Characterization and Treatment Technologies Applied for Produced Water in Qatar
Abstract
:1. Introduction
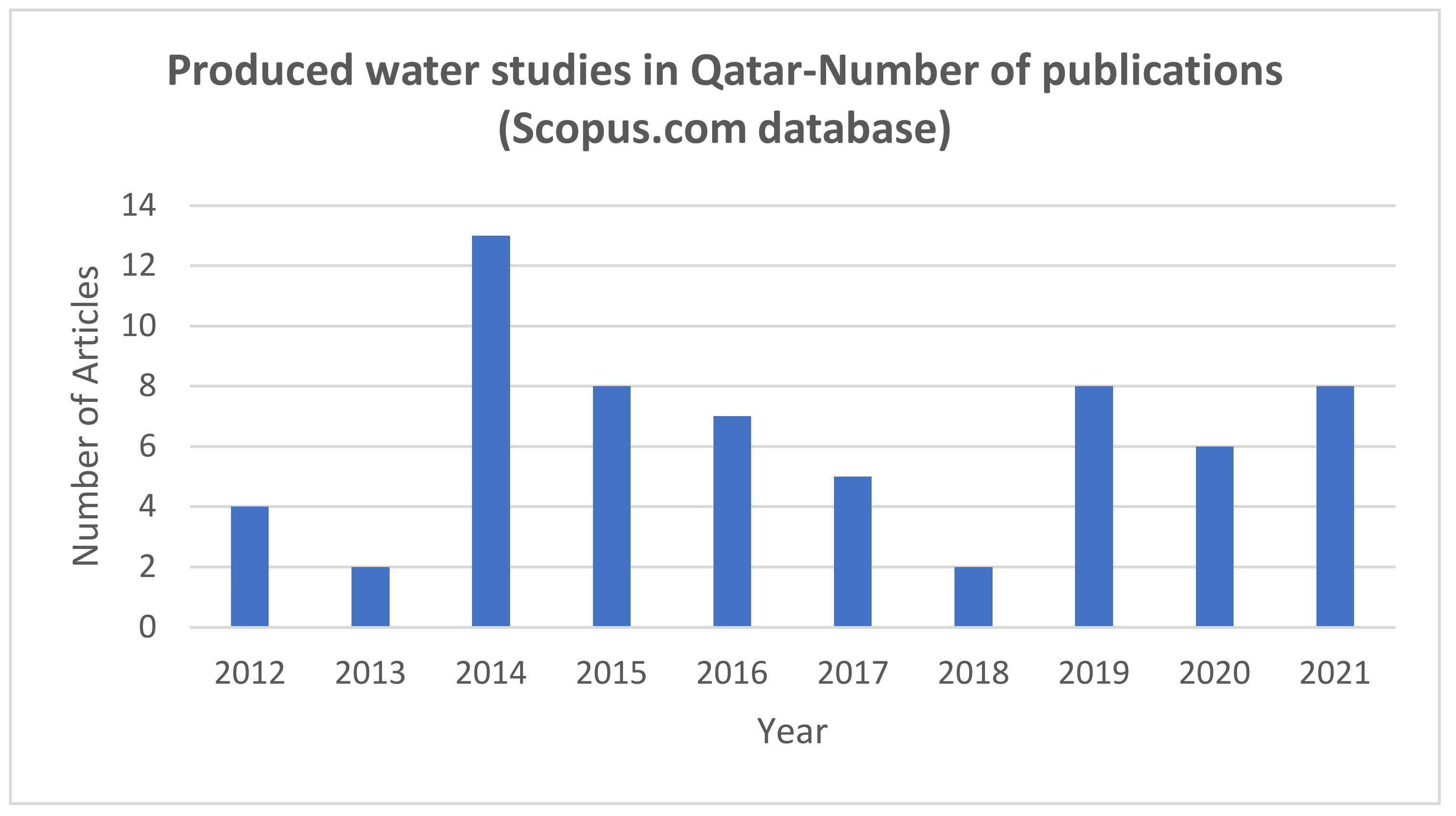
2. Methodology for the Literature Review
3. Produced Water in Qatar
4. Onshore and Offshore Produced Water Production
5. Factors Affecting Production Volume of Produced Water
6. Produced Water Characterization
6.1. pH
6.2. Chemical Oxygen Demand (COD)
6.3. Total Organic Carbon (TOC)
6.4. Biochemical Oxygen Demand (BOD)
6.5. Conductivity and Salinity
6.6. Ions and Inorganic Constituents
6.7. Total Suspended Solids (TSS)
6.8. Heavy Metal
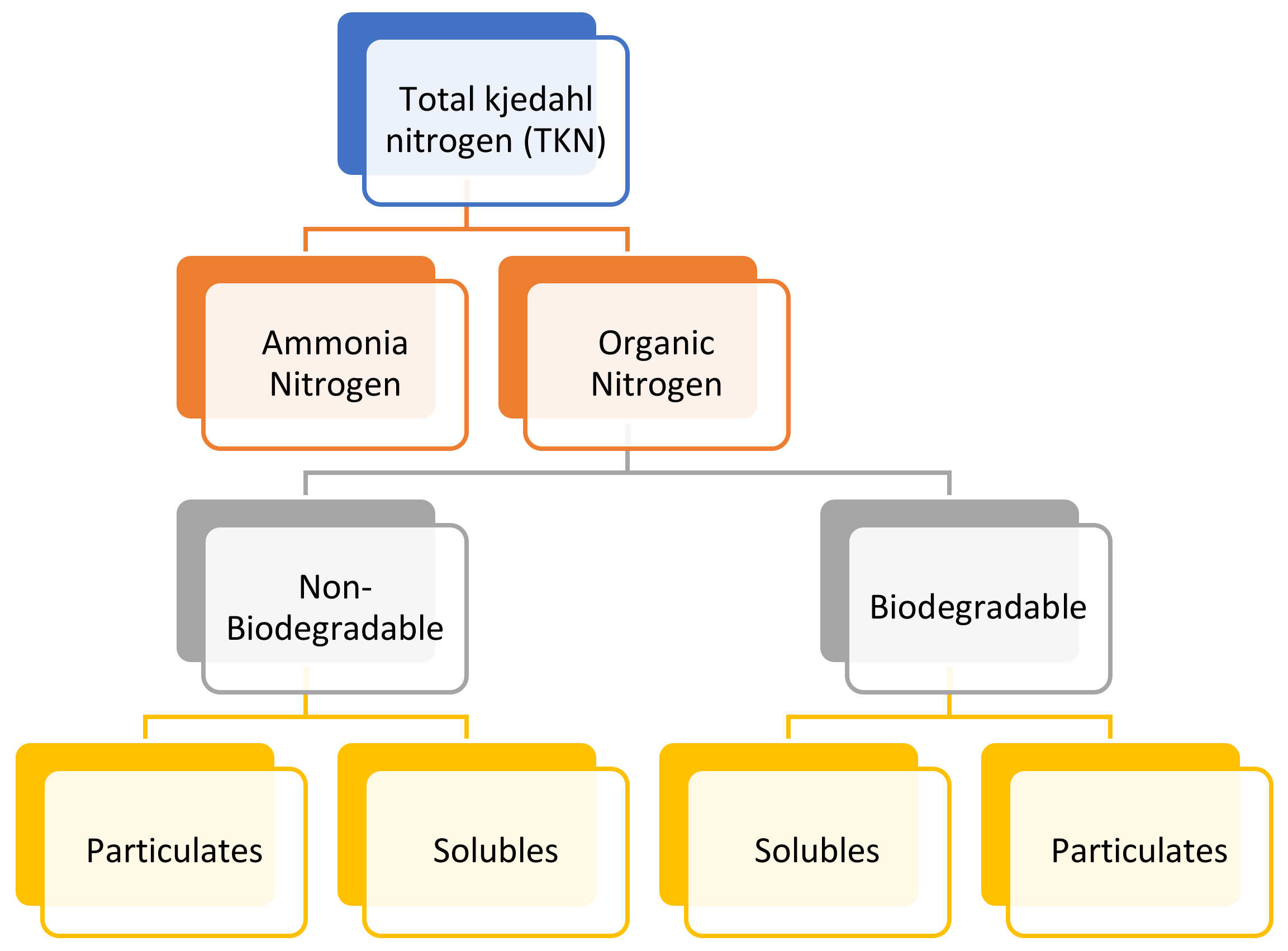
6.9. Total Kjeldahl Nitrogen (TKN)
6.10. Total Petroleum Hydrocarbon (TPH)
6.11. Total Nitrogen (TN)
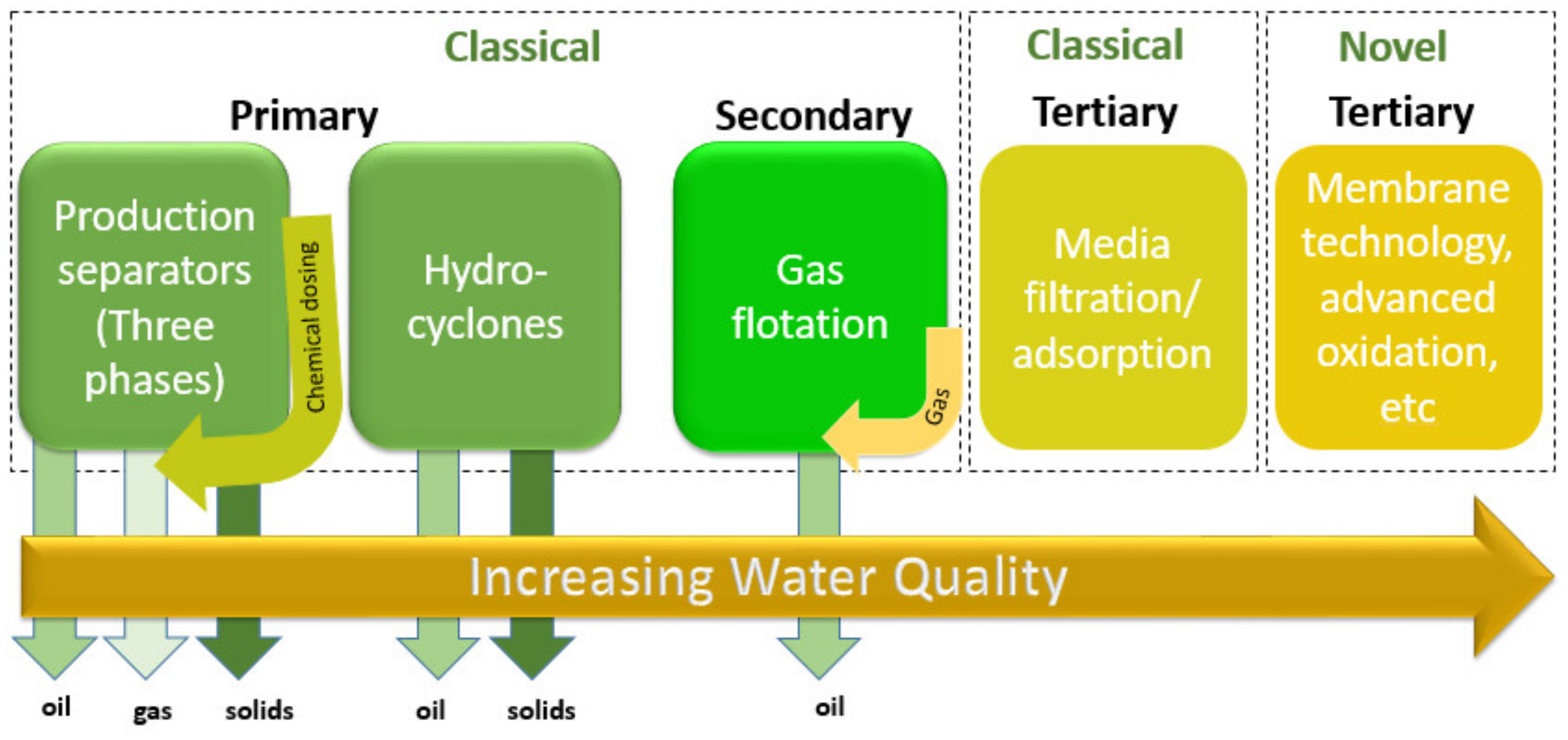
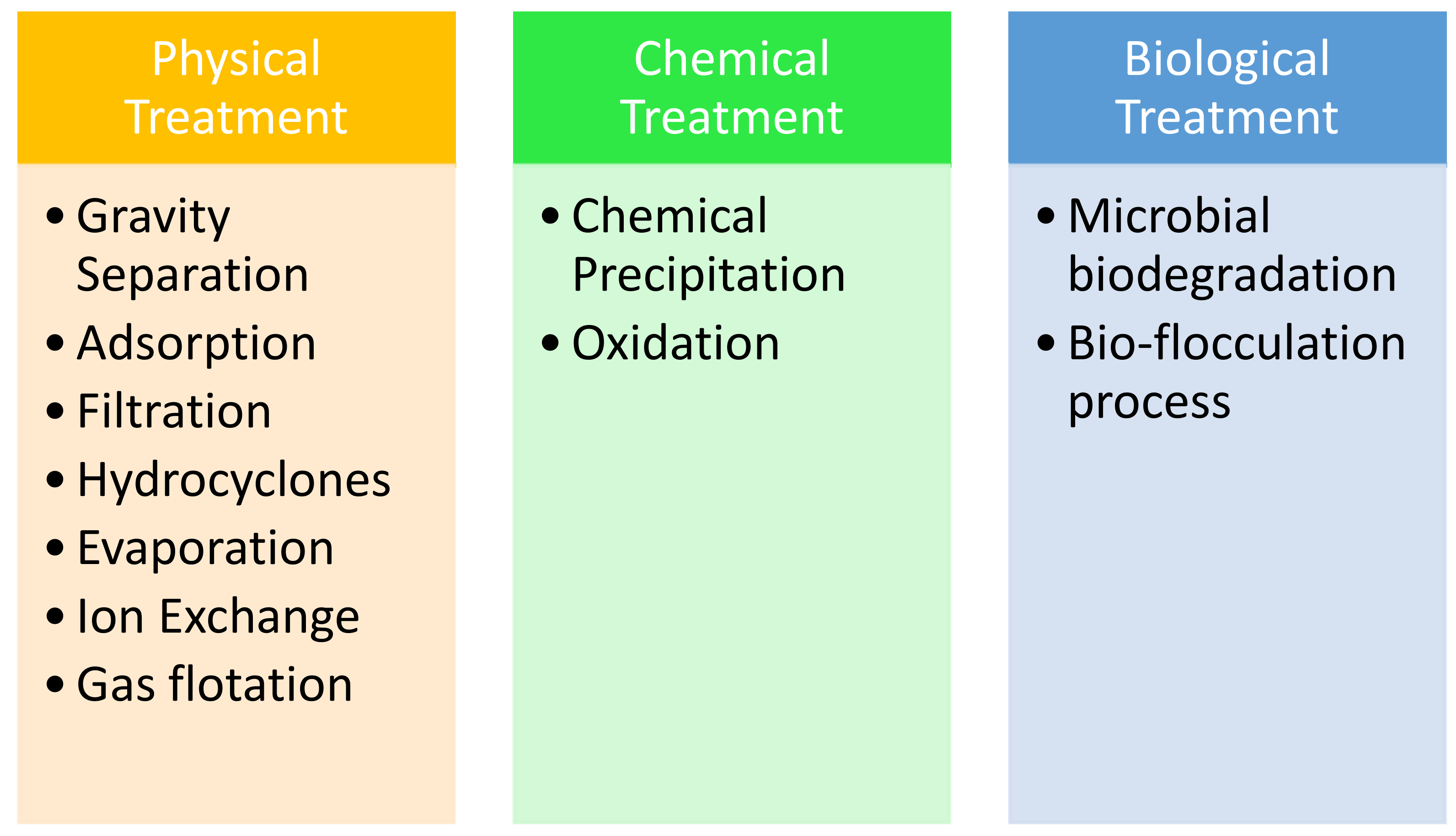
7. Treatment Processes
7.1. Gravity Separation and Adsorption
7.2. Hydrocyclones Separator
7.3. Filtration and Membrane
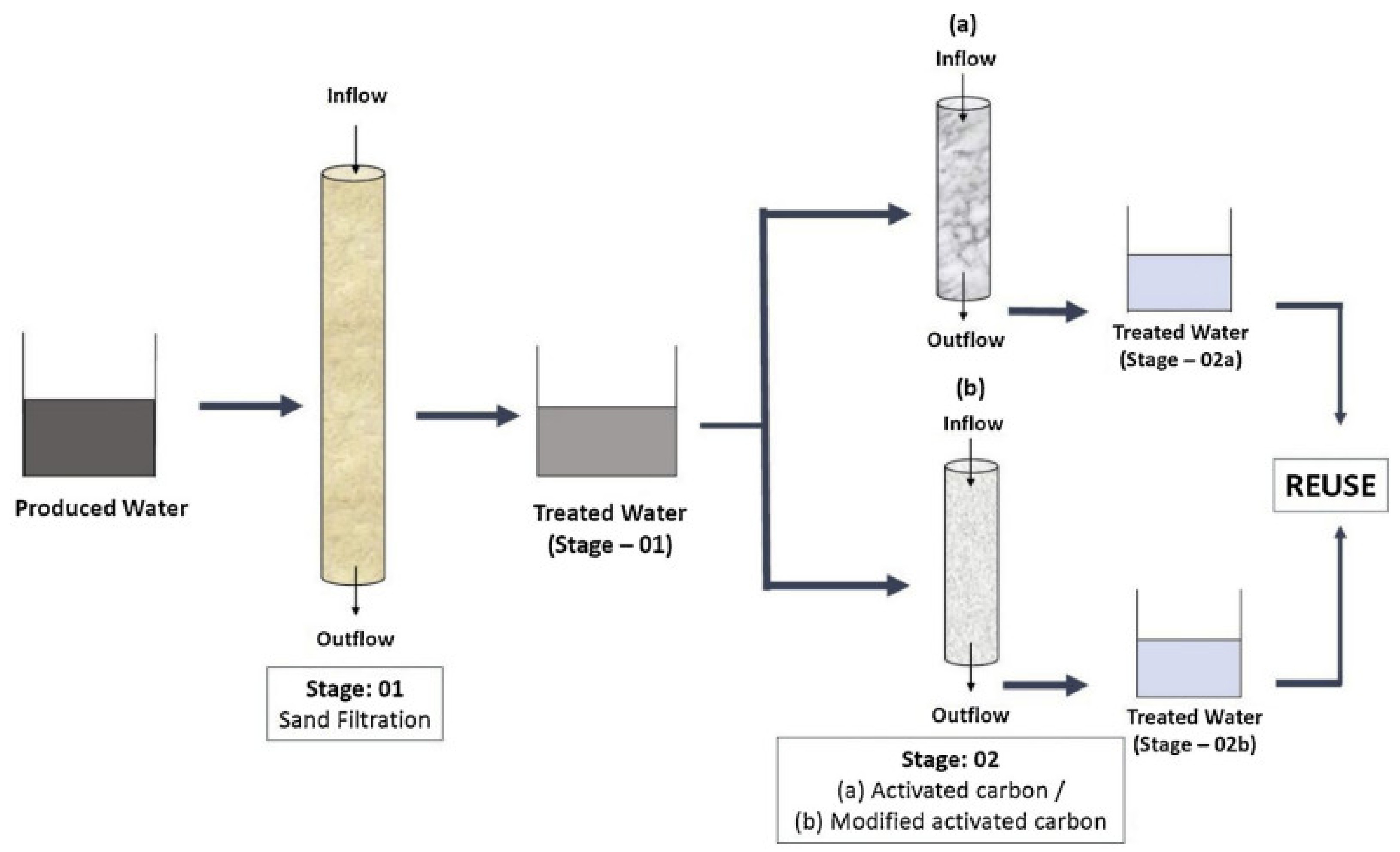
7.3.1. Sand Filtration (SF)
7.3.2. Membrane Process (MP)
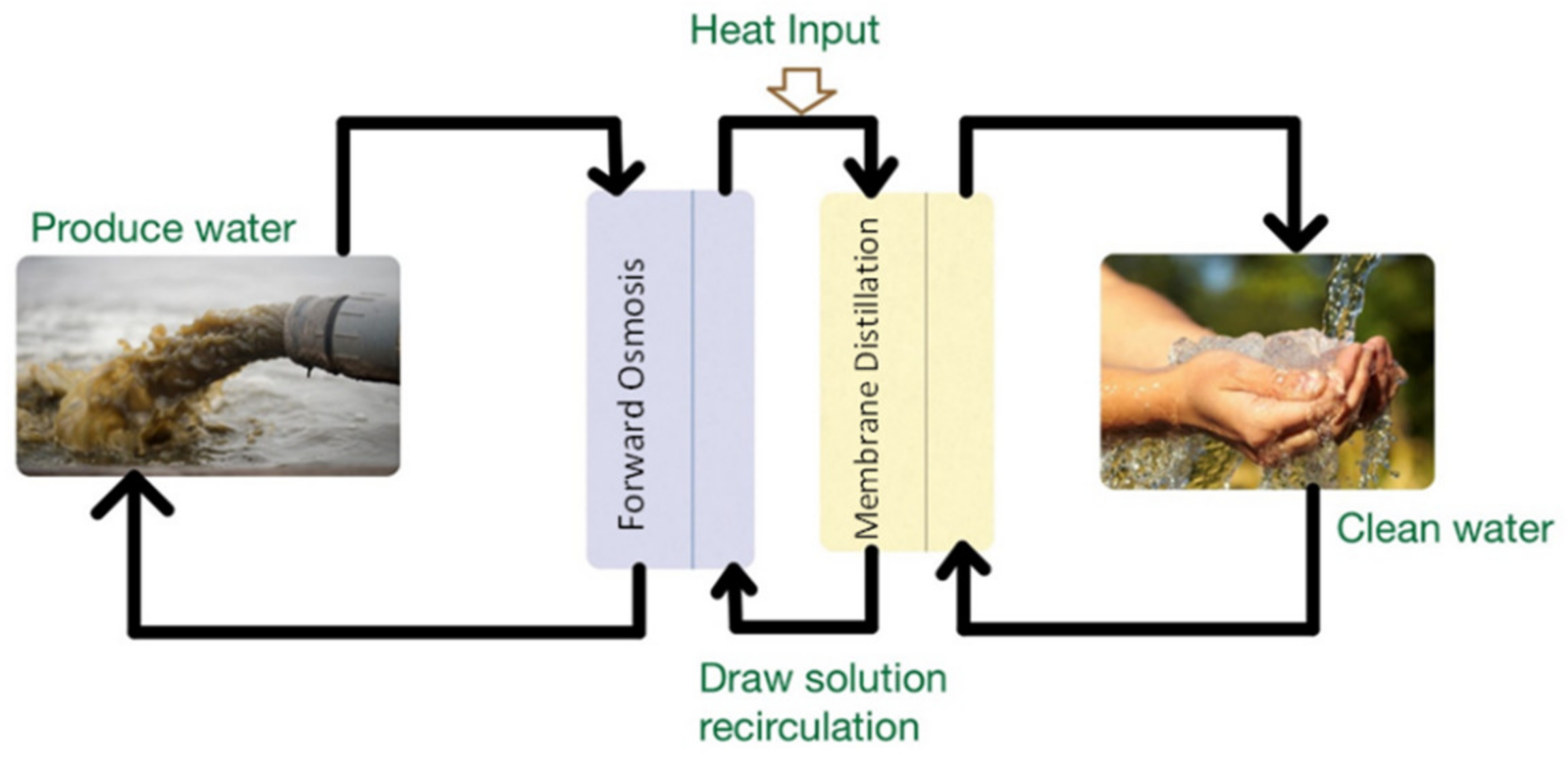
7.3.3. Membrane Distillation (MD)
7.3.4. Membrane Bioreactors (MBRs)
7.3.5. Ceramic Membrane
7.3.6. Hybrid and Asymmetric Membranes
7.3.7. Other Emerging Membrane-Based Processes
Forward Osmosis
Electrodialysis
7.4. Hydrate Inhibitors (HI)
7.5. Demulsification
7.6. Coalescing
7.7. Thermal Evaporators and Advanced Oxidation Processes (AOPs)
7.8. Surfactant Application
7.9. Activated and Modified Activated Carbon Filtration (AC and MAC)
7.10. Adsorbents
7.11. Biological Treatments
7.12. Electrocoagulation (EC)
7.13. Steel Slag Treatment
8. Case Studies of PW Treatment in Qatar
9. Future Outlook
10. Current Challenges and Environmental Issues of Produced Water
11. Reuse of Produced Water
12. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khader, E.H.; Mohammed, T.J.; Mirghaffari, N.; Salman, A.D.; Juzsakova, T.; Abdullah, T.A. Removal of organic pollutants from produced water by batch adsorption treatment. Clean Technol. Environ. Policy 2021, 23, 1–8. [Google Scholar] [CrossRef]
- Alzahrani, S.; Mohammad, A.W. Challenges and trends in membrane technology implementation for produced water treatment: A review. J. Water Process Eng. 2014, 4, 107–133. [Google Scholar] [CrossRef]
- Dickhout, J.M.; Moreno, J.; Biesheuvel, P.M.; Boels, L.; Lammertink, R.G.H.; De Vos, W.M. Produced water treatment by membranes: A review from a colloidal perspective. J. Colloid Interface Sci. 2017, 487, 523–534. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Deng, Y.; Pelusi, D. Alternatives selection for produced water management: A network-based methodology. Eng. Appl. Artif. Intell. 2020, 91, 103556. [Google Scholar] [CrossRef]
- Alammar, A.; Park, S.H.; Williams, C.J.; Derby, B.; Szekely, G. Oil-in-water separation with graphene-based nanocomposite membranes for produced water treatment. J. Membr. Sci. 2020, 603, 118007. [Google Scholar] [CrossRef]
- Ganiyu, S.O.; Sable, S.; El-Din, M.G. Advanced oxidation processes for the degradation of dissolved organics in produced water: A review of process performance, degradation kinetics and pathway. Chem. Eng. J. 2021, 429, 132492. [Google Scholar] [CrossRef]
- Hendges, L.T.; Costa, T.C.; Temochko, B.; González, S.Y.G.; Mazur, L.P.; Marinho, B.A.; da Silva, A.; Weschenfelder, S.E.; de Souza, A.A.U.; de Souza, S.M.G.U. Adsorption and desorption of water-soluble naphthenic acid in simulated offshore oilfield produced water. Process Saf. Environ. Prot. 2021, 145, 262–272. [Google Scholar] [CrossRef]
- McLaughlin, M.C.; Blotevogel, J.; Watson, R.A.; Schell, B.; Blewett, T.A.; Folkerts, E.J.; Goss, G.G.; Truong, L.; Tanguay, R.L.; Argueso, J.L.; et al. Mutagenicity assessment downstream of oil and gas produced water discharges intended for agricultural beneficial reuse. Sci. Total Environ. 2020, 715, 136944. [Google Scholar] [CrossRef]
- Babu, P.; Bollineni, C.; Daraboina, N. Energy Analysis of Methane-Hydrate-Based Produced Water Desalination. Energy Fuels 2021, 35, 2514–2519. [Google Scholar] [CrossRef]
- Ali, S.; Ijaola, A.O.; Asmatulu, E. Multifunctional water treatment system for oil and gas-produced water. Sustain. Water Resour. Manag. 2021, 7, 89. [Google Scholar] [CrossRef]
- Madadizadeh, A.; Sadeghein, A.; Riahi, S. A Comparison of Different Nanoparticles’ Effect on Fine Migration by Low Salinity Water Injection for Oil Recovery: Introducing an Optimum Condition. J. Energy Resour. Technol. 2021, 144, 013005. [Google Scholar] [CrossRef]
- Tale, F.; Kalantariasl, A.; Malayeri, M.R. Estimating transition time from deep filtration of particles to external cake during produced water re-injection and disposal. Part. Sci. Technol. 2021, 39, 312–321. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, M.; Bo, Z.; Bedrikovetsky, P.; Le-Hussain, F. Effect of temperature on mineral reactions and fines migration during low-salinity water injection into Berea sandstone. J. Pet. Sci. Eng. 2021, 202, 108482. [Google Scholar] [CrossRef]
- Zheng, J.; Chen, B.; Thanyamanta, W.; Hawboldt, K.; Zhang, B.; Liu, B. Offshore produced water management: A review of current practice and challenges in harsh/Arctic environments. Mar. Pollut. Bull. 2016, 104, 7–19. [Google Scholar] [CrossRef]
- Torres, L.; Yadav, O.P.; Khan, E. A review on risk assessment techniques for hydraulic fracturing water and produced water management implemented in onshore unconventional oil and gas production. Sci. Total Environ. 2016, 539, 478–493. [Google Scholar] [CrossRef]
- Sosa-Fernandez, P.; Miedema, S.; Bruning, H.; Leermakers, F.; Post, J.; Rijnaarts, H. Effects of feed composition on the fouling on cation-exchange membranes desalinating polymer-flooding produced water. J. Colloid Interface Sci. 2021, 584, 634–646. [Google Scholar] [CrossRef]
- Simona, C.; Raluca, I.; Anita-Laura, R.; Andrei, S.; Raluca, S.; Bogdan, T.; Elvira, A.; Catalin-Ilie, S.; Claudiu, F.R.; Daniela, I.-E.; et al. Synthesis, characterization and efficiency of new organically modified montmorillonite polyethersulfone membranes for removal of zinc ions from wastewasters. Appl. Clay Sci. 2017, 137, 135–142. [Google Scholar] [CrossRef]
- Su, Y.N.; Lin, W.S.; Hou, C.H.; Den, W. Performance of integrated membrane filtration and electrodialysis processes for copper recovery from wafer polishing wastewater. J. Water Process. Eng. 2014, 4, 149–158. [Google Scholar] [CrossRef]
- Eljamal, O.; Jinno, K.; Hosokawa, T. Modeling of solute transport and biological sulfate reduction using low cost electron donor. J. Environ. Geol. 2009, 56, 1605–1613. [Google Scholar] [CrossRef]
- Maamoun, I.; Eljamal, O.; Falyouna, O.; Eljamal, R.; Sugihara, Y.J.W.S. Stimulating effect of magnesium hydroxide on aqueous characteristics of iron nanocomposites. Water Sci. Technol. 2019, 80, 1996–2002. [Google Scholar] [CrossRef]
- Chekioua, A.; Delimi, R. Purification of H2SO4 of pickling bath contaminated by Fe (II) ions using electrodialysis process. Energy Procedia 2015, 74, 1418–1433. [Google Scholar] [CrossRef] [Green Version]
- Tristán Teja, C.; Fallanza Torices, M.; Ibáñez Mendizábal, R.; Ortiz Uribe, I. Reverse electrodialysis: Potential reduction in energy and emissions of desalination. Appl. Sci. 2020, 10, 7317. [Google Scholar] [CrossRef]
- Căprărescu, S.; Zgârian, R.G.; Tihan, G.T.; Purcar, V.; Totu, E.E.; Modrogan, C.; Chiriac, A.-L.; Nicolae, C.A. Biopolymeric membrane enriched with chitosan and silver for metallic ions removal. Polymers 2020, 12, 1792. [Google Scholar] [CrossRef]
- Caprarescu, S.; Radu, A.L.; Purcar, V.; Sarbu, A.; Vaireanu, D.I.; Ianchis, R.; Ghiurea, M. Removal of copper ions from simulated wastewaters using different bicomponent polymer membranes. Water Air Soil Pollut. 2014, 225, 2079. [Google Scholar] [CrossRef]
- Obotey Ezugbe, E.; Rathilal, S. Membrane technologies in wastewater treatment: A review. Membranes 2020, 10, 89. [Google Scholar] [CrossRef]
- Eljamal, R.; Eljamal, O.; Maamoun, I.; Yilmaz, G.; Sugihara, Y. Enhancing the characteristics and reactivity of nZVI: Polymers effect and mechanisms. J. Mol. Liq. 2020, 315, 113714. [Google Scholar] [CrossRef]
- Osama, E.; Junya, O.; Kazuaki, H. Removal of phosphorus from water using marble dust as sorbent material. J. Environ. Prot. Sci. 2012, 2012, 21755. [Google Scholar]
- Abd Halim, N.S.; Wirzal, M.D.H.; Hizam, S.M.; Bilad, M.R.; Nordin, N.A.H.M.; Sambudi, N.S.; Putra, Z.A.; Yusoff, A.R.M. Recent development on electrospun nanofiber membrane for produced water treatment: A review. J. Environ. Chem. Eng. 2021, 9, 104613. [Google Scholar] [CrossRef]
- Liang, H.; Zou, C.; Tang, W. Development of novel polyether sulfone mixed matrix membranes to enhance antifouling and sustainability: Treatment of oil sands produced water (OSPW). J. Taiwan Inst. Chem. Eng. 2021, 118, 215–222. [Google Scholar] [CrossRef]
- Weschenfelder, S.E.; Fonseca, M.J.C.; Borges, C.P. Treatment of produced water from polymer flooding in oil production by ceramic membranes. J. Pet. Sci. Eng. 2021, 196, 108021. [Google Scholar]
- Wu, M.; Zhai, M.; Li, X. Adsorptive removal of oil drops from ASP flooding-produced water by polyether polysiloxane-grafted ZIF-8. Powder Technol. 2021, 378, 76–84. [Google Scholar] [CrossRef]
- Qi, P.; Sun, D.; Gao, J.; Liu, S.; Wu, T.; Li, Y. Demulsification and bio-souring control of alkaline-surfactant-polymer flooding produced water by Gordonia sp. TD-4. Sep. Purif. Technol. 2021, 263, 118359. [Google Scholar] [CrossRef]
- Husveg, R.; Husveg, T.; van Teeffelen, N.; Ottestad, M.; Hansen, M.R. Variable Step Size P&O Algorithms for Coalescing Pump/Deoiling Hydrocyclone Produced Water Treatment System. Model. Identif. Control 2020, 41, 13–27. [Google Scholar]
- Jin, Y.; Davarpanah, A. Using photo-fenton and floatation techniques for the sustainable management of flow-back produced water reuse in shale reservoirs exploration. Water Air Soil Pollut. 2020, 231, 441. [Google Scholar] [CrossRef]
- Simões, A.J.A.; Macêdo-Júnior, R.O.; Santos, B.L.P.; Silva, D.P.; Ruzene, D.S. A bibliometric study on the application of advanced oxidation processes for produced water treatment. Water Air Soil Pollut. 2021, 232, 297. [Google Scholar] [CrossRef]
- Coha, M.; Farinelli, G.; Tiraferri, A.; Minella, M.; Vione, D. Advanced oxidation processes in the removal of organic substances from produced water: Potential, configurations, and research needs. Chem. Eng. J. 2021, 414, 128668. [Google Scholar] [CrossRef]
- Hammack, R.; Mosser, M.H. Produced water management. In Coal Bed Methane; Elsevier: Amsterdam, The Netherlands, 2020; pp. 349–369. [Google Scholar]
- Camarillo, M.K.; Stringfellow, W.T. Biological treatment of oil and gas produced water: A review and meta-analysis. Clean Technol. Environ. Policy 2018, 20, 1127–1146. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Lu, H.; Li, Y.; Xu, H.; Pan, Z.; Dai, P.; Wang, H.; Yang, Q. A review of treatment technologies for produced water in offshore oil and gas fields. Sci. Total Environ. 2021, 775, 145485. [Google Scholar] [CrossRef]
- Duraisamy, R.T.; Beni, A.H.; Henni, A. State of the art treatment of produced water. In Water Treatment; Books on Demand: Norderstedt, Germany, 2013; pp. 199–222. [Google Scholar]
- Scanlon, B.R.; Reedy, R.C.; Xu, P.; Engle, M.; Nicot, J.P.; Yoxtheimer, D.; Yang, Q.; Ikonnikova, S. Can we beneficially reuse produced water from oil and gas extraction in the US? Sci. Total Environ. 2020, 717, 137085. [Google Scholar] [CrossRef]
- Global Produced Water Treatment Industry (2021 Report). Available online: https://www.reportlinker.com/p06032674/Global-Produced-Water-Treatment-Industry.html?utm_source=GNW (accessed on 24 October 2021).
- The U.S. Energy Information Agency. Qatar: Oil Production. Available online: https://www.theglobaleconomy.com/Qatar/oil_production/ (accessed on 24 October 2021).
- Kabyl, A.; Yang, M.; Abbassi, R.; Li, S. A risk-based approach to produced water management in offshore oil and gas operations. Process Saf. Environ. Prot. 2020, 139, 341–361. [Google Scholar] [CrossRef]
- Santos, K.A.; Gomes, T.M.; Rossi, F.; Kushida, M.M.; Del Bianchi, V.L.; Ribeiro, R.; Alves, M.S.M.; Tommaso, G. Water reuse: Dairy effluent treated by a hybrid anaerobic biofilm baffled reactor and its application in lettuce irrigation. Water Supply 2021, 21, 1980–1993. [Google Scholar] [CrossRef]
- Rahman, A.; Agrawal, S.; Nawaz, T.; Pan, S.; Selvaratnam, T. A review of algae-based produced water treatment for biomass and biofuel production. Water 2020, 12, 2351. [Google Scholar] [CrossRef]
- Lahlou, F.Z.; Mackey, H.R.; McKay, G.; Onwusogh, U.; Al-Ansari, T. Water planning framework for alfalfa fields using treated wastewater fertigation in Qatar: An energy-water-food nexus approach. Comput. Chem. Eng. 2020, 141, 106999. [Google Scholar] [CrossRef]
- Ahmad, A.Y.; Al-Ghouti, M.A. Approaches to achieve sustainable use and management of groundwater resources in Qatar: A review. Groundw. Sustain. Dev. 2020, 11, 100367. [Google Scholar] [CrossRef]
- Kondash, A.J.; Redmon, J.H.; Lambertini, E.; Feinstein, L.; Weinthal, E.; Cabrales, L.; Vengosh, A. The impact of using low-saline oilfield produced water for irrigation on water and soil quality in California. Sci. Total Environ. 2020, 733, 139392. [Google Scholar] [CrossRef]
- Minier-matar, J.; Hussain, A.; Janson, A.; Wang, R.; Fane, A.G.; Adham, S. Application of Forward Osmosis for Reducing Volume of Produced/Process Water from Oil and Gas Operations. Desalination 2015, 376, 1–8. [Google Scholar] [CrossRef]
- Minier-Matar, J.; Santos, A.; Hussain, A.; Janson, A.; Wang, R.; Fane, A.G.; Adham, S. Application of Hollow Fiber Forward Osmosis Membranes for Produced and Process Water Volume Reduction: An Osmotic Concentration Process. Environ. Sci. Technol. 2016, 50, 6044–6052. [Google Scholar] [CrossRef]
- Janson, A.; Katebah, M.; Santos, A.; Minier-Matar, J.; Hussain, A.; Adham, S.; Judd, S. Assessing the Biotreatability of Produced Water from a Qatari Gas Field. Int. Pet. Technol. 2015, 20, 1113–1119. [Google Scholar] [CrossRef]
- Zhao, P.; Gao, B.; Xu, S.; Kong, J.; Ma, D.; Shon, H.K.; Yue, Q.; Liu, P. Polyelectrolyte-promoted forward osmosis process for dye wastewater treatment—Exploring the feasibility of using polyacrylamide as draw solute. Chem. Eng. J. 2015, 264, 32–38. [Google Scholar] [CrossRef]
- Jansona, A.F.; Santosa, A.; Hussaina, A.; Minier-matara, J.; Juddb, S.; Adhama, S. Application of Membrane Bioreactor Technology for Produced Water Treatment. In International Gas Processing Symposium; Elsevier: Doha, Qatar, 2015. [Google Scholar]
- Adham, S.; Hussain, A.; Matar, J.M.; Janson, A.; Gharfeh, S.; Hussain, A.; Matar, J.M.; Janson, A.; Gharfeh, S.; Adham, S. Screening of Advanced Produced Water Treatment Technologies: Overview and Testing Results Screening of Advanced Produced Water Treatment Technologies: Overview and Testing Results. IDA J. Desalin. Water Reuse 2016, 5, 7953. [Google Scholar]
- Hussain, A.; Janson, A.; Adham, S. Treatment of Produced Water from Unconventional Resources by Membrane Distillation. In Proceedings of the Internatioal Petroleum Technoloty Conference, Doha, Qatar, 19–22 January 2014. [Google Scholar]
- Dores, R.; Hussain, A.; Katebah, M.; Adham, S.S. Using Advanced Water Treatment Technologies to Treat Produced Water from The Petroleum Industry. In Proceedings of the SPE International Production and Operations Conference & Exhibition, Doha, Qatar, 14–16 May 2012. [Google Scholar]
- Zsirai, T.; Al-Jaml, A.K.; Qiblawey, H.; Al-Marri, M.; Ahmed, A.; Bach, S.; Watson, S.; Judd, S. Ceramic Membrane Filtration of Produced Water: Impact of Membrane Module. Purif. Technol. 2016, 165, 214–221. [Google Scholar] [CrossRef] [Green Version]
- Zsirai, T.; Qiblawey, H.; Buzatu, P.; Al-Marri, M.; Judd, S.J. Cleaning of Ceramic Membranes for Produced Water Filtration. J. Pet. Sci. Eng. 2018, 166, 283–289. [Google Scholar] [CrossRef] [Green Version]
- Abdallaa, M.; Nasser, M.; Fard, A.K.; Qiblawey, H.; Benamor, A.; Judd, S. Impact of Combined Oil-in-Water Emulsions and Particulate Suspensions on Ceramic Membrane Fouling and Permeability Recovery. Purif. Technol. 2018, 212, 215–222. [Google Scholar] [CrossRef] [Green Version]
- Fard, A.K.; Mckay, G.; Atieh, M.A. Hybrid Separator -Adsorbent Inorganic Membrane for Oil–Water Separation. In Proceedings of the 3rd World Congress on Civil, Structural, and Environmental Engineering (CSEE’18), Budapest, Hungary, 8–10 April 2018; pp. 2–5. [Google Scholar]
- Judd, S.; Qiblawey, H.; Al-Marri, M.; Clarkin, C.; Watson, S.; Ahmed, A.; Bach, S. The Size and Performance of Offshore Produced Water Oil-Removal Technologies for Reinjection. Sep. Purif. Technol. 2014, 134, 241–246. [Google Scholar] [CrossRef]
- Qin, D.; Liu, Z.; Liu, Z.; Bai, H.; Sun, D.D. Superior Antifouling Capability of Hydrogel Forward Osmosis Membrane for Treating Wastewaters with High Concentration of Organic Foulants. Environ. Sci. Technol. 2018, 52, 1421–1428. [Google Scholar] [CrossRef]
- Janson, A.; Santos, A.; Hussain, A.; Global, C.; Judd, S. Biotreatment of Hydrate-Inhibitor Containing Produced Waters at Low PH. SPE J. 2015, 20, 1254–1260. [Google Scholar] [CrossRef]
- Onwusogh, U. Feasibility of Produced Water Treatment and Reuse—Case Study of a GTL. In Proceedings of the Internatioal Petroleum Technoloty Conference, Doha, Qatar, 6–9 December 2015. [Google Scholar]
- Al Redoua, A.; Hamid, S.A.; Limited, D.E. Industrial Application for the Removal of Kinetic Hydrate Inhibitor (KHI) Co-Polymers from Produced Water Streams. In Proceedings of the International Petroleum Technology Conference, Doha, Qatar, 6–9 December 2015. [Google Scholar]
- Moussa, D.T.; El-naas, M.H.; Nasser, M.; Al-Marri, M.J.A. Comprehensive Review of Electrocoagulation for Water Treatment: Potentials and Challenges. J. Environ. Manag. 2016, 186, 24–41. [Google Scholar] [CrossRef]
- Aly, D.T.A. A Novel Electrocoagulation System for Produced Water Treatment. Master’s Thesis, Qatar University, Doha, Qatar, 2018. Available online: http://hdl.handle.net/10576/11221 (accessed on 4 December 2021).
- Alghoul, M. Treatment of Produced Water Using an Enhanced Electrocoagulation Process. Master’s Thesis, Qatar University, Doha, Qatar, 2017. [Google Scholar]
- Abu-dieyeh, M.H.; Shaikh, S.S.; Naemi, F.; AlAlghouti, M.; Youssef, T.A. The Influences of Produced Water Irrigation on Soil Microbial Succession and Turfgrass Grass Establishment in Qatar. In Proceedings of the Qatar Foundation Annual Research Conference, Doha, Qatar, 1 March 2018; Hamad bin Khalifa University Press: Doha, Qatar, 2018. [Google Scholar]
- Das, P.; Abdulquadir, M.; Thaher, M.; Khan, S.; Chaudhary, A.K.; Alghasal, G.; Al-Jabri, H.M.S.J. Microalgal Bioremediation of Petroleum-Derived Low Salinity and Low PH Produced Water. J. Appl. Phycol 2019, 31, 435–444. [Google Scholar] [CrossRef]
- Abdul Hakim, M.A. Potential Application of Micronalgea in Produced Water Treatment. Desalin. Water Treat. 2016, 135, 47–58. Available online: https://qspace.qu.edu.qa:8443/bitstream/handle/10576/5087/443354_pdf_F0D5787A-36CD-11E6-9423-32BB4D662D30.pdf?sequence=1&isAllowed=n (accessed on 4 December 2021). [CrossRef] [Green Version]
- Abdul, M.A.; Al-ghouti, M.A.; Das, P.; Abu-dieyeh, M.; Ahmed, T.A.; Mohammed, H.; Aljabri, S.J. Potential Application of Microalgae in Produced Water Treatment. Desalin. Water Treat 2018, 135, 23146. [Google Scholar] [CrossRef] [Green Version]
- Shaikh, S. Grass Establishment, Weed Populations and Soil Microbial Succession in Turf Grass System as Influenced by Produced Water Irrigation. Master’s Thesis, Qatar University, Doha, Qatar, 2017. [Google Scholar]
- Atia, F.A.; Al-Ghouti, M.A.; Al-Naimi, F.; Abu-Dieyeh, M.; Ahmed, T.; Al-Meer, S.H. Removal of toxic pollutants from produced water by phytoremediation: Applications and mechanistic study. J. Water Process. Eng. 2019, 32, 100990. [Google Scholar] [CrossRef]
- Al-Kaabi, M.A.J.I. Enhancing Produced Water Quality Using Modified Activiated Carbon. Master’s Thesis, Qatar University, Doha, Qatar, 2016. [Google Scholar]
- Al-Kaabi, M.; Ghazi, A.B.; Qunnaby, R.; Dawwas, F.; Al-Hadrami, H.; Khatir, Z.; Yousif, M.; Ahmed, T. Enhancing the Quality of “Produced Water” by Activated Carbon. Qatar Found. Annu. Res. Conf. Proc. 2016, 22, 16–17. [Google Scholar]
- Almarouf, H.M.N.; Al-Marri, M.J.; Khraisheh, M.; Onaizi, S.A. Demulsification of Stable Emulsions From Produced Water Using A Phase Separator With Inclined Parallel Arc Coalescing Plates. J. Pet. Sci. Eng. 2015, 135, 16–21. [Google Scholar] [CrossRef]
- Benamor, A.; Talkhan, A.G.; Nasser, M.; Hussein, I.; Okonkwo, P.C. Effect of Temperature and Fluid Speed on the Corrosion Behavior of Carbon Steel Pipeline in Qatari Oilfield Produced Water. J. Electroanal. Chem. 2018, 808, 218–227. [Google Scholar] [CrossRef]
- Shehzad, F.; Hussein, I.A.; Kamal, M.S.; Ahmad, W.; Sultan, A.S.; Nasser, M.S.; Shehzad, F.; Hussein, I.A.; Kamal, M.S.; Ahmad, W. Polymeric Surfactants and Emerging Alternatives Used in the Demulsification of Produced Water: A Review Polymeric Surfactants and Emerging Alternatives Used. Polym. Rev. 2017, 58, 63–101. [Google Scholar]
- Ahan, J.A. Characterization of Produced Water from Two Offshore Oil Fields in Qatar; Environmental Engineering College of Engineering: Doha, Qatar, 2014; Available online: https://qspace.qu.edu.qa/handle/10576/3287/restricted-resource?bitstreamId=0fb37632-1e3b-4851-a058-80c438f15419 (accessed on 4 December 2021).
- Ma, H.; Sultan, A.S.; Shawabkeh, R.; Fahd, K.; Mustafa, S. Destabilization and Treatment of Produced Water-Oil Emulsions Using Anionic Polyacrylamide with Electrolyate of Aluminum Sulphate and Ferrous Sulphate. In Proceedings of the Abu Dhabi International Petroleum Exhibition & Conference, Abu Dhabi, United Arab Emirates, 7–10 November 2016. [Google Scholar]
- Ray, J.P.; Engelhardt, F.R. Produced Water: Technological/Environmental Issues and Solutions; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1993. [Google Scholar]
- Karanisa, T.; Amato, A.; Richer, R.; Abdul Majid, S.; Skelhorn, C.; Sayadi, S. Agricultural Production in Qatar’s Hot Arid Climate. Sustainability 2021, 13, 4059. [Google Scholar] [CrossRef]
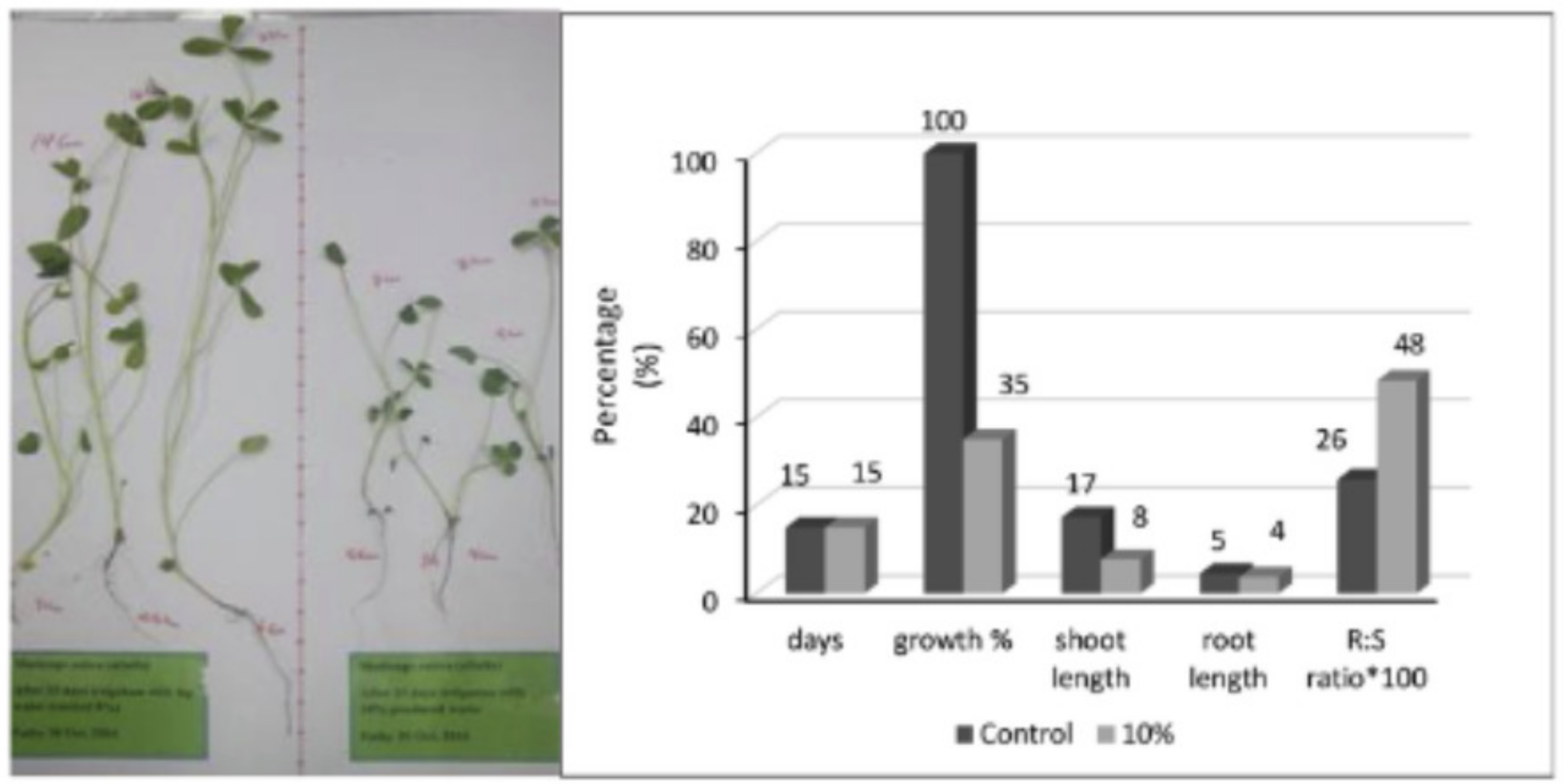
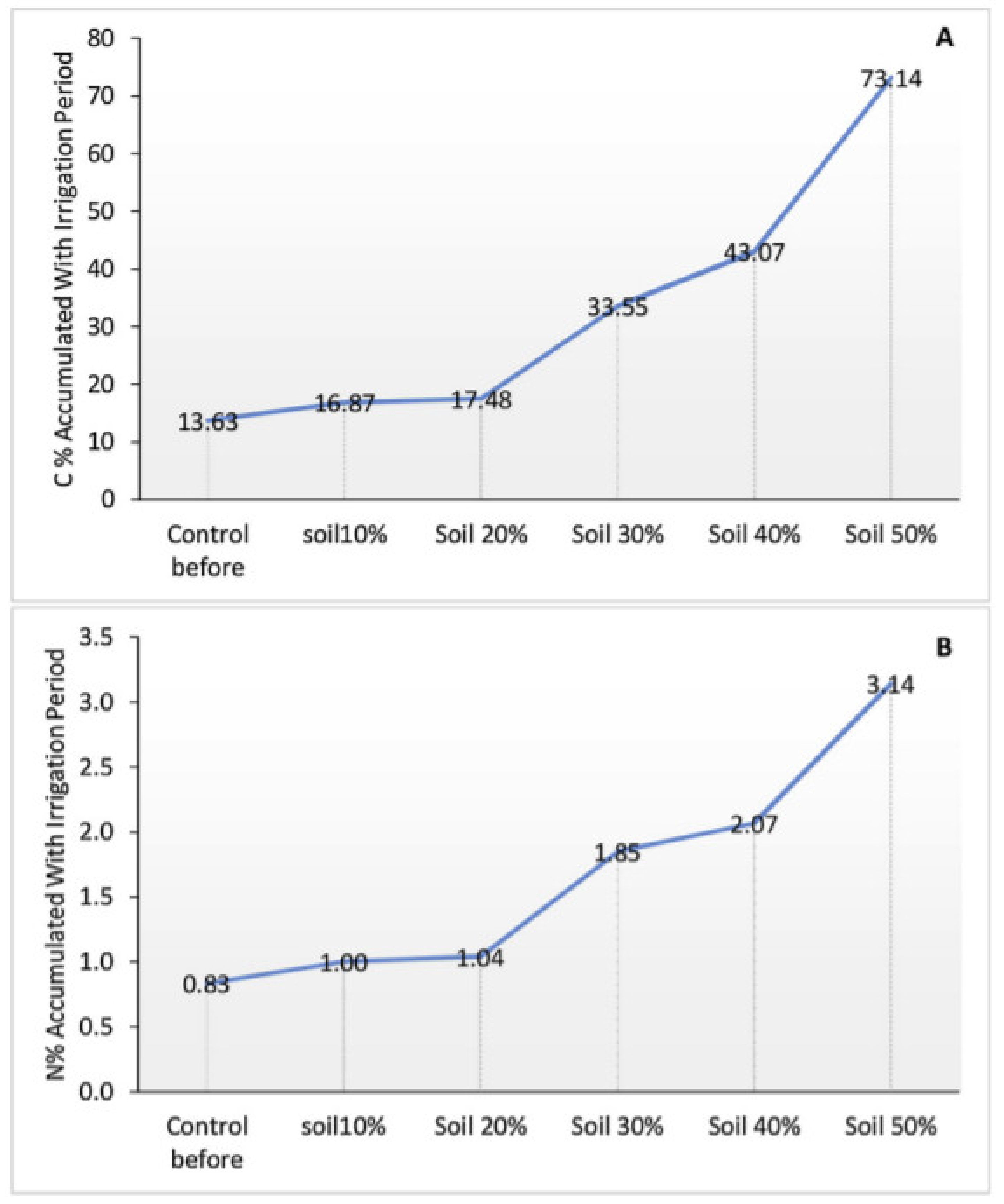
- Echchelh, A.; Hess, T.; Sakrabani, R.; Prigent, S.; Stefanakis, A.I. Towards agro-environmentally sustainable irrigation with treated produced water in hyper-arid environments. Agric. Water Manag. 2021, 243, 106449. [Google Scholar] [CrossRef]
- Hedar, Y. Pollution impact and alternative treatment for produced water. E3S Web Conf. 2018, 31, 03004. [Google Scholar]
- Natural Gas Operation Annual Report. Available online: https://s3.amazonaws.com/rgi-documents/97e32b78209f56efc6d4ec2ee5458fa89c264783.pdf (accessed on 23 October 2021).
- James, L.; Ahmed, N. The Pioneer; The Public Relations Department, Qatar Gas Operating Company Limited: Doha, Qatar, 2011; Available online: https://www.qatargas.com/english/MediaCenter/ThePioneer/ThePioneer-August2011.pdf (accessed on 3 December 2021).
- Plebon, M.J.; Saad, M.A.; Al-Kuwari, N. Application of an Enhanced Produced Water De-Oiling Technology to Middle East Oilfields. In Produced Water—Best Management Practices; TORR Canada Inc.: Montreal, QC, Canada; Qatar Petroleum: Doha, Qatar, 2007. [Google Scholar]
- Khalifa, O.M.; Rais, T.A.M.; El Maadi, A.; Sani, R.A.; Mahrous, E.K.; Al-Marri, B. Coupling Integrated Data Management with Reservoir Surveillance Workflows for Giant Mature Field in Qatar. In Proceedings of the Internatioal Petroleum Technoloty Conference, Doha, Qatar, 6–9 December 2015. [Google Scholar]
- Al-ghouti, M.A.; Al-kaabi, M.A.; Ashfaq, M.Y.; Adel, D. Produced Water Characteristics, Treatment and Reuse: A Review. J. Water Process Eng. 2019, 28, 222–239. [Google Scholar] [CrossRef]
- Yaqub, A.; Isa, M.H.; Ajab, H. Electrochemical degradation of polycyclic aromatic hydrocarbons in synthetic solution and produced water using a Ti/SnO2-Sb2O5-RuO2 anode. J. Environ. Eng. 2015, 141, 04014074. [Google Scholar] [CrossRef]
- Macheca, A.D.; Uwiragiye, B. Application of Nanotechnology in Oil and Gas Industry: Synthesis and Characterization of Organo-modified Bentonite from Boane Deposit and its Application in Produced Water Treatment. Chem. Eng. Trans. 2020, 81, 1081–1086. [Google Scholar]
- McLaughlin, M.C.; Borch, T.; McDevitt, B.; Warner, N.R.; Blotevogel, J. Water quality assessment downstream of oil and gas produced water discharges intended for beneficial reuse in arid regions. Sci. Total Environ. 2020, 713, 136607. [Google Scholar] [CrossRef]
- Konur, O. Characterization and properties of biooils: A review of the research. Biodiesel Fuels 2021, 1, 137–152. [Google Scholar]
- Fillo, J.P.; Koraido, S.M.; Evans, J.M. Sources, characteristics, and management of produced waters from natural gas production and storage operations. In Produced Water; Springer: Boston, MA, USA, 1992; pp. 151–161. [Google Scholar]
- Stephenson, M.T. A survey of produced water studies. In Produced Water; Springer: Boston, MA, USA, 1992; pp. 1–11. [Google Scholar]
- Ahmad, N.A.; Goh, P.S.; Yogarathinam, L.T.; Zulhairun, A.K.; Ismail, A.F. Current advances in membrane technologies for produced water desalination. Desalination 2020, 493, 114643. [Google Scholar] [CrossRef]
- Jacobs, R.P.W.M.; Grant, R.O.H.; Kwant, J.; Marquenie, J.M.; Mentzer, E. The composition of produced water from Shell operated oil and gas production in the North Sea. In Produced Water; Springer: Boston, MA, USA, 1992; pp. 13–21. [Google Scholar]
- Fillo, J.P.; Evans, J.M. Characterization and Management of Produced Waters from Underground Natural Gas Storage Reservoirs. Am. Gas Assoc. Oper. Sect. Proc. 1990, 1, 448–459. [Google Scholar]
- Healy, R.W.; Bartos, T.T.; Rice, C.A.; McKinley, M.P.; Smith, B.D. Groundwater chemistry near an impoundment for produced water, Powder River Basin, Wyoming, USA. J. Hydrol. 2011, 403, 37–48. [Google Scholar] [CrossRef]
- Terrens, G.W.; Tait, R.D. Monitoring Ocean Concentrations of Aromatic Hydrocarbons from Produced Formation Water Discharges to Bass Strait. In Proceedings of the International Conference on Health, Society of Petroleum Engineers Australia, New Orleans, LA, USA, 6 June 1996; pp. 739–747. [Google Scholar]
- Zhao, S.; Minier-Matar, J.; Chou, S.; Wang, R.; Fane, A.G.; Adham, S. Gas field produced/process water treatment using forward osmosis hollow fiber membrane: Membrane fouling and chemical cleaning. Desalination 2017, 402, 143–151. [Google Scholar] [CrossRef]
- Ray, J.P.; Engelhardt, F.R. Produced Water Technological Environmental Issues and Solution. Environ. Sci. Res. 1993, 46, 96–230. [Google Scholar]
- Dyakowski, T.; Kraipech, W.; Nowakowski, A.P.; Williams, R. A Three Dimensional Simulation of Hydrocyclone Behaviour. In Proceedings of the Second International Conference, Sydney, NSW, Australia, 9–11 November 1999; pp. 205–210. [Google Scholar]
- Du, Z.; Ding, P.; Tai, X.; Pan, Z.; Yang, H. Facile preparation of Ag-coated superhydrophobic/superoleophilic mesh for efficient oil/water separation with excellent corrosion resistance. Langmuir 2018, 34, 6922–6929. [Google Scholar] [CrossRef]
- Chen, S.; Lv, C.; Hao, K.; Jin, L.; Xie, Y.; Zhao, W.; Sun, S.; Zhang, X.; Zhao, C. Multifunctional negatively-charged poly (ether sulfone) nanofibrous membrane for water remediation. J. Colloid Interface Sci. 2019, 538, 648–659. [Google Scholar] [CrossRef]
- Wang, K.; Han, D.S.; Yiming, W.; Ahzi, S.; Abdel-Wahab, A.; Liu, Z. A windable and stretchable three-dimensional all-inorganic membrane for efficient oil/water separation. Sci. Rep. 2017, 7, 16081. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z. Efficient and Fast Separation of Emulsified Oil/Water Mixtures with a Novel Micro/Nanofiber Network Membrane. In Proceedings of the Qatar Foundation Annual Research Conference, Doha, Qatar, 19–20 March 2018. [Google Scholar]
- Wang, K.; Yiming, W.; Saththasivam, J.; Liu, Z. A flexible, robust and antifouling asymmetric membrane based on ultra-long ceramic/polymeric fibers for high-efficiency separation of oil/water emulsions. Nanoscale 2017, 9, 9018–9025. [Google Scholar] [CrossRef]
- Lee, S.; Boo, C.; Elimelech, M.; Hong, S. Comparison of fouling behavior in forward osmosis (FO) and reverse osmosis (RO). J. Membr. Sci. 2010, 365, 34–39. [Google Scholar] [CrossRef]
- Chen, S.; Li, L.; Zhao, C.; Zheng, J. Surface hydration: Principles and applications toward low-fouling/nonfouling biomaterials. Polymer 2010, 51, 5283–5293. [Google Scholar] [CrossRef] [Green Version]
- Qin, D.; Liu, Z.; Bai, H.; Sun, D.D. Three-dimensional architecture constructed from a graphene oxide nanosheet–polymer composite for high-flux forward osmosis membranes. J. Mater. Chem. A 2017, 5, 12183–12192. [Google Scholar] [CrossRef]
- Minier-Matar, J.; Hussain, A.; Santos, A.; Janson, A.; Wang, R.; Fane, A.G.; Adham, S. Advances in Application of Forward Osmosis Technology for Volume Reduction of Produced/Process Water from Gas-Field Operations. In Proceedings of the International Petroleum Technology Conference, Doha, Qatar, 6–9 December 2015. [Google Scholar]
- Awad, A.M.; Jalab, R.; Nasser, M.S.; El-Naas, M.; Hussein, I.A.; Minier-Matar, J.; Adham, S. Evaluation of cellulose triacetate hollow fiber membrane for volume reduction of real industrial effluents through an osmotic concentration process: A pilot-scale study. Environ. Technol. Innov. 2021, 24, 101873. [Google Scholar] [CrossRef]
- Sosa-Fernandez, P.A.; Post, J.W.; Ramdlan, M.S.; Leermakers, F.A.M.; Bruning, H.; Rijnaarts, H.H.M. Improving the performance of polymer-flooding produced water electrodialysis through the application of pulsed electric field. Desalination 2020, 484, 114424. [Google Scholar] [CrossRef]
- Sosa-Fernandez, P.A.; Post, J.W.; Leermakers, F.A.M.; Rijnaarts, H.H.M.; Bruning, H. Removal of divalent ions from viscous polymer-flooding produced water and seawater via electrodialysis. J. Membr. Sci. 2019, 589, 117251. [Google Scholar] [CrossRef]
- Kumar, P.; Mitra, S. Challenges in Uninterrupted Production and Supply of Gas to Mega LNG Trains. In Proceedings of the Internatioal Petroleum Technology Conference, Doha, Qatar, 20–22 January 2014. [Google Scholar]
- Adham, S.; Gharfeh, S.; Hussain, A.; Minier-Matar, J.; Janson, A. Kinetic Hydrate Inhibitor Removal by Physical, Chemical and Biological Processes. In Proceedings of the Offshore Technology Conference—Asia, Kuala Lumpur, Malaysia, 18 March 2014. [Google Scholar]
- Cobham, A.; Way, H. The treatment of oily water by coalescing. Filtr. Sep. 1992, 29, 295–300. [Google Scholar]
- Al-Shamrani, A.A.; James, A.; Xiao, H. Destabilisation of oil–water emulsions and separation by dissolved air flotation. Water Res. 2002, 36, 1503–1512. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Al-Degs, Y.S.; Khalili, F.I. Minimisation of organosulphur compounds by activated carbon from commercial diesel fuel: Mechanistic study. Chem. Eng. J. 2010, 162, 669–676. [Google Scholar] [CrossRef]
- Al-Kaabi, M.A.; Al-Ghouti, M.A.; Ashfaq, M.Y.; Ahmed, T.; Zouari, N. An integrated approach for produced water treatment using microemulsions modified activated carbon. J. Water Process. Eng. 2019, 31, 100830. [Google Scholar] [CrossRef]
- Upadhyayula, V.K.; Deng, S.; Mitchell, M.C.; Smith, G.B. Application of carbon nanotube technology for removal of contaminants in drinking water: A review. Sci. Total Environ. 2009, 408, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Ali, K.; Kwon, K.R.; Hyun, M.T.; Choi, K.H. Electrohydrodynamic atomization approach to graphene/zinc oxide film fabrication for application in electronic devices. J. Mater. Sci. Mater. Electron. 2014, 25, 1097–1104. [Google Scholar] [CrossRef]
- Fard, A.K.; Rhadfi, T.; Mckay, G.; Al-marri, M.; Abdala, A.; Hilal, N.; Hussien, M.A. Enhancing oil removal from water using ferric oxide nanoparticles doped carbon nanotubes adsorbents. Chem. Eng. J. 2016, 293, 90–101. [Google Scholar] [CrossRef] [Green Version]
- Rasool, K.; Pandey, R.P.; Rasheed, P.A.; Buczek, S.; Gogotsi, Y.; Mahmoud, K.A. Water treatment and environmental remediation applications of two-dimensional metal carbides (MXenes). Mater. Today 2019, 30, 80–102. [Google Scholar] [CrossRef]
- Ihsanullah, I. MXenes (two-dimensional metal carbides) as emerging nanomaterials for water purification: Progress, challenges and prospects. Chem. Eng. J. 2020, 388, 124340. [Google Scholar] [CrossRef]
- Fard, A.K.; Mckay, G.; Chamoun, R.; Rhadfi, T.; Preud’Homme, H.; Atieh, M.A. Barium removal from synthetic natural and produced water using MXene as two dimensional (2-D) nanosheet adsorbent. Chem. Eng. J. 2017, 317, 331–342. [Google Scholar] [CrossRef]
- Strømgren, T.; Sørstrøm, S.E.; Schou, L.; Kaarstad, I.; Aunaas, T.; Brakstad, O.G.; Johansen, O. Acute toxic effects of produced water in relation to chemical composition and dispersion. Mar. Environ. Res. 1995, 40, 147–169. [Google Scholar] [CrossRef]
- Das, P.; Thaher, M.I.; Hakim, M.A.Q.M.A.; Al-Jabri, H.M.S.; Alghasal, G.S.H. A comparative study of the growth of Tetraselmis sp. in large scale fixed depth and decreasing depth raceway ponds. Bioresour. Technol. 2016, 216, 114–120. [Google Scholar] [CrossRef]
- Fakhru’l-Razi, A.; Pendashteh, A.; Abdullah, L.C.; Biak, D.R.A.; Madaeni, S.S.; Abidin, Z.Z. Review of technologies for oil and gas produced water treatment. J. Hazard. Mater. 2009, 170, 530–551. [Google Scholar] [CrossRef]

| Treatment Technologies | Advantages | Disadvantages |
|---|---|---|
| Membrane separation |
|
|
| Combined fiber coalescence |
|
|
| Tubular separation |
|
|
| Media filtration |
|
|
| Hydrocyclone |
|
|
| Gravity and enhanced gravity sedimentation |
|
|
| Technology | Commercial System | Treatments | Water Recovery | Reference |
|---|---|---|---|---|
| Reverse Osmosis | CDM Technology | Combination of 3 major processes such as ion exchange process, RO, and evaporation | 50–90% | [40] |
| Reverse Osmosis | Veolia:OPUSTM | Acidification, Degasification and followed by MultifloTM chemical softening and Reverse Osmosis | Higher than 90% | [41] |
| Reverse Osmosis | Eco-sphere: OzonixTM | Activated carbon cartridge filtration and RO | 75% | [40] |
| Reverse Osmosis | GeoPure water technologies | Combination of pretreatment, UF and RO | 50% | [40] |
| Ion-Exchange (IX) based processes | EMIT: Higgins Loop | Continuous counter current ion exchange contactor for liquid phase separations of ionic components. | 99% | [37] |
| Ion-Exchange (IX) based processes | Drake: Continuous selective IX process | 3-phase, continuous fluidized bed system | 97% | [40] |
| Ion-Exchange (IX) based processes | Eco-Tech: Recoflo® compressed-bed IX process | Extension of standardpacked bed IX processes | - | [40] |
| Sl. No. | Treatments Used | Methods Used | Merits/Demerits | Main Characterization | PW Characteristics/Target Contaminants Removed | Significance of the Study | References |
|---|---|---|---|---|---|---|---|
| 1 | Membrane processes (UF, NF, and RO), thermal evaporations and advanced oxidation process | Membrane-based | - | TDS, COD, KHI, salinity, conductivity | Monovalent and divalent ions (Calcium, magnesium, potassium) | PW use in irrigation | [49] |
| 2 | Forward osmosis | Membrane-based | FO offers ecofriendly dilution before discharge. | pH, TOC, TC, inorganic carbon, water flux, anion and cations, ions, metals, salinity, organic and inorganic contents | Inorganic carbons, Monovalent and divalent ions | Efficiency of FO process | [50] |
| 3 | Hollow Fiber Forward Osmosis Membranes | Membrane-based | FO process leads to environmentally friendly dilution before discharge | Conductivity, pH, anion and cation, TDS, TN, TOC, IC, and osmotic pressure | Chloride, sodium, calcium, magnesium, bromide, sulfate, potassium, phosphate, TOC, TN | Efficiency of FO process | [51] |
| 4 | Membrane bioreactors (MBRs) | Membrane-based | Economical and good separation performance | COD, conductivity, pH TSS, VSS, TN, TPH, TOG, DO, and TOC | Organic carbons, potassium, ammonium, phosphate | Efficiency of MBRs | [52] |
| 5 | Forward osmosis membranes sing thin-film composite FO hollow fiber membranes | Membrane-based | FO process leads to environmentally friendly dilution before discharge | TDS, TOC, inorganic carbon, conductivity, alkalinity, turbidity, pH | Organic carbons | PW use in irrigation | [53] |
| 6 | MBRs | Membrane-based | Economical and good separation performance | COD, TOC, TN, anion and cation, oil and grease, TPH, thiosulfate, conductivity | Anion and cation, oil and grease, TPH, thiosulfate | Efficiency of MBRs | [54] |
| 7 | Membrane processes, membrane bioreactors, membrane distillation and ozonation | Membrane-based | Economical and good separation performance | TDS, COD, KHI, salinity, conductivity, ions, and cations | Cations, TDS | Efficiency of MB process, MD, ozonation, and membrane bioreactors | [55] |
| 8 | Membrane distillation | Membrane-based | TDS, DOC, TOC, COD, conductivity, phenol, oil and grease | Grease, oil, phenol | MD efficiency for PW treatment | [56] | |
| 9 | Direct membrane filtration, biological such as MBRs, advanced oxidation processes (AOPs) and thermal evaporators | Membrane-based | Economical and good separation performance | TDS, DOC, TOC, COD, conductivity, phenol, oil and grease | Oil, grease, phenol | Efficiency of MBRs and AOPs in PW treatment | [57] |
| 10 | Crossflow multi-channel ceramic membrane (TiO2 and SiC) | Membrane-based | Permits produced water re-Injection even in difficult reservoirs with no loss in injectivity | pH, conductivity, sulphide, TS, TOC, hardness, iron, O and G | Iron, oil, grease | Membrane process effectiveness in PW treatment | [58] |
| 11 | Chemical cleaning in place (CIP) between ceramic membrane | Membrane-based | pH, calcium, barium, and iron alkaline, oil and inorganic reagents, turbidity and oil and grease | Oil, grease, calcium, barium, inorganic reagents | Ceramic microfiltration membrane | [59] | |
| 12 | Ceramic membrane | Membrane-based | Permits produced water re-Injection even in difficult reservoirs wit no loss in injectivity | Particulate solids, Feed and permeate oil concentration, TOC, COD, turbidity | Organic carbons and suspended solids | Crossflow ceramic microfiltration (CFCMF) to the removal of emulsified oil produced water | [60] |
| 13 | Hybrid Separator-Adsorbent Inorganic Membrane (Al2O3 and AC) | Membrane-based | Salinity, oil removal efficiency, water flux | Oil removal, Salt removal | Inorganic Membrane for PW management | [61] | |
| 14 | Crossflow membrane filtration (CMF), media (nutshell) filtration (NSF induced gas flotation (IGF), and Hydrocyclones (HCs) | Membrane-based | Efficiency of removing the suspended matters- | Suspended matters removal | application of an effective chemical clean | [62] | |
| 15 | Hydrogel Forward Osmosis Membrane | Membrane-based | FO process leads to environmentally friendly dilution before discharge | TOC | Organic carbons, oil and grease | Efficiency of FO process for PW treatment | [63] |
| 16 | Biotreatment of Hydrate-Inhibitor with activated sludge process | Membrane-based | Most economical approach for organics removal | Ammonium, phosphate, potassium, COD, conductivity, pH TSS, VSS, TN, TPH, TOG, DO and TOC | Organic carbons, Ammonium, phosphate, potassium | PW use in irrigation | [64] |
| 17 | Flocculation flotation unit, biotreatment, membrane filtration (UF, RO units), evaporation and crystallization processes | Membrane-based | Permits produced water re-Injection even in difficult reservoirs with no loss in injectivity | pH, conductivity, TOC, ion chromatography, metal, COD, TDS, Ca, Mg, Ba and heavy metal | Heavy metals, organic carbons, dissolved solids, divalent ions | Cooling and power generation among different uses | [65] |
| 18 | Coagulation, dissolved air flotation and Evaporation technology | Coagulation | Energy saving process | COD, KHI, ions and cations, Total hardness, TKN, TOC, O and G, TSS, Cl, TDS | Grease, oil, organic carbons | Removal of KHI co-polymers application | [66] |
| 19 | Electrocoagulation | Coagulation | Highly efficient and energy saving process | Ammonium, phosphate, potassium, COD, conductivity, cations and anions, ionic-liquid, pH TSS, VSS, TN, TPH, TOG, DO and TOC | Phosphate, potassium, organic carbons | Efficiency of electrocoaguation for PW treatment | [67] |
| 20 | Electrocoagulation | Coagulation | Energy saving process | COD, TOC, TPH, O and G and sludge | Oil, grease | - | [68] |
| 21 | Electrocoagulation and steel slag | Coagulation | Energy saving process | Oil and grease removal, turbidity, TSS | Suspended solids, oil and grease | Efficiency of electrocoagulation and steel slag for PW treatment | [69] |
| 22 | - | Biological treatment | Produces huge amount of biomass that could be employed as feedstock for many products | Bacterial colony-forming units (CFU) | Chloride, sulfate, bromide, sodium, magnesium, calcium, and potassium, strontium and boron | 1—Irrigation of 2 turfgrass species, Paspalum sp. and Cynodon dactylon. 2—Studying the impact of PW irrigation on established grasses, heavy metal accumulation, microbial succession, and germination tests for weeds and turf grass seeds | [70] |
| 23 | Microalgae strains | Biological treatment | Most economical approach for organics removal. | Salinity, pH, TOC, TN, TP | Salts, phosphorus | Use of biomass as feedstock | [71] |
| 24 | Microscopic microalgae; screening, 5 species of microalgae strains Dictyosphaerium, Scenedesmus, Chlorella, Monoraphidium, Neochloris. | Biological treatment | Most economical approach for organics removal. | TP, BTEX, Fe, Al, TOC, TN, COD, TKN, turbidity, salinity, pH, and ammonium | Organic carbons, phosphorus, salts, ammonium | Application of microalgae for PW treatment | [72] |
| 25 | Five microalgae strains used for water treatment: Monoraphidium, Chlorella, Neochloris, Scenedesmus, Dictyosphaerium, Chlorella and Dictyosphaerium species | Biological treatment | Most economical approach for organics removal. | Organic carbon, nitrogen removal and phosphorus and various metals, removal efficiencies, TOC and BTEX | Nitrogen, metals, organic carbons, phosphorus, salts, ammonium | Application of microalgae for PW treatment | [73] |
| 26 | - | Biological treatment | Most economical approach for organics removal. | Salinity, bacterial and fungal CFUs | Salts and microorganisms | Irrigation of turf grass—Paspalum sp. and Cynodon dactylon | [74] |
| 27 | - | TDS, boron, sodium, chloride ions, sodium adsorption, and organic contents | Organic compounds, boron, and salt | Plant irrigation in greenhouse for Salsola baryosma, Phramites australis Sorghum bicolor, Medicago sativa, Helianthus annus and Zea mays | [75] | ||
| 28 | Sand filtration activated carbon filtration (ACF) as well as modified activated carbon filtration. | Activated Carbon filtration | Increased removal of COD | Cations, metals, inorganic anions, BTEX, phenolic, organic acids, oil and grease, sulfides, hardness, alkalinity, conductivity, BOD, TOC, COD, and pH | Phenolic, organic acids, oil and grease, sulfides, cations, metals, inorganic anions, BTEX | - | [76] |
| 29 | Activated carbon filtration and microemulsions modified AC | Activated Carbon filtration | Increased removal of COD | Heavy metals, salts, toxic organic components, and TDS, BTEX, pH, COD, TOC, TN, TDS, conductivity, alkalinity, hardness and sever all metals | Toxic organic components, heavy metals, salts, dissolved solids | Irrigation application of PW | [77] |
| 30 | Series of inclined multiple arc coalescence plates | Coalescing | - | Salinity, oil removal | Oil | Removal of stable oil emulsions from PW | [78] |
| 31 | Electrochemical methods | Chemical method | Highest TPH and COD removal efficiency | Corrosion study and scaling study | Examine the impact of PW from the Ras Laffan (North Oilfield) Qatar on corrosion as well as scaling of carbon steel. | [79] | |
| 32 | Chemical demulsification | Chemical method | - | Cations and anions, ionic liquid | Anions and cations | Efficacy of chemical demulsification for PW treatment | [80] |
| 33 | Site 1: 2 phase separation tanks combined with filtration unit as well as chemical injection, and finally the large gravitational separation tanks. Site 2: begins with 2-stage separation with chemical injection, 2 phases succeeded by 3 phase separation tanks combined with hydrocyclone succeeded by surge drum. | Chemical method | - | Total sulfides, dissolved CO2, concentration of ions, phosphates, ammonia nitrogen, concentration of total Kjeldahl nitrogen, concentration of metals, total dissolved solids, total suspended solids, biodegradable COD, total COD, Phenol concentration, BTEX concentration, total amount of hydrocarbons, conductivity, pH, and oil droplet size distribution | Phosphates, ammonia nitrogen, sulfides | - | [81] |
| 34 | Anionic polyacrylamide (PAMs) with electrolyte of aluminum sulphate and ferrous sulphate | Chemical method | - | Turbidity, viscosity, and COD | - | [82] |
| Different Parameters | PW Characteristics | |
|---|---|---|
| Filtered Water | Raw PW | |
| Xylene (mg/L) | 3.11 | 3.43 |
| Ethyl benzene (mg/L) | 1.05 | 1.22 |
| Toluene (mg/L) | 3.21 | 3.8 |
| Benzene (mg/L) | 16.1 | 21 |
| Total phosphorus (µg/L) | 180 | 277.78 |
| Total Nitrogen (mg/L) | 27.6 | 35.77 |
| Total organic carbon (mg/L) | 317 | 389.1 |
| Parameter | Concentration (mg/L) |
|---|---|
| Total dissolved solid | 1000–400,000 |
| Total suspended solid | 98–116 |
| Potassium | 10–12 |
| Sodium | 5462–5836 |
| Chlorine | 8475–9219 |
| Total organic carbon | 45–71 |
| Magnesium | 114–118 |
| Calcium | 356–372 |
| Sulfate radical | 61–68 |
| Total nitrogen | 23–26 |
| Title | Authors/Date/Reference | Parameters | Unit | Value | Parameters | Value |
|---|---|---|---|---|---|---|
| Advanced Technologies for Produced water treatment | Hussain, A., Minier-Matar, J., et al./2014 [49] | Produced Water Source and Composition | Process Water Source and Composition | |||
| COD | mg/L | 1572 | COD | 397 mg/L | ||
| TOC | mg/L | 491 | TOC | 114 mg/L | ||
| TN | mg/L | 43 | TN | 31 mg/L | ||
| Oil & grease | mg/L | 47 | Oil & grease | 10 mg/L | ||
| TPH | mg/L | 45 | TPH | 9 mg/L | ||
| Chloride | mg/L | 2265 | Chloride | 17 mg/L | ||
| Sodium | mg/L | 1030 | Sodium | 359 mg/L | ||
| Calcium | mg/L | 329 | Calcium | 3 mg/L | ||
| Sulfide | mg/L | 307 | Sulfide | 307 mg/L | ||
| Magnesium | mg/L | 61 | Magnesium | 0.2 mg/L | ||
| Bromide | mg/L | 51 | Bromide | <0.5 mg/L | ||
| Sulfate | mg/L | 54 | Sulfate | 9 mg/L | ||
| Potassium | mg/L | 44 | Potassium | 1.5 mg/L | ||
| Thiosulfate | mg/L | 14 | Thiosulfate | 43 mg/L | ||
| Acetate | mg/L | 347 | Acetate | 3.2 mg/L | ||
| Ammonium | mg/L | 11 | Ammonium | 11 mg/L | ||
| Conductivity | µS/cm | 7200 | Conductivity | 1761 µS/cm | ||
| TDS | mg/L | 5189 | TDS | 1491 mg/L | ||
| Produced and process water (PPW) | ||||||
| Application of forward osmosis for reducing volume of produced/Process water from oil and gas operations Gas field produced/process water treatment using forward osmosis hollow fiber membrane: Membrane fouling and chemical cleaning | Minier-Matar, J., et al./2015 [50] Zhao, S., Minier-Matar, J., and et al./2017 [103] | TOC | mg/L | 33 | ||
| Chloride | mg/L | 286 | ||||
| Sodium | mg/L | 329 | ||||
| Calcium | mg/L | 38 | ||||
| Sulfate | mg/L | 349 | ||||
| Magnesium | mg/L | 8.7 | ||||
| Bromide | mg/L | 5.6 | ||||
| Potassium | mg/L | 4.7 | ||||
| Ammonium | mg/L | 8.5 | ||||
| Alkalinity | mg/L | 223 | ||||
| PH | 8 | |||||
| Conductivity | µS/cm | 1810 | ||||
| TDS | mg/L | 1526 | ||||
| Turbidity | NTU | 32 | ||||
| Produced and process water (PPW) | ||||||
| Application of Hollow Fiber Forward Osmosis Membranes for Produced and Process Water Volume Reduction: An Osmotic Concentration Process | Minier-Matar, J., Santos, A., et al./2016 [51] | TOC | mg/L | 120 | ||
| Chloride | mg/L | 284 | ||||
| Sodium | mg/L | 345 | ||||
| Calcium | mg/L | 38 | ||||
| Sulfate | mg/L | 347 | ||||
| Magnesium | mg/L | 8 | ||||
| Bromide | mg/L | 5 | ||||
| Potassium | mg/L | 4.5 | ||||
| Phosphate | mg/L | <0.1 | ||||
| Total nitrogen | mg/L | 28 | ||||
| Inorganic carbon | mg/L | 31 | ||||
| PH | 8 | |||||
| Conductivity | µS/cm | 1725 | ||||
| TDS | mg/L | 1550 | ||||
| Osmotic pressure (25°) | bar | 1 | ||||
| Produced water | ||||||
| Assessing the Biotreatability of Produced Water from a Qatari Gas Field | Janson, A., et al./2015 [52] | COD | mg/l | 1572 | ||
| TOC | mg/L | 491 | ||||
| TN | mg/L | 34 | ||||
| Oil & grease | mg/L | 47 | ||||
| TPH | mg/L | 45 | ||||
| Chloride | mg/L | 2265 | ||||
| Sodium | mg/L | 1030 | ||||
| Calcium | mg/L | 329 | ||||
| Sulfide | mg/L | 828 | ||||
| Magnesium | mg/L | 61 | ||||
| Bromide | mg/L | 51 | ||||
| Sulfate | mg/L | 54 | ||||
| Potassium | mg/L | 44 | ||||
| Thiosulfate | mg/L | 14 | ||||
| Acetate | mg/L | 347 | ||||
| Ammonium | mg/L | 11 | ||||
| Conductivity | µS/cm | 7200 | ||||
| Total Dissolved solids | mg/L | 5189 | ||||
| PH | 4.3 | |||||
| Trace Metals | Filtered Water (ppb) | Feed Water (ppb) | % Removal | Microalgae Species |
|---|---|---|---|---|
| Cd | 0.06 | 0.09 | 97.37 | Chlorella |
| Ni | 3.71 | 7.83 | 92.29 | Dictyosphaerium |
| Cr | 17.2 | 24.09 | 19.36 | Dictyosphaerium sp. |
| Fe | 100.19 | 287.94 | 100 | Neochloris sp.; Chlorella sp. |
| Mn | 318.56 | 318.56 | 87.8 | Neochloris sp. |
| Sr | 105.73 × 102 | 111.98 × 102 | 21.23 | Dictyosphaerium sp. |
| K | 677.40 × 102 | 736.18 × 102 | 11.27 | Scenedesmus sp. |
| Ba | 43.35 | 55.69 | 13.06 | Monoraphidium sp. |
| V | 1.46 | 1.87 | 36.26 | Scenedesmus |
| Al | 13.68 | 114.41 | 100 | Neochloris sp. |
| Mg | 392.57 × 102 | 417.15 × 102 | 13.9 | Dictyosphaerium sp. |
| Cu | 180.78 | 224.97 | 91.65 | Dictyosphaerium sp. |
| B | 374.7 × 102 | 425.9 × 102 | 20.23 | Dictyosphaerium sp. |
| Parameters | Mean Values | Parameters | Mean Values |
|---|---|---|---|
| Major parameters | Metals | ||
| TSS (ppm) | 21.34 ± 3.51 | Strontium (ppb) | 13,181 ± 114 |
| pH | 4.43 ± 0.01 | Vanadium (ppb) | 2.55 ± 0.04 |
| Conductivity | 7035 ± 56 | Sodium (ppb) | 1,198,167 ± 16,526 |
| COD (ppm) | 10,496 ± 162 | Zinc (ppb) | 4.97 ± 0.28 |
| Salinity (ppt) | 4502 ± 36 | ||
| BOD (ppm) | 1034 ± 42 | Other pollutants | |
| TOC (ppm) | 2405 ± 16 | Propionate (ppm) | 17.37 ± 1.04 |
| BTEX | % KHI | 0.27 ± 0.05 | |
| Ethyl benzene (ppb) | 4648 ± 688 | Phenol (ppm) | 1.96 ± 0.07 |
| Xylene (ppb) | 1156 ± 88 | HEM (ppm) | 40.54 ± 4.20 |
| Benzene (ppb) | 11,170 ± 4298 | Formate (ppm) | 0.35 ± 0.04 |
| Toluene (ppb) | 278.2 ± 14.3 | Corrosion Inhibitor (ppm) | 623.3 ± 15.5 |
| Metals | Acetate (ppm) | 368.7 ± 4.04 | |
| Potassium (ppb) | 100,922 ± 122 | TN (ppm) | 47.41 ± 0.25 |
| Nickel (ppb) | 7.08 ± 0.28 | % MEG | 0.33 ± 0.07 |
| Molybdenum (ppb) | 5.52 ± 0.02 | Other Ions | |
| Manganese (ppb) | 258.3 ± 2.7 | Sulfide (ppm) | 326.3 ± 21.1 |
| Iron (ppb) | 4144 ± 114 | Sulphate (ppm) | 46.13 ± 0.19 |
| Aluminum (ppb) | 10.28 ± 6.75 | Silica (ppm) | 2.0 ± 0.1 |
| Arsenic (ppb) | 7.24 ± 1.89 | Phosphate (ppm) | 2.06 ± 0.08 |
| Chromium (ppb) | 30.31 ± 0.37 | Magnesium (ppb) | 45,064 ± 1223 |
| Barium (ppb) | 60.51 ± 0.45 | Chloride (ppm) | 2921 ± 10 |
| Cobalt (ppb) | 7.04 ± 0.70 | Boron (ppb) | 5744 ± 95 |
| Copper (ppb) | 0.62 ± 0.05 | Calcium (ppb) | 285,565 ± 2205 |
| Cadmium (ppb) | 0.05 ± 0.01 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dawoud, H.D.; Saleem, H.; Alnuaimi, N.A.; Zaidi, S.J. Characterization and Treatment Technologies Applied for Produced Water in Qatar. Water 2021, 13, 3573. https://doi.org/10.3390/w13243573
Dawoud HD, Saleem H, Alnuaimi NA, Zaidi SJ. Characterization and Treatment Technologies Applied for Produced Water in Qatar. Water. 2021; 13(24):3573. https://doi.org/10.3390/w13243573
Chicago/Turabian StyleDawoud, Hana D., Haleema Saleem, Nasser Abdullah Alnuaimi, and Syed Javaid Zaidi. 2021. "Characterization and Treatment Technologies Applied for Produced Water in Qatar" Water 13, no. 24: 3573. https://doi.org/10.3390/w13243573
APA StyleDawoud, H. D., Saleem, H., Alnuaimi, N. A., & Zaidi, S. J. (2021). Characterization and Treatment Technologies Applied for Produced Water in Qatar. Water, 13(24), 3573. https://doi.org/10.3390/w13243573






