Improvement of Log Reduction Values Design Equations for Helminth Egg Management in Recycled Water
Abstract
:1. Introduction
- Quantification of HE concentrations in both the sewage and recycled water of the relevant treatment process in the WWTP (measured concentrations needed to be greater than the detection limit), and
- The associated HRT.
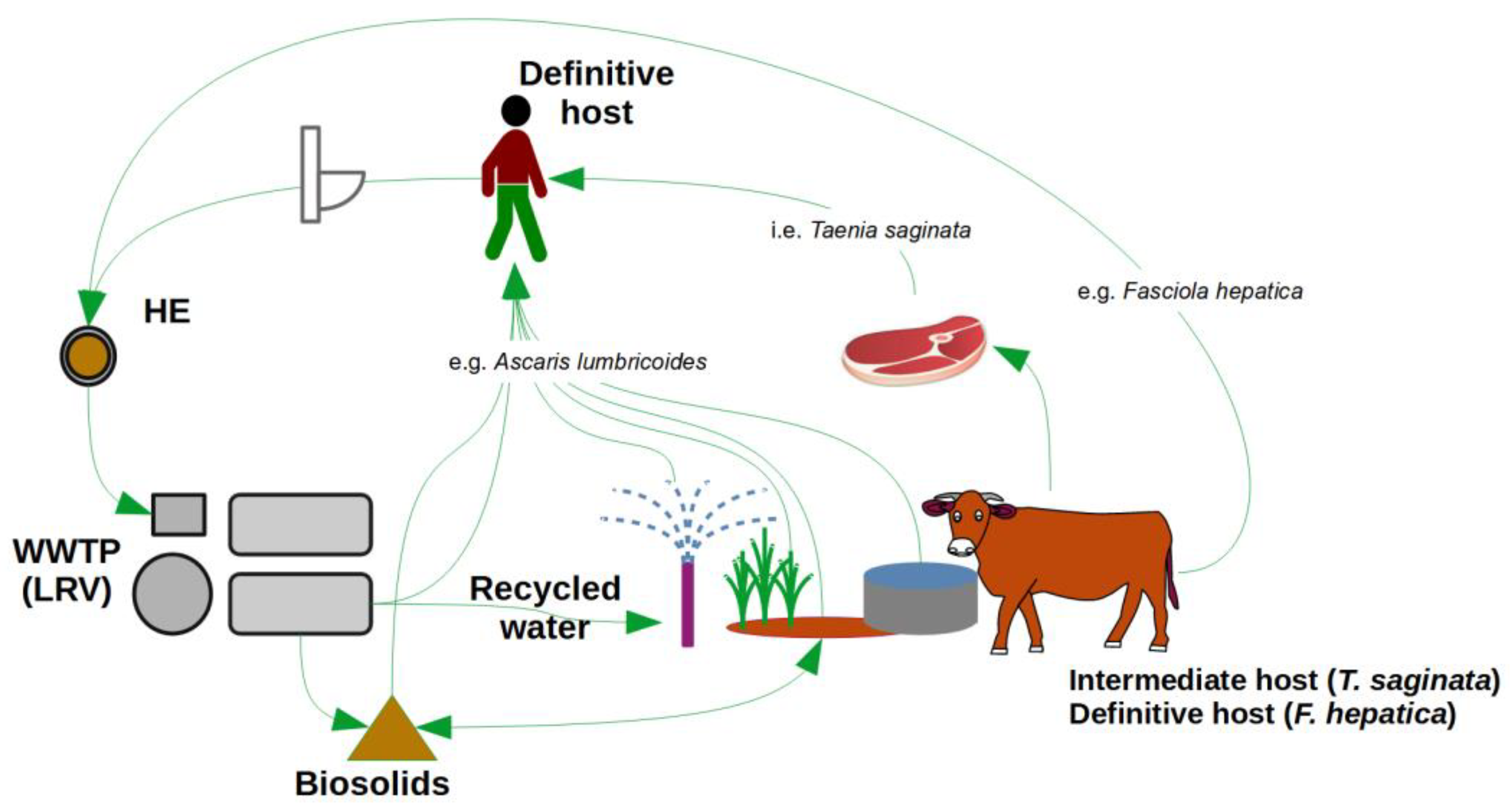
2. Helminth Management
2.1. Helminth Infection
2.2. Helminth Egg Loads Entering the Sewer
2.2.1. Mass Drug Administration
2.2.2. Population Migration and Movement
2.3. Controlling HE Movement into Non-Endemic Regions
2.4. Helminth Egg Removal from Sewage
2.4.1. Removal of HE via Activated Sludge Plants and Lagoons
2.4.2. Removal of HE via Sand Filtration
2.4.3. Other Filtration Approaches
3. Guidelines for Helminth Eggs in Recycled Water
3.1. International Guidelines for Human Health
3.2. Australian Guidelines for Helminth Management in Sewage
3.3. Animal Health
4. Limitations to the LRV Design Equation
4.1. Detection Limits and Recovery of HE
4.2. Data Limitation
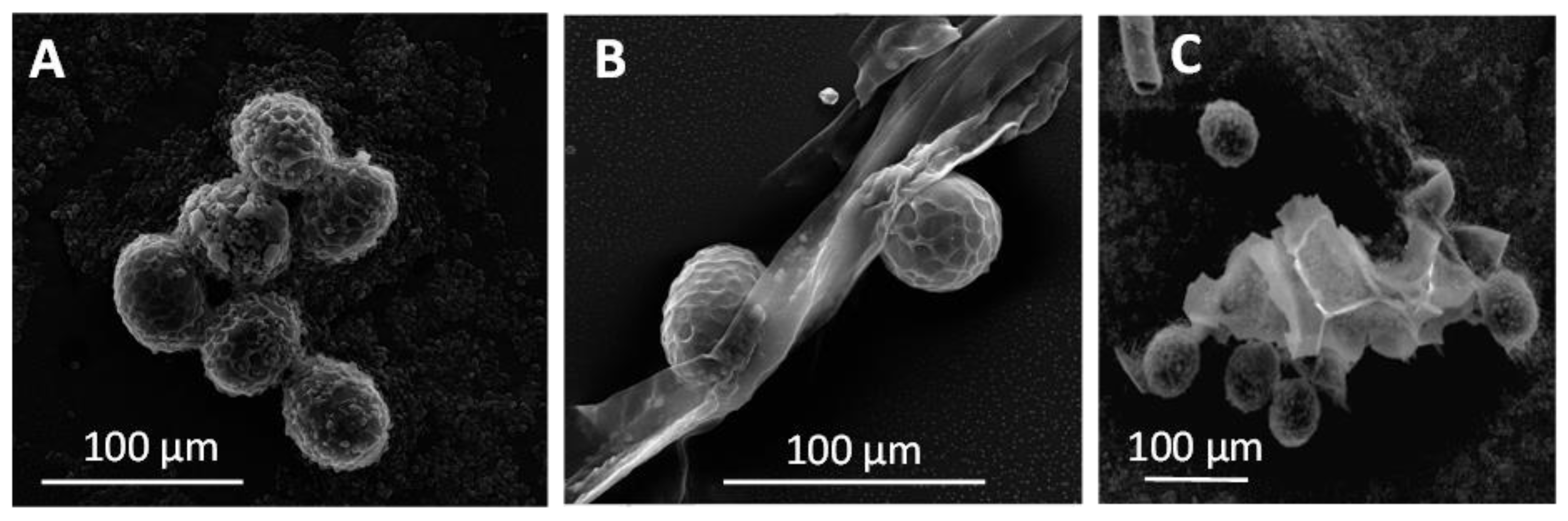
4.3. Analytical Methods Relying on Visual Identification
4.4. ASP and Lagoon Maintenance and Management
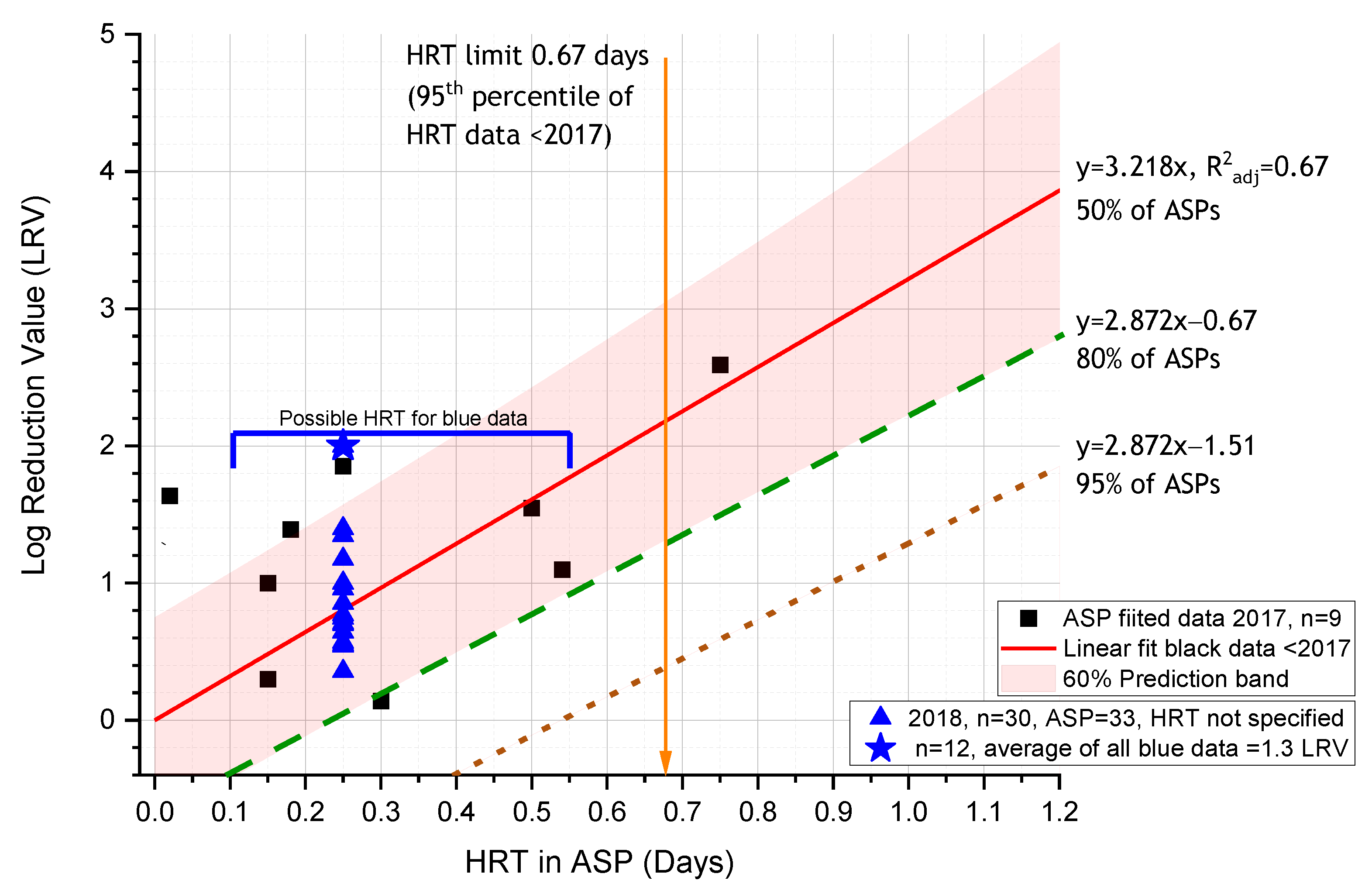
4.4.1. ASP Removal of HE
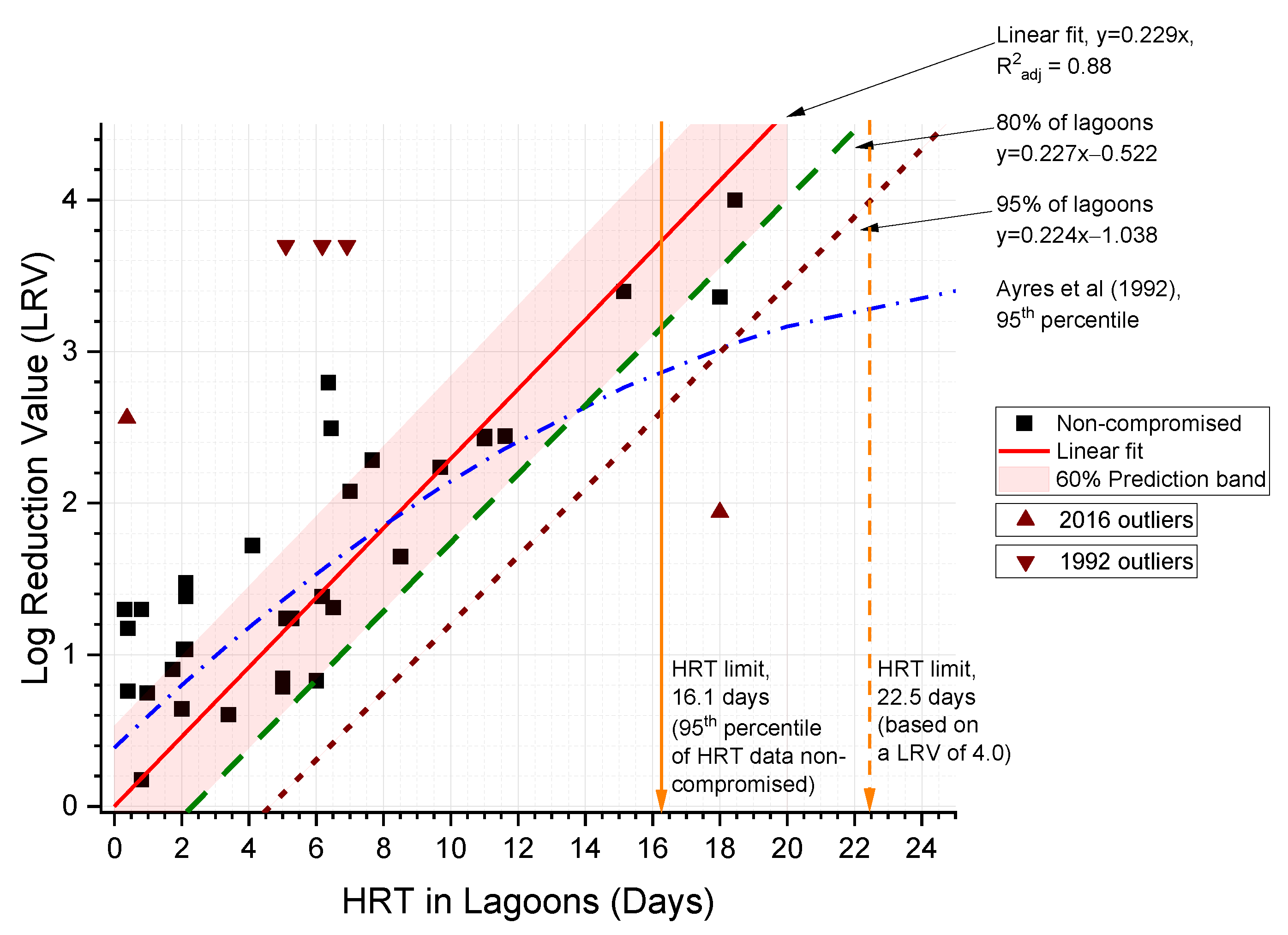
4.4.2. Lagoon Removal of HE
4.4.3. Conceptual Method for LRV Estimates Considering ASP and Lagoon Maintenance and Management
- Removal of solids with HE attached (e.g., ASP),
- Settling, as HE have a greater density than water (e.g., lagoon system), and
- Filtration, as HE are larger than most pathogens with a diameter > 25 µm.
4.5. Viability of Helminth Eggs
4.5.1. Conventional Methods
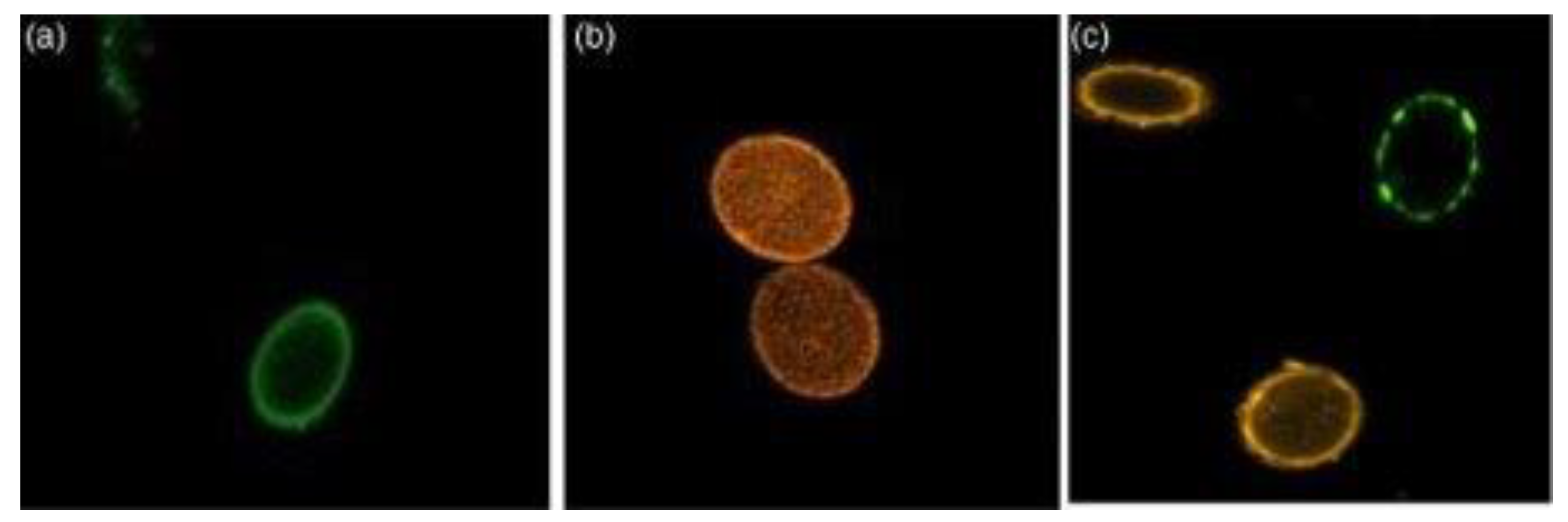
Microscope-Based Methods
Vital Stains
BacLight Bacterial Viability Stain
4.5.2. PCR-Based Methods
4.6. Surrogates
4.7. Sedimentation of Helminth Eggs
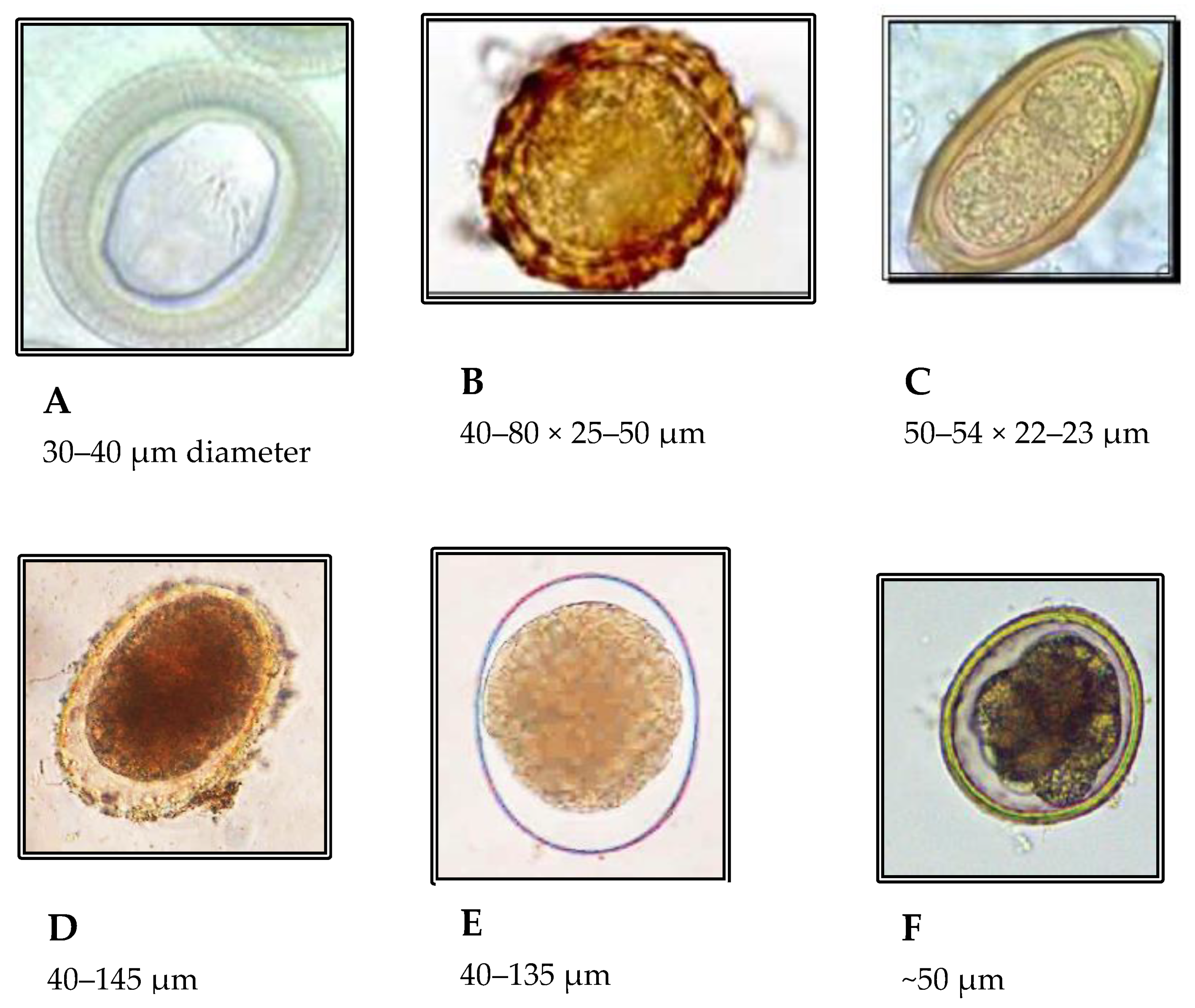
4.8. Helminth Species of Interest
4.9. Reliance on an Assumed Sewage Concentration of HEs
5. Research Required to Improve the LRV Design Equation
- A cost-effective method for the detection of viable HE concentrations is required with detection levels sufficient to estimate LRV of each relevant treatment process within WWTPs. Such a method should improve detection limits, improve recovery rates, decrease variability and assess viability, with a focus on cost-effective routine analysis.
- Quantification of the mechanisms and rate of HE removal through conventional sewage treatment processes under a range of operating conditions, with a view to deriving an improved LRV design relationship between key operational parameters and HE removal.
- Investigate the application of suitable surrogates for viable HE for application in estimating the settling velocity and impact of settling rates in controlled conditions and field dosing experiments.
- Challenge testing of lagoon systems with HE or a suitable surrogate to validate the LRV design equation. However, owing to the costs and complexity involved, it will be difficult to carry out experiments at the plant-scale. Consequently, bench-top or scaled-down pilot plants, that reflect full-scale WWTP function, may provide significant savings in costs and time.
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Radcliffe, J.C.; Page, D. Water reuse and recycling in Australia—History, current situation and future perspectives. Water Cycle 2020, 1, 19–40. [Google Scholar] [CrossRef]
- WRC. California WateReuse Action Plan. Available online: https://watereuse.org/wp-content/uploads/2019/07/WateReuse-CA-Action-Plan_July-2019_r5-2.pdf (accessed on 18 August 2020).
- Madaeni, S. The application of membrane technology for water disinfection. Water Res. 1999, 33, 301–308. [Google Scholar] [CrossRef]
- Bennett, A. Pathogen Removal from Water—Technologies and Techniques. Available online: https://www.filtsep.com/water-and-wastewater/features/pathogen-removal-from-water-technologies-and (accessed on 12 July 2020).
- WHO. Soil-Transmitted Helminth Infections [WWW Document]. Available online: https://www.who.int/news-room/fact-sheets/detail/soil-transmitted-helminth-infections (accessed on 18 August 2020).
- WHO. Crossing the Billion. Preventive Chemotherapy for Neglected Tropical Diseases; Department of Control of Neglected Tropical Diseases: Geneva, Switzerland, 2017; pp. 1–55. [Google Scholar]
- Natural Resource Management Ministerial Council; Environment Protection and Heritage Council; National Health and Medical Research Council. Australian Guidelines for Water Recycling: Managing Health and Environmental Risks. Phase 1. National Water Quality Management Strategy 21; Environment Protection and Heritage Council, the National Health and Medical Research Council and the Natural Resource Management Ministerial Council: Canberra, NSW, Australia, 2006. [Google Scholar]
- Yaya-Beas, R.-E.; Cadillo-La-Torre, E.-A.; Kujawa-Roeleveld, K.; van Lier, J.B.; Zeeman, G. Presence of helminth eggs in domestic wastewater and its removal at low temperature UASB reactors in Peruvian highlands. Water Res. 2016, 90, 286–293. [Google Scholar] [CrossRef]
- WHO. Guidelines for the Safe Use of Wastewater, Excreta and Greywater. Wastewater Use in Agriculture; World Health Organization: Geneva, Switzerland, 2006; Available online: https://www.who.int/water_sanitation_health/publications/gsuweg2/en (accessed on 19 July 2020).
- Gordon, C.A.; Kurscheid, J.; Jones, M.K.; Gray, D.J.; McManus, D.P. Soil-transmitted helminths in tropical Australia and Asia. Trop. Med. Infect. Dis. 2017, 2, 56. [Google Scholar] [CrossRef] [Green Version]
- Ravindran, V.B.; Surapaneni, A.; Crosbie, N.D.; Schmidt, J.; Shahsavari, E.; Haleyur, N.; Soni, S.K.; Ball, A.S. A modified approach to recover and enumerate Ascaris ova in wastewater and sludge. PLoS Negl. Trop. Dis. 2019, 13, e0007020. [Google Scholar] [CrossRef] [Green Version]
- Stevens, D.P.; Surapaneni, A.; Thodupunuri, R.; O’Connor, N.A.; Smith, D. Helminth log reduction values for recycling water from sewage for the protection of human and stock health. Water Res. 2017, 125, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Wharton, D.A. The production and functional morphology of helminth egg-shells. Parasitology 1983, 86, 85–97. [Google Scholar] [CrossRef]
- Guadagnini, R.A.; dos Santos, L.U.; Franco, R.M.B.; Guimarães, J.R. Inactivation of bacteria and helminth in wastewater treatment plant effluent using oxidation processes. Water Sci. Technol. 2013, 68, 1825–1829. [Google Scholar] [CrossRef] [PubMed]
- Ayres, R.; Alabaster, G.; Mara, D.; Lee, D. A design equation for human intestinal nematode egg removal in waste stabilization ponds. Water Res. 1992, 26, 863–865. [Google Scholar] [CrossRef]
- Zacharia, A.; Ahmada, W.; Outwater, A.H.; Ngasala, B.; Van Deun, R. Evaluation of occurrence, concentration, and removal of pathogenic parasites and fecal coliforms in three waste stabilization pond systems in Tanzania. Sci. World J. 2019, 2019, 3415617. [Google Scholar] [CrossRef] [PubMed]
- Gazzinelli, A.; Correa-Oliveira, R.; Yang, G.-J.; Boatin, B.A.; Kloos, H. A research agenda for helminth diseases of humans: Social Ecology, environmental determinants, and health systems. PLoS Negl. Trop. Dis. 2012, 6, e1603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amoah, I.D.; Singh, G.; Stenström, T.A.; Reddy, P. Detection and quantification of soil-transmitted helminths in environmental samples: A review of current state-of-the-art and future perspectives. Acta Trop. 2017, 169, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Gyawali, P. Infectious helminth ova in wastewater and sludge: A review on public health issues and current quantification practices. Water Sci. Technol. 2018, 77, 1048–1061. [Google Scholar] [CrossRef] [PubMed]
- Amoah, I.D.; Reddy, P.; Seidu, R.; Stenström, T.A. Removal of helminth eggs by centralized and decentralized wastewater treatment plants in South Africa and Lesotho: Health implications for direct and indirect exposure to the effluents. Environ. Sci. Pollut. Res. 2018, 25, 12883–12895. [Google Scholar] [CrossRef] [Green Version]
- Verbyla, M.E.; Oakley, S.M.; Lizima, L.A.; Zhang, J.; Iriarte, M.; Tejada-Martinez, A.E.; Mihelcic, J.R. Taenia eggs in a stabilization pond system with poor hydraulics: Concern for human cysticercosis? Water Sci. Technol. 2013, 68, 2698–2703. [Google Scholar] [CrossRef]
- Navarro, I.; Jiménez, B.; Lucario, S.; Cifuentes, E. Application of Helminth ova infection dose curve to estimate the risks associated with biosolid application on soil. J. Water Health 2008, 7, 31–44. [Google Scholar] [CrossRef]
- Trang, D.T.; Mølbak, K.; Cam, P.D.; Dalsgaard, A. Helminth infections among people using wastewater and human excreta in peri-urban agriculture and aquaculture in Hanoi, Vietnam. Trop. Med. Int. Health 2007, 12, 82–90. [Google Scholar] [CrossRef]
- O’Sullivan, A.; Sheffrin, S.M. Economics: Principles in Action; Prentice Hall: Needham, MA, USA, 2003. [Google Scholar]
- Webster, J.P.; Molyneux, D.H.; Hotez, P.J.; Fenwick, A. The contribution of mass drug administration to global health: Past, present and future. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130434. [Google Scholar] [CrossRef] [Green Version]
- LCNTDR. The London Declaration. London Centre for Neglected Tropical Disease Research (LCNTDR) [WWW Document]. Available online: https://www.londonntd.org/about-us/the-rationale-for-the-lcntdr/the-london-declaration (accessed on 19 July 2020).
- WHO. Eliminating Soil-Transmitted Helminthiases as a Public Health Problem in Children. Progress Report 2001−2010 and Strategic Plan 2011−2020; WHO: Geneva, Switzerland, 2012; pp. 1–78. [Google Scholar]
- Dunn, J.C.; Turner, H.C.; Tun, A.; Anderson, R.M. Epidemiological surveys of, and research on, soil-transmitted helminths in Southeast Asia: A systematic review. Parasites Vectors 2016, 9, 31. [Google Scholar] [CrossRef] [Green Version]
- Musuva, R.M.; Matey, E.; Masaku, J.; Odhiambo, G.; Mwende, F.; Thuita, I.; Kihara, J.; Njomo, D. Lessons from implementing mass drug administration for soil transmitted helminths among pre-school aged children during school based deworming program at the Kenyan coast. BMC Public Health 2017, 17, 575. [Google Scholar] [CrossRef] [Green Version]
- Bah, Y.M.; Bah, M.S.; Paye, J.; Conteh, A.; Saffa, S.; Tia, A.; Sonnie, M.; Veinoglou, A.; Amon, J.J.; Hodges, M.H.; et al. Soil-transmitted helminth infection in school age children in Sierra Leone after a decade of preventive chemotherapy interventions. Infect. Dis. Poverty 2019, 8, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- WHO. Research Priorities for Helminth Infections: Technical Report of the TDR Disease Reference Group on Helminth Infections; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Sunish, I.P.; Rajendran, R.; Munirathinam, A.; Kalimuthu, M.; Kumar, V.A.; Nagaraj, J.; Tyagi, B.K. Impact on prevalence of intestinal helminth infection in school children administered with seven annual rounds of diethyl carbamazine (DEC) with albendazole. Indian J. Med. Res. 2015, 141, 330–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ABS. Australian Demographic Statistics: Australian Bureau of Statistics. Available online: https://www.abs.gov.au/AUSSTATS/abs@.nsf/mf/3412.0.2020. (accessed on 19 July 2020).
- Collender, P.A.; Kirby, A.E.; Addiss, D.G.; Freeman, M.C.; Remais, J.V. Methods for quantification of soil-transmitted helminths in environmental media: Current techniques and recent advances. Trends Parasitol. 2015, 31, 625–639. [Google Scholar] [CrossRef] [Green Version]
- De Silva, S.; Saykao, P.; Kelly, H.; MacIntyre, C.R.; Ryan, N.; Leydon, J.; Biggs, B.A. Chronic Strongyloides stercoralis infection in Laotian immigrants and refugees 7-20 years after resettlement in Australia. Epidemiol. Infect. 2002, 128, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Caruana, S.R.; Kelly, H.A.; Ngeow, J.Y.Y.; Ryan, N.J.; Bennett, C.M.; Chea, L.; Nuon, S.; Bak, N.; Skull, S.A.; Biggs, B.A. Undiagnosed and potentially lethal parasite infections among immigrants and refugees in Australia. J. Travel Med. 2006, 13, 233–239. [Google Scholar] [CrossRef] [PubMed]
- DIBP. Health Examinations; Department of Immigration and Border Protection, Australian Government: Canberra, NSW, Australia. Available online: http://www.border.gov.au/Trav/Visa/Heal/meeting-the-health-requirement/health-examinations (accessed on 8 February 2017).
- Hanieh, S.; Ryan, N.; Biggs, B.A. Assessing enteric helminths in refugees, asylum seekers and new migrants. Microbiol. Aust. 2016, 37, 15–19. [Google Scholar] [CrossRef] [Green Version]
- Nissan, B.; Lachish, T.; Schwartz, E. The effectiveness of empirical anti-parasitic treatment in returning travellers with persistent abdominal symptoms. J. Travel Med. 2017, 25, tax083. [Google Scholar] [CrossRef]
- Rahmanian, H.; MacFarlane, A.C.; Rowland, K.E.; Einsiedel, L.J.; Neuhaus, S.J. Seroprevalence of Strongyloides stercoralis in a South Australian Vietnam veteran cohort. Aust. N. Zealand J. Public Health 2015, 39, 331–335. [Google Scholar] [CrossRef]
- Speare, R.; Miller, A.; Page, W.A. Strongyloidiasis: A case for notification in Australia? Med. J. Aust. 2015, 202, 523–524. [Google Scholar] [CrossRef]
- Schwartzbrod, J.; Stien, J.L.; Bouhoum, K.; Baleux, B. Impact of wastewater treatment on helminth eggs. Water Sci. Technol. 1989, 21, 295–297. [Google Scholar] [CrossRef]
- Tyagi, V.K.; Sahoo, B.K.; Khursheed, A.; Kazmi, A.A.; Ahmad, Z.; Chopra, A.K. Fate of coliforms and pathogenic parasite in four full-scale sewage treatment systems in India. Environ. Monit. Assess. 2011, 181, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Hatam-Nahavandi, K.; Mahvi, A.H.; Mohebali, M.; Keshavarz, H.; Mobedi, I.; Rezaeian, M. Detection of parasitic particles in domestic and urban wastewaters and assessment of removal efficiency of treatment plants in Tehran, Iran. J. Environ. Health Sci. Eng. 2015, 13, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayres, R.M.; Mara, D.D. Analysis of Wastewater for Use in Agriculture—A Laboratory Manual of Parasitological and Bacteriological Techniques; WHO: Geneva, Switzerland, 1996. [Google Scholar]
- Newton, W.L.; Bennett, H.J.; Figgat, W.B. Observations on the effects of various sewage treatment processes upon eggs of taenia saginata1. Am. J. Epidemiol. 1949, 49, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Landa, H.; Capella, A.; Jiménez, B. Particle size distribution in an efluent from an advanced primary treatment and its removal during filtration. Water Sci. Technol. 1997, 36, 159–165. [Google Scholar] [CrossRef]
- Jiménez-Cisneros, B.E.; Maya-Rendon, C.; Salgado-Velázquez, G. The elimination of helminth ova, faecal coliforms, Salmonella and protozoan cysts by various physicochemical processes in wastewater and sludge. Water Sci. Technol. 2001, 43, 179–182. [Google Scholar] [CrossRef]
- Jimenez, B. Helminth ova removal from wastewater for agriculture and aquaculture reuse. Water Sci. Technol. 2007, 55, 485–493. [Google Scholar] [CrossRef]
- Stott, R. Fate and behaviour of parasites in wastewater treatment systems A2. In Handbook of Water and Wastewater Microbiology; Mara, D., Horan, N., Eds.; Academic Press: London, UK, 2003. [Google Scholar]
- Dufour, A.; Snozzi, M.; Koster, W.; Bartram, J.; Ronchi, E.; Fewtrell, L.; World Health Organization. Assessing Microbial Safety of Drinking Water: Improving Approaches and Methods; Al Dufour, M., Ed.; WHO Drinking-water Quality Series; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Düppenbecker, B.; Maya, C.; Kneidl, S.; Meza, C.; Lucario, E.; Echagüe, V.; Cornel, P. Removal of Helminth Eggs by Surface Filtration. In Proceedings of the 9th International Conference on Water Reuse, Windhoek, Namibia, 27–31 October 2013; p. 31. [Google Scholar]
- CDC. CDC—DPDx—Ascariasis WWW Document. Available online: https://www.cdc.gov/dpdx/ascariasis/index.html (accessed on 19 July 2020).
- Sanz, J.; Strube, R.; Quinzaños, S.; Dahl, C.P.; Montoliu, I.; Gracenea, M.; Mujeriego Sahuquillo, R. Helminth eggs removal in water reclamation: Disc filtration as an effective barrier. In Proceedings of the International Desalination Association World Congress, International Desalination Association, Dubai, United Arabic Emirates, 7–12 November 2009; pp. 1–14. [Google Scholar]
- Quinzaños, S.; Dahl, C.; Strube, R.; Mujeriego, R. Helminth eggs removal by microscreening for water reclamation and reuse. Water Sci. Technol. 2008, 57, 715–720. [Google Scholar] [CrossRef]
- Maya, C.; Jimenez, B.; Schwartzbrod, J. Comparison of Techniques for the Detection of Helminth Ova in Drinking Water and Wastewater. Water Environ. Res. 2006, 78, 118–124. [Google Scholar] [CrossRef]
- Feachem, R.G.; Bradley, D.J.; Garelick, H.; Mara, D.D. Sanitation and Disease: Health Aspects of Excreta and Wastewater Management; World Bank Studies in Water Supply and Sanitation; John Wiley Sons: New York, NY, USA, 1983; Volume 3. [Google Scholar]
- Kiermeier, A.; Hamilton, D.; Pointon, A.J.M.R.A. Quantitative risk assessment for human Taenia saginata infection from consumption of Australian beef. Microb. Risk Anal. 2019, 12, 1–10. [Google Scholar] [CrossRef]
- USEPA. Guidelines for Water Reuse; USA Environmental Protection Agency: Washington, DC, USA, 2012.
- Sanz, L.A.; Gawlik, B.M. Water Reuse in Europe—Relevant Guidelines, Needs for and Barriers to Innovation; European Commission: Brussels, Belgium, 2014. [Google Scholar]
- Stevens, D.; Surapaneni, A.; Deere, D.; O’Connor, N.; Crosbie, N.; Keegan, A.; Stackpole, L.; Robards, M. The probability of cysticercus bovis detection in livestock from exposure to recycled water in non-endemic countries. Microb. Risk Anal. 2021, 18, 100164. [Google Scholar] [CrossRef]
- CDC. Parasites—Taeniasis; CDC—Centre for Disease Control and Prevention, U.S. Department of Health and Human Services: Washington, DC, USA, 2016.
- Dettrick, D.; Gallagher, S. Environmental Guidelines for the Use of Recycled Water in Tasmania; Department of Primary Industries, Water and the Environment, Tasmanian Government: Hobart, Tasmania, 2002.
- MLA. Meat and Livestock Australia. The Cattle Parasite Atlas: A Regional Guide to Cattle Parasite Control in Australia; Meat & Livestock Australia: North Sydney, Australia, 2005. [Google Scholar]
- Bowman, D.D.; Little, M.D.; Reimers, R.S. Precision and accuracy of an assay for detecting Ascaris eggs in various biosolid matrices. Water Res. 2003, 37, 2063–2072. [Google Scholar] [CrossRef]
- Da Rocha, M.C.V.; Barés, M.E.; Braga, M.C.B. Quantification of viable helminth eggs in samples of sewage sludge. Water Res. 2016, 103, 245–255. [Google Scholar] [CrossRef]
- Verbyla, M.E.; Iriarte, M.M.; Mercado Guzmán, A.; Coronado, O.; Almanza, M.; Mihelcic, J.R. Pathogens and fecal indicators in waste stabilization pond systems with direct reuse for irrigation: Fate and transport in water, soil and crops. Sci. Total Environ. 2016, 551–552, 429–437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shahsavari, E.; Schmidt, J.; Aburto-Medina, A.; Khallaf, B.; Balakrishnan, V.; Crosbie, N.D.; Surapaneni, A.; Ball, A.S. A modified assay for the enumeration of ascaris eggs in fresh raw sewage. MethodsX 2017, 4, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, V.B.; Khallaf, B.; Surapaneni, A.; Crosbie, N.D.; Soni, S.K.; Ball, A.S. Detection of helminth ova in wastewater using recombinase polymerase amplification coupled to lateral flow strips. Water 2020, 12, 691. [Google Scholar] [CrossRef] [Green Version]
- Naidoo, D.; Archer, C.; Louton, B.; Rodda, N. Testing household disinfectants for the inactivation of helminth eggs on surfaces and in spills during pit latrine emptying. Water SA 2016, 42, 560–570. [Google Scholar] [CrossRef]
- Stevens, D.; Australian Water Quality Centre, Adelaide, South Australia, Australia. Personal Communication, 2018.
- Pinto, H.A.; Mati VL, T.; Melo, A.L.D. Toxocara cati (Nematoda: Ascarididae) in Didelphis albiventris (Marsupialia: Didelphidae) from Brazil: A case of pseudoparasitism. J. Rev. Bras. Parasitol. Veterinária 2014, 23, 522–525. [Google Scholar] [CrossRef] [Green Version]
- Ben Ayed, L.; Schijven, J.; Alouini, Z.; Jemli, M.; Sabbahi, S. Presence of parasitic protozoa and helminth in sewage and efficiency of sewage treatment in Tunisia. Parasitol. Res. 2009, 105, 393. [Google Scholar] [CrossRef]
- Sengupta, M.E.; Thamsborg, S.M.; Andersen, T.J.; Olsen, A.; Dalsgaard, A. Sedimentation of helminth eggs in water. Water Res. 2011, 45, 4651–4660. [Google Scholar] [CrossRef]
- Zacharia, A.; Outwater, A.H.; Ngasala, B.; Van Deun, R. Pathogenic parasites in raw and treated wastewater in Africa: A Review. Resour. Environ. 2018, 8, 232–240. [Google Scholar]
- Sabbahi, S.; Trad, M.; Ben Ayed, L.; Marzougui, N. Occurrence of intestinal parasites in sewage samples and efficiency of wastewater treatment systems in Tunisia. Water Qual. Res. J. 2018, 53, 86–101. [Google Scholar] [CrossRef]
- Ravindran, V.B.; Shahsavari, E.; Soni, S.K.; Ball, A.S. Viability determination of Ascaris ova in raw wastewater: A comparative evaluation of culture-based, BacLight Live/Dead staining and PMA-qPCR methods. Water Sci. Technol. 2019, 80, 817–826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- WHO. Integrated Guide to Sanitary Parasitology. Amman-Jordan: World Health Organization, Regional Office for the Eastern Mediterranean and Regional Centre for Environmental Health Activities. Available online: https://www.who.int/water_sanitation_health/publications/gsuweg2/en/2004 (accessed on 19 July 2020).
- Karkashan, A.; Khallaf, B.; Morris, J.; Thurbon, N.; Rouch, D.; Smith, S.R.; Deighton, M. Comparison of methodologies for enumerating and detecting the viability of Ascaris eggs in sewage sludge by standard incubation-microscopy, the BacLight Live/Dead viability assay and other vital dyes. Water Res. 2015, 68, 533–544. [Google Scholar] [CrossRef]
- Jarmey-Swan, C.; Gibbs, R.A.; Ho, G.E.; Bailey, I.W.; Howgrave-Graham, A.R. A novel method for detection of viable Giardia cysts in water samples. Water Res. 2000, 34, 1948–1951. [Google Scholar] [CrossRef]
- Zarlenga, D.S.; Trout, J.M. Concentrating, purifying and detecting waterborne parasites. Vet. Parasitol. 2004, 126, 195–217. [Google Scholar] [CrossRef]
- Sidhu, J.P.S.; Toze, S.G. Human pathogens and their indicators in biosolids: A literature review. Environ. Int. 2009, 35, 187–201. [Google Scholar] [CrossRef]
- Pascaud, A.; Amellal, S.; Soulas, M.-L.; Soulas, G. A fluorescence-based assay for measuring the viable cell concentration of mixed microbial communities in soil. J. Microbiol. Methods 2009, 76, 81–87. [Google Scholar] [CrossRef]
- Dobrowsky, P.H.; Khan, S.; Cloete, T.E.; Khan, W. Molecular detection of Acanthamoeba spp., Naegleria fowleri and Vermamoeba (Hartmannella) vermiformis as vectors for Legionella spp. in untreated and solar pasteurized harvested rainwater. Parasites Vectors 2016, 9, 539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gyawali, P.; Sidhu, J.P.S.; Ahmed, W.; Jagals, P.; Toze, S. Comparison of culture-based, vital stain and PMA-qPCR methods for the quantitative detection of viable hookworm ova. Water Sci. Technol. 2017, 75, 2615–2621. [Google Scholar] [CrossRef] [Green Version]
- Reyneke, B.; Ndlovu, T.; Khan, S.; Khan, W. Comparison of EMA-, PMA- and DNase qPCR for the determination of microbial cell viability. Appl. Microbiol. Biotechnol. 2017, 101, 7371–7383. [Google Scholar] [CrossRef]
- Zhu, X.; Chilton, N.B.; Jacobs, D.E.; Boes, J.; Gasser, R.B. Characterisation of Ascaris from human and pighosts by nuclear ribosomal DNA sequencesfn1fn1Note: Nucleotide sequence data reported in this paperare available in the embl, GenBankTM and DDJBdatabases under the accession numbersAJ000894–AJ000896, AJ001506, AJ001507 andY09491. Int. J. Parasitol. 1999, 29, 469–478. [Google Scholar] [CrossRef]
- Nejsum, P.; Thamsborg, S.M.; Jørgensen, C.; Fredholm, M.; Roepstorff, A. A novel technique for identification of Ascaris suum cohorts in pigs. Vet. Parasitol. 2008, 154, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Steinbaum, L.; Kwong, L.H.; Ercumen, A.; Negash, M.S.; Lovely, A.J.; Njenga, S.M.; Boehm, A.B.; Pickering, A.J.; Nelson, K.L. Detecting and enumerating soil-transmitted helminth eggs in soil: New method development and results from field testing in Kenya and Bangladesh. PLOS Negl. Trop. Dis. 2017, 11, e0005522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DHV. Guidelines for Validating Treatment Processes for Pathogen Reduction: Supporting Class a Recycled Water Schemes in Victoria. Melbourne, Australia: Victorian Government, Department of Health. Available online: https://www2.health.vic.gov.au:443/about/publications/policiesandguidelines/GuidelinesforvalidatingtreatmentprocessesforpathogenreductionSupportingClassArecycledwaterschemesinVictoria2013 (accessed on 19 July 2020).
- Emelko, M.B.; Huck, P.M. Microspheres as surrogates for cryptosporidium filtration. J. Am. Water Work. Assoc. 2004, 96, 94–105. [Google Scholar] [CrossRef]
- Murakami, E.G.; Jackson, L.; Madsen, K.; Schickedanz, B. Factors affecting the ultraviolet inactivation of Escherichia coli K12 in apple juice and a model system. J. Food Process. Eng. 2006, 29, 53–71. [Google Scholar] [CrossRef]
- Hmaied, F.; Jebri, S. Use of bacteriophages as surrogate indicators of viruses in water. Asia Pac. J. Life Sci. 2013, 7, 109–122. [Google Scholar]
- Arthur, B.K.; Nettmann, E.; Rademacher, A.; Lübken, M.; Marschner, B.; Wichern, M. Microspheres as surrogate helminth eggs: A Comparative Labscale Sedimentation Study for Tap- and Wastewater. Water 2018, 10, 1192. [Google Scholar] [CrossRef] [Green Version]
- Sengupta, M.E.; Andersen, T.J.; Dalsgaard, A.; Olsen, A.; Thamsborg, S.M. Resuspension and settling of helminth eggs in water: Interactions with cohesive sediments. Water Res. 2012, 46, 3903–3912. [Google Scholar] [CrossRef]
- David, E.D.; Lindquist, W.D. Determination of the Specific Gravity of Certain Helminth Eggs Using Sucrose Density Gradient Centrifugation. J. Parasitol. 1982, 68, 916–919. [Google Scholar] [CrossRef]
- CDC. CDC–DPDx—Taeniasis. WWW Document. Available online: https://www.cdc.gov/dpdx/taeniasis/index.html2019 (accessed on 19 July 2020).
- Winkler, M.K.H.; Bassin, J.P.; Kleerebezem, R.; van der Lans, R.G.J.M.; van Loosdrecht, M.C.M. Temperature and salt effects on settling velocity in granular sludge technology. Water Res. 2012, 46, 3897–3902. [Google Scholar] [CrossRef]
- Chaoua, S.; Boussaa, S.; Khadra, A.; Boumezzough, A. Efficiency of two sewage treatment systems (activated sludge and natural lagoons) for helminth egg removal in Morocco. J. Infect. Public Health 2018, 11, 197–202. [Google Scholar] [CrossRef] [PubMed]
| Country | Economic Status A | Helminth Eggs in Sewage (HE/L) | References |
|---|---|---|---|
| Australia | Developed | ≤1.0 | [12] |
| France | 9 | [19] | |
| UK | <1 | [21] | |
| USA | 1–8 | [21] | |
| Bolivia | Developing | 306–3006 | [8] |
| Brazil | 166–202 | [8] | |
| Jordan | 300 | [19] | |
| Mexico | 6–330 | [19] | |
| Morocco | 840 | [19] | |
| Ukraine | 60 | [22] | |
| Vietnam | 5730 | [23] |
| Exposure Scenario for Unrestricted Irrigation | Log10 Pathogen Reduction Required A | Number of Helminth Eggs (HE/L) |
|---|---|---|
| Lettuce | 6 | ≤1.0 |
| Onion | 7 | ≤1.0 |
| Restricted irrigation | ||
| Highly mechanised | 3 | ≤1.0 |
| Labour intensive | 4 | ≤1.0 |
| Localised (Drip Irrigation) | ||
| High-growing crops | 2 | No recommendation, no crops to be pick up from soil |
| Low-growing crops | 4 | ≤1.0 |
| Number of Helminth Eggs in Sewage (HE/L) | Required HE Reduction by Treatment (log10 Units) | Verification Monitoring for Recycled Water (HE/L) | Comments |
|---|---|---|---|
| 1000 | 3 | ≤1.0 | Treatment should be shown to achieve this concentration reliably |
| 100 | 2 | ≤1.0 | |
| 10 | 1 | ≤1.0 | |
| ≤1.0 | none, see comments column | ≤1.0 | The target of ≤1.0 HE/L is automatically achieved |
| Treatment Process | Level of Design and Management of the Treatment System Level | LRV Credit | |
|---|---|---|---|
| High A | Low B | ||
| ASP | HRT (d) | HRT (d) | LRV |
| 0.41 | 0.70 | 0.5 | |
| 0.60 | 0.88 | 1.0 | |
| Lagoon system | HRT | HRT | LRV |
| 12 | 14 | 2.0 | |
| 14 | 16 | 2.5 | |
| 16 | 18 | 3.0 | |
| 18 | 21 | 3.5 | |
| 20 | 23 | 4.0 | |
| Helminth | Helminth Egg Size A | Helminth Shape or Eccentricity | Specific Gravity (g/cm3) | Surface | Settle Velocity (mm/s) | Reference | ||
|---|---|---|---|---|---|---|---|---|
| Length | Width | Theoretical | Measured | |||||
| Ascaris | 67.2 (52.2, 84.1) | 55.4 (46.8, 64.6) | 1.22 | 1.12 | Mamillated | 0.275 | 0.0612 (TW) | [74] |
| 75 | 45 | [53] | ||||||
| 1.23 | [96] A. suum | |||||||
| 60 | 45 | 1.11 | 0.214 (20 °C) | [50] | ||||
| 65 | 45 | 1.13 | 0.264 | [78] A. suum | ||||
| 55 | 40 | Ellipse-shaped to round | 1.11 | Thick, rough albuminous outer wall | 0.119 | [78] A. lumbricoides | ||
| Trichuris | 62.2 (54.2, 68) | 30.8 (26.7, 38.3) | 1.56 | 1.10 | Smooth | 0.129 | 0.149 (TW) | [74]. |
| 1.15 to 1.23 | [96] T. vulpis and T. suis, | |||||||
| 50 | 22 | 1.15 | 0.203 (20 °C) | [50] | ||||
| 52 | 22 | Lemon-shaped, barrel-shaped | 1.15 | Thick shell with smooth surface, | 0.133 | [78] | ||
| Taenia | 30 | 35 | Radially-striated | [97] | ||||
| 1.23 | [96] | |||||||
| 40 | 30 | 1.23 | 0.200 (20 °C) | [50] | ||||
| 40 | 35 | Round, | 1.3 | Thick, smooth shell with radially striated embryophore | 0.231 | [78] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stevens, D.P.; Daniel, V.; Shahsavari, E.; Aburto-Medina, A.; Soni, S.K.; Khudur, L.S.; Khallaf, B.; Surapaneni, A.; Schmidt, J.; Keegan, A.; et al. Improvement of Log Reduction Values Design Equations for Helminth Egg Management in Recycled Water. Water 2021, 13, 3149. https://doi.org/10.3390/w13223149
Stevens DP, Daniel V, Shahsavari E, Aburto-Medina A, Soni SK, Khudur LS, Khallaf B, Surapaneni A, Schmidt J, Keegan A, et al. Improvement of Log Reduction Values Design Equations for Helminth Egg Management in Recycled Water. Water. 2021; 13(22):3149. https://doi.org/10.3390/w13223149
Chicago/Turabian StyleStevens, Daryl P., Vivek Daniel, Esmaeil Shahsavari, Arturo Aburto-Medina, Sarvesh K. Soni, Leadin S. Khudur, Basma Khallaf, Aravind Surapaneni, Jonathan Schmidt, Alexandra Keegan, and et al. 2021. "Improvement of Log Reduction Values Design Equations for Helminth Egg Management in Recycled Water" Water 13, no. 22: 3149. https://doi.org/10.3390/w13223149
APA StyleStevens, D. P., Daniel, V., Shahsavari, E., Aburto-Medina, A., Soni, S. K., Khudur, L. S., Khallaf, B., Surapaneni, A., Schmidt, J., Keegan, A., Crosbie, N. D., Blackbeard, J., Hampton, J., Deere, D., O’Connor, N., & Ball, A. S. (2021). Improvement of Log Reduction Values Design Equations for Helminth Egg Management in Recycled Water. Water, 13(22), 3149. https://doi.org/10.3390/w13223149







