Investigation and Optimization of Chitosan Performance in Flocculating Kaolin Suspensions Using a Real-Time Suspending Solid Concentration Measuring Method
Abstract
1. Introduction
2. Experiment
2.1. Materials
2.2. Preparation of Kaolin Suspension
2.3. Preparation of Chitosan Flocculant
2.4. Jar Test
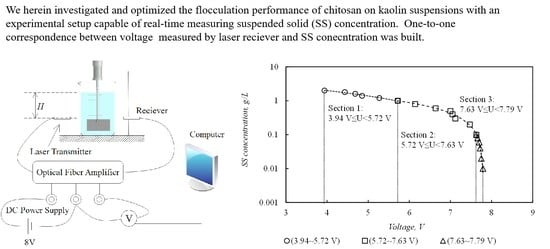
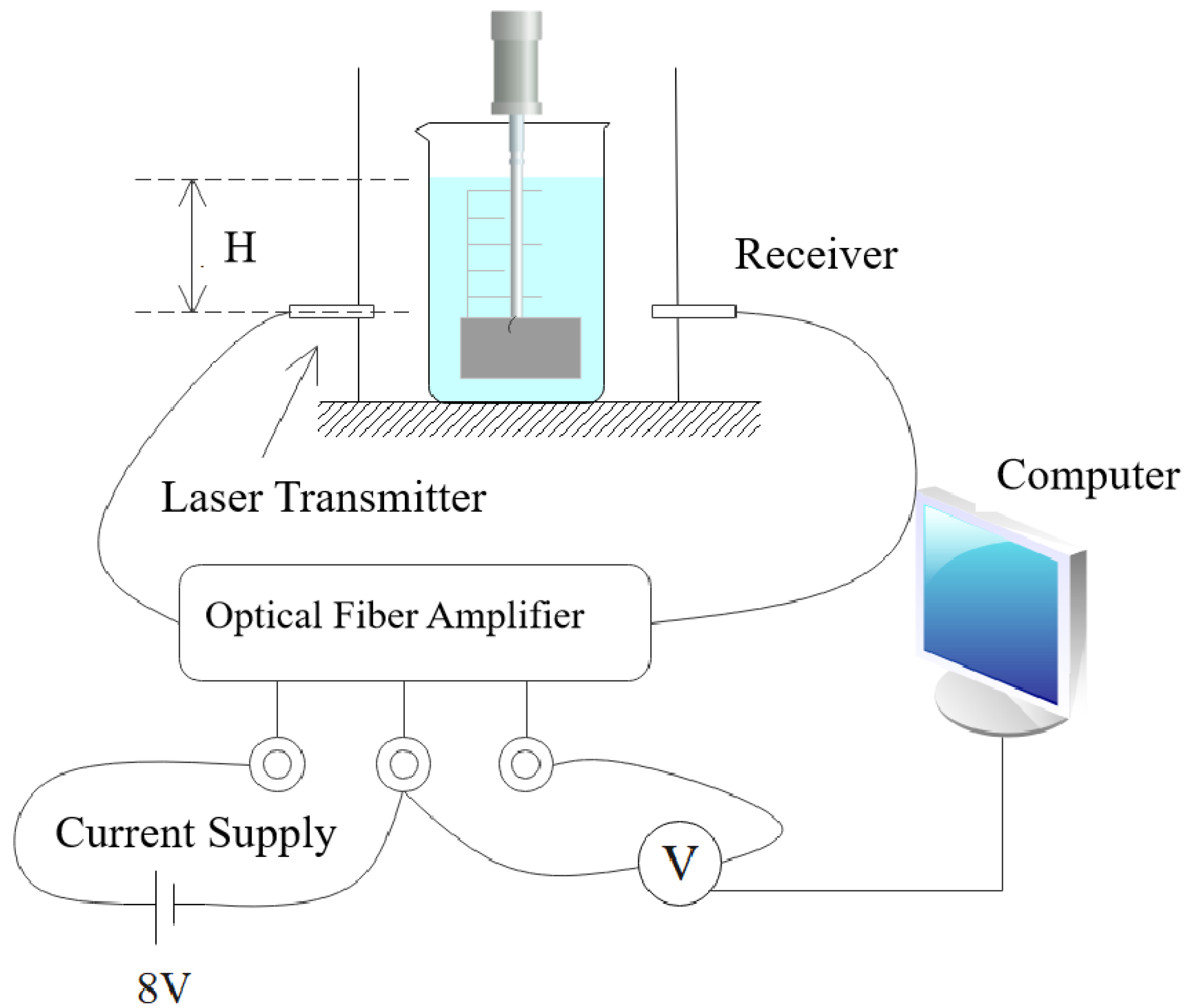
2.5. Measurement of SS Concentration
3. Results and Discussion
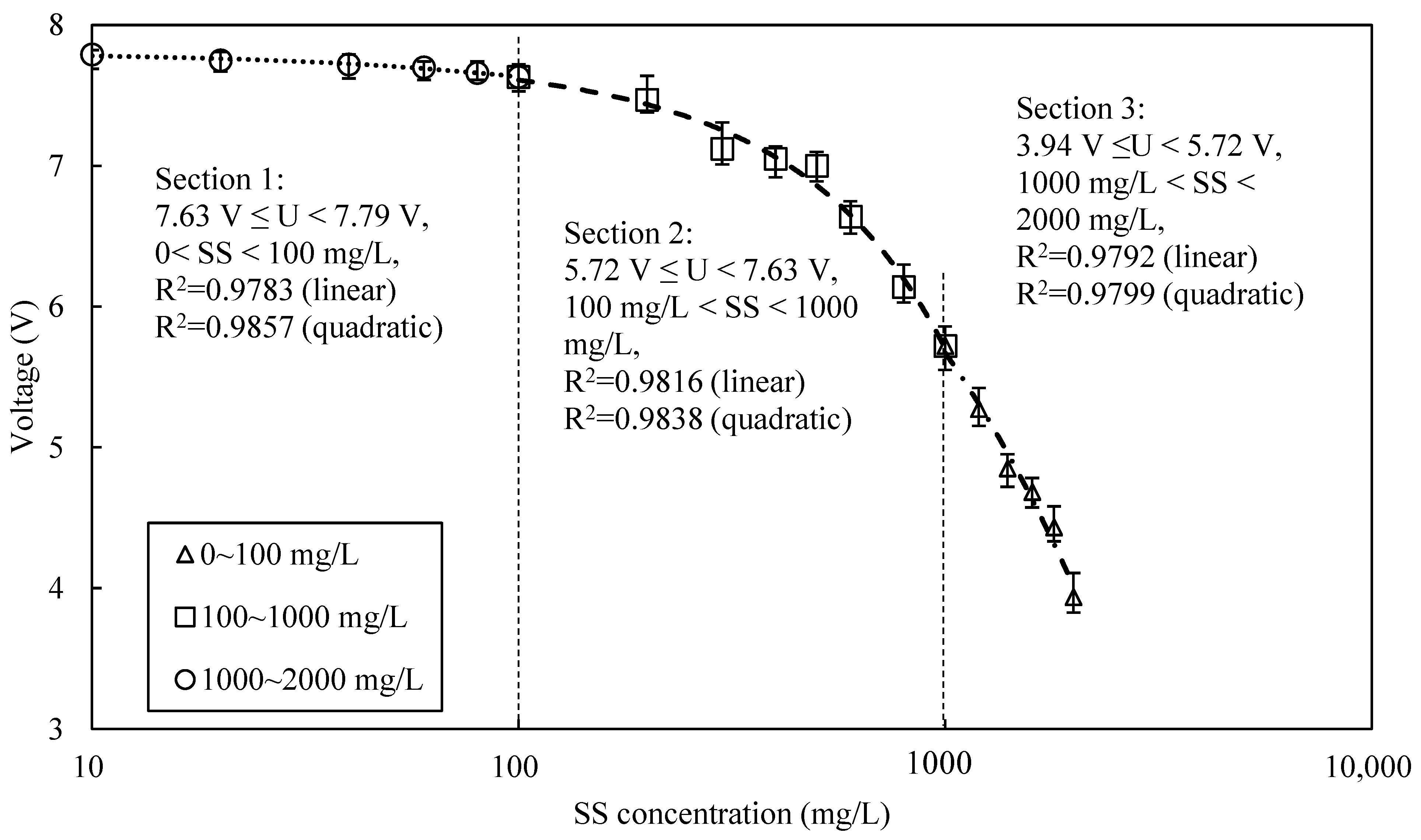
3.1. Calibration Curve
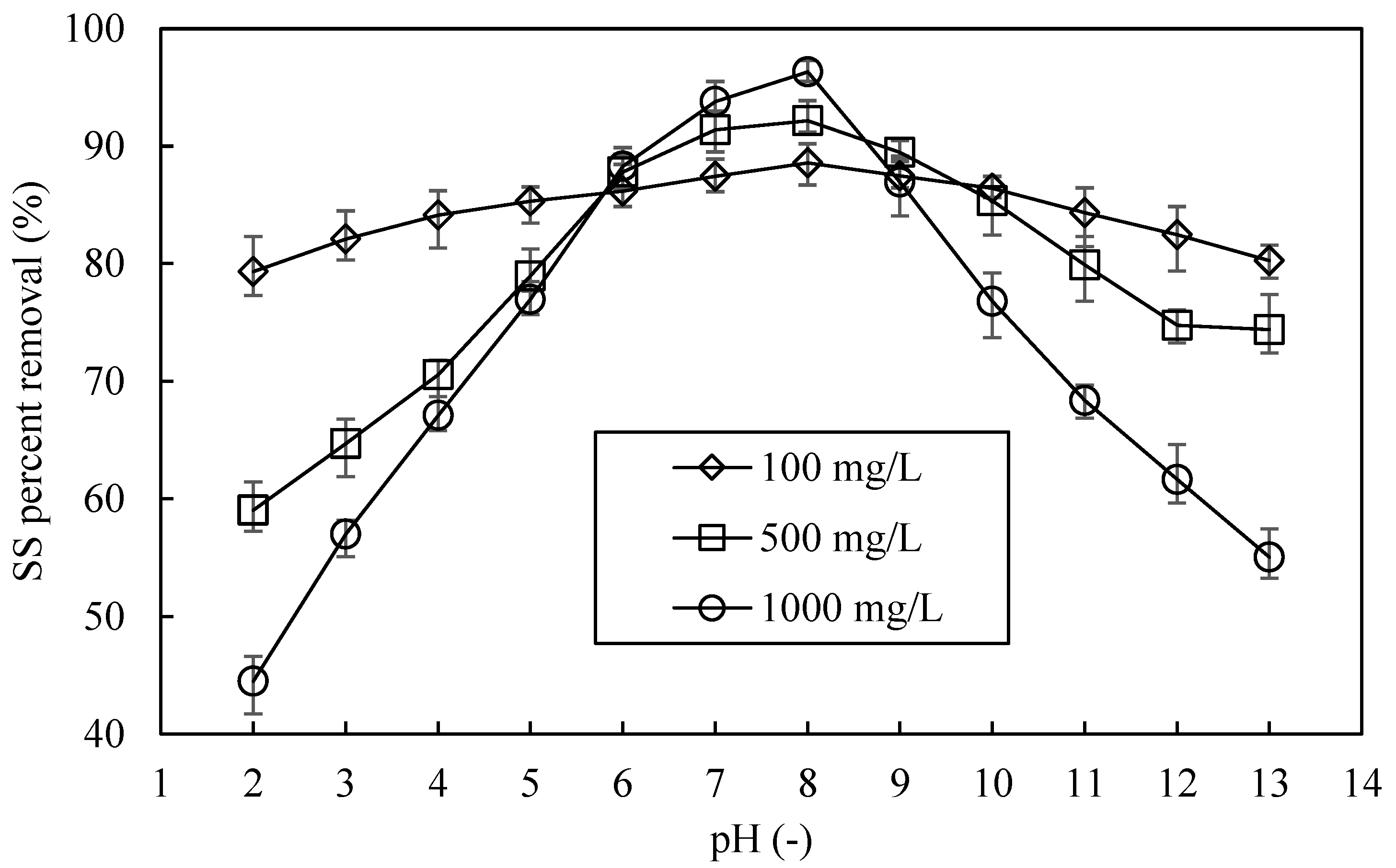
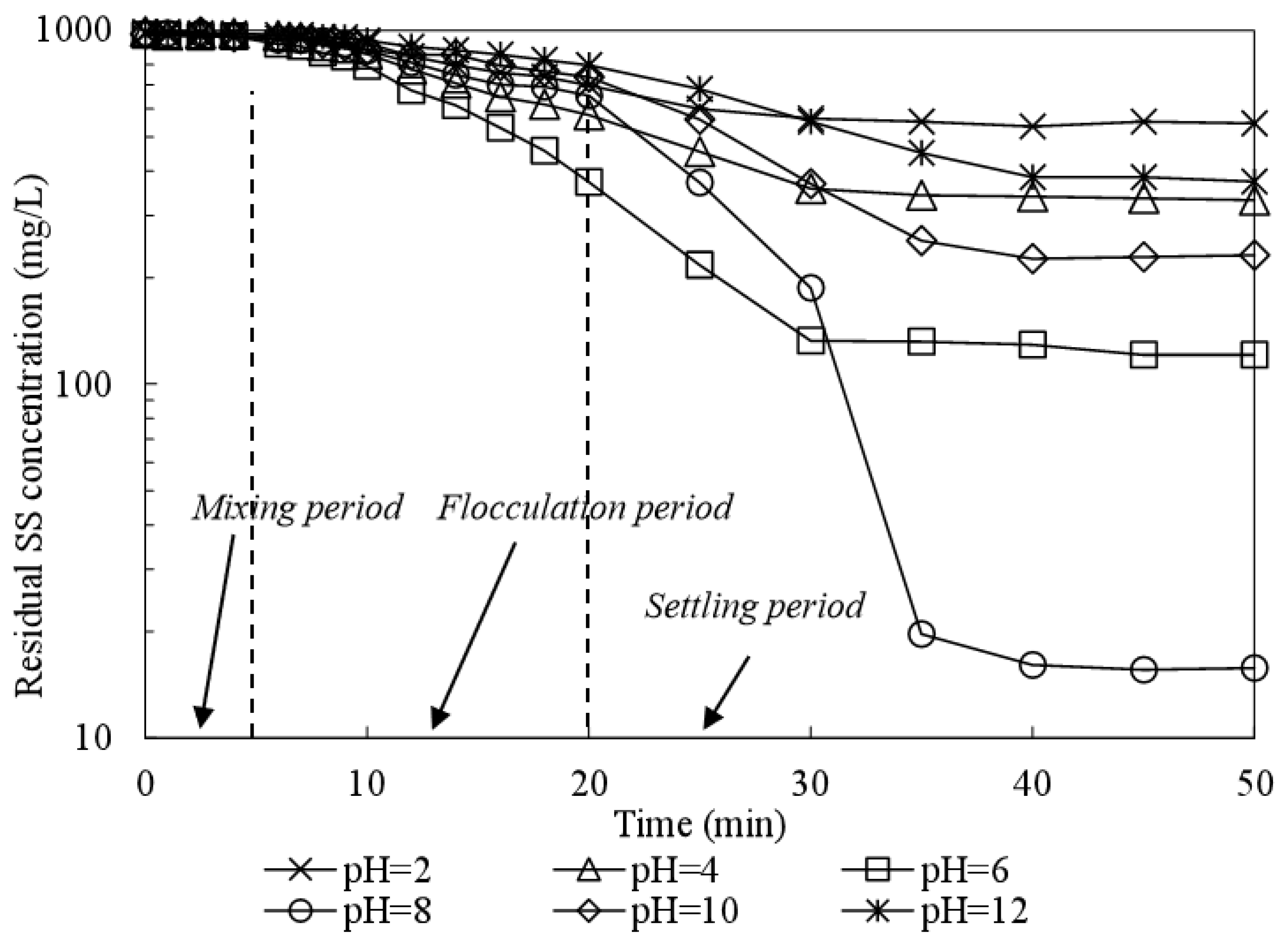
3.2. Influence of pH on SS Decantation
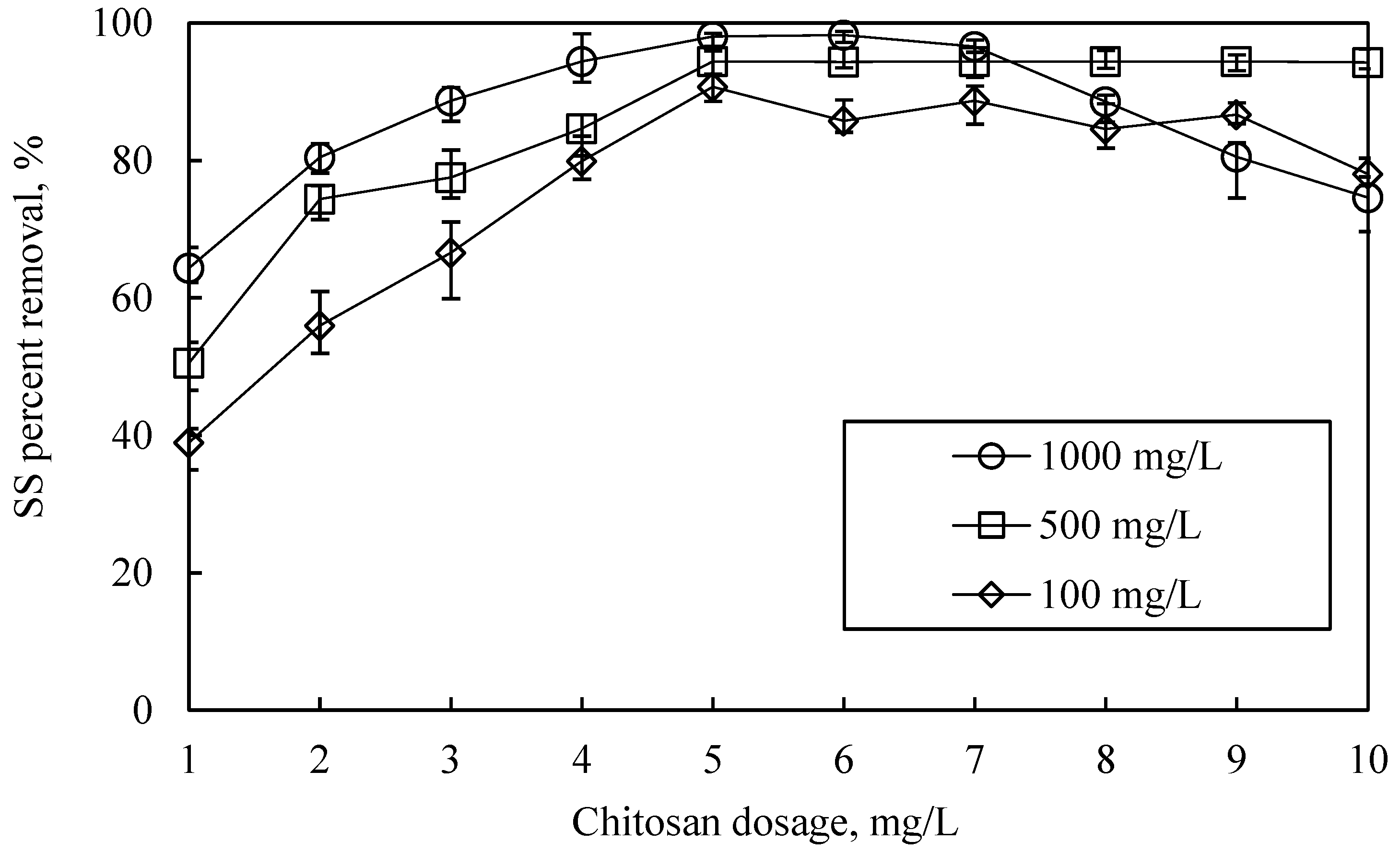
3.3. Optimization of Chitosan Dosage
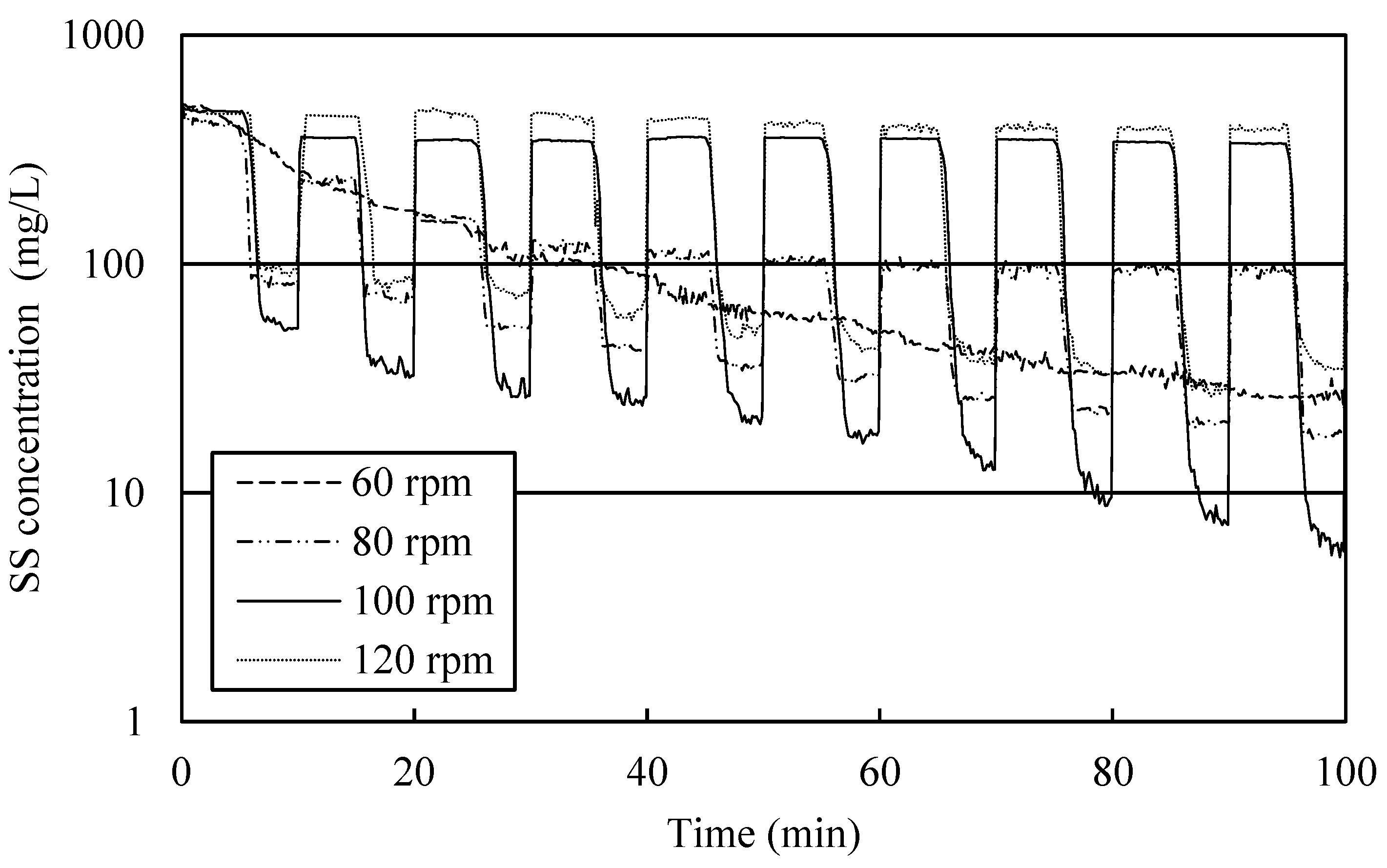
3.4. Optimization of Hydraulic Conditions
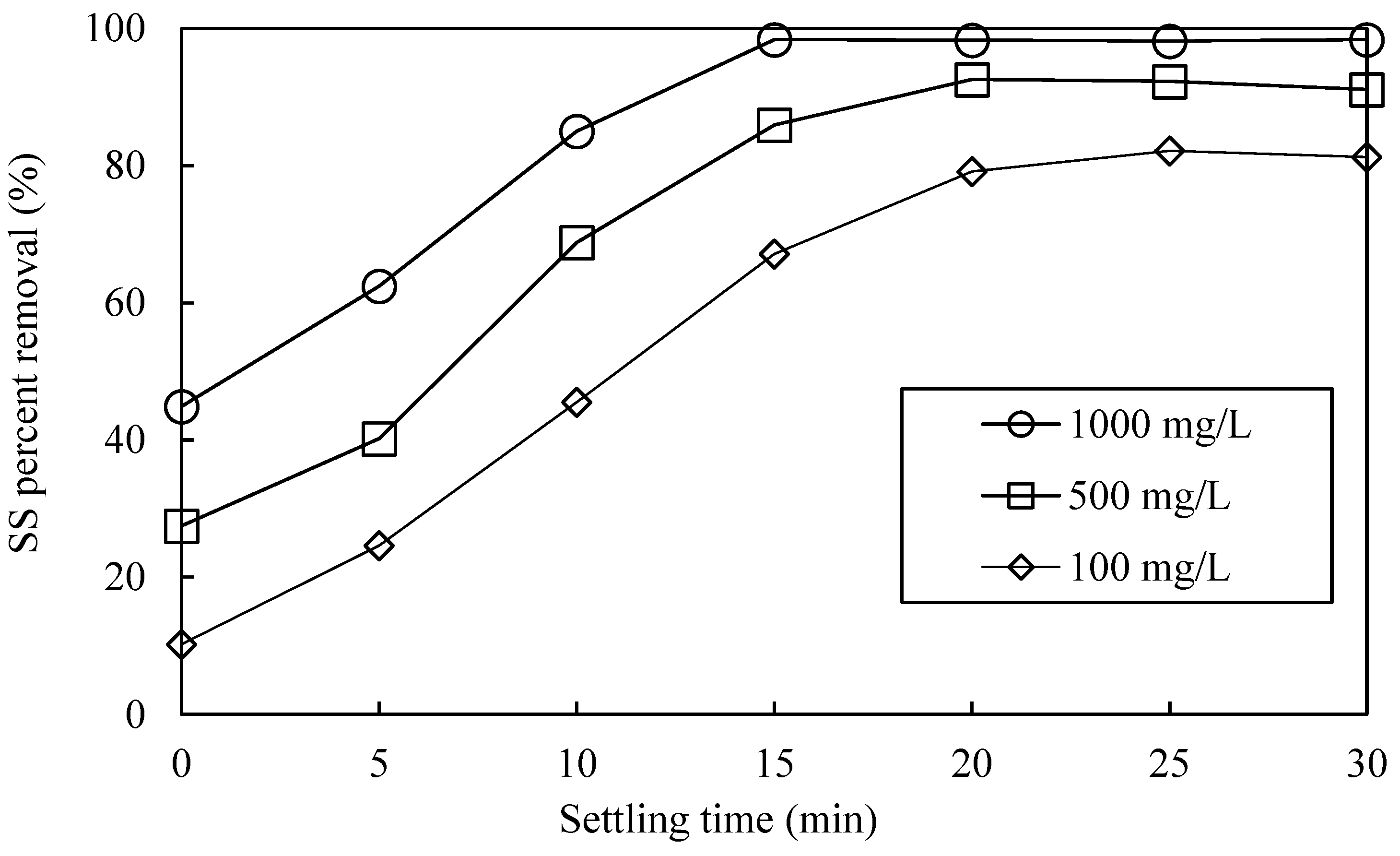
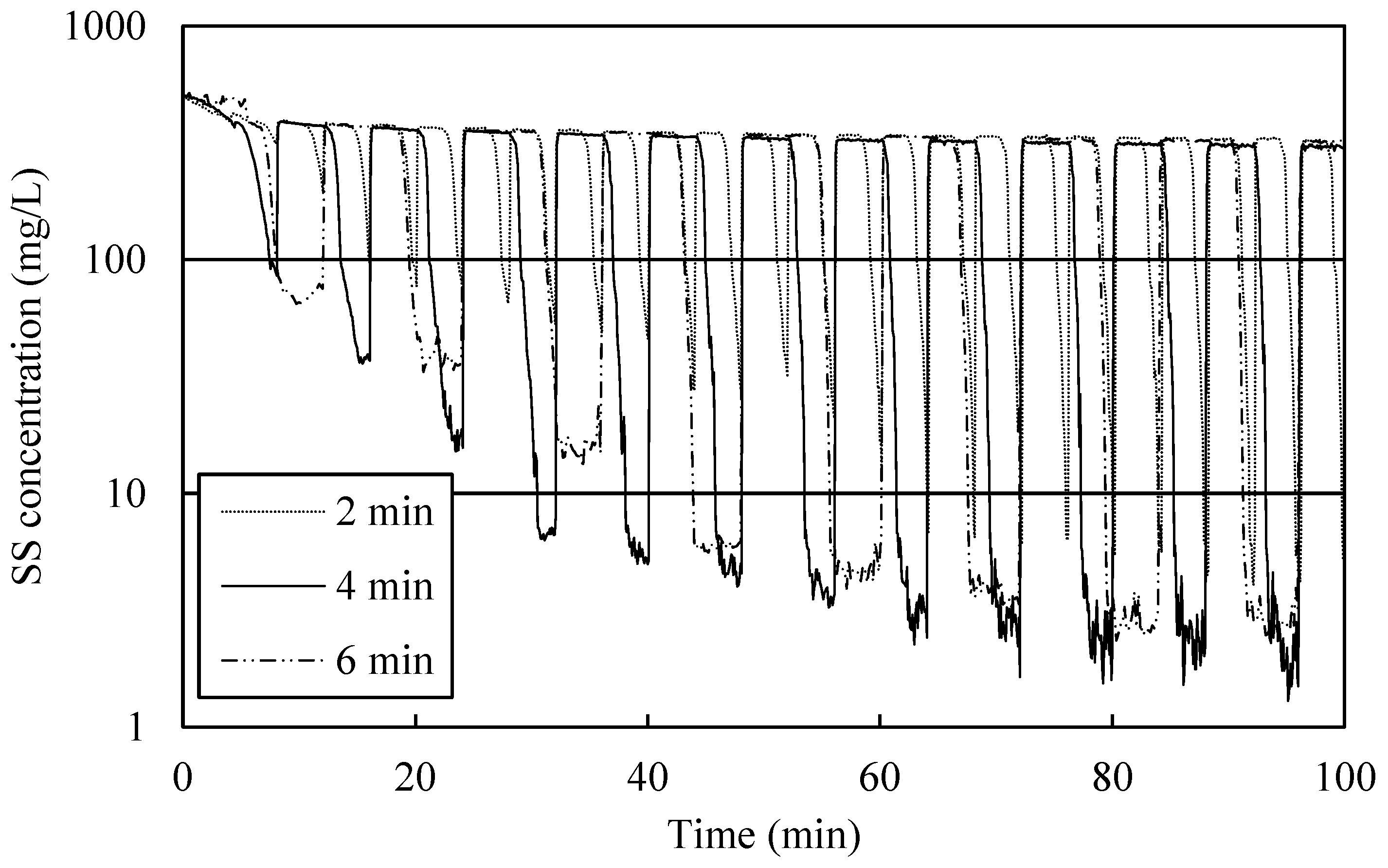
3.5. Settling Time
3.6. Influence of Intermittent Agitation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Edzwald, J.K. Coagulation in Drinking Water Treatment: Particles, Organics and Coagulants. Water Sci. Technol. 1993, 27, 21–35. [Google Scholar] [CrossRef]
- Rondeau, V.; Commenges, D. Chapter 3—The epidemiology of aluminium and Alzheimer’s disease. In Aluminium and Alzheimer’s Disease; Exley, C., Ed.; Elsevier: Amsterdam, The Netherlands, 2001; pp. 59–73. [Google Scholar]
- Campbell, A. The potential role of aluminium in Alzheimer’s disease. Nephrol. Dial. Transplant. 2002, 17, 17–20. [Google Scholar] [CrossRef]
- Renault, F.; Sancey, B.; Badot, P.-M.; Crini, G. Chitosan for coagulation/flocculation processes—An eco-friendly approach. Eur. Polym. J. 2009, 45, 1337–1348. [Google Scholar] [CrossRef]
- Pal, P. Industrial Water Treatment Process Technology; Butterworth-Heinemann: Oxford, UK, 2017. [Google Scholar]
- Bolto, B.; Gregory, J. Organic polyelectrolytes in water treatment. Water Res. 2007, 41, 2301–2324. [Google Scholar] [CrossRef]
- Bratby, J. Coagulation and Flocculation in Water and Wastewater Treatment; IWA Publishing: London, UK, 2006. [Google Scholar]
- Guibal, E.; Van Vooren, M.; Dempsey, B.A.; Roussy, J. A Review of the Use of Chitosan for the Removal of Particulate and Dissolved Contaminants. Sep. Sci. Technol. 2006, 41, 2487–2514. [Google Scholar] [CrossRef]
- Dao, V.H.; Cameron, N.R.; Saito, K. Synthesis, properties and performance of organic polymers employed in flocculation applications. Polym. Chem. 2015, 7, 11–25. [Google Scholar] [CrossRef]
- Zhong, Q.-Q.; Yue, Q.-Y.; Li, Q.; Gao, B.-Y.; Xu, X. Removal of Cu(II) and Cr(VI) from wastewater by an amphoteric sorbent based on cellulose-rich biomass. Carbohydr. Polym. 2014, 111, 788–796. [Google Scholar] [CrossRef]
- Krentz, D.-O.; Lohmann, C.; Schwarz, S.; Bratskaya, S.; Liebert, T.; Laube, J.; Heinze, T.; Kulicke, W.-M. Properties and Flocculation Efficiency of Highly Cationized Starch Derivatives. Starch-Stärke 2006, 58, 161–169. [Google Scholar] [CrossRef]
- Yang, R.; Li, H.; Huang, M.; Yang, H.; Li, A. A review on chitosan-based flocculants and their applications in water treatment. Water Res. 2016, 95, 59–89. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Dunn, E.; Grandmaison, E.; Goosen, M. Applications and Properties of Chitosan. J. Bioact. Compat. Polym. 1992, 7, 370–397. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [PubMed]
- Kettles, G.J.; Kanyuka, K. Dissecting the Molecular Interactions between Wheat and the Fungal Pathogen Zymoseptoria tritici. Front. Plant Sci. 2016, 7, 508. [Google Scholar] [CrossRef]
- Kramer, K.J.; Koga, D. Insect chitin. Insect Biochem. 1986, 16, 851–877. [Google Scholar] [CrossRef]
- Muzzarelli, R.; Muzzarelli, C. Chitosan chemistry: Relevance to the biomedical sciences, In Polysaccharides I.; Heinze, T., Ed.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 151–209. [Google Scholar]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Roussy, J.; Van Vooren, M.; Guibal, E. Influence of chitosan characteristics on coagulation and flocculation of organic suspensions. J. Appl. Polym. Sci. 2005, 98, 2070–2079. [Google Scholar] [CrossRef]
- Roussy, J.; Chastellan, P.; Van Vooren, M.; Guibal, E. Treatment of ink-containing wastewater by coagulation/flocculation using biopolymers. Water SA 2007, 31, 369–376. [Google Scholar] [CrossRef]
- Roussy, J.; Van Vooren, M.; Guibal, E. Chitosan for the Coagulation and Flocculation of Mineral Colloids. J. Dispers. Sci. Technol. 2005, 25, 663–677. [Google Scholar] [CrossRef]
- Onsøyen, E.; Skaugrud, O.; Onsosyen, E. Metal recovery using chitosan. J. Chem. Technol. Biotechnol. 2007, 49, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Zeng, D.; Wu, J.; Kennedy, J.F. Application of a chitosan flocculant to water treatment. Carbohydr. Polym. 2008, 71, 135–139. [Google Scholar] [CrossRef]
- Tran, N.V.N.; Yu, Q.J.; Nguyen, T.P.; Wang, S.-L. Coagulation of Chitin Production Wastewater from Shrimp Scraps with By-Product Chitosan and Chemical Coagulants. Polymers 2020, 12, 607. [Google Scholar] [CrossRef]
- Leea, M.D.; Leea, P.S.; Chongb, K.H. Treatment performance of palm oil mill effluent by utilizing Chitosan and ferric chloride coupled with activated carbon and ultrasound bath. Desalin. Water Treat. 2020, 174, 136–142. [Google Scholar] [CrossRef]
- Chen, F.; Liu, W.; Pan, Z.; Wang, Y.; Guo, X.; Sun, S.; Jia, R. Characteristics and mechanism of chitosan in flocculation for water coagulation in the Yellow River diversion reservoir. J. Water Process. Eng. 2020, 34, 101191. [Google Scholar] [CrossRef]
- Vidal, R.R.L.; Moraes, J.S. Removal of organic pollutants from wastewater using chitosan: A literature review. Int. J. Environ. Sci. Technol. 2019, 16, 1741–1754. [Google Scholar] [CrossRef]
- Huang, C.; Chen, S.; Pan, J.R. Optimal condition for modification of chitosan: A biopolymer for coagulation of colloidal particles. Water Res. 2000, 34, 1057–1062. [Google Scholar] [CrossRef]
- Chen, L.; Chen, D.; Wu, C. A New Approach for the Flocculation Mechanism of Chitosan. J. Polym. Environ. 2003, 11, 87–92. [Google Scholar] [CrossRef]
- Roussy, J.; Van Vooren, M.; Dempsey, B.A.; Guibal, E. Influence of chitosan characteristics on the coagulation and the flocculation of bentonite suspensions. Water Res. 2005, 39, 3247–3258. [Google Scholar] [CrossRef]
- Li, J.; Song, X.; Pan, J.; Zhong, L.; Jiao, S.; Ma, Q. Adsorption and flocculation of bentonite by chitosan with varying degree of deacetylation and molecular weight. Int. J. Biol. Macromol. 2013, 62, 4–12. [Google Scholar] [CrossRef]
- Li, J.; Jiao, S.; Zhong, L.; Pan, J.; Ma, Q. Optimizing coagulation and flocculation process for kaolinite suspension with chitosan. Colloids Surf. A Physicochem. Eng. Asp. 2013, 428, 100–110. [Google Scholar] [CrossRef]
- Likens, G.; Benbow, M.; Burton, T.; van Donk, E.; Downing, J.; Gulati, R. Encyclopedia of Inland Waters; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Altaher, H. The use of chitosan as a coagulant in the pre-treatment of turbid sea water. J. Hazard. Mater. 2012, 233–234, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Hannouche, A.; Chebbo, G.; Ruban, G.; Tassin, B.; Lemaire, B.J.; Joannis, C. Relationship between turbidity and total suspended solids concentration within a combined sewer system. Water Sci. Technol. 2011, 64, 2445–2452. [Google Scholar] [CrossRef] [PubMed]
- Daphne, L.H.X.; Utomo, H.D.; Kenneth, L.Z.H. Correlation between turbidity and total suspended solids in Singapore rivers. J. Water Sustain. 2011, 1, 313–322. [Google Scholar]
- Fujisaki, K. Comparison of Flocculation Properties with Use of a New Flocculation Tester. Mod. Environ. Sci. Eng. 2016, 1, 304–310. [Google Scholar] [CrossRef]
- Fujisaki, K. Experimental study on flocculation performance of Chitosan-Based Flocculant using a Novel Jar Tester. J. Civ. Eng. Environ. Sci. 2018, 4, 038–043. [Google Scholar] [CrossRef]
- Qin, C.; Li, H.; Xiao, Q.; Liu, Y.; Zhu, J.; Du, Y. Water-solubility of chitosan and its antimicrobial activity. Carbohydr. Polym. 2006, 63, 367–374. [Google Scholar] [CrossRef]
- Gupta, V.; Miller, J.D. Surface force measurements at the basal planes of ordered kaolinite particles. J. Colloid Interface Sci. 2010, 344, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Varga, G. The structure of kaolinite and metakaolinite. Epitoanyag 2007, 59, 6–9. [Google Scholar] [CrossRef]
- Yang, Z.; Yang, H.; Jiang, Z.; Cai, T.; Li, H.; Li, H.; Li, A.; Cheng, R. Flocculation of both anionic and cationic dyes in aqueous solutions by the amphoteric grafting flocculant carboxymethyl chitosan-graft-polyacrylamide. J. Hazard. Mater. 2013, 254–255, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Gregory, J. Coagulation by hydrolysing metal salts. Adv. Colloid Interface Sci. 2003, 100, 475–502. [Google Scholar] [CrossRef]
- Yang, Z.; Shang, Y.; Huang, X.; Chen, Y.; Lu, Y.; Chen, A.; Jiang, Y.; Gu, W.; Qian, X.; Yang, H.; et al. Cationic content effects of biodegradable amphoteric chitosan-based flocculants on the flocculation properties. J. Environ. Sci. 2012, 24, 1378–1385. [Google Scholar] [CrossRef]
- Yang, Z.; Shang, Y.; Lu, Y.; Chen, Y.; Huang, X.; Chen, A.; Jiang, Y.; Gu, W.; Qian, X.; Yang, H.; et al. Flocculation properties of biodegradable amphoteric chitosan-based flocculants. Chem. Eng. J. 2011, 172, 287–295. [Google Scholar] [CrossRef]
- Sun, J.; Qin, L.; Li, G.; Kang, Y. Effect of hydraulic conditions on flocculation performances and floc characteristics in Chinese herbal extracts by chitosan and chitosan hydrochloride. Chem. Eng. J. 2013, 225, 641–649. [Google Scholar] [CrossRef]
- Wheeler, R.E.; Atkinson, A.C.; Donev, A.N. Optimum Experimental Designs. Technometrics 1993, 35, 450. [Google Scholar] [CrossRef]
- Weissman, S.A.; Anderson, N.G. Design of Experiments (DoE) and Process Optimization. A Review of Recent Publications. Org. Process. Res. Dev. 2015, 19, 1605–1633. [Google Scholar] [CrossRef]
- Aydın, Y.A.; Aksoy, N.D. Adsorption of chromium on chitosan: Optimization, kinetics and thermodynamics. Chem. Eng. J. 2009, 151, 188–194. [Google Scholar] [CrossRef]
- Maazinejad, B.; Mohammadnia, O.; Ali, G.A.; Makhlouf, A.S.; Nadagouda, M.N.; Sillanpää, M.; Asiri, A.M.; Agarwal, S.; Gupta, V.K.; Sadegh, H. Taguchi L9 (34) orthogonal array study based on methylene blue removal by single-walled carbon nanotubes-amine: Adsorption optimization using the experimental design method, kinetics, equilibrium and thermodynamics. J. Mol. Liq. 2020, 298, 112001. [Google Scholar] [CrossRef]
- Bratskaya, S.; Avramenko, V.; Schwarz, S.; Philippova, I. Enhanced flocculation of oil-in-water emulsions by hydrophobically modified chitosan derivatives. Colloids Surf. A Physicochem. Eng. Asp. 2006, 275, 168–176. [Google Scholar] [CrossRef]
- Gupta, V. Surface Charge Features of Kaolinite Particles and their Interactions. Ph.D. Thesis, The University of Utah, Salt Lake City, UT, USA, 2011. [Google Scholar]
| Levels | Factors | |||
|---|---|---|---|---|
| Paddle Rotation Speed in the Mixing Period (RS1) | Mixing Period Time (T1) | Paddle Rotation Speed in the Flocculation Period (RS2) | Flocculation Period Time (T2) | |
| A (rpm) | B (min) | C (rpm) | D (min) | |
| 1 | 100 | 3 | 30 | 10 |
| 2 | 150 | 5 | 50 | 15 |
| 3 | 200 | 7 | 70 | 20 |
| Test Number | Factors | Percent Removal of SS (%) | |||||
|---|---|---|---|---|---|---|---|
| A | B | C | D | 1 | 2 | Sum | |
| 1 | 1 | 1 | 1 | 1 | 85.83 | 85.31 | 171.14 |
| 2 | 1 | 2 | 2 | 2 | 86.52 | 86.14 | 172.66 |
| 3 | 1 | 3 | 3 | 3 | 87.56 | 88.30 | 175.86 |
| 4 | 2 | 1 | 2 | 3 | 97.46 | 97.51 | 194.97 |
| 5 | 2 | 2 | 3 | 1 | 98.54 | 98.66 | 197.20 |
| 6 | 2 | 3 | 1 | 2 | 98.22 | 98.12 | 196.34 |
| 7 | 3 | 1 | 3 | 2 | 87.92 | 87.74 | 175.66 |
| 8 | 3 | 2 | 1 | 3 | 88.63 | 88.54 | 177.17 |
| 9 | 3 | 3 | 2 | 1 | 85.43 | 85.11 | 170.54 |
| K1j | 519.66 | 541.77 | 544.65 | 538.88 | 1631.54 | ||
| K2j | 588.51 | 547.03 | 538.17 | 544.66 | |||
| K3j | 523.37 | 542.74 | 548.72 | 548.00 | |||
| Range | 68.85 | 5.26 | 10.55 | 9.12 | |||
| Source | Sum of Squares | Degrees of Freedom | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| A | 499.85 | 2 | 249.93 | 3967.06 | ** <0.0001 |
| B | 2.61 | 2 | 1.31 | 20.71 | ** 0.0004 |
| C | 9.44 | 2 | 4.72 | 74.92 | ** <0.0001 |
| D | 7.10 | 2 | 3.55 | 56.35 | ** <0.0001 |
| Error | 0.57 | 9 | |||
| Total | 519.57 | 17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Y.; Gan, K.; Liang, S.; Liu, H.; Wang, Q. Investigation and Optimization of Chitosan Performance in Flocculating Kaolin Suspensions Using a Real-Time Suspending Solid Concentration Measuring Method. Water 2021, 13, 513. https://doi.org/10.3390/w13040513
Xu Y, Gan K, Liang S, Liu H, Wang Q. Investigation and Optimization of Chitosan Performance in Flocculating Kaolin Suspensions Using a Real-Time Suspending Solid Concentration Measuring Method. Water. 2021; 13(4):513. https://doi.org/10.3390/w13040513
Chicago/Turabian StyleXu, Yan, Kemin Gan, Siyi Liang, Haitao Liu, and Qunhui Wang. 2021. "Investigation and Optimization of Chitosan Performance in Flocculating Kaolin Suspensions Using a Real-Time Suspending Solid Concentration Measuring Method" Water 13, no. 4: 513. https://doi.org/10.3390/w13040513
APA StyleXu, Y., Gan, K., Liang, S., Liu, H., & Wang, Q. (2021). Investigation and Optimization of Chitosan Performance in Flocculating Kaolin Suspensions Using a Real-Time Suspending Solid Concentration Measuring Method. Water, 13(4), 513. https://doi.org/10.3390/w13040513