J. Evaluating Cost Trade-Offs between Hydropower and Fish Passage Mitigation. Sustainability 2020, 12, 8520. [Google Scholar] [CrossRef]
- Cubanová, L. Methodology for Fish Passes Design. “Ovidius” University Annals Constantza. Ser. Civil Eng. 2013, 15, 233–240. [Google Scholar]
- Weyand, M.; Kühlmann, M. River Continuity Under Difficult Boundary Conditions–Useful Steps and Practical Hints for Designing a Fish Way. Water Pract. Technol. 2016, 11, 228–233. [Google Scholar] [CrossRef]
- Calles, E.O.; Greenberg, L.A. The Use of Two Nature-Like Fishways by Some Fish Species in the Swedish River Emån. Ecol. Fish 2007, 16, 183–190. [Google Scholar] [CrossRef]
- Baki, A.B.M.; Zhu, D.Z.; Rajaratnam, N. Flow Simulation in a Rock-Ramp Fish pass. J. Hydraul. Eng. 2016, 142, 04016031. [Google Scholar] [CrossRef]
- Meulenbroek, P.; Drexler, S.; Nagel, C.; Geistler, M.; Waidbacher, H. The Importance of a Constructed Near-Nature-Like Danube Fish by-Pass as a Lifecycle Fish Habitat for Spawning, Nurseries, Growing and Feeding: A Long-Term View with Remarks on Management. Mar. Freshw. Res. 2018, 69, 1857–1869. [Google Scholar] [CrossRef]
- Katopodis, C.; Kells, J.A.; Acharya, M. Nature-Like and Conventional Fishways: Alternative Concepts? Can. Water Resour. J. 2001, 26, 211–232. [Google Scholar] [CrossRef] [Green Version]
- Bretón, F.; Baki, A.B.M.; Link, O.; Zhu, D.Z.; Rajaratnam, N. Flow in Nature-Like Fishway and its Relation to Fish Behaviour. Can. J. Civil. Eng. 2013, 40, 567–573. [Google Scholar] [CrossRef]
- EU Commission. Towards Sustainable Water Management in the European Union. First Stage in the Implementation of the Water Framework Directive 2000⁄60⁄EC; Commission Staff Working Document, Accompanying Document to the Communication Forum from the Commission to the European Parliament and the Council COM 2007; EU Commission: Brussels, Belgium, 2007; p. 128. [Google Scholar]
- Kondolf, M.G.; Boulton, A.J.; O’Daniel, S.; Geoffrey, C.; Poole, G.C.; Rahel, F.J.; Stanley, E.H.; Wohl, E.; Bång, A.; Carlstrom, J.; et al. Process-Based Ecological River Restoration: Visualizing Three-Dimensional Connectivity and Dynamic Vectors to Recover Lost Linkages. Ecol. Soc. 2006, 11, 1–17. Available online: https://www.jstor.org/stable/26266026 (accessed on 6 April 2021). [CrossRef]
- Beechie, T.J.; Sear, D.A.; Olden, J.D.; Pess, G.R.; Buffington, J.M.; Moir, H.; Roni, P.; Pollock, M.M. Process-Based Principles for Restoring River Ecosystems. BioScience 2010, 60, 209–222. [Google Scholar] [CrossRef] [Green Version]
- Palmer, M.; Ruhi, A. Linkages between Flow Regime, Biota, and Ecosystem Processes: Implications for River Restoration. Science 2019, 365, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pander, J.; Geist, J. Ecological Indicators for Measuring Stream Restoration Success. Ecol. Indic. 2013, 30, 106–118. [Google Scholar] [CrossRef]
- Pander, J.; Geist, J. The Contribution of Different Restored Habitats to Fish Diversity and Population Development in a Highly Modified River: A Case Study from the River Günz. Water 2018, 10, 1202. [Google Scholar] [CrossRef] [Green Version]
- Forseth, T.; Harby, A. Handbook for Environmental Design in Regulated Salmon Rivers; NINA Special Report; Norsk Institutt for Naturforskning: Trondheim, Norway, 2014. [Google Scholar]
- Noonan, M.J.; Grant, J.W.; Jackson, C.D. A Quantitative Assessment of Fish Passage Efficiency. Fish. Fish 2012, 13, 450–464. [Google Scholar] [CrossRef]
- Pander, J.; Mueller, M.; Geist, J. Ecological Functions of Fish Bypass Channels in Streams: Migration Corridor and Habitat for Rheophilic Species. River Res. Appl. 2013, 29, 441–450. [Google Scholar] [CrossRef]
- DWA-M 509. Fischaufstiegsanlagen und Fischpassierbare Bauwerke–Gestaltung, Bemessung, Qualitätssicherung; Deutsche Vereinigung für Wasserwirtschaft, Abwasser und Abfall e.V.: Hennef, Germany, 2014; p. 334. ISBN 978-3-942964-91-3. [Google Scholar]
- Wolter, C.; Schomaker, C. Fish Passes Design Discharge Requirements for Successful Operation. River Res. Appl. 2019, 35, 1697–1701. [Google Scholar] [CrossRef]
- Nannini, M.A.; Goodrich, J.; Dettmers, J.M.; Soluk, D.A.; Wahl, D.H. Larval and Early Juvenile Fish Dynamics in Main Channel and Backwater Lake Habitats of the Illinois River Ecosystem. Ecol. Freshw. Fish 2012, 21, 499–509. [Google Scholar] [CrossRef]
- Nicolas, Y.; Pont, D. Hydrosedimentary Classification of Natural and Engineered Backwaters of a Large River, the Lower Rhône: Possible Applications for the Maintenance of High Fish Biodiversity. Regul. Rivers: Res. Manag. An. Int. J. Devoted River Res. Manag. 1997, 13, 417–431. [Google Scholar] [CrossRef]
- Pander, J.; Mueller, M.; Geist, J. Succession of Fish Diversity After Reconnecting a Large Floodplain to the Upper Danube River. Ecol. Eng. 2015, 75, 41–50. [Google Scholar] [CrossRef]
- Garrett-Walker, J.; Collier, K.J.; Daniel, A.; Hicks, B.J.; Klee, D. Design Features of Constructed Floodplain Ponds Influence Waterbird and Fish Communities in Northern New Zealand. Freshw. Biol. 2020, 65, 2066–2080. [Google Scholar] [CrossRef]
- Pander, J.; Mueller, M.; Knott, J.; Egg, L.; Geist, J. Is it Worth the Money? The Functionality of Engineered Shallow Stream Banks as Habitat for Juvenile Fishes in Heavily Modified Water Bodies. River Res. Appl. 2017, 33, 63–72. [Google Scholar] [CrossRef]
- Schmall, B.; Ratschan, C. Die historische und aktuelle Fischfauna der Salzach–ein Vergleich mit dem Inn. Beitr. Naturk. Oberösterreichs 2011, 21, 55–191. [Google Scholar]
- Arthington, A.H.; Bhaduri, A.; Bunn, S.E.; Jackson, S.E.; Tharme, R.E.; Tickner, D.; Young, B.; Acreman, M.; Baker, N.; Capon, S.; et al. The Brisbane Declaration and Global Action Agenda on Environmental Flows. Front. Environ. Sci. 2018, 6, 15. [Google Scholar] [CrossRef] [Green Version]
- Pander, J.; Geist, J. Seasonal and Spatial Bank Habitat Use by Fish in Highly Altered Rivers-a Comparison of Four Different Restoration Measures. Ecol. Freshw. Fish 2010, 19, 127–138. [Google Scholar] [CrossRef]
- CEN. Water Quality–Sampling Fish with Electricity; CEN prEN: Brussels, Belgium, 2003; p. 16. [Google Scholar]
- Dußling, U. Handbuch zu fiBS; Schriftenreihe des Verbandes Deutscher Fischereiverwaltungsbeamter und Fischereiwissenschaftler e.V.: Rostock, Germany, 2009; p. 72. [Google Scholar]
- Mueller, M.; Pander, J.; Geist, J. Comprehensive Analysis of > 30 Years of Data on Stream Fish Population Trends and Conservation Status in Bavaria, Germany. Biol. Conserv. 2018, 226, 311–320. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N.; Somerfield, P.J.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 3rd ed.; PRIMER-E: Plymouth, UK, 2014; p. 144. [Google Scholar]
- Bray, J.R.; Curtis, J.T. An Ordination of the Upland Forest Communities of Southern Wisconsin. Ecol. Monogr. 1957, 27, 325–349. [Google Scholar] [CrossRef]
- Clarke, K.R. Non-Parametric Multivariate Analyses of Changes in Community Structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Clarke, K.R.; Somerfield, P.J.; Chapman, M.G. On Resemblance Measures for Ecological Studies, Including Taxonomic Dissimilarities and a Zero-Adjusted Bray–Curtis Coefficient for Denuded Assemblages. J. Exp. Mar. Biol. Ecol. 2006, 330, 55–80. [Google Scholar] [CrossRef]
- Hartig, F. DHARMa: Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models. R Package Version 0.3.3.0. 2020. Available online: http://florianhartig.github.io/DHARMa/ (accessed on 18 February 2021).
- R Core Team R. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 18 February 2021).
- Fox, J.; Weisberg, S. An R Companion to Applied Regression, 2nd ed.; Sage Publications: Thousand Oaks, CA, USA, 2018; p. 571. [Google Scholar]
- Bohl, E.; Kleisinger, H.; Leuner, E. Rote Liste gefährdeter Fische (Pisces) und Rundmäuler (Cyclostomata) Bayern. Bayerisches Landesamt für Umwelt, BayLfU/166/2003. 2003. Available online: https://www.sf-kemmern.de/pdf_Dateien/rote_liste.pdf (accessed on 18 February 2021).
- Council of the European Communities. Council Directive 92/43/EEC of 21 May 1992 on the Conservation of Natural Habitats and of Wild Fauna and Flora. Off. J. Eur. Union 1992, 206, 7–50. [Google Scholar]
- Zauner, G.; Eberstaller, J. Klassifizierungsschema der Österreichischen Flußfischfauna in Bezug auf Deren Lebensraumansprüche. Osterr. Fischerei 1999, 52, 198–205. [Google Scholar]
- Bohlin, T.; Hamrin, S.; Heggberget, T.G.; Rasmussen, G.; Saltveit, S.J. Electrofishing—Theory and Practice with Special Emphasis on Salmonids. Hydrobiologia 1989, 173, 9–43. [Google Scholar] [CrossRef]
- Mollenhauer, R.; Logue, D.; Brewer, S.K. Quantifying Seining Detection Probability for Fishes of Great Plains Sand-Bed Rivers. Trans. Am. Fish. Soc. 2018, 147, 329–341. [Google Scholar] [CrossRef]
- Cushing, D.H. Plankton Production and Year-Class Strength in Fish Populations: An Update of the Match/Mismatch Hypothesis. Adv. Mar. Biol. 1990, 26, 249–293. [Google Scholar]
- Sternecker, K.; Denic, M.; Geist, J. Timing Matters: Species-Specific Interactions between Spawning Time, Substrate Quality, and Recruitment Success in Three Salmonid Species. Ecol. Evol. 2014, 4, 2749–2758. [Google Scholar] [CrossRef]
- Reinartz, R. Untersuchungen zur Gefährdungssituation der Fischart Nase (Chondrostoma nasus L.) in Bayerischen Gewässern. Ph.D. Thesis, Technische Universität München, München, Germany, 1997. [Google Scholar]
- Reckendorfer, W.; Keckeis, H.; Tiitu, V.; Winkler, G.; Zornig, H. Diet shifts in 0+ nase, Chondrostoma nasus: Size-Specific Differences and the Effect of Food. Arch. Hydrobiol. Suppl. 2001, 13512, 425–440. [Google Scholar] [CrossRef]
- Holcčík, J. Threatened Fishes of the World: Hucho Hucho (Linnaeus, 1758) (Salmonidae). Environ. Biol. Fishes 1995, 43, 105–106. [Google Scholar] [CrossRef]
- Nagel, C.; Mueller, M.; Pander, J.; Stoeckle, B.C.; Kuehn, R.; Geist, J. Going with the Flow: Spatio-Temporal Drift Patterns of Larval Fish in a Large Alpine River. Freshw. Biol. 2021. Under Review. [Google Scholar]
- Britton, J.R.; Davies, G.D.; Harrod, C. Trophic Interactions and Consequent Impacts of the Invasive Fish Pseudorasbora Parva in a Native Aquatic Foodweb: A Field Investigation in the UK. Biol. Invasions 2010, 12, 1533–1542. [Google Scholar] [CrossRef]
- Zangl, L.; Jung, M.; Gessl, W.; Koblmueller, S.; Ratschan, C. Oriental or Not: First Record of an Alien Weatherfish (Misgurnus) Species in Austria Verified by Molecular Data. BioInvasions Rec. 2020, 9, 375–383. [Google Scholar] [CrossRef]
- Stoeckle, B.C.; Belle, C.C.; Geist, J.; Oehm, J.; Effenberger, M.; Heiss, M.; Seifert, K.; Kuehn, R. Molecular Confirmation of the Large-Scale Loach Paramisgurnus Dabryanus Dabry de Thiersant, 1872 (Cypriniformes, Cobitidae) in Europe. BioInvasions Rec. 2019, 8, 419–426. [Google Scholar] [CrossRef]
- Schomaker, C.; Wolter, C. The Contribution of Long-Term Isolated Water Bodies to Floodplain Fish Diversity. Freshw. Biol. 2011, 56, 1469–1480. [Google Scholar] [CrossRef]
- Tockner, K.; Ward, J.V.; Arscott, D.B.; Edwards, P.J.; Kollmann, J.; Gurnell, A.M.; Petts, G.E.; Maiolini, B. The Tagliamento River: A Model Ecosystem of European Importance. Aquat. Sci. 2003, 65, 239–253. [Google Scholar] [CrossRef] [Green Version]
- Van Der Nat, D.; Tockner, K.; Edwards, P.J.; Ward, J.V.; Gurnell, A.M. Habitat Change in Braided Flood Plains (Tagliamento, NE-Italy). Freshw. Biol. 2003, 48, 1799–1812. [Google Scholar] [CrossRef]
- Schiemer, F.; Drescher, A.; Hauer, C.; Schwarz, U. The Vjosa River Corridor: A Riverine Ecosystem of European Significance. Acta Zoo Bot. Austria 2018, 155, 1–40. [Google Scholar]
- Telcean, I.C.; Cupşa, D. Fishfauna from the Lowland Mureş River (Romania) and the Floodplain Natural Park Area (Western Romania). An. Univ. Oradea Fasc. Biol. 2009, 16, 131–135. [Google Scholar]
- Britton, J.R.; Pegg, J. Ecology of European barbel Barbus Barbus: Implications for River, Fishery, and Conservation Management. Rev. Fish. Sci. 2011, 19, 321–330. [Google Scholar] [CrossRef]
- Kottelat, M.; Freyhof, J. Handbook of European Freshwater Fishes; Publications Kottelat: Cornol, Switzerland, 2007; p. 646. [Google Scholar]


| Fish Pass | Year Built | Gradient [‰] | Length [m] | Height Difference [m] | Discharge [L s−1] | Flow-Course [m] |
|---|---|---|---|---|---|---|
| Gars | 2015 | 10 | 670 | 7.5 | 900 | 670 |
| Stammham | 2016 | 5.5 | 1500 | 8.5 | 350 | 1500 |
| Fluvial habitats | Width [m] | Depth [m] | CSB [ms−1] | CSS [ms−1] | Area [m²] | MA [%] | DW [%] | Veg [%] | B [%] | G [%] | S [%] | F [%] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gars | 4.83 | 0.43 | 0.43 | 0.61 | 146 | 2 | 5 | 7 | 34 | 67 | 23 | 11 |
| 3.10–7.00 | 0.10–1.12 | 0.00–1.44 | 0.00–2.07 | 120–177 | 0–5 | 0–10 | 0–25 | 25–50 | 50–70 | 15–35 | 5–15 | |
| Stammham | 2.89 | 0.34 | 0.36 | 0.41 | 89 | 3 | 5 | 46 | 4 | 60 | 26 | 14 |
| 1.83–4.32 | 0.08–0.75 | 0.00–1.09 | 0.01–1.10 | 75–107 | 0–10 | 0–10 | 0–90 | 0–25 | 45–70 | 20–35 | 10–20 |
| Floodplain Ponds | N | Length [m] | Width [m] | Depth Base-Flow [m] | Depth E-Flow [m] | Area Base-Flow [m²] | Area E-Flow [m²] | MA [%] | DW [%] | Veg [%] | B [%] | G [%] | S [%] | F [%] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gars | 5 | 15.52 | 8.10 | 0.32 | 0.50 | 66 | 115 | 38 | 10 | 4 | 2 | 7 | 28 | 65 |
| 9.55–25.89 | 6.60–9.52 | 0.06–0.93 | 0.25–1.08 | 44–130 | 66–181 | 0–90 | 5–20 | 0–20 | 0–10 | 0–15 | 0–40 | 50–90 | ||
| Stammham | 4 | 26.00 | 11.70 | 0.51 | NA | 377 | NA | 58 | 8 | 4 | 3 | 13 | 26 | 61 |
| 11.47–39.00 | 2.89–30.00 | 0.17–1.65 | NA | 67–1127 | NA | 10–95 | 5–15 | 0–10 | 0–10 | 0–40 | 20–30 | 30–75 |
| Gars Fluvial Habitats | Gars Ponds | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| T [°C] | O2 [mgl−1] | EC [µscm−1] | pH | Turbidity [NTU] | T [°C] | O2 [mgl−1] | EC [µscm−1] | pH | Turbidity [NTU] | |
| March | 6.4 | 12.1 | 385 | 8.2 | 16.3 | 7.6 | 14.2 | 358 | 8.5 | 12.3 |
| 6.4–6.5 | 11.8–12.8 | 380–388 | 8.2–8.3 | 12.1–24.8 | 6.6–8.4 | 11.0–16.6 | 352–416 | 8.0–8.9 | 2.6–19.6 | |
| May | 9.1 | 11.1 | 276 | 8.3 | 36.0 | 10.7 | 12.3 | 272 | 8.5 | 19.1 |
| 8.6–9.4 | 11.0–11.1 | 273–279 | 8.2–8.5 | 28.5–45.1 | 8.8–12.5 | 11.0–15.3 | 263–280 | 8.3–8.9 | 6.9–35.3 | |
| June | 13.8 | 10.2 | 194 | NA | 117.0 | 16.1 | 10.4 | 191 | NA | 60.5 |
| 13.6–14.1 | 10.1–10.4 | 195–199 | NA | 101.6–134.9 | 14.3–20.5 | 9.3–11.2 | 169–197 | NA | 12.5–113.0 | |
| August | 18.6 | 9.1 | 224 | 8.0 | 167.7 | 22.0 | 10.0 | 208 | 8.5 | 78 |
| 18.2–19.1 | 7.8–9.7 | 218–231 | 7.9–8.1 | 139.0–215.2 | 18.9–27.2 | 7.2–13.3 | 150–225 | 7.6–9.7 | 5.5–141.8 | |
| October | 11.8 | 10.9 | 330 | 8.1 | 16,4 | 13.3 | 11.6 | 303 | 8.4 | 7.3 |
| 11.5–12.1 | 10.3–11.1 | 327–333 | 7.9–8.2 | 11.1–23.2 | 12.7–14.8 | 4.0–18.3 | 241–386 | 7.8–8.9 | 2.7–17.3 | |
| December | 4.8 | 11.7 | 388 | 8.1 | 15.7 | 3.0 | 13.4 | 336 | 8.6 | 10.1 |
| 4.7–4.9 | 11.1–11.9 | 359–393 | 7.9–8.2 | 10.1–25.6 | 2.4–3.4 | 10.3–16.2 | 275–362 | 8.1–8.8 | 0.5–22.9 | |
| Stammham Fluvial Habitats | Stammham Ponds | |||||||||
| T [°C] | O2 [mgl−1] | EC [µscm−1] | pH | Turbidity [NTU] | T [°C] | O2 [mg−1] | EC [µscm−1] | pH | Turbidity [NTU] | |
| March | 7.0 | 11.2 | 425 | 8.0 | 16.0 | 7.4 | 7.7 | 425 | 7.8 | 3.5 |
| 6.6–7.1 | 11.0–11.7 | 379–441 | 7.9–8.1 | 10.6–26.3 | 6.7–9.1 | 1.2–13.0 | 405–442 | 6.7–8.5 | 0.2–8.7 | |
| May | 9.0 | 11.0 | 327 | 8.2 | 20.3 | 13.3 | 10.7 | 369 | 8.2 | 4.4 |
| 8.7–9.3 | 10.0–11.3 | 304–365 | 8.1–8.3 | 12.5–27.7 | 9.3–19.6 | 3.8–16.9 | 319–418 | 7.8–8.6 | 1.8–11.4 | |
| June | 14.6 | 9.9 | 253 | NA | 72.2 | 17.9 | 9.3 | 295 | NA | 25.8 |
| 14.0–14.8 | 9.7–10.1 | 234–288 | NA | 52.0–90.3 | 12.5–24.5 | 4.4–13.1 | 207–354 | NA | 2.2–74.6 | |
| August | 19.1 | 8.8 | 264 | 8.0 | 79.9 | 23.3 | 12.7 | 269 | 8.6 | 27.9 |
| 18.3–19.7 | 7.7–9.0 | 245–295 | 7.9–8.9 | 59.4–107.0 | 15.1–30.0 | 4.5–18.4 | 203–328 | 8.0–9.5 | 1.9–128.7 | |
| October | 12.8 | 10.3 | 355 | 8.1 | 9.9 | 13.9 | 8.1 | 335 | 8.0 | 3.2 |
| 12.3–13.2 | 9.5–11.0 | 342–379 | 7.9–8.2 | 5.8–17.1 | 11.6–15.8 | 1.8–10.9 | 297–376 | 7.5–8.5 | 1.1–5.6 | |
| December | 5.1 | 11.2 | 407 | 8.1 | 17.1 | 5.6 | 8.0 | 388 | 7.9 | 7.4 |
| 4.7–6.1 | 10.5–11.8 | 400–423 | 8.0–8.1 | 7.9–29.2 | 4.4–9.8 | 2.6–11.0 | 373–421 | 7.6–8.2 | 0.3–20.9 | |
| ANOSIM | SIMPER | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Comparisons | R-Value | p-Value | df | AVDIS | Bleak | Stonel | Barbel | Bullh | Nase | Stickle | Dace | Lamprey Larvae | Stomo | Bitter | Chub | Grayl |
| FP–River Inn | 0.482 | <0.001 | 1 | 88.42 | 9.44–17.89 | NA | NA | 47.46–3.02 | NA | 52.95–0.30 | NA | NA | NA | NA | 76.57–7.13 | 12.96–0.71 |
| FP Gars–FP Stammham | 0.07 | <0.001 | 1 | 83.28 | NA | NA | NA | 20.15–6.31 | 3.36–7.46 | 5.71–25.49 | NA | 0.00–6.04 | NA | NA | 18.71–25.36 | 2.26–5.29 |
| FP spring–FP summer | 0.134 | <0.001 | 2 | 82.97 | 7.85–13.92 | NA | NA | NA | 1.17 –14.38 | 3.17–40.40 | NA | 2.46–5.44 | NA | NA | 2.73–58.19 | 1.69–5.15 |
| FP summer–FP late fall | 0.051 | <0.01 | 2 | 81.22 | NA | NA | NA | 13.92–16.19 | 14.38–1.21 | 40.40–5.71 | 6.83–3.54 | NA | NA | NA | 58.19–6.02 | 5.15–4.88 |
| FP spring– FP late fall | 0.023 | >0.05 | 2 | 82.31 | NA | NA | NA | 7.85–16.19 | NA | 3.17–5.71 | NA | 2.46–1.92 | NA | NA | 2.73–6.02 | 1.69–4.88 |
| FS–PO | 0.631 | <0.001 | 1 | 92.62 | NA | NA | NA | 19.97–0.46 | 4.19–7.91 | 1.81–40.78 | NA | NA | NA | NA | 4.42–52.13 | 6.18–0.11 |
| PO Gars–PO Stammham | 0.122 | <0.01 | 3 | 84.05 | NA | 3.70–0.58 | NA | NA | 6.23–10.00 | 12.53–76.08 | 7.63–5.46 | NA | NA | NA | 39.40–68.04 | NA |
| FS Gars–FS Stammham | 0.208 | <0.001 | 3 | 72.10 | NA | NA | NA | 36.25–9.11 | NA | NA | NA | 0.00–8.50 | NA | NA | 1.47–6.39 | 3.97–7.65 |
| PO Gars–FS Gars | 0.795 | <0.001 | 3 | 93.64 | NA | NA | NA | 0.83–36.25 | NA | 12.53–0.03 | NA | NA | NA | NA | 39.40–1.47 | NA |
| PO Stammham–FS Stammham | 0.635 | <0.001 | 3 | 93.34 | NA | NA | NA | 0.00–9.11 | 10.00–6.33 | 76.08–3.00 | NA | 0.50–8.50 | NA | 9.42–0.13 | 68.04–6.39 | 0.00–7.65 |
| FS spring–FS summer | 0.181 | <0.001 | 5 | 71.43 | NA | NA | 0.03–4.30 | 12.40–21.77 | 0.83–9.83 | NA | NA | 3.83–8.60 | NA | NA | 0.97–7.13 | 2.50–8.23 |
| FS summer–FS late fall | 0.009 | >0.05 | 5 | 65.93 | 3.30–4.67 | NA | NA | 21.77–25.73 | 9.83–1.90 | NA | NA | 8.60–2.87 | NA | NA | 7.13–5.17 | 8.23 –7.80 |
| FS spring–FS late fall | 0.119 | <0.001 | 5 | 70.00 | NA | NA | NA | 12.40–25.73 | NA | NA | NA | 3.83–2.87 | NA | NA | 0.97–5.17 | 2.50–7.80 |
| PO spring–PO summer | 0.201 | <0.001 | 5 | 82.62 | NA | NA | NA | NA | 1.72–21.94 | 8.06–105.22 | NA | NA | NA | NA | 5.67 143.28 | NA |
| PO summer–PO late fall | 0.207 | <0.01 | 5 | 85.21 | NA | NA | NA | NA | 21.94–0.06 | 105.22–9.06 | NA | NA | NA | NA | 143.28–7.44 | NA |
| PO spring–PO late fall | 0.017 | >0.05 | 5 | 81.34 | NA | 2.56–1.72 | NA | NA | NA | 8.06–9.06 | NA | NA | 1.00–1.33 | 1.11–2.50 | 5.67–7.44 | NA |
| PO spring–FS spring | 0.699 | <0.001 | 5 | 92.69 | NA | 2.56–0.70 | NA | 0.28–12.40 | NA | 8.06–0.23 | NA | 0.17–3.83 | NA | NA | 5.67–0.97 | NA |
| PO summer–FS summer | 0.644 | <0.001 | 5 | 90.50 | NA | NA | NA | 0.83–21.77 | 21.94–9.83 | 105.22–1.50 | NA | NA | NA | NA | 143.28–7.13 | 0.00–8.23 |
| PO late fall–FS late fall | 0.723 | <0.001 | 5 | 93.79 | NA | NA | NA | 0.28–25.73 | NA | 9.06–3.70 | 5.83–2.17 | NA | NA | NA | 7.44–5.17 | 0.00–7.80 |
| Species | Common Names | Abbreviation | INN | FP | Gars | Stammham | Family | CP | RL | RLB | FFH |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Anguilla anguilla | European eel | Eel | 225 | 0 | 0 | 0 | Anguillidae | indifferent | 3 | F | NL |
| Abramis brama | Freshwater bream | Bream | 162 | 5 | 4 | 1 | Cyprinidae | indifferent | * | NL | NL |
| Alburnoides bipunctatus | Spirlin | Spirlin | 85 | 19 | 7 | 12 | Cyprinidae | rheophil | V | 2 | NL |
| Alburnus alburnus | Bleak | Bleak | 1830 | 434 | 26 | 408 | Cyprinidae | indifferent | * | V | NL |
| Aspius aspius | Asp | Asp | 10 | 0 | 0 | 0 | Cyprinidae | indifferent | 3 | 3 | Annex II |
| Barbatula barbatula | Stone loach | Stonel | 70 | 305 | 191 | 114 | Nemacheilidae | rheophil | * | V | NL |
| Barbus barbus | Barbel | Barbel | 112 | 152 | 28 | 124 | Cyprinidae | rheophil | * | 3 | Annex V |
| Blicca bjoerkna | White bream | Whitebr | 1 | 1 | 0 | 1 | Cyprinidae | indifferent | * | NL | NL |
| Carassius gibelio | Prussian carp | Cruci | 1 | 2 | 0 | 2 | Cyprinidae | indifferent | * | NL | NL |
| Chondrostoma nasus | Common nase | Nase | 211 | 786 | 222 | 564 | Cyprinidae | rheophil | V | 2 | NL |
| Cottus gobio | Bullhead | Bullh | 507 | 1822 | 1330 | 492 | Cottidae | rheophil | * | V | Annex II |
| Cyprinus carpio | Common carp | Carp | 7 | 5 | 5 | 0 | Cyprinidae | indifferent | * | NL | NL |
| Esox lucius | Northern pike | Pike | 110 | 32 | 4 | 28 | Esocidae | indifferent | * | NL | NL |
| Gasterosteus aculeatus | Three-spined stickleback | Stickle | 15 | 2365 | 377 | 1988 | Gasterosteidae | limnophil | * | V | NL |
| Gobio gobio | Gudgeon | Gudge | 23 | 125 | 0 | 125 | Cyprinidae | rheophil | * | V | NL |
| Gymnocephalus cernua | Ruffe | Ruffe | 19 | 0 | 0 | 0 | Percidae | indifferent | * | V | NL |
| Hucho hucho | Huchen | Huchen | 22 | 6 | 3 | 3 | Salmonidae | rheophil | 2 | 3 | Annex II |
| NA | Brook lamprey | Blamp | 20 | 532 | 15 | 517 | Petromyzontidae | rheophil | * | 1 | Annex II |
| Lepomis gibbosus | Pumpkinseed | Pumpk | 0 | 9 | 2 | 7 | Percidae | indifferent | * | NL | NL |
| Leucaspius delineatus | Belica | Belica | 0 | 1 | 0 | 1 | Cyprinidae | limnophil | * | 3 | NL |
| Leuciscus idus | Ide | Ide | 26 | 0 | 0 | 0 | Cyprinidae | indifferent | 3 | V | NL |
| Leuciscus leuciscus | Common dace | Dace | 230 | 506 | 246 | 260 | Cyprinidae | indifferent | * | V | NL |
| Lota lota | Burbot | Burb | 235 | 2 | 2 | 0 | Lotidae | indifferent | V | 2 | NL |
| Misgurnus anguillicaudatus | Pond loach | Pondl | 0 | 2 | 2 | 0 | Cobitidae | limnophil | ◊ | NL | NL |
| Oncorhynchus mykiss | Rainbow trout | Raintr | 110 | 23 | 14 | 9 | Salmonidae | rheophil | ◊ | NL | NL |
| Perca fluviatilis | European perch | Perch | 89 | 76 | 29 | 47 | Percidae | indifferent | * | NL | NL |
| Phoxinus phoxinus | Eurasian minnow | Minnow | 28 | 6 | 0 | 6 | Cyprinidae | indifferent | * | 3 | NL |
| Pseudorasbora parva | Stone moroko | Stomo | 4 | 44 | 43 | 1 | Cyprinidae | indifferent | ◊ | NL | NL |
| Rhodeus amarus | European bitterling | Bitter | 5 | 233 | 0 | 233 | Cyprinidae | limnophil | * | 2 | Annex II |
| Romanogobio vladykovi | Gudgeon | Wgudge | 15 | 0 | 0 | 0 | Cyprinidae | rheophil | 2 | 2 | Annex II |
| Rutilus rutilus | Roach | Roach | 267 | 526 | 44 | 482 | Cyprinidae | indifferent | * | NL | NL |
| Salmo trutta fario | Brown trout | Browntr | 331 | 77 | 16 | 61 | Salmonidae | rheophil | * | V | NL |
| Salvelinus fontinalis | Brook trout | Brooktr | 4 | 1 | 1 | 0 | Salmonidae | rheophil | ◊ | NL | NL |
| Sander lucioperca | Pike-pearch | Pikepe | 12 | 1 | 0 | 1 | Percidae | indifferent | * | NL | NL |
| Scardinius erythrophthalmus | Rudd | Rudd | 4 | 2 | 1 | 1 | Cyprinidae | limnophil | * | NL | NL |
| Silurus glanis | Wels catfish | Wels | 3 | 0 | 0 | 0 | Siluridae | indifferent | 2 | V | NL |
| Squalius cephalus | Chub | Chub | 837 | 3213 | 1235 | 1978 | Cyprinidae | indifferent | * | NL | NL |
| Thymallus thymallus | Grayling | Grayl | 238 | 562 | 149 | 413 | Salmonidae | rheophil | 2 | 2 | Annex V |
| Tinca tinca | Tench | Tench | 1 | 29 | 6 | 23 | Cyprinidae | limnophil | * | NL | NL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pander, J.; Nagel, C.; Geist, J. Integration of Constructed Floodplain Ponds into Nature-Like Fish Passes Supports Fish Diversity in a Heavily Modified Water Body. Water 2021, 13, 1018. https://doi.org/10.3390/w13081018
Pander J, Nagel C, Geist J. Integration of Constructed Floodplain Ponds into Nature-Like Fish Passes Supports Fish Diversity in a Heavily Modified Water Body. Water. 2021; 13(8):1018. https://doi.org/10.3390/w13081018
Chicago/Turabian StylePander, Joachim, Christoffer Nagel, and Juergen Geist. 2021. "Integration of Constructed Floodplain Ponds into Nature-Like Fish Passes Supports Fish Diversity in a Heavily Modified Water Body" Water 13, no. 8: 1018. https://doi.org/10.3390/w13081018
APA StylePander, J., Nagel, C., & Geist, J. (2021). Integration of Constructed Floodplain Ponds into Nature-Like Fish Passes Supports Fish Diversity in a Heavily Modified Water Body. Water, 13(8), 1018. https://doi.org/10.3390/w13081018






