Prospects in Cadmium-Contaminated Water Management Using Free-Living Cyanobacteria (Oscillatoria sp.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganism and Culture Conditions
2.2. Stock Solution of Cd
2.3. Cd Removal Studies and Elemental Analysis
2.4. Metal Removal Kinetics
2.5. FTIR-ATR Analysis
2.6. Analytical Analysis
2.6.1. Microbial Growth
2.6.2. Protein Content
2.6.3. Lipid Peroxidation (MDA Accumulation)
2.6.4. Dehydrogenase Activity
2.6.5. Physiological Parameters
Quantification of Photosynthetic Pigments
2.6.6. Carbohydrate Determination
2.7. Statistical Analysis
3. Results
3.1. Cd Removal Efficiency
3.2. Evaluation of Cd Toxicity in Oscillatoria sp.
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, X.; Cobbina, S.J.; Mao, G.; Xu, H.; Zhang, Z.; Yang, L. A review of toxicity and mechanisms of individual and mixtures of heavy metals in the environment. Environ. Sci. Pollut. Res. 2016, 23, 8244–8259. [Google Scholar] [CrossRef]
- Anyanwu, B.O.; Ezejiofor, A.N.; Igweze, Z.N.; Orisakwe, O.E. Heavy metal mixture exposure and effects in developing nations: An update. Toxics 2018, 6, 65. [Google Scholar] [CrossRef]
- Idrees, N.; Tabassum, B.; Abd Allah, E.F.; Hashem, A.; Sarah, R.; Hashim, M. Groundwater contamination with cadmium concentrations in some West U.P. Regions, India. Saudi J. Biol. Sci. 2018, 25, 1365–1368. [Google Scholar] [CrossRef] [PubMed]
- Hayat, M.T.; Nauman, M.; Nazir, N.; Ali, S.; Bangash, N. Environmental hazards of cadmium: Past, present, and future. In Cadmium Toxicity and Tolerance in Plants: Agronomic, Genetic, Molecular and Omic Approaches, 1st ed.; Hasanuzzaman, M., Prasad, M.N.V., Fujita, M., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 163–183. [Google Scholar] [CrossRef]
- Chunhabundit, R. Cadmium exposure and potential health risk from foods in contaminated area, Thailand. Toxicol. Res. 2016, 32, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Rahimzadeh, M.R.; Kazemi, S.; Moghadamnia, A.A. Cadmium toxicity and treatment: An update. Caspian J. Intern. Med. 2017, 8, 135–145. [Google Scholar] [CrossRef]
- Qu, R.J.; Wang, X.H.; Feng, M.B.; Li, Y.; Liu, H.X.; Wang, L.S.; Wang, Z.Y. The toxicity of cadmium to three aquatic organisms (Photobacterium phosphoreum, Daphnia magna and Carassius auratus) under different pH levels. Ecotox. Environ. Saf. 2013, 95, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Zaki, M.S.; Zakaria, A.; Eissa, I.A.; Eldeen, A.I. Effect of cadmium toxicity on Vertebrates. Electron. Physician 2016, 8, 1964–1965. [Google Scholar] [CrossRef]
- WHO. Chemical Safety—Activity Report; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Azimi, A.; Azari, A.; Rezakazemi, M.; Ansarpour, M. Removal of heavy metals from industrial wastewaters: A review. ChemBioEng. Rev. 2017, 4, 37–59. [Google Scholar] [CrossRef]
- Barakat, M.A. New trends in removing heavy metals from industrial wastewater. Arab. J. Chem. 2011, 4, 361–377. [Google Scholar] [CrossRef]
- Azubuike, C.C.; Chikere, C.B.; Okpokwasili, G.C. Bioremediation techniques–classification based on site of application: Principles, advantages, limitations and prospects. World J. Microbiol. Biotechnol. 2016, 32, 180–198. [Google Scholar] [CrossRef]
- Dhir, B. Potential of biological materials for, removing heavy metals from wastewater. Environ. Sci. Pollut. Res. 2014, 21, 1614–1627. [Google Scholar] [CrossRef]
- Dixit, R.; Wasiullah; Malaviya, D.; Pandiyan, K.; Singh, U.B.; Sahu, A.; Shukla, R.; Singh, B.P.; Rai, J.P.; Sharma, P.K.; et al. Bioremediation of heavy metals from soil and aquatic environment: An overview of principles and criteria of fundamental processes. Sustainability 2015, 7, 2189–2212. [Google Scholar] [CrossRef]
- Azizi, S.N.; Colagar, A.H.; Hafeziyan, S.M. Removal of Cd(II) from aquatic system using Oscillatoria sp. biosorbent. Sci. World J. 2012, 1–7. [Google Scholar] [CrossRef]
- Katircioǧlu, H.; Aslim, B.; Rehber Türker, A.; Atici, T.; Beyatli, Y. Removal of cadmium(II) ion from aqueous system by dry biomass, immobilized live and heat-inactivated Oscillatoria sp. H1 isolated from freshwater (Mogan Lake). Bioresour. Technol. 2008, 99, 4185–4191. [Google Scholar] [CrossRef]
- Rao, K.S.; Mohapatra, M.; Anand, S.; Venkateswarlu, P. Review on cadmium removal from aqueous solutions. Int. J. Eng. Sci. Technol. 2010, 2, 81–103. [Google Scholar] [CrossRef]
- Satya, A.; Harimawan, A.; Haryani, G.S.; Johir, M.A.H.; Vigneswaran, S.; Ngo, H.H.; Setiadi, T. Batch study of cadmium biosorption by carbon dioxide enriched Aphanothece sp. dried biomass. Water 2020, 12, 264. [Google Scholar] [CrossRef]
- Cassier-Chauvat, C.; Chauvat, F. Responses to oxidative and heavy metal stresses in cyanobacteria: Recent advances. Int. J. Mol. Sci. 2015, 16, 871–886. [Google Scholar] [CrossRef]
- Singh, S. Biosorption of heavy metals by cyanobacteria: Potential of live and dead cells in bioremediation. In Microbial Bioremediation & Biodegradation; Shah, M., Ed.; Springer: Singapore, 2020; pp. 409–423. [Google Scholar] [CrossRef]
- Riskuwa-Shehu, M.; Ismail, H.; Sulaiman, M. Biosorption of heavy metals by Oscillatoria species. Microbiol. Res. J. Int. 2019, 27, 1–8. [Google Scholar] [CrossRef]
- Barkia, I.; Saari, N.; Manning, S.R. Microalgae for high-value products towards human health and nutrition. Mar. Drugs 2019, 17, 304. [Google Scholar] [CrossRef] [PubMed]
- Sevda, S.; Garlapi, V.G.; Sharma, S.; Bhattacharya, S.; Mishra, S.; Sreekrishnan, T.R.; Pant, D. Microalgae at niches of bioelectrochemical systems: A new platform for sustainable energy production coupled industrial effluent treatment. Bioresour. Technol. Rep. 2019, 7, 100290. [Google Scholar] [CrossRef]
- Zahra, Z.; Choo, D.H.; Lee, H.; Parveen, A. Cyanobacteria: Review of current potentials and applications. Environments 2020, 7, 13. [Google Scholar] [CrossRef]
- Bejjanki, D.; Muthukumar, K.; Radhakrishnan, T.K.; Alagarsamy, A.; Pugazhendhi, A.; Mohamed, S.N. Simultaneous bioelectricity generation and water desalination using Oscillatoria sp. as biocatalyst in photosynthetic microbial desalination cell. Sci. Total. Environ. 2021, 754, 142215. [Google Scholar] [CrossRef] [PubMed]
- Kokabian, B.; Gude, V.G. Photosynthetic microbial desalination cells (PMDCs) for clean energy, water and biomass production. Environ. Sci. Process. Impacts 2013, 15, 2178–2185. [Google Scholar] [CrossRef]
- Emiliani, J.; Llatance Oyarce, W.G.; Bergara, C.D.; Salvatierra, L.M.; Novo, L.A.B.; Pérez, L.M. Variations in the phytoremediation efficiency of metal-polluted water with Salvinia biloba: Prospects and toxicological impacts. Water 2020, 12, 1737. [Google Scholar] [CrossRef]
- Tello Zevallos, W.; Salvatierra, L.M.; Loureiro, D.B.; Morató, J.; Pérez, L.M. Evaluation of the autochthonous free-floating macrophyte Salvinia biloba Raddi for use in the phytoremediation of water contaminated. Desalin. Water Treat. 2018, 103, 282–289. [Google Scholar] [CrossRef]
- Zheng, X.; Bi, C.; Li, Z.; Podariu, M.; Hage, D.S. Analytical methods for kinetic studies of biological interactions: A review. J. Pharm. Biomed. Anal. 2015, 113, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Balaji, S.; Kalaivani, T.; Shalini, M.; Gopalakrishnan, M.; Muhammad, M.A.R.; Rajasekaran, C. Sorption sites of microalgae possess metal binding ability towards Cr(VI) from tannery effluents—A kinetic and characterization study. Desalin. Water Treat. 2016, 57, 14518–14529. [Google Scholar] [CrossRef]
- Lowry, O.; Rosebroug, H.; Farr, A.; Randall, R. Protein measurement with the Folin-phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Piotrowska-Niczyporuk, A.; Bajguz, A.; Zambrzycka, E.; Godlewska-Żyłkiewicz, B. Phytohormones as regulators of heavy metal biosorption and toxicity in green alga Chlorella vulgaris (Chlorophyceae). Plant Physiol. Biochem. 2012, 52, 52–65. [Google Scholar] [CrossRef]
- Capasso, J.M.; Cossío, B.R.; Berl, T.; Rivard, C.J.; Jiménez, C. A colorimetric assay for determination of cell viability in algal cultures. Biomol. Eng. 2003, 20, 133–138. [Google Scholar] [CrossRef]
- Miazek, K.; Ledakowicz, S. Chlorophyll extraction from leaves, needles and microalgae: A kinetic approach. Int. J. Agric. Biol. Eng. 2013, 6, 107–115. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.; Hamilton, J.; Rebers, P.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Hall, J.L. Cellular mechanisms for heavy metal detoxification and tolerance. J. Exp. Bot. 2002, 53, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cobbett, C.; Goldsbrough, P. Phytochelatins and metallothioneins: Roles in heavy metal detoxification and homeostasis. Annu. Rev. Plant Biol. 2002, 53, 159–182. [Google Scholar] [CrossRef]
- Ziller, A.; Fraissinet-Tachet, L. Metallothionein diversity and distribution in the tree of life: A multifunctional protein. Metallomics 2018, 10, 1549–1559. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Nakashima, S.; Hirose, K.; Uemura, Y.; Shibasaka, M.; Katsuhara, M.; Kasamo, K. A metallothionein and CPx-ATPase handle heavy-metal tolerance in the filamentous cyanobacterium Oscillatoria brevis 1. FEBS Lett. 2003, 542, 159–163. [Google Scholar] [CrossRef]
- Çelekli, A.; Gültekin, E.; Bozkurt, H. Morphological and biochemical responses of Spirogyra setiformis exposed to Cadmium. Clean-Soil Air Water 2016, 44, 256–262. [Google Scholar] [CrossRef]
- Mohadi, R.; Setiawan, D.; Zulkifli, H. Biosorption of Cu(II) onto algae biomass (Oscillatoria splendida) isolated from swamp water ecosystem in Palembang, South Sumatera. J. Phys. Conf. Ser. 2019, 1282, 012066. [Google Scholar] [CrossRef]
- Alam, M.A.; Wan, C.; Zhao, X.Q.; Chen, L.J.; Chang, J.S.; Bai, F.W. Enhanced removal of Zn2+ or Cd2+ by the flocculating Chlorella vulgaris JSC-7. J. Hazard. Mater. 2015, 289, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Qiu, H.; Chang, Z.; Jiang, Z.; Yin, W. The effect of cadmium on the growth and antioxidant response for freshwater algae Chlorella vulgaris. SpringerPlus 2016, 5, 1290. [Google Scholar] [CrossRef] [PubMed]
- Jamers, A.; Lenjou, M.; Deraedt, P.; Van Bockstaele, D.; Blust, R.; de Coen, W. Flow cytometric analysis of the cadmium-exposed green alga Chlamydomonas reinhardtii (Chlorophyceae). Eur. J. Phycol. 2009, 44, 541–550. [Google Scholar] [CrossRef]
- Berridge, M.V.; Herst, P.M.; Tan, A.S. Tetrazolium dyes as tools in cell biology: New insights into their cellular reduction. Biotechnol. Annu. Rev. 2005, 11, 127–152. [Google Scholar] [CrossRef] [PubMed]
- Ghaly, A.E.; Mahmoud, N. Optimum conditions for measuring dehydrogenase activity of Aspergillus niger using TTC. Am. J. Biochem. Biotechnol. 2006, 2, 186–194. [Google Scholar] [CrossRef]
- Pérez, L.M.; López Álvarez, B.L.; Codony, F.; Fittipaldi, M.; Adrados, B.; Peñuela, G.; Morató, J. A new microtiter plate screening method for evaluating the viability of aerobic respiring bacteria in high surface biofilms. Lett. Appl. Microbiol. 2010, 51, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Hirschberg, J.; Chamovitz, D. Carotenoids in Cyanobacteria. In The Molecular Biology of Cyanobacteria. Advances in Photosynthesis; Bryant, D.A., Ed.; Springer: Dordrecht, The Netherlands, 1994; Volume 1, pp. 559–579. [Google Scholar] [CrossRef]
- Havaux, M. Carotenoid oxidation products as stress signals in plants. Plant J. 2014, 79, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Ramel, F.; Mialoundama, A.S.; Havaux, M. Nom-enzymic carotenoid oxidation and photooxidative stress signaling in plants. J. Exp. Bot. 2013, 64, 799–805. [Google Scholar] [CrossRef]
- Castillo Loría, K.; Emiliani, J.; Bergara, C.D.; Herrero, M.S.; Salvatierra, L.M.; Pérez, L.M. Effect of daily exposure to Pb-contaminated water on Salvinia biloba physiology and phytoremediation performance. Aquat. Toxicol. 2019, 210, 158–166. [Google Scholar] [CrossRef]
- Prado, C.; Rodríguez-Montelongo, L.; González, J.A.; Pagano, E.A.; Hilal, M.; Prado, F.E. Uptake of chromium by Salvinia minima: Effect on plant growth, leaf respiration and carbohydrate metabolism. J. Hazard. Mater. 2010, 177, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Rosa, M.; Prado, C.; Podazza, G.; Interdonato, R.; González, A.; Prado, F. Soluble sugars-metabolism, sensing and abiotic stress. Plant Signal. Behav. 2009, 4, 388–393. [Google Scholar] [CrossRef] [PubMed]
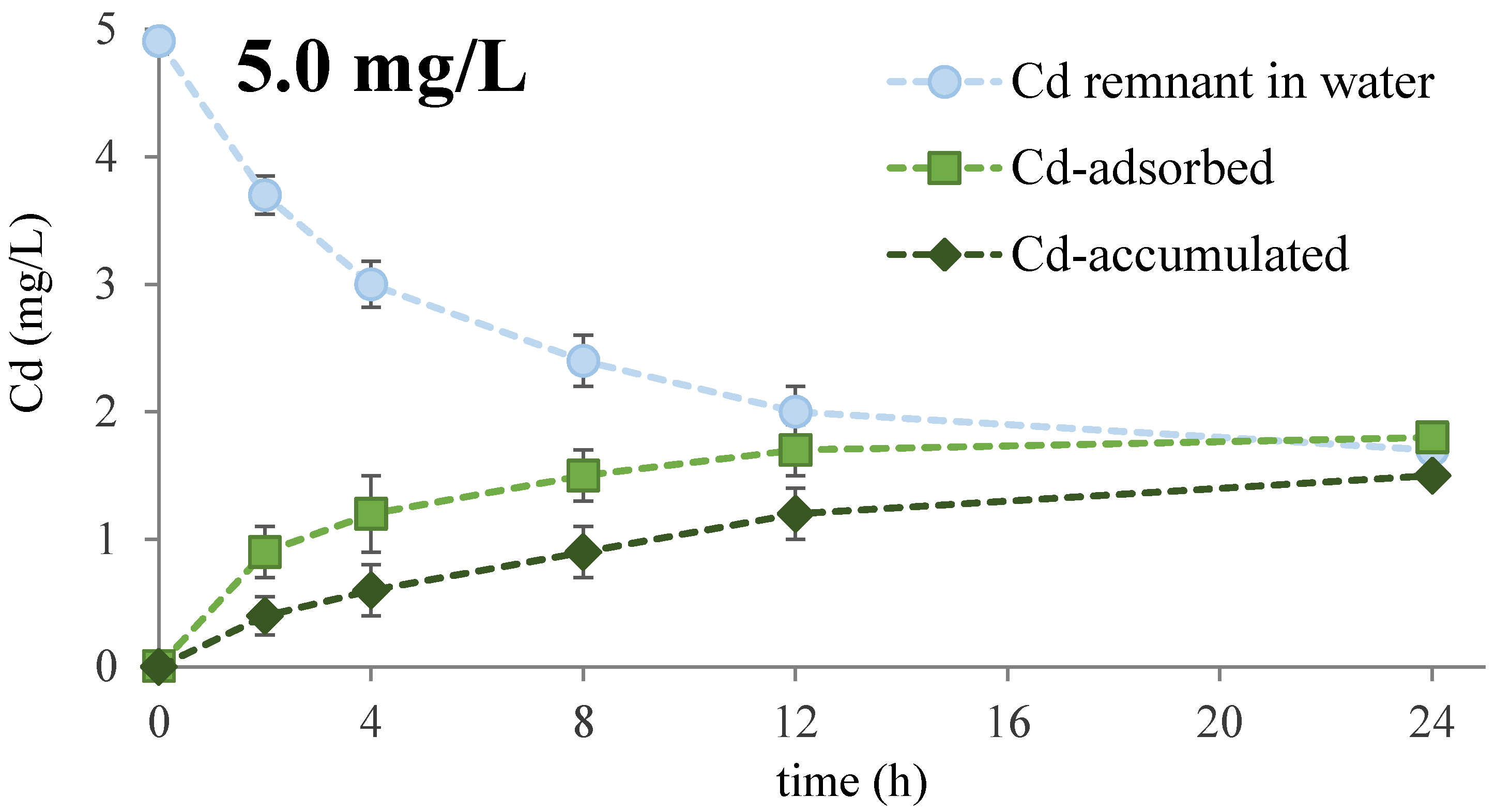
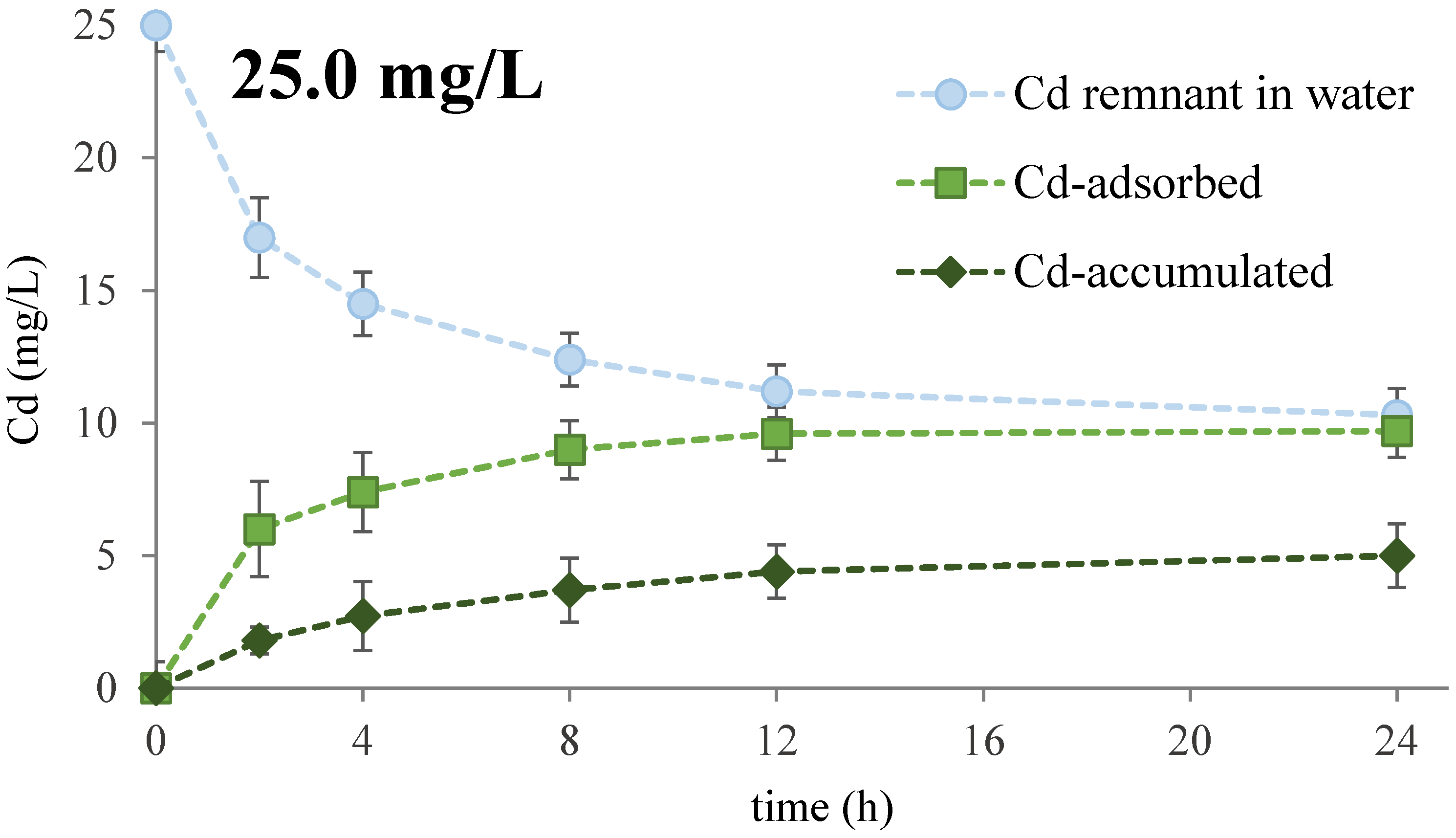
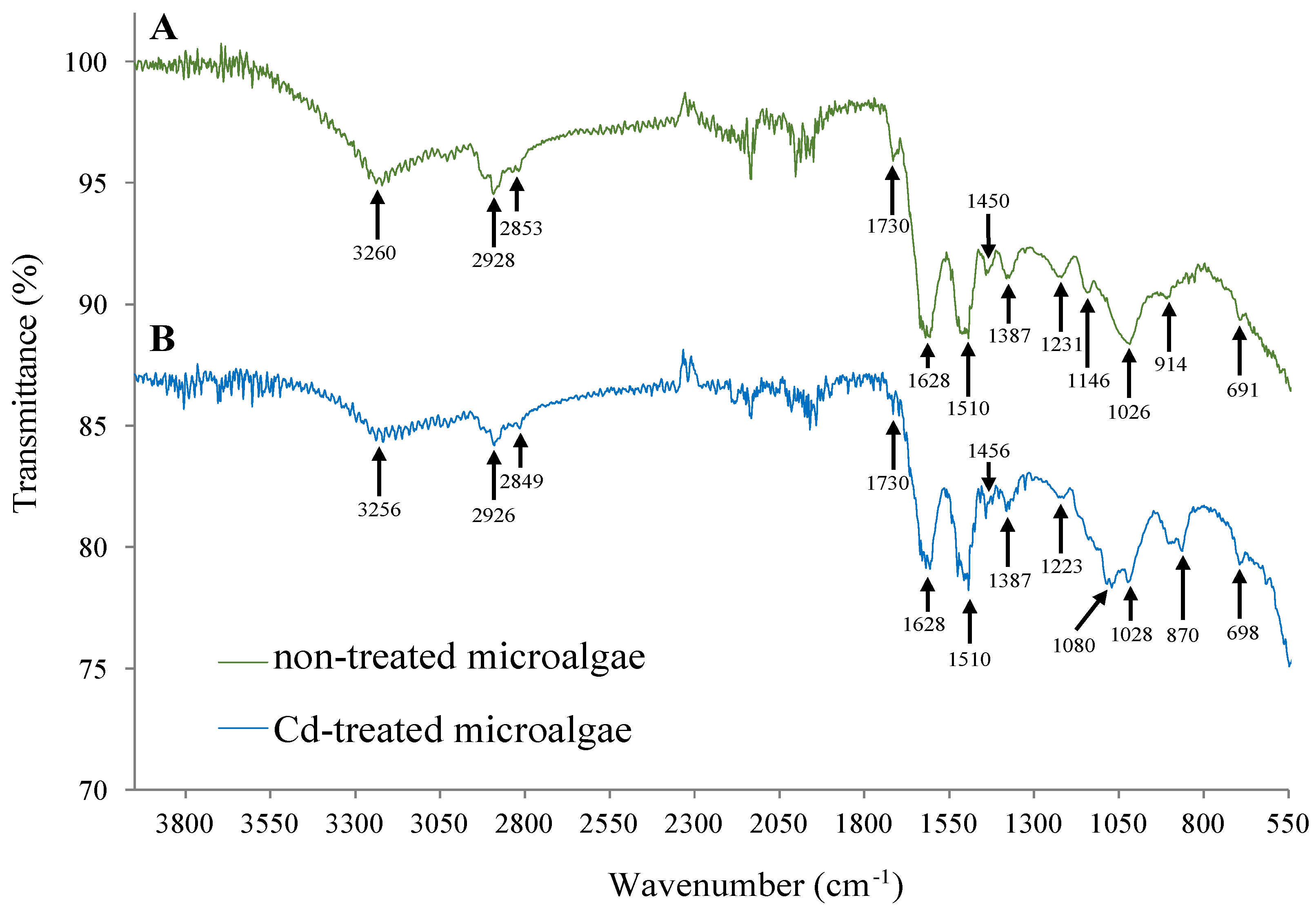
| Cd (mg/L) | First-Order Kinetics | Second-Order Kinetics | ||
|---|---|---|---|---|
| K1 (1/h) | R2 | K2 (L/mg h) | R2 | |
| 5.0 | 0.0712 | 0.9237 | 0.0242 | 0.9981 |
| 25.0 | 0.0597 | 0.8329 | 0.0038 | 0.9815 |
| Parameters | Cd (mg/L) | ||
|---|---|---|---|
| 0.0 (Control) | 5.0 | 25.0 | |
| MDA content (µmol/mg) | 7.35 ± 0.31 a | 8.42 ± 0.16 b | 9.41 ± 0.04 c |
| DHase activity (UA/mg) | 17.7 ± 0.1 a | 14.6 ± 0.1 b | 14.4 ± 0.4 b |
| Cell density (O.D.560 nm) | 0.567 ± 0.023 a | 0.558 ± 0.027 a | 0.528 ± 0.040 a |
| Total proteins (mg/L) | 118.9 ± 6.7 a | 118.0 ± 10.0 a | 113.8 ± 6.5 a |
| chl-a (µg/mg) | 53.6 ± 3.6 a | 53.7 ± 1.7 a | 51.2 ± 3.4 a |
| Carotenoids (µg/mg) | 139.3 ± 7.4 a | 119.8 ± 2.1 b | 117.6 ± 6.0 b |
| Carbohydrates (µg/mg) | 710 ± 85 a | 879 ± 36 b | 909 ± 21 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bon, I.C.; Salvatierra, L.M.; Lario, L.D.; Morató, J.; Pérez, L.M. Prospects in Cadmium-Contaminated Water Management Using Free-Living Cyanobacteria (Oscillatoria sp.). Water 2021, 13, 542. https://doi.org/10.3390/w13040542
Bon IC, Salvatierra LM, Lario LD, Morató J, Pérez LM. Prospects in Cadmium-Contaminated Water Management Using Free-Living Cyanobacteria (Oscillatoria sp.). Water. 2021; 13(4):542. https://doi.org/10.3390/w13040542
Chicago/Turabian StyleBon, Iván Carralero, Lucas M. Salvatierra, Luciana D. Lario, Jordi Morató, and Leonardo M. Pérez. 2021. "Prospects in Cadmium-Contaminated Water Management Using Free-Living Cyanobacteria (Oscillatoria sp.)" Water 13, no. 4: 542. https://doi.org/10.3390/w13040542
APA StyleBon, I. C., Salvatierra, L. M., Lario, L. D., Morató, J., & Pérez, L. M. (2021). Prospects in Cadmium-Contaminated Water Management Using Free-Living Cyanobacteria (Oscillatoria sp.). Water, 13(4), 542. https://doi.org/10.3390/w13040542







