Variation of Diatoms at Different Scales in the Brazilian Pantanal Basin
Abstract
1. Introduction
- (1)
- Do communities differ more than expected at random levels between local Pantanal habitats (i.e., ponds), or the difference is just a mathematical artifact?
- (2)
- If differences exist, which species characterize these differences? Does the different species composition a consequence of the environmental characteristics of the ponds?
- (3)
- Is there a homogeneous or heterogeneous spatial composition in the ponds?
- (4)
- Which is the scale that most contributes to the total diversity of diatoms: the samples made at each site, the compositional variation within the pond, or the compositional variation between different ponds? Which of these contributions are the greater, and which are less than expected by a null model?
2. Materials and Methods
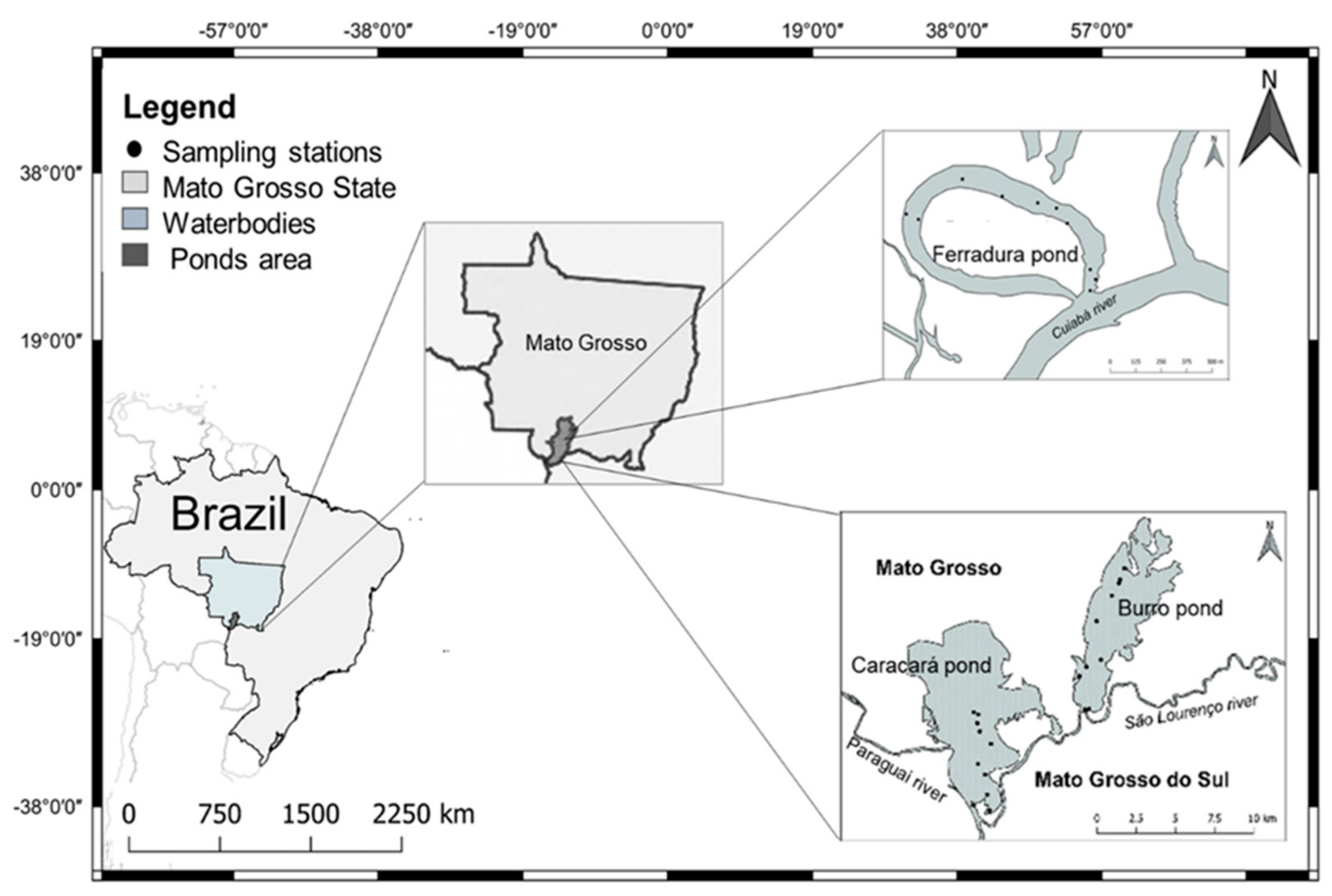
2.1. Study Area
- -
- Ferradura Pond (P1) connects with the Cuiabá River; has an average width of 300 m, with an approximate length of 1200 m; the pond depth is of 270–650 cm; is located 107 km from the city of Cuiabá. The Cuiabá River is impacted by the discharge of sewage, as well as by the fish farming tanks that discharge their effluents with high levels of nitrogen [29].
- -
- Burro Pond (P2) connects with the São Lourenço river; has an average width of 1000 m and 5000 m in length and a depth of 140–280 cm; is located 173 km from P1. The São Lourenço River is influenced by agricultural activities and also by heavy metals—mainly mercury due to gold mining activities [29].
- -
- Caracará Pond (P3) connects with the Paraguay River, has 3000 m width and 3600 m length, has a depth of 120–290 cm, is located 7 km from P2. The Paraguay River is influenced by agricultural, livestock and heavy metals activities—mainly mercury due to gold mining activities [29]. Sampling locations, geographical coordinates of the three ponds listed in Table 1.
2.2. Experimental Design
2.3. Diatoms
2.4. Physical and Chemical Variables
2.5. Data Analysis
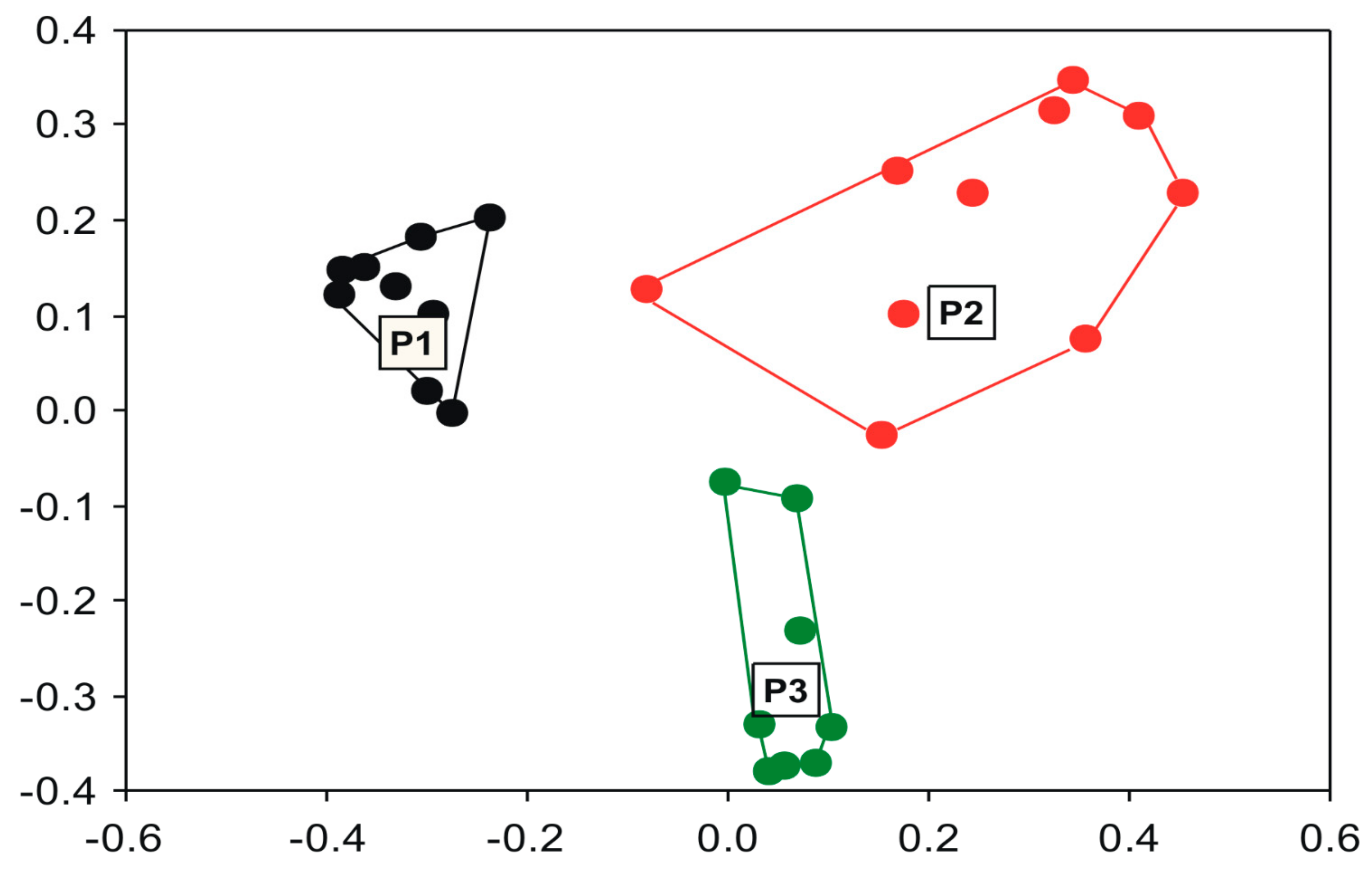
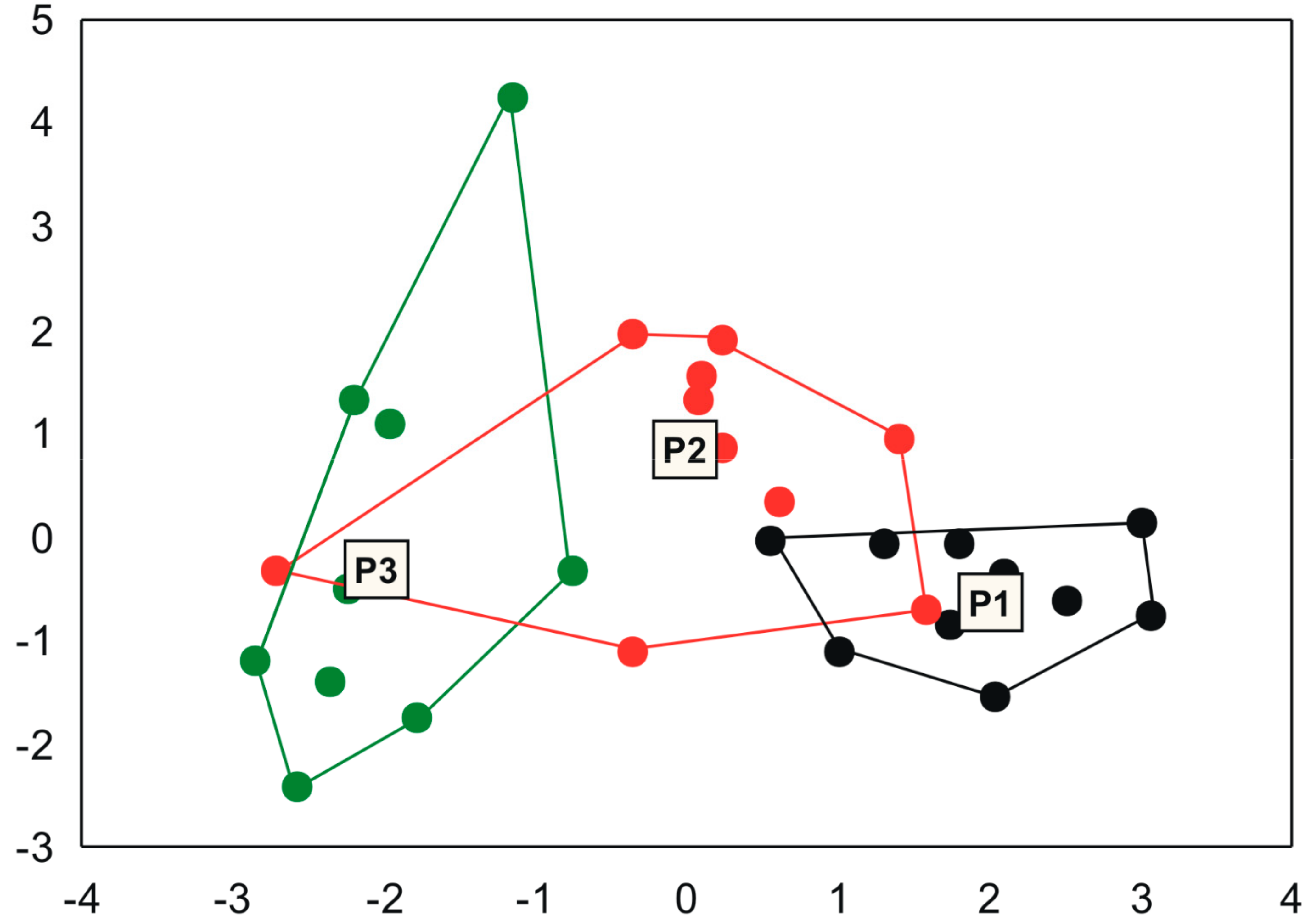
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leibold, M.A.; Norberg, J. Biodiversity in metacommunities, Plankton as complex adaptive systems? Limnol. Oceanogr. 2004, 49, 1278–1289. [Google Scholar] [CrossRef]
- Algarte, V.M.; Siqueira, N.S.; Murakami, E.A.; Rodrigues, L. Effects of hydrological regime and connectivity on the interannual variation in taxonomic similarity of periphytic algae. Braz. J. Biol. 2009, 69, 609–616. [Google Scholar] [CrossRef]
- Naselli-Flores, L.; Termine, R.; Barone, R. Phytoplankton colonization patterns. Is species richness depending on distance among freshwaters and on their connectivity? Hydrobiologia 2015, 764, 103–113. [Google Scholar] [CrossRef]
- Zorzal-Almeida, S.; Soininen, J.; Bini, L.M.; Bicudo, D.C. Local environment and connectivity are the main drivers of diatom species composition and trait variation in a set of tropical reservoirs. Freshw. Biol. 2017, 62, 1551–1563. [Google Scholar] [CrossRef]
- Hubbell, S.P. The neutral theory of biodiversity and biogeography, and Stephen Jay Gould. Paleobiology 2005, 31, 122–132. [Google Scholar] [CrossRef]
- Holyoak, M.; Leibold, M.A.; Holt, R.D. Metacommunities, Spatial Dynamics and Ecological Communities, 1st ed.; Hoopes, M.F., Holt, R.D., Holyoak, M., Eds.; The University of Chicago Press: Chicago, IL, USA, 2005. [Google Scholar]
- Algarte, V.M.; Siqueira, T.; Landeiro, V.L.; Rodrigues, L.; Bonecker, C.C.; Rodrigues, L.C.; Santana, N.F.; Thomaz, S.M.; Bini, L.M. Main predictors of periphyton species richness depend on adherence strategy and cell size. PLoS ONE 2017, 12, e181720. [Google Scholar] [CrossRef]
- Thompson, P.L.; Guzman, L.M.; De Meester, L.; Horváth, Z.; Ptacnik, R.; Vanschoenwinkel, B.; Viana, D.S.; Chase, J.M. A process-based metacommunity framework linking local and regional scale community ecology. Ecol. Lett. 2020, 23, 1314–1329. [Google Scholar] [CrossRef]
- Crump, B.C.; Adams, H.E.; Hobbie, J.E.; Kling, G.W. Biogeography of bacterioplankton in lakes and streams of an arctic tundra catchment. Ecology 2007, 88, 1365–1378. [Google Scholar] [CrossRef]
- Soininen, J. Environmental and spatial control of freshwater diatoms—A Review. Diatom Res. 2007, 22, 473–490. [Google Scholar] [CrossRef]
- Verleyen, E.; Vyverman, W.; Sterken, M.; Hodgson, D.A.; De Wever, A.; Juggins, S.; Van de Vijver, B.; Jones, V.J.; Vanormelingen, P.; Roberts, D.; et al. The importance of dispersal related and local factors in shaping the taxonomic structure of diatom metacommunities. Oikos 2009, 118, 1239–1249. [Google Scholar] [CrossRef]
- Padial, A.A.; Ceschin, F.; Declerck, S.A.J.; De Meester, L.; Bonecker, C.C.; Lansac-Tôha, F.A.; Rodrigues, L.; Rodrigues, L.C.; Train, S.; Velho, L.F.M.; et al. Dispersal ability determines the role of environmental, spatial and temporal drivers of metacommunity structure. PLoS ONE 2014, 9, e111227. [Google Scholar] [CrossRef]
- Vilmi, A.; Tolonen, K.T.; Karjalainen, S.M.; Heino, J. Metacommunity structuring in a highly-connected aquatic system, Effects of dispersal, abiotic environment and grazing pressure on microalgal guilds. Hydrobiologia 2017, 790, 125–140. [Google Scholar] [CrossRef]
- Leland, H.V.; Porter, S.D. Distribution of benthic algae in the upper Illinois River basin in relation to geology and land use. Freshw. Biol. 2000, 44, 279–301. [Google Scholar] [CrossRef]
- Pommier, T.; Canbäck, B.; Riemann, L.; Boström, K.H.; Simu, K.; Lundberg, P.; Tunlid, A.; Hagström, A. Global patterns of diversity and community structure in marine bacterioplankton. Mol. Ecol. 2007, 16, 867–880. [Google Scholar] [CrossRef] [PubMed]
- Logue, J.B.; Mouquet, N.; Peter, H.; Hillebrand, H. Empirical approaches to metacommunities, A review and comparison with theory. Trends Ecol. Evol. 2011, 26, 482–491. [Google Scholar] [CrossRef]
- Astorga, A.; Oksanen, J.; Luoto, M.; Soininen, J.; Virtanen, R.; Muotka, T. Distance decay of similarity in freshwater communities, Do macro- and microorganisms follow the same rules? Glob. Ecol. Biogeogr. 2012, 21, 365–375. [Google Scholar] [CrossRef]
- Heino, J.; Melo, A.S.; Bini, L.M. Reconceptualising the beta diversity-environmental heterogeneity relationship in running water systems. Freshw. Biol. 2015. [Google Scholar] [CrossRef]
- Dong, X.; Li, B.; He, F.; Gu, Y.; Sun, M.; Zhang, H.; Tan, L.; Xiao, W.; Liu, S.; Cai, Q. Flow directionality, mountain barriers and functional traits determine diatom metacommunity structuring of high mountain streams. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Keck, F.; Franc, A.; Kahlert, M. Disentangling the processes driving the biogeography of freshwater diatoms, A multiscale approach. J. Biogeogr. 2018, 45, 1582–1592. [Google Scholar] [CrossRef]
- Keck, F.; Kahlert, M. Community phylogenetic structure reveals the imprint of dispersal-related dynamics and environmental filtering by nutrient availability in freshwater diatoms. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lobo, E.A.; Heinrich, C.G.; Schuch, M.; Düpont, A.; da Costa, A.B.; Wetzel, C.E.; Ector, L. Índice Trófico de Qualidade da Água, Guia Ilustrado Para Sistemas Lóticos Subtropicais e Temperados Brasileiros, 1st ed.; Lobo, E., Ed.; Acta Universitatis Agriculturae et Silviculturae Mendelianae Brunensis: Santa Cruz do Sul, Edunisc, Brazil, 2016; Available online: http://publications.lib.chalmers.se/records/fulltext/245180/245180.pdf%0Ahttps://hdl.handle.net/20.500.12380/245180%0Ahttp://dx.doi.org/10.1016/j.jsames.2011.03.003%0Ahttps://doi.org/10.1016/j.gr.2017.08.001%0Ahttp://dx.doi.org/10.1016/j.precamres.2014.12 (accessed on 14 April 2016).
- Smol, J.P.; Stoermer, E.F. The Diatoms, Applications for the Environmental and Earth Sciences, 2nd ed.; Cambridge University Press: New york, NY, USA, 2010; ISBN 978-0-521-50996-1. [Google Scholar]
- Soininen, J. Local and regional coexistence of diatoms—on the mechanisms promoting high local diatom species richness. Diatom Res. 2006, 21, 217–223. [Google Scholar] [CrossRef]
- Soininen, J.; Jamoneau, A.; Rosebery, J.; Passy, S.I. Global patterns and drivers of species and trait composition in diatoms. Glob. Ecol. Biogeogr. 2016, 25. [Google Scholar] [CrossRef]
- Remmer, C.R.; Robichaud, C.D.; Polowyk, H.; Rooney, R. The role of ecological drift in structuring periphytic diatom communities. J. Freshw. Ecol. 2019, 34, 363–377. [Google Scholar] [CrossRef]
- Soininen, J.; Teittinen, A. Fifteen important questions in the spatial ecology of diatoms. Freshw. Biol. 2019, 64, 2071–2083. [Google Scholar] [CrossRef]
- De Oliveira, M.D.; Calheiros, D.F.; Padovani, C.R. Mapeamento e Descrição das Áreas de Ocorrência dos Eventos de Decoada no Pantanal, 1st ed.; de Salis, S.M., Ed.; Corumbá, Embrapa Pantanal: Mato Grosso do sul, Brazil, 2013; Available online: https://www.infoteca.cnptia.embrapa.br/infoteca/handle/doc/958791 (accessed on 10 August 2016).
- Tabarelli, M.; da Rocha, C.F.D.; Romanowski, H.P.; Rocha, O.; de Lacerda, L.D. PELD–CNPq Dez Anos do Programa de Pesquisas Ecológicas de Longa Duração no Brasil, Achados, Lições e Perspectivas; Tabarelli, M., da Rocha, C.F.D., Romanowski, H.P., Rocha, O., de Lacerda, L.D., Eds.; Recife, Editora Universitária UFPE: Recife, Brazil, 2013. [Google Scholar]
- Assine, M.L.; Merino, E.R.; do Nascimento Pupim, F.; de Azevedo Macedo, H.; dos Santos, M.G.M.; Assine, M.L. The Quaternary alluvial systems tract of the Pantanal Basin, Brazil. Braz. J. Geol. 2015, 45, 475–489. [Google Scholar] [CrossRef]
- Fontaneto, D.; Brodie, J. Why Biogeography of Microorganisms? Biogeography of Microscopic Organisms Is Everything Small Everywhere? Academic Press: Cambridge, MA, USA, 2011; pp. 3–10. [Google Scholar] [CrossRef]
- Incagnone, G.; Marrone, F.; Barone, R.; Robba, L.; Naselli-Flores, L. How do freshwater organisms cross the “dry ocean”? A review on passive dispersal and colonization processes with a special focus on temporary ponds. Hydrobiologia 2015, 750, 103–123. [Google Scholar] [CrossRef]
- Padisák, J.; Vasas, G.; Borics, G. Phycogeography of freshwater phytoplankton, Traditional knowledge and new molecular tools. Hydrobiologia 2015, 764, 3–27. [Google Scholar] [CrossRef]
- Cox, C.B.; Moore, P.D.; Ladle, R.J. Biogeography, an Ecological and Evolutionary Approach, 9th ed.; John Wiley & Sons: Chichester, UK; Hoboken, NJ, USA, 2016. Available online: http://lccn.loc.gov/2016000665 (accessed on 1 February 2021).
- Jackson, D.A.; Peres-Neto, P.R.; Olden, J.D. What controls who is where in freshwater fish communities—The roles of biotic, abiotic, and spatial factors. Can. J. Fish. Aquat. Sci. 2001, 58, 157–170. [Google Scholar] [CrossRef]
- Da Silva, L.P.B.; Hussein, H. Production of scale in regional hydropolitics, An analysis of La Plata River Basin and the Guarani Aquifer System in South America. Geoforum 2019, 99, 42–53. [Google Scholar] [CrossRef]
- Hussein, H. The Guarani Aquifer System, highly present but not high profile, A hydropolitical analysis of transboundary groundwater governance. Environ. Sci. Policy 2018, 83, 54–62. [Google Scholar] [CrossRef]
- Andrade, R.G.; Sediyama, G.C.; da Paz, A.R.; de Paiva Lima, E.; Facco, A.G. Geotecnologias aplicadas á avaliação de parametros biofísicos do Pantanal. Pesqui. Agropecu. Bras. 2012, 47, 1227–1234. [Google Scholar] [CrossRef]
- Viana, D.R.; dos Alvalá, R.C.S. Vegetation Index Performance for the Pantanal Region During Both Dry and Rainy Seasons. Geografia 2011, 36, 143–158. [Google Scholar]
- Battarbee, R.W.; Jones, V.J.; Birks, H.J.B.; Last, W.M. Diatom In Rastreando Mudança Ambiental Usando Sedimentos Lake, 3rd ed.; Smol, J.P., Birks, H.J.B., Last, W., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; pp. 155–202. [Google Scholar]
- Round, F.E.; Crawford, R.M.; Mann, D.G. The Diatoms, 4th ed.; Round, F.E., Crawford, R.M., Mann, D.G., Eds.; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Lecointe, C.; Coste, M.; Prygiel, J. “Omnidia”, Software for taxonomy, calculation of diatom indices and inventories management. Hydrobiologia 1993, 269–270, 509–513. [Google Scholar] [CrossRef]
- Valderrama, J.C. The simultaneous analysis of total nitrogen and total phosphorus in natural waters. Mar. Chem. 1981, 10, 109–122. [Google Scholar] [CrossRef]
- Marker, A.F.H. Chlorophyll a SCA Method Revision, 1st ed.; National Rivers Authority: Bristol, UK, 1994. [Google Scholar]
- Lamparelli, M.C. Graus de Trofia em Corpos D’água do Estado de São Paulo, Avaliação dos Métodos de Monitoramento; Universidade de São Paulo; Tese em Ciências da área de Ecossistema Terrestres e Aquáticos, Departamento de Ecologia: São Paulo, Brazil, 2004. [Google Scholar] [CrossRef]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Gower, J.C. Some distance properties of latent root and vector methods used in multivariate analysis. Biometrika 1966, 53, 325. [Google Scholar] [CrossRef]
- Dufrêne, M.; Legendre, P. species assemblages and indicator species, the need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]
- Anderson, M.J.; Ellingsen, K.E.; McArdle, B.H. Multivariate dispersion as a measure of beta diversity. Ecol. Lett. 2006, 9, 683–693. [Google Scholar] [CrossRef]
- Mantel, N. The Detection of Disease Clustering and a Generalized Regression Approach. Cancer Res. 1967, 27, 209–220. [Google Scholar] [CrossRef][Green Version]
- Crist, T.O.; Veech, J.A.; Gering, J.C.; Summerville, K.S. Partitioning Species Diversity across Landscapes and Regions, A Hierarchical Analysis of α, β, and γ Diversity. Am. Nat. 2003, 162, 734–743. [Google Scholar] [CrossRef]
- Leibold, M.A.; Mikkelson, G.M. Coherence, species turnover, and boundary clumping, Elements of meta-community structure. Oikos 2002, 97, 237–250. [Google Scholar] [CrossRef]
- Tudesque, L.; Pool, T.K.; Chevalier, M. Planktonic diatom community dynamics in a tropical flood-pulse lake, The Tonle Sap (Cambodia). Diatom Res. 2019, 34, 1–22. [Google Scholar] [CrossRef]
- Lindholm, M.; Grönroos, M.; Hjort, J.; Karjalainen, S.M.; Tokola, L.; Heino, J. Different species trait groups of stream diatoms show divergent responses to spatial and environmental factors in a subarctic drainage basin. Hydrobiologia 2018, 816, 213–230. [Google Scholar] [CrossRef]
- Paes, E.T.; Blinder, P.B. Modelos nulos e processos de aleatorização, algumas aplicações em ecologia de comunidades. Oecologia Bras. 1995, 2, 119–139. [Google Scholar] [CrossRef]
- Shurin, J.B. Dispersal Limitation, Invasion Resistance, and the Structure of Pond Zooplankton Communities. Ecology 2000, 81, 3074–3086. [Google Scholar] [CrossRef]
- Moro, R.S.; Furstenberger, C.B. (Eds.) Catálogo dos prinCipais Parâmetros Ecológicos de Diatomáceas Não-Marinhas; UEPG-Universidade Estadual de Ponta Grossa: Ponta Grossa, Brazil, 1997. [Google Scholar]
- Ferrari, F.; Ludwig, T.A.V. Coscinodiscophyceae, Fragilariophyceae e Bacillariophyceae (Achnanthales) dos rios Ivaí, São João e dos Patos, bacia hidrográfica do rio Ivaí, município de Prudentópolis, Brasil. Acta Bot. Bras. 2007, 21, 421–441. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Q.; Fu, C. Taxonomy and distribution of diatoms in the genus Eunotia from the Da’erbin Lake and Surrounding Bogs in the Great Xing’an Mountains, China. Nova Hedwig. 2011, 92, 205–232. [Google Scholar] [CrossRef]
- Costa, L.F.; Wetzel, C.E.; Lange-Bertalot, H.; Ector, L.; Bicudo, D.C. Taxonomy and Ecology of Eunotia Species (Bacillariophyta) in Southeastern Brazilian Reservoirs; Costa, L.F., Wetzel, C.E., Lange-Bertalot, H., Ector, L., Bicudo, D.C., Eds.; Schweizerbart Science Publishers: Stuttgart, Germany, 2017. [Google Scholar]
- Siver, P.A.; Kling, H. Morphological observations of Aulacoseira using scanning electron microscopy. Can. J. Bot. Rev. Can. Bot. 1997, 75, 1807–1835. [Google Scholar] [CrossRef]
- Crawford, R.M.; Likhoshway, Y.V.; Jahn, R. Morphology and Identity of Aulacoseira Italica and Typification of Aulacoseira (Bacillariophyta). Diatom Res. 2003, 18, 1–19. [Google Scholar] [CrossRef]
- Nardelli, M.S.; Bueno, N.C.; Ludwig, T.A.V.; Guimarães, A.T.B. Structure and dynamics of the planktonic diatom community in the Iguassu River, Paraná State, Brazil. Braz. J. Biol. 2016, 76, 374–386. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, N.; Marins, M.A.; Tundisi, J.G. Synchronous growth of a freshwater diatom Melosira italica under natural environment. Oecologia 1976, 23, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L. Contribuição ao Connhecimento das Diatómaceas, Do Rio Tubarão-Santa Catarina-Brasil. Ínsula 1984, 14, 47–120. [Google Scholar]
- Bicudo, D.C.; Tremarin, P.I.; Almeida, P.D.; Zorzal-Almeida, S.; Wengrat, S.; Faustino, S.B. Ecology and distribution of Aulacoseira species (Bacillariophyta) in tropical reservoirs from Brazil. Diatom Res. 2016, 31, 199–215. [Google Scholar] [CrossRef]
- Shmida, A.; Wilson, M.V. Biological determinants of species diversity. J. Biogeogr. 1985, 12, 1–20. [Google Scholar] [CrossRef]
- Holt, R.D.; Barfield, M.; Gonzalez, A. Impacts of environmental variability in open populations and communities, “inflation” in sink environments. Theor. Popul. Biol. 2003, 64, 315–330. [Google Scholar] [CrossRef]
- Chen, G.; Dalton, C.; Leira, M.; Taylor, D. Diatom-based total phosphorus (TP) and pH transfer functions for the Irish Ecoregion. J. Paleolimnol. 2008, 40, 143–163. [Google Scholar] [CrossRef]
- Houk, V.; Klee, R. Atlas of freshwater centric diatoms with a brief key and descriptions, Part II Melosiraceae and Aulacoseiraceae. Fottea 2007, 7, 85–255. [Google Scholar]
- Taylor, J.C.; Harding, W.R.; Archibald, C.G.M. An Illustrated Guide to Some Common Diatom Species from South Africa; Report to the Water Research Commission: Brussels, Belgium, 2007. [Google Scholar]
- Tuji, A.; Williams, D.M. Type Examination of Japanese Diatoms Described by Friedrich Meister (1913) from Lake Suwa. Bull. Natl. Sci. Mus. Ser. B 2007, 33, 69–79. [Google Scholar]
- Tremarin, P.I.; Ludwig, T.A.V.; Torgan, L.C. Four new Aulacoseira species (Coscinodiscophyceae) from Matogrossense Pantanal, Brazil. Diatom Res. 2014, 29, 183–199. [Google Scholar] [CrossRef]
- Cermeño, P.; Falkowski, P.G. Controls on diatom biogeography in the ocean. Science 2009. [Google Scholar] [CrossRef]
- Wojciechowski, J.; Heino, J.; Bini, L.M.; Padial, A.A. Temporal variation in phytoplankton beta diversity patterns and metacommunity structures across subtropical reservoirs. Freshw. Biol. 2017, 62, 751–766. [Google Scholar] [CrossRef]
- Chust, G.; Irigoien, X.; Chave, J.; Harris, R.P. Latitudinal phytoplankton distribution and the neutral theory of biodiversity. Glob. Ecol. Biogeogr. 2012, 22, 531–543. [Google Scholar] [CrossRef]
- Leibold, M.A.; Holyoak, M.; Mouquet, N.; Amarasekare, P.; Chase, J.M.; Hoopes, M.F.; Holt, R.D.; Shurin, J.B.; Law, R.; Tilman, D.; et al. The metacommunity concept, A framework for multi-scale community ecology. Ecol. Lett. 2004, 7, 601–613. [Google Scholar] [CrossRef]
- Leibold, M.A. Ecology, return of the niche. Nature 2008, 454, 39–41. [Google Scholar] [CrossRef]
- Howeth, J.G.; Leibold, M.A. Species dispersal rates alter diversity and ecosystem stability in pond metacommunities. Ecology 2010, 91, 2727–2741. [Google Scholar] [CrossRef]
- Loreau, M.; Mouquet, N.; Gonzalez, A. Biodiversity as spatial insurance in heterogeneous landscapes. Proc. Natl. Acad. Sci. USA 2003, 100, 12765–12770. [Google Scholar] [CrossRef] [PubMed]
- Butchart, S.; Dieme-Amting, E.; Gitay, H.; Raaymakers, S.; Taylor, D. Ecosystems Wetlands Well-Being, And Human and Water-Synthesis; Sarukhán, J., Whyte, A., Board, M., Eds.; Green Chemistry Letters and Reviews; World Resources Institute: Washington, DC, USA, 2005. [Google Scholar] [CrossRef]
| * Ferradura Pond (P1) | * Burro Pond (P2) | * Caracará Pond (P3) | ||||||
|---|---|---|---|---|---|---|---|---|
| Site | Latitude | Longitude | Site | Latitude | Longitude | Site | Latitude | Longitude |
| 1 | 16°31′35″ S | 56°23′26″ O | 11 | 17°50′24″ S | 57°23′53″ O | 21 | 17°53′32″ S | 57°27′55″ O |
| 2 | 16°31′34″ S | 56°23′25″ O | 12 | 17°50′22″ S | 57°23′44″ O | 22 | 17°53′42″ S | 57°27′19″ O |
| 3 | 16°31′32″ S | 56°23′26″ O | 13 | 17°49′18″ S | 57°24′05″ O | 23 | 17°53′11″ S | 57°27′25″ O |
| 4 | 16°31′29″ S | 56°23′26″ O | 14 | 17°49′00″ S | 57°23′49″ O | 24 | 17°52′32″ S | 57°27′29″ O |
| 5 | 16°31′23″ S | 56°23′32″ O | 15 | 17°48′46″ S | 57°23′18″ O | 25 | 17°52′11″ S | 57°27′45″ O |
| 6 | 16°31′22″ S | 56°23′35″ O | 16 | 17°47′30″ S | 57°23′28″ O | 26 | 17°51′32″ S | 57°27′17″ O |
| 7 | 16°31′21″ S | 56°23′41″ O | 17 | 17°46′40″ S | 57°22′55″ O | 27 | 17°51′07″ S | 57°27′40″ O |
| 8 | 16°31′19″ S | 56°23′47″ O | 18 | 17°46′16″ S | 57°22′39″ O | 28 | 17°50′51″ S | 57°27′46″ O |
| 9 | 16°31′24″ S | 56°23′57″ O | 19 | 17°46′09″ S | 57°22′37″ O | 29 | 17°50′34″ S | 57°27′44″ O |
| 10 | 16°31′25″ S | 56°23′55″ O | 20 | 17°45′47″ S | 57°22′28″ O | 30 | 17°50′29″ S | 57°27′54″ O |
| Index | Ferradura Pond | Burro Pond | Caracará Pond |
|---|---|---|---|
| Species richness | 83 | 72 | 78 |
| Simpson 1-D | 0.810 | 0.923 | 0.902 |
| Shannon H | 2.580 | 3.126 | 3.099 |
| Menhinick | 2.626 | 2.276 | 2.466 |
| Fisher-alpha | 21.500 | 17.790 | 19.780 |
| Berger–Parker | 0.397 | 0.189 | 0.205 |
| Site | Depth | T °C | pH | TDS ug·L−1 | Cond μS·cm−1 | TU | N ug·L−1 | P ug·L−1 | DO | TSI | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| P1 | Min | 320 | 28.2 | 5.8 | 29 | 0.04 | 12.1 | 1612.8 | 20.3 | 27.7 | 49.8 |
| Max | 650 | 29.1 | 6.4 | 31 | 0.05 | 27.1 | 2822.4 | 36.8 | 41.4 | 51.4 | |
| SD | 111 | 0.3 | 0.2 | 0.7 | 0 | 4.2 | 395.1 | 5.4 | 5.3 | 0.5 | |
| CV (%) | 25 | 0.9 | 3.4 | 2.3 | 2.17 | 25.3 | 18.8 | 20 | 15.4 | 1 | |
| P2 | Min | 160 | 28.1 | 5.8 | 24 | 0.04 | 0 | 1478.4 | 16.6 | 29.7 | 56.3 |
| Max | 380 | 29.5 | 6.7 | 32 | 0.05 | 47.2 | 2419.2 | 49.3 | 55.4 | 59.1 | |
| SD | 59 | 0.4 | 0.3 | 2.9 | 0 | 15.3 | 276.7 | 8.3 | 7.5 | 0.7 | |
| CV (%) | 27 | 1.4 | 4.4 | 10.2 | 9.86 | 70.3 | 15.1 | 26.4 | 18.2 | 1.2 | |
| P3 | Min | 120 | 29.2 | 5.9 | 23 | 0.04 | 5.5 | 1478.4 | 13 | 48.6 | 53.7 |
| Max | 270 | 30.2 | 6.8 | 38 | 0.06 | 61.6 | 2688 | 41.9 | 93.3 | 56.7 | |
| SD | 48 | 0.3 | 0.3 | 4.8 | 0.01 | 13.4 | 442.7 | 10.2 | 13 | 1.1 | |
| CV (%) | 25 | 0.9 | 4.7 | 17 | 17.14 | 34.7 | 22.7 | 42.7 | 20.7 | 1.9 | |
| P1 | P2 | P3 | ||||||
|---|---|---|---|---|---|---|---|---|
| Species Code | Indictor Value | p | Species Code | Indictor Value | p | Species Code | Indictor Value | p |
| AUIT | 0.81 | 0.001 | AAMB | 0.90 | 0.001 | SGOU | 0.99 | 0.001 |
| EMET | 0.67 | 0.001 | AUSI | 0.90 | 0.001 | EDMG | 0.98 | 0.001 |
| EDNT | 0.60 | 0.002 | ETFG | 0.85 | 0.001 | ECUT | 0.66 | 0.008 |
| DCOF | 0.45 | 0.016 | AUVE | 0.81 | 0.002 | PSP2 | 0.60 | 0.002 |
| PACR | 0.45 | 0.048 | ASP2 | 0.80 | 0.001 | PHUC | 0.60 | 0.001 |
| EFLX | 0.40 | 0.030 | AUMN | 0.70 | 0.001 | EREL | 0.59 | 0.001 |
| EYBE | 0.40 | 0.026 | AUPU | 0.70 | 0.001 | GNAV | 0.59 | 0.003 |
| NAIK | 0.40 | 0.026 | AUGR | 0.60 | 0.008 | EMAI | 0.53 | 0.003 |
| ACOP | 0.36 | 0.031 | DSTE | 0.58 | 0.003 | ISPL | 0.50 | 0.005 |
| SRUD | 0.34 | 0.038 | AUHE | 0.55 | 0.006 | UULN | 0.42 | 0.014 |
| - | - | - | EPAP | 0.40 | 0.031 | GYAC | 0.40 | 0.033 |
| - | - | - | SLDB | 0.40 | 0.025 | PSP1 | 0.40 | 0.027 |
| - | - | - | EFRM | 0.36 | 0.025 | EURS | 0.35 | 0.036 |
| - | - | - | - | - | - | FFRA | 0.35 | 0.049 |
| Observed Contribution | Standardized Effect Size | Expected Contribution | p | |
|---|---|---|---|---|
| Alpha | 27.5 | 18.195 | 22.666 | 0.001 |
| Beta 1 | 50.167 | −14.182 | 70.378 | 0.001 |
| Beta 2 | 41.333 | 10.778 | 25.956 | 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nardelli, M.S.; Padial, A.A.; Bicudo, D.C.; Cordovil, C.M.d.S.; Sampaio, S.C. Variation of Diatoms at Different Scales in the Brazilian Pantanal Basin. Water 2021, 13, 823. https://doi.org/10.3390/w13060823
Nardelli MS, Padial AA, Bicudo DC, Cordovil CMdS, Sampaio SC. Variation of Diatoms at Different Scales in the Brazilian Pantanal Basin. Water. 2021; 13(6):823. https://doi.org/10.3390/w13060823
Chicago/Turabian StyleNardelli, Margaret S., André A. Padial, Denise C. Bicudo, Claudia M. d. S. Cordovil, and Silvio C. Sampaio. 2021. "Variation of Diatoms at Different Scales in the Brazilian Pantanal Basin" Water 13, no. 6: 823. https://doi.org/10.3390/w13060823
APA StyleNardelli, M. S., Padial, A. A., Bicudo, D. C., Cordovil, C. M. d. S., & Sampaio, S. C. (2021). Variation of Diatoms at Different Scales in the Brazilian Pantanal Basin. Water, 13(6), 823. https://doi.org/10.3390/w13060823








