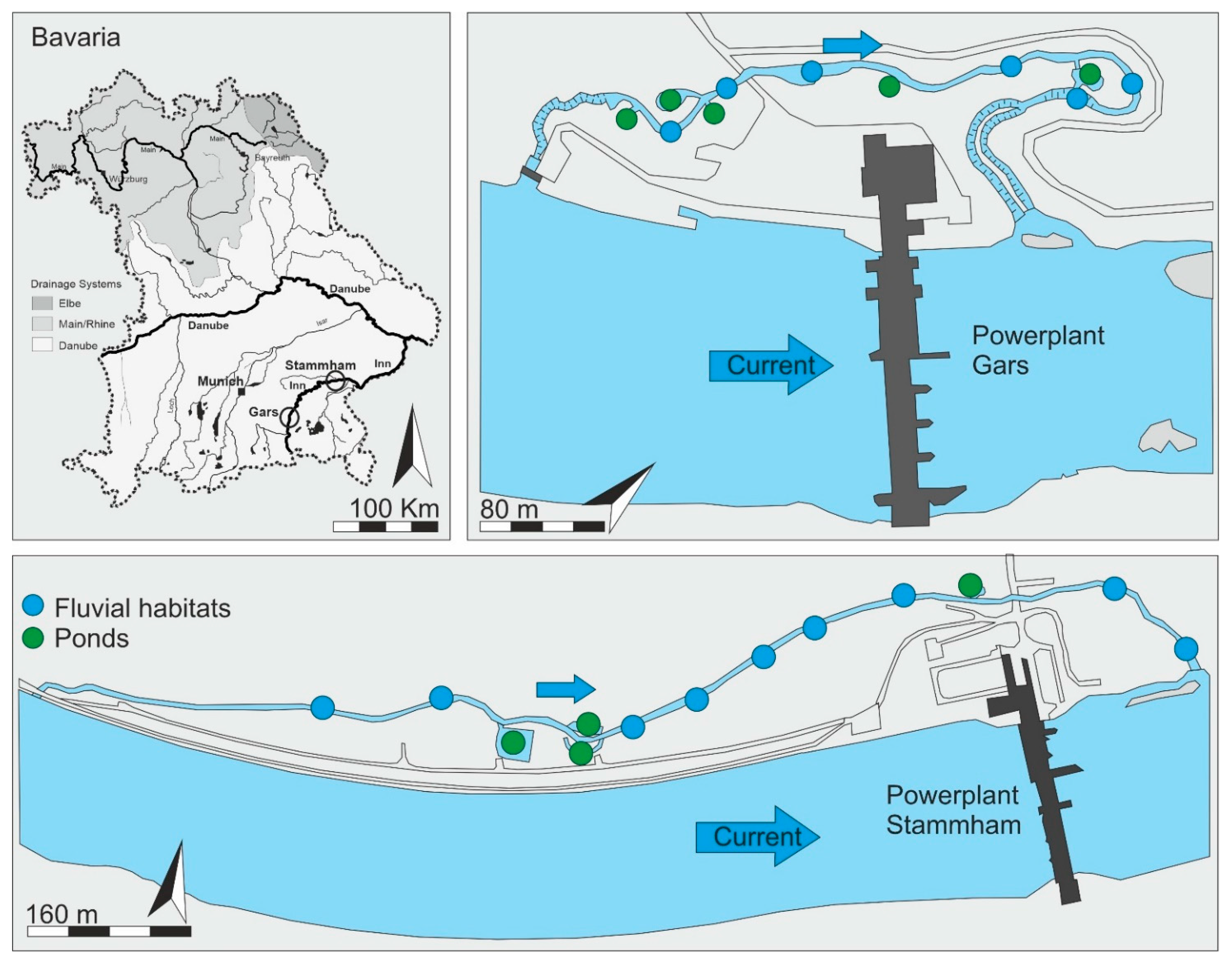
1. Introduction
Habitat degradation and fragmentation of river systems caused by dams, weirs, and other barriers is considered a major challenge in restoring European fish populations [
1]. For instance, impoundments that restrict river dynamic processes, such as sediment transport or other matter fluxes, have been identified as one of the major factors related to the decline of riverine fish species in the headwater areas of Elbe, Main, and Danube rivers [
2,
3]. Besides restoring habitat quality of rivers by improving structural richness, sediment transport and deadwood dynamics, the restoration of fish passage in fragmented rivers is high on the agenda [
4,
5,
6]. Different techniques to restore fish migration, such as technical fish passes (e.g., vertical slot-pass; [
7,
8]) or solutions with nature-like construction schemes [
9,
10,
11], have been applied in practice. These artificial structures have widely proven their valuable contributions to restore fish migration [
12,
13]. However, the restoration of fish communities in degraded rivers seems to be highly challenging and not only dependent on restoring connectivity, due to the manifold other restrictions in heavily modified water bodies (HMWB; [
14]). In these waters, the natural river dynamic processes are strongly impaired [
15,
16,
17], limiting the chances of restoring complete life cycles of specialized riverine fish species, such as
Thymallus thymallus L.,
Hucho hucho L.,
Salmo trutta L.,
Chondrostoma nasus L., and
Barbus barbus L. [
18,
19]. It has been proposed to restore fish habitats in highly degraded rivers by designing separate, parallel flowing river courses or channels outside the degraded systems using a portion of their natural discharge [
19,
20] as it is required for nature-like fish passes that initially were built to restore fish migration (reviewed in [
21]). This initiated a discussion between ecologists, river managers and users, to which extend fish passes can provide important fish habitats potentially compensating the existing habitat loss in the main river. First studies on the contribution of nature-like fish passes to overall biodiversity of fishes indicated that these structures can be highly valuable refuges for rheophilic fishes [
13,
19,
22], e.g., providing key habitats for reproduction [
11,
12].
Many countries developed extensive guidelines how fish passes have to be built (e.g., Germany; [
23]), often primarily focussing on their role for fish passage. Nature-like solutions are generally built to mimic small streams with discharges between 0.3 m³/s and 30 m³/s comprising runs, riffles and pools [
23,
24], with a special focus on providing a widely functional migration corridor for the target species of conservation throughout the year [
23]. Besides the fluvial habitats, nature-like solutions increasingly consider the lateral connectivity to stagnant waters in the floodplain. These additional aquatic habitats can comprise large structures, such as backwaters that evolved from ancient cut-off meanders or small floodplain ponds [
25,
26,
27,
28]. If such structures are absent, they can also be newly constructed.
In this study, we investigated the value of fluvial habitats and newly created and connected small floodplain ponds (further referred to as ponds) as habitat for the fish community in two nature-like fish passes in relation to their characteristics. In particular, we hypothesized that (i) fish community composition in the nature-like fish passes reflects the fish community composition of the main stem, and (ii) nature-like fish passes integrating stagnant waters provide habitat for both rheophilic and stagnophilic fish species, thus contributing to greater fish diversity. Furthermore, we hypothesized that (iii) fish pass systems can provide important juvenile habitats for rheophilic and stagnophilic species, as well.
3. Results
In the two fish passes 11,893 individuals of fish and lampreys were caught during the sampling survey. The 33 fish species detected belonged to nine families with Cyprinidae (55% of all species) dominating, followed by Salmonidae (15%) and Percidae (9%). The Families Esocidae, Nemacheilidae, Cottidae, Cobitidae, and Gasterosteidae comprised only one species each (
Table 6). Most abundant species were
S. cephalus (27% of all individuals), followed by
Gasterosteus aculeatus L. (20%),
C. gobio (15%),
C. nasus (7%),
T. thymallus (5%),
A. alburnus (4%),
Leuciscus leuciscus L. (4%),
R. rutilus (4%),
Barbatula barbatula L. (3%), and
Rhodeus amarus Bloch (2%). All other species only contributed less than 1% to the individuals (
Table 6).
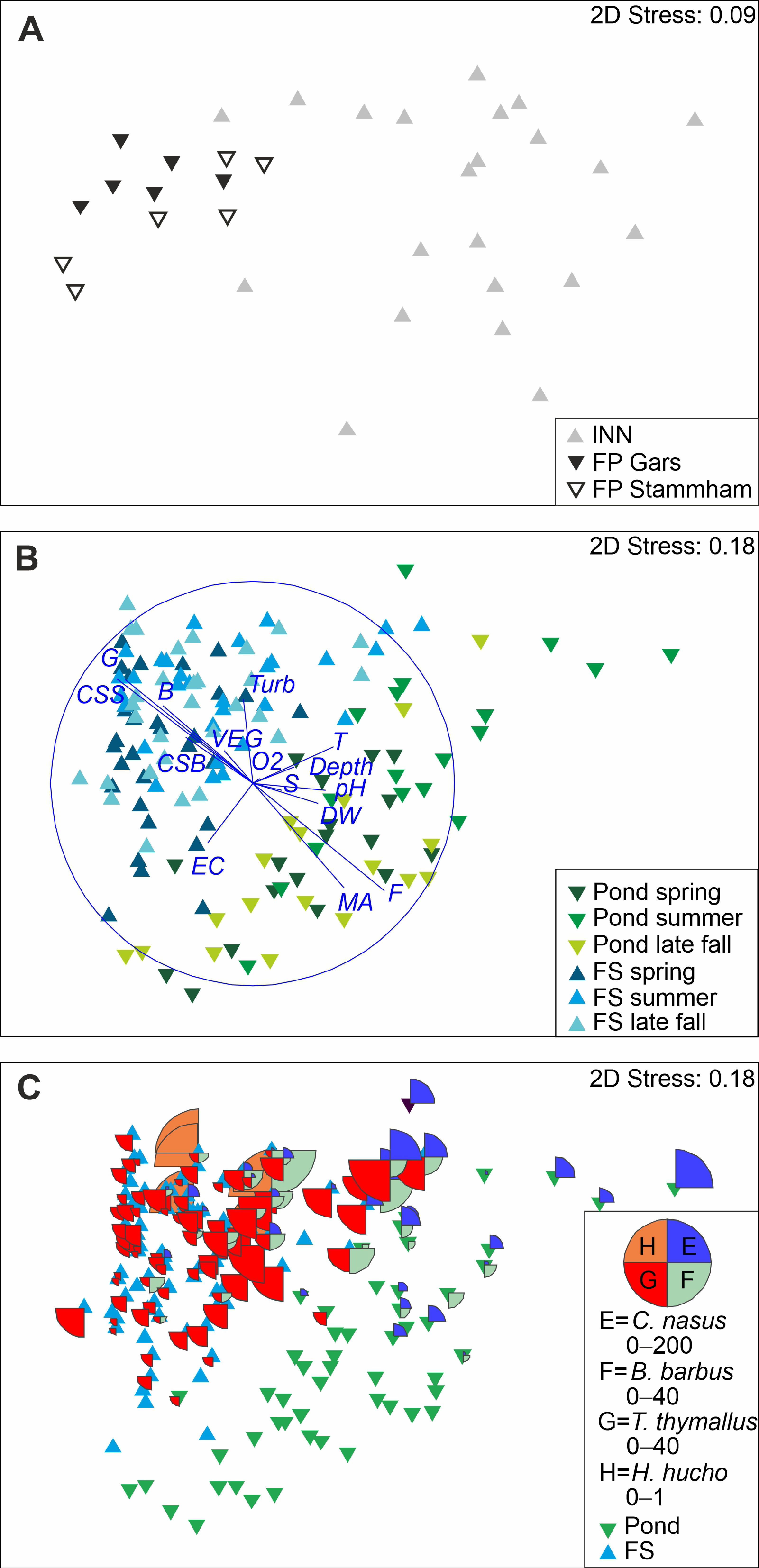
Fish community composition in the fish passes based on pooled data over all sampling time points differed significantly from the River Inn (
Figure 4, ANOSIM, R = 0.48,
p < 0.001). According to SIMPER analysis, these differences were mainly attributed to the higher abundance of
C. gobio,
S. cephalus,
G. aculeatus,
T. thymallus, and Petromyzontidae in the fish passes and higher abundance of
A. alburnus in the main river (
Table 5). Species richness in the River Inn was higher (40 species) than in the fish passes with six more species being caught. These were mainly ubiquitous species except for the Danube-endemic rheophilic specialist
Romanogobio vladykovi Fang. Three additional species (
Lepomis gibbosus L.,
Misgurnus bipartitus Dybowski, and
Leucaspius delineates Heckel) were exclusively caught in the fish passes (
Table 6).
The community composition of the FP Gars differed significantly from the FP Stammham; however, the R-value was very low for this comparison (ANOSIM, R = 0.07,
p < 0.001). According to SIMPER, the same set of species was identified as for the comparison Inn and FP (
Table 5), with
C. gobio being 3-fold more abundant in Gars and
S. cephalus,
G. aculeatus,
T. thymallus, and Petromyzontidae being more abundant in Stammham. Throughout the year, fish community composition in FP changed significantly (ANOSIM, R = 0.058,
p < 0.001) between spring (March and May), summer (June and August), and late fall (October and December) (
Figure 4,
Table 5). For example, SIMPER detected that species, such as
C. gobio, increased in abundance from spring to late fall, whilst
S. cephalus,
G. aculeatus, and
C. nasus reached their abundance peak in summer.
T. thymallus was also detected with highest densities in summer, but, in contrast to the other species, it was still present in high abundances in the FP during late fall.
Fluvial habitats of the fish passes differed significantly from ponds in community composition (
Figure 4, ANOSIM, R = 0.631,
p < 0.001). BEST analysis detected macrophyte coverage, gravel, boulders, temperature and current speed at the surface as variables best fitting the ordination of abundance data in the nMDS (R
2 = 0.74,
Figure 4). There was a higher proportion of macrophytes and a higher water temperature in ponds, and more gravel, boulders and higher current speed in fluvial habitats. According to SIMPER, differences in fish community structure among habitat types were mainly attributed to higher abundances of
T. thymallus (56-fold),
C. gobio (40-fold),
C. nasus (1.9-fold) in fluvial habitats, and
G. aculeatus (22-fold) and
S. cephalus (12-fold) in ponds (
Table 5). Five stagnophilic or indifferent species, such as
L. gibbosus,
Scardinius erythrophthalmus L.,
M. bipartitus,
Cyprinus carpio L., and
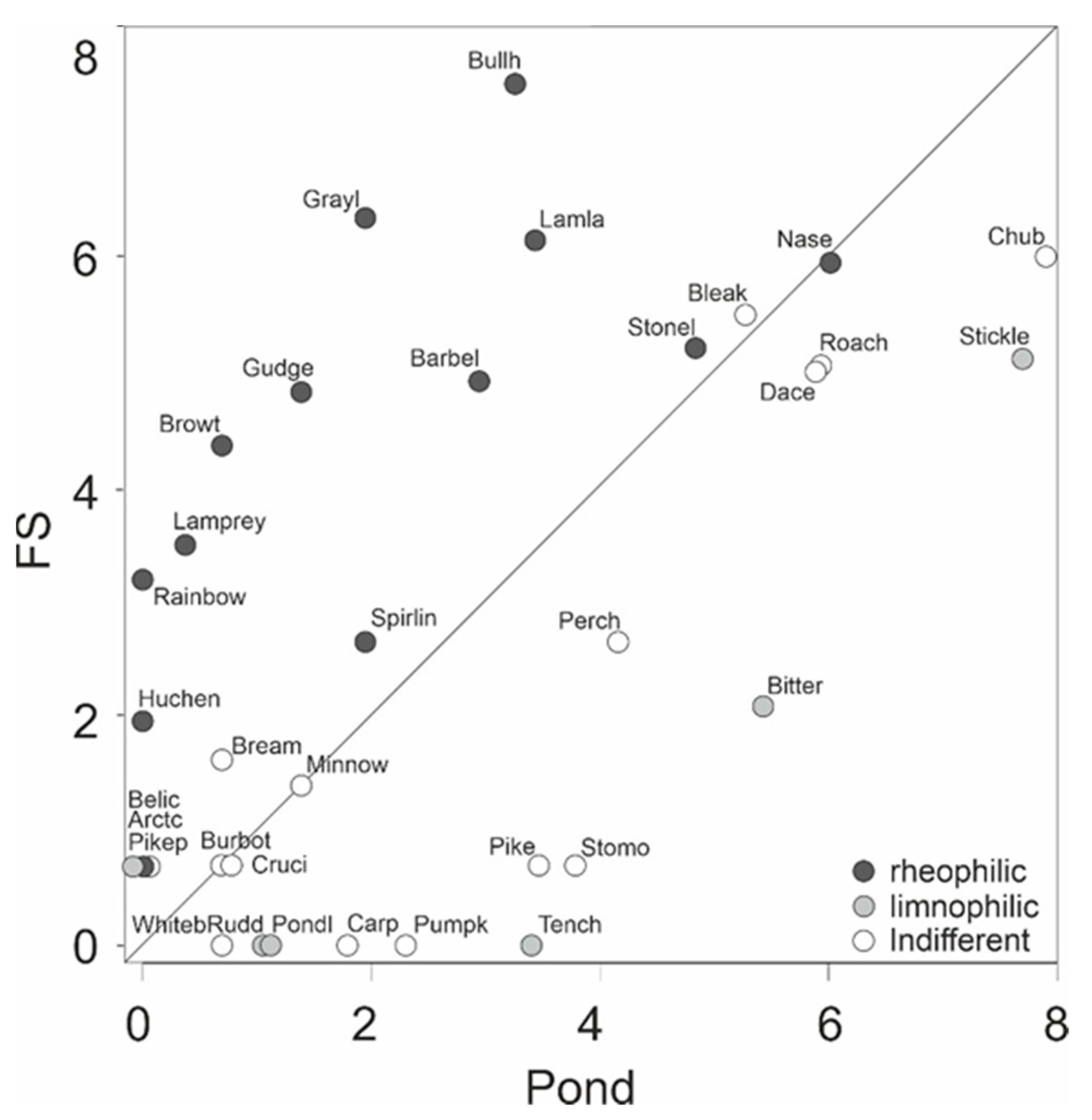
Blicca bjoerkna L., were exclusively detected in ponds (overall number of species here 26), whilst 6 mostly rheophilic species (e.g.,
Salvelinus fontinalis Mitchill,
Gobio gobio L.,
O. mykiss,
H. Hucho) were exclusively detected in fluvial habitats (28 species) of the FP (
Figure 5,
Table 6).
C. gobio (99% individuals),
S. trutta (99%),
T. thymallus (99%),
B. barbus (95%), and Petromyzontidae (97%) were almost exclusively detected in the fluvial habitats, and
R. amarus, as well as
Pseudorasbora parva Temmink & Schlegel, almost exclusively in ponds (
Figure 5,
Table 6).
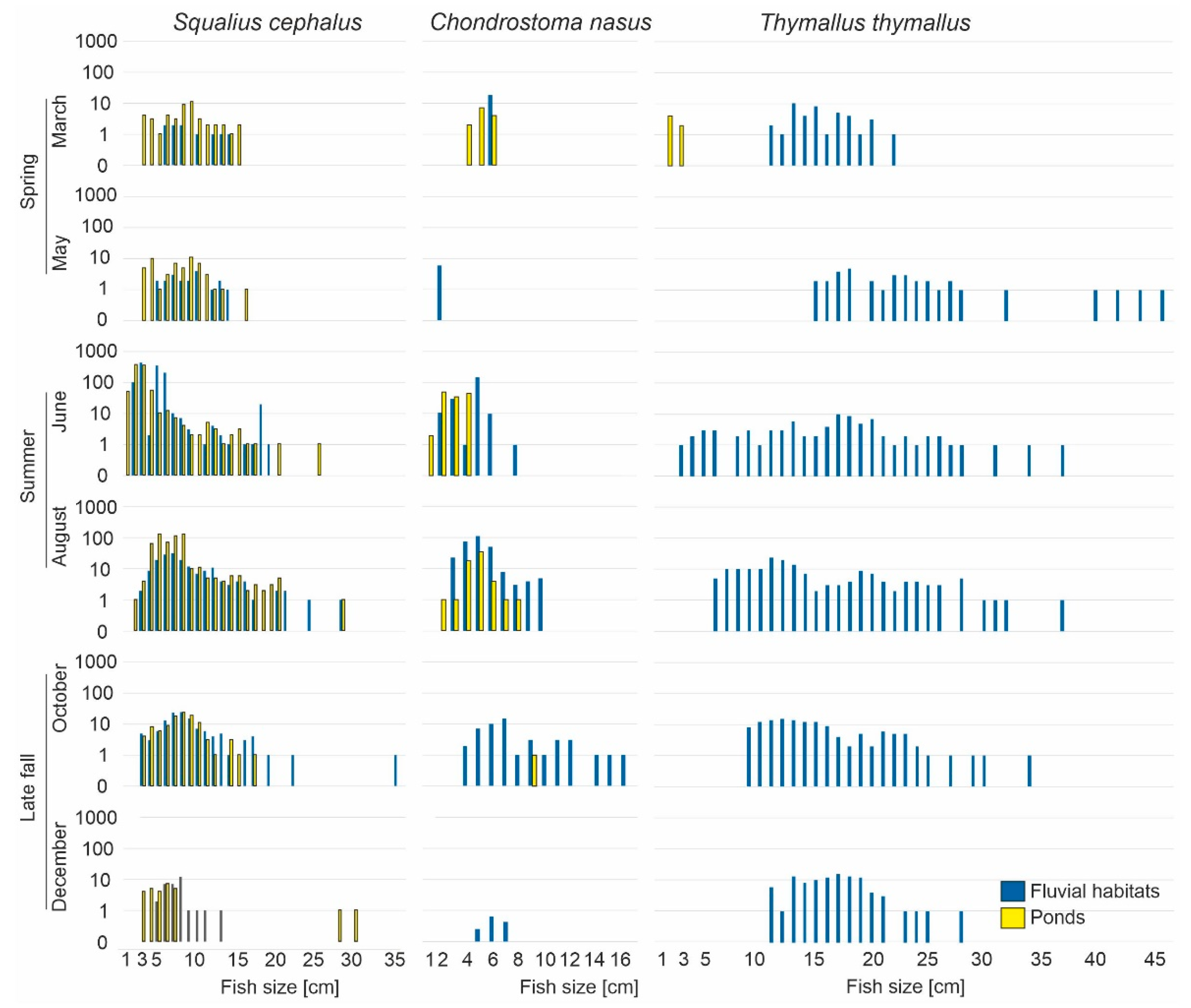
However, it has to be noted that the abundance of species in fluvial habitats or ponds was strongly governed by seasonal effects (
Figure 6). This was particularly true for species that are known to switch habitats during different stages of their life cycle, such as
C. nasus. This species was very abundant in ponds during summer and disappeared almost entirely from this habitat in late fall (
Figure 6 and
Figure 7). In contrast, other species, like
S. cephalus, used ponds and fluvial habitats equally throughout the year in almost all size classes (
Figure 7), whilst
R. amarus and
P. parva only used ponds comprising a population structure, including all size classes. In contrast, all size classes of the reophilic species
T. thymallus were exclusively found in fluvial habitats. LM detected that species richness in the ponds was significantly influenced by the factors macrophyte coverage (
p < 0.05), deadwood coverage (
p < 0.01) and water depth (
p < 0.05). For all detections of significances, the relationship between the variable and the response species richness was not linear with highest species diversity for macrophyte coverage at 75%, deadwood coverage between 5% and 10%, and water depth about 0.5 m to 1.0 m.
4. Discussion
The findings of this study confirm the positive contribution of diverse habitats within constructed fish passes to the fish diversity of the River Inn. Fish diversity within the fish passes (33 species) largely resembled the species inventory of the River Inn (36 species), except for some rare species, such as
R. vladykovi, that also occur at low densities in the main river [
30]. Still, there were major differences in abundances of species between the fish passes and the main river.
As demonstrated herein, nature- like fish passes of high habitat diversity, comprising both fluvial habitats and stagnant waters, can at least partially contribute to the restoration of the formerly present habitat mosaic in this alpine river. In such large degraded alpine systems as the River Inn, the restoration of river dynamic processes, which govern high habitat and species diversity, is almost impossible due to the many restrictions in densely populated areas. From a fish conservation point of view, this makes near-natural watercourses that can at least partially fulfil these functions most valuable.
As expected, fluvial habitats within the fish passes comprised a rheophilic-dominated fish community, whilst ponds were largely dominated by stagnophilic fish species, or by species with indifferent current preferences. Consequently, both habitats contributed to a high species diversity in that system. However, it has to be considered that the fish community sampling in these small structures within a fish pass is likely more effective than in the larger main stem [
46], potentially resulting in a bias towards an underestimation of species richness in the River Inn. This is also in compliance with the findings from Mollenhauer et al. [
47], who demonstrated that fish detection probability can be affected by water depth and turbidity.
The populations of species, such as
T. thymallus,
B. barbus, and
C. nasus, are under pressure in almost all rivers across their distribution range, particularly in the densest populated areas of central Europe, with their names on national and international red lists alike [
43]. This makes them prominent target species for conservation. The fact that
T. thymallus was detected in all size classes and that
C. nasus and
B. barbus were present in their most sensitive small size classes indicate that fluvial habitats and ponds in nature-like fish passes can provide important juvenile habitats, which are scarce in the main river. This should encourage river managers to follow the pathway of creating additional fluvial habitats and small ponds adjacent to the heavily modified and exploited main channel of rivers such as the Inn. The presence of early life stages of those target species and their seasonal habitat use, as detected herein for
C. nasus, indicates that, besides habitat quality, connectivity between the source and all other habitats relevant for subsequent life stages is mandatory. A high degree of connectivity is particularly crucial when species have to change habitats for spawning or during larval drift. Thereby, it is crucial to match the timing of, e.g., spawning season [
48,
49] or emergence, with the accessibility of those life stage specific habitats. This is particularly important if species during their ontogeny change food sources, which happens usually simultaneously with a shift in habitat requirements. Such a situation is typical for
C. nasus [
50,
51], which obviously moved seasonally between Inn, fluvial habitats in the fish passes and the ponds in our study. Other species may follow their food sources into the habitats, such as the apex predator
H. hucho [
52], or use the fish pass for spawning (
T. thymallus), as previously proposed by Nagel et al. [
53].
Ponds contributed to fish diversity by providing habitat for additional species, which prefer stagnant water conditions, such as
Tinca tinca L.,
S. erythrophthalmus, the strictly protected
R. amarus [
43,
44], or the invasive
P. parva [
54], and
M. bipartitus [
55]. Whilst the presence of additional species, such as
R. amarus, can be accomplishable, the occurrence of
M. bipartitus is not always desired since it is known to be an invasive species potentially replacing the indigenous
Misgurnus fossilis L. [
55,
56]. In addition to the contribution to the overall diversity of the FP that was highest during the summer months, ponds provided habitat for species of high conservation value which has previously received little attention. This peak in diversity during summer can likely be attributed to higher structural density caused by macrophytes and bank vegetation, as well as more available habitat area, due to higher water levels, particularly in the ponds of fish pass Gars, where, during summer, the e-flow regime is applied. Besides the small life stages of the rheophilic
C. nasus, the stagnophilic
R. amarus [
45] had peak densities in these ponds during summer. Usually, in braided high-energy rivers, like the River Inn, its distribution range is restricted to backwaters and small floodplain ponds [
57], which all underlie strong river dynamic processes of habitat change [
58,
59,
60]. In the historically (early 1960s) highly altered River Inn, the remaining backwaters have meanwhile undergone a successional development peaking often in a terminal phase. Some of those backwaters shifted towards terrestrial habitats, and others are being used for recreational purposes, such as angling or bathing lakes. Due to the restricted river dynamic processes, which consistently would have created new backwaters and floodplain ponds, such habitats are today widely lacking. Therefore, the construction of such structures can be important. However, it has to be noted that also artificially created floodplain ponds undergo rapid successional changes, which need to be mitigated in order to preserve high species richness. Particularly macrophyte coverage increased rapidly in the rather shallow ponds since their construction, restricting habitat area for at least some species. This creates the need for a partial maintenance by clear-cutting when macrophyte coverage exceeds 75% of pond area on a regular basis.
The rich species inventory in the system must not obscure the fact that fish community composition in the highly degraded River Inn of today comprises a reduced set of riverine specialists that typically dominated the fish community in the past, originally comprising historically widespread and now undetected species, such as
Zingel streber Siebold and
Zingel zingel L. [
30]. This underscores the finding that as good as the habitat quality of a fish pass might be, the species inventory largely depends on the source and if the source habitat is severely altered, an ecological integer fish community composition in restored areas or fish passes might be limited [
27]. To date, it is not fully clear if the smaller size and discharge of a fish pass compared to the main river can still provide a suitable compensatory habitat, since it has not yet been proven if all riverine species can benefit from fish passes as permanent habitat or at least habitat for certain life stages. For example,
B. barbus,
Z. zingel, and
Z. streber are known to prefer the strong flowing main current of rivers in their adult stages and only use riverbanks during early ontogeny [
61,
62]. In our study,
B. barbus was present in the fish passes but only in small size-classes.
Z. zingel and
Z. streber could not be detected at all due to the earlier mentioned reasons for habitat degradation in the main river. If these rare species will respond positively to the installation of nature-like fish passes, they can only be determined on a long-term basis because it needs some time for them to reach a critical population size that can be detected reliably by fish population assessments (e.g., [
18,
63]).
5. Conclusions
This study underlines the important habitat role of fish passes, which can largely contribute to overall fish diversity in rivers, comprising typically rheophilic species in fluvial habitats and stagnophilic species in stagnant waters, such as the created floodplain ponds. Both, rheophilc and stagnophilic, species of high conservation value, such as, e.g., T. thymallus, C. nasus, B. barbus, H. hucho, and R. amarus, are rare in the main river. Some of those species, e.g., T. thymallus, even occurred in large numbers across all size classes in the fish passes, suggesting that fish passes can host a complete population of this species. Other species, which continue declining in the main river, were present in high numbers of small life stages in fluvial habitats, as well as ponds, indicating potential functionality of both habitat types as juvenile habitats. Provided regular maintenance to keep the small floodplain ponds open, high species diversity can be achieved in combination with fish passes.