New Technology for Degradation of Wastewater Containing High Concentrations of COD and Boron Based on Manganese-Iron Oxide Catalytic Support
Abstract
:Highlights:
- The method for synthesis of manganese-iron oxide catalytic support was researched.
- A combined treatment method was researched to efficiently degrade COD and boron.
- The high stability and reproducibility of this method provides the potential for its practical application.
1. Introduction
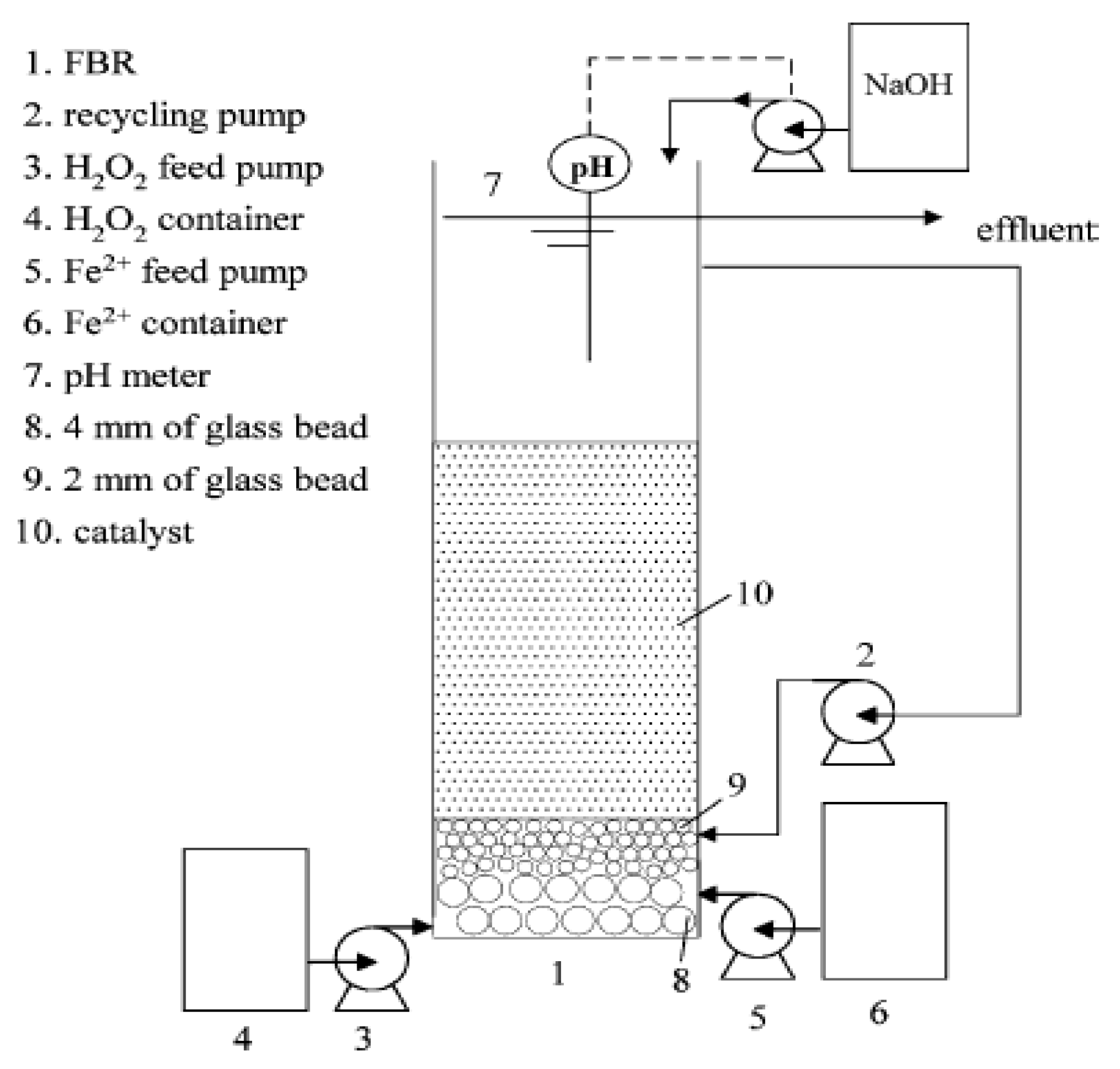
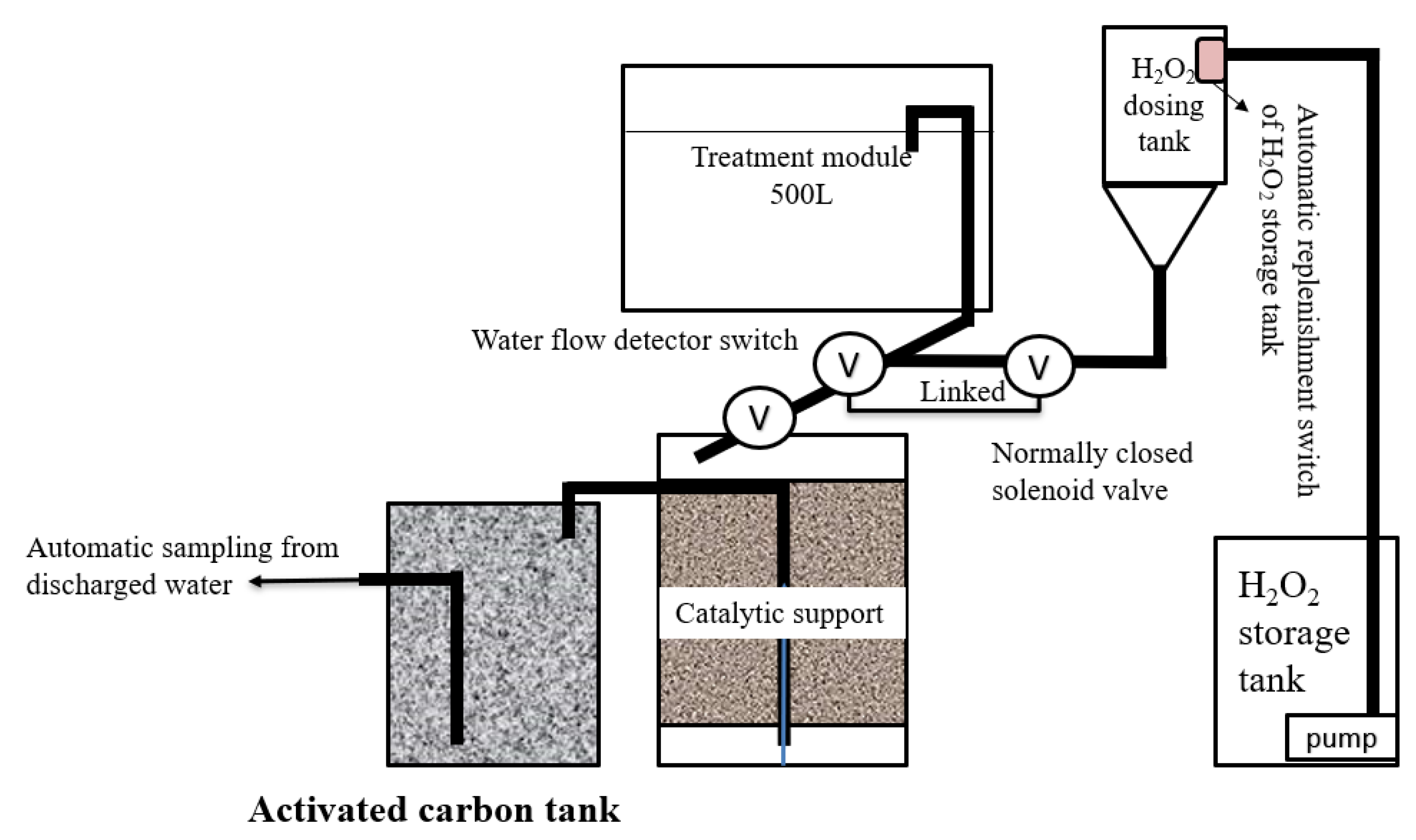
2. Materials and Methodology
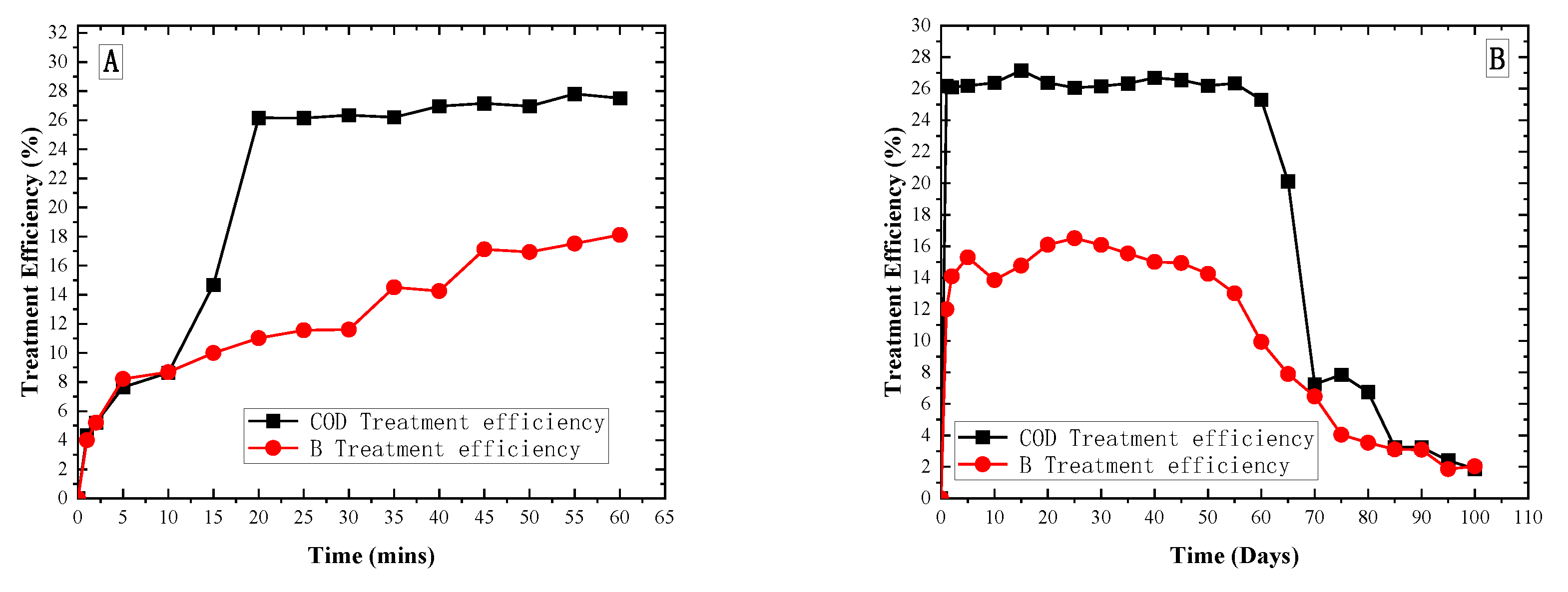
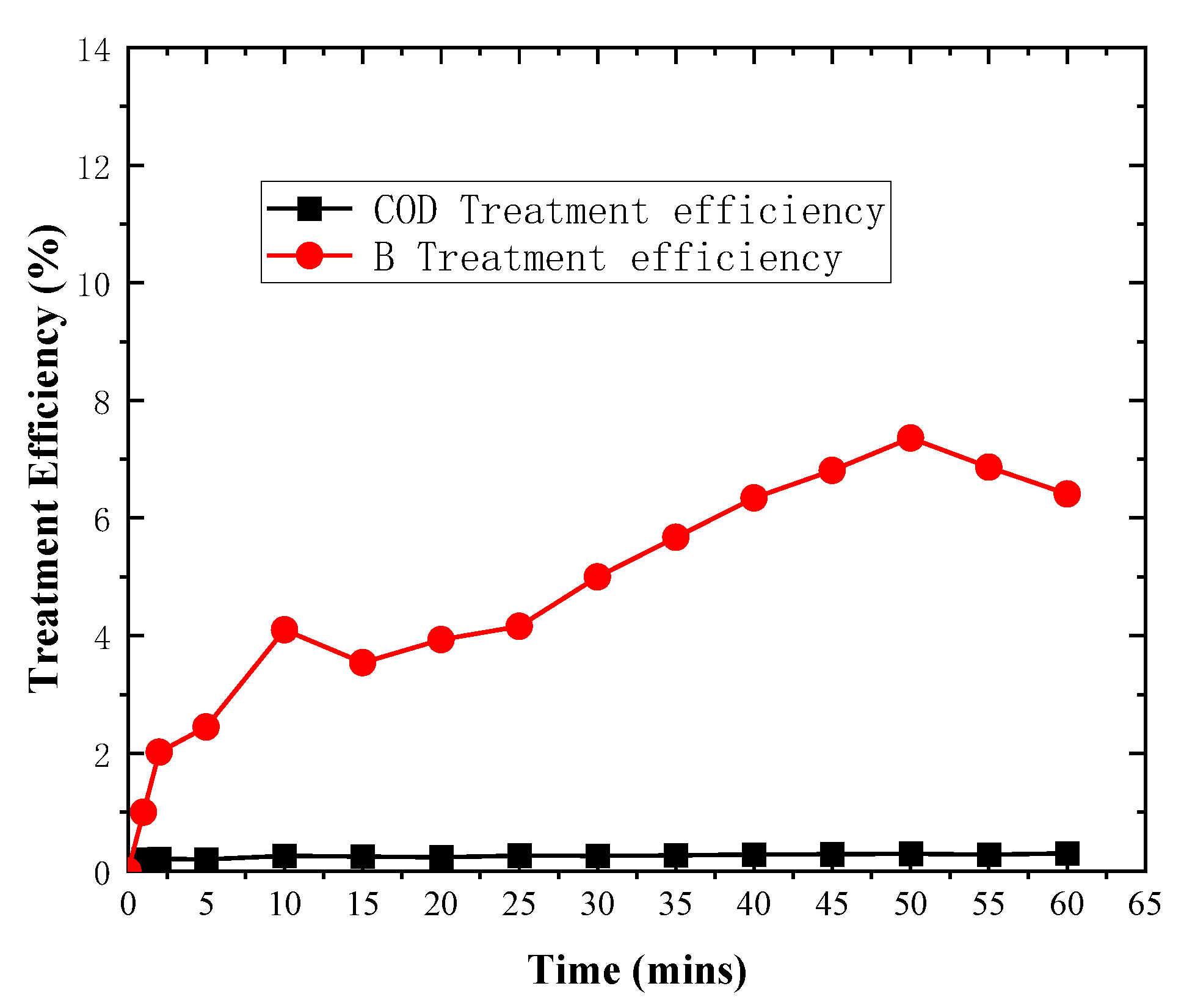
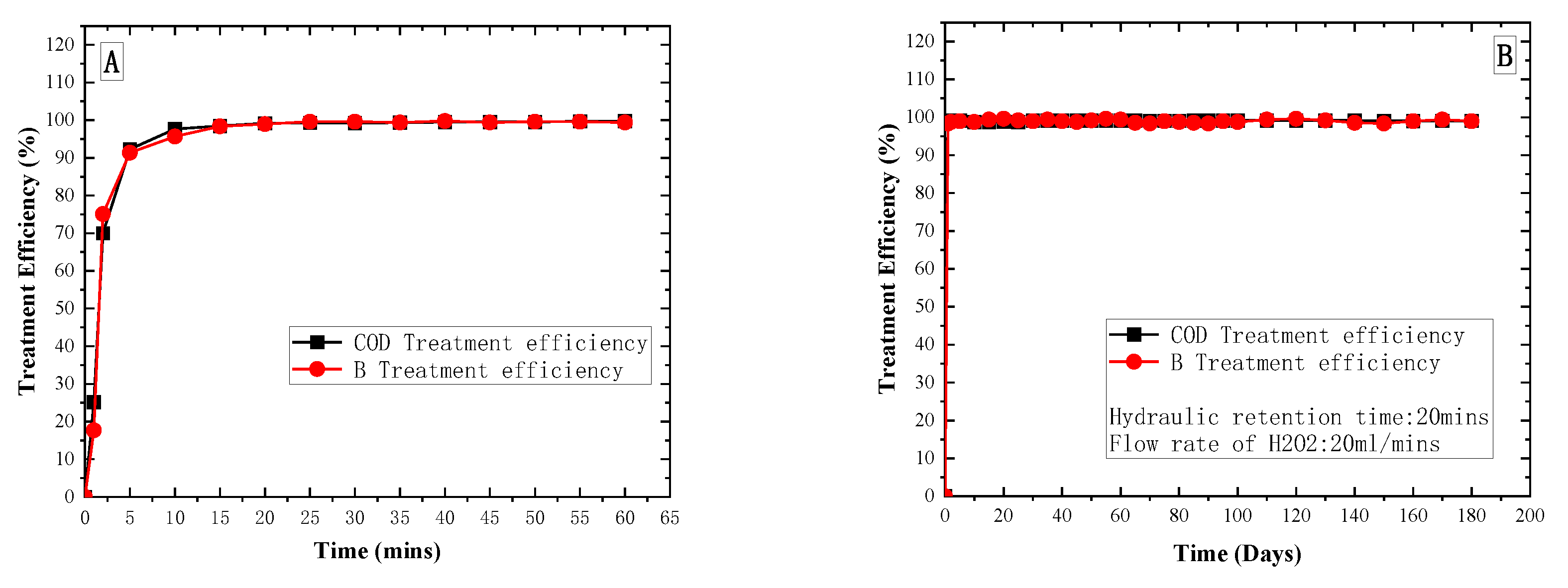
3. Results and Discussion
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shih, Y.-J.; Liu, C.-H.; Lan, W.-C.; Huang, Y.-H. A novel chemical oxo-precipitation (COP) process for efficient remediation of boron wastewater at room temperature. Chemosphere 2014, 111, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Wolska, J.; Bryjak, M. Methods for boron removal from aqueous solutions—A review. Desalination 2013, 310, 18–24. [Google Scholar] [CrossRef]
- Hilal, N.; Kim, G.; Somerfield, C. Boron removal from saline water: A comprehensive review. Desalination 2011, 273, 23–35. [Google Scholar] [CrossRef]
- Schoderboeck, L.; Mühlegger, S.; Losert, A.; Gausterer, C.; Hornek, R. Effects assessment: Boron compounds in the aquatic environment. Chemosphere 2011, 82, 483–487. [Google Scholar] [CrossRef]
- Yilmaz, A.E.; Boncukcuoğlu, R.; Kocakerim, M.M. A quantitative comparison between electrocoagulation and chemical coagula-tion for boron removal from boron-containing solution. J. Hazard. Mater. 2007, 149, 475–481. [Google Scholar] [CrossRef]
- Kentjono, L.; Liu, J.; Chang, W.; Irawan, C. Removal of boron and iodine from optoelectronic wastewater using Mg–Al (NO3) layered double hydroxide. Desalination 2010, 262, 280–283. [Google Scholar] [CrossRef]
- Miyazaki, T.; Takeda, Y.; Akane, S.; Itou, T.; Hoshiko, A.; En, K. Role of boric acid for a poly (vinyl alcohol) film as a cross-linking agent: Melting behaviors of the films with boric acid. Polymer 2010, 51, 5539–5549. [Google Scholar] [CrossRef]
- Kabay, N.; Sarp, S.; Yuksel, M.; Arar, Ö.; Bryjak, M. Removal of boron from seawater by selective ion exchange resins. React. Funct. Polym. 2007, 67, 1643–1650. [Google Scholar] [CrossRef]
- Wei, Y.-T.; Zheng, Y.-M.; Chen, J.P. Design and fabrication of an innovative and environmental friendly adsorbent for boron removal. Water Res. 2011, 45, 2297–2305. [Google Scholar] [CrossRef]
- Tombuloglu, H.; Semizoglu, N.; Sakcali, S.; Kekec, G. Boron induced expression of some stress-related genes in tomato. Chemosphere 2012, 86, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Öztürk, N.; Köse, T.E. Boron removal from aqueous solutions by ion-exchange resin: Batch studies. Desalination 2008, 227, 233–240. [Google Scholar] [CrossRef]
- Bryjak, M.; Wolska, J.; Kabay, N. Removal of boron from seawater by adsorption–membrane hybrid process: Implementation and challenges. Desalination 2008, 223, 57–62. [Google Scholar] [CrossRef]
- Chorghe, D.; Sari, M.A.; Chellam, S. Boron removal from hydraulic fracturing wastewater by aluminum and iron coagulation: Mechanisms and limitations. Water Res. 2017, 126, 481–487. [Google Scholar] [CrossRef]
- Palencia, M.; Vera, M.; Rivas, B.L. Modification of ultrafiltration membranes via interpenetrating polymer networks for removal of boron from aqueous solution. J. Membr. Sci. 2014, 466, 192–199. [Google Scholar] [CrossRef]
- Tang, Y.P.; Luo, L.; Thong, Z.; Chung, T.S. Recent advances in membrane materials and technologies for boron removal. J. Membr. Sci. 2017, 541, 434–446. [Google Scholar] [CrossRef]
- Hou, D.; Dai, G.; Wang, J.; Fan, H.; Luan, Z.; Fu, C. Boron removal and desalination from seawater by PVDF flat-sheet membrane through direct contact membrane distillation. Desalination 2013, 326, 115–124. [Google Scholar] [CrossRef]
- Morisada, S.; Rin, T.; Ogata, T.; Kim, Y.-H.; Nakano, Y. Adsorption removal of boron in aqueous solutions by amine-modified tannin gel. Water Res. 2011, 45, 4028–4034. [Google Scholar] [CrossRef] [PubMed]
- Polowczyk, I.; Ulatowska, J.; Koźlecki, T.; Bastrzyk, A.; Sawiński, W. Studies on removal of boron from aqueous solution by fly ash agglomerates. Desalination 2013, 310, 93–101. [Google Scholar] [CrossRef]
- Oishi, K.; Maehata, Y. Removal properties of dissolved boron by glucomannan gel. Chemosphere 2013, 91, 302–306. [Google Scholar] [CrossRef]
- Wang, H.; Huang, W.; Zhang, Y.; Wang, C.; Jiang, H. Unique metalloid uptake on microplastics: The interaction between boron and microplastics in aquatic environment. Sci. Total Environ. 2021, 800, 149668. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C.-T.; Wu, C.-Y.; Chu, Y.-L.; Huang, W.-J. Effects of high turbidity seawater on removal of boron and transparent exopolymer particles by chemical oxo-precipitation. J. Taiwan Inst. Chem. Eng. 2019, 94, 109–118. [Google Scholar] [CrossRef]
- Tsai, H.-C.; Lo, S.-L. Boron removal and recovery from concentrated wastewater using a microwave hydrothermal method. J. Hazard. Mater. 2011, 186, 1431–1437. [Google Scholar] [CrossRef]
- Ay, F.; Catalkaya, E.C.; Kargi, F. A statistical experiment design approach for advanced oxidation of Direct Red azo-dye by pho-to-Fenton treatment. J. Hazard. Mater. 2009, 162, 230–236. [Google Scholar] [CrossRef]
- Chou, S.; Huang, C. Application of a supported iron oxyhydroxide catalyst in oxidation of benzoic acid by hydrogen peroxide. Chemosphere 1999, 38, 2719–2731. [Google Scholar] [CrossRef] [Green Version]
- Ren, Y.; Lin, L.; Ma, J.; Yang, J.; Feng, J.; Fan, Z. Sulfate radicals induced from peroxymonosulfate by magnetic ferrospinel MFe2O4 (M=Co, Cu, Mn, and Zn) as hetero-geneous catalysts in the water. Appl. Catal. B Environ. 2015, 165, 572–578. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, H.; Zhang, C.; Feng, J.; Pu, S.; Ren, Y.; Wang, Y. Enhanced catalytic ozonation treatment of dibutyl phthalate enabled by porous magnetic Ag-doped ferrospinel MnFe2O4 materials: Performance and mechanism. Chem. Eng. J. 2018, 354, 42–52. [Google Scholar] [CrossRef]
- Zhou, Y.; Xiao, B.; Liu, S.-Q.; Meng, Z.; Chen, Z.-G.; Zou, C.-Y.; Liu, C.-B.; Chen, F.; Zhou, X. Photo-Fenton degradation of ammonia via a manganese–iron double-active component catalyst of graphene–manganese ferrite under visible light. Chem. Eng. J. 2016, 283, 266–275. [Google Scholar] [CrossRef]
- Ma, Q.; Zhang, H.; Zhang, X.; Li, B.; Guo, R.; Cheng, Q.; Cheng, X. Synthesis of magnetic CuO/MnFe2O4 nanocompisite and its high activity for degradation of levofloxacin by activation of persulfate. Chem. Eng. J. 2019, 360, 848–860. [Google Scholar] [CrossRef]
- Rashmi, S.K.; Naik, H.S.B.; Jayadevappa, H.; Sudhamani, C.N.; Patil, S.B.; Naik, M.M. Influence of Sm3+ ions on structural, optical and solar light driven photocatalytic activity of spinel MnFe2O4 nanoparticles. J. Solid State Chem. 2017, 255, 178–192. [Google Scholar] [CrossRef]
- Wang, M.; Zhao, Z.; Zhang, Y. Disposal of Fenton sludge with anaerobic digestion and the roles of humic acids involved in Fenton sludge. Water Res. 2019, 163, 114900. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-Y.; Shih, Y.-J.; Chen, P.-Y.; Huang, Y.-H. Potential Chemical Oxo-precipitation (COP) for Remediating Wastewater with a High Boron Concentration using H2O2/Ba(OH)2 at Room Temperature. Energy Procedia 2014, 61, 349–352. [Google Scholar] [CrossRef] [Green Version]
- Goren, A.; Okten, H. Energy production from treatment of industrial wastewater and boron removal in aqueous solutions using microbial desalination cell. Chemosphere 2021, 285, 131370. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-Y.; Mahasti, N.N.; Huang, Y.-H. Recent advances in adsorption and coagulation for boron removal from wastewater: A comprehensive review. J. Hazard. Mater. 2021, 407, 124401. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.-F. New Technology for Degradation of Wastewater Containing High Concentrations of COD and Boron Based on Manganese-Iron Oxide Catalytic Support. Water 2021, 13, 3020. https://doi.org/10.3390/w13213020
Yang Y-F. New Technology for Degradation of Wastewater Containing High Concentrations of COD and Boron Based on Manganese-Iron Oxide Catalytic Support. Water. 2021; 13(21):3020. https://doi.org/10.3390/w13213020
Chicago/Turabian StyleYang, Ya-Fei. 2021. "New Technology for Degradation of Wastewater Containing High Concentrations of COD and Boron Based on Manganese-Iron Oxide Catalytic Support" Water 13, no. 21: 3020. https://doi.org/10.3390/w13213020




