The Effect of Exogenous Oxytetracycline on High-Temperature Anaerobic Digestion of Elements in Swine Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Swine Wastewater and Granular Sludge
2.2. Experimental Design
2.3. Analysis Instruments and Methods
2.3.1. Instruments
2.3.2. Solid Phase Extraction
2.3.3. Liquid Chromatography Conditions
2.3.4. Mass Spectrometry Conditions
2.3.5. Standard Curve
2.3.6. Microbial Community Analysis
3. Results and Discussion
3.1. Kinetic Degradation Model
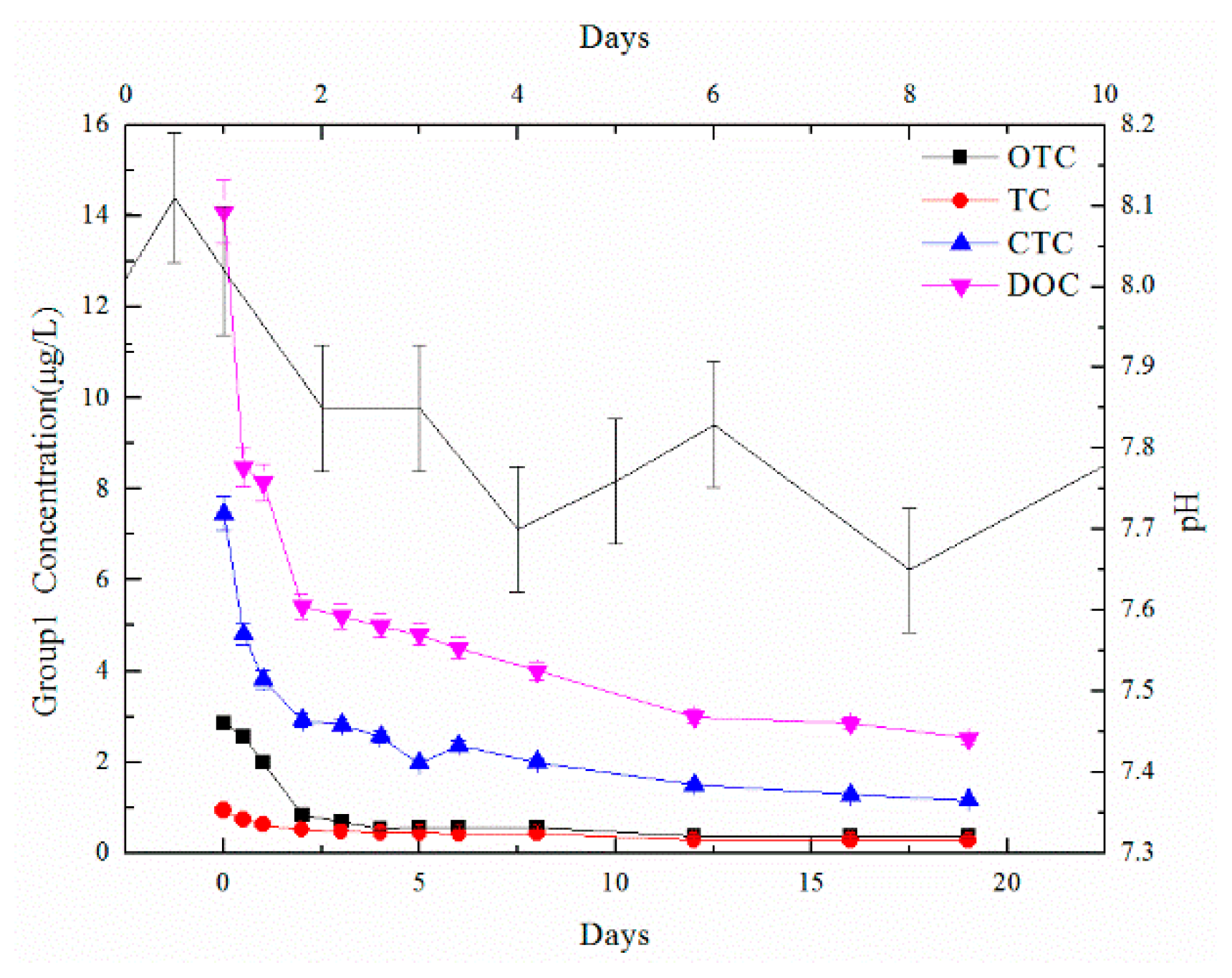
3.2. Degradation Results of TCs
3.3. The Effect of Exogenous OTC in Different Time Periods
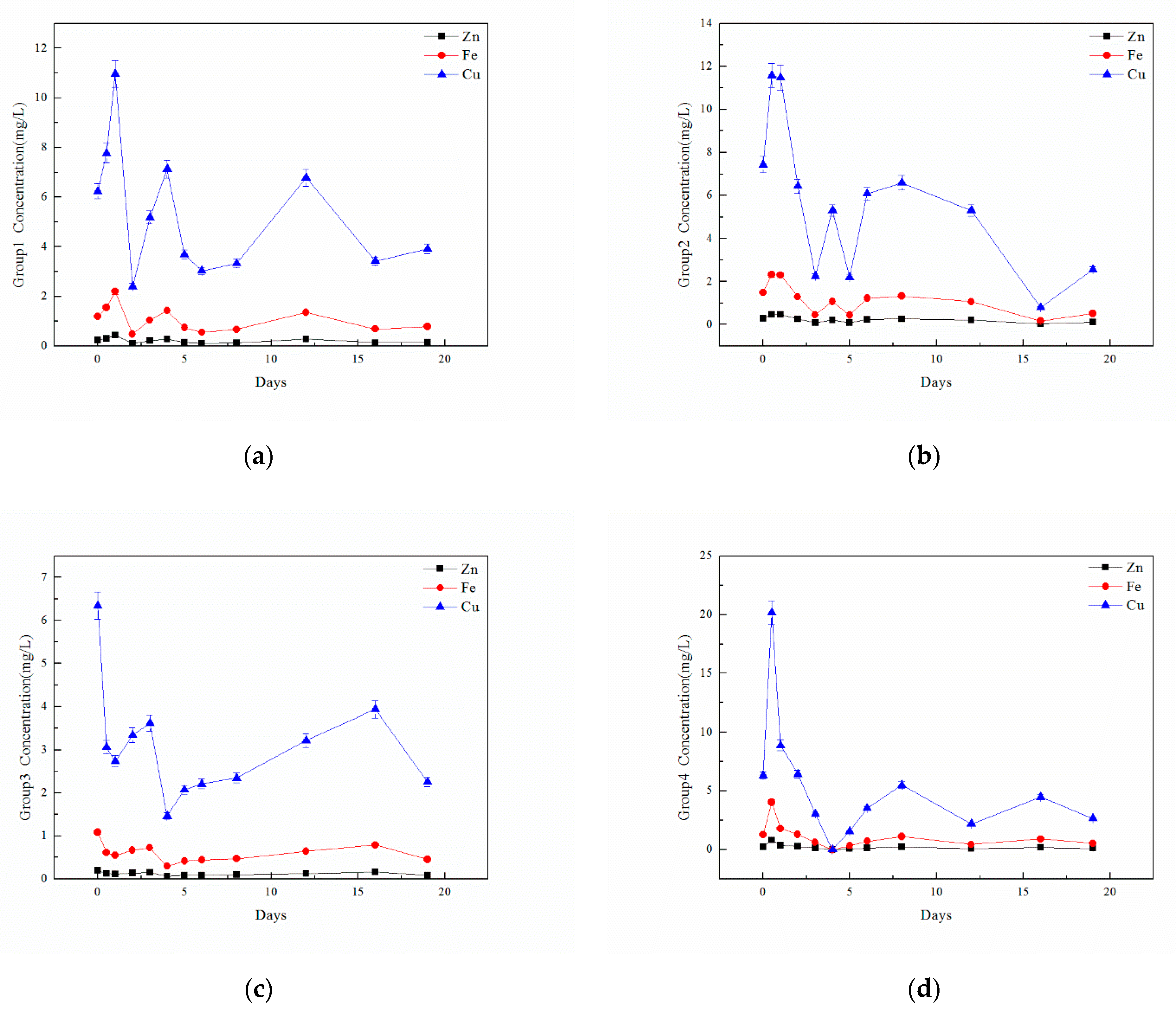
3.4. The Effect of OTC Addition on Heavy Metals
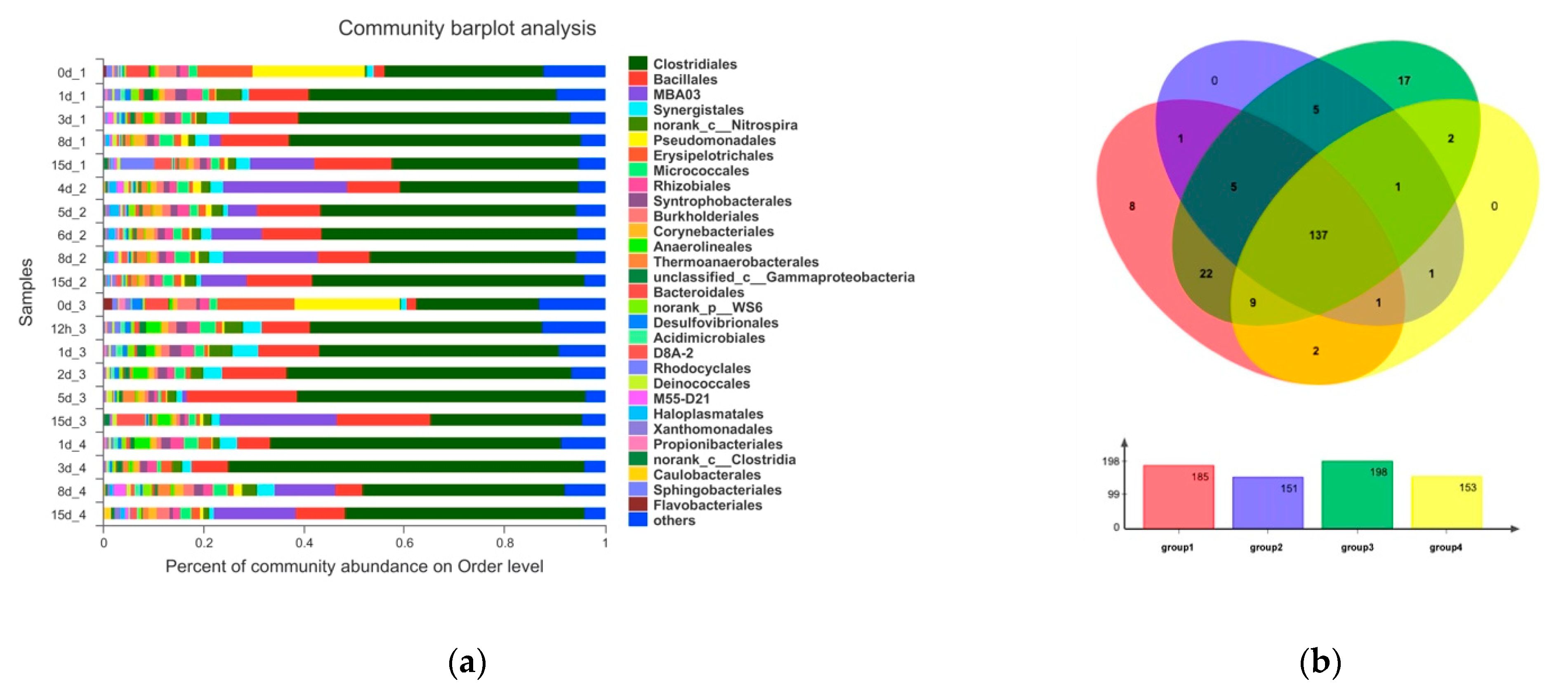
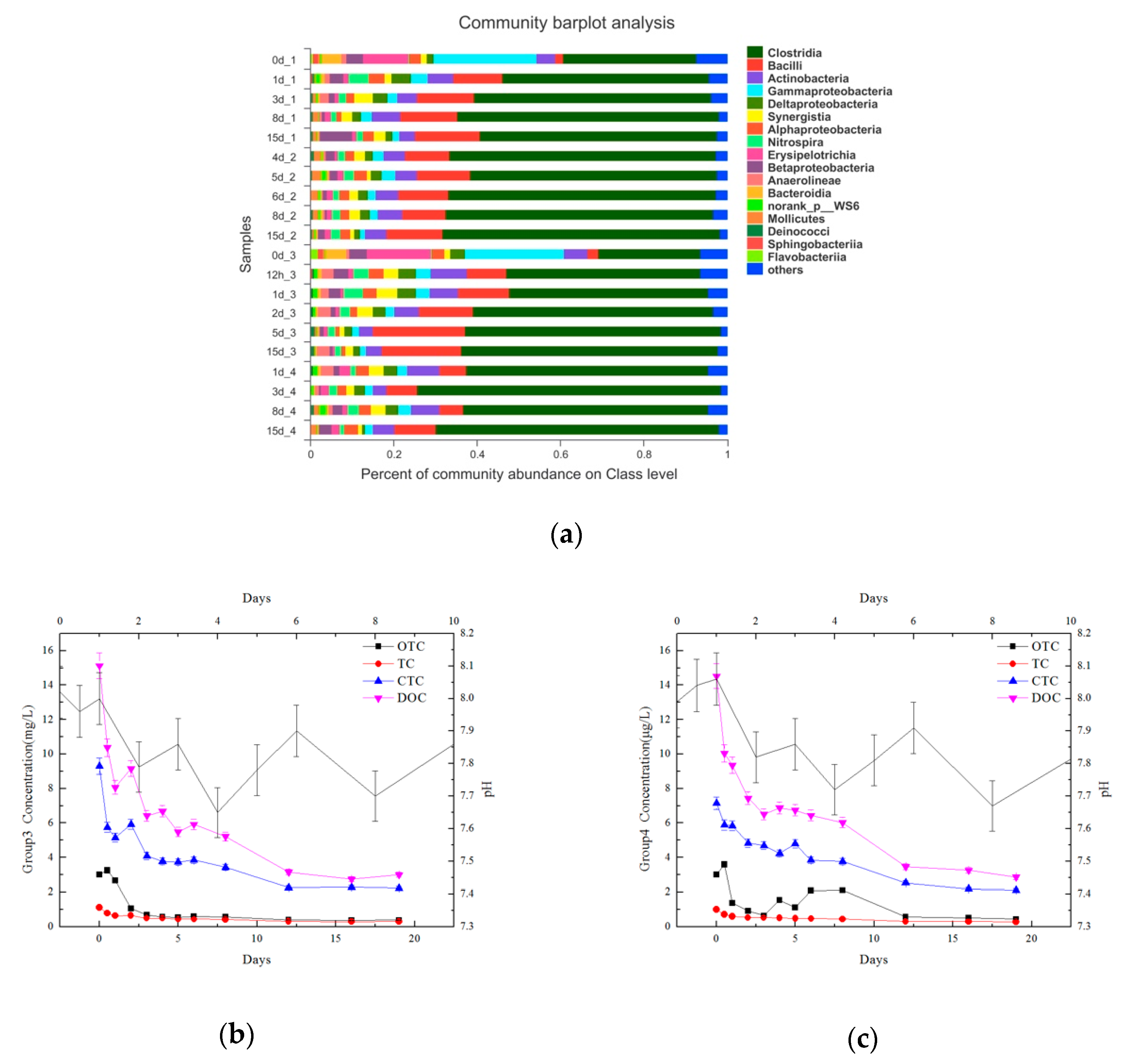
3.5. The Effects of Adding OTC on Microbial Community
3.6. Interaction with TCs
3.6.1. Relationship between TCs, Heavy Metals and Microorganisms
3.6.2. Relationship between TCs, pH, and Microorganisms
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tsai, W.T. Regulatory Promotion and Benefit Analysis of Biogas-Power and Biogas-Digestate from Anaerobic Digestion in Taiwan’s Livestock Industry. Fermentation 2018, 4, 57. [Google Scholar] [CrossRef] [Green Version]
- Song, M.; Liu, L.J.; Wang, Z.; Nanseki, T. Consumers’ Attitudes to Food Traceability System in China-Evidences from the Pork Market in Beijing. J. Fac. Agric. Kyushu Univ. 2008, 53, 569–574. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, X.X.; Zhou, Z.G. Impacts of Small-Scale Industrialized Swine Farming on Local Soil, Water and Crop Qualities in a Hilly Red Soil Region of Subtropical China. Int. J. Environ. Res. Public Health 2017, 14, 1524. [Google Scholar] [CrossRef] [Green Version]
- Kwon, G.; Nam, J.H.; Kim, D.M.; Song, C.; Jahng, D. Growth And Nutrient Removal of Chlorella Vulgaris in Ammonia-Reduced Raw and Anaerobically-Digested Piggery Wastewaters. Environ. Eng. Res. 2020, 25, 135–146. [Google Scholar] [CrossRef]
- Xie, W.Y.; Shen, Q.; Zhao, F.J. Antibiotics and Antibiotic Resistance from Animal Manures to Soil: A Review. Eur. J. Soil Sci. 2018, 69, 181–195. [Google Scholar] [CrossRef] [Green Version]
- Van Boeckel, T.P.; Brower, C.; Gilbert, M.; Grenfell, B.T.; Levin, S.A.; Robinson, T.P.; Teillant, A.; Laxminarayan, R. Global trends in antimicrobial use in food animals. Proc. Natl. Acad. Sci. USA 2015, 112, 5649–5654. [Google Scholar] [CrossRef] [Green Version]
- Jia, S.Y.; Zhang, X.X.; Miao, Y.; Zhao, Y.T.; Ye, L.; Li, B.; Zhang, T. Fate Of Antibiotic Resistance Genes And Their Associations With Bacterial Community In Livestock Breeding Wastewater And Its Receiving River Water. Water Res. 2017, 124, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.L.; Ngo, H.H.; Guo, W.S.; Liu, Y.W.; Zhou, J.L.; Chang, S.W.; Nguyen, D.D.; Bui, X.T.; Zhang, X.B. Bioprocessing for elimination antibiotics and hormones from swine wastewater. Sci. Total Environ. 2018, 621, 1664–1682. [Google Scholar] [CrossRef]
- Martinez, J.L. Environmental Pollution by Antibiotics and by Antibiotic Resistance Determinants. Environ. Pollut. 2009, 157, 2893–2902. [Google Scholar] [CrossRef]
- Boeckel, T.; Pires, J.; Silvester, R.; Zhao, C.; Song, J.; Criscuolo, N.G.; Gilbert, M.; Bonhoeffer, S.; Laxminarayan, R. Global Trends in Antimicrobial Resistance in Animals in Low- and Middle-Income Countries. Science 2019, 365, 6459. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Du, M.M.; Jiang, H.Y.; Zhang, D.D.; Lin, L.F.; Yea, H.; Zhang, X. Occurrence, Seasonal Variation and Removal Efficiency of Antibiotics and Their Metabolites in Wastewater Treatment Plants, Jiulongjiang River Basin, South China. Environ. Sci. Process. Impacts 2015, 17, 225–234. [Google Scholar] [CrossRef]
- Zhang, M.; He, L.-Y.; Liu, Y.-S.; Zhao, J.-L.; Liu, W.-R.; Zhang, J.-N.; Chen, J.; He, L.-K.; Zhang, Q.-Q.; Ying, G.-G. Fate of Veterinary Antibiotics During Animal Manure Composting. Sci. Total Environ. 2019, 650, 1363–1370. [Google Scholar] [CrossRef]
- Park, H.; Choung, Y.K. Degradation of antibiotics (tetracycline, sulfathiazole, ampicillin) using enzymes of glutathion S-transferase. Hum. Ecol. Risk Assess. 2007, 13, 1147–1155. [Google Scholar] [CrossRef]
- Yanghui, X.; Moustapha, H.; Pei-Ying, H. Performance and microbial community variations of anaerobic digesters under increasing tetracycline concentrations. Appl. Microbiol. Biotechnol. 2017, 101, 5505–5517. [Google Scholar]
- Arikan, O.A. Degradation and metabolization of chlortetracycline during the anaerobic digestion of manure from medicated calves. J. Hazard. Mater. 2008, 158, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Rui, W.; Wei, Y.; Zhao, G.; Zhou, H.; Xin, Z.; Zhong, W. Effects of Residual Tetracyclines and Copper on Swine Manure Anaerobic Digestion. Asian J. Ecotoxicol. 2015, 7, 110–116. [Google Scholar]
- Jian, W.; Weiwei, B.; Yu, Z.; Min, Y.; Zhimin, Q. Effects of thermophilic composting on oxytetracycline, sulfamethazine, and their corresponding resistance genes in swine manure. Environ. Sci. Process. Impacts 2015, 17, 1654–1660. [Google Scholar]
- Ma, Y.; Wilson, C.A.; Novak, J.T.; Riffat, R.; Aynur, S.; Murthy, S.; Pruden, A. Effect of Various Sludge Digestion Conditions on Sulfonamide, Macrolide, and Tetracycline Resistance Genes and Class I Integrons. Environ. Sci. Technol. 2011, 45, 7855–7861. [Google Scholar] [CrossRef] [PubMed]
- Diehl, D.L.; LaPara, T.M. Effect of Temperature on the Fate of Genes Encoding Tetracycline Resistance and the Integrase of Class 1 Integrons within Anaerobic and Aerobic Digesters Treating Municipal Wastewater Solids. Environ. Sci. Technol. 2010, 44, 9128–9133. [Google Scholar] [CrossRef]
- Burch, T.R.; Sadowsky, M.J.; LaPara, T.M. Modeling the fate of antibiotic resistance genes and class 1 integrons during thermophilic anaerobic digestion of municipal wastewater solids. Appl. Microbiol. Biotechnol. 2016, 100, 1437–1444. [Google Scholar] [CrossRef]
- Zhen, L.S.; Gu, J.; Hu, T.; Chen, Z.X. Effects of compost containing oxytetracycline on enzyme activities and microbial communities in maize rhizosphere soil. Environ. Sci. Pollut. Res. 2018, 25, 29459–29467. [Google Scholar] [CrossRef] [PubMed]
- Akyol, C.; Aydin, S.; Ince, O.; Ince, B. A comprehensive microbial insight into single-stage and two-stage anaerobic digestion of oxytetracycline-medicated cattle manure. Chem. Eng. J. 2016, 303, 675–684. [Google Scholar] [CrossRef]
- Chen, Y.S.; Zhang, H.B.; Luo, Y.M.; Song, J. Occurrence and dissipation of veterinary antibiotics in two typical swine wastewater treatment systems in east China. Environ. Monit. Assess. 2012, 184, 2205–2217. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.F.; Wei, Y.S.; Zheng, J.X.; Zhao, X.; Zhong, W.K. The behavior of tetracyclines and their degradation products during swine manure composting. Bioresour. Technol. 2011, 102, 5924–5931. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Feng, F.; Chai, Y.F.; Meng, X.S.; Sui, Q.W.; Chen, M.X.; Wei, Y.S.; Qi, K.M. Screening and quantitation of residual antibiotics in two different swine wastewater treatment systems during warm and cold seasons. Sci. Total Environ. 2019, 660, 1542–1554. [Google Scholar] [CrossRef] [PubMed]
- Kasumba, J.; Appala, K.; Agga, G.E.; Loughrin, J.H.; Conte, E.D. Anaerobic digestion of livestock and poultry manures spiked with tetracycline antibiotics. J. Environ. Sci. Health Part B-Pestic. Food Contam. Agric. Wastes 2020, 55, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Pu, C.J.; Yu, X.L.; Sun, Y.; Chen, J.H. Removal of tetracyclines, sulfonamides, and quinolones by industrial-scale composting and anaerobic digestion processes. Environ. Sci. Pollut. Res. 2018, 25, 35835–35844. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Lee, Y.W.; Jahng, D. Anaerobic co-digestion of food waste and piggery wastewater: Focusing on the role of trace elements. Bioresour. Technol. 2011, 102, 5048–5059. [Google Scholar] [CrossRef]
- Zhou, M.M.; Yan, B.H.; Wong, J.W.C.; Zhang, Y. Enhanced volatile fatty acids production from anaerobic fermentation of food waste: A mini-review focusing on acidogenic metabolic pathways. Bioresour. Technol. 2018, 248, 68–78. [Google Scholar] [CrossRef]
- Jiang, L.; Hu, X.; Xu, T.; Zhang, H.; Sheng, D.; Yin, D. Prevalence of antibiotic resistance genes and their relationship with antibiotics in the Huangpu River and the drinking water sources, Shanghai, China. Sci. Total Environ. 2013, 458, 267–272. [Google Scholar] [CrossRef]
- Graham, D.W.; Olivares-Rieumont, S.; Knapp, C.W.; Lima, L.; Werner, D.; Bowen, E. Antibiotic Resistance Gene Abundances Associated with Waste Discharges to the Almendares River near Havana, Cuba. Environ. Sci. Technol. 2011, 45, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Li, H.J.; Song, D.; Lin, X.G.; Wang, Y.M. Influence of zeolite and superphosphate as additives on antibiotic resistance genes and bacterial communities during factory-scale chicken manure composting. Bioresour. Technol. 2018, 263, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Tan, Y.; Guo, Y.; Gu, X.; Wang, X.; Zhang, Y. Interactions of tetracycline with Cd (II), Cu (II) and Pb (II) and their cosorption behavior in soils. Environ. Pollut. 2013, 180, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Fan, N.-S.; Zhu, X.-L.; Wu, J.; Tian, Z.; Bai, Y.-H.; Huang, B.-C.; Jin, R.-C. Deciphering the microbial and genetic responses of anammox biogranules to the single and joint stress of zinc and tetracycline. Environ. Int. 2019, 132, 105097. [Google Scholar] [CrossRef]
- Uivarosi, V. Metal Complexes of Quinolone Antibiotics and Their Applications: An Update. Molecules 2013, 18, 11153–11197. [Google Scholar] [CrossRef]
- Chiang, P.N.; Chiu, C.Y.; Wang, M.K.; Chen, B.T. Low-Molecular-Weight Organic Acids Exuded by Millet (Setaria italica (L.) Beauv.) Roots and Their Effect on the Remediation of Cadmium-Contaminated Soil. Soil Sci. 2011, 176, 33–38. [Google Scholar] [CrossRef]
- Aydin, S.; Shahi, A.; Ozbayram, E.G.; Ince, B.; Ince, O. Use of PCR-DGGE based molecular methods to assessment of microbial diversity during anaerobic treatment of antibiotic combinations. Bioresour. Technol. 2015, 192, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Dash, S.; Ng, C.Y.; Maranas, C.D. Metabolic modeling of clostridia: Current developments and applications. Fems Microbiol. Lett. 2016, 363, fnw004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duda, R.M.; Vantini, J.D.; Martins, L.S.; Varani, A.D.; Franco Lemos, M.V.; Tiraboschi Ferro, M.I.; de Oliveira, R.A. A balanced microbiota efficiently produces methane in a novel high-rate horizontal anaerobic reactor for the treatment of swine wastewater. Bioresour. Technol. 2015, 197, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.-H.; Cheng, C.-H.; Guan, D.-W.; Wang, S.-T.; Hsu, S.-C.; Liang, C.-M.; Lin, C.-Y. Interactions between Clostridium sp. and other facultative anaerobes in a self-formed granular sludge hydrogen-producing bioreactor. Int. J. Hydrogen Energy 2011, 36, 8704–8711. [Google Scholar] [CrossRef]
- Leng, L.; Yang, P.; Singh, S.; Zhuang, H.; Xu, L.; Chen, W.-H.; Dolfing, J.; Li, D.; Zhang, Y.; Zeng, H.; et al. A review on the bioenergetics of anaerobic microbial metabolism close to the thermodynamic limits and its implications for digestion applications. Bioresour. Technol. 2018, 247, 1095–1106. [Google Scholar] [CrossRef]
- Cheng, Z.; Hu, X.; Sun, Z.R. Microbial community distribution and dominant bacterial species analysis in the bio-electrochemical system treating low concentration cefuroxime. Chem. Eng. J. 2016, 303, 137–144. [Google Scholar] [CrossRef]
- Schloss, P.D.; Gevers, D.; Westcott, S.L. Reducing the Effects of PCR Amplification and Sequencing Artifacts on 16S rRNA-Based Studies. PLoS ONE 2011, 6, e27310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, C.; Huang, Z.; Chen, M.; Liu, F.; Lu, Y.; Nong, Z.; Zhu, X. Effects of infiltrator structure and hydraulic loading rates on pollutant removal efficiency and microbial community in a modified two-stage constructed rapid infiltration systems treating swine wastewater. Environ. Prog. Sustain. Energy 2019, 38, 13218. [Google Scholar] [CrossRef]
- Reith, F.; Lengke, M.F.; Falconer, D.; Craw, D.; Southam, G. The geomicrobiology of gold. Isme J. 2007, 1, 567–584. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Tarighi, S.; Doetsch, A.; Haeussler, S.; Muesken, M.; Wright, V.J.; Camara, M.; Williams, P.; Haenen, S.; Boerjan, B.; et al. Phenotypic and Genome-Wide Analysis of an Antibiotic-Resistant Small Colony Variant (SCV) of Pseudomonas aeruginosa. PLoS ONE 2011, 6, e29276. [Google Scholar] [CrossRef]
- Elkhatib, W.; Noreddin, A. In Vitro Antibiofilm Efficacies of Different Antibiotic Combinations with Zinc Sulfate against Pseudomonas aeruginosa Recovered from Hospitalized Patients with Urinary Tract Infection. Antibiotics 2014, 3, 64–84. [Google Scholar] [CrossRef] [Green Version]
- Ghasemi, F.; Jalal, R. Antimicrobial action of zinc oxide nanoparticles in combination with ciprofloxacin and ceftazidime against multidrug-resistant Acinetobacter baumannii. J. Glob. Antimicrob. Resist. 2016, 6, 118–122. [Google Scholar] [CrossRef]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371–384. [Google Scholar] [CrossRef]
- Sui, Q.W.; Jiang, C.; Yu, D.W.; Chen, M.X.; Zhang, J.Y.; Wang, Y.W.; Wei, Y.S. Performance of a sequencing-batch membrane bioreactor (SMBR) with an automatic control strategy treating high-strength swine wastewater. J. Hazard. Mater. 2018, 342, 210–219. [Google Scholar] [CrossRef]
| Indicator | Value (Unit) |
|---|---|
| pH | 8.01 ± 0.1 |
| TOC | 2598.8 ± 130.0 mg/L |
| TN | 1374.0 ± 54.4 mg/L |
| TP | 112.8 ± 5.3 mg/L |
| Group | 0 | 2 | 4 | 6 | 8 | 10 | 12 | 14 | 16 | 18 | 20 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2 | 1.00 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 3 | 0.33 | 0 | 0.33 | 0 | 0.33 | 0 | 0 | 0 | 0 | 0 | 0 |
| 4 | 0 | 0 | 1.00 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
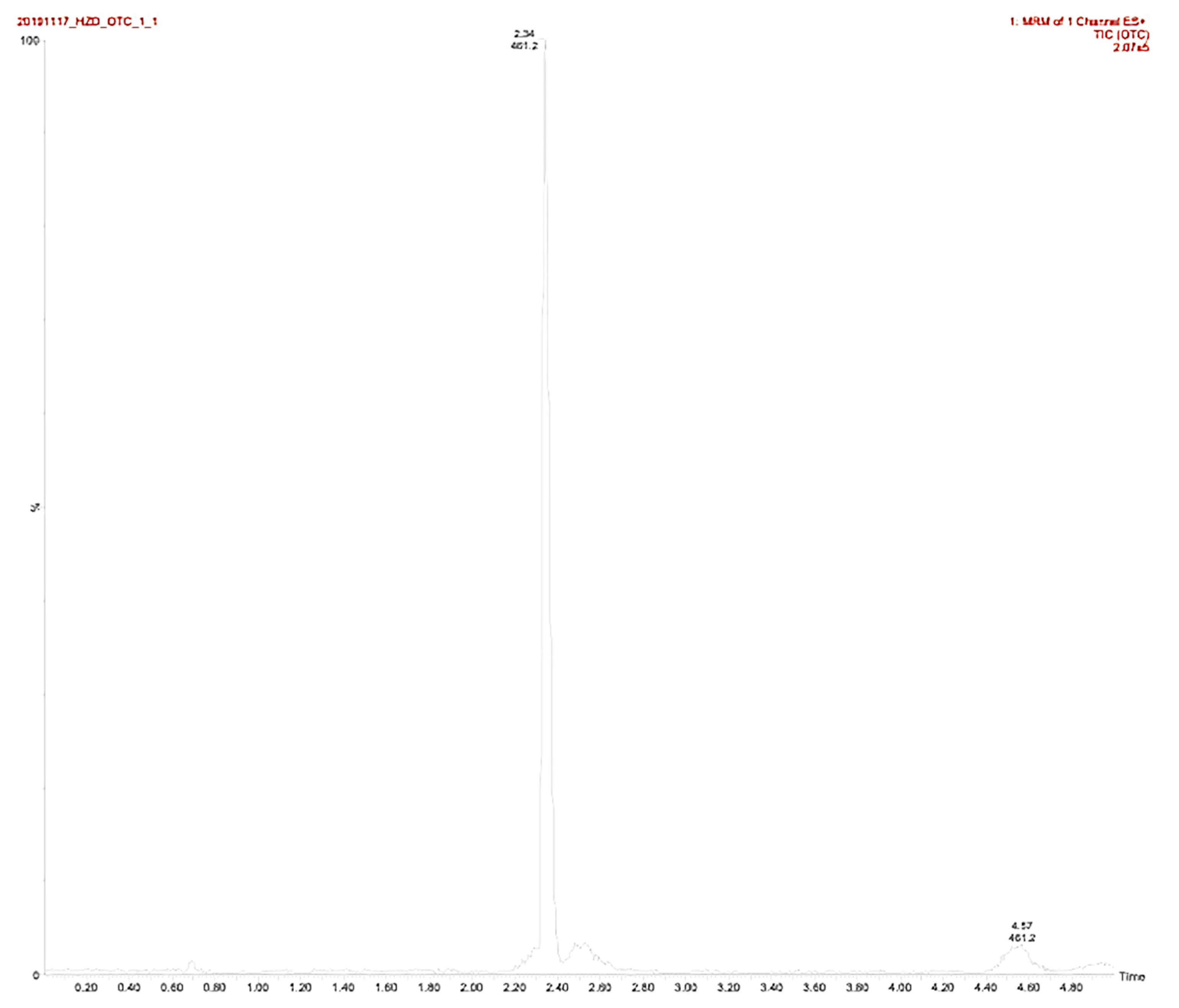
| Compound | Retention Time (min) | Formula | Parent (m/z) | Daughters (m/z) | Cone Voltage (V) | Collision Energy (eV) |
|---|---|---|---|---|---|---|
| OTC | 2.34 | C22H24N2O8 | 444.45 | 409.97 | 32 | 28 |
| TC | 2.35 | C22H24N2O9 | 460.45 | 426.00 | 32 | 18 |
| CTC | 2.47 | C22H23ClN2O8 | 479.02 | 443.98 | 40 | 30 |
| DOX | 2.52 | C22H24N2O8 | 445.06 | 153.96 | 40 | 30 |
| Antibiotic | Group | Initial Concentration C0 (ug/L) | Rate Constant k (d−1) | Equation | Half-Life t1/2 (d) | R2 |
|---|---|---|---|---|---|---|
| OTC | 1 | 3.0518 ± 0.2328 | 0.6418 ± 0.0929 | y = 2.6532 × e(−x/1.5580) + 0.3986 | 1.0799 ± 0.1562 | 0.9644 |
| 2 | 4.6182 ± 1.6674 | 0.1500 ± 0.0876 | y = 4.5725 × e(−x/6.6670) + 0.0456 | 4.6212 ± 2.6984 | 0.8940 | |
| 3 | 3.4847 ± 0.4583 | 0.5374 ± 0.1267 | y = 3.1303 × e(−x/1.8607) + 0.3543 | 1.2898 ± 0.3041 | 0.9095 | |
| 4 | 3.4199 ± 1.0734 | 1.0366 ± 0.7981 | y = 2.3494 × e(−x/0.9647) + 1.0705 | 0.9647 ± 0.7428 | 0.4217 | |
| TC | 1 | 0.8804 ± 0.0800 | 0.4569 ± 0.1057 | y = 0.5423 × e(−x/2.1888) + 0.3381 | 1.5172 ± 0.3509 | 0.9119 |
| 2 | 0.8571 ± 0.0872 | 1.3607 ± 0.4106 | y = 0.4898 × e(−x/0.7349) + 0.3673 | 0.5094 ± 0.1537 | 0.8459 | |
| 3 | 1.0108 ± 0.1096 | 0.4890 ± 0.1282 | y = 0.6610 × e(−x/2.0450) + 0.3499 | 1.4175 ± 0.3716 | 0.8895 | |
| 4 | 0.8667 ± 0.1189 | 0.3506 ± 0.1169 | y = 0.5330 × e(−x/2.8526) + 0.3337 | 1.9773 ± 0.6596 | 0.8291 | |
| CTC | 1 | 7.1269 ± 0.6777 | 0.8531 ± 0.1835 | y = 5.2933 × e(−x/1.1723) + 1.8336 | 0.8126 ± 0.1748 | 0.9224 |
| 2 | 5.7384 ± 0.9615 | 0.9616 ± 0.4567 | y = 3.4088 × e(−x/1.0400) + 2.3296 | 0.7209 ± 0.3424 | 0.6919 | |
| 3 | 7.8822 ± 1.2341 | 0.3613 ± 0.1260 | y = 5.3175 × e(−x/2.7678) + 2.5647 | 1.9185 ± 0.6693 | 0.8153 | |
| 4 | 6.5028 ± 1.0580 | 0.1362 ± 0.0385 | y = 4.7480 × e(−x/7.3450) + 1.7548 | 5.0912 ± 1.4401 | 0.9293 | |
| DOC | 1 | 13.3652 ± 1.3394 | 0.8698 ± 0.2043 | y = 9.5854 × e(−x/1.1497) + 3.7798 | 0.7969 ± 0.1872 | 0.9084 |
| 2 | 14.2782 ± 1.7099 | 1.0783 ± 0.3013 | y = 10.3165 × e(−x/0.9274) + 3.9617 | 0.6428 ± 0.1796 | 0.8699 | |
| 3 | 12.9623 ± 1.9475 | 0.3157 ± 0.0945 | y = 9.5809 × e(−x/3.1674) + 3.3814 | 2.1955 ± 0.6568 | 0.8602 | |
| 4 | 12.3889 ± 1.9377 | 0.2785 ± 0.0893 | y = 8.7491 × e(−x/3.5902) + 3.6398 | 2.4885 ± 0.7980 | 0.8462 |
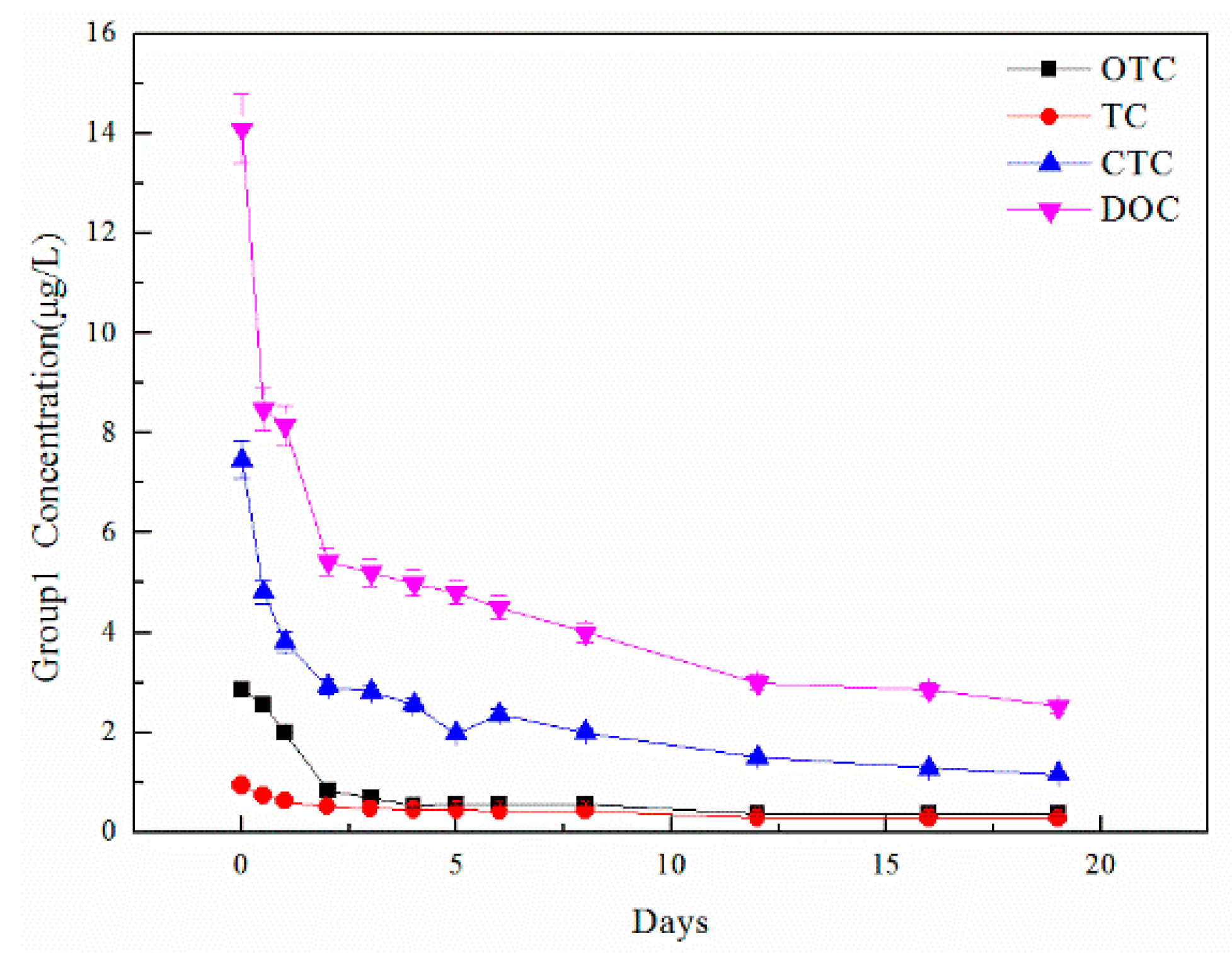
| TCs | Initial Concentration C0 (ug/L) | Day 20 Concentration C20 (ug/L) | Removal Efficiency |
|---|---|---|---|
| TC | 0.94 | 0.28 | 70.21% |
| OTC | 2.87 | 0.37 | 87.11% |
| CTC | 7.47 | 1.16 | 84.47% |
| DOC | 14.10 | 2.52 | 82.13% |
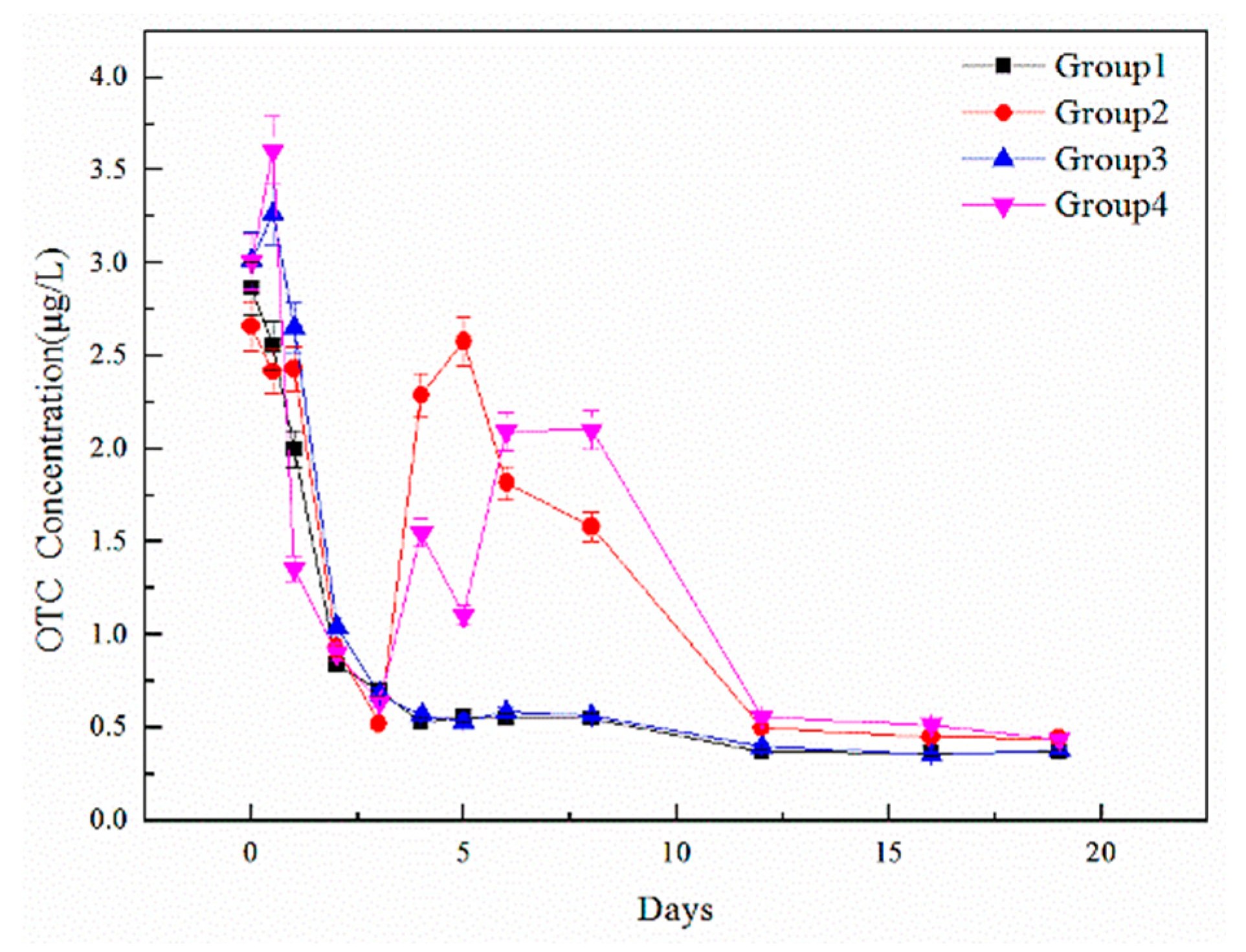
| TCs | Group | Initial Concentration C0 (ug/L) | Adding Concentration (ug/L) | Day 20 Concentration C20 (ug/L) | Removal Efficiency |
|---|---|---|---|---|---|
| OTC | 1 | 2.87 | 0.00 | 0.37 | 87.11% |
| 2 | 2.66 | 1.00 | 0.45 | 87.70% | |
| 3 | 3.02 | 1.00 | 0.38 | 90.55% | |
| 4 | 3.01 | 1.00 | 0.44 | 89.03% | |
| TC | 1 | 0.94 | 0.00 | 0.28 | 70.21% |
| 2 | 0.88 | 0.00 | 0.29 | 67.05% | |
| 3 | 1.12 | 0.00 | 0.30 | 73.21% | |
| 4 | 1.00 | 0.00 | 0.28 | 72.00% | |
| CTC | 1 | 7.47 | 0.00 | 1.16 | 84.47% |
| 2 | 6.12 | 0.00 | 1.53 | 75.00% | |
| 3 | 9.29 | 0.00 | 2.23 | 76.00% | |
| 4 | 7.15 | 0.00 | 2.10 | 70.63% | |
| DOC | 1 | 14.10 | 0.00 | 2.52 | 82.13% |
| 2 | 14.82 | 0.00 | 2.30 | 84.46% | |
| 3 | 15.13 | 0.00 | 3.00 | 80.17% | |
| 4 | 14.53 | 0.00 | 2.87 | 80.25% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Z.; Fan, Z.; Song, Q.; Khatoon, R.; Zhang, M.; Wang, N.; Luo, X. The Effect of Exogenous Oxytetracycline on High-Temperature Anaerobic Digestion of Elements in Swine Wastewater. Water 2021, 13, 3497. https://doi.org/10.3390/w13243497
Hu Z, Fan Z, Song Q, Khatoon R, Zhang M, Wang N, Luo X. The Effect of Exogenous Oxytetracycline on High-Temperature Anaerobic Digestion of Elements in Swine Wastewater. Water. 2021; 13(24):3497. https://doi.org/10.3390/w13243497
Chicago/Turabian StyleHu, Zhongda, Zijing Fan, Qixuan Song, Rabia Khatoon, Mei Zhang, Ning Wang, and Xingzhang Luo. 2021. "The Effect of Exogenous Oxytetracycline on High-Temperature Anaerobic Digestion of Elements in Swine Wastewater" Water 13, no. 24: 3497. https://doi.org/10.3390/w13243497
APA StyleHu, Z., Fan, Z., Song, Q., Khatoon, R., Zhang, M., Wang, N., & Luo, X. (2021). The Effect of Exogenous Oxytetracycline on High-Temperature Anaerobic Digestion of Elements in Swine Wastewater. Water, 13(24), 3497. https://doi.org/10.3390/w13243497







