Seasonal and Spatial Distribution and Pollution Assessment of Nitrogen and Phosphorus in Sediments from One of the World’s Largest Tidal Reservoirs
Abstract
1. Introduction
2. Materials and Methods
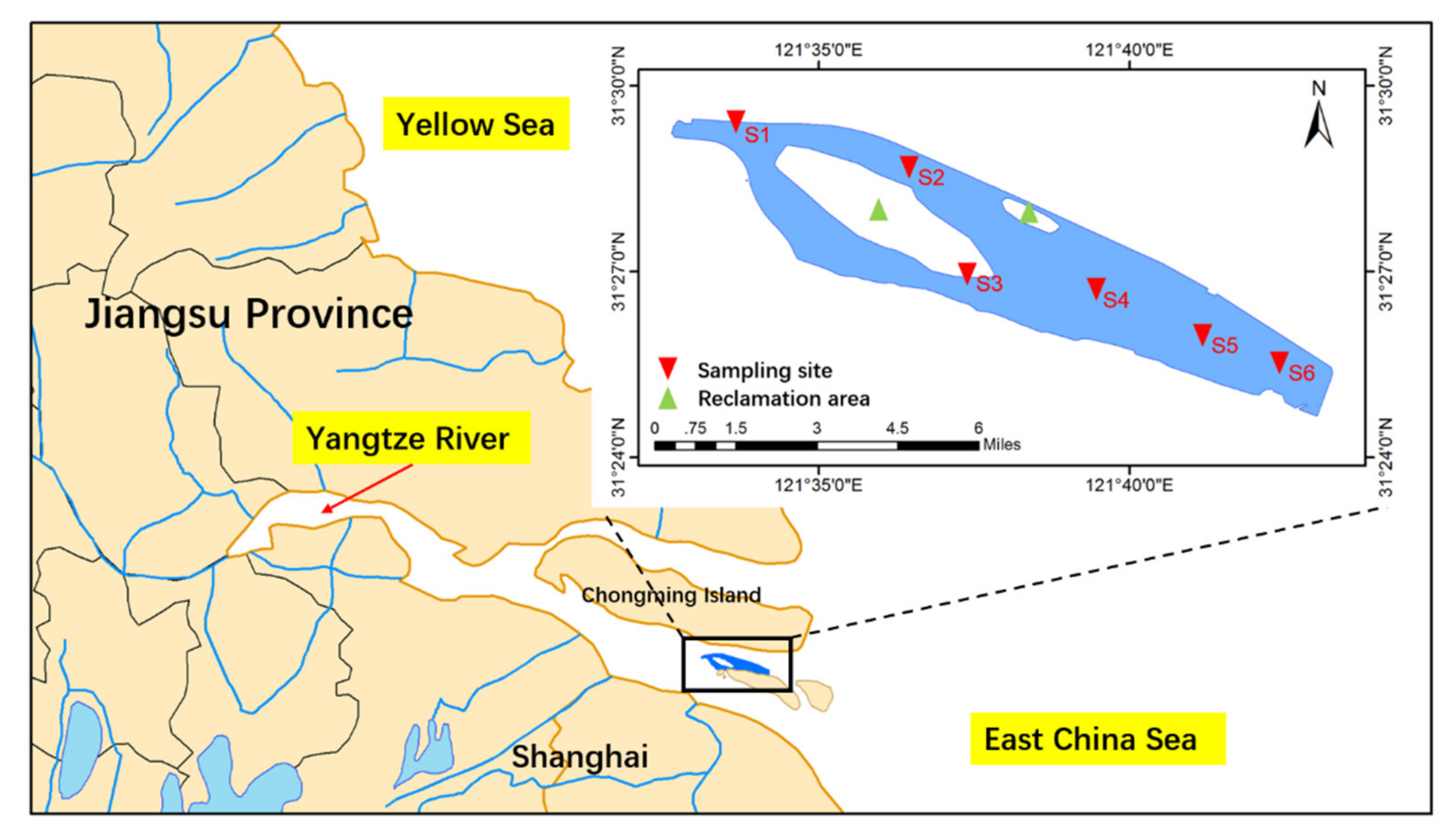
2.1. Study Area and Sediment Sampling
2.2. Determination of Different Species of N and P in Sediments
2.3. Assessment of Nutrient Pollution in Sediments
2.3.1. Pollution Index
2.3.2. Nemerow Pollution Index
2.4. Data Analysis
3. Results and Discussion
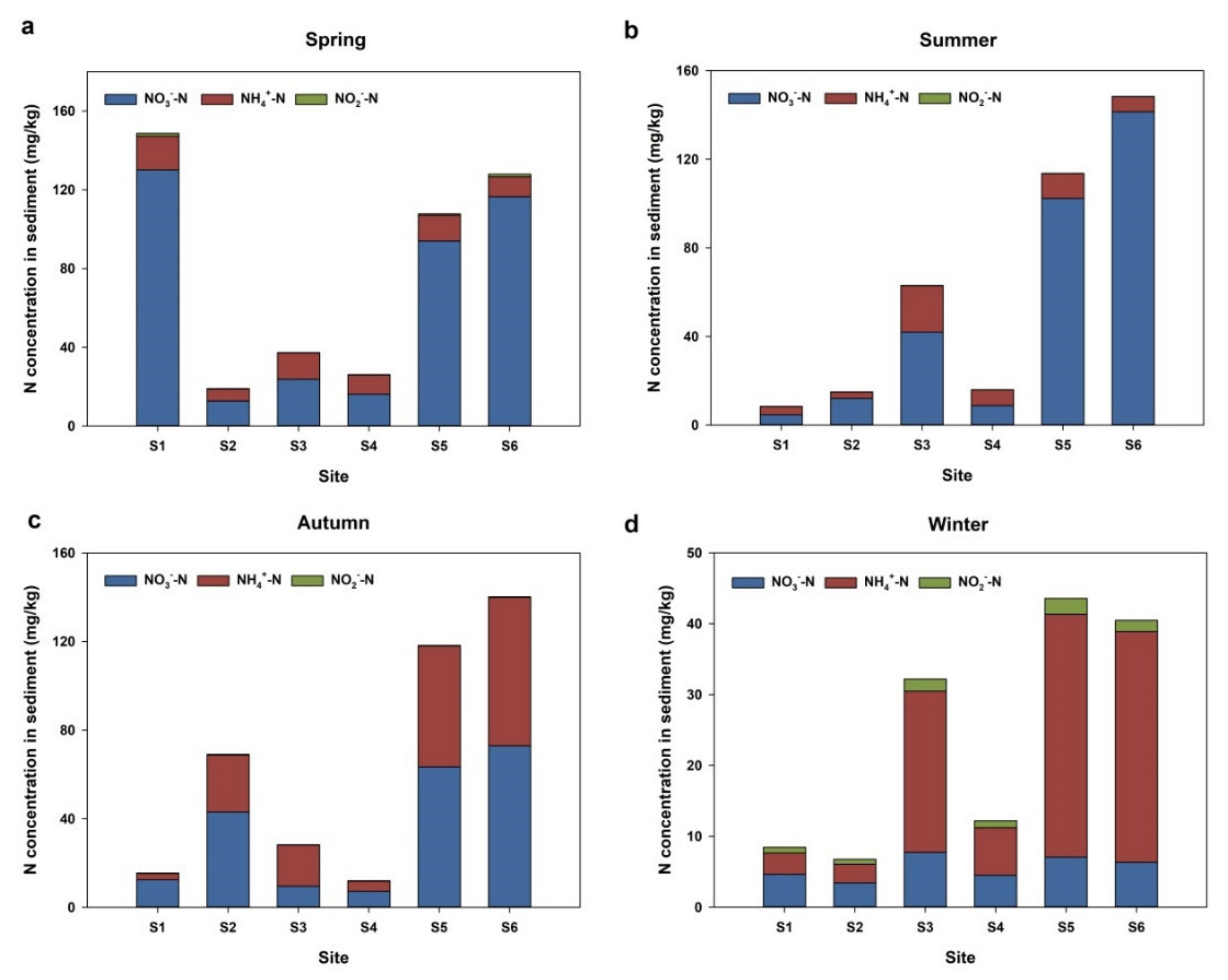
3.1. Seasonal Variation and Spatial Distribution of Total Nitrogen in Surface Sediments
3.2. Seasonal and Spatial Variations of Different Inorganic Nitrogen in Sediments
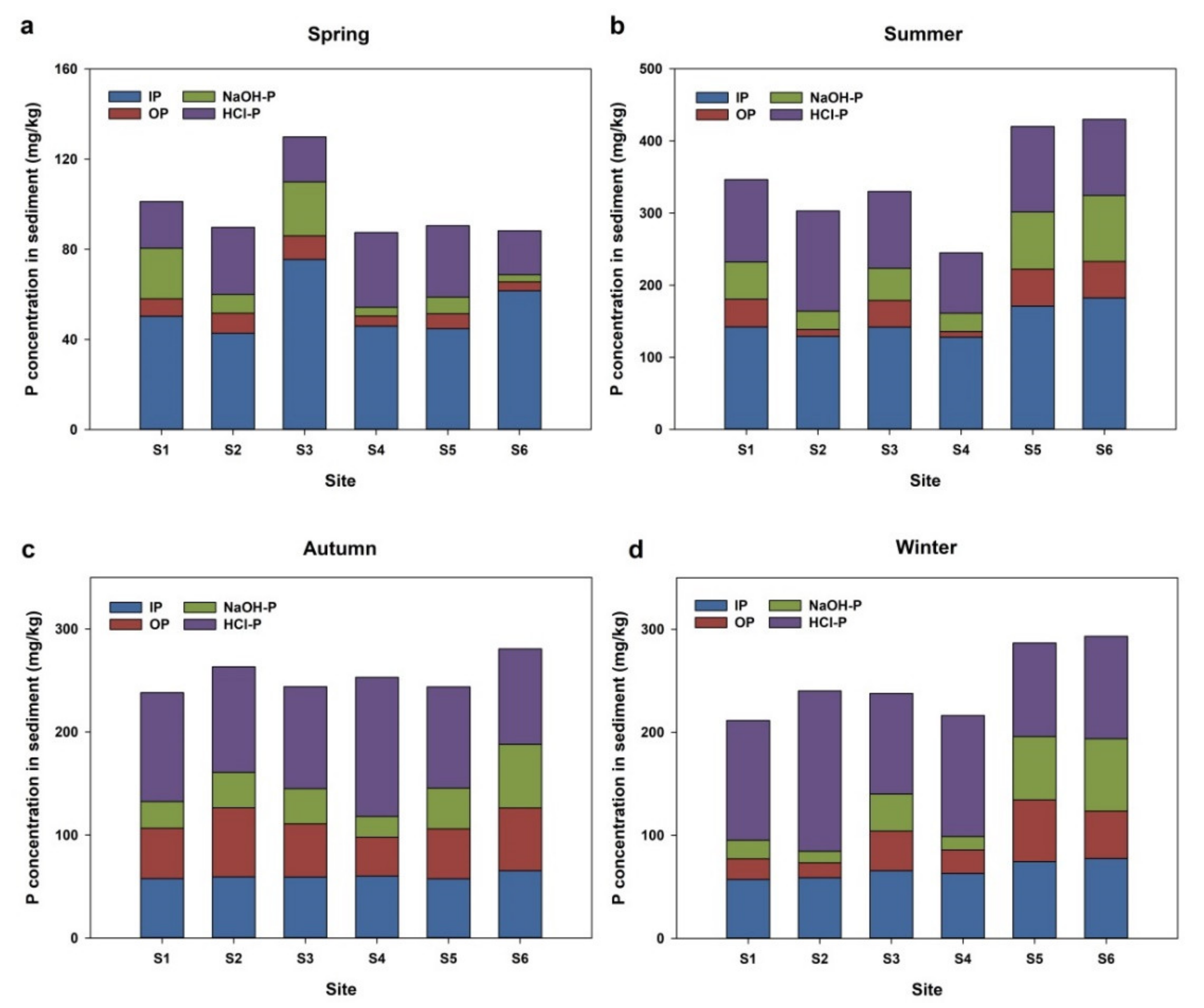
3.3. Seasonal and Spatial Variations of Total Phosphorus in Surface Sediments
3.4. Spatial Distribution and Seasonal Variation of Different Forms of Phosphorus in Sediments
3.5. Assessment of N and P Pollution in Sediments
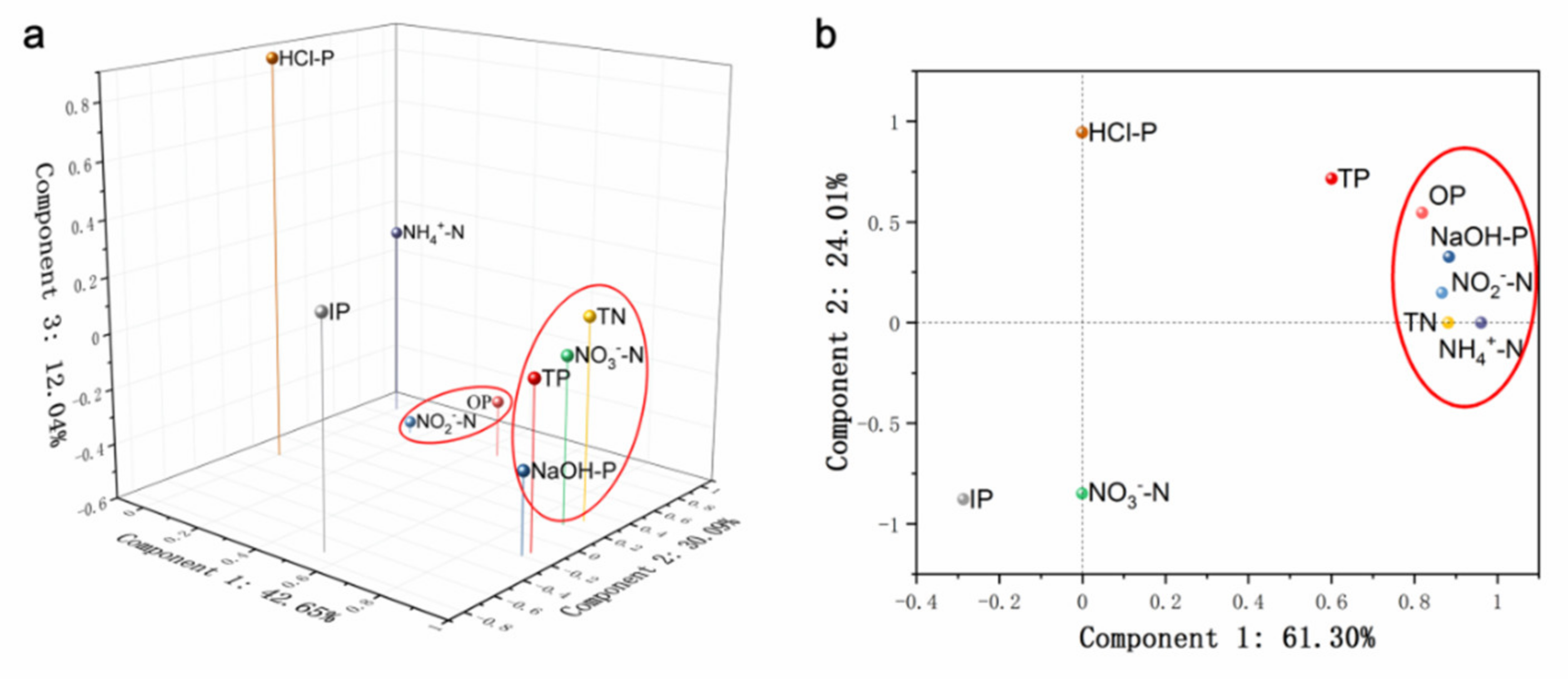
3.6. Identification of Pollution Sources Based on Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chon, H.S.; Ohandja, D.G.; Voulvoulis, N. The role of sediments as a source of metals in river catchments. Chemosphere 2012, 88, 1250–1256. [Google Scholar] [CrossRef]
- He, J.; Deng, W.; Chen, C.; Xu, X.; Wang, S.; Liu, W.; Wu, X. Ammonia nitrogen adsorption and release characteristics of surface sediments in Dianchi Lake, China. Environ. Earth Sci. 2015, 74, 3917–3927. [Google Scholar]
- Duan, L.; Song, J.; Xu, Y.; Li, X.; Zhang, Y. The distribution, enrichment and source of potential harmful elements in surface sediments of Bohai Bay, North China. J. Hazard. Mater. 2010, 183, 155–164. [Google Scholar] [CrossRef]
- Yang, Y.; Gao, B.; Hao, H.; Zhou, H.; Lu, J. Nitrogen and phosphorus in sediments in China: A national-scale assessment and review. Sci. Total Environ. 2017, 576, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Jin, X.; Liu, D.; Lang, C.; Shan, B. Temporal and spatial variation of nitrogen and phosphorus and eutrophication assessment for a typical arid river-Fuyang River in northern China. J. Environ. Sci. 2017, 55, 41–48. [Google Scholar] [CrossRef]
- Rozpondek, K.; Rozpondek, R.; Pachura, P. Characteristics of Spatial Distribution of Phosphorus and Nitrogen in the Bottom Sediments of the Water Reservoir Poraj. J. Ecol. Eng. 2017, 18, 178–184. [Google Scholar] [CrossRef]
- Sun, S.; Huang, S.; Sun, X.; Wen, W. Phosphorus fractions and its release in the sediments of Haihe River, China. J. Environ. Sci. 2009, 21, 291–295. [Google Scholar] [CrossRef]
- Zhu, M.; Zhu, G.; Li, W.; Zhang, Y.; Zhao, L.; Gu, Z. Estimation of the algal-available phosphorus pool in sediments of a large, shallow eutrophic lake (Taihu, China) using profiled SMT fractional analysis. Environ. Pollut. 2013, 173, 216–223. [Google Scholar] [CrossRef]
- Młynarczyk, N.; Bartoszek, M.; Polak, J.; Sułkowski, W.W. Forms of phosphorus in sediments from the Goczałkowice Reservoir. Appl. Geochem. 2013, 37, 87–93. [Google Scholar] [CrossRef]
- Zhang, Z.; Lv, Y.; Zhang, W.; Zhang, Y.; Sun, C.; Marhaba, T. Phosphorus, organic matter and nitrogen distribution characteristics of the surface sediments in Nansi Lake, China. Environ. Earth Sci. 2014, 73, 5669–5675. [Google Scholar] [CrossRef]
- Nikolai, S.J.; Dzialowski, A.R. Effects of internal phosphorus loading on nutrient limitation in a eutrophic reservoir. Limnologica 2014, 49, 33–41. [Google Scholar] [CrossRef]
- Zhang, C.; Lai, S.; Gao, X.; Xu, L. Potential impacts of climate change on water quality in a shallow reservoir in China. Environ. Sci. Pollut. Res. Int. 2015, 22, 14971–14982. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Deng, W.; Xu, X.; He, J.; Wang, S.; Jiao, L.; Zhang, Y. Phosphorus adsorption and release characteristics of surface sediments in Dianchi Lake, China. Environ. Earth Sci. 2015, 74, 3689–3700. [Google Scholar] [CrossRef]
- Kundzewicz, Z.W.; Mata, L.J.; Arnell, N.W.; Döll, P.; Jimenez, B.; Miller, K.; Oki, T.; Şen, Z.; Shiklomanov, I. The implications of projected climate change for freshwater resources and their management. Hydrol. Sci. J. 2009, 53, 3–10. [Google Scholar] [CrossRef]
- Sondergaard, M.; Bjerring, R.; Jeppesen, E. Persistent internal phosphorus loading during summer in shallow eutrophic lakes. Hydrobiologia 2013, 710, 95–107. [Google Scholar] [CrossRef]
- Zhao, Y.; Zheng, B.; Jia, H.; Chen, Z. Determination sources of nitrates into the Three Gorges Reservoir using nitrogen and oxygen isotopes. Sci. Total Environ. 2019, 687, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Tang, H.-W.; Xiao, Y.; Ji, M. Hydrodynamic characteristics and sediment transport of a tidal river under influence of wading engineering groups. China Ocean. Eng. 2014, 27, 829–842. [Google Scholar] [CrossRef]
- Cao, L.; Liu, J.; Shi, X.; He, W.; Chen, Z. Source-to-sink processes of fluvial sediments in the northern South China Sea: Constraints from river sediments in the coastal region of South China. J. Asian Earth Sci. 2019, 185. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, S.; Yi, L. The Sediment Source and Transport Trends around the Abandoned Yellow River Delta, China. Mar. GeoResour. Geotechnol. 2015, 34, 440–449. [Google Scholar] [CrossRef]
- Liu, N.; Zhang, D.; Cen, K.; Cui, L.; Li, J.; Liu, Q.; Lu, J.; Lin, X. Influence of anthropogenic activities on the temporal and spatial variation of polycyclic aromatic hydrocarbons in the sediments of Jiangsu coastal zone, China. Cont. Shelf Res. 2018, 170, 11–20. [Google Scholar] [CrossRef]
- Li, M.; Chen, J.; Finlayson, B.; Chen, Z.; Webber, M.; Barnett, J.; Wang, M. Freshwater Supply to Metropolitan Shanghai: Issues of Quality from Source to Consumers. Water 2019, 11, 2176. [Google Scholar] [CrossRef]
- Guan, X.; Wu, C.; Dai, B.; Liu, X. Study on water availability in Qingcaosha Reservoir under extremely low water conditions. Water Resour. Prot. 2013, 29, 75–78. (In Chinese) [Google Scholar]
- Jin, X.; He, Y.; Kirumba, G.; Hassan, Y.; Li, J. Phosphorus fractions and phosphate sorption-release characteristics of the sediment in the Yangtze River estuary reservoir. Ecol. Eng. 2013, 55, 62–66. [Google Scholar] [CrossRef]
- Liu, H.; Pan, D.; Chen, P. A two-year field study and evaluation of water quality and trophic state of a large shallow drinking water reservoir in Shanghai, China. Desalination Water Treat. 2015, 57, 13829–13838. [Google Scholar] [CrossRef]
- Xu, C.; Zhang, J.; Bi, X.; Xu, Z.; He, Y.; Gin, K.Y. Developing an integrated 3D-hydrodynamic and emerging contaminant model for assessing water quality in a Yangtze Estuary Reservoir. Chemosphere 2017, 188, 218–230. [Google Scholar] [CrossRef]
- Yuan, P.; Wu, X.; Xia, Y.; Peng, C.; Tong, H.; Liu, J.; Jiang, L.; Wang, X. Spatial and seasonal variations and risk assessment for heavy metals in surface sediments of the largest river-embedded reservoir in China. Environ. Sci Pollut. Res. Int. 2020. [Google Scholar] [CrossRef]
- Wang, H.; Gilbert, J.A.; Zhu, Y.; Yang, X. Salinity is a key factor driving the nitrogen cycling in the mangrove sediment. Sci. Total Environ. 2018, 631–632, 1342–1349. [Google Scholar] [CrossRef]
- Weatherburn, M.W. Phenol-hypochlorite reaction for determination of ammonia. Anal. Chem. 1967, 39, 971–974. [Google Scholar] [CrossRef]
- Kelso, B.H.L.; Smith, R.V.; Laughlin, R.J.; Lennox, S.D. Dissimilatory nitrate reduction in anaerobic sediments leading to river nitrite accumulation. Appl. Environ. Microbiol. 1997, 63, 4679–4685. [Google Scholar] [CrossRef]
- Rice, E.W.; Baird, R.B.; Eaton, A.D.; Clesceri, L.S. (Eds.) Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
- Dong, L.; Yang, Z.; Liu, X. Phosphorus fractions, sorption characteristics, and its release in the sediments of Baiyangdian Lake, China. Environ. Monit. Assess. 2011, 179, 335–345. [Google Scholar] [CrossRef]
- Ruban, V.; López-Sánchez, J.F.; Pardo, P.; Rauret, G.; Muntau, H.; Quevauviller, P. Harmonized protocol and certified reference material for the determination of extractable contents of phosphorus in freshwater sediments—A synthesis of recent works. Fresenius J. Anal. Chem. 2001, 370, 224–228. [Google Scholar] [CrossRef] [PubMed]
- Mudroch, A.; Azcue, J.M. Manual of Aquatic Sediment Sampling; CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Chi, Q.; Yan, M. Handbook of Elemental Abundance for Applied Geochemistry; Geological Publishing House: Beijing, China, 2007. (In Chinese) [Google Scholar]
- Ji, X.; Dahlgren, R.A.; Zhang, M. Comparison of seven water quality assessment methods for the characterization and management of highly impaired river systems. Environ. Monit. Assess. 2016, 188, 15. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Heilig, G.K.; Chen, J.; Heino, M. Interactions between economic growth and environmental quality in Shenzhen, China’s first special economic zone. Ecol. Econ. 2007, 62, 559–570. [Google Scholar] [CrossRef]
- Da Silva, G.S.; de Sousa, E.R.; Konrad, C.; Carla, C.B.; Pauli, J.; Pereirac, A. Phosphorus and Nitrogen in Waters of the Ocoí River Sub-basin, Itaipu Reservoir Tributary. J. Braz. Chem. Soc. 2009, 20, 1580–1588. [Google Scholar] [CrossRef][Green Version]
- Li, N.X.; Xu, J.F.; Yin, W.; Chen, Q.Z.; Wang, J.; Shi, Z.H. Effect of local watershed landscapes on the nitrogen and phosphorus concentrations in the waterbodies of reservoir bays. Sci. Total Environ. 2020, 716, 137132. [Google Scholar] [CrossRef]
- Le, Q.; Guan, X.; Liu, X.; Wang, Z.; Liu, C. Study on the water intake site selection and water intake mode of Qingcaosha Reservoir. Water Wastewater Eng. 2009, 35, 46–51. (In Chinese) [Google Scholar] [CrossRef]
- Zheng, M.; Sheng, Y.; Sun, R.; Tian, C.; Zhang, H.; Ning, J.; Sun, Q.; Li, Z.; Bottrell, S.H.; Mortimer, R.J. Identification and Quantification of Nitrogen in a Reservoir, Jiaodong Peninsula, China. Water Environ. Res. 2017, 89, 369–377. [Google Scholar] [CrossRef]
- Guo, J.; Peng, Y.; Wang, S.; Zheng, Y.; Huang, H.; Wang, Z. Long-term effect of dissolved oxygen on partial nitrification performance and microbial community structure. Bioresour. Technol. 2009, 100, 2796–2802. [Google Scholar] [CrossRef]
- Wall, L.G.; Tank, J.L.; Royer, T.V.; Bernot, M.J. Spatial and Temporal Variability in Sediment Denitrification within an Agriculturally Influenced Reservoir. Biogeochemistry 2005, 76, 85–111. [Google Scholar] [CrossRef]
- Keys, T.A.; Caudill, M.F.; Scott, D.T. Storm effects on nitrogen flux and longitudinal variability in a river–reservoir system. River Res. Appl. 2019. [Google Scholar] [CrossRef]
- Carvalho, L.; Miller, C.; Spears, B.M.; Gunn, I.D.M.; Bennion, H.; Kirika, A.; May, L. Water quality of Loch Leven: Responses to enrichment, restoration and climate change. Hydrobiologia 2011, 681, 35–47. [Google Scholar] [CrossRef]
- Yang, Z.; Liu, D.; Ji, D.; Xiao, S. Influence of the impounding process of the Three Gorges Reservoir up to water level 172.5 m on water eutrophication in the Xiangxi Bay. Sci. China Technol. Sci. 2010, 53, 1114–1125. [Google Scholar] [CrossRef]
- Scott, J.T.; Stanley, J.K.; Doyle, R.D.; Forbes, M.G.; Brooks, B.W. River–reservoir transition zones are nitrogen fixation hot spots regardless of ecosystem trophic state. Hydrobiologia 2009, 625, 61–68. [Google Scholar] [CrossRef]
- Tang, X.Q.; Wu, M.; Dai, X.C.; Chaia, P.H. Phosphorus storage dynamics and adsorption characteristics for sediment from a drinking water source reservoir and its relation with sediment compositions. Ecol. Eng. 2014, 64, 276–284. [Google Scholar] [CrossRef]
- Kumar, B.; Anshumali. Phosphorus fractionation in surface water and sediments of industrially polluted freshwater reservoir, India. Chem. Ecol. 2018, 35, 219–234. [Google Scholar] [CrossRef]
- Wang, Y.; Li, K.; Liang, R.; Han, S.; Li, Y. Distribution and Release Characteristics of Phosphorus in a Reservoir in Southwest China. Int. J. Environ. Res. Public Health 2019, 16, 303. [Google Scholar] [CrossRef]
- Quan, W.; Shi, L.; Han, J.; Ping, X.; Shen, A.; Chen, Y. Spatial and temporal distributions of nitrogen, phosphorus and heavy metals in the intertidal sediment of the Chang jiang River Estuary in China. Acta Oceanol. Sin. 2010, 29, 108–115. [Google Scholar] [CrossRef]
- Wu, X.; Ni, B. Analysis and evaluation of heavy metal in sediment of Qingcaosha reservoir. Water Purif. Techonol. 2018, 37, 5–8. (In Chinese) [Google Scholar]
- Mhamdi, B.A.; Azzouzi, A.; Mhamdi, M.A.; Sime-Ngando, T. Dynamics of the relative nitrogen and phosphorus concentrations in a reservoir situated in a semi-arid zone (Sahela, Morocco). Water Resour. Manag. 2006, 21, 983–995. [Google Scholar] [CrossRef]
- Gin, K.Y.-H.; Ramaswamy, U.; Gopalakrishnan, A.P. Comparison of Nutrient Limitation in Freshwater and Estuarine Reservoirs in Tropical Urban Singapore. J. Environ. Eng. 2011, 137, 913–919. [Google Scholar] [CrossRef]
- Xu, X.; Zhou, G.; Ding, J.; Cao, L.; Zhang, S. Pollution status of surface sediments and assessment of its potential ecological risk in the intertidal flats of Changxing Island. Mar. Sci. Bull. 2012, 31, 223–227. (In Chinese) [Google Scholar]
- Huang, Q.; Shen, H.; Wang, Z.; Liu, X.; Fu, R. Influences of natural and anthropogenic processes on the nitrogen and phosphorus fluxes of the Yangtze Estuary, China. Reg. Environ. Chang. 2006, 6, 125–131. [Google Scholar] [CrossRef]
- Tong, Y.; Bu, X.; Chen, J.; Zhou, F.; Chen, L.; Liu, M.; Tan, X.; Yu, T.; Zhang, W.; Mi, Z.; et al. Estimation of nutrient discharge from the Yangtze River to the East China Sea and the identification of nutrient sources. J. Hazard. Mater. 2017, 321, 728–736. [Google Scholar] [CrossRef] [PubMed]
| Season | Site | Pi-TN | Pollution Level of Nitrogen | Pi-TP | Pollution Level of Phosphorus | PN | Pollution Level |
|---|---|---|---|---|---|---|---|
| Spring | S1 | 0.55 | Unpolluted | 0.20 | Unpolluted | 0.47 | Unpolluted |
| S2 | 0.79 | Unpolluted | 0.14 | Unpolluted | 0.65 | Unpolluted | |
| S3 | 1.36 | Slightly polluted | 0.25 | Unpolluted | 1.12 | Slightly polluted | |
| S4 | 0.29 | Unpolluted | 0.13 | Unpolluted | 0.25 | Unpolluted | |
| S5 | 2.86 | Moderately polluted | 0.16 | Unpolluted | 2.29 | Slightly polluted | |
| S6 | 2.45 | Slightly polluted | 0.13 | Unpolluted | 1.96 | Slightly polluted | |
| Summer | S1 | 1.59 | Slightly polluted | 0.30 | Unpolluted | 1.31 | Slightly polluted |
| S2 | 0.23 | Unpolluted | 0.24 | Unpolluted | 0.24 | Unpolluted | |
| S3 | 1.48 | Slightly polluted | 0.39 | Unpolluted | 1.24 | Slightly polluted | |
| S4 | 0.18 | Unpolluted | 0.23 | Unpolluted | 0.22 | Unpolluted | |
| S5 | 2.72 | Moderately polluted | 0.56 | Slightly polluted | 2.24 | Slightly polluted | |
| S6 | 2.22 | Slightly polluted | 0.43 | Unpolluted | 1.83 | Slightly polluted | |
| Autumn | S1 | 0.69 | Unpolluted | 0.25 | Unpolluted | 0.59 | Unpolluted |
| S2 | 1.44 | Slightly polluted | 0.28 | Unpolluted | 1.19 | Slightly polluted | |
| S3 | 1.07 | Slightly polluted | 0.27 | Unpolluted | 0.89 | Slightly polluted | |
| S4 | 0.35 | Unpolluted | 0.30 | Unpolluted | 0.34 | Unpolluted | |
| S5 | 1.25 | Slightly polluted | 0.29 | Unpolluted | 1.04 | Slightly polluted | |
| S6 | 2.05 | Slightly polluted | 0.34 | Unpolluted | 1.68 | Slightly polluted | |
| Winter | S1 | 1.28 | Slightly polluted | 0.28 | Unpolluted | 1.06 | Slightly polluted |
| S2 | 0.76 | Unpolluted | 0.32 | Unpolluted | 0.66 | Unpolluted | |
| S3 | 2.59 | Moderately polluted | 0.02 | Unpolluted | 2.05 | Slightly polluted | |
| S4 | 1.20 | Slightly polluted | 0.25 | Unpolluted | 0.99 | Slightly polluted | |
| S5 | 3.98 | Moderately polluted | 0.38 | Unpolluted | 3.21 | Moderately polluted | |
| S6 | 3.64 | Moderately polluted | 0.36 | Unpolluted | 2.94 | Moderately polluted |
| TN | NH4+−N | NO3−−N | NO2−−N | TP | OP | IP | NaOH−P | HCl−P | |
|---|---|---|---|---|---|---|---|---|---|
| Flood Seasons | |||||||||
| TN | 1.000 | ||||||||
| NH4+−N | 0.331 | 1.000 | |||||||
| NO3−−N | 0.802 ** | 0.293 | 1.000 | ||||||
| NO2−−N | 0.245 | 0.462 | 0.195 | 1.000 | |||||
| TP | 0.779 ** | −0.163 | 0.683 * | 0.003 | 1.000 | ||||
| OP | 0.537 | 0.451 | 0.458 | 0.729 ** | 0.362 | 1.000 | |||
| IP | 0.318 | −0.491 | 0.361 | −0.478 | 0.535 | −0.443 | 1.000 | ||
| NaOH−P | 0.709 ** | −0.250 | 0.747 ** | 0.239 | 0.825 ** | 0.419 | 0.546 | 1.000 | |
| HCl−P | 0.092 | −0.130 | −0.055 | −0.369 | 0.147 | −0.255 | 0.200 | −0.124 | 1.000 |
| Dry Seasons | |||||||||
| TN | 1.000 | ||||||||
| NH4+−N | 0.787 ** | 1.000 | |||||||
| NO3−−N | −0.026 | −0.041 | 1.000 | ||||||
| NO2−−N | 0.720 ** | 0.754 ** | 0.094 | 1.000 | |||||
| TP | 0.508 | 0.553 | −0.561 | 0.585* | 1.000 | ||||
| OP | 0.731 ** | 0.791 ** | −0.506 | 0.771 ** | 0.856 ** | 1.000 | |||
| IP | −0.297 | −0.212 | 0.615 * | −0.507 | −0.712 ** | −0.703 * | 1.000 | ||
| NaOH−P | 0.715 ** | 0.898 ** | −0.333 | 0.707 * | 0.819 ** | 0.906 ** | −0.458 | 1.000 | |
| HCl−P | 0.132 | −0.005 | −0.652* | 0.303 | 0.737 ** | 0.532 | −0.907 ** | 0.318 | 1.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, Y.; Peng, C.; Yuan, P.; Wu, X.; Jiang, L.; Chen, S.; Song, X. Seasonal and Spatial Distribution and Pollution Assessment of Nitrogen and Phosphorus in Sediments from One of the World’s Largest Tidal Reservoirs. Water 2021, 13, 395. https://doi.org/10.3390/w13040395
Shen Y, Peng C, Yuan P, Wu X, Jiang L, Chen S, Song X. Seasonal and Spatial Distribution and Pollution Assessment of Nitrogen and Phosphorus in Sediments from One of the World’s Largest Tidal Reservoirs. Water. 2021; 13(4):395. https://doi.org/10.3390/w13040395
Chicago/Turabian StyleShen, Yihao, Cheng Peng, Peng Yuan, Xuefei Wu, Lei Jiang, Si Chen, and Xinshan Song. 2021. "Seasonal and Spatial Distribution and Pollution Assessment of Nitrogen and Phosphorus in Sediments from One of the World’s Largest Tidal Reservoirs" Water 13, no. 4: 395. https://doi.org/10.3390/w13040395
APA StyleShen, Y., Peng, C., Yuan, P., Wu, X., Jiang, L., Chen, S., & Song, X. (2021). Seasonal and Spatial Distribution and Pollution Assessment of Nitrogen and Phosphorus in Sediments from One of the World’s Largest Tidal Reservoirs. Water, 13(4), 395. https://doi.org/10.3390/w13040395







