Assessing Chlorophyll a Spatiotemporal Patterns Combining In Situ Continuous Fluorometry Measurements and Landsat 8/OLI Data across the Barataria Basin (Louisiana, USA)
Abstract
1. Introduction
2. Materials and Methods
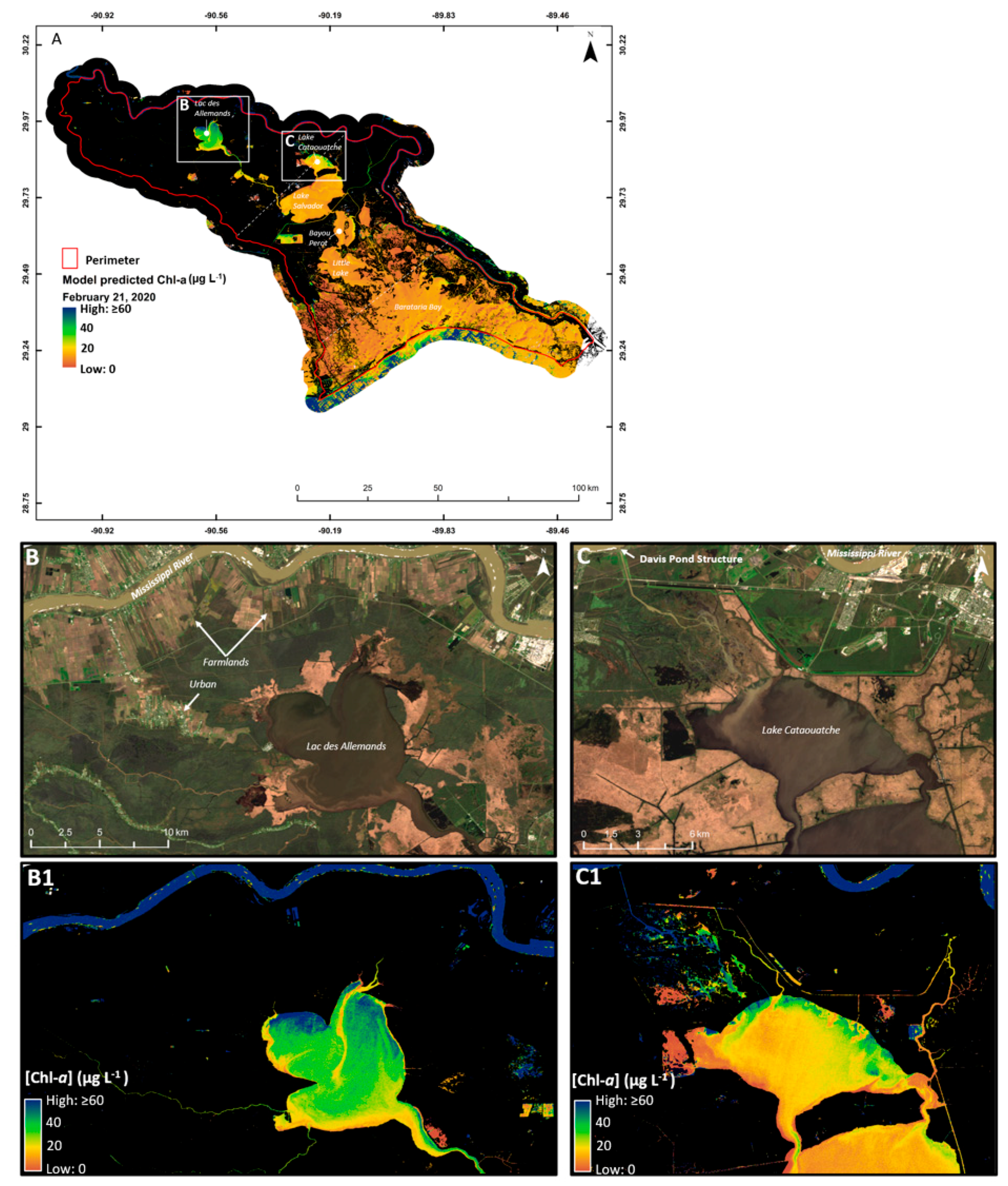
2.1. Area Description
2.2. Field and Laboratory Methods
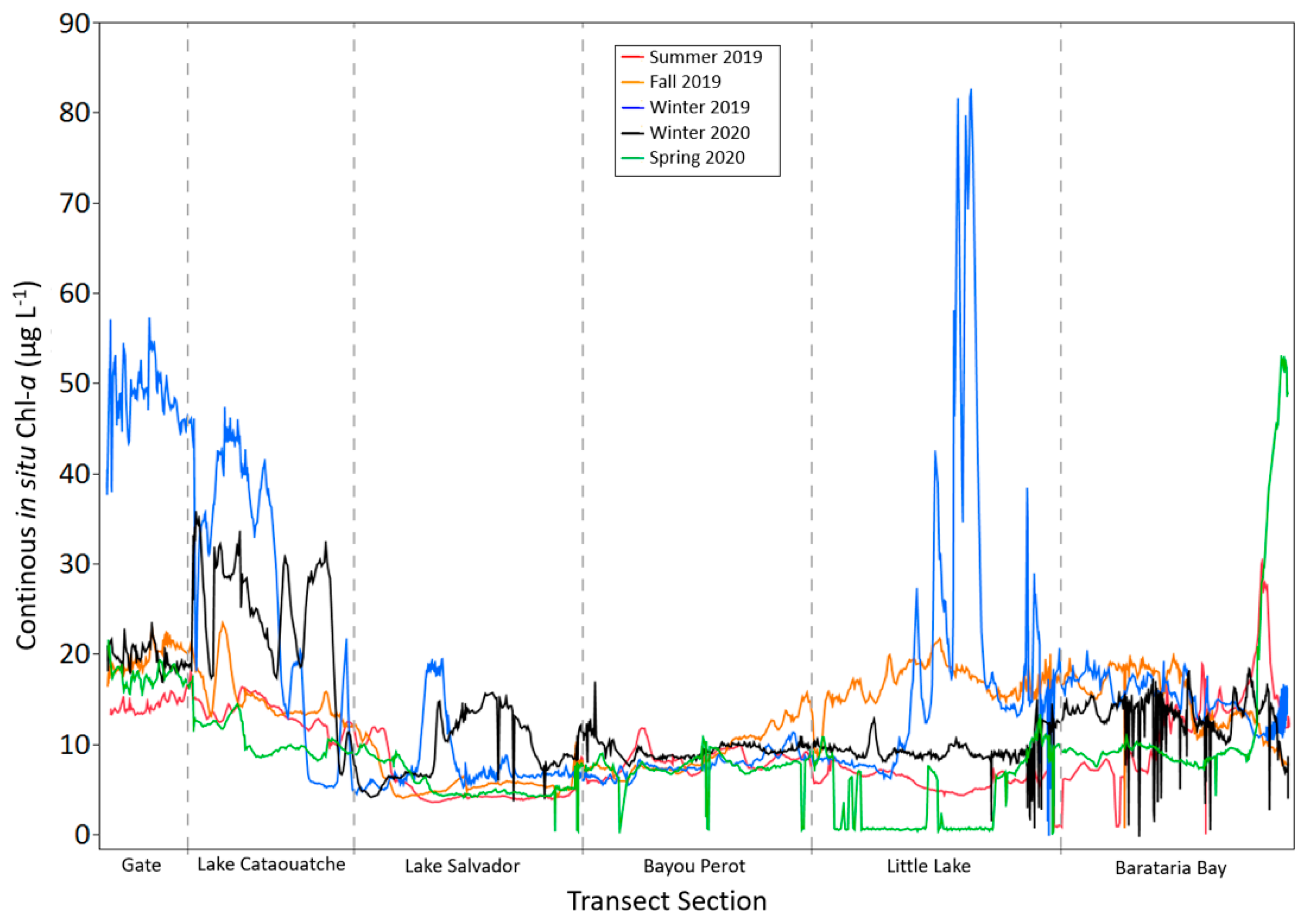
2.2.1. In-Situ Continuous Sampling
2.2.2. In-Situ Discrete Sampling
2.2.3. Satellite Imagery Acquisition and Processing
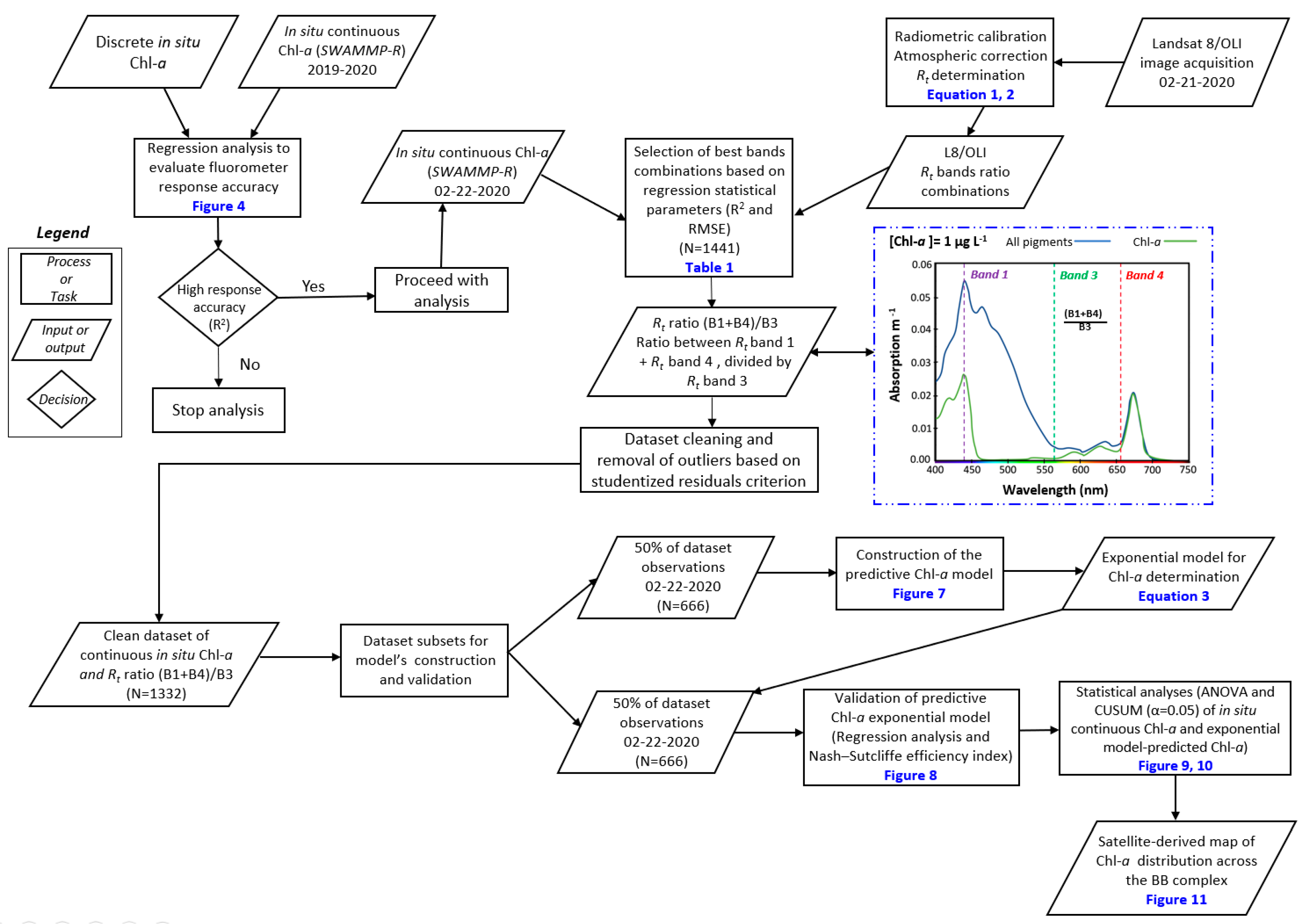
2.2.4. Remote Sensing-based Algorithms to Estimate Chl-a Concentrations
3. Results
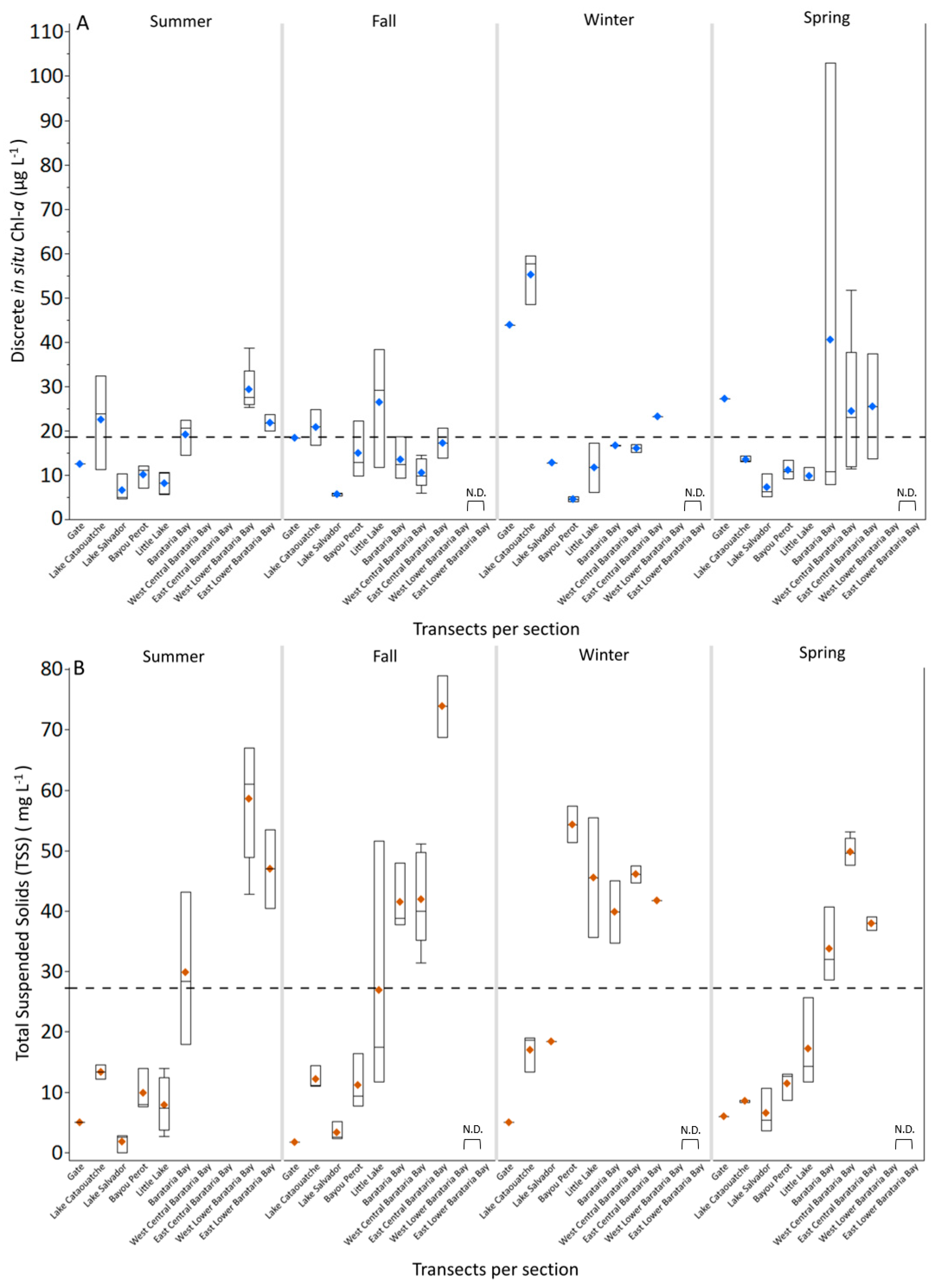
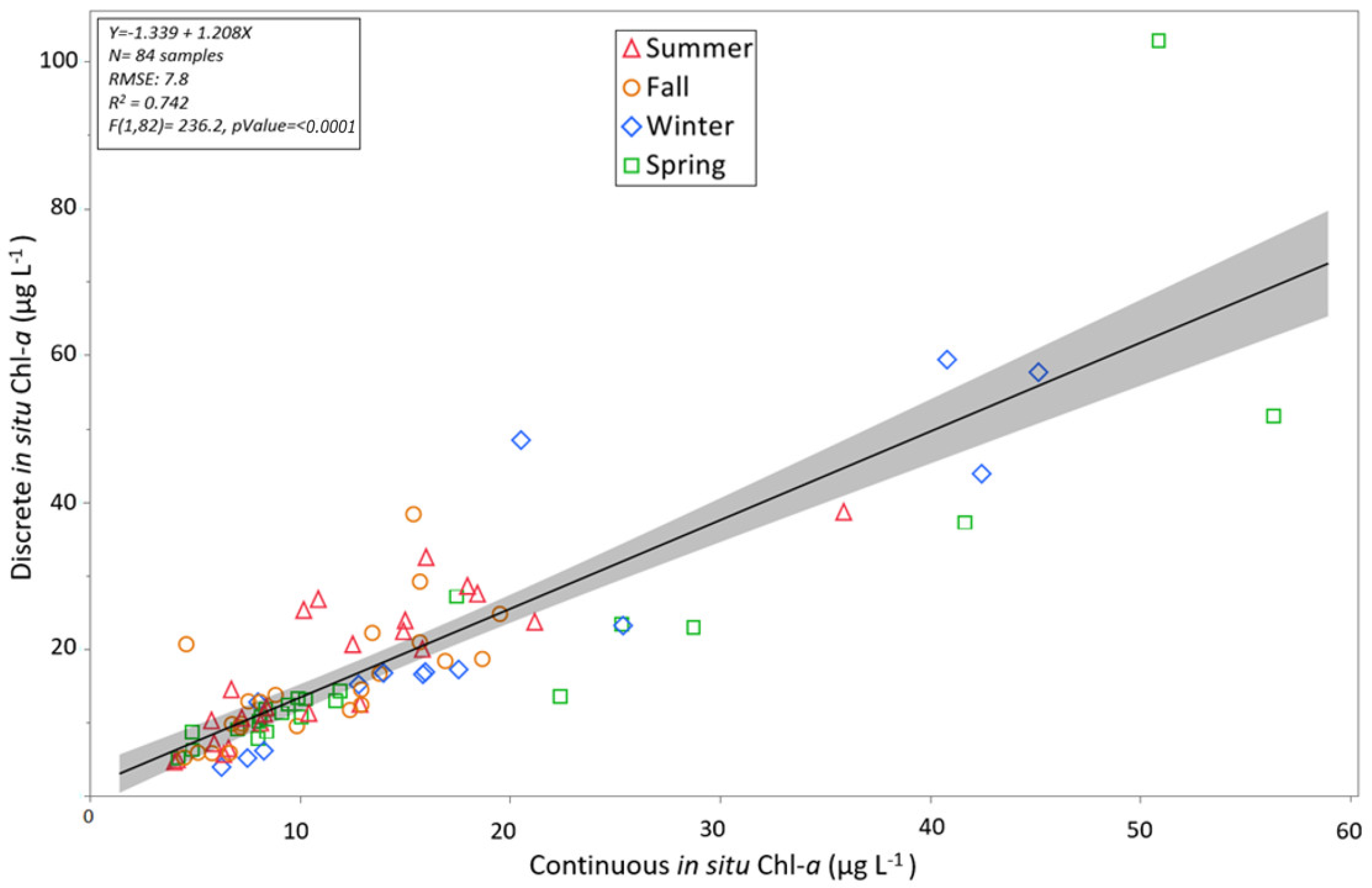
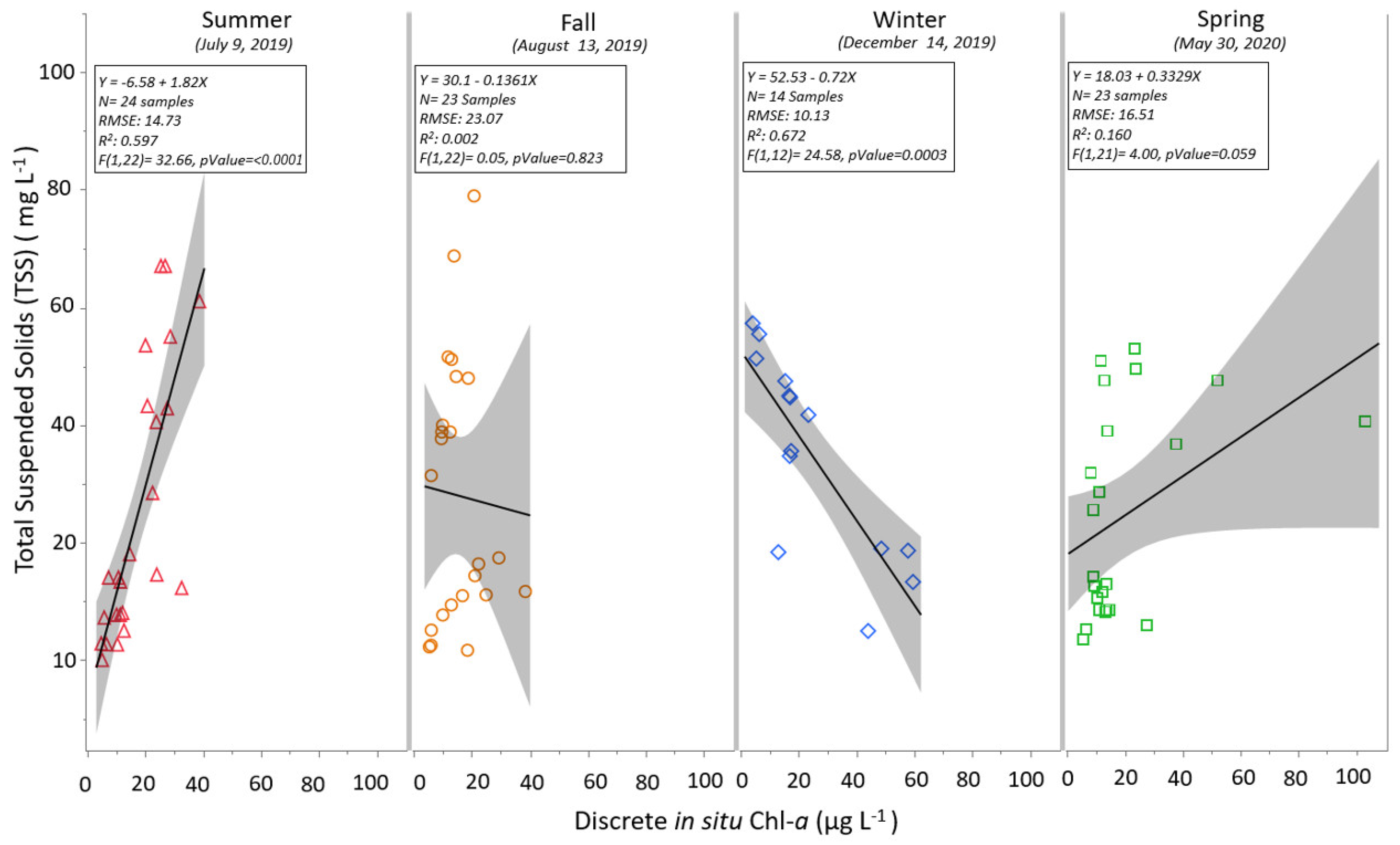
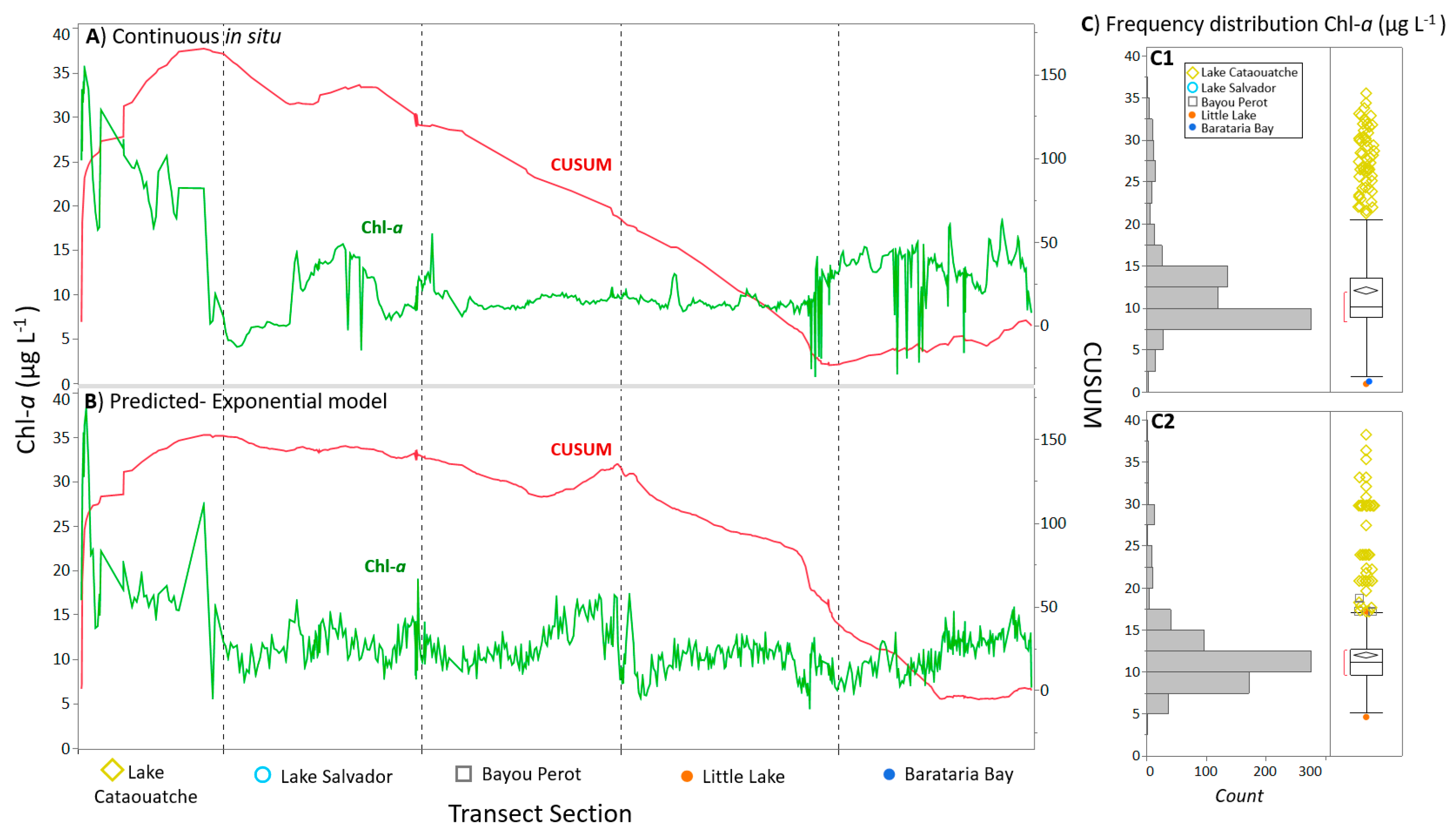
3.1. Discrete and Continuous Chl-a Data Statistical Summary
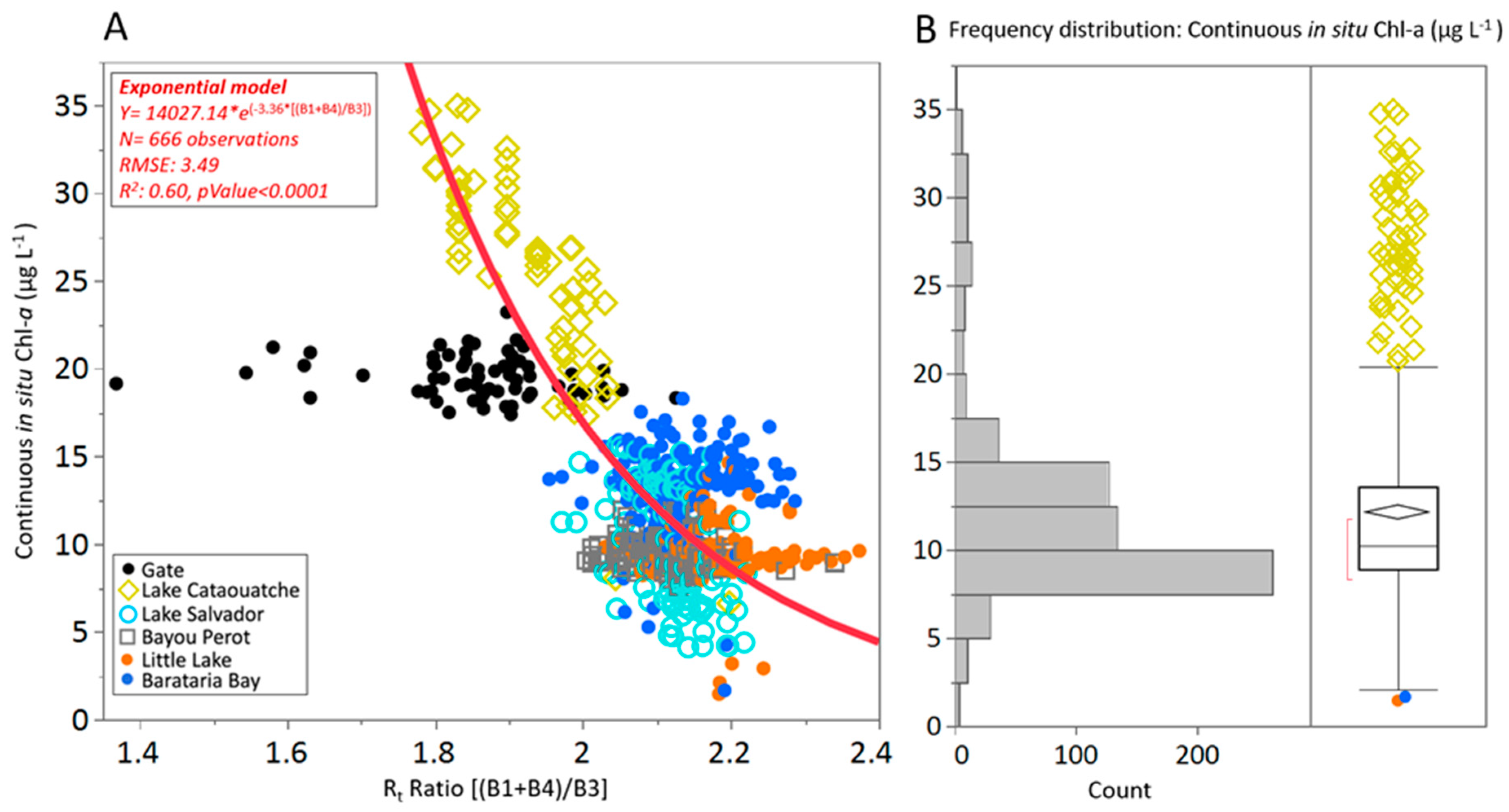
3.2. Determining the Best Band Ratio and Algorithm Development Application
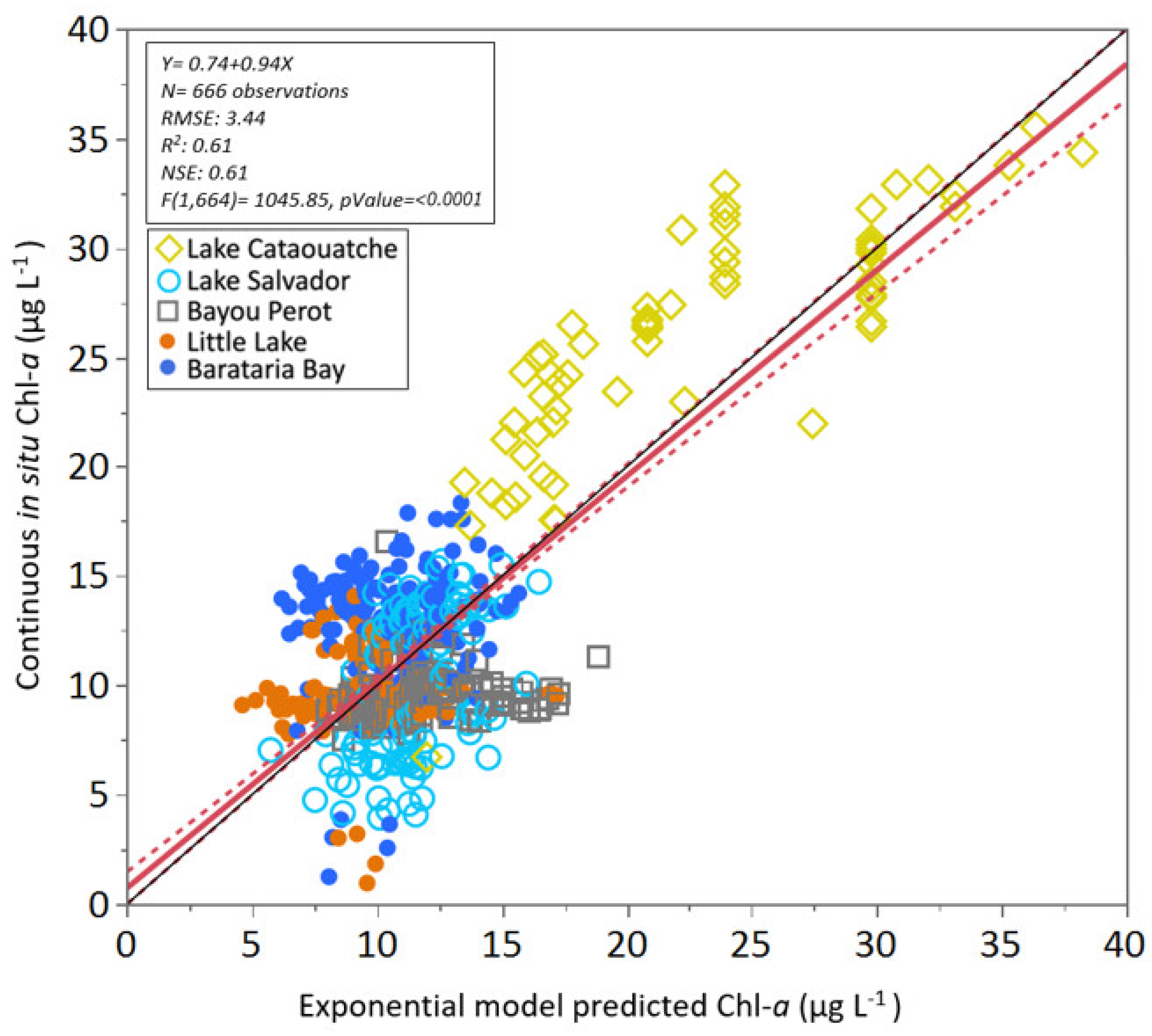
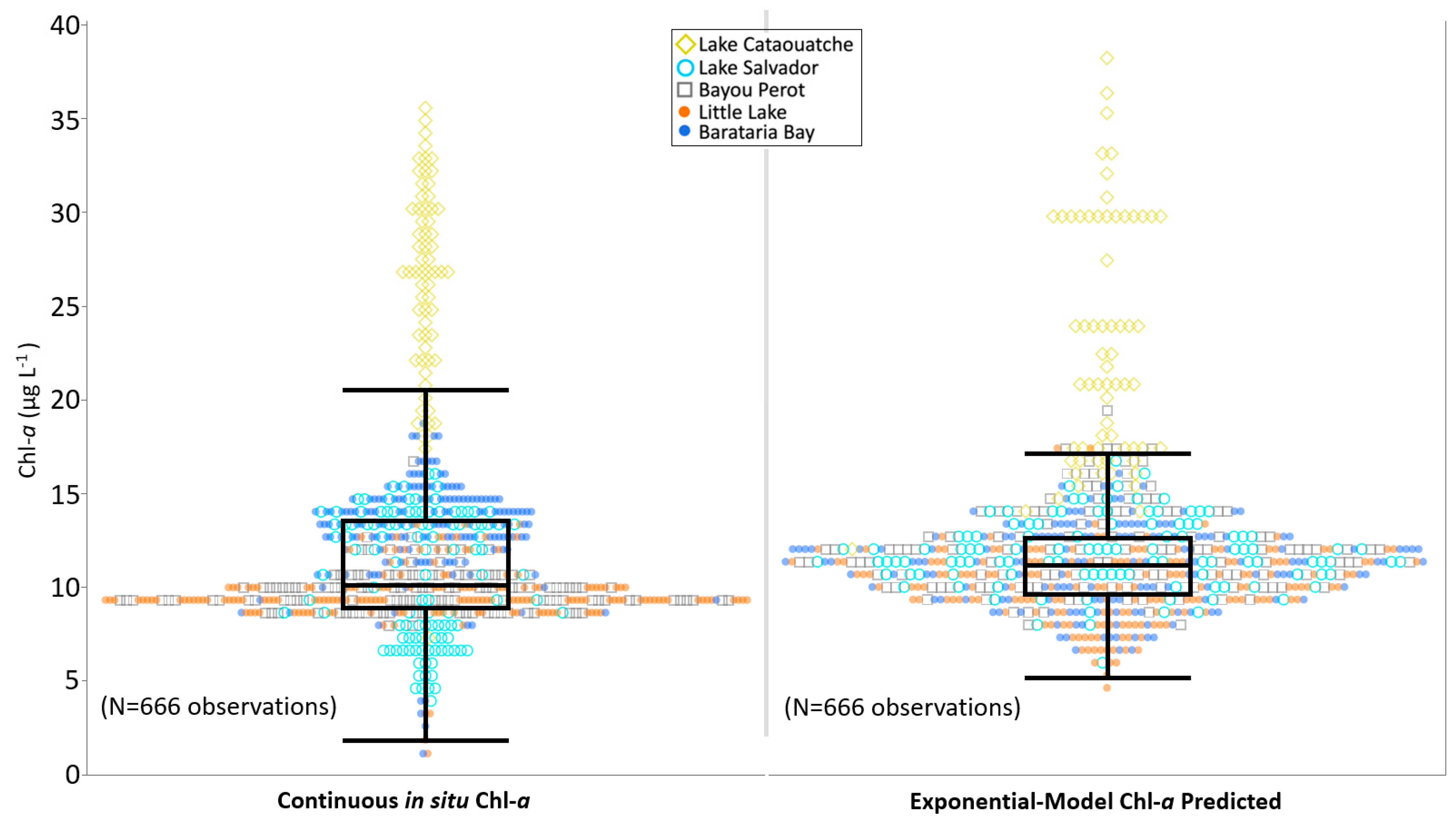
3.3. Algorithm Validation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Bianchi, T.S. Biogeochemistry of Estuaries; Oxford University Press on Demand: Oxford, UK, 2007. [Google Scholar]
- Teichert, N.; Borja, A.; Chust, G.; Uriarte, A.; Lepage, M. Restoring fish ecological quality in estuaries: Implication of interactive and cumulative effects among anthropogenic stressors. Sci. Total Environ. 2016, 542, 383–393. [Google Scholar] [CrossRef]
- Vasconcelos, R.P.; Reis-Santos, P.; Fonseca, V.; Maia, A.; Ruano, M.; França, S.; Vinagre, C.; Costa, M.J.; Cabral, H. Assessing anthropogenic pressures on estuarine fish nurseries along the Portuguese coast: A multi-metric index and conceptual approach. Sci. Total Environ. 2007, 374, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Arend, K.K.; Beletsky, D.; DePinto, J.V.; Ludsin, S.A.; Roberts, J.J.; Rucinski, D.K.; Scavia, D.; Schwab, D.J.; Höök, T.O. Seasonal and interannual effects of hypoxia on fish habitat quality in central Lake Erie. Freshw. Biol. 2011, 56, 366–383. [Google Scholar] [CrossRef]
- O’Hare, M.T.; Baattrup-Pedersen, A.; Baumgarte, I.; Freeman, A.; Gunn, I.D.; Lázár, A.N.; Sinclair, R.; Wade, A.J.; Bowes, M.J. Responses of aquatic plants to eutrophication in rivers: A revised conceptual model. Front. Plant Sci. 2018, 9, 451. [Google Scholar] [CrossRef]
- Smith, V.H. Eutrophication of freshwater and coastal marine ecosystems a global problem. Environ. Sci. Pollut. Res. 2003, 10, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.E.; Rabalais, N.N. Coastal eutrophication near the Mississippi river delta. Nature 1994, 368, 619–621. [Google Scholar] [CrossRef]
- Walker, N.D.; Rabalais, N.N. Relationships among satellite chlorophylla, river inputs, and hypoxia on the Louisiana Continental shelf, Gulf of Mexico. Estuaries Coasts 2006, 29, 1081–1093. [Google Scholar] [CrossRef]
- Bricker, S.; Longstaff, B.; Dennison, W.; Jones, A.; Boicourt, K.; Wicks, C.; Woerner, J. Effects of nutrient enrichment in the nation’s estuaries: A decade of change. In NOAA Coastal Ocean Program Decision Analysis Series No. 26; National Ocean Service: Silver Spring, MD, USA, 2007; p. 328. [Google Scholar]
- Bricker, S.; Clement, C.; Pirhalla, D.; Orlando, S.; Farrow, D. National estuarine eutrophication assessment. In Effects of Nutrient Enrichment in the Nation’s Estuaries; NOAA, National Ocean Service: Silver Spring, MD, USA, 1999; p. 71. [Google Scholar]
- Howarth, R.W.; Swaney, D.P.; Butler, T.J.; Marino, R. Rapid communication: Climatic control on eutrophication of the Hudson River Estuary. Ecosystems 2000, 3, 210–215. [Google Scholar] [CrossRef]
- James, N.C.; van Niekerk, L.; Whitfield, A.K.; Potts, W.M.; Götz, A.; Paterson, A.W. Effects of climate change on South African estuaries and associated fish species. Clim. Res. 2013, 57, 233–248. [Google Scholar] [CrossRef]
- Statham, P.J. Nutrients in estuaries—an overview and the potential impacts of climate change. Sci. Total Environ. 2012, 434, 213–227. [Google Scholar] [CrossRef]
- Bricker, S.B.; Longstaff, B.; Dennison, W.; Jones, A.; Boicourt, K.; Wicks, C.; Woerner, J. Effects of nutrient enrichment in the nation’s estuaries: A decade of change. Harmful Algae 2008, 8, 21–32. [Google Scholar] [CrossRef]
- Painting, S.J.; Devlin, M.J.; Malcolm, S.J.; Parker, E.R.; Mills, D.K.; Mills, C.; Tett, P.; Wither, A.; Burt, J.; Jones, R.; et al. Assessing the impact of nutrient enrichment in estuaries: Susceptibility to eutrophication. Mar. Pollut. Bull. 2007, 55, 74–90. [Google Scholar] [CrossRef]
- Carpenter, S.R. Phosphorus control is critical to mitigating eutrophication. Proc. Natl. Acad. Sci. USA 2008, 105, 11039–11040. [Google Scholar] [CrossRef]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Lancelot, C.; Likens, G.E. Controlling eutrophication: Nitrogen and phosphorus. Science 2009, 323, 1014–1015. [Google Scholar] [CrossRef]
- Huot, Y.; Babin, M. Overview of fluorescence protocols: Theory, basic concepts, and practice. In Chlorophyll a Fluorescence in Aquatic Sciences: Methods and Applications; Springer: Berlin/Heidelberg, Germany, 2010; pp. 31–74. [Google Scholar]
- Laney, S.R. In situ measurement of variable fluorescence transients. In Chlorophyll a Fluorescence in Aquatic Sciences: Methods and Applications; Springer: Berlin/Heidelberg, Germany, 2010; pp. 19–30. [Google Scholar]
- Pan, Y.; Qiu, L. A Submersible in-Situ Highly Sensitive Chlorophyll Fluorescence Detection System. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; p. 022065. [Google Scholar]
- Ha, N.T.T.; Koike, K.; Nhuan, M.T.; Canh, B.D.; Thao, N.T.P.; Parsons, M. Landsat 8/OLI two bands ratio algorithm for chlorophyll-a concentration mapping in hypertrophic waters: An application to West Lake in Hanoi (Vietnam). IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2017, 10, 4919–4929. [Google Scholar] [CrossRef]
- Darecki, M.; Weeks, A.; Sagan, S.; Kowalczuk, P.; Kaczmarek, S. Optical characteristics of two contrasting Case 2 waters and their influence on remote sensing algorithms. Cont. Shelf Res. 2003, 23, 237–250. [Google Scholar] [CrossRef]
- Moses, W.J.; Gitelson, A.A.; Berdnikov, S.; Povazhnyy, V. Estimation of chlorophyll-a concentration in case II waters using MODIS and MERIS data—successes and challenges. Environ. Res. Lett. 2009, 4, 045005. [Google Scholar] [CrossRef]
- Babin, M.; Morel, A.; Gentili, B. Remote sensing of sea surface sun-induced chlorophyll fluorescence: Consequences of natural variations in the optical characteristics of phytoplankton and the quantum yield of chlorophyll a fluorescence. Int. J. Remote Sens. 1996, 17, 2417–2448. [Google Scholar] [CrossRef]
- Schallenberg, C.; Lewis, M.R.; Kelley, D.E.; Cullen, J.J. Inferred influence of nutrient availability on the relationship between Sun-induced chlorophyll fluorescence and incident irradiance in the Bering Sea. J. Geophys. Res. Ocean. 2008, 113. [Google Scholar] [CrossRef]
- Xing, X.; Claustre, H.; Blain, S.; d’Ortenzio, F.; Antoine, D.; Ras, J.; Guinet, C. Quenching correction for in vivo chlorophyll fluorescence acquired by autonomous platforms: A case study with instrumented elephant seals in the Kerguelen region (Southern Ocean). Limnol. Oceanogr. Methods 2012, 10, 483–495. [Google Scholar] [CrossRef]
- Cui, T.; Zhang, J.; Wang, K.; Wei, J.; Mu, B.; Ma, Y.; Zhu, J.; Liu, R.; Chen, X. Remote sensing of chlorophyll a concentration in turbid coastal waters based on a global optical water classification system. ISPRS J. Photogramm. Remote Sens. 2020, 163, 187–201. [Google Scholar] [CrossRef]
- Schaeffer, B.A.; Hagy, J.D.; Conmy, R.N.; Lehrter, J.C.; Stumpf, R.P. An Approach to Developing Numeric Water Quality Criteria for Coastal Waters Using the SeaWiFS Satellite Data Record. Environ. Sci. Technol. 2012, 46, 916–922. [Google Scholar] [CrossRef]
- Hopkinson, C.S.; Day, J.W. Aquatic productivity and water quality at the upland-estuary interface in Barataria Basin, Louisiana. In Ecological Processes in Coastal and Marine Systems; Springer: Berlin/Heidelberg, Germany, 1979; pp. 291–314. [Google Scholar]
- Ren, L.; Rabalais, N.N.; Turner, R.E.; Morrison, W.; Mendenhall, W. Nutrient limitation on phytoplankton growth in the upper Barataria Basin, Louisiana: Microcosm bioassays. Estuaries Coasts 2009, 32, 958–974. [Google Scholar] [CrossRef]
- Bargu, S.; Justic, D.; White, J.R.; Lane, R.; Day, J.; Paerl, H.; Raynie, R. Mississippi River diversions and phytoplankton dynamics in deltaic Gulf of Mexico estuaries: A review. Estuar. Coast. Shelf Sci. 2019, 221, 39–52. [Google Scholar] [CrossRef]
- Elsey-Quirk, T.; Graham, S.A.; Mendelssohn, I.A.; Snedden, G.; Day, J.W.; Twilley, R.; Shaffer, G.; Sharp, L.; Pahl, J.; Lane, R. Mississippi river sediment diversions and coastal wetland sustainability: Synthesis of responses to freshwater, sediment, and nutrient inputs. Estuar. Coast. Shelf Sci. 2019, 221, 170–183. [Google Scholar] [CrossRef]
- Wang, H.; Steyer, G.D.; Couvillion, B.R.; Rybczyk, J.M.; Beck, H.J.; Sleavin, W.J.; Meselhe, E.A.; Allison, M.A.; Boustany, R.G.; Fischenich, C.J. Forecasting landscape effects of Mississippi River diversions on elevation and accretion in Louisiana deltaic wetlands under future environmental uncertainty scenarios. Estuar. Coast. Shelf Sci. 2014, 138, 57–68. [Google Scholar] [CrossRef]
- White, J.R.; Delaune, R.D.; Justic, D.; Day, J.W.; Pahl, J.; Lane, R.R.; Boynton, W.R.; Twilley, R.R. Consequences of Mississippi River diversions on nutrient dynamics of coastal wetland soils and estuarine sediments: A review. Estuar. Coast. Shelf Sci. 2019, 224, 209–216. [Google Scholar] [CrossRef]
- Das, A.; Justic, D.; Inoue, M.; Hoda, A.; Huang, H.; Park, D. Impacts of Mississippi River diversions on salinity gradients in a deltaic Louisiana estuary: Ecological and management implications. Estuar. Coast. Shelf Sci. 2012, 111, 17–26. [Google Scholar] [CrossRef]
- Spera, A.C.; White, J.R.; Corstanje, R. Spatial and temporal changes to a hydrologically-reconnected coastal wetland: Implications for restoration. Estuar. Coast. Shelf Sci. 2020, 238, 106728. [Google Scholar] [CrossRef]
- CPRA. Louisiana’s Comprehensive Master Plan for a Sustainable Coast; CPRA, Ed.; CPRA: Baton Rouge, LA, USA, 2017; p. 184. [Google Scholar]
- Hiatt, M.; Snedden, G.; Day, J.W.; Rohli, R.V.; Nyman, J.A.; Lane, R.; Sharp, L.A. Drivers and impacts of water level fluctuations in the Mississippi River delta: Implications for delta restoration. Estuar. Coast. Shelf Sci. 2019, 224, 117–137. [Google Scholar] [CrossRef]
- Green, M. Coastal Restoration Annual Project Reviews: December 2006. Natural Resources Department: Baton Rouge, LA, USA, 2006. [Google Scholar]
- Lohrenz, S.E.; Dagg, M.J.; Whitledge, T.E. Enhanced primary production at the plume/oceanic interface of the Mississippi River. Cont. Shelf Res. 1990, 10, 639–664. [Google Scholar] [CrossRef]
- Prasad, K.S.; Lohrenz, S.E.; Redalje, D.G.; Fahnenstiel, G.L. Primary production in the Gulf of Mexico coastal waters using “remotely-sensed” trophic category approach. Cont. Shelf Res. 1995, 15, 1355–1368. [Google Scholar] [CrossRef]
- Tunnell, J., Jr.; Felder, D.; Earle, S. The Gulf of Mexico past, present, and future: A United States, Mexico, and Cuba Collaboration. In Environmental Analysis of the Gulf of Mexico; Caso, M., Pisanty, L., Ezcurra, E., Eds.; Special Publication Series; Diagnóstico Ambiental del Golfo de México; Instituto Nacional de Ecología (INE-SEMARNAT): México, Mexico, 2004; pp. 222–229. [Google Scholar]
- Cai, W.-J.; Guo, X.; Chen, C.-T.A.; Dai, M.; Zhang, L.; Zhai, W.; Lohrenz, S.E.; Yin, K.; Harrison, P.J.; Wang, Y. A comparative overview of weathering intensity and HCO3− flux in the world’s major rivers with emphasis on the Changjiang, Huanghe, Zhujiang (Pearl) and Mississippi Rivers. Cont. Shelf Res. 2008, 28, 1538–1549. [Google Scholar] [CrossRef]
- Kammerer, J.C. Largest rivers in the United States. In Water Resources Investigations Open File Report 87-242; U.S. Geological Survey: Reston, VA, USA, 1987; p. 2. [Google Scholar]
- Milliman, J.D.; Meade, R.H. World-wide delivery of river sediment to the oceans. J. Geol. 1983, 91, 1–21. [Google Scholar] [CrossRef]
- Byrnes, M.R.; Britsch, L.D.; Berlinghoff, J.L.; Johnson, R.; Khalil, S. Recent subsidence rates for Barataria Basin, Louisiana. Geo-Mar. Lett. 2019, 39, 265–278. [Google Scholar] [CrossRef]
- Conner, W.H.; Day, J.W. The ecology of Barataria Basin, Louisiana: An Estuarine Profile; National Wetlands Research Center, US Fish and Wildlife Service, US: Washington, DC, USA, 1987; Volume 85. [Google Scholar]
- Feijtel, T.; DeLaune, R.; Patrick, W., Jr. Carbon flow in coastal Louisiana. Mar. Ecol. Prog. Ser. 1985, 24, 255–260. [Google Scholar] [CrossRef]
- Turner, R.E.; Swenson, E.M.; Milan, C.S.; Lee, J.M. Spatial variations in Chlorophyll a, C, N, and P in a Louisiana estuary from 1994 to 2016. Hydrobiologia 2019, 834, 131–144. [Google Scholar] [CrossRef]
- Liu, B.; D’Sa, E.J.; Joshi, I. Multi-decadal trends and influences on dissolved organic carbon distribution in the Barataria Basin, Louisiana from in-situ and Landsat/MODIS observations. Remote Sens. Environ. 2019, 228, 183–202. [Google Scholar] [CrossRef]
- Wissel, B.; Gaçe, A.; Fry, B. Tracing river influences on phytoplankton dynamics in two Louisiana estuaries. Ecology 2005, 86, 2751–2762. [Google Scholar] [CrossRef]
- Das, A. Modeling the Impacts of Pulsed Riverine Inflows on Hydrodynamics and Water Quality in the Barataria Bay Estuary. Ph.D. Thesis, Louisiana State University, Baton Rouge, LA, USA, 2010. [Google Scholar]
- Habib, E.; Nuttle, W.K.; Rivera-Monroy, V.H.; Gautam, S.; Wang, J.; Meselhe, E.; Twilley, R.R. Assessing effects of data limitations on salinity forecasting in Barataria basin, Louisiana, with a Bayesian analysis. J. Coast. Res. 2007, 2007, 749–763. [Google Scholar] [CrossRef]
- Li, C.; White, J.R.; Chen, C.; Lin, H.; Weeks, E.; Galvan, K.; Bargu, S. Summertime tidal flushing of Barataria Bay: Transports of water and suspended sediments. J. Geophys. Res. Ocean. 2011, 116, 1–15. [Google Scholar] [CrossRef]
- Walker, N.D.; Hammack, A.B. Impacts of winter storms on circulation and sediment transport: Atchafalaya-Vermilion Bay region, Louisiana, USA. J. Coast. Res. 2000, 16, 996–1010. [Google Scholar]
- Cao, Y.; Li, C.; Dong, C. Atmospheric Cold Front-Generated Waves in the Coastal Louisiana. J. Mar. Sci. Eng. 2020, 8, 900. [Google Scholar] [CrossRef]
- Mortimer, E.B.; Johnson, G.A.; Lau, H.W. Major Arctic outbreaks affecting Louisiana. Natl. Weather Dig. 1988, 13, 5–14. [Google Scholar]
- Swenson, E.; Swarzenski, C. Water levels and salinity in the Barataria-Terrebonne estuarine system. In Status and Historical Trends of Hydrologic Modification, Reduction in Sediment Availability, and Habitat Loss/Modification in the Barataria and Terrebonne Estuarine Systems; BTNEP: Thibodaux, LA, USA, 1995; Volume 20, pp. 129–201. [Google Scholar]
- Snedden, G. River, Tidal and Wind Interactions in a Deltaic Estuarine System. Ph.D. Thesis, Louisiana State University, Baton Rouge, LA, USA, 2006. [Google Scholar]
- Sorourian, S.; Huang, H.; Li, C.; Justic, D.; Payandeh, A.R. Wave dynamics near Barataria Bay tidal inlets during spring–summer time. Ocean Model. 2020, 147, 101553. [Google Scholar] [CrossRef]
- Wiseman, W.; Swenson, E.M.; Power, J. Salinity trends in Louisiana estuaries. Estuaries 1990, 13, 265–271. [Google Scholar] [CrossRef]
- Marmer, H.A. The Currents in Barataria Bay; Texas A&M Research Foundation: College Station, TX, USA, 1948. [Google Scholar]
- Bianchi, T.S.; Cook, R.L.; Perdue, E.M.; Kolic, P.E.; Green, N.; Zhang, Y.; Smith, R.W.; Kolker, A.S.; Ameen, A.; King, G. Impacts of diverted freshwater on dissolved organic matter and microbial communities in Barataria Bay, Louisiana, USA. Mar. Environ. Res. 2011, 72, 248–257. [Google Scholar] [CrossRef]
- Kesel, R. Human modifications to the sediment regime of the Lower Mississippi River flood plain. Geomorphology 2003, 56, 325–334. [Google Scholar] [CrossRef]
- Couvillion, B.R.; Beck, H.; Schoolmaster, D.; Fischer, M. Land Area Change in Coastal Louisiana (1932 to 2016); 2329-132X; US Geological Survey: Reston, VA, USA, 2017.
- Day, J.; Clark, H.; Chang, C.; Hunter, R.; Norman, C. Life cycle of oil and gas fields in the Mississippi River Delta: A review. Water 2020, 12, 1492. [Google Scholar] [CrossRef]
- Jenkins, J.A.; Olivier, H.M.; Draugelis-Dale, R.O.; Kaller, M.D. Davis Pond Freshwater Diversion Biomonitoring: Prediversion and Postdiversion Freshwater Fish Data; 2327-638X; US Geological Survey: Reston, VA, USA, 2012.
- Penland, S.; Beall, A.D.; Britsch III, L.D. Geologic classification of coastal land loss between 1932 and 1990 in the Mississippi River Delta Plain, southeastern Louisiana. Trans. Gulf Coast Assoc. Geol. Soc. 2002, 52, 799–807. [Google Scholar]
- Turner, R.E. Wetland loss in the northern Gulf of Mexico: Multiple working hypotheses. Estuaries 1997, 20, 1–13. [Google Scholar] [CrossRef]
- Zhang, W.; White, J.; DeLaune, R. Diverted Mississippi River sediment as a potential phosphorus source affecting coastal Louisiana water quality. J. Freshw. Ecol. 2012, 27, 575–586. [Google Scholar] [CrossRef]
- Lane, R.R.; Day Jr, J.W.; Marx, B.D.; Reyes, E.; Hyfield, E.; Day, J.N. The effects of riverine discharge on temperature, salinity, suspended sediment and chlorophyll a in a Mississippi delta estuary measured using a flow-through system. Estuar. Coast. Shelf Sci. 2007, 74, 145–154. [Google Scholar] [CrossRef]
- Madden, C.J.; Day, J.W. An instrument system for high-speed mapping of chlorophyll a and physico-chemical variables in surface waters. Estuaries 1992, 15, 421–427. [Google Scholar] [CrossRef]
- Rivera-Monroy, V.H.; Twilley, R.R.; Mancera-Pineda, J.E.; Madden, C.J.; Alcantara-Eguren, A.; Moser, E.B.; Jonsson, B.F.; Castañeda-Moya, E.; Casas-Monroy, O.; Reyes-Forero, P. Salinity and chlorophyll a as performance measures to rehabilitate a mangrove-dominated deltaic coastal region: The Ciénaga Grande de Santa Marta–Pajarales Lagoon Complex, Colombia. Estuaries Coasts 2011, 34, 1–19. [Google Scholar] [CrossRef]
- Stachelek, J.; Madden, C.J. Application of inverse path distance weighting for high-density spatial mapping of coastal water quality patterns. Int. J. Geogr. Inf. Sci. 2015, 29, 1240–1250. [Google Scholar] [CrossRef]
- Carroll, M.; Chigounis, D.; Gilbert, S.; Gundersen, K.; Hayashi, K.; Janzen, C.; Johengen, T.; Koles, T.; Laurier, F.; McKissack, T.; et al. Performance Verification Statement for the TURNER Designs CYCLOPS-7 fluorometer. Solomonsmdalliance Coast. Technol. 2005, 37. [Google Scholar] [CrossRef]
- Arar, E.J.; Collins, G.B. Method 445.0: In Vitro Determination of Chlorophyll a and Pheophytin a in Marine and Freshwater Algae by Fluorescence; United States Environmental Protection Agency, Office of Research and Development: Cincinnati, OH, USA, 1997.
- Chen, J.; Zhu, W.-N.; Tian, Y.Q.; Yu, Q. Estimation of colored dissolved organic matter from Landsat-8 imagery for complex inland water: Case study of Lake Huron. IEEE Trans. Geosci. Remote Sens. 2017, 55, 2201–2212. [Google Scholar] [CrossRef]
- Joshi, I.; D’Sa, E.J. Seasonal variation of colored dissolved organic matter in Barataria Bay, Louisiana, using combined Landsat and field data. Remote Sens. 2015, 7, 12478–12502. [Google Scholar] [CrossRef]
- Kutser, T.; Pierson, D.C.; Kallio, K.Y.; Reinart, A.; Sobek, S. Mapping lake CDOM by satellite remote sensing. Remote Sens. Environ. 2005, 94, 535–540. [Google Scholar] [CrossRef]
- Nezhad, M.M.; Groppi, D.; Marzialetti, P.; Laneve, G. A sediment detection analysis with multi sensor satellites: Caspian Sea and Persian Gulf case studies. In Proceedings of the 4th World Congress on Civil, Structural, and Environmental Engineering, CSEE, Rome, Italy, 7–9 April 2019. [Google Scholar]
- Flores-Anderson, A.I.; Griffin, R.; Dix, M.; Romero-Oliva, C.S.; Ochaeta, G.; Skinner-Alvarado, J.; Ramirez Moran, M.V.; Hernandez, B.; Cherrington, E.; Page, B.; et al. Hyperspectral satellite remote sensing of water quality in Lake Atitlán, Guatemala. Front. Environ. Sci. 2020, 8. [Google Scholar] [CrossRef]
- Watanabe, F.S.Y.; Alcântara, E.; Rodrigues, T.W.P.; Imai, N.N.; Barbosa, C.C.F.; Rotta, L.H.d.S. Estimation of chlorophyll-a concentration and the trophic state of the Barra Bonita hydroelectric reservoir using OLI/Landsat-8 images. Int. J. Environ. Res. Public Health 2015, 12, 10391–10417. [Google Scholar] [CrossRef]
- JMP Pro 15.1.0.; SAS Institute Inc.: Cary, NC, USA, 1989–2019.
- Wang, H.; Meselhe, E.A.; Waldon, M.G.; Harwell, M.C.; Chen, C. Compartment-based hydrodynamics and water quality modeling of a Northern Everglades Wetland, Florida, USA. Ecol. Model. 2012, 247, 273–285. [Google Scholar] [CrossRef]
- Zhao, X.; Rivera-Monroy, V.H.; Wang, H.; Xue, Z.G.; Tsai, C.-F.; Willson, C.S.; Castañeda-Moya, E.; Twilley, R.R. Modeling soil porewater salinity in mangrove forests (Everglades, Florida, USA) impacted by hydrological restoration and a warming climate. Ecol. Model. 2020, 436, 109292. [Google Scholar] [CrossRef]
- Regier, P.; Briceño, H.; Boyer, J.N. Analyzing and comparing complex environmental time series using a cumulative sums approach. MethodsX 2019, 6, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Schalles, J.F. Optical remote sensing techniques to estimate phytoplankton chlorophyll a concentrations in coastal. In Remote Sensing of Aquatic Coastal Ecosystem Processes; Springer: Berlin/Heidelberg, Germany, 2006; pp. 27–79. [Google Scholar]
- Day, J.W.; Li, B.; Marx, B.D.; Zhao, D.; Lane, R.R. Multivariate Analyses of Water Quality Dynamics Over Four Decades in the Barataria Basin, Mississippi Delta. Water 2020, 12, 3143. [Google Scholar] [CrossRef]
- Kemp, M.U. Spatial and Temporal Distribution of Solar Radiation in Louisiana. Master Thesis, Louisiana State University, Baton Rouge, LA, USA, 2007. [Google Scholar]
- Turner, R.E.; Rabalais, N.N.; Alexander, R.B.; McIsaac, G.; Howarth, R.W. Characterization of nutrient, organic carbon, and sediment loads and concentrations from the Mississippi River into the northern Gulf of Mexico. Estuaries Coasts 2007, 30, 773–790. [Google Scholar] [CrossRef]
- Ren, L.; Rabalais, N.N.; Turner, R.E. Effects of Mississippi River water on phytoplankton growth and composition in the upper Barataria estuary, Louisiana. Hydrobiologia 2020, 847, 1831–1850. [Google Scholar] [CrossRef]
- Liu, B.; D’Sa, E.J.; Maiti, K.; Rivera-Monroy, V.H.; Xue, Z. Biogeographical trends in phytoplankton community size structure using adaptive sentinel 3-OLCI chlorophyll a and spectral empirical orthogonal functions in the estuarine-shelf waters of the northern Gulf of Mexico. Remote Sens. Environ. 2021, 252, 112154. [Google Scholar] [CrossRef]
- Day, J.W.; Conner, W.H.; DeLaune, R.D.; Hopkinson, C.S.; Hunter, R.G.; Shaffer, G.P.; Kandalepas, D.; Keim, R.F.; Kemp, G.P.; Lane, R.R.; et al. A Review of 50 Years of Study of Hydrology, Wetland Dynamics, Aquatic Metabolism, Water Quality and Trophic Status, and Nutrient Biogeochemistry in the Barataria Basin, Mississippi Delta—System Functioning, Human Impacts and Restoration Approaches. Water. in review.
- Rivera-Monroy, V.H.; Branoff, B.; Meselhe, E.; McCorquodale, A.; Dortch, M.; Steyer, G.D.; Visser, J.; Wang, H. Landscape-level estimation of nitrogen removal in coastal Louisiana wetlands: Potential sinks under different restoration scenarios. J. Coast. Res. 2013, 67, 75–87. [Google Scholar] [CrossRef]
- Upetri, K.; Rivera-Monroy, V.H.; Maiti, K.; Giblin, A.; Geaghan, J.P. Emerging Wetlands from River Diversions Can Sustain High Denitrification Rates in a Coastal Delta. J. Geophys. Res. Biogeochem. in review.
- Wong, W.H.; Rabalais, N.N.; Turner, R.E. Abundance and ecological significance of the clam Rangia cuneata (Sowerby, 1831) in the upper Barataria Estuary (Louisiana, USA). Hydrobiologia 2010, 651, 305–315. [Google Scholar] [CrossRef]
- Van Der Wiel, K.; Kapnick, S.B.; Van Oldenborgh, G.J.; Whan, K.; Philip, S.; Vecchi, G.A.; Singh, R.K.; Arrighi, J.; Cullen, H. Rapid attribution of the August 2016 flood-inducing extreme precipitation in south Louisiana to climate change. Hydrol. Earth Syst. Sci. 2017, 21, 897–921. [Google Scholar] [CrossRef]
- Shen, F.; Zhou, Y.-X.; Li, D.-J.; Zhu, W.-J.; Suhyb Salama, M. Medium resolution imaging spectrometer (MERIS) estimation of chlorophyll-a concentration in the turbid sediment-laden waters of the Changjiang (Yangtze) Estuary. Int. J. Remote Sens. 2010, 31, 4635–4650. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Gurlin, D.; Moses, W.J.; Barrow, T. A bio-optical algorithm for the remote estimation of the chlorophyll-a concentration in case 2 waters. Environ. Res. Lett. 2009, 4, 045003. [Google Scholar] [CrossRef]
- Lesht, B.M.; Barbiero, R.P.; Warren, G.J. A band-ratio algorithm for retrieving open-lake chlorophyll values from satellite observations of the Great Lakes. J. Great Lakes Res. 2013, 39, 138–152. [Google Scholar] [CrossRef]
- Cannizzaro, J.P.; Carder, K.L. Estimating chlorophyll a concentrations from remote-sensing reflectance in optically shallow waters. Remote Sens. Environ. 2006, 101, 13–24. [Google Scholar] [CrossRef]
- O’Reilly, J.E.; Maritorena, S.; Mitchell, B.G.; Siegel, D.A.; Carder, K.L.; Garver, S.A.; Kahru, M.; McClain, C. Ocean color chlorophyll algorithms for SeaWiFS. J. Geophys. Res. Ocean. 1998, 103, 24937–24953. [Google Scholar] [CrossRef]
- Werdell, P.J.; Bailey, S.W. An improved in-situ bio-optical data set for ocean color algorithm development and satellite data product validation. Remote Sens. Environ. 2005, 98, 122–140. [Google Scholar] [CrossRef]
- Markogianni, V.; Kalivas, D.; Petropoulos, G.; Dimitriou, E. An Appraisal of the Potential of Landsat 8 in Estimating Chlorophyll-a, Ammonium Concentrations and Other Water Quality Indicators. Remote Sens. 2018, 10, 1018. [Google Scholar] [CrossRef]
- Liu, K.; Chen, Q.; Hu, K.; Xu, K.; Twilley, R.R. Modeling hurricane-induced wetland-bay and bay-shelf sediment fluxes. Coast. Eng. 2018, 135, 77–90. [Google Scholar] [CrossRef]
- Swenson, E.M. Assessing the potential climate change impact on salinity in the northern Gulf of Mexico estuaries: A test case in the Barataria estuarine system. In Integrated Assessment of the Climate Change Impacts on the Gulf Coast Region; GCRCC & LSU Graphic Series; GCRCC: Baton Rouge, LA, USA, 2003; pp. 131–150. [Google Scholar]
- NIDIS. Drought in Louisiana from 2000–2020. Available online: https://www.drought.gov/drought/states/louisiana (accessed on 15 November 2020).

| Bands Ratios | Average Spectrum Wavelength (nm) | Linear | Quadratic | Cubic | Quartic | Exponential | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| R2 | RMSE | R2 | RMSE | R2 | RMSE | R2 | RMSE | R2 | RMSE | ||
| B3/B1 | 560/443 | 0.23 | 5.75 | 0.27 | 5.59 | 0.32 | 5.42 | 0.37 | 5.20 | 0.26 | 5.66 |
| B5/B2 | 865/480 | 0.17 | 5.99 | 0.19 | 5.91 | 0.21 | 5.84 | 0.23 | 5.75 | 0.19 | 5.93 |
| B5/B3 | 865/560 | 0.15 | 6.03 | 0.19 | 5.89 | 0.21 | 5.84 | 0.24 | 5.73 | 0.18 | 5.96 |
| B5/B4 | 865/655 | 0.12 | 6.17 | 0.17 | 5.99 | 0.19 | 5.92 | 0.20 | 5.86 | 0.14 | 6.09 |
| B4/B2 | 655/480 | 0.01 | 6.53 * | 0.01 | 6.53 * | 0.03 | 6.47 | 0.04 | 6.43 | 0.01 | 6.53 |
| B3/B2 | 560/480 | 0.05 | 6.39 | 0.09 | 6.25 | 0.09 | 6.25 | 0.11 | 6.21 | 0.06 | 6.37 |
| B2/B1 | 480/443 | 0.16 | 6.03 | 0.18 | 5.93 | 0.19 | 5.93 | 0.19 | 5.93 | 0.18 | 5.96 |
| B4/B1 | 655/443 | 0.02 | 6.48 | 0.04 | 6.43 | 0.05 | 6.41 | 0.08 | 6.30 | 0.03 | 6.47 |
| B4/B3 | 655/560 | 0.29 | 5.53 | 0.32 | 5.42 | 0.33 | 5.40 | 0.33 | 5.37 | 0.31 | 5.44 |
| B5/B1 | 865/443 | 0.12 | 6.17 | 0.16 | 6.02 | 0.19 | 5.92 | 0.19 | 5.92 | 0.13 | 6.11 |
| B2/B3 | 480/560 | 0.04 | 6.42 | 0.08 | 6.29 | 0.10 | 6.24 | 0.10 | 6.24 | 0.05 | 6.40 |
| B1/B2 | 443/480 | 0.14 | 6.08 | 0.17 | 5.97 | 0.19 | 5.92 | 0.19 | 5.93 | 0.17 | 5.99 |
| B1/B3 | 443/560 | 0.19 | 5.89 | 0.29 | 5.53 | 0.29 | 5.52 | 0.35 | 5.32 | 0.25 | 5.68 |
| (B1 + B4)/B3 | (443 + 655)/560 | 0.39 | 5.12 | 0.50 | 4.66 | 0.50 | 4.63 | 0.54 | 4.47 | 0.46 | 4.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vargas-Lopez, I.A.; Rivera-Monroy, V.H.; Day, J.W.; Whitbeck, J.; Maiti, K.; Madden, C.J.; Trasviña-Castro, A. Assessing Chlorophyll a Spatiotemporal Patterns Combining In Situ Continuous Fluorometry Measurements and Landsat 8/OLI Data across the Barataria Basin (Louisiana, USA). Water 2021, 13, 512. https://doi.org/10.3390/w13040512
Vargas-Lopez IA, Rivera-Monroy VH, Day JW, Whitbeck J, Maiti K, Madden CJ, Trasviña-Castro A. Assessing Chlorophyll a Spatiotemporal Patterns Combining In Situ Continuous Fluorometry Measurements and Landsat 8/OLI Data across the Barataria Basin (Louisiana, USA). Water. 2021; 13(4):512. https://doi.org/10.3390/w13040512
Chicago/Turabian StyleVargas-Lopez, Ivan A., Victor H. Rivera-Monroy, John W. Day, Julie Whitbeck, Kanchan Maiti, Christopher J. Madden, and Armando Trasviña-Castro. 2021. "Assessing Chlorophyll a Spatiotemporal Patterns Combining In Situ Continuous Fluorometry Measurements and Landsat 8/OLI Data across the Barataria Basin (Louisiana, USA)" Water 13, no. 4: 512. https://doi.org/10.3390/w13040512
APA StyleVargas-Lopez, I. A., Rivera-Monroy, V. H., Day, J. W., Whitbeck, J., Maiti, K., Madden, C. J., & Trasviña-Castro, A. (2021). Assessing Chlorophyll a Spatiotemporal Patterns Combining In Situ Continuous Fluorometry Measurements and Landsat 8/OLI Data across the Barataria Basin (Louisiana, USA). Water, 13(4), 512. https://doi.org/10.3390/w13040512










