Utilization of Biomass Derived from Cyanobacteria-Based Agro-Industrial Wastewater Treatment and Raisin Residue Extract for Bioethanol Production
Abstract
1. Introduction
| Substrate | Pretreatment Method | Initial Sugar Concentration (g L−1) | Maximum EtOH Concentration (g L−1) | EtOH Yield on Sugars Consumed (%)/(g/g) | References |
|---|---|---|---|---|---|
| Wheat straw | 0.2% H2SO4, 300 bar, 190 °C, 10 min | 43.0–46.0 | 38.0 | 69.0/0.35 | [18] |
| Corn stover | Steam 50 °C 24 h, pH 4.8 | ~160.8 | - | 72.3/- | [19] |
| Molasses Molasses with 20% olive mill wastewaters in a batch bioreactor | 1 M HCl, 100 °C, 30 min KOH 1 M | ~100.0 ~100.0 ~135.0 ∼150.0 | 37.3 44.4 52.4 50.6 | -/0.44 -/0.49 -/0.48 -/0.45 | [20] |
| Glucose-enriched olive mill wastewaters | ~75.0 ~75.0 ~115.0 | 26.1 33.9 52.0 | -/0.36 -/0.45 -/0.46 | [21] | |
| Chlorococcum sp. | Lipid-extracted microalgae | ~100.0 | 3.58 | - | [22] |
| Spirogyra sp. | Untreated | - | 8 g/100 g substrate | - | [23] |
| Chlamydomonas reinhardtii | Enzymatic | - | 11.73 | - | [24] |
| Dunaliella sp. | 1% H2SO4 121 °C, 15 min | - | 7.26 | - | [25] |
| Scenedesmus sp. | 0.3 N H2SO4 121 °C, 15 psi, 20 min | 15.0 | 6.6 | 86.0/- | [26] |
| Mixed algal biomass | 10% H2SO4 121 °C 15 psi, 120 min | - | - | 61.0/- | [27] |
| Chlamydomonas reinhardtii | 3% H2SO4 110 °C, 30 min | 28.5 | 14.6 | - | [28] |
| Chlorella vulgaris FSP-E | 1% H2SO4 121 °C, 20 min | 22.0–24.0 | 11.7 Zymomonas mobilis | - | [4] |
| Scenedesmus obliquus | 2 N H2SO4 120 °C, 30 min | 14.4 (28.6% g/g DW) | - | - | [29] |
| Chlorococcum sp. | 1% H2SO4 140 °C, 0 min, 3% H2SO4 160 °C, 15 min | - | 7.20 6.0 | - | [30] |
| Scenedesmus obliquus YSW15 in swine wastewater effluent | Ultra-sonication 15–60 min | 10.5–10.8 | 6.7 | - | [31] |
| Microalgae biomass from wastewater | 1 M H2SO4 90 °C, 30 min and 5 M NaOH 90 °C, 30 min | ~35.0 | 0.53 Clostridium saccharo-perbutylacetonicum | - | [32] |
| Microalgae biomass from wastewater | 1 M H2SO4 80–90 °C, 120 min | 166.1 g/kg dry algae | 0.53 Clostridium saccharo-perbutylacetonicum | - | [33] |
| Microalgae biomass from wastewater | 1 M H2SO4 autoclaved | 33.8 | 4.6 Clostridium phytofermentans | - | [34] |
| Chlorella sorokiniana, Nannochloropsis gaditana, Scenedesmus almeriensis | 4% H2SO4 121 °C, 90 min 5 M NaOH 90 °C, 30 min, acid and enzymatic | 136 mg/g dry algae 15 mg/g dry algae 129 mg/g dry algae | - - - | - - - | [35] |
| Scenedesmus obliquus | 5% H2SO4 120 °C, 30 min | 63.2 | 11.7 Kluyveromyces marxianus | - | [36] |
| Chlorella vulgaris | Bead-beating and enzymatic | ~1.15 | ~0.5 | 89.0/- | [37] |
| Marine brown algae | Acid and enzymatic | 90.0 | 25.8 E. coli KO11 | -/0.41 | [38] |
| Molasses | Sterilized | 110.0 120.0 | 46.6 50.6 | - - | [39] |
| Anabaena sp. | Genetically modified | - | - | 70.2/- | [17] |
| Synechococcus sp. | Freezing and enzymatic | ~65.0 | 30.0 | 0.27 g EtOH/ g DW | [14] |
| Synechococcus elongatus (recombinant) | 2% H2SO4 autoclaved | - | 7.2 Zymomonas mobilis | 91.0/0.45 | [15] |
| Arthrospira platensis (Spirulina) | 0.5 N H2SO4, 80 °C, 180 min 1 N H2SO4, 60 °C, 90 min | - | - | 53.3/- 30.4/- | [40] |
| Arthrospira platensis (Spirulina) | Enzymatic | - | 6.5 | 86.0/- | [41] |
| Anabaena variabilis, Microcystis aeruginosa | 2 N H2SO4 autoclaved | 342.4 283.2 | 152.5 121.8 | 28.2 g EtOH/g DW 23.9 g EtOH/g DW | [16] |
| Microalgae biomass and raisin extract | 2.5 N H2SO4 (6.6%) 120 min autoclaved | 258.6 | 111.1 | 85.9/0.43 | This study |
| Microalgae biomass from mixed wastewater | 2.5 N H2SO4 120 min autoclaved | 87.2 | 32.2 | 73.8/0.37 | This study |
| Microalgae biomass from mixed wastewater | 2.5 N H2SO4 180 min autoclaved | 85.3 | 32.7 | 76.5/0.38 | This study |
| Microalgae biomass from dairy wastewater | 2.5 N H2SO4 120 min autoclaved | 87.2 | 31.5 | 70.7/0.36 | This study |
| Microalgae biomass from winery wastewater | 2.5 N H2SO4 120 min autoclaved | 76.0 | 21.0 | 55.2/0.28 | This study |
2. Materials and Methods
2.1. Biomass Origin and Harvesting
2.2. Biomass Pretreatment
2.3. Raisin Residue Extract Production
2.4. Yeast Strain and Bioethanol Analysis
3. Results and Discussion
3.1. Biomass Pretreatment
3.2. Yeast Growth Conditions and Bioethanol Production
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Valdés, G.; Mendonça, R.T.; Aggelis, G. Lignocellulosic biomass as a substrate for oleaginous microorganisms: A review. Appl. Sci. 2020, 10, 7698. [Google Scholar] [CrossRef]
- Bhagea, R.; Bhoyroo, V.; Puchooa, D. Microalgae: The next best alternative to fossil fuels after biomass. A review. Microbiol. Res. 2019, 10, 7936. [Google Scholar] [CrossRef]
- John, R.P.; Anisha, G.S.; Nampoothiri, K.M.; Pandey, A. Micro and macroalgal biomass: A renewable source for bioethanol. Bioresour. Technol. 2011, 102, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.-H.; Huang, S.-W.; Chen, C.-Y.; Hasunuma, T.; Kondo, A.; Chang, J.-S. Bioethanol production using carbohydrate-rich microalgae biomass as feedstock. Bioresour. Technol. 2013, 135, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Kokkinos, N.; Lazaridou, A.; Stamatis, N.; Orfanidis, S.; Mitropoulos, A.C.; Christoforidis, A.; Nikolaou, N. Biodiesel production from selected microalgae strains and determination of its properties and combustion specific characteristics. J. Eng. Sci. Technol. Rev. 2015, 8, 1–6. [Google Scholar] [CrossRef]
- Santhosh, S.; Dhandapani, R.; Hemalatha, N. Bioactive compounds from microalgae and its different applications-a review. Adv. Appl. Sci. Res. 2016, 7, 153–158. [Google Scholar]
- Savvides, A.L.; Moisi, K.; Katsifas, E.A.; Karagouni, A.D.; Hatzinikolaou, D.G. Lipid production from indigenous Greek microalgae: A possible biodiesel source. Biotechnol. Lett. 2019, 41, 533–545. [Google Scholar] [CrossRef]
- Li, K.; Liu, S.; Liu, X. An overview of algae bioethanol production. Int. J. Energy Res. 2014, 38, 965–977. [Google Scholar] [CrossRef]
- Bibi, R.; Ahmad, Z.; Imran, M.; Hussain, S.; Ditta, A.; Mahmood, S.; Khalid, A. Algal bioethanol production technology: A trend towards sustainable development. Renew. Sustain. Energy Rev. 2017, 71, 976–985. [Google Scholar] [CrossRef]
- Hossain, N.; Mahlia, T.M.I.; Zaini, J.; Saidur, R. Techno-economics and sensitivity analysis of microalgae as commercial feedstock for bioethanol production. Environ. Prog. Sustain. Energy 2019, 38, 13157. [Google Scholar] [CrossRef]
- Espinosa-Gonzalez, I.; Parashar, A.; Bressler, D.C. Heterotrophic growth and lipid accumulation of Chlorella protothecoides in whey permeate, a dairy by-product stream, for biofuel production. Bioresour. Technol. 2014, 155, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Ueda, R.; Hirayama, S.; Sugata, K.; Nakayama, H. Process for the Production of Ethanol from Microalgae. U.S. Patent 5578472, 26 November 1996. [Google Scholar]
- Chen, P.; Min, M.; Chen, Y.; Wang, L.; Li, Y.; Chen, Q.; Wang, C.; Wan, Y.; Wang, X.; Cheng, Y. Review of biological and engineering aspects of algae to fuels approach. Int. J. Agric. Biol. Eng. 2010, 2, 1–30. [Google Scholar]
- Möllers, K.B.; Cannella, D.; Jørgensen, H.; Frigaard, N.U. Cyanobacterial biomass as carbohydrate and nutrient feedstock for bioethanol production by yeast fermentation. Biotechnol. Biofuels 2014, 7, 1–11. [Google Scholar] [CrossRef]
- Chow, T.-J.; Su, H.-Y.; Tsai, T.-Y.; Chou, H.-H.; Lee, T.-M.; Chang, J.-S. Using recombinant cyanobacterium (Synechococcus elongatus) with increased carbohydrate productivity as feedstock for bioethanol production via separate hydrolysis and fermentation process. Bioresour. Technol. 2015, 184, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Deb, D.; Mallick, N.; Bhadoria, P.B.S. Analytical studies on carbohydrates of two cyanobacterial species for enhanced bioethanol production along with poly-β-hydroxybutyrate, C-phycocyanin, sodium copper chlorophyllin, and exopolysaccharides as co-products. J. Clean. Prod. 2019, 221, 695–709. [Google Scholar] [CrossRef]
- Sanz Smachetti, M.E.; Perez Cenci, M.; Salerno, G.L.; Curatti, L. Ethanol and protein production from minimally processed biomass of a genetically-modified cyanobacterium over-accumulating sucrose. Bioresour. Technol. Rep. 2019, 5, 230–237. [Google Scholar] [CrossRef]
- Olofsson, K.; Palmqvist, B.; Lidén, G. Improving simultaneous saccharification and co-fermentation of pretreated wheat straw using both enzyme and substrate feeding. Biotechnol. Biofuels 2010, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Fu, X. Industrial technologies for bioethanol production from lignocellulosic biomass. Renew. Sustain. Energy Rev. 2016, 57, 468–478. [Google Scholar] [CrossRef]
- Sarris, D.; Matsakas, L.; Aggelis, G.; Koutinas, A.A.; Papanikolaou, S. Aerated vs non-aerated conversions of molasses and olive mill wastewaters blends into bioethanol by Saccharomyces cerevisiae under non-aseptic conditions. Ind. Crops Prod. 2014, 56, 83–93. [Google Scholar] [CrossRef]
- Sarris, D.; Giannakis, M.; Philippoussis, A.; Komaitis, M.; Koutinas, A.A.; Papanikolaou, S. Conversions of olive mill wastewater-based media by Saccharomyces cerevisiae through sterile and non-sterile bioprocesses. J. Chem. Technol. Biotechnol. 2013, 88, 958–969. [Google Scholar] [CrossRef]
- Harun, R.; Danquah, M.K.; Forde, G.M. Microalgal biomass as a fermentation feedstock for bioethanol production. J. Chem. Technol. Biotechnol. 2010, 85, 199–203. [Google Scholar] [CrossRef]
- Eshaq, F.S.; Ali, M.N.; Mohd, M.K. Spirogyra biomass a renewable source for biofuel (bioethanol) production. Int. J. Eng. Sci. Technol. 2010, 2, 7045–7054. [Google Scholar]
- Choi, S.P.; Nguyen, M.T.; Sim, S.J. Enzymatic pretreatment of Chlamydomonas reinhardtii biomass for ethanol production. Bioresour. Technol. 2010, 101, 5330–5336. [Google Scholar] [CrossRef]
- Karatay, S.E.; Erdoğan, M.; Dönmez, S.; Dönmez, G. Experimental investigations on bioethanol production from halophilic microalgal biomass. Ecol. Eng. 2016, 95, 266–270. [Google Scholar] [CrossRef]
- Sivaramakrishnan, R.; Incharoensakdi, A. Utilization of microalgae feedstock for concomitant production of bioethanol and biodiesel. Fuel 2018, 217, 458–466. [Google Scholar] [CrossRef]
- Kumar, V.; Nanda, M.; Joshi, H.C.; Singh, A.; Sharma, S.; Verma, M. Production of biodiesel and bioethanol using algal biomass harvested from fresh water river. Renew. Energy 2018, 116, 606–612. [Google Scholar] [CrossRef]
- Nguyen, M.T.; Choi, S.P.; Lee, J.; Lee, J.H.; Sim, S.J. Hydrothermal acid pretreatment of Chlamydomonas reinhardtii biomass for ethanol production. J. Microbiol. Biotechnol. 2009, 19, 161–166. [Google Scholar]
- Miranda, J.R.; Passarinho, P.C.; Gouveia, L. Pre-treatment optimization of Scenedesmus obliquus microalga for bioethanol production. Bioresour. Technol. 2012, 104, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Harun, R.; Danquah, M.K. Influence of acid pre-treatment on microalgal biomass for bioethanol production. Process Biochem. 2011, 46, 304–309. [Google Scholar] [CrossRef]
- Choi, J.-A.; Hwang, J.-H.; Dempsey, B.A.; Abou-Shanab, R.A.I.; Min, B.; Song, H.; Lee, D.S.; Kim, J.R.; Cho, Y.; Hong, S. Enhancement of fermentative bioenergy (ethanol/hydrogen) production using ultrasonication of Scenedesmus obliquus YSW15 cultivated in swine wastewater effluent. Energy Environ. Sci. 2011, 4, 3513–3520. [Google Scholar] [CrossRef]
- Ellis, J.T.; Hengge, N.N.; Sims, R.C.; Miller, C.D. Acetone, butanol, and ethanol production from wastewater algae. Bioresour. Technol. 2012, 111, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Castro, Y.A.; Ellis, J.T.; Miller, C.D.; Sims, R.C. Optimization of wastewater microalgae saccharification using dilute acid hydrolysis for acetone, butanol, and ethanol fermentation. Appl. Energy 2015, 140, 14–19. [Google Scholar] [CrossRef]
- Fathima, A.A.; Sanitha, M.; Kumar, T.; Iyappan, S.; Ramya, M. Direct utilization of waste water algal biomass for ethanol production by cellulolytic Clostridium phytofermentans DSM1183. Bioresour. Technol. 2016, 202, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Hernández, D.; Riaño, B.; Coca, M.; García-González, M.C. Saccharification of carbohydrates in microalgal biomass by physical, chemical and enzymatic pre-treatments as a previous step for bioethanol production. Chem. Eng. J. 2015, 262, 939–945. [Google Scholar] [CrossRef]
- Miranda, J.R.; Passarinho, P.C.; Gouveia, L. Bioethanol production from Scenedesmus obliquus sugars: The influence of photobioreactors and culture conditions on biomass production. Appl. Microbiol. Biotechnol. 2012, 96, 555–564. [Google Scholar] [CrossRef]
- Kim, K.H.; Choi, I.S.; Kim, H.M.; Wi, S.G.; Bae, H.-J. Bioethanol production from the nutrient stress-induced microalga Chlorella vulgaris by enzymatic hydrolysis and immobilized yeast fermentation. Bioresour. Technol. 2014, 153, 47–54. [Google Scholar] [CrossRef]
- Kim, N.-J.; Li, H.; Jung, K.; Chang, H.N.; Lee, P.C. Ethanol production from marine algal hydrolysates using Escherichia coli KO11. Bioresour. Technol. 2011, 102, 7466–7469. [Google Scholar] [CrossRef]
- Kopsahelis, N.; Bosnea, L.; Bekatorou, A.; Tzia, C.; Kanellaki, M. Alcohol production from sterilized and non-sterilized molasses by Saccharomyces cerevisiae immobilized on brewer’s spent grains in two types of continuous bioreactor systems. Biomass Bioenergy 2012, 45, 87–94. [Google Scholar] [CrossRef]
- Markou, G.; Angelidaki, I.; Nerantzis, E.; Georgakakis, D. Bioethanol production by carbohydrate-enriched biomass of Arthrospira (Spirulina) platensis. Energies 2013, 6, 3937–3950. [Google Scholar] [CrossRef]
- Aikawa, S.; Joseph, A.; Yamada, R.; Izumi, Y.; Yamagishi, T.; Matsuda, F.; Kawai, H.; Chang, J.S.; Hasunuma, T.; Kondo, A. Direct conversion of Spirulina to ethanol without pretreatment or enzymatic hydrolysis processes. Energy Environ. Sci. 2013, 6, 1844–1849. [Google Scholar] [CrossRef]
- Behera, S.; Singh, R.; Arora, R.; Sharma, N.K.; Shukla, M.; Kumar, S. Scope of algae as third generation biofuels. Front. Bioeng. Biotechnol. 2015, 2, 90. [Google Scholar] [CrossRef]
- Bellou, S.; Baeshen, M.N.; Elazzazy, A.M.; Aggeli, D.; Sayegh, F.; Aggelis, G. Microalgal lipids biochemistry and biotechnological perspectives. Biotechnol. Adv. 2014, 32, 1476–1493. [Google Scholar] [CrossRef]
- Gonçalves, A.L.; Pires, J.C.M.; Simões, M. A review on the use of microalgal consortia for wastewater treatment. Algal Res. 2017, 24, 403–415. [Google Scholar] [CrossRef]
- Tsolcha, O.N.; Tekerlekopoulou, A.G.; Akratos, C.S.; Aggelis, G.; Genitsaris, S.; Moustaka-Gouni, M.; Vayenas, D.V. Biotreatment of raisin and winery wastewaters and simultaneous biodiesel production using a Leptolyngbya-based microbial consortium. J. Clean. Prod. 2017, 148, 185–193. [Google Scholar] [CrossRef]
- Tsolcha, O.N.; Tekerlekopoulou, A.G.; Akratos, C.S.; Aggelis, G.; Genitsaris, S.; Moustaka-Gouni, M.; Vayenas, D.V. Agroindustrial wastewater treatment with simultaneous biodiesel production in attached growth systems using a mixed microbial culture. Water 2018, 10, 1693. [Google Scholar] [CrossRef]
- Tsolcha, O.N.; Tekerlekopoulou, A.G.; Akratos, C.S.; Antonopoulou, G.; Aggelis, G.; Genitsaris, S.; Moustaka-Gouni, M.; Vayenas, D.V. A Leptolyngbya-based microbial consortium for agro-industrial wastewaters treatment and biodiesel production. Environ. Sci. Pollut. Res. 2018, 25, 17957–17966. [Google Scholar] [CrossRef]
- Papadopoulos, K.P.; Economou, C.N.; Dailianis, S.; Charalampous, N.; Stefanidou, N.; Moustaka-Gouni, M.; Tekerlekopoulou, A.G.; Vayenas, D.V. Brewery wastewater treatment using cyanobacterial-bacterial settleable aggregates. Algal Res. 2020, 49, 101957. [Google Scholar] [CrossRef]
- Luo, L.; van der Voet, E.; Huppes, G. An energy analysis of ethanol from cellulosic feedstock-Corn stover. Renew. Sustain. Energy Rev. 2009, 13, 2003–2011. [Google Scholar] [CrossRef]
- Palmqvist, E.; Hahn-Hägerdal, B. Fermentation of lignocellulosic hydrolysates. II: Inhibitors and mechanisms of inhibition. Bioresour. Technol. 2000, 74, 25–33. [Google Scholar] [CrossRef]
- Parawira, W.; Tekere, M. Biotechnological strategies to overcome inhibitors in lignocellulose hydrolysates for ethanol production: Review. Crit. Rev. Biotechnol. 2011, 31, 20–31. [Google Scholar] [CrossRef]
- Velazquez-Lucio, J.; Rodríguez-Jasso, R.M.; Colla, L.M.; Sáenz-Galindo, A.; Cervantes-Cisneros, D.E.; Aguilar, C.N.; Fernandes, B.D.; Ruiz, H.A. Microalgal biomass pretreatment for bioethanol production: A review. Biofuel Res. J. 2018, 5, 780–791. [Google Scholar] [CrossRef]
- Yoo, G.; Park, M.S.; Yang, J.-W. Chemical pretreatment of algal biomass. In Pretreatment of Biomass; Elsevier: Amsterdam, The Netherlands, 2015; pp. 227–258. [Google Scholar]
- Łukajtis, R.; Kucharska, K.; Hołowacz, I.; Rybarczyk, P.; Wychodnik, K.; Słupek, E.; Nowak, P.; Kamiński, M. Comparison and optimization of saccharification conditions of alkaline pre-treated triticale straw for acid and enzymatic hydrolysis followed by ethanol fermentation. Energies 2018, 11, 639. [Google Scholar] [CrossRef]
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.T.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Argiriou, T.; Kaliafas, A.; Psarianos, K.; Kanellaki, M.; Voliotis, S.; Koutinas, A.A. Psychrotolerant Saccharomyces cerevisiae strains after an adaptation treatment for low temperature wine making. Process Biochem. 1996, 31, 639–643. [Google Scholar] [CrossRef]
- Mehdikhani, P.; Bari, M.R.; Hovsepyan, H. Screening of Saccharomyces cerevisiae for high tolerance of ethanol concentration and temperature. Afr. J. Microbiol. Res. 2011, 5, 2654–2660. [Google Scholar]
- Wyman, C. Handbook on Bioethanol: Production and Utilization; CRC Press: Boca Raton, FL, USA, 1996. [Google Scholar]
- Lee, S.; Oh, Y.; Kim, D.; Kwon, D.; Lee, C.; Lee, J. Converting carbohydrates extracted from marine algae into ethanol using various ethanolic Escherichia coli strains. Appl. Biochem. Biotechnol. 2011, 164, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Schneider, R.C.; Bjerk, T.R.; Gressler, P.D.; Souza, M.P.; Corbellini, V.A.; Lobo, E.A. Potential production of biofuel from microalgae biomass produced in wastewater. In Biodiesel—Feedstocks, Production and Applications; Bjerk, T.R., Ed.; IntechOpen: Rijeka, Croatia, 2013. [Google Scholar]
- Badger, P.C. Ethanol from cellulose: A general review. Trends New Crop. New Uses 2002, 14, 17–21. [Google Scholar]
- Guimarães, P.M.R.; Teixeira, J.A.; Domingues, L. Fermentation of lactose to bio-ethanol by yeasts as part of integrated solutions for the valorisation of cheese whey. Biotechnol. Adv. 2010, 28, 375–384. [Google Scholar] [CrossRef]
- Kasavi, C.; Finore, I.; Lama, L.; Nicolaus, B.; Oliver, S.G.; Oner, E.T.; Kirdar, B. Evaluation of industrial Saccharomyces cerevisiae strains for ethanol production from biomass. Biomass Bioenergy 2012, 45, 230–238. [Google Scholar] [CrossRef]
- Laplace, J.M.; Delgenès, J.-P.; Moletta, R.; Navarro, J.M. Combined alcoholic fermentation of D-xylose and D-glucose by four selected microbial strains: Process considerations in relation to ethanol tolerance. Biotechnol. Lett. 1991, 13, 445–450. [Google Scholar] [CrossRef]
- Palmqvist, E.; Grage, H.; Meinander, N.Q.; Hahn-Hägerdal, B. Main and interaction effects of acetic acid, furfural, and p- hydroxybenzoic acid on growth and ethanol productivity of yeasts. Biotechnol. Bioeng. 1999, 63, 46–55. [Google Scholar] [CrossRef]
- Klinke, H.B.; Thomsen, A.B.; Ahring, B.K. Inhibition of ethanol-producing yeast and bacteria by degradation products produced during pre-treatment of biomass. Appl. Microbiol. Biotechnol. 2004, 66, 10–26. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.P.; Mulako, I.; Tuffin, M.; Cowan, D. Understanding physiological responses to pre-treatment inhibitors in ethanologenic fermentations. Biotechnol. J. 2012, 7, 1169–1181. [Google Scholar] [CrossRef]
- Jönsson, L.J.; Alriksson, B.; Nilvebrant, N.-O. Bioconversion of lignocellulose: Inhibitors and detoxification. Biotechnol. Biofuels 2013, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- de Souza, R.R.; Bergamasco, R.; da Costa, S.C.; Feng, X.; Faria, S.H.B.; Gimenes, M.L. Recovery and purification of lactose from whey. Chem. Eng. Process. Process Intensif. 2010, 49, 1137–1143. [Google Scholar] [CrossRef]
- Najafpour, G.; Younesi, H.; Ku Ismail, K.S. Ethanol fermentation in an immobilized cell reactor using Saccharomyces cerevisiae. Bioresour. Technol. 2004, 92, 251–260. [Google Scholar] [CrossRef]
- Plessas, S.; Bekatorou, A.; Koutinas, A.A.; Soupioni, M.; Banat, I.M.; Marchant, R. Use of Saccharomyces cerevisiae cells immobilized on orange peel as biocatalyst for alcoholic fermentation. Bioresour. Technol. 2007, 98, 860–865. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Chang, K.-S.; Chen, C.-Y.; Hsu, C.-L.; Chang, T.-C.; Jang, H.-D. Enhancement of the efficiency of bioethanol production by Saccharomyces cerevisiae via gradually batch-wise and fed-batch increasing the glucose concentration. Fermentation 2018, 4, 45. [Google Scholar] [CrossRef]
- Bely, M.; Stoeckle, P.; Masneuf-Pomarède, I.; Dubourdieu, D. Impact of mixed Torulaspora delbrueckii–Saccharomyces cerevisiae culture on high-sugar fermentation. Int. J. Food Microbiol. 2008, 122, 312–320. [Google Scholar] [CrossRef]
- Sarris, D.; Kotseridis, Y.; Linga, M.; Galiotou-Panayotou, M.; Papanikolaou, S. Enhanced ethanol production, volatile compound biosynthesis and fungicide removal during growth of a newly isolated Saccharomyces cerevisiae strain on enriched pasteurized grape musts. Eng. Life Sci. 2009, 9, 29–37. [Google Scholar] [CrossRef]
- Kopsahelis, N.; Agouridis, N.; Bekatorou, A.; Kanellaki, M. Comparative study of spent grains and delignified spent grains as yeast supports for alcohol production from molasses. Bioresour. Technol. 2007, 98, 1440–1447. [Google Scholar] [CrossRef] [PubMed]
- Lakatos, G.E.; Ranglová, K.; Manoel, J.C.; Grivalský, T.; Kopecký, J.; Masojídek, J. Bioethanol production from microalgae polysaccharides. Folia Microbiol. 2019, 64, 627–644. [Google Scholar] [CrossRef] [PubMed]
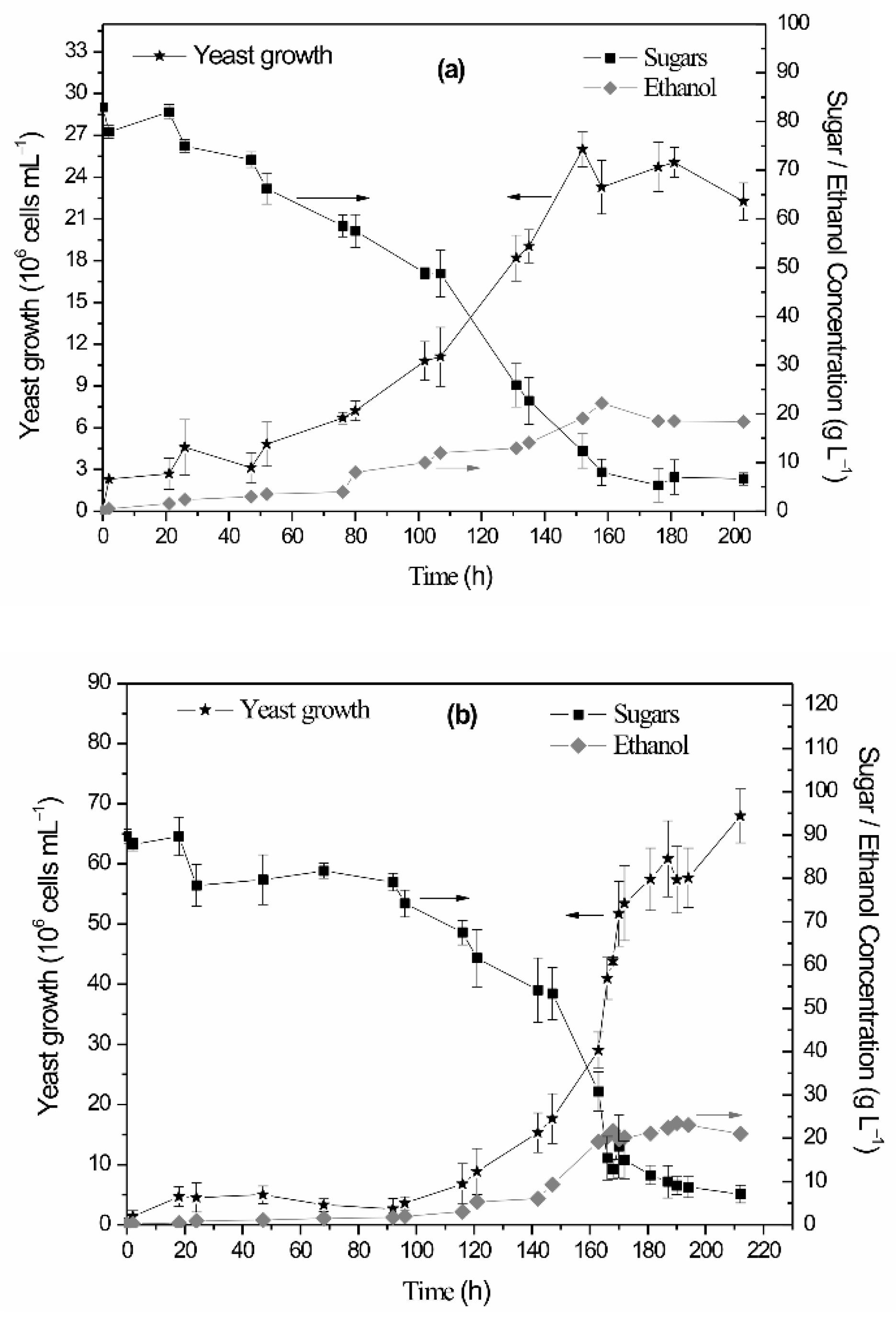
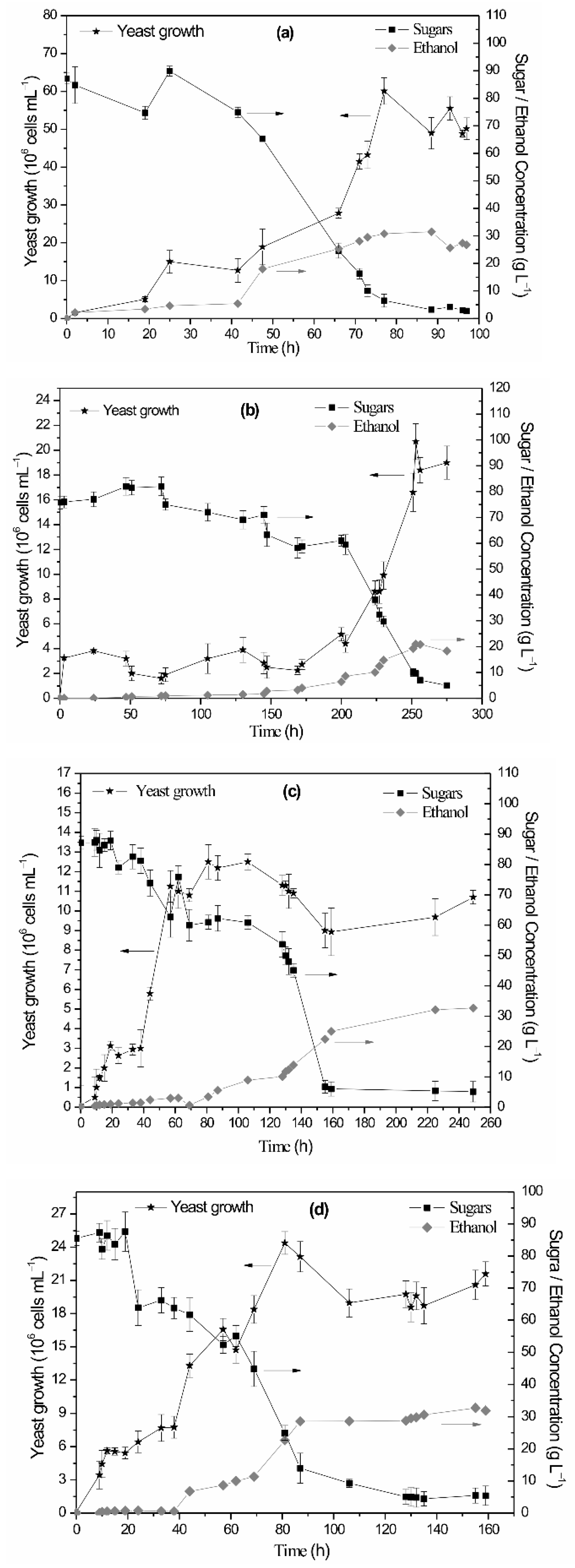
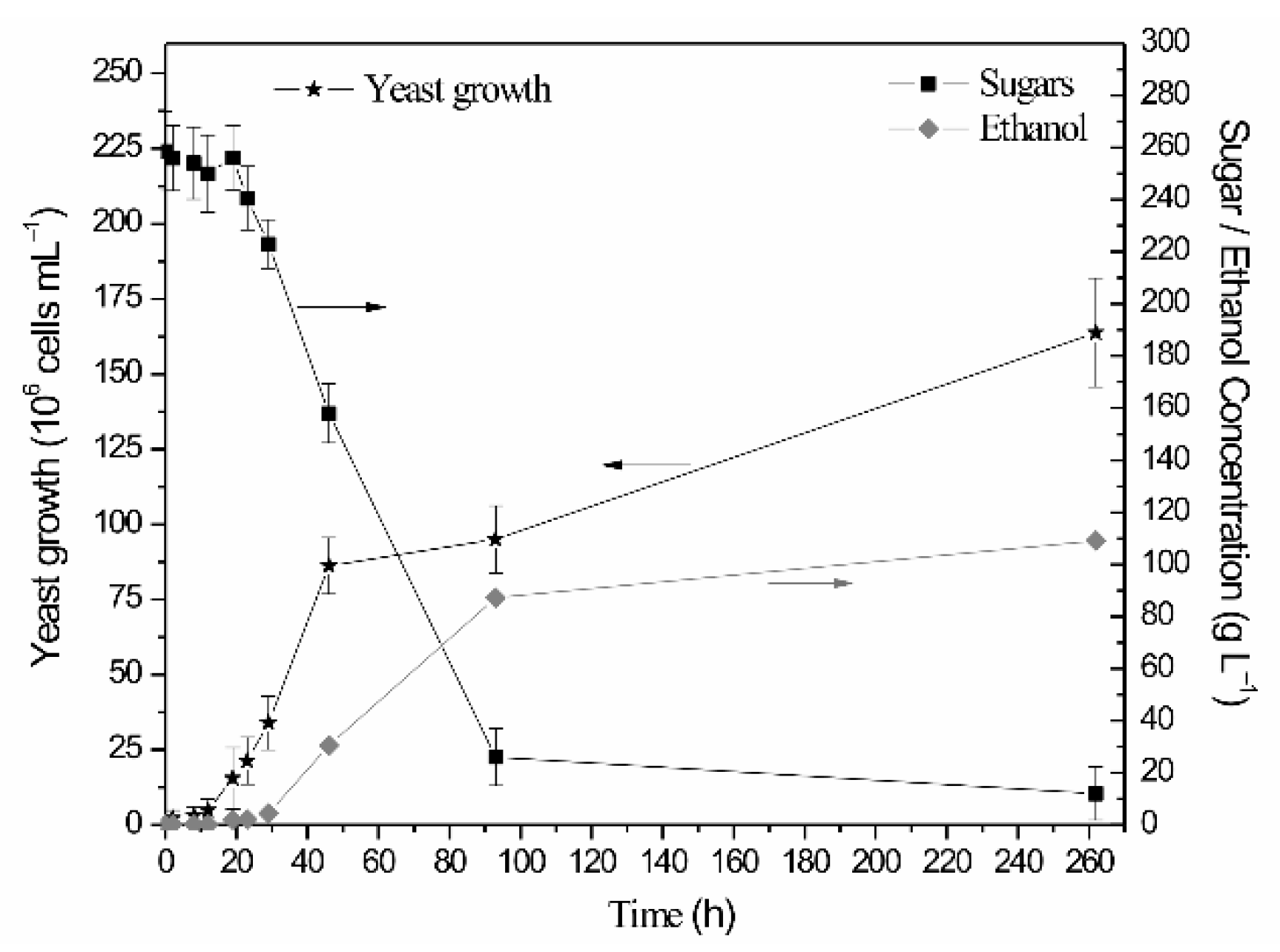
| Cyanobacterial Growth Substrate | Percentage Reducing/Total Sugars (%) per Acid Pretreatment Duration | Sulfuric Acid Concentration | |||
|---|---|---|---|---|---|
| 30 min | 60 min | 120 min | 180 min | ||
| Synthetic medium | 86.8 ± 4.0 86.7 ± 0.9 | 80.0 ± 6.8 91.3 ± 4.3 | 89.8 ± 4.2 94.4 ± 4.1 | 51.8 ± 2.5 47.0 ± 5.4 | 1.5 N 2.5 N |
| Cyanobacterial Growth Substrate | Percentage Reducing/Total Sugars (%) per Acid Pretreatment Duration | Sulfuric Acid Concentration | |||
|---|---|---|---|---|---|
| 30 min | 60 min | 120 min | 180 min | ||
| Dairy wastewater | ND ND | ND 85.3 ± 2.0 | 83.0 ± 6.0 93.0 ± 7.0 | ND 50.3 ± 2.5 | 1.5 N 2.5 N |
| Winery wastewater | 91.0 ± 0.3 87.5 ± 2.0 | ND 91.5 ± 1.5 | ND 98.0 ± 1.0 | ND 51.8 ± 3.0 | 1.5 N 2.5 N |
| Mixed wastewater (winery and raisin) | ND 84.3 ± 2.9 | ND 89.5 ± 2.2 | 46.6 ± 3.0 98.1 ± 1.9 | ND 60.7 ± 1.0 | 1.5 N 2.5 N |
| Substrate | g Sugars/g Dry Biomass (%) | Initial Sugar Concentration (g L−1) | Ethanol Concentration (g L−1) | Ethanol Yield in Sugars (%) |
|---|---|---|---|---|
| SM-60 | 21.5 | 83.0 | 22.2 | 53.5 |
| SM-120 | 31.8 | 89.7 | 22.8 | 62.0 |
| DW-120 | 26.1 | 87.2 | 31.5 | 70.7 |
| WW-120 | 20.6 | 75.8 | 21.0 | 55.2 |
| MW-120 | 19.0 | 87.2 | 32.2 | 73.8 |
| MW-180 | - | 85.3 | 32.7 | 76.5 |
| RR; SM-120 | - | 258.6 | 111.1 | 85.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsolcha, O.N.; Patrinou, V.; Economou, C.N.; Dourou, M.; Aggelis, G.; Tekerlekopoulou, A.G. Utilization of Biomass Derived from Cyanobacteria-Based Agro-Industrial Wastewater Treatment and Raisin Residue Extract for Bioethanol Production. Water 2021, 13, 486. https://doi.org/10.3390/w13040486
Tsolcha ON, Patrinou V, Economou CN, Dourou M, Aggelis G, Tekerlekopoulou AG. Utilization of Biomass Derived from Cyanobacteria-Based Agro-Industrial Wastewater Treatment and Raisin Residue Extract for Bioethanol Production. Water. 2021; 13(4):486. https://doi.org/10.3390/w13040486
Chicago/Turabian StyleTsolcha, Olga N., Vasiliki Patrinou, Christina N. Economou, Marianna Dourou, George Aggelis, and Athanasia G. Tekerlekopoulou. 2021. "Utilization of Biomass Derived from Cyanobacteria-Based Agro-Industrial Wastewater Treatment and Raisin Residue Extract for Bioethanol Production" Water 13, no. 4: 486. https://doi.org/10.3390/w13040486
APA StyleTsolcha, O. N., Patrinou, V., Economou, C. N., Dourou, M., Aggelis, G., & Tekerlekopoulou, A. G. (2021). Utilization of Biomass Derived from Cyanobacteria-Based Agro-Industrial Wastewater Treatment and Raisin Residue Extract for Bioethanol Production. Water, 13(4), 486. https://doi.org/10.3390/w13040486











