Preliminary Evaluation of the Possible Occurrence of Pesticides in Groundwater Contaminated with Nitrates—A Case Study from Southern Poland
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jankowska, M. Pesticides in natural water. Ochr. Śr. 1998, 68, 13–16. [Google Scholar]
- Biziuk, M. Pesticides-Occurrence, Determination and Disposal; Wydawnictwo Naukowo-Techniczne: Warsaw, Poland, 2009; ISBN 83-2042-663-4. [Google Scholar]
- Lindahl, A.M.L.; Bockstaller, C. An indicator of pesticide leaching risk to groundwater. Ecol. Indic. 2012, 23, 95–108. [Google Scholar] [CrossRef]
- McManus, S.; Coxon, C.E.; Mellander, P.; Danaher, M.; Richards, K.G. Hydrogeological characteristics influencing the occurrence of pesticides and pesticide metabolites in groundwater across the Republic of Ireland. Sci. Total Environ. 2017, 601, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, T.D.; Cifoni, M.; Fiasca, B.; Cioccio, A.D.; Galassi, D.M.P. Ecological risk assessment of pesticide mixtures in the alluvial aquifers of central Italy: Toward more realistic scenarios for risk mitigation. Sci. Total Environ. 2018, 644, 167–172. [Google Scholar] [CrossRef]
- Demir, A.E.A.; Dilek, F.B.; Yetis, U. A new screening index for pesticides leachability to groundwater. J. Environ. Manag. 2019, 231, 1193–1202. [Google Scholar] [CrossRef] [PubMed]
- Haddad, K.; Gheid, A.; Haddad, D.; Oulmi, K. Experimental and numerical study on the leaching of pesticides into the groundwater through a porous medium: Effect of transport parameters. Environ. Technol. Inno. 2019, 13, 244–256. [Google Scholar] [CrossRef]
- Kjær, J.; Rosenbom, A.; Brüsch, W.; Juhler, R.K.; Gudmundsson, L.; Plauborg, F.; Grant, R.; Olsen, P. The Danish Pesticide Leaching Assessment Programme. Monitoring results May 1999–June 2010; Kjær, J., Ed.; Geological Survey of Denmark and Greenland: Copenhagen, Dennmark, 2011; ISBN 978-87-7871-312-4. [Google Scholar]
- Velasco, A.; Hernández, S.; Ramírez, M.; Ortíz, I. Detection of residual organochlorine and organophosphorus pesticides in agricultural soil in Rio Verde region of San Luis Potosí, Mexico. J. Environ. Sci. Health B 2014, 49, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, A.G.P.; López, M.I.R.; Casillas, Á.D.; Leónc, J.A.A.; Banik, S.D. Impact of pesticides in karst groundwater. Review of recent trends in Yucatan, Mexico. Groundw. Sustain. 2018, 7, 20–29. [Google Scholar] [CrossRef]
- Köck-Schulmeyer, M.; Ginebreda, A.; Postigo, C.; Garridob, T.; Fraile, J.; Alda, M.L.; Barceló, D. Four-year advanced monitoring program of polar pesticides in groundwater of Catalonia (NE-Spain). Sci. Total Environ. 2014, 470, 1087–1098. [Google Scholar] [CrossRef] [PubMed]
- Close, M.E.; Humphries, B. The 2014 national survey of pesticides in groundwater in New Zealand. J. Hydrol. 2016, 55, 73–88. [Google Scholar]
- Squillace, P.J.; Scott, J.C.; Moran, M.J.; Nolan, B.T.; Kolpin, D.W. VOCs, pesticides, nitrate, and their mixtures in groundwater used for drinking water in the United States. Environ. Sci. Technol. 2002, 36, 1923–1930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marouane, B.; Dahchour, A.; Dousset, S.; Hajjaji, S.E. Monitoring of nitrate and pesticide pollution in Mnasra, Morocco soil and groundwater. Water Environ. Res. 2015, 87, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Minnesota Department of Agriculture, Monitoring Unit of Pesticide and Fertilizer Management Division—Analysis of the Co-occurrence of Nitrate-Nitrogen and Pesticides in Minnesota Groundwater. Available online: https://www.leg.mn.gov/docs/2008/other/080926.pdf (accessed on 5 May 2021).
- Maanen, J.M.S.V.; Vaan, M.A.J.D.; Veldstra, A.W.F.; Hendrix, W.P.A.M. Pesticides and nitrate in groundwater and rainwater in the province of Limburg in The Netherlands. Environ. Monit. Assess. 2001, 72, 95–114. [Google Scholar] [CrossRef]
- Kraft, G.J.; Browne, B.A.; DeVita, W.M.; Mechenich, D.J. Nitrate and Pesticide Residue Penetration into a Wisconsin Central Sand Plain Aquifer; University of Wisconsin—Stevens Point: Stevens Point, Wisconsin, USA, 2004; Available online: https://www.uwsp.edu/cnr-ap/watershed/documents/penetration_sandplain.pdf (accessed on 5 May 2021).
- Baroudi, M.; Bakkour, H.; Halwani, J.; Taha, S.; El Osmani, R.; Mouneimne, A.H. Determination of pesticides, nitrates and nitrites level in groundwater of Akkar plain in Northern Lebanon. J. Appl. Sci. Res. 2012, 8, 4663–4667. [Google Scholar]
- Yu, G.; Wang, J.; Liu, L.; Li, Y.; Zhang, Y.; Wang, S. The analysis of groundwater nitrate pollution and health risk assessment in rural areas of Yantai, China. BMC Public Health 2020, 20, 437. [Google Scholar] [CrossRef]
- Zufiaurre, R.; Martín-Ramos, P.; Cuchí, J.A. Nitrates in groundwater of small shallow aquifers in the western side of Hoya de Huesca (NE Spain). Agronomy 2019, 10, 22. [Google Scholar] [CrossRef] [Green Version]
- Suthar, S.; Bishnoi, P.; Singh, S.; Mutiyar, P.K.; Nema, A.K.; Patil, N.S. Nitrate contamination in groundwater of some rural areas of Rajasthan, India. J. Hazard. Mater. 2009, 171, 189–199. [Google Scholar] [CrossRef]
- Balla, D.; Zichar, M.; Tóth, R.; Kiss, E.; Karancsi, G.; Mester, T. Geovisualization techniques of spatial environmental data using different visualization tools. Appl. Sci. 2020, 10, 6701. [Google Scholar] [CrossRef]
- Mester, T.; Szabó, G.; Balla, D. Assessment of shallow groundwater purification processes after the construction of a municipal sewerage network. Water 2021, 13, 1946. [Google Scholar] [CrossRef]
- Mester, T.; Balla, D.; Karancsi, G.; Bessenyei, E.; Szabó, G. Effects of nitrogen loading from domestic wastewater on groundwater quality. Water SA 2019, 45, 349–358. [Google Scholar] [CrossRef] [Green Version]
- Burrow, K.R.; Shelton, J.L.; Dubrovsky, N.M. Occurrence of Nitrate and Pesticides in Ground Water Beneath Three Agricultural Land-Use Settings in the Eastern San Joaquin Valley, California, 1993–1995; Water-Resourses Investigations Report 97-4298; US Geological Survey: Sacramento, CA, USA, 1998.
- Witczak, S.; Kania, J.; Kmiecik, E. Catalog of Selected Physical and Chemical Indicators of Groundwater Pollution and Methods of Their Determination; Biblioteka Monitoringu Środowiska: Warsaw, Poland, 2013; ISBN 978-83-61227-13-7. [Google Scholar]
- Łyp, B. Civilization-Related Pollution of Groundwater in Poland; Wydawnictwo Seidel-Przywecki: Warsaw, Poland, 2019; ISBN 978-83-60956-61-8. [Google Scholar]
- Our World in Data—Pesticides. Available online: http://ourworldindata.org/pesticides (accessed on 22 June 2020).
- Statistics Poland—Means of Production in Agriculture in the 2017/18 Farming Year. Available online: https://stat.gov.pl/en/topics/agriculture-forestry/agriculture/means-of-production-in-agriculture-in-the-20172018-farming-year,6,3.html (accessed on 22 June 2020).
- Cabalska, J.; Mikołajczyk, A.; Polak-Mazur, D.; Wołkowicz, W. Occurrence of pesticides in the measurement points of groundwater chemical status monitoring. Prz. Geol. 2015, 63, 635–638. [Google Scholar]
- Dragon, K.; Górski, J.; Kruc´, R.; Drożdżyński, D.; Grischek, T. Removal of natural organic matter and organic micropollutants during riverbank filtration in Krajkowo, Poland. Water 2018, 10, 1457. [Google Scholar] [CrossRef] [Green Version]
- Regulation of the Minister of the Environment of 9 October 2019 on the Forms and Methods of Surface Water and Groundwater Bodies Monitoring. Available online: http://isap.sejm.gov.pl/isap.nsf/download.xsp/WDU20190002147/O/D20192147.pdf (accessed on 5 May 2021).
- Czekaj, J.; Jakóbczyk-Karpierz, S.; Rubin, H.; Sitek, S.; Witkowski, A.J. Identification of nitrate sources in groundwater and potential impact on drinking water reservoir (Goczałkowice reservoir, Poland). Phys. Chem. Earth 2016, 94, 35–46. [Google Scholar] [CrossRef]
- Czekaj, J.; Skrzypczak, M.; Siudy, A. Implementation of Best Practices for Water Protection in Pilot Actions. Partner-Specific Pilot Action Documentations; The Report of PROLINE-CE Workpackage T2 (D.T2.2.2); Interreg Central Europe PROLINE-CE. Available online: https://www.interreg-central.eu/Content.Node/CE110-PROLINE-CE-T2-D.T2.2.2-9-pilot-action-reports.zip (accessed on 5 May 2021).
- Chief Inspectorate of Environmental Protection—Status Assessment of River and Dam Reservoirs in 2017–2018. Available online: https://www.gios.gov.pl/images/dokumenty/pms/monitoring_wod/Klasyfikacja_i_ocena_stanu_RW_2017_2018x.xlsx (accessed on 22 June 2020).
- Sołtysiak, M.; Witkowski, A.J.; Kaźmierczak, J. Hydrogeological aspects of realization of project “integrated system supporting management and protection of Dammed Reservoir (ZiZoZap)”—An example of Goczałkowice reservoir. Biul. Państwowego Inst. Geol. 2011, 445, 651–660. [Google Scholar]
- Ulańczyk, R.; Kożuch, B. Monitoring środowiska wodnego i badania modelowe realizowane w obszarze zlewni zbiornika Kozłowa Góra. Gospod. Wodna 2020, 2, 19–22. [Google Scholar]
- Topografic Map of Poland. Available online: https://mapy.geoportal.gov.pl/wss/service/img/guest/TOPO/MapServer/WMSServer (accessed on 5 May 2021).
- CLC 2018. CORINE Land Cover 2018. Available online: https://land.copernicus.eu/pan-european/corine-land-cover/clc2018 (accessed on 28 October 2021).
- Kempa, J.; Winckiewicz, A. Hydrogeological Map of Poland 1:50 000 “First Aquifer-Groundwater Vulnerability” Sheet Koziegłowy 878; Państwowy Instytut Geologiczny: Warsaw, Poland, 2010.
- Kempa, J.; Winckiewicz, A. Hydrogeological Map of Poland 1:50 000 “First Aquifer-Groundwater Vulnerability” Sheet Wojkowice 911; Państwowy Instytut Geologiczny: Warsaw, Poland, 2010.
- Sołtysiak, M.; Rubin, K. Hydrogeological Map of Poland 1:50 000 “First Aquifer-Groundwater Vulnerability” Sheet Pszczyna 992; Państwowy Instytut Geologiczny: Warsaw, Poland, 2013.
- Czekaj, J.; Witkowski, A.J. Structural model of the quaternary aquifer in the area of Goczałkowice reservoir. Biul. Państwowego Inst. Geol. 2012, 451, 27–33. [Google Scholar]
- Rodzoch, A.; Mutek, K.; Karwacka, K.; Pazio-Urbanowicz, K.; Grodzka, M.; Jeleniewicz, G. Hydrogeological Documentation Establishing the Disposable Groundwater Resources of the Biała Przemsza and Przemsza River Basins; HYDROEKO—Biuro Poszukiwań i Ochrony Wód: Warsaw, Poland, 2012. [Google Scholar]
- Kajewski, I. Assessment of groundwater vulnerability to pollution by organic pesticides in environmental conditions of agricultural catchment. Infrastrukt. Ekol. Teren. Wiej. 2010, 8, 69–78. [Google Scholar]
- PN EN 12918: Water Quality—Determination of Parathion, Parathion-Methyl and Some other Organophosphorus Compounds in Water by Dichloromethane Extraction and Gas Chromatographic Analysis; Polish Committee for Standardization: Warsaw, Poland, 2013.
- PN-EN ISO 6468:2002: Water Quality—Determination of Certain Organochlorine Insecticides, Polychlorinated Biphenyls and Chlorobenzenes—Gas Chromatographic Method after Liquid-Liquid Extraction; Swedish Standard Institution: Stockholm, Sweden, 2012.
- Macioszczyk, A.; Dobrzyński, D. Hydrogeochemistry of an Active Groundwater Exchange Zone; Wydawnictwo Naukowe PWN: Warsaw, Poland, 2007; ISBN 978-83-01151-35-5. [Google Scholar]
- Świderska-Bróż, M. Micropollutants and Their Removal from Natural Waters; Wydawnictwo Politechniki Wrocławskiej: Wrocław, Poland, 1993; ISBN 83-7085-046-4. [Google Scholar]
- Regulation of the Minister of Health of 7 December 2017 on Water Quality Intended for Human Consumption. Available online: http://isap.sejm.gov.pl/isap.nsf/download.xsp/WDU20170002294/O/D20172294.pdf (accessed on 5 May 2021).
- Blarasin, M.; Cabrera, A.; Matiatos, I.; Quinodóz, F.B.; Albo, J.G.; Lutri, V.; Matteos, E.; Panarello, H. Comparative evaluation of urban versus agricultural nitrate sources and sinks in an unconfined aquifer by isotopic and multivariate analyses. Sci. Total Environ. 2020, 741, 140374. [Google Scholar] [CrossRef]
- Buttler, T.; Martinkovic, W.; Nesheim, O.N. Factors Influencing Pesticide Movement to Ground Water; University of Florida, Institute of Food and Agricultural Sciences: Gainesville, FL, USA, 1998. [Google Scholar]
- Affum, A.O.; Acquaah, S.O.; Osae, S.D.; Kwaansa-Ansah, E.E. Distribution and risk assessment of banned and other current-use pesticides in surface and groundwaters consumed in an agricultural catchment dominated by coca crops in the Ankobra Basin, Ghana. Sci. Total Environ. 2018, 633, 360–640. [Google Scholar] [CrossRef]
- Fenner, K.; Canonica, S.; Wackett, L.P.; Elsner, M. Evaluating pesticide degradation in the environment: Blind spots and emerging opportunities. Science 2014, 341, 725–758. [Google Scholar] [CrossRef] [Green Version]
- Sasikala, C.; Jiwal, S.; Rout, P.; Ramya, M. Biodegradation of chlorpyrifos by bacterial consortium isolated from agriculture soil. World J. Microbiol. Biotechnol. 2012, 28, 1301–1308. [Google Scholar] [CrossRef]
- Sirotkina, M.; Lyagin, I.; Efremenko, E. Hydrolysis of organophosphorus pesticides in soil: New opportunities with ecocompatible immobilized His6-OPH. Int. Biodeterior. Biodegrad. 2012, 68, 18–23. [Google Scholar] [CrossRef]
- Dyguda-Kazimierowicz, E.; Roszak, S.; Sokalski, A. Alkaline hydrolysis of organophosphorus pesticides: The dependence of the reaction mechanism on the incoming group conformation. J. Phys. Chem. B 2014, 118, 7277–7289. [Google Scholar] [CrossRef]
- Fishel, F. Effects of Water pH on the Stability of Pesticides. Integrated Pest Management 1017:1-2. MU Extension, University of Missouri, Columbia, USA. Available online: https://extension.missouri.edu/media/wysiwyg/Extensiondata/Pub/pdf/agguides/pests/ipm1017.pdf (accessed on 5 May 2021).
- Draper, W.; Wolfe, N. Abiotic Processes in the Degradation of Pesticides in Natural Waters; US Environmental Protection Agency: Washington, DC, USA, 2002.
- Katagi, T. Abiotic hydrolysis of pesticides in the aquatic environment. Rev. Environ. Contam. Toxicol. 2002, 175, 79–261. [Google Scholar]
- Živančev, N.; Kovačević, S.; Perović, P.; Čalenić, A.; Dimkić, M. Influence of oxic and anoxic groundwater conditions on occurrence of selected agrochemicals. Water Supply 2019, 20, 487–498. [Google Scholar] [CrossRef]
- Papiernik, S.K.; Spalding, R.F. Atrazine, deethylatrazine, and deisopropylatrazine persistence measured in groundwater in situ under low-oxygen conditions. J. Agric. Food Chem. 1998, 46, 749–754. [Google Scholar] [CrossRef] [Green Version]
- Włodarczyk-Makuła, M. Selected Organic Micropollutants in Waters and Soil; Komitet Inżynierii Środowiska PAN: Warsaw, Poland, 2013; ISBN 978-83-63714-03-1. [Google Scholar]
- Carbo, L.; Souza, V.; Dores, E.F.G.C.; Ribeiro, M.L. Determination of pesticides multi-residues in shallow groundwater in a cotton-growing region of Mato Grosso, Brazil. J. Braz. Chem. Soc. 2008, 19, 1111–1117. [Google Scholar]
- Mathys, W. Pesticide pollution of groundwater and drinking water by the processes of artificial groundwater enrichment or coastal filtration: Underrated sources of contamination. Zentralbl. Hyg. Umweltmed. 1994, 196, 338–359. [Google Scholar]
- Burrow, K.R.; Stork, S.V.; Dubrovsky, N.M. Nitrate and Pesticides in Ground Water in the Eastern San Joaquin Valley, California: Occurrence and Trends; Water-Resourses Investigations Report 98-4040; US Geological Survey: Sacramento, CA, USA, 1998.
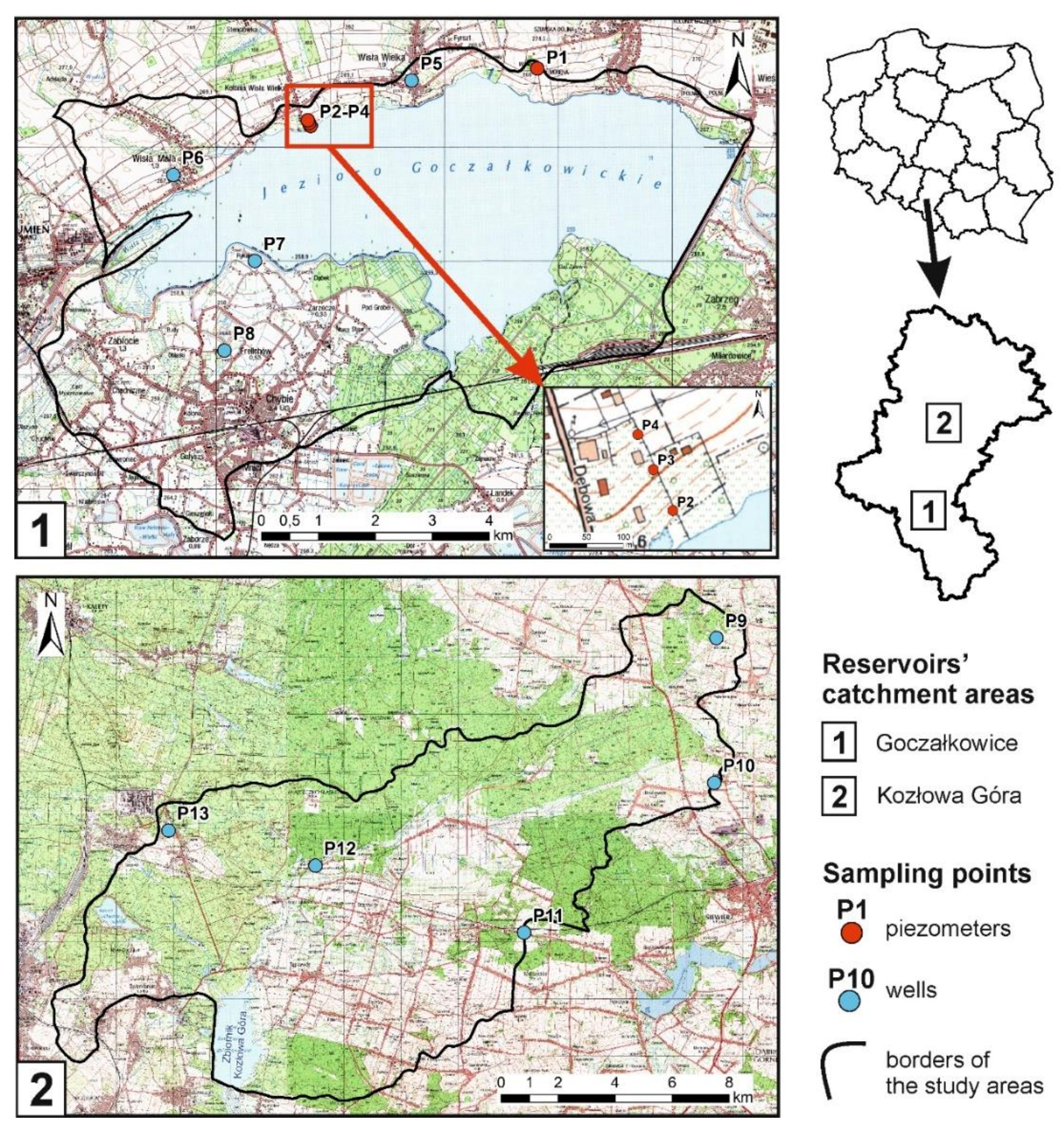
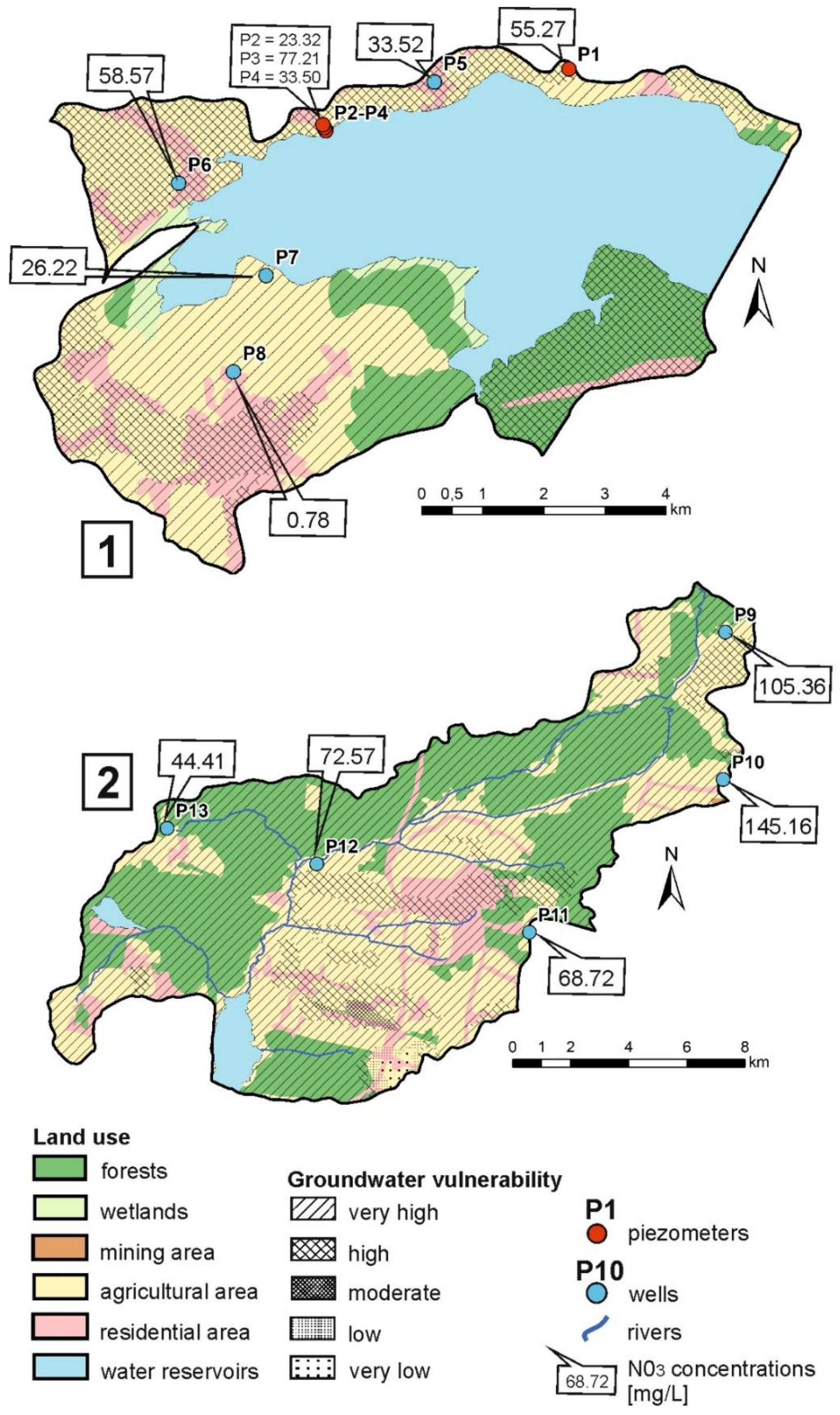
| Sampling Point | Type of Well 1 | Surface Elevation (m a.s.l.) | Well Depth (m) | Water Table Elevation (m a.s.l.) | Water Table Depth (m) | Land Use Type 2 | Details on Potential Pollution Sources |
|---|---|---|---|---|---|---|---|
| The catchment area NO. 1—Goczałkowice | |||||||
| P1 | P | 266.6 | 6.8 | 262.7 | 3.9 | A | Immediate surroundings of arable lands |
| P2 | P | 257.1 | 7.0 | 256.7 | 0.4 | A/R | 100 m from arable lands; 250 m from dwellings |
| P3 | P | 260.0 | 7.0 | 256.1 | 3.9 | A/R | 100 m from arable lands; 250 m from dwellings |
| P4 | P | 262.1 | 8.3 | 256.8 | 5.3 | A/R | 100 m from arable lands; 250 m from dwellings |
| P5 | DW | 268.5 | 30.0 | 257.9 | 10.6 | A/R | 50 m from arable lands; 200 m from dwellings |
| P6 | DW | 266.9 | 8.3 | 259.3 | 7.6 | A/R | Immediate surroundings of arable lands; 250 m from dwellings |
| P7 | DW | 256.4 | 3.8 | 253.8 | 2.6 | A | Immediate surroundings of arable lands |
| P8 | DW | 258.8 | 4.7 | 256.2 | 2.7 | A/R | 50 m from arable lands; 150 m from dwellings |
| The catchment area NO. 2—Kozłowa Góra | |||||||
| P9 | DW | 341.6 | 3.3 | 339.0 | 2.7 | A/F | Immediate surroundings of arable lands; 200 m from forests |
| P10 | DW | 336.3 | 5.3 | 333.5 | 2.9 | A/R | 50 from arable lands; 200 m from dwellings |
| P11 | DW | 309.7 | 4.1 | 307.2 | 2.6 | A/R | 100 m from arable lands; 400 m from dwellings |
| P12 | DW | 286.3 | 5.3 | 282.2 | 4.1 | A/F | Immediate surroundings of arable lands; 300 m from forests |
| P13 | DW | 300.2 | 4.7 | 297.3 | 2.9 | A/R | 200 m from arable lands; 250 m from dwellings |
| Organophosphate pesticides | ||||||||
|---|---|---|---|---|---|---|---|---|
| Compound | USE 1 | LOQ2 | Compound | USE 1 | LOQ 2 | Compound | USE 1 | LOQ 2 |
| Dichlorvos | I | <0.050 | Cypermethrin | I | <0.050 | Pirimiphos-ethyl | I. A | <0.050 |
| Diazinon | I | <0.10 | Deltamethrin | I | <0.050 | Mecarbam | I. A | <0.050 |
| Parathion-ethyl | I, A | <0.050 | Fluopicolide | F | <0.050 | Methidathion | I | <0.050 |
| Parathion-methyl | I | <0.050 | Captan | F | <0.050 | Fensulfothion | I | <0.050 |
| Malathion | I | <0.050 | Mefenpyr-diethyl | H | <0.050 | Azinphos-methyl | I | <0.050 |
| Chlorpyrifos-ethyl | I | <0.030 | Oxyfluorfen | H | <0.050 | Phosalone | I. A | <0.050 |
| Chlorpyrifos-methyl | I | <0.030 | Trifloxystrobin | F | <0.050 | Isoproturon | H | <0.050 |
| Chlorpyrifos | I | <0.030 | λ-cyhalothrin | I | <0.050 | Fenvalerate | I | <0.050 |
| Chlorfenvinfos | I, A | <0.050 | β-cyfluthrin | I | <0.050 | Fenpropathrin | I | <0.050 |
| Fenitrothion | I | <0.050 | Bifenthrin | I | <0.050 | Permethrin | I | <0.050 |
| Malaoxon | I | <0.050 | Procymidone | F | <0.050 | Metribuzin | H | <0.050 |
| Pyrazophos | I, F | <0.050 | Ethoprophos | I. N | <0.050 | Diflufenican | H | <0.050 |
| Triazofos | I, A, N | <0.050 | Pirimiphos-methyl | I | <0.050 | |||
| Organochlorine pesticides | ||||||||
| Compound | USE 1 | LOQ 2 | Compound | USE 1 | LOQ 2 | Compound | USE 1 | LOQ 2 |
| Aldrin | I | <0.010 | α-HCH | I | <0.010 | Endosulfan I | I. A | <0.010 |
| Dieldrin | I | <0.010 | β-HCH | I | <0.010 | Endosulfan II | I. A | <0.010 |
| Endrin | I, R | <0.010 | δ-HCH | I | <0.010 | Endrin aldehyde | I. R | <0.010 |
| Isodrin | I | <0.010 | γ-HCH. lindane | I | <0.010 | Methoxychlor (DMDT) | I | <0.010 |
| o.p-DDT | I | <0.010 | Hexachlorocyclohexane (HCH) | I | <0.010 | Trifluralin | H | <0.010 |
| p.p-DDT | I | <0.010 | Total HCH | I | <0.010 | Alachlor | H | <0.010 |
| o.p’-DDE | I | <0.010 | Heptachlor epoxide B | I | <0.010 | Endosulfan sulfate | I. A | <0.010 |
| p.p’-DDE | I | <0.010 | Heptachlor | I | <0.010 | Hexachlorobenzene (HCB) | F | <0.010 |
| o.p’-DDD | I | <0.010 | α-chlordan | I | <0.010 | |||
| p.p’-DDD | I | <0.010 | γ-chlordan | I | <0.010 | |||
| The catchment area NO. 1—Goczałkowice | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Temp. | pH | PEW | Eh | O2 | HCO3− | SO42− | Cl− | Ca2+ | Mg2+ | Na+ | K+ | NH4+ | NO2− | NO3− |
| °C | - | µS/cm | mV | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |
| P1 | 13.3 | 5 | 311 | 241.1 | 6.77 | 9.20 | 48.20 | 34.75 | 32.54 | 10.34 | 8.58 | 2.05 | 0 | 0.08 | 55.27 |
| P2 | 11.8 | 5.35 | 282 | 228.5 | 9.25 | 21.40 | 79.82 | 17.73 | 31.49 | 8.13 | 10.59 | 2.24 | 0 | 0.07 | 23.32 |
| P3 | 11.9 | 5.47 | 658 | 235.4 | 2.31 | 33.60 | 208.94 | 37.35 | 79.88 | 18.58 | 22.37 | 14.33 | 0 | 0.29 | 77.21 |
| P4 | 12.5 | 5.3 | 494 | 252.5 | 4.03 | 27.40 | 172.54 | 27.35 | 54.32 | 14.89 | 24.01 | 4.35 | 0 | 0.14 | 33.50 |
| P5 | 12.9 | 7.37 | 785 | 114.2 | 12.41 | 329.50 | 147.81 | 25.34 | 120.83 | 13.92 | 37.27 | 32.99 | 1.76 | 0.30 | 33.52 |
| P6 | 13.1 | 6.33 | 457 | 200.6 | 8.34 | 152.60 | 57.69 | 15.56 | 58.68 | 11.92 | 17.15 | 14.64 | 0 | 0.15 | 58.57 |
| P7 | 17.6 | 6.59 | 213 | 197.2 | 4.31 | 85.40 | 11.43 | 3.25 | 22.86 | 3.23 | 3.26 | 20.16 | 0 | 0.11 | 26.22 |
| P8 | 13.9 | 6.66 | 563 | −52.5 | 7.99 | 250.20 | 78.23 | 30.54 | 88.69 | 8.37 | 29.42 | 1.89 | 0.44 | 0.22 | 0.78 |
| The catchment area NO. 2—Kozłowa Góra | |||||||||||||||
| Sample | Temp. | pH | PEW | Eh | O2 | HCO3− | SO42− | Cl− | Ca2+ | Mg2+ | Na+ | K+ | NH4+ | NO2− | NO3− |
| °C | - | µS/cm | mV | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |
| P9 | 15.1 | 6.21 | 390 | 173.2 | 3.46 | 91.50 | 38.16 | 11.64 | 53.30 | 6.51 | 9.79 | 18.26 | 0 | 0.16 | 105.36 |
| P10 | 15 | 7.04 | 721 | 144.2 | 4.04 | 305.10 | 43.81 | 20.87 | 131.03 | 10.67 | 11.03 | 23.53 | 0 | 0.29 | 145.16 |
| P11 | 16.9 | 6.64 | 462 | 136.8 | 0.89 | 118.90 | 75.87 | 18.09 | 66.32 | 5.39 | 16.00 | 11.73 | 0 | 1.15 | 68.72 |
| P12 | 13.5 | 7.08 | 434 | 105.5 | 3.98 | 106.80 | 64.18 | 21.42 | 64.81 | 7.12 | 12.40 | 10.81 | 0 | 0.23 | 72.57 |
| P13 | 18 | 6.57 | 1033 | 148.7 | 4.06 | 122.00 | 409.65 | 105.21 | 154.29 | 35.56 | 63.08 | 10.33 | 0 | 0.38 | 44.41 |
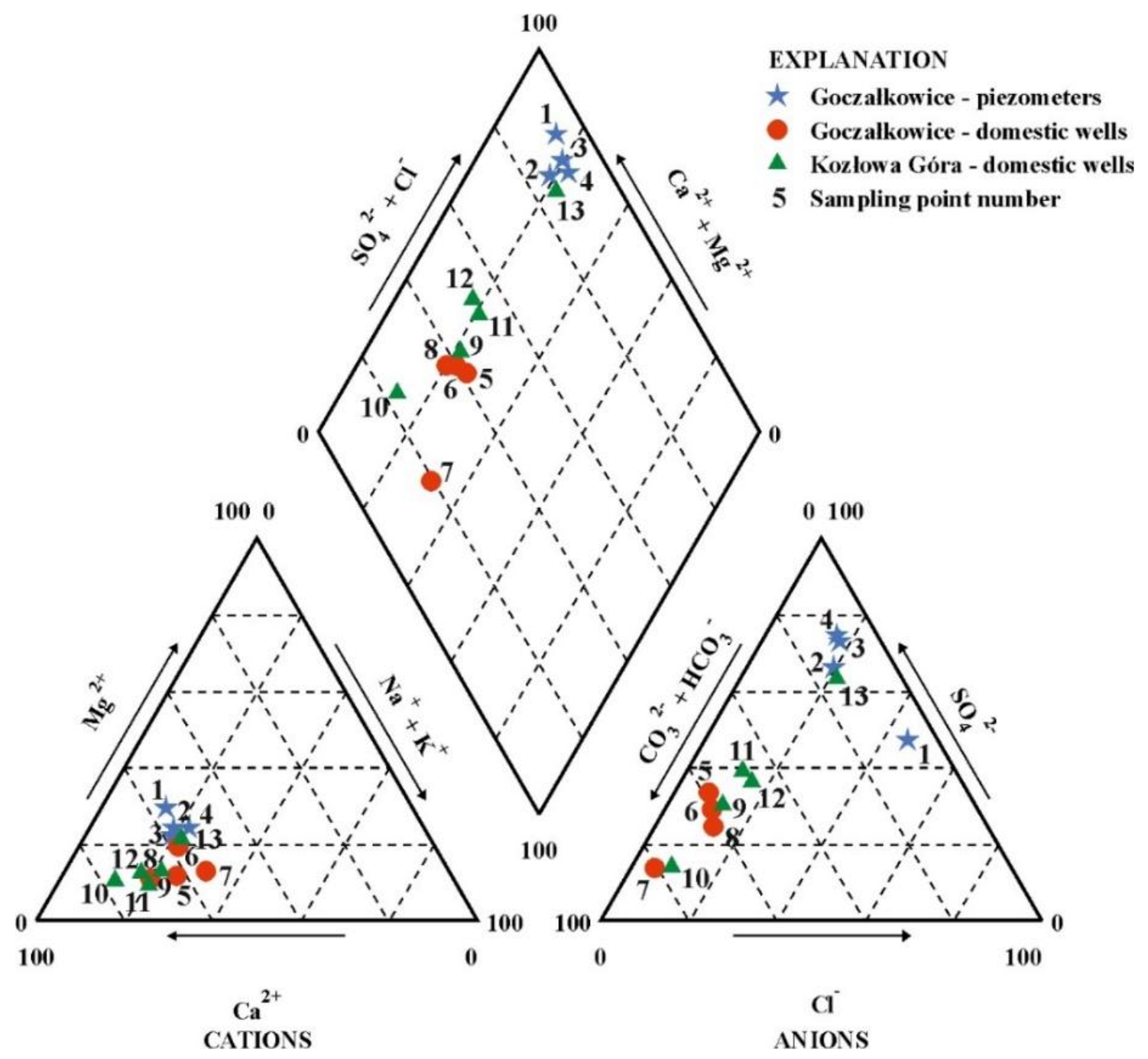
| Sampling Point | Hydrogeochemical Type | |
|---|---|---|
| Szczukariew-Prikłoński | Modified Version (Incl. NO3) | |
| The catchment area NO. 1—Goczałkowice | ||
| P1 | SO4–Cl–Ca–Mg | SO4–Cl–NO3–Ca–Mg |
| P2 | SO4–Ca–Mg | SO4–Ca–Mg |
| P3 | SO4–Ca–Mg | SO4–NO3–Ca–Mg |
| P4 | SO4–Ca–Mg–Na | SO4–Ca–Mg–Na |
| P5 | HCO3–SO4–Ca | HCO3–SO4–NO3–Ca |
| P6 | HCO3–SO4–Ca | HCO3–SO4–Ca |
| P7 | HCO3–Ca | HCO3–NO3–Ca |
| P8 | HCO3–SO4–Ca | HCO3–SO4–Ca |
| The catchment area NO. 2—Kozłowa Góra | ||
| P9 | HCO3–SO4–Ca | NO3–HCO3–SO4–Ca |
| P10 | HCO3–Ca | HCO3–NO3–Ca |
| P11 | HCO3–SO4–Ca | HCO3–SO4–NO3–Ca |
| P12 | HCO3–SO4–Ca | HCO3–SO4–NO3–Ca |
| P13 | SO4–Cl–Ca–Mg–Na | SO4–Cl–Ca–Mg–Na |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ślósarczyk, K.; Witkowski, A.J. Preliminary Evaluation of the Possible Occurrence of Pesticides in Groundwater Contaminated with Nitrates—A Case Study from Southern Poland. Water 2021, 13, 3091. https://doi.org/10.3390/w13213091
Ślósarczyk K, Witkowski AJ. Preliminary Evaluation of the Possible Occurrence of Pesticides in Groundwater Contaminated with Nitrates—A Case Study from Southern Poland. Water. 2021; 13(21):3091. https://doi.org/10.3390/w13213091
Chicago/Turabian StyleŚlósarczyk, Kinga, and Andrzej Jarosław Witkowski. 2021. "Preliminary Evaluation of the Possible Occurrence of Pesticides in Groundwater Contaminated with Nitrates—A Case Study from Southern Poland" Water 13, no. 21: 3091. https://doi.org/10.3390/w13213091
APA StyleŚlósarczyk, K., & Witkowski, A. J. (2021). Preliminary Evaluation of the Possible Occurrence of Pesticides in Groundwater Contaminated with Nitrates—A Case Study from Southern Poland. Water, 13(21), 3091. https://doi.org/10.3390/w13213091