Ontogenetic Structure and Temporal Patterns of Summer Ichthyoplankton in Upper Course of the Xijiang River, SW China
Abstract
:1. Introduction
2. Materials and Methods
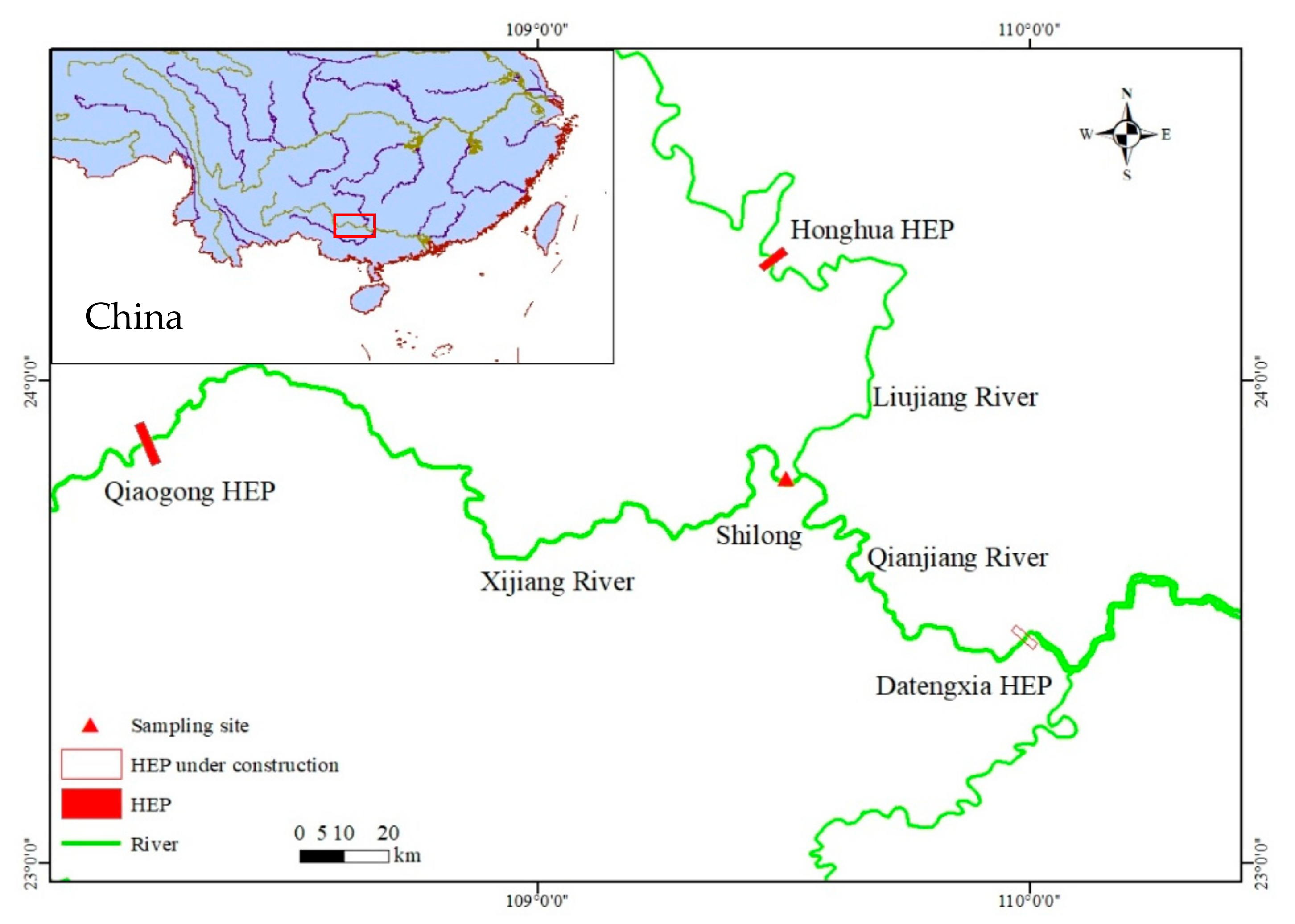
2.1. Study Area
2.2. Sampling and Data Collection
2.3. Data Analysis
3. Results
3.1. Environmental Factors
3.2. Species Composition of Ichthyoplankton
3.3. Temporal Variation of Ichthyoplankton
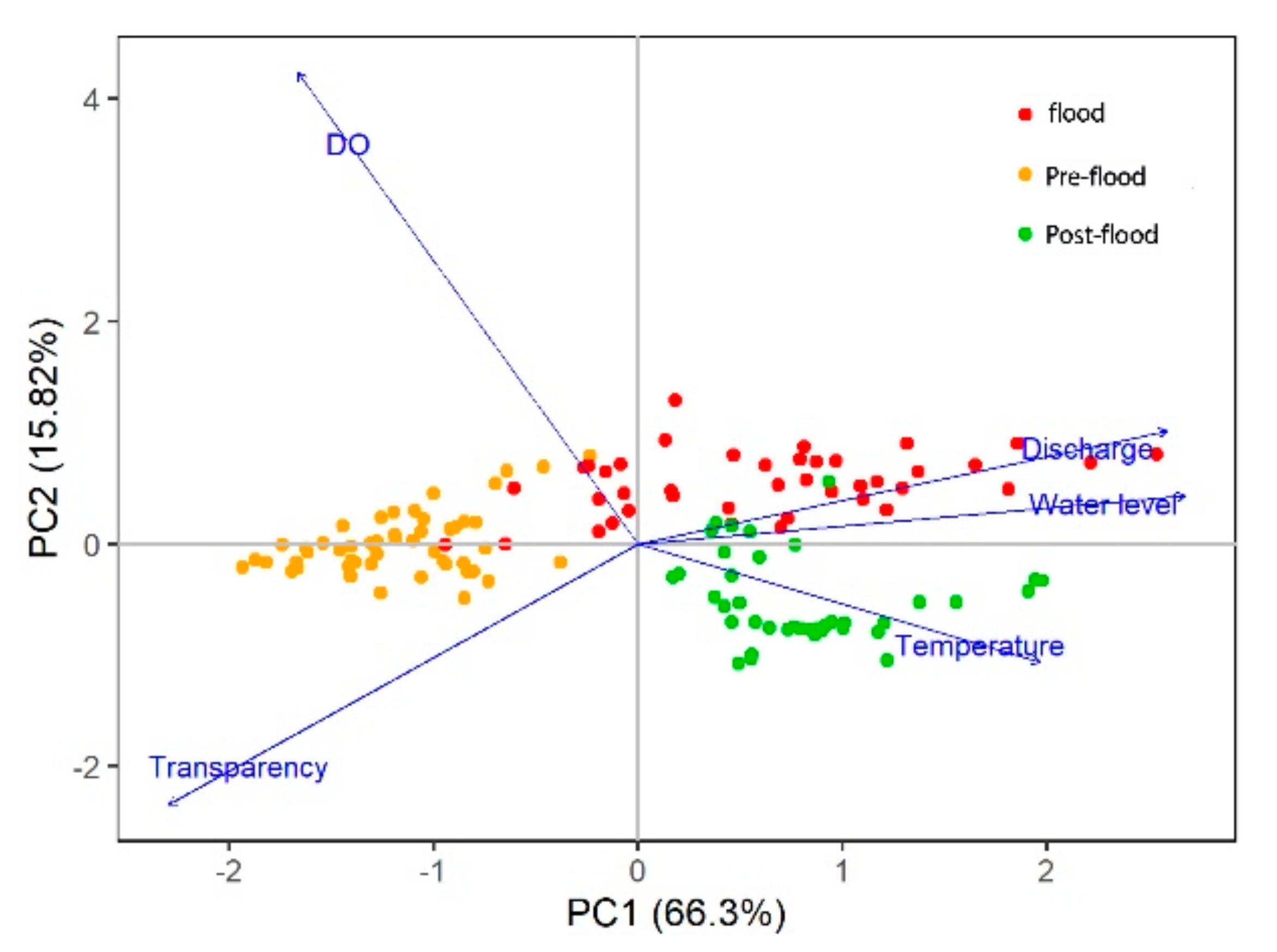
3.4. Relationships between Temporal Patterns and Environmental Factors
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Diaz, S.; Settele, J.; Brondizio, E.S.; Ngo, H.T.; Agard, J.; Arneth, A.; Balvanera, P.; Brauman, K.A.; Butchart, S.H.M.; Chan, K.M.A.; et al. Pervasive human-driven decline of life on Earth points to the need for transformative change. Science 2019, 366, 3100. [Google Scholar] [CrossRef] [Green Version]
- Sala, O.E.; Chapin, F.S.; Armesto, J.J.; Berlow, E.; Bloomfield, J.; Dirzo, R.; Huber-Sanwald, E.; Huenneke, L.F.; Jackson, R.B.; Kinzig, A.; et al. Global Biodiversity Scenarios for the Year 2100. Science 2000, 278, 1770–1774. [Google Scholar] [CrossRef]
- Dudgeon, D. Large-Scale Hydrological Changes in Tropical Asia: Prospects for Riverine Biodiversity. Bioscience 2000, 50, 753–762. [Google Scholar] [CrossRef]
- Yam, R.S.W.; Dudgeon, D. Genetic differentiation of Caridina cantonensis (Decapoda:Atyidae) in Hong Kong streams. J. N. Am. Benthol. Soc. 2005, 24, 845–857. [Google Scholar] [CrossRef]
- Grill, G.; Lehner, B.; Thieme, M.; Geenen, B.; Tickner, D.; Antonelli, F.; Babu, S.; Borrelli, P.; Cheng, L.; Crochetiere, H.; et al. Mapping the world’s free-flowing rivers. Nature 2019, 569, 215–221. [Google Scholar] [CrossRef]
- Mu, H.; Li, M.; Liu, H.; Cao, W. Analysis of fish eggs and larvae flowing into the Three Gorges Reservoir on the Yangtze River, China. Fish. Sci. 2014, 80, 505–515. [Google Scholar] [CrossRef]
- Shuai, F.; Li, X.; Li, Y.; Li, J.; Yang, J.; Lek, S. Temporal Patterns of Larval Fish Occurrence in a Large Subtropical River. PLoS ONE 2016, 11, e0146441. [Google Scholar] [CrossRef] [Green Version]
- Rankin, T.L.; Sponaugle, S. Characteristics of settling coral reef fish are related to recruitment timing and success. PLoS ONE 2014, 9, e108871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sexton, S.C.; Ward, T.M.; Huveneers, C. Characterising the spawning patterns of Jack Mackerel (Trachurus declivis) off eastern Australia to optimise future survey design. Fish. Res. 2017, 186, 223–236. [Google Scholar] [CrossRef]
- Klimova, T.; Podrezova, P. Seasonal distribution of the Black Sea ichthyoplankton near the Crimean Peninsula. Reg. Stud. Mar. Sci. 2018, 24, 260–269. [Google Scholar] [CrossRef]
- Randall, J.R.; Busby, M.S.; Spear, A.H.; Mier, K.L. Spatial and temporal variation of late summer ichthyoplankton assemblage structure in the eastern Chukchi Sea: 2010–2015. Polar Biol. 2019, 42, 1811–1824. [Google Scholar] [CrossRef]
- Humphries, P.; King, A.J.; Koehn, J.D. Fish, Flows and Flood Plains: Links between Freshwater Fishes and their Environment in the Murray-Darling River System Australia. Environ. Boil. Fishes 1999, 56, 129–151. [Google Scholar] [CrossRef]
- Reynalte-Tataje, D.A.; Agostinho, A.A.; Bialetzki, A.; Hermes-Silva, S.; Fernandes, R.; Zaniboni-Filho, E. Spatial and temporal variation of the ichthyoplankton in a subtropical river in Brazil. Environ. Biol. Fishes 2011, 94, 403–419. [Google Scholar] [CrossRef]
- Suzuki, H.; Agostinho, A.; Bailly, D.; Gimenes, M.; Júlio-Junior, H.; Gomes, L. Inter-annual variations in the abundance of young-of-the-year of migratory fishes in the Upper Paraná River floodplain: Relations with hydrographic attributes. Braz. J. Biol. 2009, 69, 649–660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, R.; Agostinho, A.A.; Ferreira, E.A.; Pavanelli, C.S.; Suzuki, H.I.; Lima, D.P.; Gomes, L.C. Effects of the hydrological regime on the ichthyofauna of riverine environments of the Upper Paraná River floodplain. Braz. J. Biol. 2009, 69, 669–680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, J.; Lei, J.; Chen, S.; Kong, B. Investigation on spawning grounds of fish in the lower reaches of the Hongshui River. Guangxi Fish. Sci. Technol. 2005, 12, 1–5. [Google Scholar]
- Cai, Y.; Ng, P.K.L. Freshwater shrimps from karst caves of southern China, with descriptions of seven new species and the identity of Typhlocaridina linyunensis Li and Luo, 2001 (Crustacea: Decapoda: Caridea). Zool. Stud. 2018, 57, 27. [Google Scholar] [CrossRef]
- Zhou, J.; Zhang, C. Freshwater Fishes of Guangxi, China; Guangxi People’s Publishing House: Nanning, China, 2005.
- Tan, X.; Li, X.; Lek, S.; Li, Y.; Wang, C.; Li, J.; Luo, J. Annual dynamics of the abundance of fish larvae and its relationship with hydrological variation in the Pearl River. Environ. Biol. Fishes 2010, 88, 217–225. [Google Scholar] [CrossRef]
- Chong, W.; Yanhong, F.; Yinquan, L.; Daoming, H. Eggs and larval and Taxonomic Diversity in Main Stream of Hongshui River. Chin. J. Fish. 2019, 32, 48–53. [Google Scholar]
- Ye, Q. Early Morphogenesis and Species Identification of Fishes in Yangtze River Doctor; Institute of Hydrobiology, Chinese Academy of Sciences: Wuhan, China, 2005.
- Xichang, A.; Yuefei, L.; Chao, W.; Jie, L.; Shixun, P.; Xinhui, L. Early morphogenesis and larval resources of squaliobarbus curriculus in the Pearl River. J. Huazhong Agric. Univ. 2009, 28, 609–613. [Google Scholar]
- Gotelli, N.J.; Ellison, A.M. A Primer of Ecological Statistics; Sinauer Associates Publishers: Sunderland, UK, 2004. [Google Scholar]
- Legendre, P.; Legendre, L. Numerical Ecology, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 1998. [Google Scholar]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Song, Y.; Cheng, F.; Ren, P.; Wang, Z.; Xie, S. Longitudinal recovery gradients of drifting larval fish assemblages in the middle reach of the Yangtze River: Impact of the Three Gorges Dam and conservation implementation. Can. J. Fish. Aquat. Sci. 2019, 76, 2256–2267. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F.; Prentice, I.C. A Theory of Gradient Analysis. Adv. Ecol. Res. 1988, 18, 271–317. [Google Scholar] [CrossRef]
- Makarenkov, V.; Legendre, P. Nonlinear redundancy analysis and canonical correspondence analysis based on polynomial regression. Ecology 2002, 83, 1146–1161. [Google Scholar] [CrossRef]
- Chong, W.; Shan, X.; Wen-xing, X.; Xiu-ling, C.; Dao-ming, H. Investigation on fish resources in the mainstream of Hongshuihe River after the formation of the cascade reservoirs. Freshw. Fish. 2015, 45, 30–36. [Google Scholar]
- Deters, J.E.; Chapman, D.C.; McElroy, B. Location and timing of Asian carp spawning in the Lower Missouri River. Environ. Biol. Fishes 2012, 96, 617–629. [Google Scholar] [CrossRef]
- Bolu, Y. He Distribution, Natural Conditions, and Breeding Production of the Spawning Ground of Four Famous Freshwater Fishes on the Main Stream of the Yangtze River; Hubei Science and Technology Press: Wuhan, China, 1988.
- Winemiller, K.O. Patterns of variation in life history among South American fishes in seasonal environments. Oecologia 1989, 81, 225–241. [Google Scholar] [CrossRef] [PubMed]
- Xuan, Q.; Jun, C.; Fang, X. Impact of Cascaded Hydroelectric on Reproduction of Fish with Pelagic Eggs in Middle and Lower Reaches of Hanjiang River. Environ. Sci. Technol. 2014, 37, 501–506. [Google Scholar] [CrossRef]
- Martins, E.G.; Hinch, S.G.; Patterson, D.A.; Hague, M.J.; Cooke, S.J.; Miller, K.M.; Robichaud, D.; English, K.K.; Farrell, A.P.; Jonsson, B. High river temperature reduces survival of sockeye salmon (Oncorhynchus nerka) approaching spawning grounds and exacerbates female mortality. Can. J. Fish. Aquat. Sci. 2012, 69, 330–342. [Google Scholar] [CrossRef] [Green Version]
- Fincham, J.I.; Rijnsdorp, A.D.; Engelhard, G.H. Shifts in the timing of spawning in sole linked to warming sea temperatures. J. Sea Res. 2013, 75, 69–76. [Google Scholar] [CrossRef]
- Kurita, Y.; Fujinami, Y.; Amano, M. The effect of temperature on the duration of spawning markers-migratory-nucleus and hydrated oocytes and postovulatory follicles-in the multiple-batch spawner Japanese flounder (Paralichthys olivaceus). Fish. Bull. 2011, 109, 79–89. [Google Scholar]
- Li, S.; Gao, H.; Hao, X.; Zhu, L.; Li, T.; Zhang, H.; Zhou, Y.; Xu, X.; Yang, G.; Chen, B. Seasonal, Lunar and Tidal Influences on Habitat Use of Indo-Pacific Humpback Dolphins in Beibu Gulf, China. Zool. Stud. 2018, 57, e1. [Google Scholar] [CrossRef]
- Winemiller, K.O. Life history strategies, population regulation, and implications for fisheries management. Can. J. Fish. Aquat. Sci. 2005, 62, 872–885. [Google Scholar] [CrossRef]
- Chu, C.; Loh, K.H.; Ng, C.C.; Ooi, A.L.; Konishi, Y.; Huang, S.P.; Chong, V.C. Using DNA Barcodes to Aid the Identification of Larval Fishes in Tropical Estuarine Waters (Malacca Straits, Malaysia). Zool. Stud. 2019, 58, e30. [Google Scholar] [CrossRef] [PubMed]




| Period | Water Temperature (℃) | Discharge (m3/s) | Water Levels (m) | DO (mg/L) | Water Transparency (cm) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Range | Mean ± SD | Range | Mean ± SD | Range | Mean ± SD | Range | Mean ± SD | Range | Mean ± SD | |
| Pre-flood | 15–24.5 | 19.17 ± 4.07 | 1310–3070 | 1836.67 ± 346.67 | 63.08–67.88 | 64.82 ± 0.96 | 6.32–6.83 | 6.55 ± 0.16 | 100–125 | 111.97 ± 7.60 |
| Flood | 22.6–25.8 | 23.63 ± 0.71 | 1420–8520 | 3707.78 ± 1579.71 | 63.5–76.46 | 68.77 ± 2.93 | 6.12–7.02 | 6.56 ± 0.26 | 20–125 | 70.44 ± 27.32 |
| Post-flood | 24.3–27.2 | 26.15 ± 0.91 | 3100–7230 | 4240 ± 1095.30 | 68.46–75.29 | 70.48 ± 1.89 | 5.64–5.98 | 5.78 ± 0.12 | 54–105 | 76.25 ± 13.99 |
| Taxon | Egg Type | Life Stages of Fish | Pre-Flood | Flood | Post-Flood | |||
|---|---|---|---|---|---|---|---|---|
| Density (ind.·100−3) | IRI | Density (ind.·100−3) | IRI | Density (ind.·100−3) | IRI | |||
| SALMONIFORMES | ||||||||
| Salangidae | ||||||||
| Neosalanx taihuensis Chen, 1956 | O | larval | 0.0012 | 0.0144 | 0.0106 | 0.0122 | 0.0059 | 2.7429 |
| CYPRINIFORMES | ||||||||
| Cobitidae | ||||||||
| Sinibotia robusta (Wu, 1939) | P | eggs | 1.4776 | 1048.72 ** | 0.3131 | 45.77 | 0.1475 | 101.98 * |
| larval | 0.0044 | 0.0078 | 0.0216 | |||||
| Sinibotia pulchra (Wu, 1939) | P | eggs | 0.3240 | 98.84 | 0.3784 | 165.56 * | 0.4778 | 753.54 * |
| Sinibotia zebra (Wu, 1939) | P | eggs | 0.1680 | 53.65 | 0.1668 | 66.60 | 0.1193 | 135.01 * |
| larval | 0.0011 | 0.0072 | ||||||
| Squaliobarbus curriculus (Richardson, 1846) | P | eggs | 0.3596 | 167.79 * | 0.8156 | 508.90 * | 0.4444 | 845.43 * |
| larval | 0.0251 | 0.0106 | 0.1079 | |||||
| Hemiculter leucisculus (Basilewsky, 1855) | A | eggs | 1.1124 | 794.57 * | 0.9909 | 774.69 * | 0.7626 | 1261.47 ** |
| larval | 0.0284 | 0.0026 | 0.0144 | |||||
| Chanodichthys erythropterus (Basilewsky, 1855) | A | eggs | 0.4861 | 225.37 | 0.3904 | 224.85 * | 0.0283 | 15.79 |
| larval | 0.0321 | |||||||
| Sinibrama macrops (Günther, 1868) | P | eggs | 0.7246 | 274.67 * | 1.1307 | 731.14 * | 0.1159 | 118.28 * |
| larval | 0.0016 | |||||||
| Hypophthalmichthys nobilis (Richardson, 1845) | A | larval | 0.0023 | 0.09 | ||||
| Squalidus argentatus (Sauvage and Dabry de Thiersant, 1874) | P | eggs | 3.8542 | 2922.07 ** | 5.9786 | 4440.72 ** | 2.6720 | 1481.5 ** |
| larval | 0.0087 | 0.0086 | 0.0504 | |||||
| Saurogobio dabryi Bleeker, 1871 | P | eggs | 0.2826 | 124.16 * | 0.3370 | 179.50 * | 0.0201 | 13.98 |
| Rhodeus ocellatus (Kner, 1866) | O | larval | 0.0035 | 0.08 | 0.0272 | 4.65 | 0.0059 | 2.47 |
| Acrossocheilus kreyenbergii (Regan, 1908) | A | larval | 0.0059 | 0.07 | 0.0686 | 19.56 | 0.0035 | 1.48 |
| Onychostoma gerlachi (Peters, 1881) | A | larval | 0.0177 | 7.41 | ||||
| Cirrhinus molitorella (Valenciennes, 1844) | A | eggs | 0.1326 | 22.51 | 0.4508 | 281.44 * | 0.2873 | 382.47 * |
| larval | 0.0093 | 0.0026 | 0.0072 | |||||
| Osteochilus salsburyi Nichols and Pope, 1927 | O | eggs | 0.1750 | 32.03 | 0.5854 | 311.76 * | 0.0981 | 81.95 |
| Ptychidio jordani Myers, 1930 | O | larval | 0.0141 | 4.61 | ||||
| Cyprinus carpio Linnaeus, 1758 | A | eggs | 0.0213 | 2.59 | ||||
| Carassius auratus (Linnaeus, 1758) | A | eggs | 0.0130 | 0.95 | ||||
| Homalopteridae | ||||||||
| Sinogastromyzon wui Fang, 1930 | P | eggs | 4.0345 | 2854.91 ** | 1.6949 | 741.37 * | 1.0668 | 1632.92 ** |
| larval | 0.0072 | |||||||
| SILURIFORMES | ||||||||
| Clariidae | ||||||||
| Clarias fuscus (Lacepède, 1803) | A | larval | 0.0094 | 1.08 | 0.0012 | 0.5485 | ||
| Bagridae | ||||||||
| Tachysurus fulvidraco (Richardson, 1846) | A | larval | 0.0023 | 0.06 | 0.0485 | 11.98 | 0.0047 | 2.19 |
| Hemibagrus guttatus (Lacepède, 1803) | A | larval | 0.0012 | 0.01 | 0.0130 | 1.24 | 0.0059 | 2.74 |
| Perciformes | ||||||||
| Serranidae | ||||||||
| Siniperca chuatsi (Basilewsky, 1855) | P | larval | 0.0047 | 0.11 | 0.0283 | 6.47 | 0.0059 | 2.47 |
| Cichlidae | ||||||||
| Oreochromis mossambicus (Peters, 1852) | O | larval | 0.0047 | 0.11 | 0.1029 | 33.26 | 0.0070 | 3.62 |
| Oreochromis niloticus (Linnaeus, 1758) | O | larval | 0.0047 | 0.17 | 0.0709 | 22.94 | 0.0047 | 2.19 |
| Gobiidae | ||||||||
| Rhinogobius giurinus (Rutter, 1897) | A | eggs | 0.01 | 1.57 | 0.0812 | 3.95 | ||
| larval | 0.0011 | 0.0018 | 0.0070 | |||||
| Mastacembelidae | ||||||||
| Mastacembelus armatus (Lacepède, 1800) | O | larval | 0.0532 | 14.17 | 0.0047 | 2.19 | ||
| Taxons | Pre-Flood Versus Flood | Flood Versus Post-Flood | Pre-Flood Versus Post-Flood |
|---|---|---|---|
| Sinogastromyzon wui | 20.41 | 31.80 | 15.36 |
| Squalidus argentatus | 30.06 | 16.13 | 32.61 |
| Sinibotia robusta | 6.49 | 9.87 | 4.92 |
| Sinibrama macrops | 9.15 | 9.79 | |
| Hemiculter leucisculus | 9.64 | 5.76 | |
| Squaliobarbus curriculus | 5.83 | 8.34 | 5.78 |
| Chanodichthys erythropterus | 4.39 | ||
| Osteochilus salsburyi | 5.13 | 5.14 | |
| Cirrhinus molitorella | 4.10 | ||
| Sinibotia pulchra | 6.37 | ||
| Medium dissimilarity | 59.11 | 54.76 | 59.93 |
| RDA1 | RDA2 | RDA3 | RDA4 | RDA5 | |
|---|---|---|---|---|---|
| F | 57.68 | 18.67 | 9.3 | 3.81 | 2.31 |
| p-value | 0.001 *** | 0.001 *** | 0.01 * | 0.05 * | 0.1 |
| Eigenvalue | 0.07764 | 0.0251 | 0.0125 | 0.0051 | 0.0031 |
| Proportion Explained | 0.62842 | 0.2035 | 0.1013 | 0.0416 | 0.0252 |
| Cumulative Proportion | 0.62842 | 0.8319 | 0.9332 | 0.9748 | 1 |
| Transparency | −0.4736 | 0.0122 | −0.0237 | 0.0112 | 0.0360 |
| DO | −0.1498 | −0.2190 | −0.1180 | −0.0149 | −0.0089 |
| Temperature | 0.3631 | 0.1232 | −0.0737 | 0.0227 | 0.0412 |
| Discharge | 0.4381 | −0.0359 | 0.0862 | 0.0285 | 0.0182 |
| “Water Level” | 0.4483 | 0.0106 | 0.0783 | −0.0122 | 0.0289 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, M.; Wu, Z.; Tan, X.; Huang, L.; Feng, J.; Rad, S. Ontogenetic Structure and Temporal Patterns of Summer Ichthyoplankton in Upper Course of the Xijiang River, SW China. Water 2021, 13, 703. https://doi.org/10.3390/w13050703
Gao M, Wu Z, Tan X, Huang L, Feng J, Rad S. Ontogenetic Structure and Temporal Patterns of Summer Ichthyoplankton in Upper Course of the Xijiang River, SW China. Water. 2021; 13(5):703. https://doi.org/10.3390/w13050703
Chicago/Turabian StyleGao, Minghui, Zhiqiang Wu, Xichang Tan, Liangliang Huang, Jie Feng, and Saeed Rad. 2021. "Ontogenetic Structure and Temporal Patterns of Summer Ichthyoplankton in Upper Course of the Xijiang River, SW China" Water 13, no. 5: 703. https://doi.org/10.3390/w13050703
APA StyleGao, M., Wu, Z., Tan, X., Huang, L., Feng, J., & Rad, S. (2021). Ontogenetic Structure and Temporal Patterns of Summer Ichthyoplankton in Upper Course of the Xijiang River, SW China. Water, 13(5), 703. https://doi.org/10.3390/w13050703







