3.1. Time Variation of CODCr and Permanganate Index in Qinghai Lake from 2010 to 2020
In order to clarify the background values of the Qinghai Lake watershed, indexes for monitoring water quality were collected and analyzed to determine the characterizations of the variations in water quality. According to the historical monitoring data on the water quality (
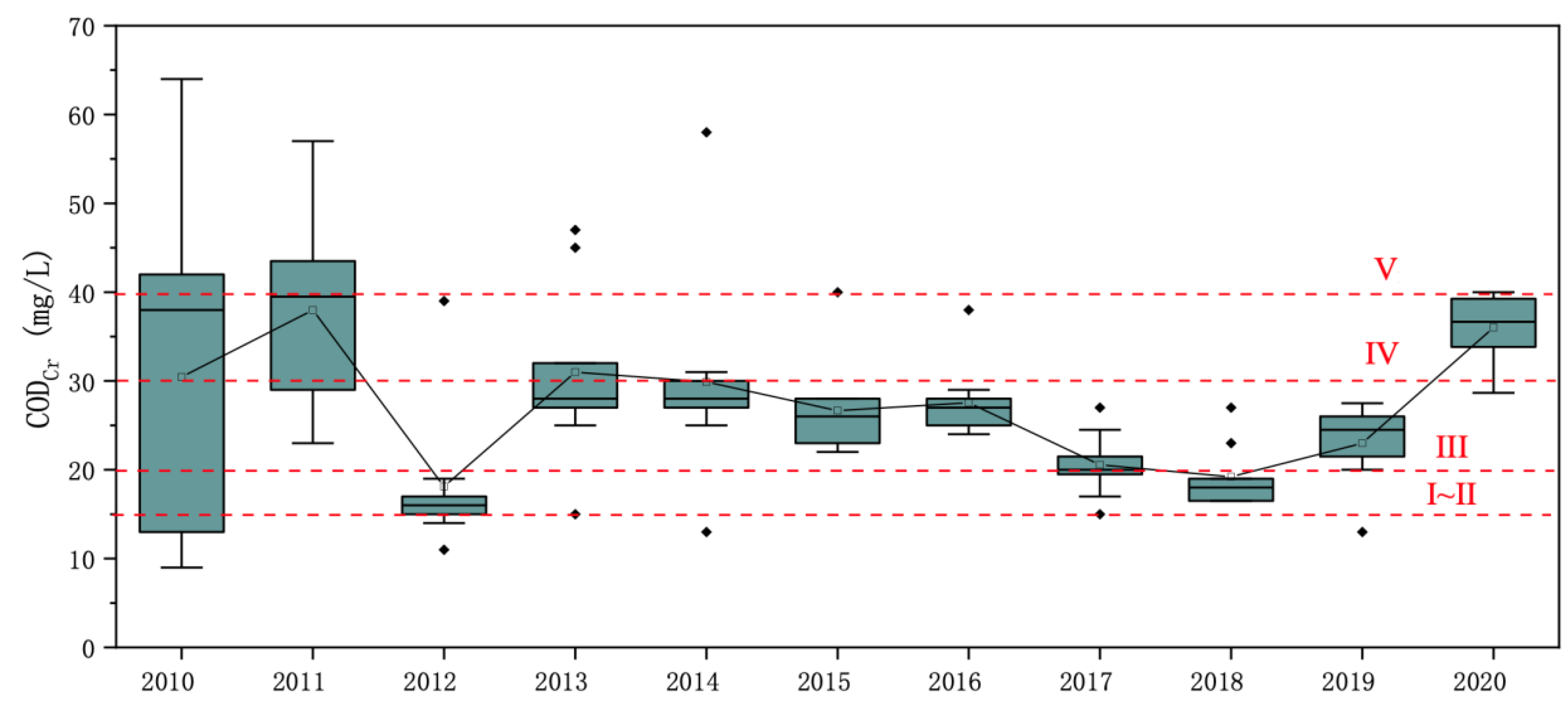
Figure 4), the COD
Cr concentration in Qinghai Lake was between 18.11 and 38.00 mg/L from 2010 to 2020, with an average of 27.10 mg/L, which was at the surface class IV level. Except for the large fluctuation of the COD
Cr concentration in 2010 and 2011, the fluctuation of the COD
Cr concentration in the water of Qinghai Lake in other years was relatively small. In terms of the mean values of the COD
Cr at each historical monitoring point, the mean value of COD
Cr at the Sha Dao point was the highest relative to the others with 43.84 mg/L.
From 2010 to 2020, the annual mean value of CODCr of the water body of Qinghai Lake first decreased and then increased; it fluctuated greatly between the class III and V levels of surface water. Among them, the average value of CODCr in 2011 was relatively the highest and was close to the upper limit of the class V water quality standard for surface water, and the annual average value of CODCr in 2012 was relatively the lowest. In 2020, the CODCr rose to the class IV water level, with an average of 36.02 mg/L.
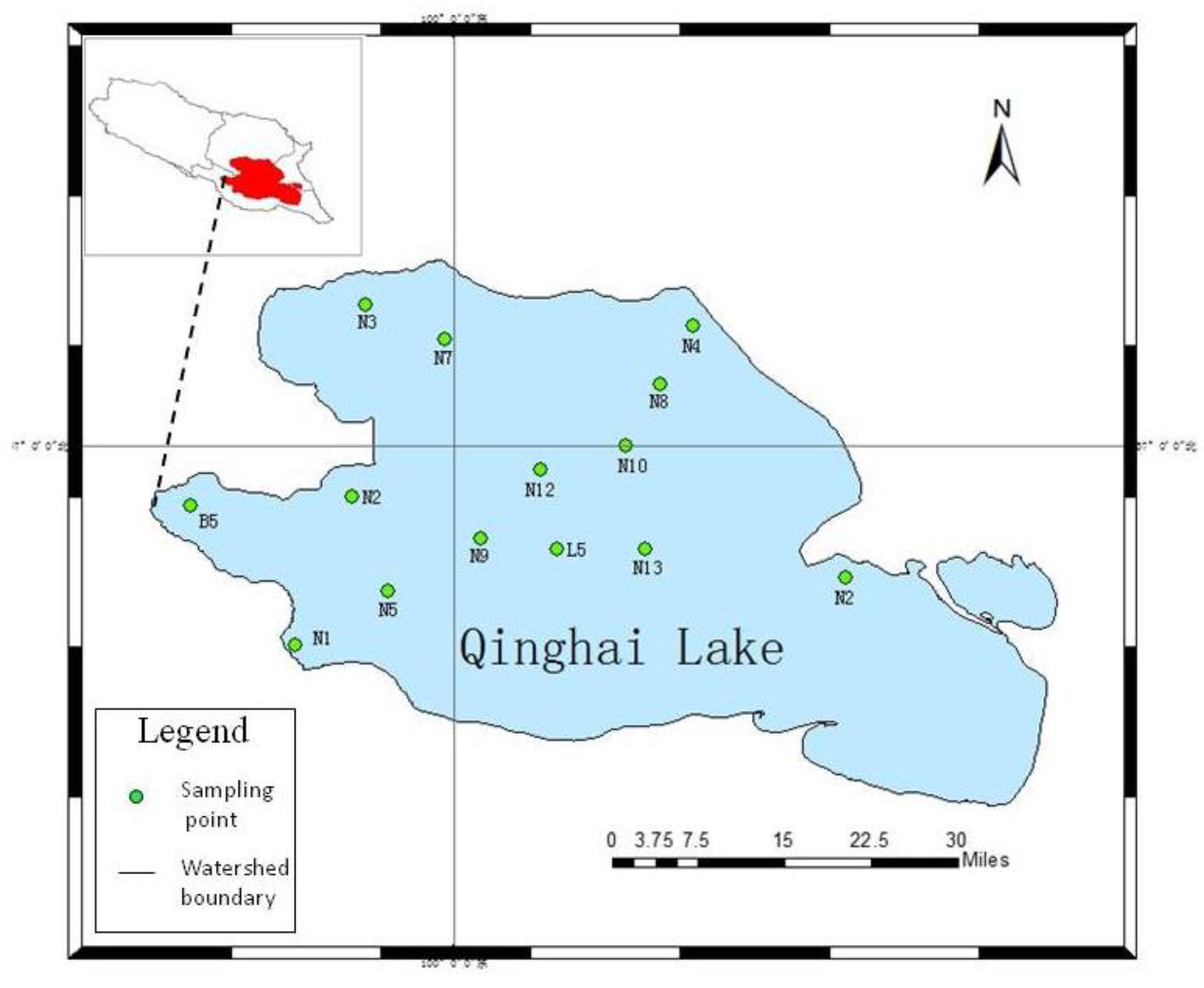
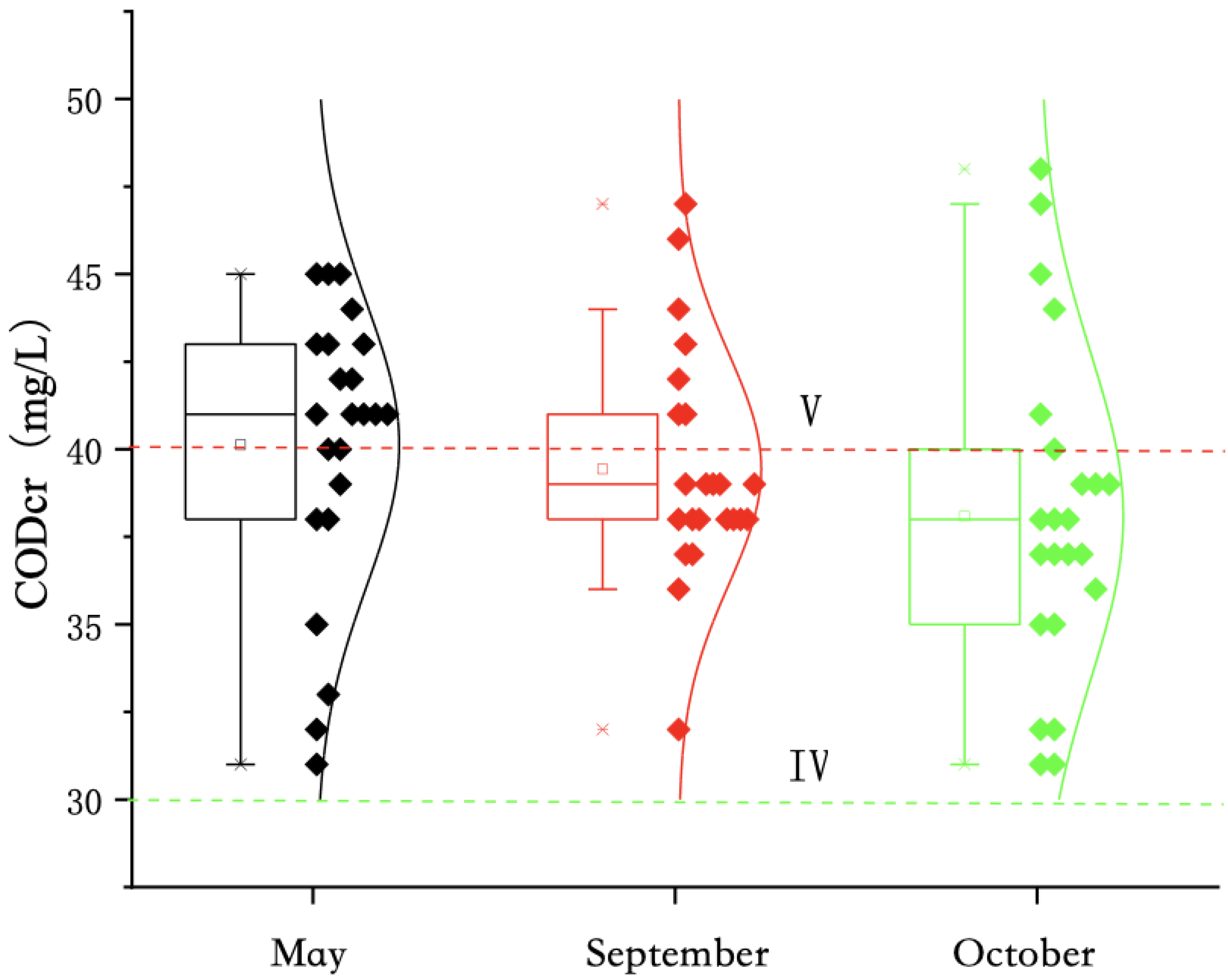
The on-site monitoring in May, September, and October 2020 showed that (
Figure 5) the annual COD
Cr of the water body of Qinghai Lake ranged from 31 to 48 mg/L, with an average of 39.21 mg/L. The COD
Cr of all points in the water body exceeded the class IV standard for surface water (threshold ≤ 30 mg/L), and the COD
Cr of at some points in the water body exceeded the class V standard for surface water (threshold ≤ 40 mg/L). From the perspective of monthly variation, the ranges of COD
Cr in the water body of Qinghai Lake were 31–45, 32–47, and 31–48 mg/L, respectively, and the average values were 40.13, 39.43, and 38.09 mg/L. On the whole, the difference in COD
Cr in the water body of Qinghai Lake in different months was small, and the COD
Cr concentration was at a high level. It can be seen from
Figure 4 that from 2018 to 2020, the water quality of the rivers entering Qinghai Lake gradually worsened from class II surface water in 2018 to class IV surface water in 2020. Combined with the detection data from the lake samples in 2020, it was found that the COD content in Qinghai Lake was similar to that in the rivers entering the lake. Though it is the largest saltwater lake in Western China, Qinghai Lake has a very small population, but there are many herdsmen living around the rivers entering the lake. Therefore, the main source of COD
Cr in Qinghai Lake may be affected by the rivers entering the lake, non-point sources in the grasslands, and human activities in the surroundings.
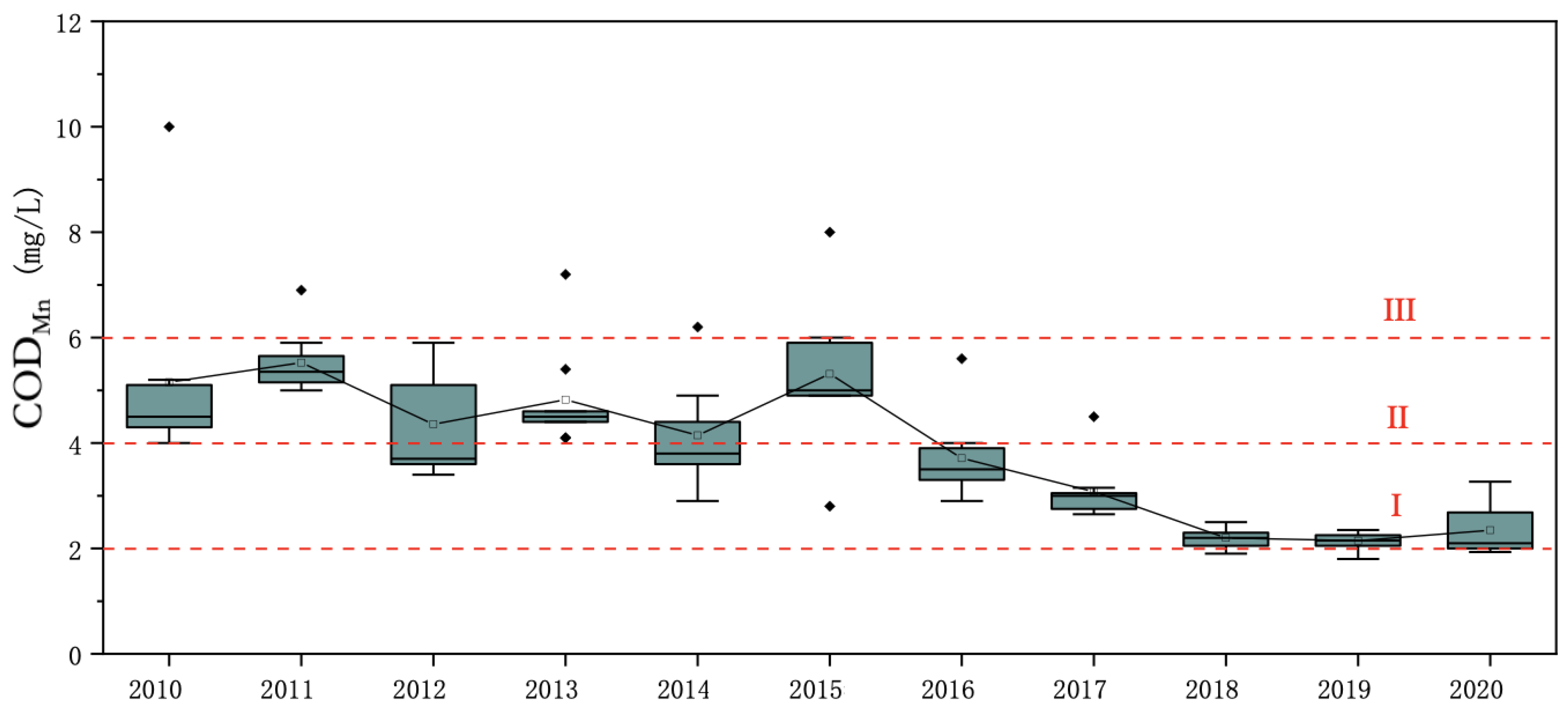
According to the historical monitoring data (
Figure 6), the permanganate concentration index in the water of Qinghai Lake from 2010 to 2020 was between 2.14 and 5.53 mg/L, with an average of 3.89 mg/L, which is at the class II level for surface water. Except for the large fluctuation of the permanganate concentration index in the water body in 2012, its fluctuation in the Qinghai Lake’s water body in other years was relatively small. Among the mean values of the permanganate index at each historical monitoring point, the mean value at the Shadao point was relatively the highest at 5.39 mg/L.
From 2010 to 2015, the annual mean value of the permanganate index in the water body of Qinghai Lake fluctuated as a whole, but all values met the class III level for surface water. From 2016 to 2020, the annual average values of the permanganate index in the water body of Qinghai Lake showed a downward trend and was basically at the class II level for surface water. The permanganate index remained at the level of 2.14–2.35 mg/L from 2018 to 2020, which was slightly higher than the limit for class I water quality. The average value of the permanganate index in 2011 was relatively the highest at 5.53 mg/L, which was close to the upper limit of the class III water quality standard for surface water, and the annual average value of the permanganate index in 2019 was relatively the lowest.
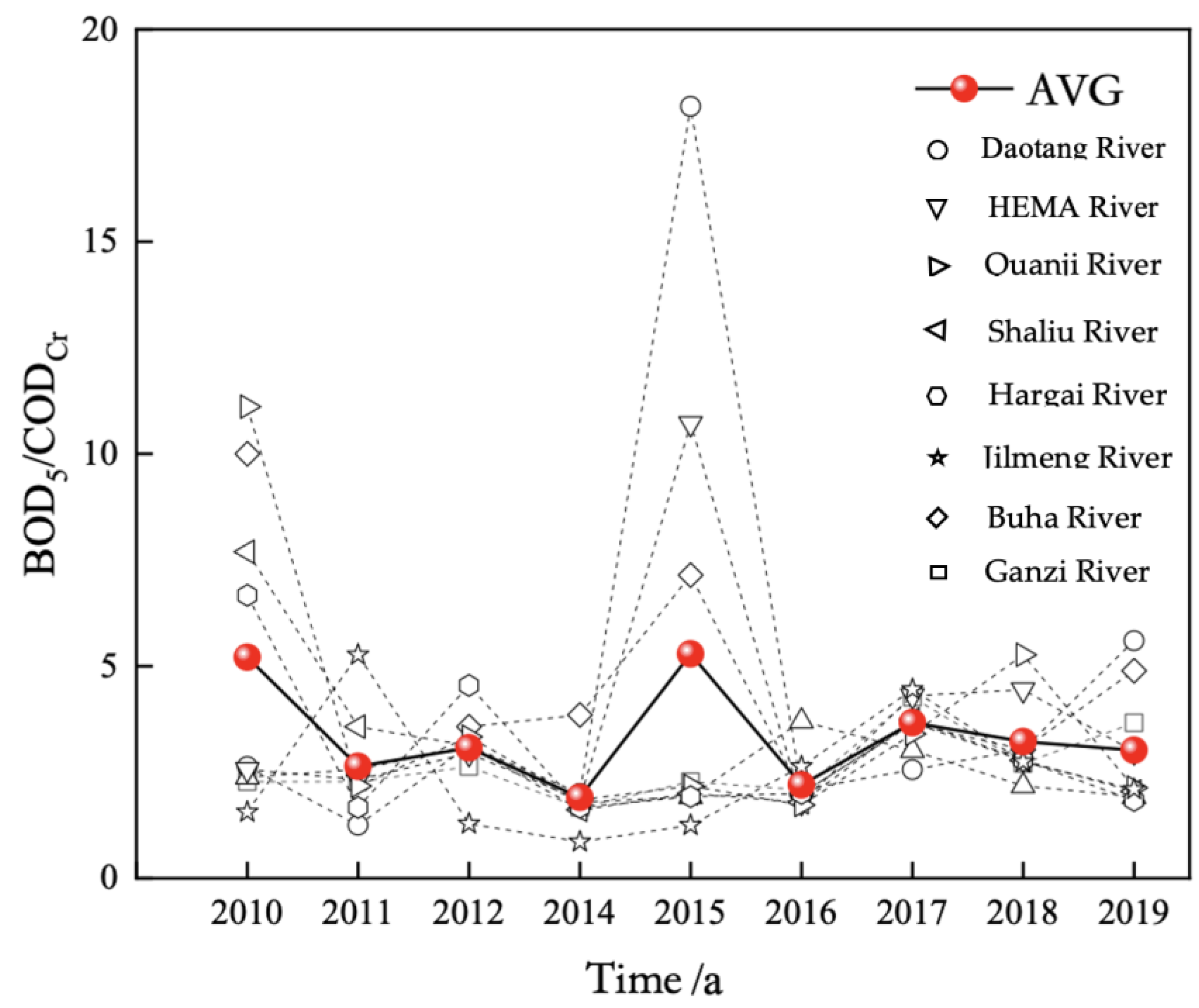
3.2. Time Variation of CODcr and Permanganate Index in the Rivers Entering Qinghai Lake from 2010 to 2020
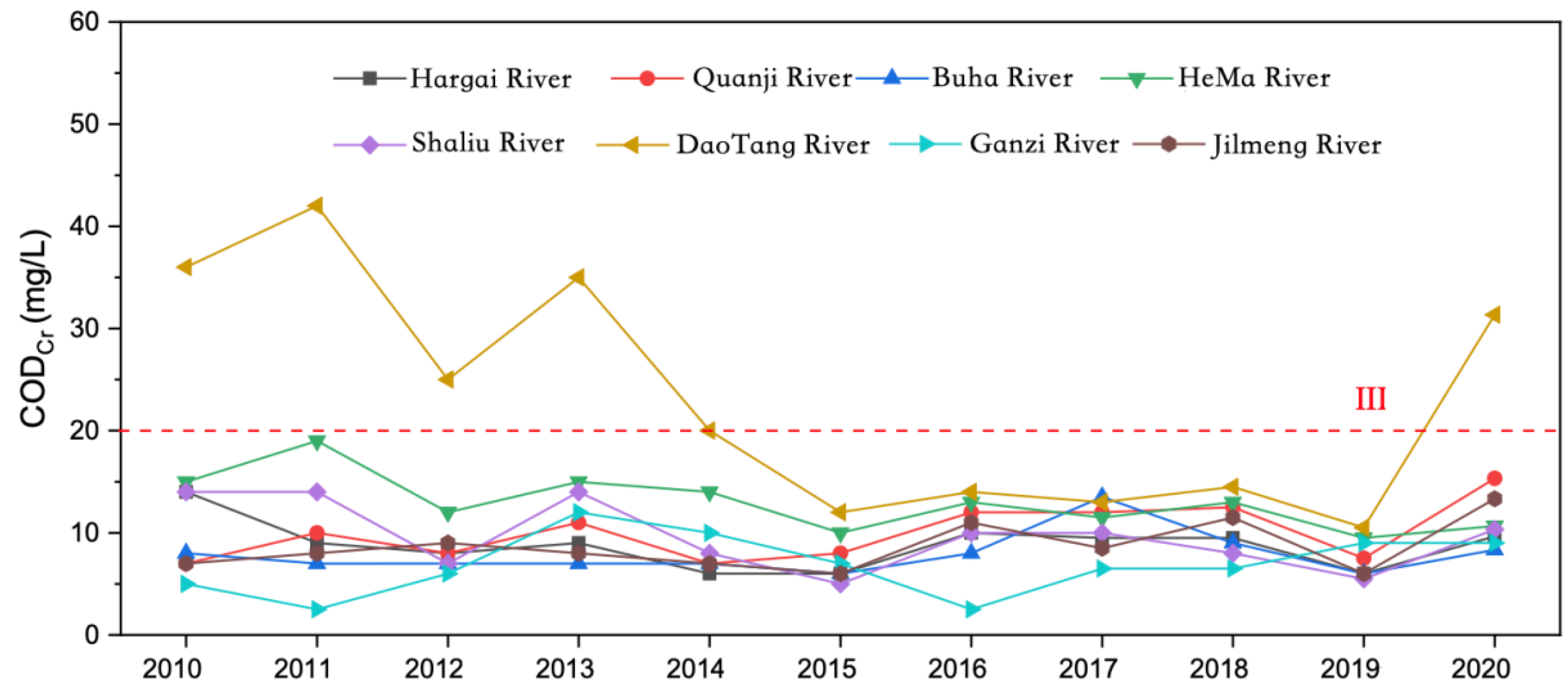
From 2010 to 2020, the COD
Cr of the main rivers entering Qinghai Lake Basin was 7.50–13.94 mg/L, with an average of 10.99 mg/L. The COD
Cr of the rivers entering the lake was lower than that of the lake as a whole (
Figure 7).
Except for the DaoTang River, the mean values of the CODCr of the major rivers were relatively similar from 2010 to 2020, and the interannual variation trend was relatively consistent. The eight main rivers entering Qinghai Lake from 2010 to 2020 were ranked in the following order according to their average CODcr: Ganzi River < Buha River < Jilmeng River < Hargai River < Shaliu River < Quanji River < HeMa River < DaoTang River.
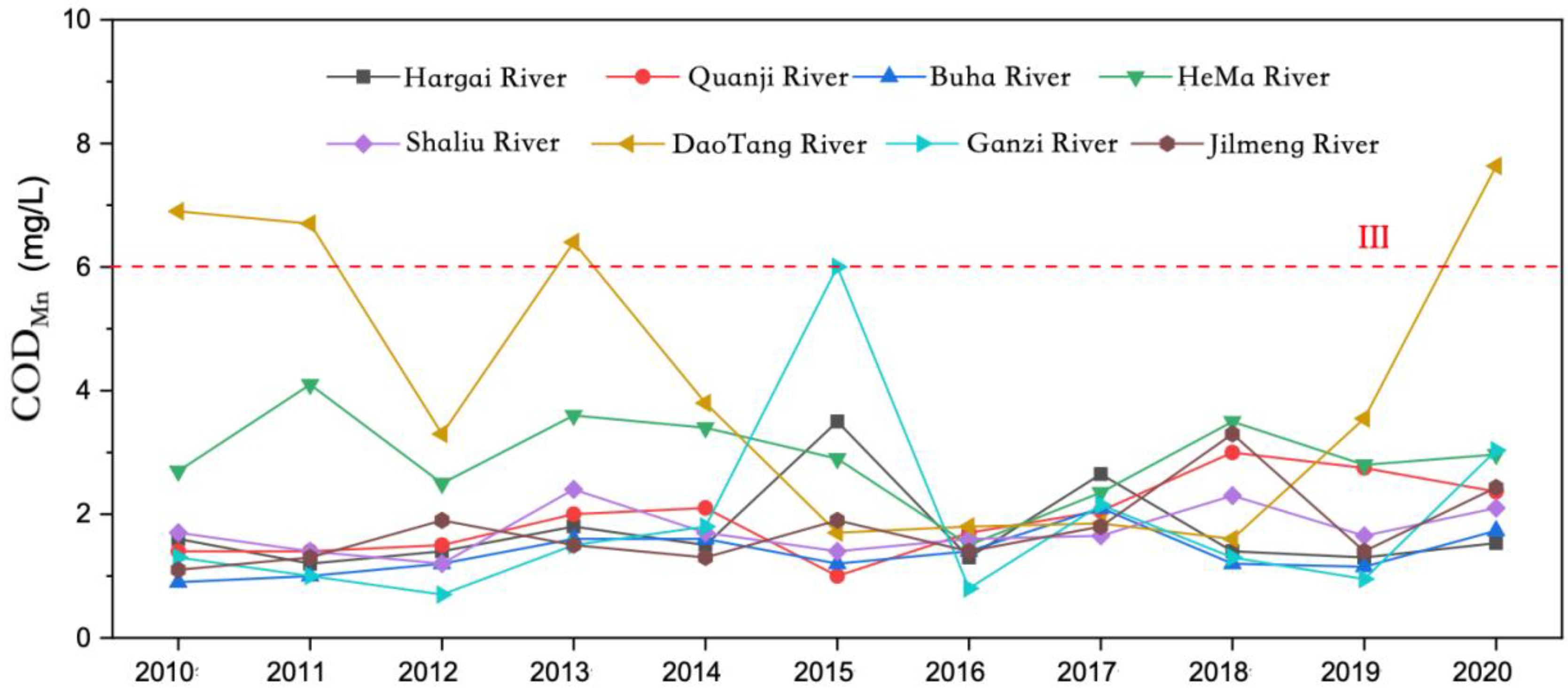
From 2010 to 2020, the permanganate index of the main rivers entering the lake in Qinghai Lake Basin was between 1.44 and 2.98 mg/L, with an average of 2.18 mg/L (
Figure 8); The permanganate index of the rivers entering the lake was lower than that of the lake as a whole.
Except for Daotang River and Heima River, the mean values of the permanganate index of the main rivers were relatively similar from 2010 to 2020, and the interannual variation trend was relatively consistent. The eight main rivers entering Qinghai Lake from 2010 to 2020 were ranked in the following order according to the average values of the permanganate index: Buha River < Shaliu River < Hargai River < Jilmeng River < Ganzi River < Quanji River < Heima River < DaoTang River. Except for Shaliu River and Ganzi River, the order of the other rivers was consistent with CODCr.
3.3. Spatial Distribution of CODCr and the Permanganate Index in Qinghai Lake
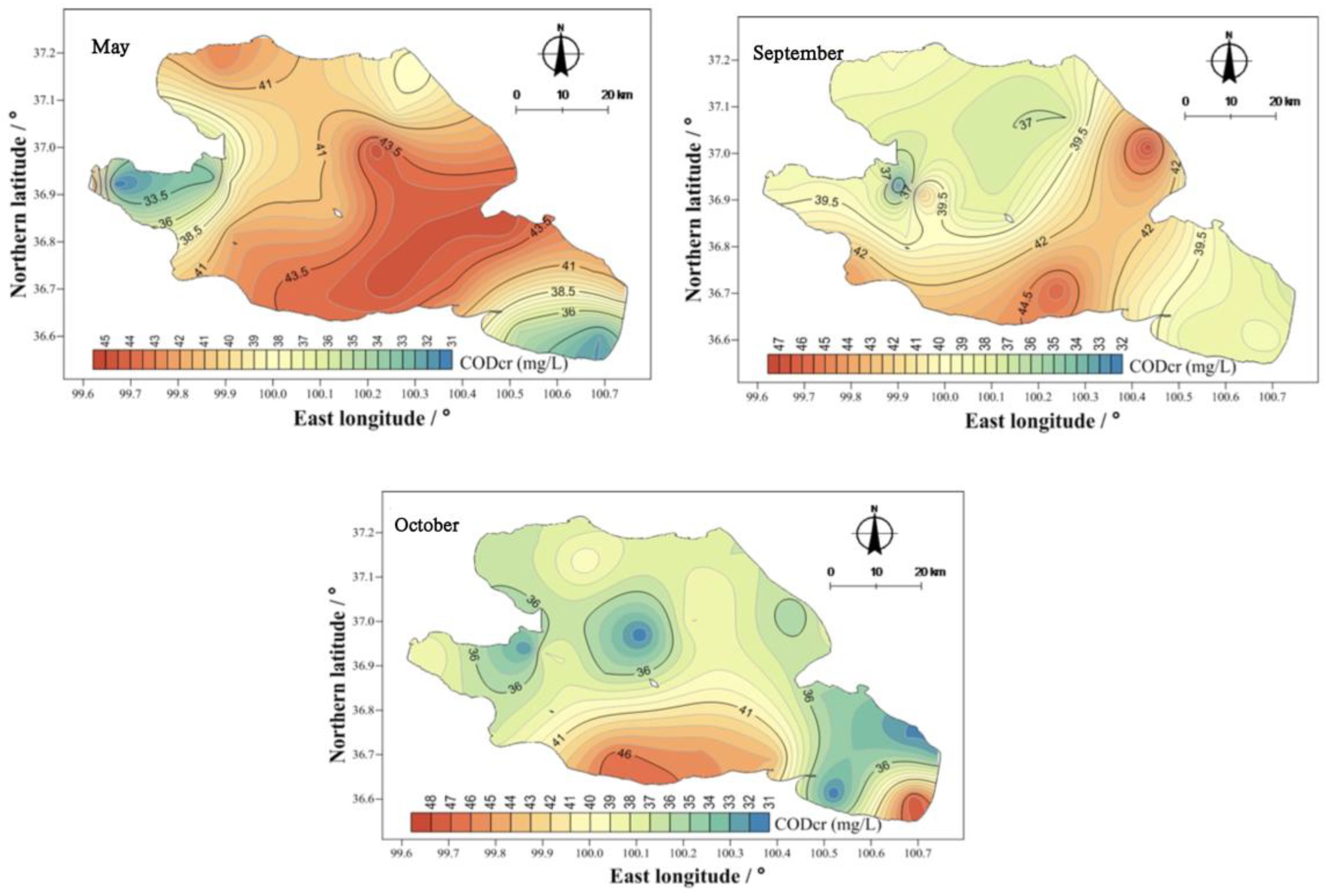
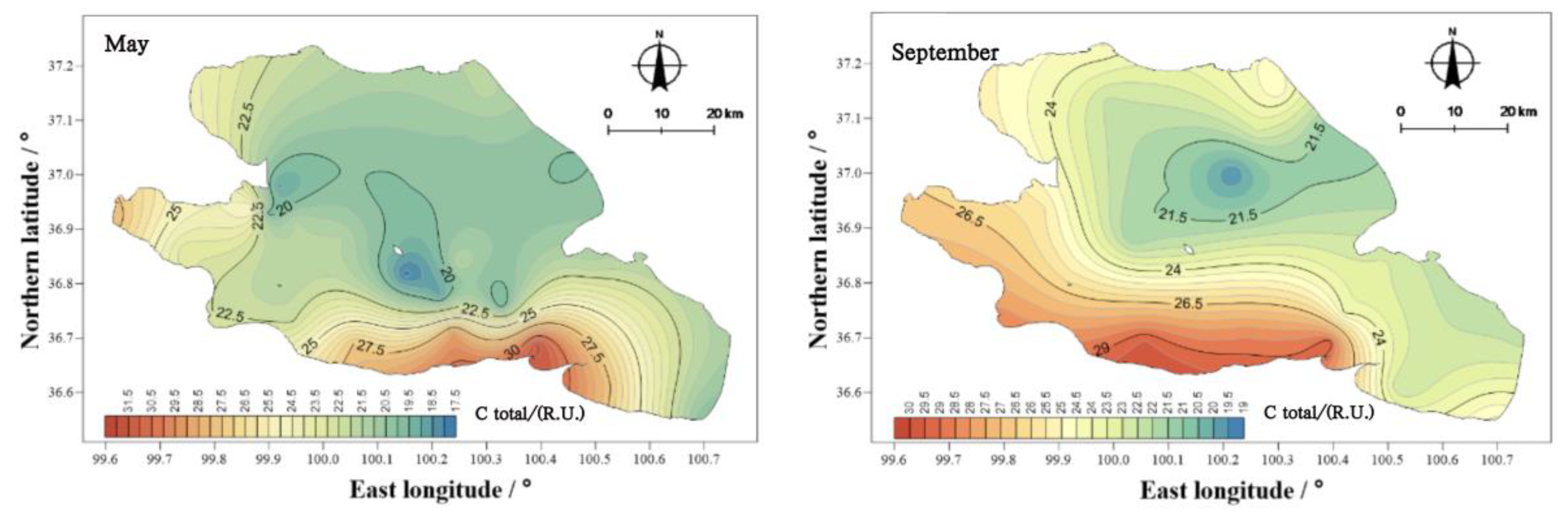
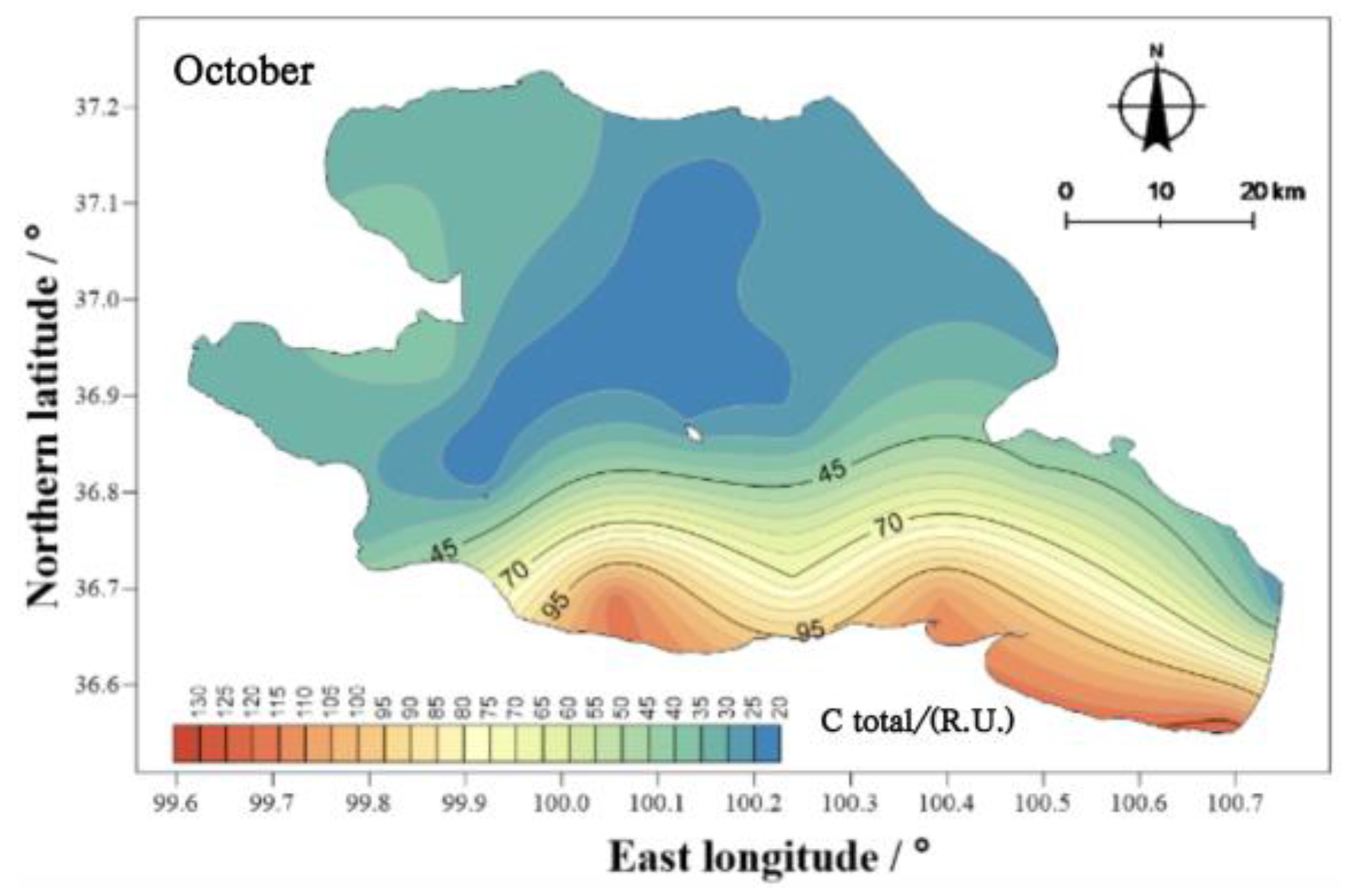
There were obvious differences in the spatial distribution of COD
Cr concentrations in Qinghai Lake (
Figure 9). In May, the COD
Cr was higher in the east than in the west, and that in the center of the lake was higher than that at the coast. In September and October, the COD
Cr of the water body showed two high areas, namely, near the Jiangxi ditch wharf and Sand Island, as well as near the Jiangxi ditch wharf and Qinghai Lake fishing ground wharf. From the perspective of the spatial distribution characteristics, the higher values in those three months were mainly concentrated at the Jiangxi ditch wharf and Qinghai Lake fishery wharf. The Jiangxi ditch wharf and Qinghai Lake fishery wharf are located in the south of Qinghai Province, and the planting industry is mainly along the south coast. Therefore, the input of external sources led to the higher concentration of CODCr in the water body in the south of Qinghai Lake than in other regions.
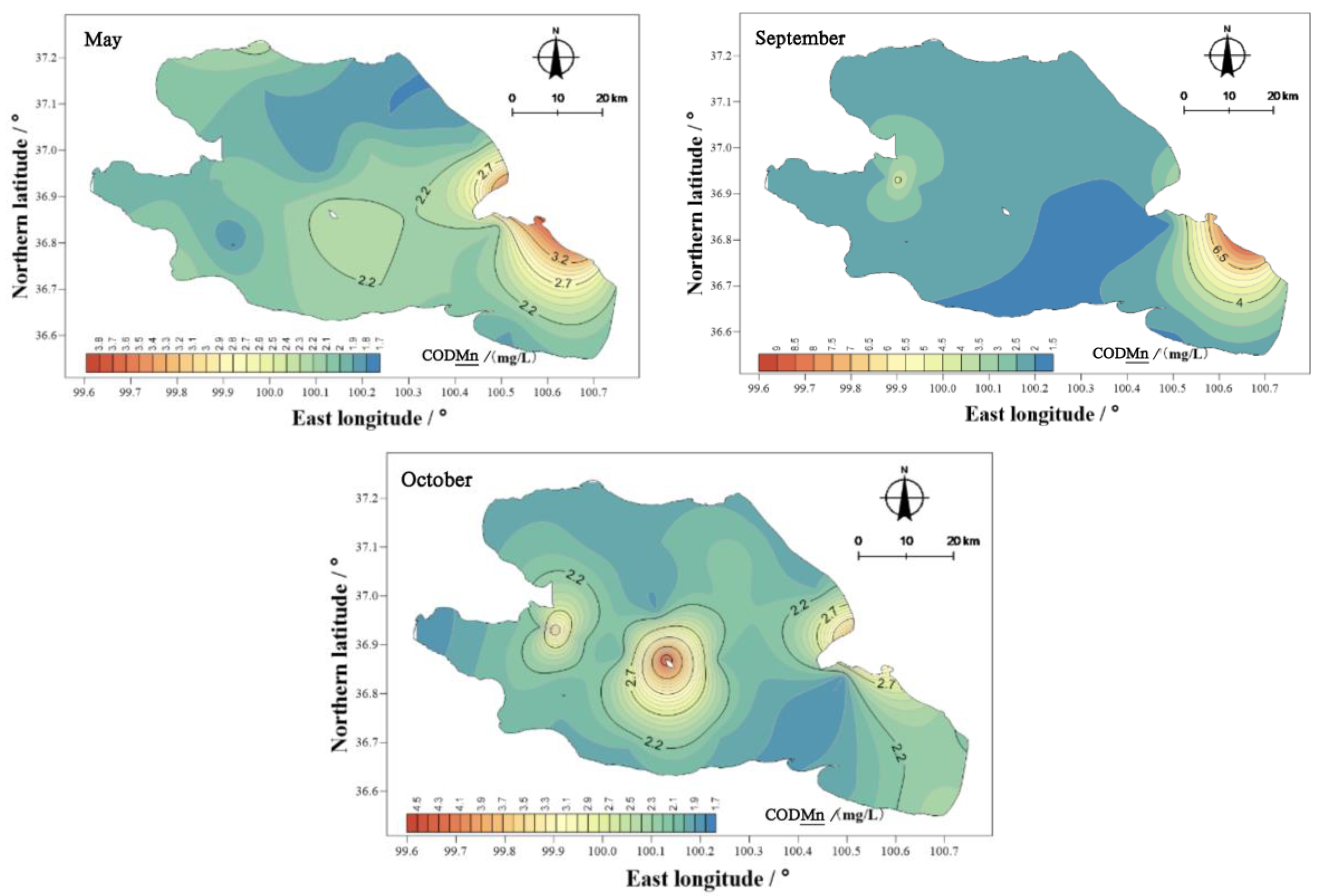
There were obvious differences in the spatial distribution of the permanganate concentration index in Qinghai Lake (
Figure 10). In May, the range of the permanganate concentration index was 1.80–4.50 mg/L, with an average of 2.21 mg/L. In September, the range of the permanganate concentration index was 1.90–14.50 mg/L, with an average of 2.83 mg/L, and in October, the range of the permanganate concentration index was 1.80–4.50 mg/L, with an average of 2.29 mg/L. In May and September, the permanganate index in the east was higher than in the west. In October, the permanganate index of the water body showed three high areas, namely, the center of the lake, the east and the west, and the shore area close to the center of the lake. From the perspective of the spatial distribution characteristics, except for the center of the lake in October, areas with high values appeared close to the shore. Therefore, external inputs may be the reason for the higher concentrations of permanganate in the water body of Qinghai Lake than in other areas.
3.4. Optical Properties of the DOM in Qinghai Lake
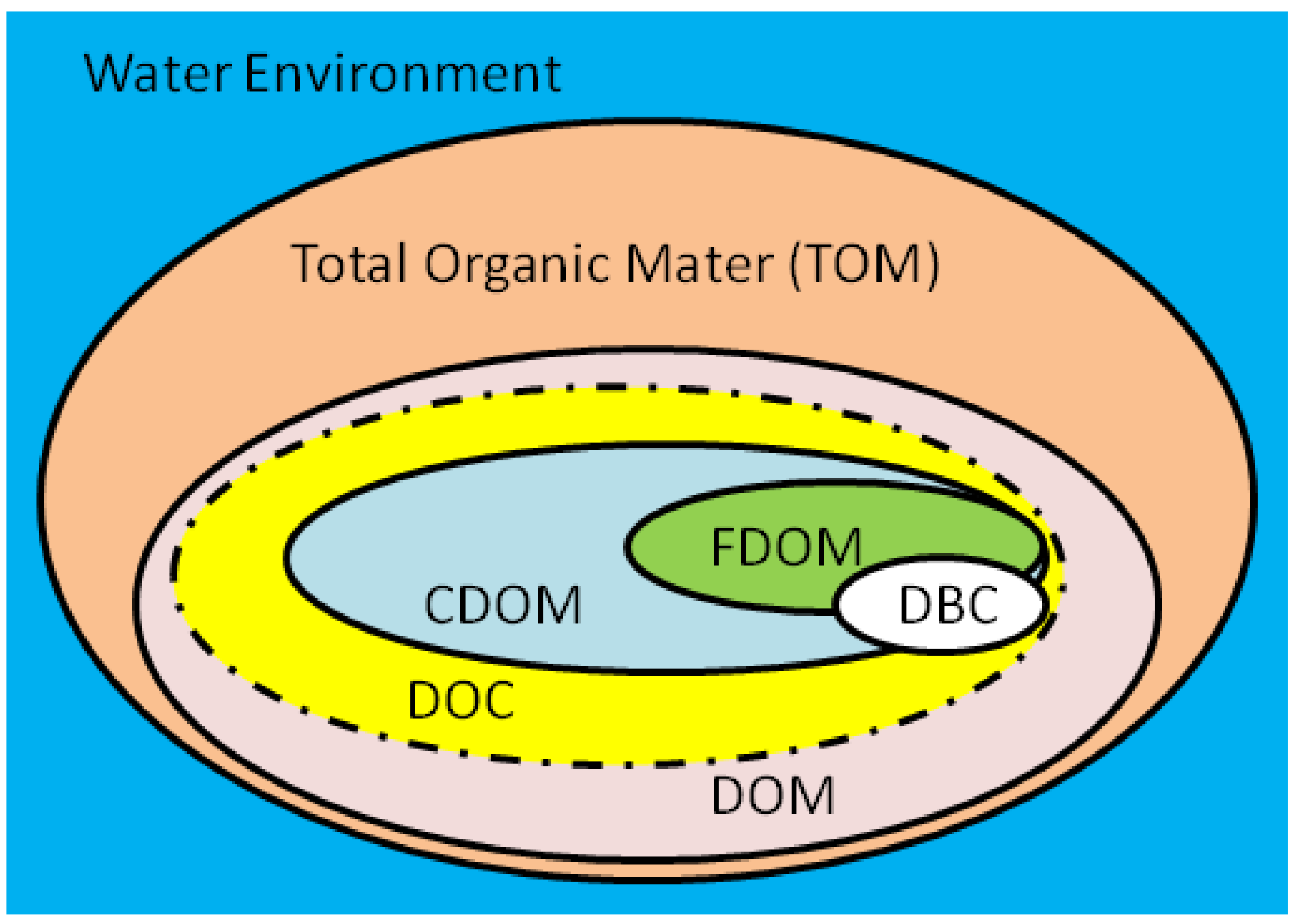
Colored soluble organic matter (CDOM) and fluorescent soluble organic matter (FDOM) are two important indicators of the optical properties of DOM. CDOM is a component of DOM that can absorb effective ultraviolet and photosynthetic radiation; FDOM is a component of DOM that can release fluorescent photons after absorbing light radiation. Therefore, CDOM and FDOM are commonly used to characterize the optical characteristics of DOM.
- (1)
Seasonal and spatial distributions of CDOM
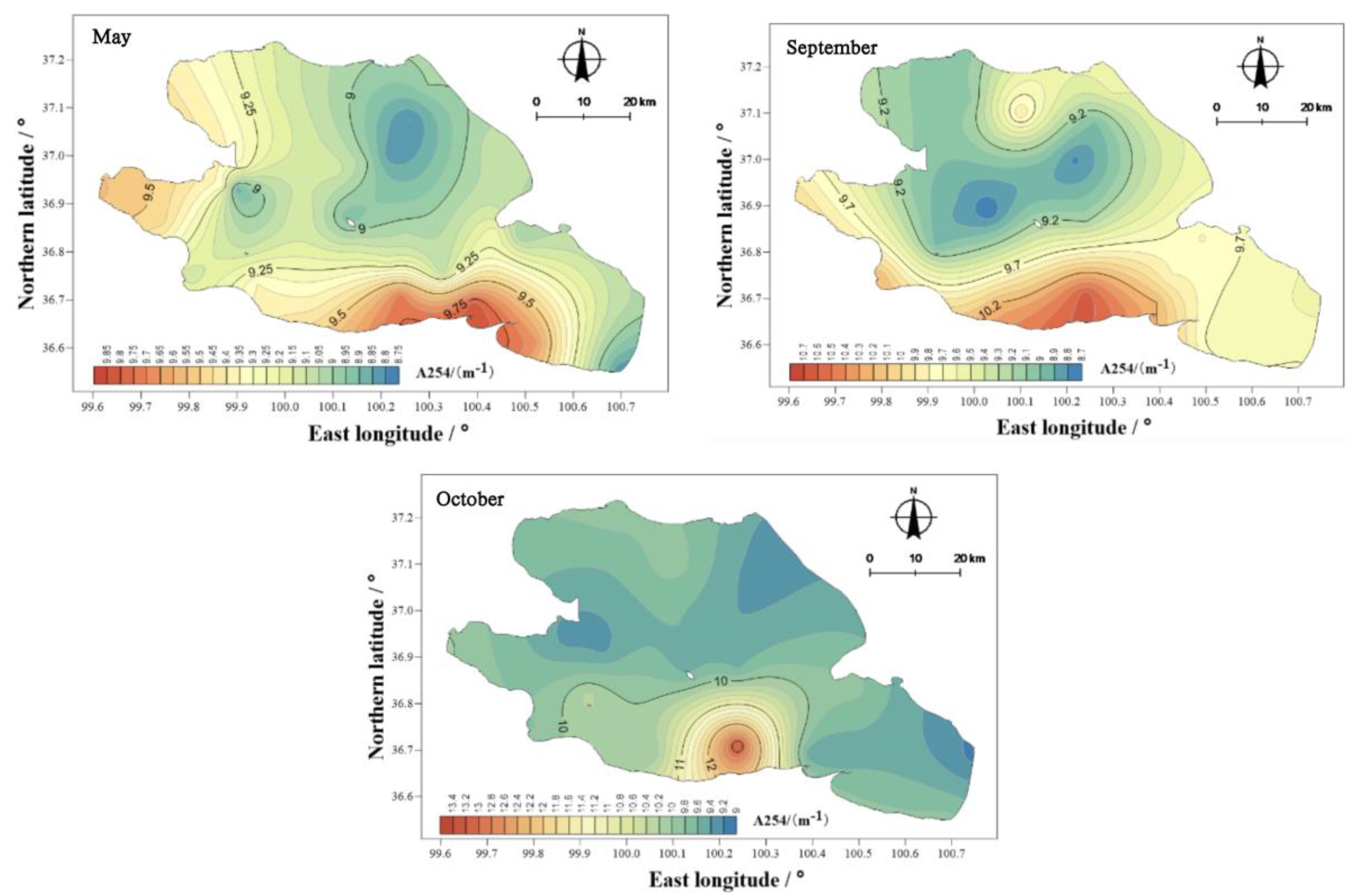
The concentration of CDOM was characterized by its absorption coefficient, a254, at 254 nm. The field monitoring data in May, September, and October 2020 show that (
Figure 11) the values of a254 in the water body of Qinghai Lake were between 8.80 and 13.29 M
−1, with obvious seasonal differences. Among them, the value of a254 in the water in May was between 8.80 and 9.83 M
−1, with an average value of 9.17 M
−1, which was significantly lower than in other seasons (
p < 0.01). In September, the value of a254 in the water body was between 8.73 and 10.64 M
−1, with an average value of 9.48 M
−1. In October, the value of a254 in the water body was between 9.14 and 13.29 M
−1, with an average value of 9.81 M
−1, showing the ranking: October > September > May. The concentration of CDOM in Qinghai Lake not only had obvious seasonal differences, but also showed different spatial differences. The highest values of CDOM at each sampling point in the water body in May, September, and October appeared near Erlangjian in the south of the lake. In addition, the value of CDOM in the water body on the west bank of Qinghai Lake in May was also high.
- (2)
Fluorescence spectra and fluorescence component characteristics of FDOM
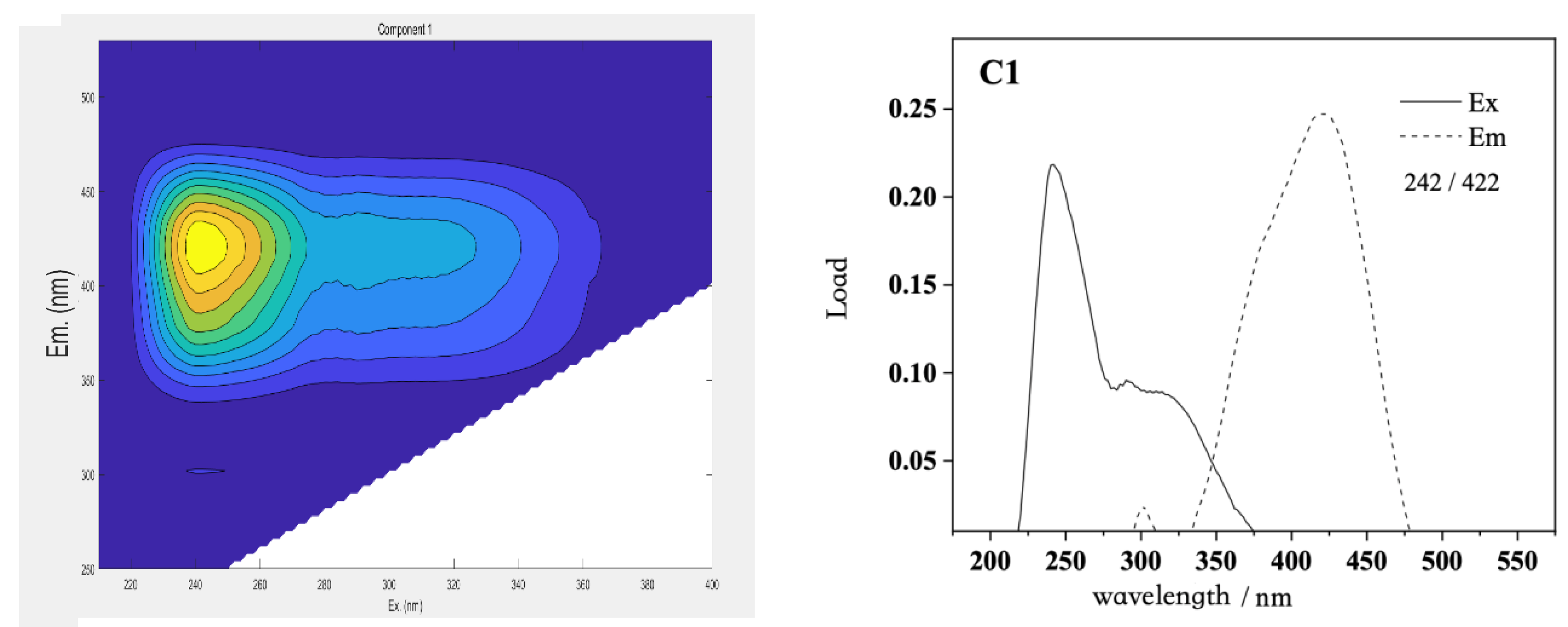
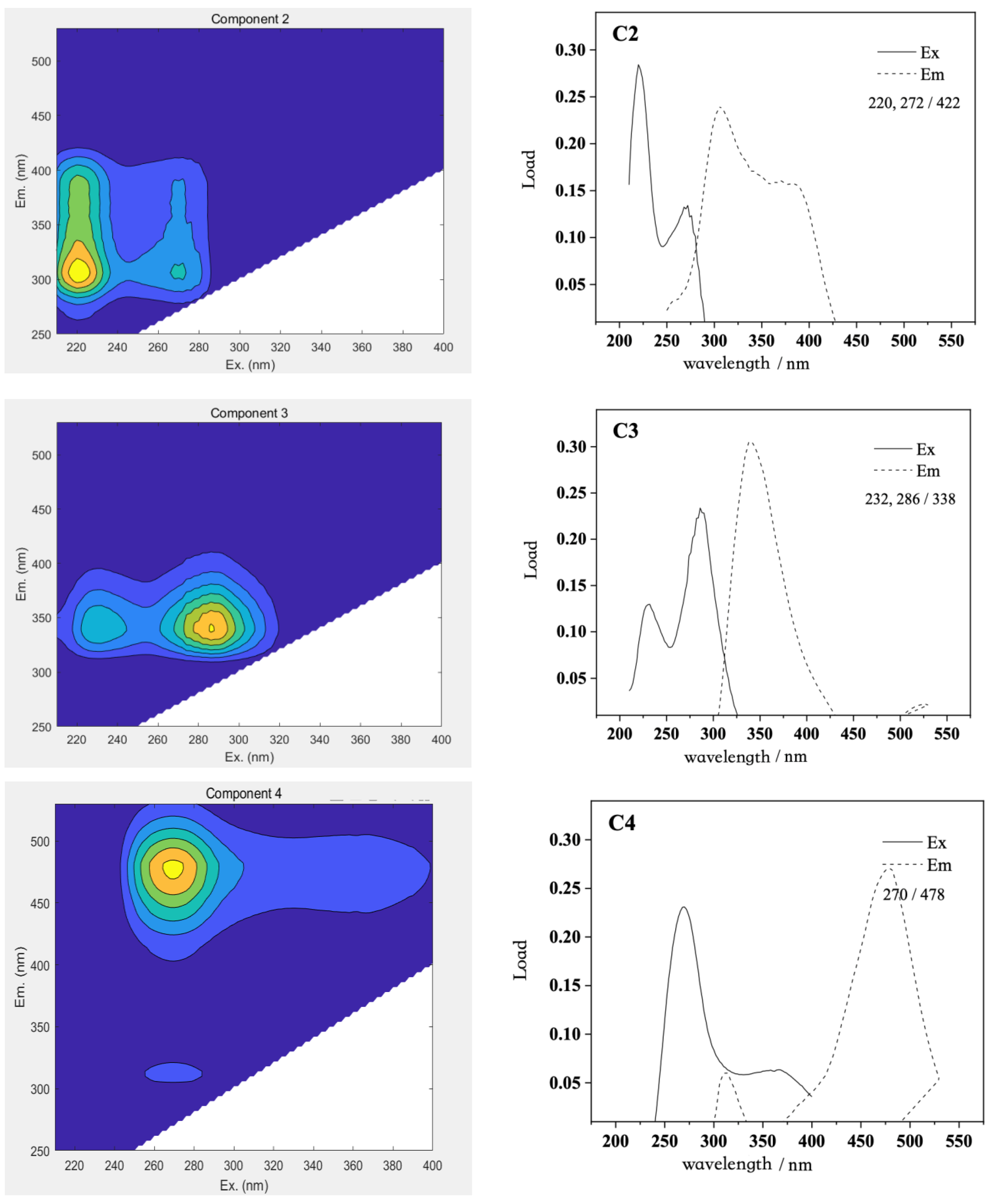
The three-dimensional fluorescence spectrum matrix data of the DOM in Qinghai Lake were analyzed using the PARAFAC model, and four DOM fluorescence components with a single maximum emission wavelength were analyzed. The distribution of the maximum excitation/emission wavelengths of the four fluorescent components and the three-dimensional fluorescence spectra of the principal components are shown in the figure below (
Figure 12).
The excitation wavelength of component C1 was at 242 nm, and the maximum emission wavelength was near 422 nm, which reflected the land-based high-molecular-weight humic acid, which was mainly from the degradation of higher plants or soil leaching. Component C2 had two obvious excitation wavelengths at 220 and 272 nm, and the maximum emission wavelength was 422 nm, which reflected the fluorescence peak of low-molecular-weight fulvic acid, which mainly came from the fluorescence peak formed by biodegradable organic compounds. Component C3 had two obvious excitation wavelengths at 232 and 286 nm, and the maximum emission wavelength was 338 nm. It had binding peaks similar to those of protein and fulvic acid, which have a red shift compared with the conventional tryptophan-like peak. Component C4 had an obvious excitation wavelength at 270 nm and the maximum emission wavelength was 478 nm, reflecting the fluorescence peak formed by the polymer humic acid.
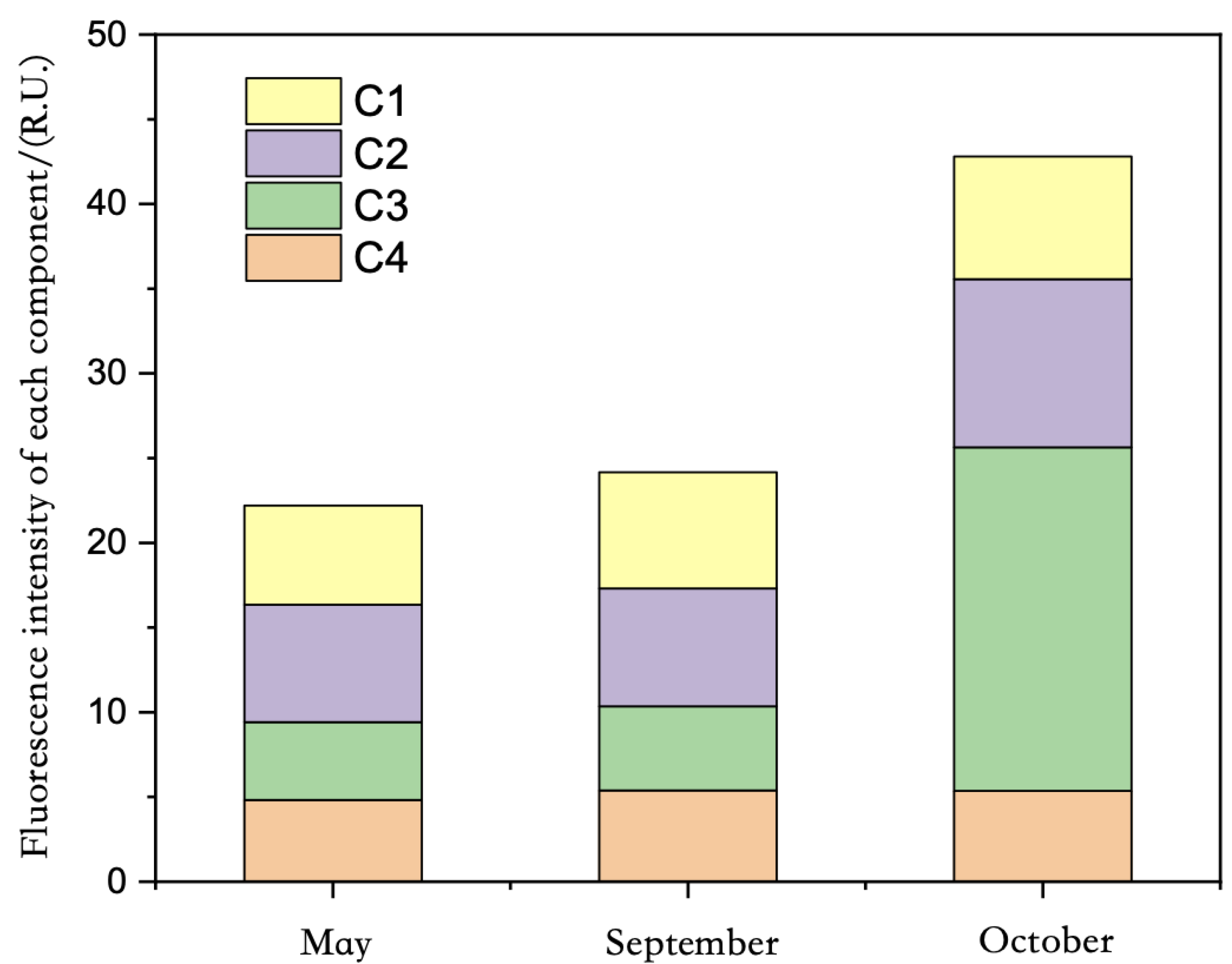
Overall, the proportion of each component in the total fluorescence intensity of FDOM in May and September was C2 > C1 > C4 > C3, and the proportion of each component in the total fluorescence intensity of FDOM in October was C3 > C2 > C1 > C4, which had certain seasonal differences (
Figure 13).
In the three sampling surveys of Qinghai Lake in May, September, and October (
Figure 14), the average total fluorescence intensity of FDOM in the water body of Qinghai Lake was 22.20, 24.17, and 42.80 R.U., showing significant seasonal differences: October > September > May. The spatial distribution of the total fluorescence intensity of FDOM in the water was also different in different seasons. The spatial difference in October was more obvious than those in May and September. However, in the three surveys, the highest value of the total fluorescence intensity of FDOM in the water appeared near Erlangjian in the south of Qinghai Province.
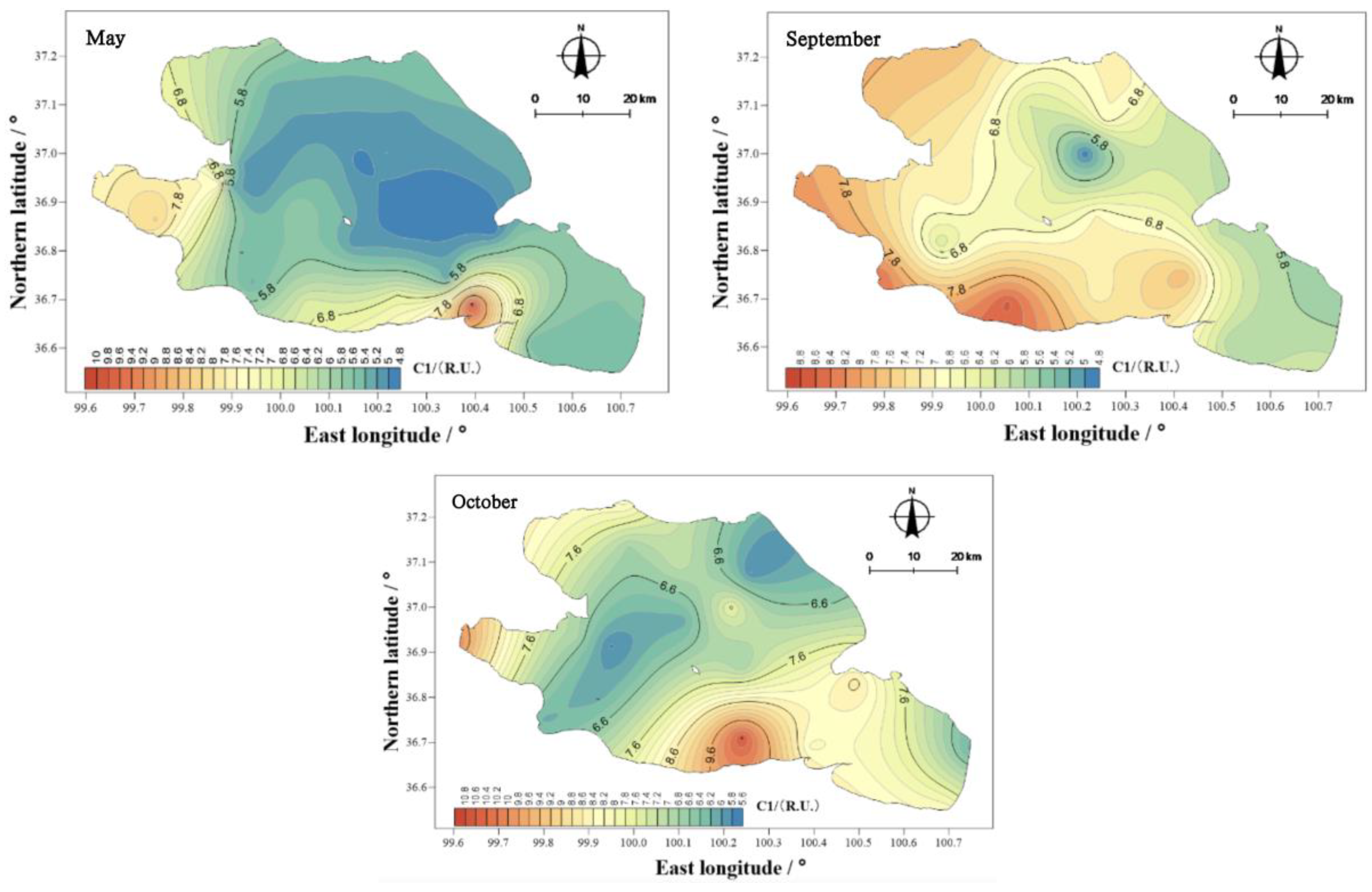
The mean values of component C1 in the overlying water of Qinghai Lake in May, September, and October were 5.85, 6.86, and 7.23 R.U. (
Figure 15), showing the ranking of October > September > May. The spatial distribution characteristics were similar to those of the total amounts, and the spatial differences were obvious. The highest value appeared near Erlangjian in the south of Qinghai, and the lowest value appeared in the center of the lake.
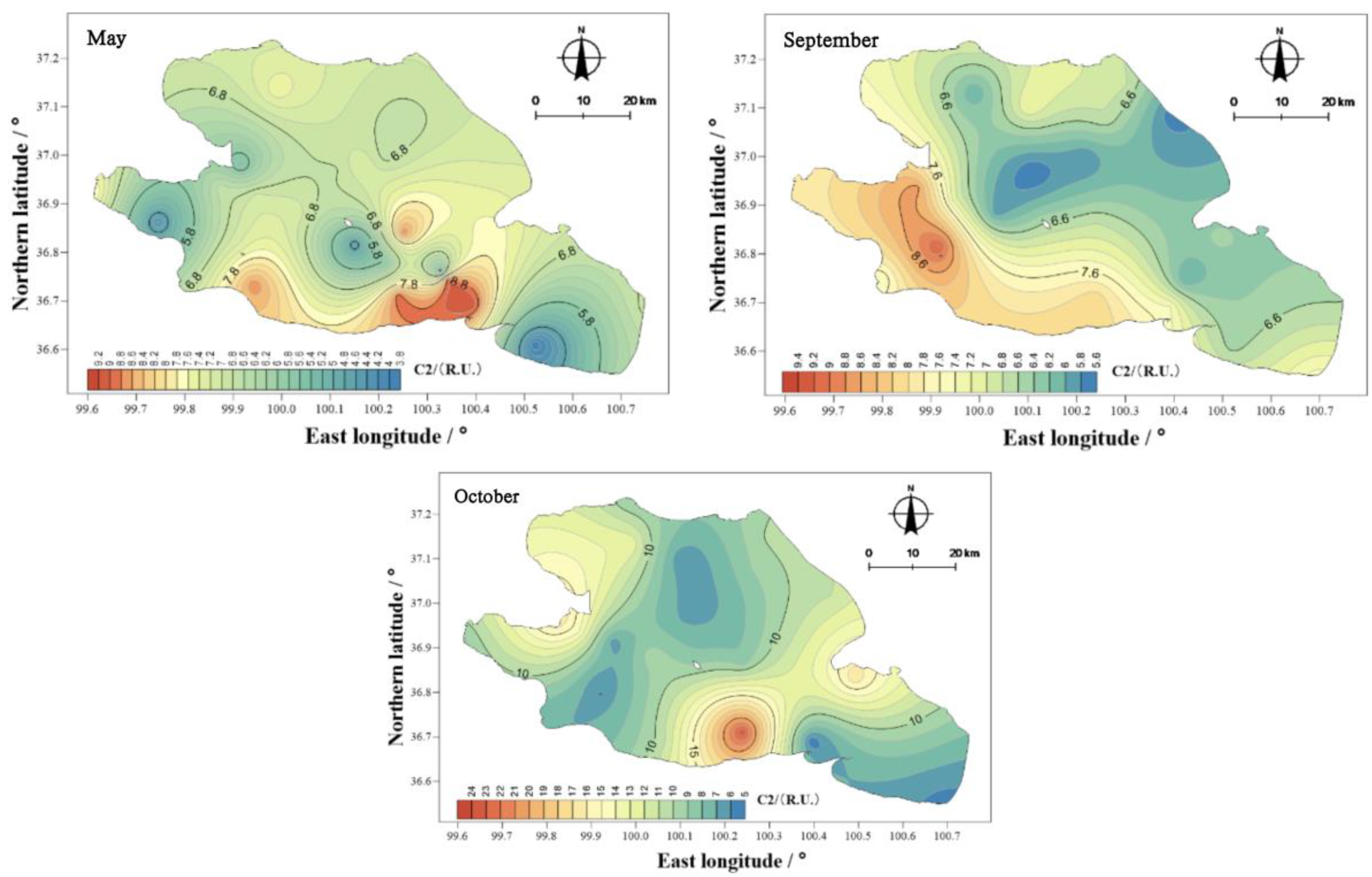
The mean values of component C2 in the water body of Qinghai Lake in May, September, and October were 6.93, 6.96, and 9.93 R.U. (
Figure 16), thus showing the ranking of October > September > May. The spatial distribution characteristics are similar to those of the total amounts, and the spatial differences were obvious. The highest values in May and October appeared in the south of Qinghai, and the highest values in September appeared in the southwest of the lake.
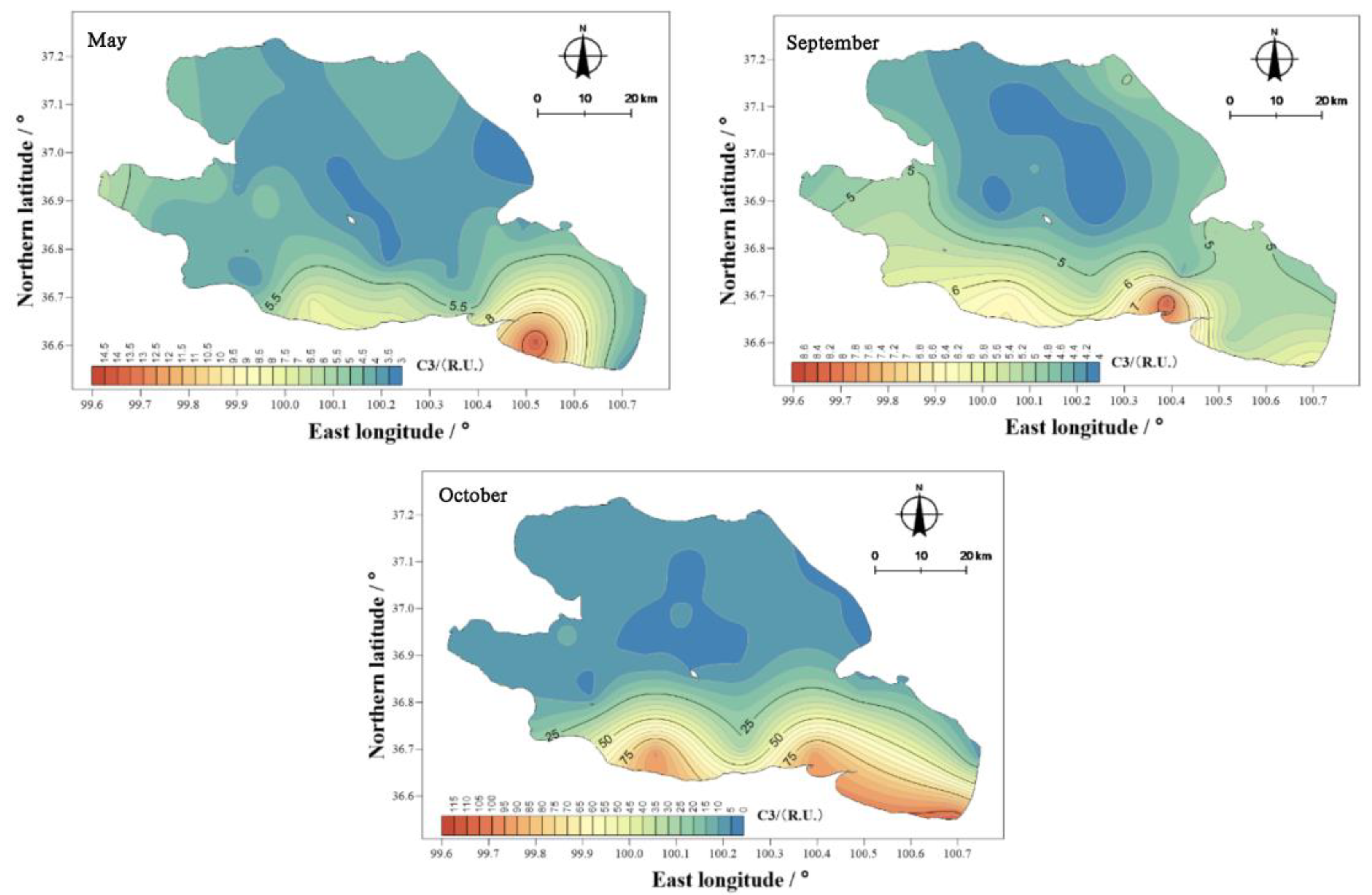
The mean values of component C3 in the water body of Qinghai Lake in May, September, and October were 4.59, 4.96, and 20.28 R.U. (
Figure 17), thus showing the ranking of October > September > May. The spatial distribution characteristics were similar to those of the total amounts, and the spatial differences were obvious. The highest values appeared near Erlangjian in the south of Qinghai and Hunan.
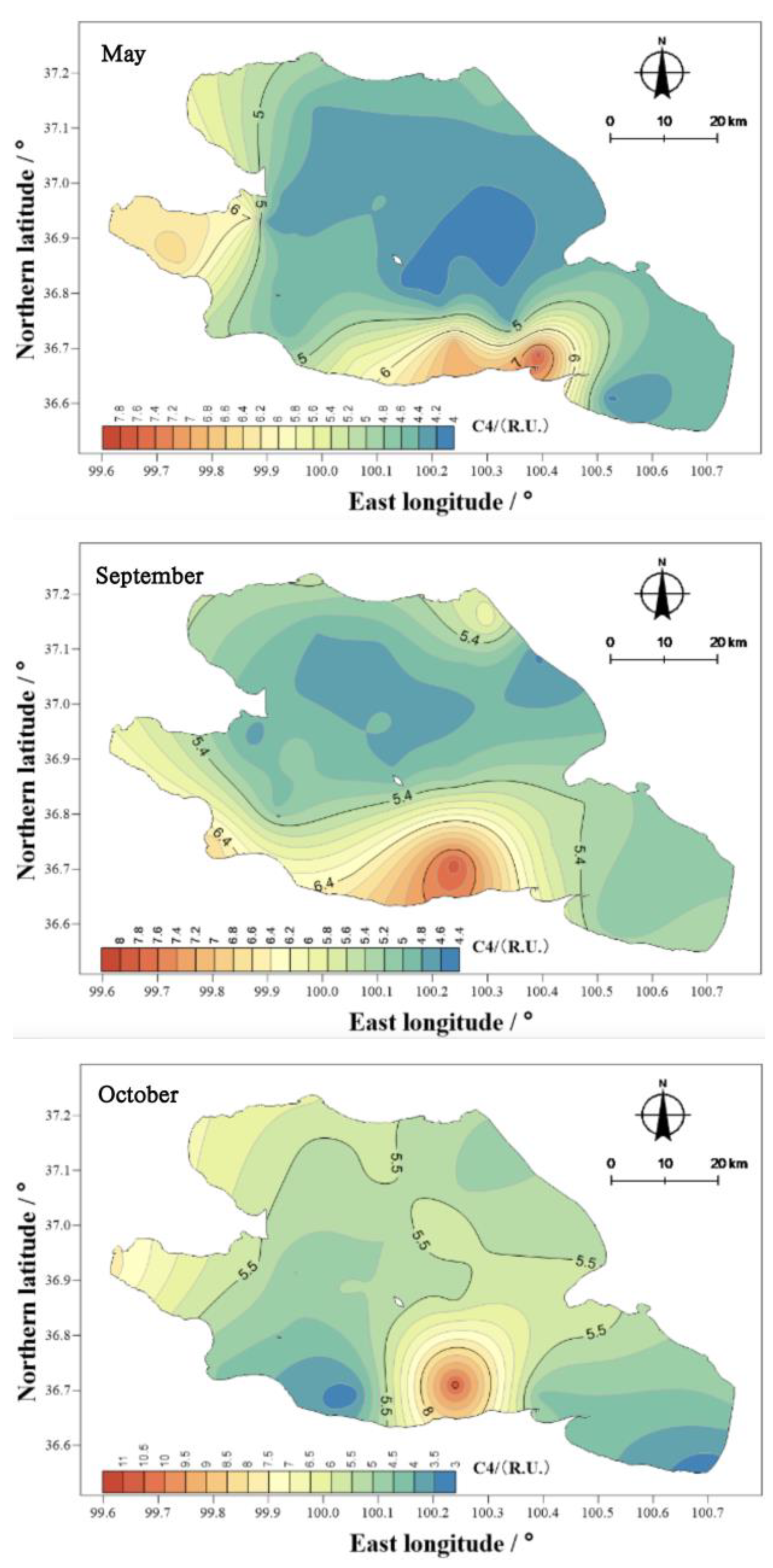
The mean values of component C4 in the water body of Qinghai Lake in May, September, and October were 4.82, 5.39, and 5.36 R.U. (
Figure 18), respectively, thus showing a different ranking from that of the other components, that is, September > October > May. The spatial distribution characteristics were similar to those of the total amounts, and the spatial differences were obvious. The highest values appeared near Erlangjian in the south of Qinghai and Hunan.