Features of the Distribution of Chlorophyll-a Concentration along the Western Coast of the Novaya Zemlya Archipelago in Spring
Abstract
:1. Introduction
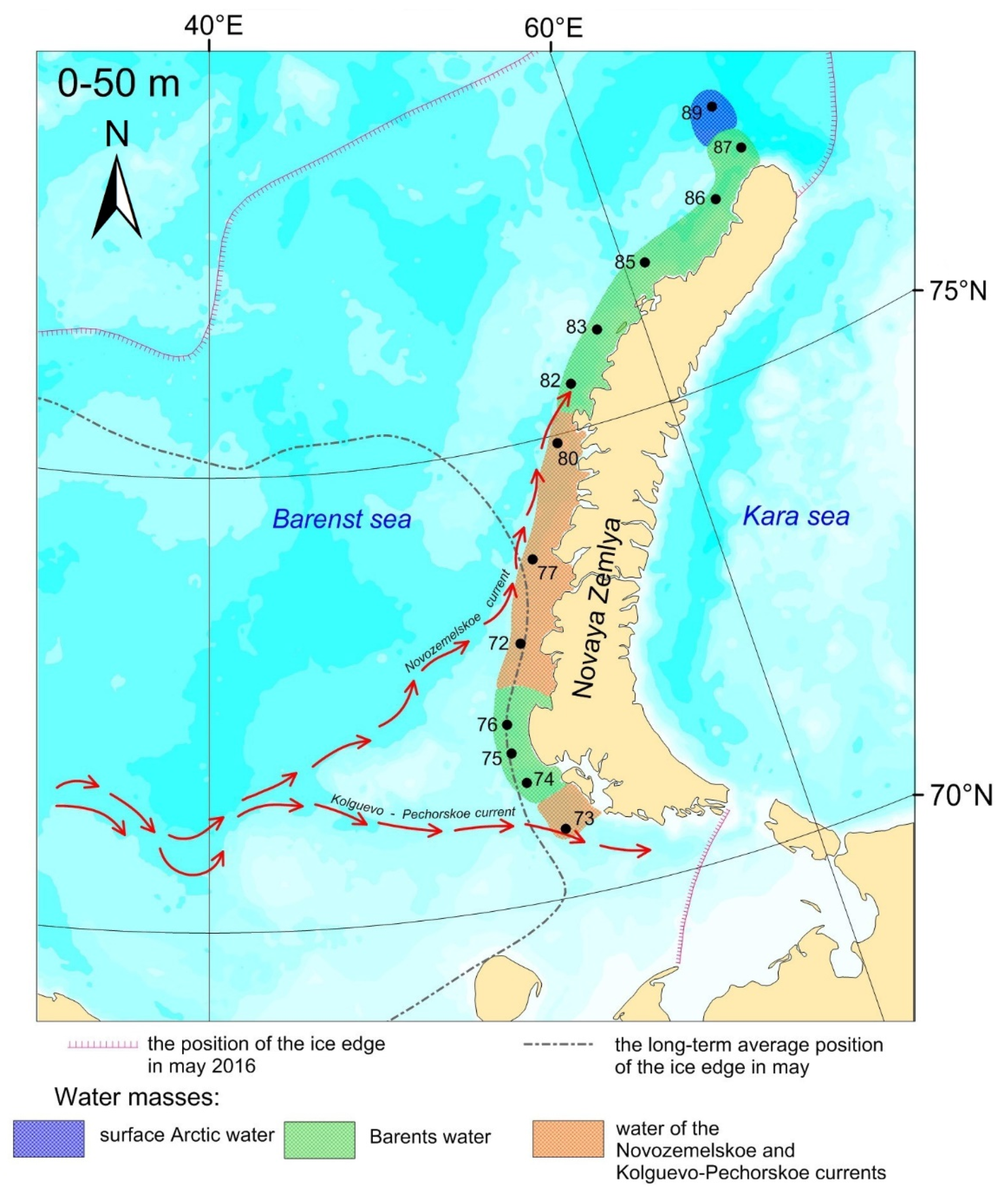
2. Materials and Methods
3. Results
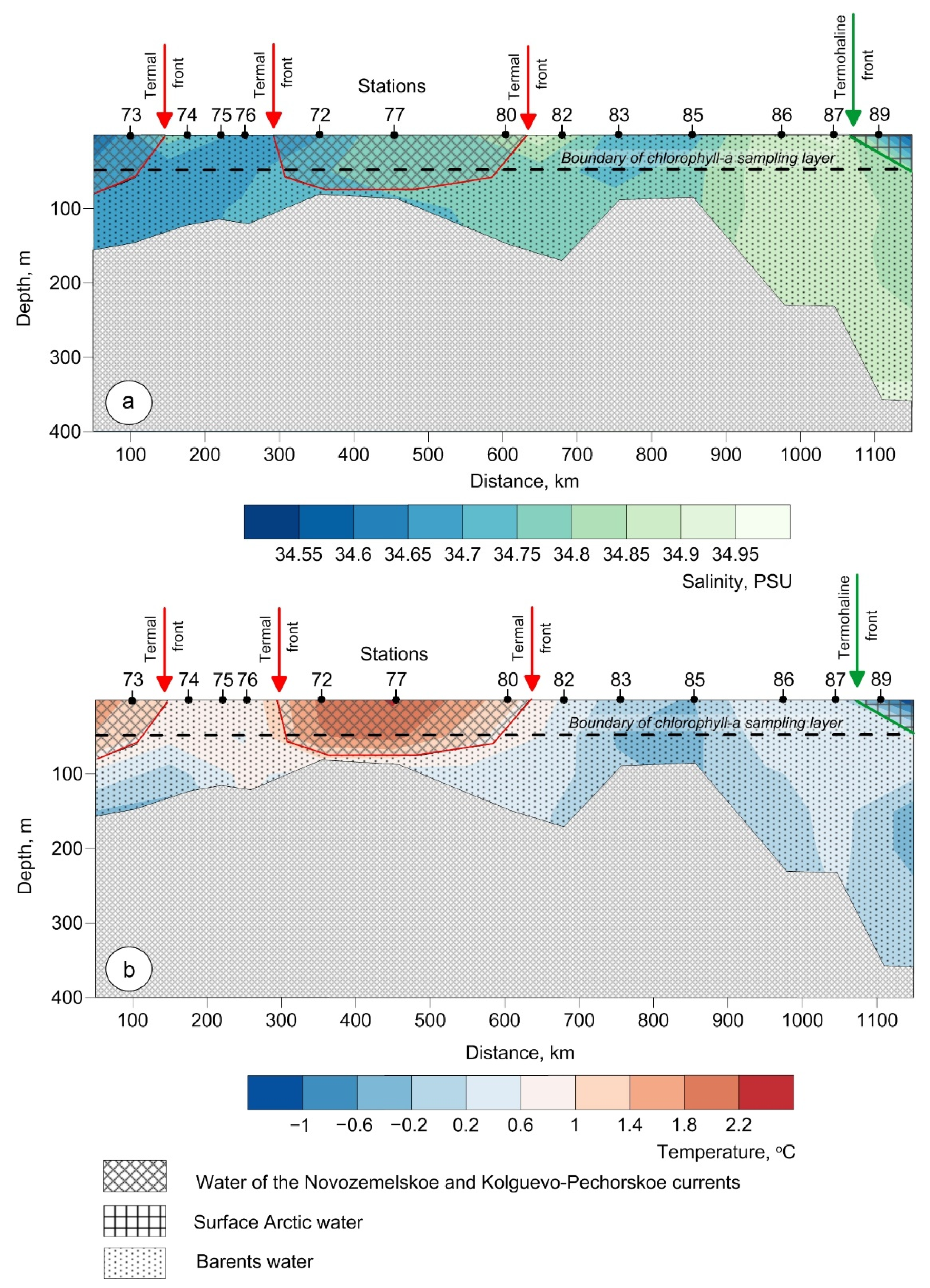
3.1. Water Masses at the Transect
3.1.1. Water of the Novozemelskoe and Kolguevo–Pechorskoe Currents
3.1.2. Barents Water
3.1.3. Arctic Surface Water
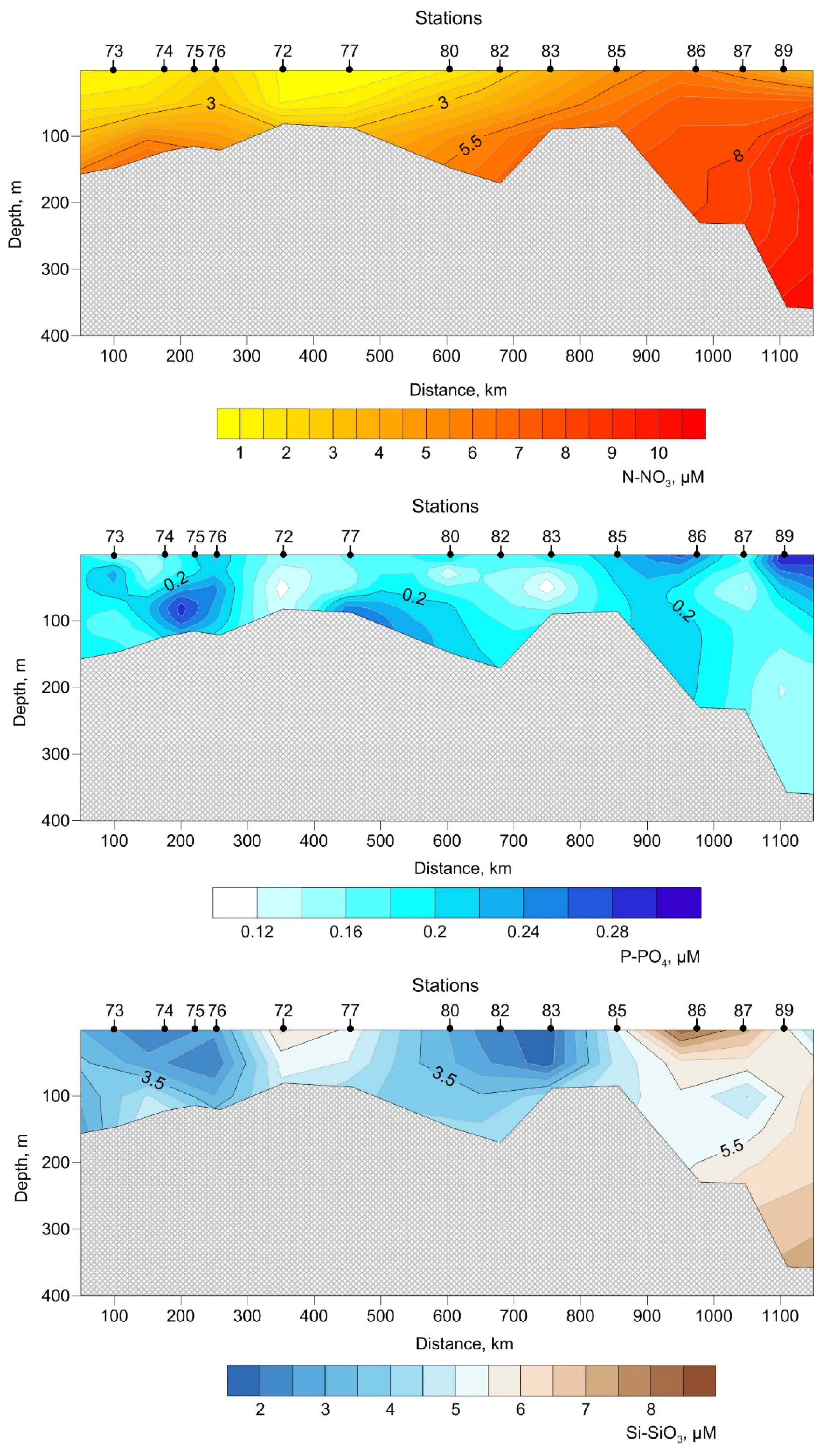
3.2. Biogeochemistry of the Transect
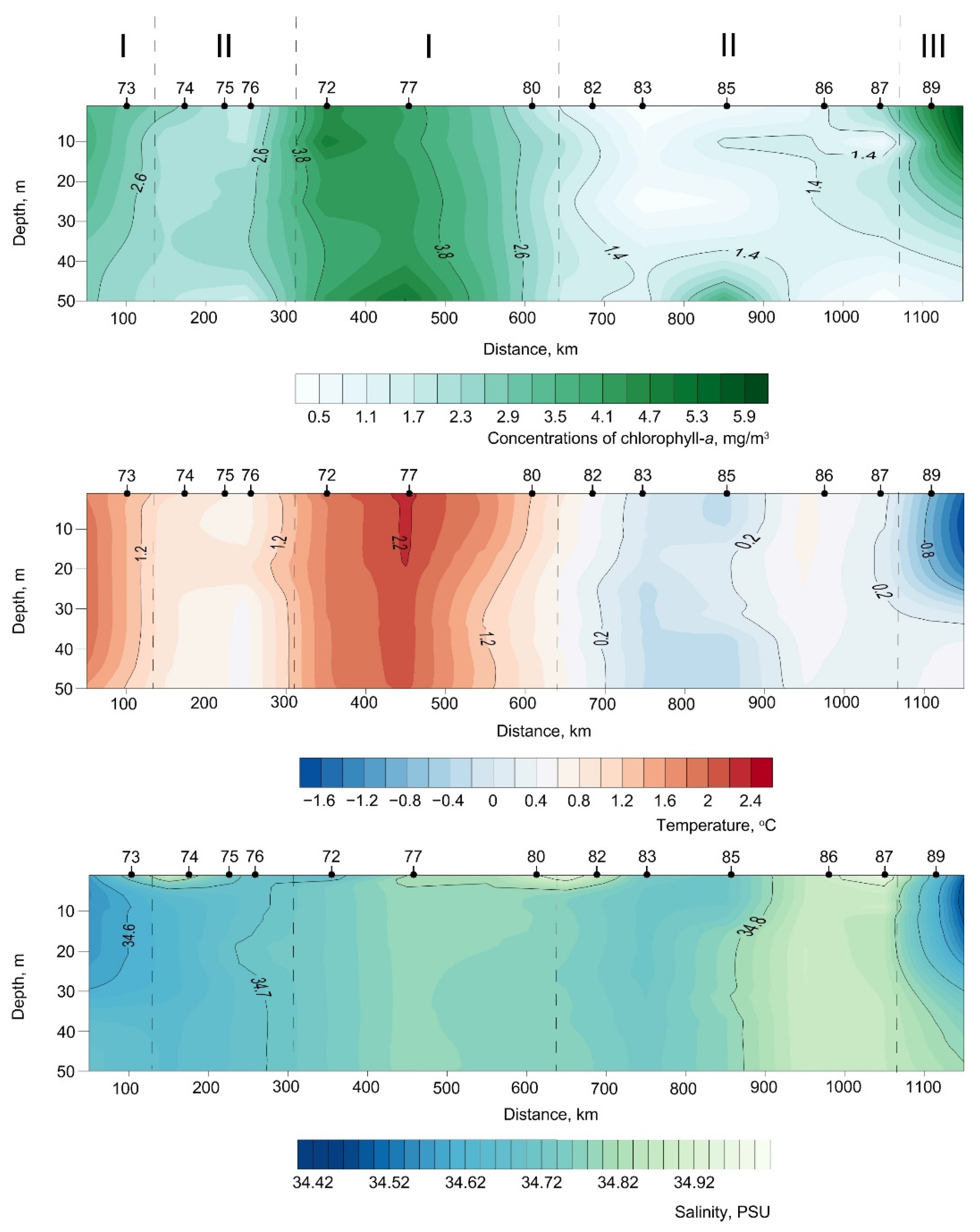
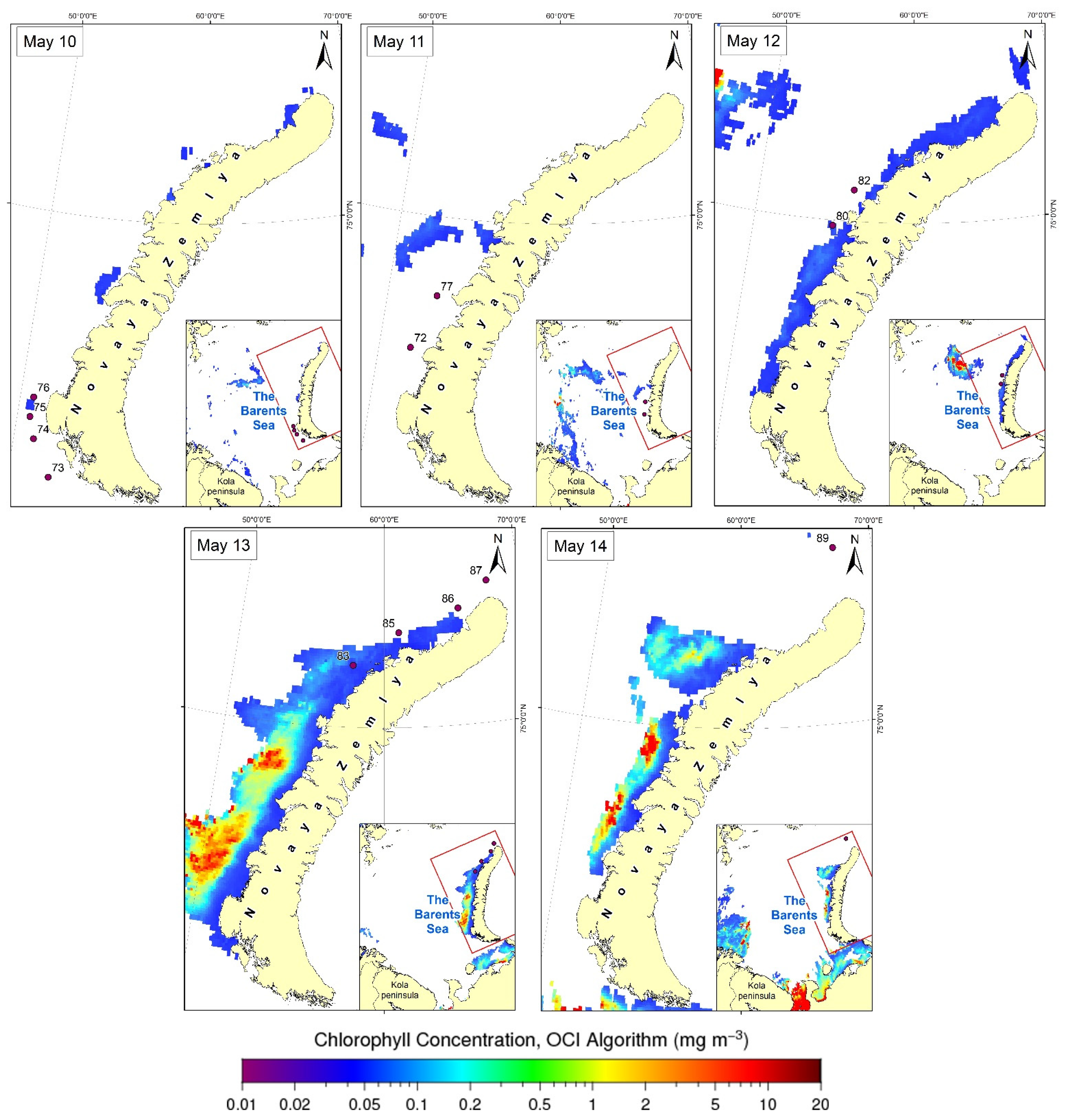
3.3. Satellite Data
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Water Masses | St | Date | Coordinates | Total Depth (m) | Chl-a (mg/m3) at Depth (m) | Satellite Data | |||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 10 | 25 | 50 | Chl-a (mg/m3) | |||||
| Water of the Novozemelskoe and Kolguevo– Pechorskoe currents | 72 | 11 May | 72°40′ N, 51°33′ E | 85 | 4.59 | 4.97 | 4.43 | 4.27 | no data |
| 73 | 10 May | 70°45′ N, 52°00′ E | 156 | 3.18 | 3.60 | 3.15 | 2.56 | no data | |
| 77 | 11 May | 73°44′ N, 52°50′ E | 89 | 3.98 | 3.83 | 4.23 | 5.08 | no data | |
| 80 | 12 May | 74°55′ N, 54°54′ E | 154 | 2.37 | 2.44 | 2.57 | 2.53 | 0.63 | |
| Mean ± std | 3.53 ± 0.97 | 3.71 ± 1.04 | 3.60 ± 0.88 | 3.61 ± 1.27 | |||||
| Barents water | 74 | 10 May | 71°20′ N, 51°01′ E | 130 | 2.58 | 1.37 | 1.68 | 1.80 | no data |
| 75 | 10 May | 71°41′ N, 50°39′ E | 114 | 1.12 | 1.26 | 1.51 | 1.41 | no data | |
| 76 | 10 May | 71°60′ N, 50°39′ E | 122 | 1.89 | 2.08 | 2.53 | 1.60 | no data | |
| 82 | 12 May | 75°30′ N, 56°07′ E | 168 | 0.52 | 1.83 | 0.88 | 1.00 | no data | |
| 83 | 13 May | 76°00′ N, 57°55′ E | 93 | 0.21 | 0.44 | 0.31 | 1.19 | 0.53 | |
| 85 | 13 May | 76°32′ N, 61°01′ E | 85 | 0.62 | 1.57 | 0.52 | 3.91 | no data | |
| 86 | 13 May | 76°53′ N, 65°16′ E | 224 | 0.95 | 1.63 | 1.54 | 0.26 | no data | |
| 87 | 13 May | 77°17′ N, 67°30′ E | 229 | 2.05 | 0.02 | 1.64 | 0.39 | no data | |
| Mean ± std | 1.24 ± 0.84 | 1.28 ± 0.70 | 1.33 ± 0.72 | 1.45 ± 1.13 | |||||
| Surface Arctic water | 89 | 14 May | 77°50′ N, 67°03′ E | 365 | 5.56 | 5.14 | 2.59 | 0.46 | no data |
References
- Comiso, J.C.; Hall, D.K. Climate trends in the Arctic as observed from space. WIREs Clim. Chang. 2014, 5, 389–409. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Kvile, Ø.K.; Stenseth, N.C.; Stige, L.C. Associations among temperature, sea ice and phytoplankton bloom dynamics in the Barents Sea. Mar. Ecol. Prog. Ser. 2020, 635, 25–36. [Google Scholar] [CrossRef]
- Neukermans, G.; Oziel, L.; Babin, M. Increased intrusion of warming Atlantic water leads to rapid expansion of temperate phytoplankton in the Arctic. Glob. Chang. Biol. 2018, 24, 2545–2553. [Google Scholar] [CrossRef]
- Ingvaldsen, R.; Loeng, H. Physical oceanography. In Ecosystem Barents Sea; Sakshaug, E., Johnsen, G., Kovacs, K.M., Eds.; Tapir Academic Press: Trondheim, Norway, 2009; pp. 33–64. [Google Scholar]
- Ardyna, M.; Arrigo, K.R. Phytoplankton dynamics in a changing Arctic Ocean. Nat. Clim. Chang. 2020, 10, 892–903. [Google Scholar] [CrossRef]
- Ardyna, M.; Mundy, C.J.; Mayot, N.; Matthes, L.C.; Oziel, L.; Horvat, C.; Leu, E.; Assmy, P.; Hill, V.; Matrai, P.A.; et al. Under-Ice Phytoplankton Blooms: Shedding Light on the “Invisible” Part of Arctic Primary Production. Front. Mar. Sci. 2020, 7, 608032. [Google Scholar] [CrossRef]
- IPCC. 2019: IPCC Special Report on the Ocean and Cryosphere in a Changing Climate. Available online: https://www.ipcc.ch/srocc/ (accessed on 26 November 2021).
- Wang, Y.; Xiang, P.; Kang, J.H.; Ye, Y.Y.; Lin, G.M.; Yang, Q.L.; Lin, M. Microphytoplankton community structure in the western Arctic Ocean: Surface layer variability of geographic and temporal considerations in summer. Hydrobiologia 2018, 811, 295–312. [Google Scholar] [CrossRef]
- Fujiwara, A.; Hirawake, T.; Suzuki, K.; Imai, I.; Saitoh, S.I. Timing of sea ice retreat can alter phytoplankton community structure in the western Arctic Ocean. Biogeosciences 2014, 11, 1705–1716. [Google Scholar] [CrossRef] [Green Version]
- Park, J.; Kug, J.; Bader, J. Amplified Arctic warming by phytoplankton under greenhouse warming. Proc. Natl. Acad. Sci. USA 2015, 112, 5921–5926. [Google Scholar] [CrossRef] [Green Version]
- Makarevich, P.R.; Druzhkova, E.I. Seasonal Cyclic Processes in Coastal Planktonic Algocenoses of Northern Seas; Rostov-on Don Publ.: Rostov-on-Don, Russia, 2010. (In Russian) [Google Scholar]
- Biological Atlas of the Arctic Seas 2000: Plankton of the Barents and Kara Seas. Available online: https://www.nodc.noaa.gov/OC5/BARPLANK/start.html (accessed on 6 November 2021).
- Korneev, O.; Titov, O.; van der Meeren, G.I.; Arneberg, P.; Tchernova, J.; Jørgensen, M.N. Final Report 2012–2015 Joint Russian-Norwegian Monitoring Project—Ocean 3. Available online: https://www.academia.edu/28647014/Final_Report_2012_2015_Joint_Russian_Norwegian_Monitoring_Project_Ocean_3 (accessed on 5 December 2021).
- Signorini, S.R.; McClain, C.R. Environmental factors controlling the Barents Sea spring-summer phytoplankton blooms. Geophys. Res. 2009, 36, 1–5. [Google Scholar] [CrossRef]
- Politova, N.V.; Shevchenko, V.P.; Zernova, V.V. Environment and Ecosystems of Novaya Zemlya. In The Archipelago and Scelf; KSC RAS Press: Apatity, Russia, 1995. (In Russian) [Google Scholar]
- Sergeeva, V.M.; Mosharov, S.A.; Nedospasov, A.A.; Polukhin, A.A. Phytoplankton community structure in the polar front of the Eastern Barents Sea at the end of the growth season. Oceanology 2018, 58, 700–709. [Google Scholar] [CrossRef]
- Wassmann, P.; Ratkova, T.; Andreassen, I.; Vernet, M.; Pedersen, G.; Rey, F. Spring bloom development in the marginal ice zone and the central Barents Sea. Mar. Ecol. 1999, 20, 321–346. [Google Scholar] [CrossRef] [Green Version]
- Perrette, M.; Yool, A.; Quartly, G.; Popova, E. Near-ubiquity of ice-edge blooms in the Arctic. Biogeosciences 2011, 8, 515–524. [Google Scholar] [CrossRef] [Green Version]
- Engelsen, O.; Hegseth, E.; Hop, H.; Hansen, E.; Falk-Petersen, S. Spatial variability of chlorophyll-a in the marginal ice zone of the Barents Sea, with relations to sea ice and oceanographic conditions. J. Mar. Syst. 2002, 35, 79–97. [Google Scholar] [CrossRef]
- Hodal, H.; Kristiansen, S. The importance of small-celled phytoplankton in spring blooms at the marginal ice zone in the northern Barents Sea. Deep-Sea Res. 2008, 55, 2176–2185. [Google Scholar] [CrossRef] [Green Version]
- Sturluson, M.; Nielsen, T.; Wassmann, P. Bacterial abundance, biomass and production during spring blooms in the northern Barents Sea. Deep-Sea Res. 2008, 55, 2186–2198. [Google Scholar] [CrossRef]
- Unified State System on the Situation in the World Ocean. Available online: http://data.oceaninfo.ru (accessed on 6 November 2021).
- Boitsov, V.D.; Karsakov, A.L.; Trofimov, A.G. Atlantic water temperature and climate in the Barents Sea, 2000–2009. ICES J. Mar. Sci. 2012, 69, 833–840. [Google Scholar] [CrossRef]
- Alekseev, G.V. Development and amplification of global warming in the Arctic. Fundam. Appl. Climatol. 2015, 1, 11–26. (In Russian) [Google Scholar]
- Zhichkin, A.P. Peculiarities of interannual and seasonal variations of the Barents Sea ice coverage anomalies. Russ. Meteorol. Hydrol. 2015, 40, 319–326. [Google Scholar] [CrossRef]
- Water. Spectrophotometric Determination of Chlorophyll-А; (State Standard: 17.1.4.02-90); Standards: Moscow, Russia, 2001. (In Russian) [Google Scholar]
- Aminot, A.; Rey, F. Standard Procedure for the Determination of Chlorophyll A by Spectroscopic Methods; International Council for the Exploration of the Sea: Copenhagen, Denmark, 2000. [Google Scholar]
- Determination of Photosynthetic Pigments in Sea-Water, Monographs on Oceanographic Methodology; UNESCO: Paris, France, 1966; Available online: https://unesdoc.unesco.org/ark:/48223/pf0000071612 (accessed on 16 November 2021).
- Mamaev, O.I. Thermohaline Analysis of the World Ocean Waters; Hydrometeoizdat: Leningrad, Russia, 1987. (In Russian) [Google Scholar]
- Wood, E.D.; Armstrong, F.A.J.; Richards, F.A. Determination of nitrate is sea water by cadmium-copper reduction to nitrite. J. Mar. Biol. Ass. UK 1967, 47, 23–31. [Google Scholar] [CrossRef] [Green Version]
- Grasshoff, K.; Kremling, K.; Ehrhardt, M. (Eds.) Methods of Seawater Analysis; Verlag Chemie: Weinheim, Germany, 1983; pp. 125–187. [Google Scholar]
- Intergovernmental Oceanographic Commission. Chemical methods for the use in marine environmental monitoring. In IOC, Manuals and Guides; UNESCO: Paris, France, 1983. [Google Scholar]
- Vuorinen, I.; Hänninen, J.; Rajasilta, M.; Laine, P.; Eklund, J.; Montesino-Pouzols, F.; Corona, F.; Junker, K.; Meier, H.M.; Dippner, J.W. Guidelines for Chemical Analysis of Marine and Fresh Water in Environmental Monitoring of Fishery Waters and Fishery Prospective Areas of the World Ocean; VNIRO Press: Moscow, Russia, 2003. (In Russian) [Google Scholar]
- Mahotin, M.; Ivanov, V. Distribution of Atlantic water masses in the Barents Sea according to observations and numerical modeling. Proc. Hydrometeorol. Res. Cent. Russ. Fed. 2016, 361, 169–191. (In Russian) [Google Scholar]
- Ivanov, V.V.; Shapiro, G.I. Formation of a dense water cascade in the marginal ice zone in the Barents Sea. Deep Sea Res. 2005, 52, 1699–1717. [Google Scholar] [CrossRef]
- Lien, V.S.; Trofimov, A.G. Formation of Barents Sea Branch Water in the north-eastern Barents Sea. Polar Res. 2013, 32, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Rudels, B.; Anderson, L.G.; Jones, E.P. Formation and evolution of the surface mixed layer and halocline of the Arctic Ocean. J. Geophys. Res. 1996, 101, 8807–8821. [Google Scholar] [CrossRef]
- Averkiev, А;.S. Transformation of Atlantic water masses in the Barents, Kara and Laptev Sea by observation data in september 2014 year. Int. Res. J. 2017, 7, 37–41. (In Russian) [Google Scholar] [CrossRef]
- Earth Observing System Data and Information System (EOSDIS) Worldview. Available online: https://worldview.earthdata.nasa.gov/ (accessed on 6 November 2021).
- All-Russian Research Institute of Hydrometeorological Information—World Data Center (RRIHI-WDC). Available online: http://aisori-m.meteo.ru/waisori/index.xhtml?idata=5 (accessed on 6 November 2021).
- Bulygina, O.N.; Razuvaev, V.N.; Aleksandrova, T.M. Opisanie Massiva Dannykh Sutochnoy Temperatury Vozdukha i Kolichestva Osadkov na Meteorologicheskikh Stantsiyakh ROSSII i Byvshego SSSR (TTTR). [Description of the Data Array of Daily Air Temperature and Precipitation at Meteorological Stations in Russia and the Former USSR (TTTR)]. Certificate of Registration of the Database No.2014620942. Available online: http://meteo.ru/data/162-temperature-precipitation#oписание-массива-данных (accessed on 29 October 2021).
- Doerffer, R.; Fiseher, J. Concentration of chlorophyll, suspended matter, and gelbstoff case II water derived from satellite coastal zone color scanner data with inverse modeling methods. J. Geophys. Res. 1994, 99, 7457–7466. [Google Scholar] [CrossRef]
- Kuznetsova, О.А;.; Kopelevich, О.V.; Shebestov, S.V.; Burenkov, V.I.; Mosharov, S.A.; Demidov, A.B. Estimation of Chlorophyll Concentration in the Kara Sea from Data of MODIS_Aqua Satellite Scanner. Earth Observ. Remote Sens. 2013, 5, 21–31. (In Russian) [Google Scholar] [CrossRef]
- Kopelevich, O.V.; Sahling, I.V.; Vazyulya, S.V.; Glukhovets, D.I.; Sheberstov, S.V.; Burenkov, V.I.; Karalli, P.G.; Yushmanova, A.V. Bio-Optical Characteristics of the Seas, Surrounding the Western Part of Russia, from Data of the Satellite Ocean Color Scanners of 1998–2017; OOO VASh FORMAT Press: Moscow, Russia, 2018. (In Russian) [Google Scholar]
- Young-Sun, S.; Hyun-cheol, K. Empirical ocean color algorithms and bio-optical properties of the western coastal waters of Svalbard, Arctic. ISPRS J. Photogramm. Remote Sens. 2018, 139, 272–283. [Google Scholar] [CrossRef]
- Grebmeier, J.M.; Rodger Harvey, H. Plankton of the Sea of the Western Arctic; KSC RAS press: Apatity, Russia, 1997. (In Russian) [Google Scholar]
- Kuznetsov, L.L.; Shoshina, E.V. Phytocenoses of the Barents Sea (Physiological and Structural Characteristics); SSC RAS Publ: Rostov-on-Don, Russia, 2003. (In Russian) [Google Scholar]
- Wassmann, P.; Reigstad, M.; Haug, T.; Rudels, B.; Carroll, M.L.; Hop, H.; Gabrielsen, G.W.; Falk-Petersen, S.; Denisenko, S.G.; Arashkevich, E.; et al. Food webs and carbon flux in the Barents Sea. Prog. Oceanogr. 2006, 71, 232–287. [Google Scholar] [CrossRef]
- Makarevich, P.; Vodopianova, V.; Bulavina, A.; Kalinka, O. Influence of the Barents Sea Frontal zones on сhlorophyll сoncentration in Spring. KnE Life Sci. 2020, 5, 536–546. [Google Scholar] [CrossRef] [Green Version]
- Makarevich, P.R.; Larionov, V.V.; Vodopyanova, V.V.; Bulavina, A.S.; Ishkulova, T.G.; Venger, M.P.; Pastukhov, I.A.; Vashchenko, A.V. Phytoplankton of the Barents Sea at the Polar Front in Spring. Okeanology 2021, 61, 930–934, in press. [Google Scholar] [CrossRef]
- Dvoretsky., V.G.; Dvoretsky, A.G. (Murmansk Marine Biological Institute, Murmansk, Russia). Personal communication, 2021. [Google Scholar]
- Dvoretsky, V.G. Zooplankton production in the Barents Sea in Summer. Izvestya TINRO 2012, 168, 169–183. (In Russian) [Google Scholar]
- Raymont, J.E.G. Plankton and Productivity in the Oceans; Pergamon Press Ltd.: Oxford, UK, 1963; p. 660. Available online: https://archive.org/details/planktonproducti0000raym/page/n7/mode/2up (accessed on 26 November 2021).
- Pasculli, L.; Piermattei, V.; Madonia, A.; Bruzzone, G.; Caccia, M.; Ferretti, R.; Odetti, A.; Marcelli, M. New Cost-Effective Technologies Applied to the Study of the Glacier Melting Influence on Physical and Biological Processes in Kongsfjorden Area (Svalbard). J. Mar. Sci. Eng. 2020, 8, 593. [Google Scholar] [CrossRef]
- Marcelli, M.; Piermattei, V.; Gerin, R.; Brunetti, F.; Pietrosemoli, E.; Addo, S.; Boudaya, L.; Coleman, R.; Nubi, O.A.; Jojannes, R.; et al. Toward the widespread application of low-cost technologies in coastal ocean observing (Internet of Things for the Ocean). Mediterr. Mar. Sci. 2021, 22, 255–269. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Makarevich, P.R.; Vodopianova, V.V.; Bulavina, A.S.; Vashchenko, P.S.; Ishkulova, T.G. Features of the Distribution of Chlorophyll-a Concentration along the Western Coast of the Novaya Zemlya Archipelago in Spring. Water 2021, 13, 3648. https://doi.org/10.3390/w13243648
Makarevich PR, Vodopianova VV, Bulavina AS, Vashchenko PS, Ishkulova TG. Features of the Distribution of Chlorophyll-a Concentration along the Western Coast of the Novaya Zemlya Archipelago in Spring. Water. 2021; 13(24):3648. https://doi.org/10.3390/w13243648
Chicago/Turabian StyleMakarevich, Pavel R., Veronika V. Vodopianova, Aleksandra S. Bulavina, Pavel S. Vashchenko, and Tatiana G. Ishkulova. 2021. "Features of the Distribution of Chlorophyll-a Concentration along the Western Coast of the Novaya Zemlya Archipelago in Spring" Water 13, no. 24: 3648. https://doi.org/10.3390/w13243648
APA StyleMakarevich, P. R., Vodopianova, V. V., Bulavina, A. S., Vashchenko, P. S., & Ishkulova, T. G. (2021). Features of the Distribution of Chlorophyll-a Concentration along the Western Coast of the Novaya Zemlya Archipelago in Spring. Water, 13(24), 3648. https://doi.org/10.3390/w13243648









