Influence of Nano- and Small Microplastics on Ciliated Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microparticles
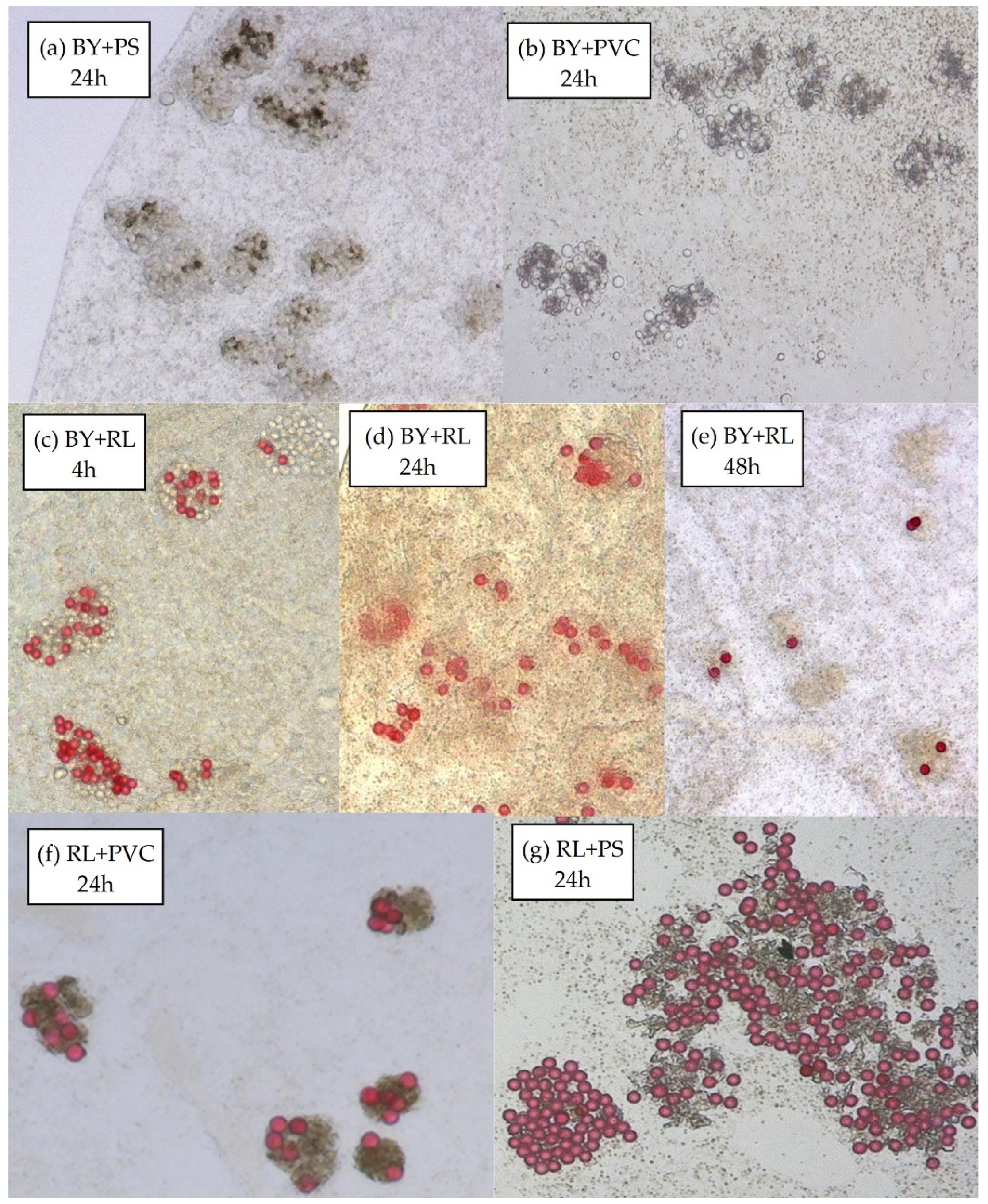
2.2. Microscopic Imaging
2.3. Protozoan S. ambiguum
2.4. Ingestion Studies
2.5. Data Treatment
3. Results
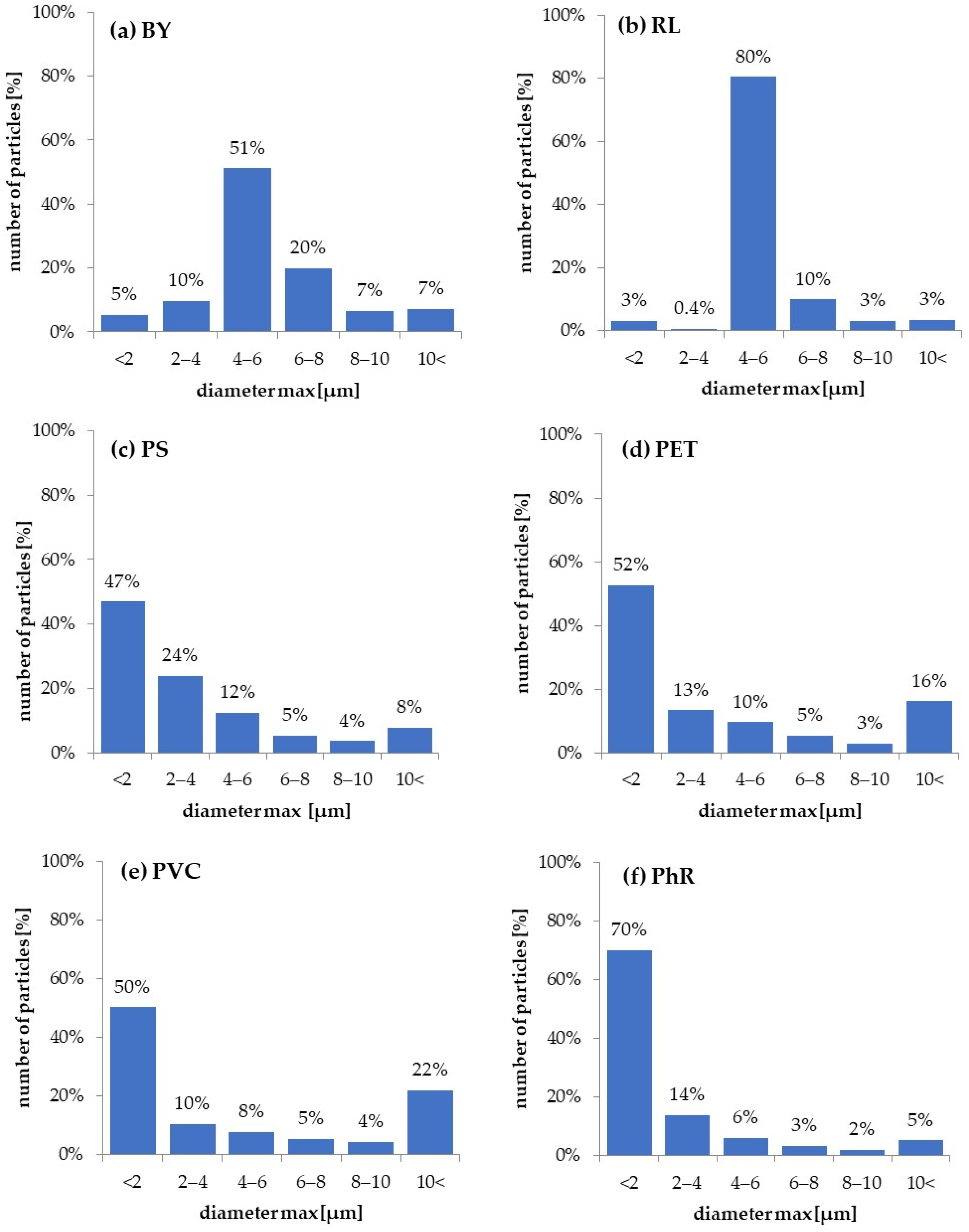
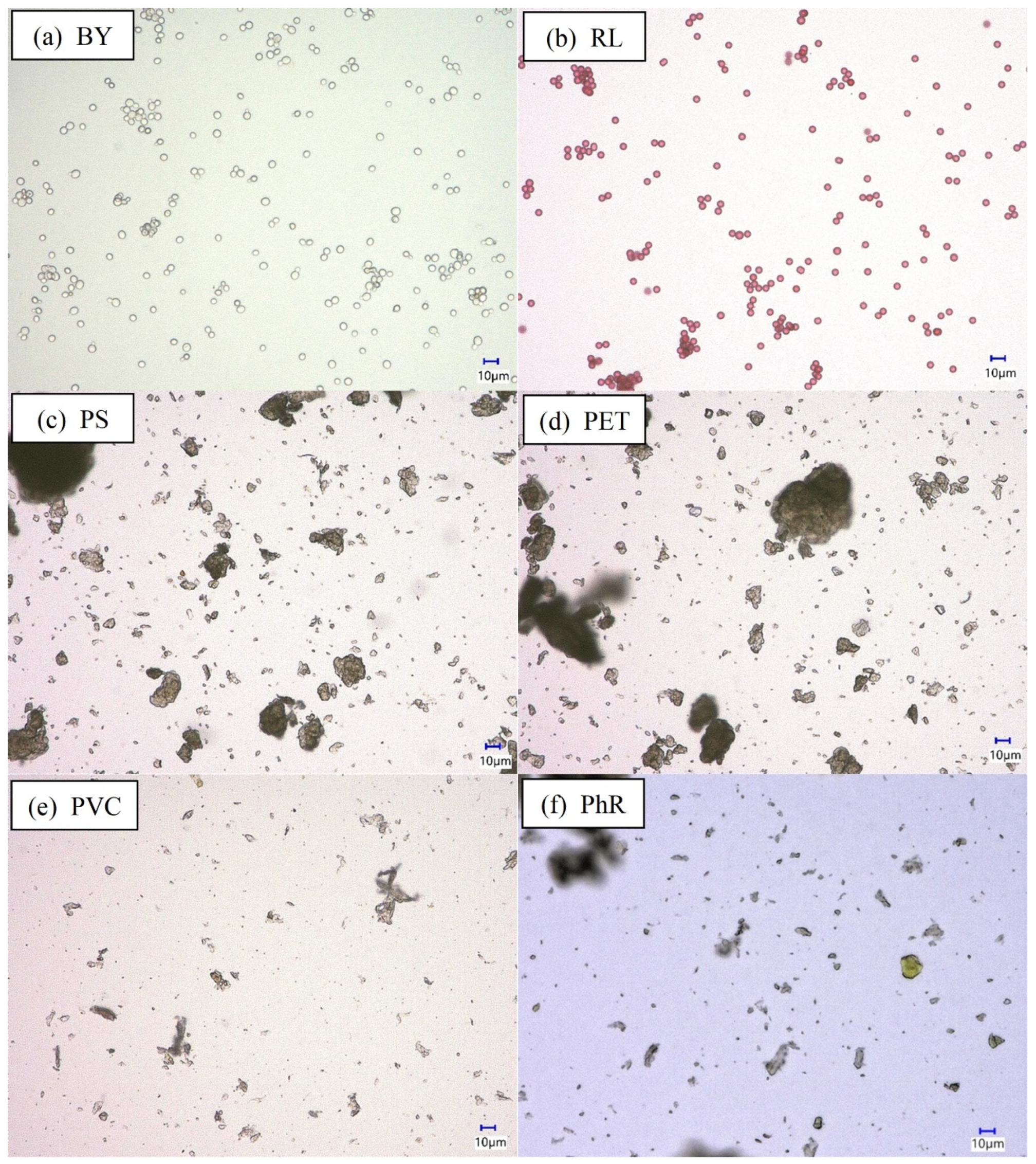
3.1. Particles
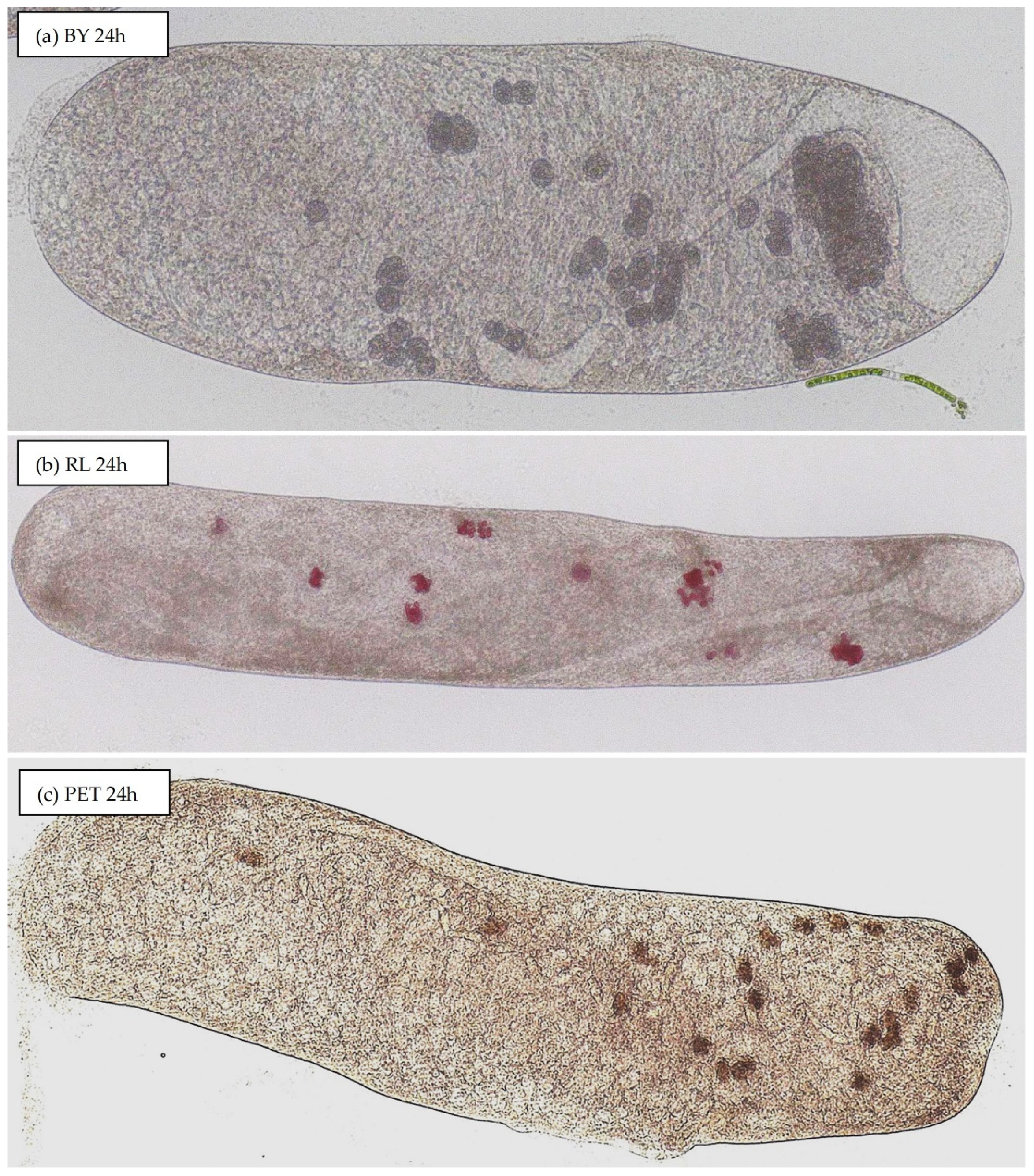
3.2. Uptake of Particles by S. ambiguum
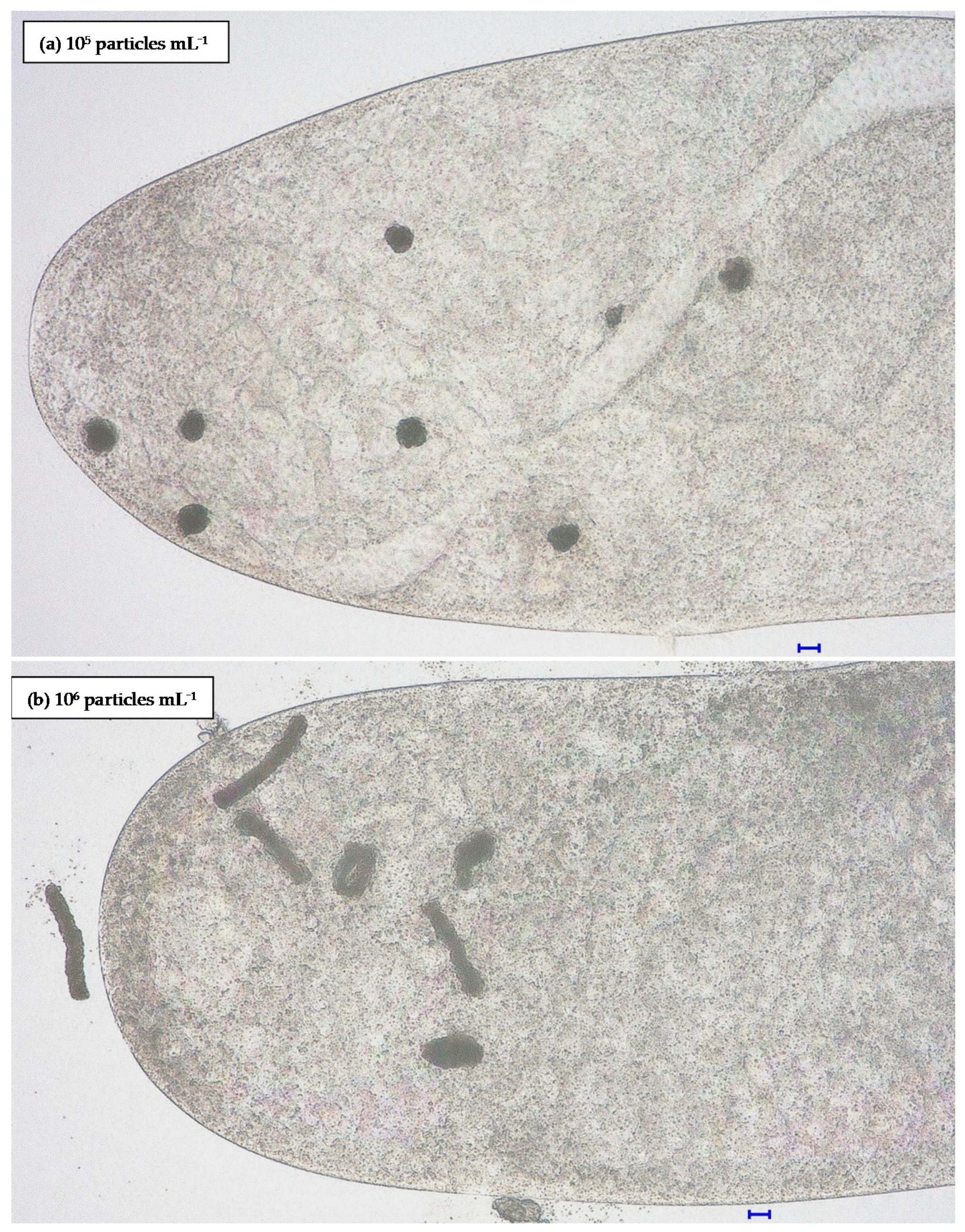
3.2.1. Influence of Particle Concentration
3.2.2. Material Composition
3.2.3. Binary Mixtures of Spherical Particles with Secondary Microplastics
4. Discussion
Ecological Consequences
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Akdogan, Z.; Guven, B. Microplastics in the environment: A critical review of current understanding and identification of future research needs. Environ. Pollut. 2019, 254, 113011. [Google Scholar] [CrossRef] [PubMed]
- Koelmans, A.A.; Nor, N.H.M.; Hermsen, E.; Kooi, M.; Mintenig, S.M.; De France, J. Microplastics in freshwaters and drinking water: Critical review and assessment of data quality. Water Res. 2019, 155, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Prokić, M.D.; Radovanovi, T.B.; Gavri, J.P.; Faggio, C. Ecotoxicological effects of microplastics: Examination of biomarkers, current state and future perspectives. Trends Anal. Chem. 2019, 111, 37–46. [Google Scholar] [CrossRef]
- Sarijan, S.; Azman, S.; Said, M.I.M.; Jamal, M.H. Microplastics in freshwater ecosystems: A recent review of occurrence, analysis, potential impacts, and research needs. Environ. Sci. Pollut. Res. 2021, 28, 1341–1356. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Zhang, Y.; Zhu, Y.; Song, B.; Zeng, G.; Hu, D.; Wen, X.; Ren, X. Recent advances in toxicological research of nanoplastics in the environment: A review. Environ. Pollut. 2019, 252, 511–521. [Google Scholar] [CrossRef]
- Anbumani, S.; Kakkar, P. Ecotoxicological effects of microplastics on biota: A review. Environ. Sci. Pollut. Res. 2018, 25, 14373–14396. [Google Scholar] [CrossRef]
- Hartmann, N.B.; Huffer, T.; Thompson, R.C.; Hassellov, M.; Verschoor, A.; Daugaard, A.E.; Rist, S.; Karlsson, T.; Brennholt, N.; Cole, M.; et al. Are we speaking the same language? Recommendations for a definition and categorization framework for plastic debris. Environ. Sci. Technol. 2019, 53, 1039–1047. [Google Scholar] [CrossRef] [Green Version]
- Yin, K.; Wang, Y.; Zhao, H.; Wang, D.; Guo, M.; Mu, M.; Liu, Y.; Nie, X.; Li, B.; Li, J.; et al. A comparative review of microplastics and nanoplastics: Toxicity hazards on digestive, reproductive and nervous system. Sci. Total Environ. 2021, 774, 145758. [Google Scholar] [CrossRef]
- Rist, S.; Baun, A.; Hartmann, N.B. Ingestion of micro- and nanoplastics in Daphnia magna—Quantification of body burdens and assessment of feeding rates and reproduction. Environ. Pollut. 2017, 228, 398–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christaki, U.; Dolan, J.R.; Pelegri, S.; Rassoulzadegan, F. Consumption of picoplankton-size particles by marine ciliates: Effects of physiological state of the ciliate and particle quality. Limnol. Oceanogr. 2003, 43, 458–464. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.; Fileman, E.; Halsband, C.; Goodhead, R.; Moger, J.; Galloway, T.S. Microplastic ingestion by zooplankton. Environ. Sci. Technol. 2013, 47, 6646–6655. [Google Scholar] [CrossRef]
- Albano, M.; Panarello, G.; Di Paola, D.; D’Angelo, G.; Granata, A.; Savoca, S.; Capillo, G. The mauve stinger Pelagia noctiluca (Cnidaria, Scyphozoa) plastics contamination, the Strait of Messina case. Int. J. Environ. Stud. 2021, 1–6. [Google Scholar] [CrossRef]
- Sorrentino, R.; Senna, A.R. A review of current approaches for the study of microplastic contamination in crustaceans. Environ. Rev. 2021, 29, 64–74. [Google Scholar] [CrossRef]
- Allen, B.; Coumoul, X.; Lacorte, S. Microplastic freshwater contamination: An issue advanced by science with public engagement. Environ. Sci. Pollut. Res. 2019, 26, 16904–16905. [Google Scholar] [CrossRef] [Green Version]
- Scherer, C.; Brennholt, N.; Reifferscheid, G.; Wagner, M. Feeding type and development drive the ingestion of microplastics by freshwater invertebrates. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Hamm, T.; Lenz, M. Negative impacts of realistic doses of spherical and irregular microplastics emerged late during a 42 weeks-long exposure experiment with blue mussels. Sci. Total Environ. 2021, 778, 146088. [Google Scholar] [CrossRef]
- Wang, S.; Hu, M.; Zheng, J.; Huang, W.; Shang, Y.; Kar-Hei Fang, J.; Shi, H.; Wang, Y. Ingestion of nano/micro plastic particles by the mussel Mytilus coruscus is size dependent. Chemosphere 2021, 263, 127957. [Google Scholar] [CrossRef] [PubMed]
- Nałęcz-Jawecki, G. Spirotox—Spirostomum ambiguum acute toxicity test—10 years of experience. Environ. Toxicol. 2004, 19, 359–364. [Google Scholar] [CrossRef]
- Fenchel, T. Suspension feeding in citiated protozoa: Functional response and particle size selection. Microb. Ecol. 1980, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Railkin, A.I. Choice of food by the ciliates Paramecium caudatum Ehrbg. and Spirostomum ambiguum Ehrbg. A Protozool. 1981, 20, 233–254. (In Russian) [Google Scholar]
- Railkin, A.I. The quantitative analysis of phagocytosis in Paramecium caudatum Ehrbg. and Spirostomum ambiguum Ehrbg. A Protozool. 1981, 20, 255–280. (In Russian) [Google Scholar]
- Müller, M.; Röhlich, P.; Törö, I. Studies on feeding and digestion in protozoa. VII. Ingestion of polystyrene latex particles and its early effect on acid phosphatase in Paramecium multimicronucleatum and Tetrahymena pyriformis. J. Eukaryot Microbiol 1965, 12, 27–34. [Google Scholar] [CrossRef]
- Lavin, D.P.; Hatzis, C.; Srienc, F.; Fredrickson, A.G. Size effects on the uptake of particles by populations of Tetrahymena pyriformis cells. J. Protozool 1990, 37, 157–163. [Google Scholar] [CrossRef]
- Bermúdez, J.R.; Metian, M.; Oberhänsli, F.; Taylor, A.; Swarzenski, P.W. Preferential grazing and repackaging of small polyethylene microplastic particles (≤5 µm) by the ciliate Sterkiella sp. Mar. Environ. Res. 2021, 166, 105260. [Google Scholar] [CrossRef] [PubMed]
- Juchelka, C.M.; Snell, T.W. Rapid toxicity assessment using ingestion rate of cladocerans and ciliates. Arch. Environ. Contam. Toxicol. 1995, 28, 508–512. [Google Scholar] [CrossRef]
- Dürichen, H.; Siegmund, L.; Burmester, A.; Fischer, M.S.; Wöstemeyer, J. Ingestion and digestion studies in Tetrahymena pyriformis based on chemically modified microparticles. Eur. J. Protistol. 2016, 52, 45–57. [Google Scholar] [CrossRef]
- Setälä, O.; Fleming-Lehtinen, V.; Lehtiniemi, M. Ingestion and transfer of microplastics in the planktonic food web. Environ. Pollut. 2014, 185, 77–83. [Google Scholar] [CrossRef]
- Corami, F.; Rosso, B.; Bravo, B.; Gambaro, A.; Barbante, C. A novel metod for purification, quantitative analysis and characterization of microplastic fibers using Micro-FTIR. Chemosphere 2020, 238, 124564. [Google Scholar] [CrossRef]
- Rowley, K.H.; Cucknell, A.; Smith, B.D.; Clark, P.F.; Morritt, D. London’s river of plastic: High levels of microplastics in the Thames water column. Sci. Total Environ. 2020, 740, 140018. [Google Scholar] [CrossRef]
- Dubaish, F.; Liebezeit, G. Suspended microplastics and black carbon particles in the Jade system, southern North Sea. Water Air Soil Pollut. 2013, 224, 1352. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G.; Sawicki, J. Toxicity of inorganic compounds in the Spirotox test—A miniaturized version of the Spirostomum ambiguum test. A Environ. Contam. Toxicol. 1998, 34, 1–5. [Google Scholar] [CrossRef]
- Ogonowski, M.; Schür, C.; Jarsén, Å.; Gorokhova, E. The Effects of Natural and Anthropogenic Microparticles on Individual Fitness in Daphnia magna. PLoS ONE 2016, 11, e0155063. [Google Scholar] [CrossRef] [PubMed]
- Krauskopf, L.-M.; Hemmerich, H.; Dsikowitzky, L.; Schwarzbauer, J. Critical aspects on off-line pyrolysis-based quantification of microplastic in environmental samples. J. Anal. Appl. Pyrolysis. 2020, 152, 104830. [Google Scholar] [CrossRef]
- Lithner, D.; Nordensvan, I.; Dave, G. Comparative acute toxicity of leachates from plastic products made of polypropylene, polyethylene, PVC, acrylonitrilebutadiene- styrene, and epoxy to Daphnia magna. Environ. Sci. Pollut. R. Int. 2012, 19, 1763–1772. [Google Scholar] [CrossRef] [PubMed]
- Renzi, M.; Grazioli, E.; Blasković, A. Effects of different microplastic types and surfactant-microplastic mixtures under fasting and feeding conditions: A case study on Daphnia magna. Bull. Environ. Contam. Toxicol. 2019, 103, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Eltemsah, Y.S.; Bøhn, T. Acute and chronic effects of polystyrene microplastics on juvenile and adult Daphnia magna. Environ. Pollut. 2019, 254, 112919. [Google Scholar] [CrossRef]
- Albano, M.; Panarello, G.; Di Paola, D.; Capparucci, F.; Crupi, R.; Gugliandolo, E.; Spanò, N.; Capillo, G.; Savoca, S. The Influence of Polystyrene Microspheres Abundance on Development and Feeding Behavior of Artemia salina (Linnaeus, 1758). Appl. Sci. 2021, 11, 3352. [Google Scholar] [CrossRef]
- Xue, Y.H.; Sun, Z.X.; Feng, L.S.; Jin, T.; Xing, J.C.; Wen, X.L. Algal density affects the influences of polyethylene microplastics on the freshwater rotifer Brachionus calyciflorus. Chemosphere 2021, 270, 128613. [Google Scholar] [CrossRef]
- Anderson, J.C.; Park, B.J.; Palace, V.P. Microplastics in aquatic environments: Implications for Canadian ecosystems. Environ. Pollut. 2016, 218, 269–280. [Google Scholar] [CrossRef] [Green Version]
- Frydkjær, C.K.; Iversen, N.; Roslev, P. Ingestion and Egestion of Microplastics by the Cladoceran Daphnia magna: Effects of Regular and Irregular Shaped Plastic and Sorbed Phenanthrene. Bull. Environ. Contam. Toxicol. 2017, 99, 655–661. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.; Fileman, E.; Halsband, C.; Galloway, T.S. The Impact of Polystyrene Microplastics on Feeding, Function and Fecundity in the Marine Copepod Calanus helgolandicus. Environ. Sci. Technol. 2015, 49, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Triebskorn, R.; Braunbeck, T.; Grummt, T.; Hanslik, L.; Huppertsberg, S.; Jekel, M.; Knepper, T.P.; Krais, S.; Muller, Y.K.; Pittroff, M.; et al. Relevance of nano- and microplastics for freshwater ecosystems: A critical review. Trends Anal. Chem. 2019, 110, 375–392. [Google Scholar] [CrossRef]
| No 1 | 103 Particles mL−1 | 104 Particles mL−1 | 105 Particles mL−1 | 106 Particles mL−1 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AVE ± SD | MIN | MAX | AVE ± SD | MIN | MAX | AVE ± SD | MIN | MAX | AVE ± SD | MIN | MAX | |||
| 2 h | BY | 13–19 | 1.1 ± 1.0 | 0 | 3 | 7.3 ± 2.3 | 4 | 11 | 9.9 ± 2.4 | 5 | 14 | 13.3 ± 3.9 | 6 | 22 |
| RL | 13–17 | 3.1 ± 1.9 | 1 | 7 | 6.5 ± 3.1 | 2 | 11 | 10.0 ± 3.5 | 5 | 15 | 10.8 ± 2.9 | 5 | 17 | |
| PS | 14–18 | 1.1 ± 1.4 | 0 | 4 | 0.3 ± 0.6 2 | 0 | 2 | 5.5 ± 2.0 2 | 2 | 9 | 7.1 ± 2.0 2 | 3 | 10 | |
| PET | 12–17 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1.1 ± 1.0 2 | 0 | 2 | |
| PVC | 13–18 | 0 | 0 | 0 | 0 | 0 | 0 | 1.2 ± 0.7 2 | 0 | 2 | 3.3 ± 1.5 2 | 1 | 6 | |
| PhR | 13–17 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.9 ± 1.0 2 | 0 | 2 | |
| 24 h | BY | 12–20 | 0.5 ± 0.9 | 0 | 2 | 8.8 ± 3.2 | 4 | 16 | 12.9 ± 3.1 | 9 | 19 | 28.0 ± 3.5 | 24 | 30 |
| RL | 13–20 | 0 | 0 | 0 | 0.8 ± 0.8 2 | 0 | 2 | 4.2 ± 1.8 2 | 2 | 7 | 14.7 ± 2.4 2 | 12 | 19 | |
| PS | 13–17 | 0 | 0 | 0 | 0 | 0 | 0 | 8.2 ± 2.1 2 | 6 | 12 | 9.2 ± 2.2 2 | 6 | 14 | |
| PET | 13–18 | 0 | 0 | 0 | 0 | 0 | 0 | 3.7 ± 1.7 2 | 2 | 8 | 14.1 ± 4.7 2 | 6 | 20 | |
| PVC | 14–17 | 0 | 0 | 0 | 0 | 0 | 0 | 6.2 ± 2.2 2 | 3 | 11 | 7.0 ± 1.4 2 | 5 | 10 | |
| PhR | 13–19 | 0 | 0 | 0 | 1.7 ± 0.9 2 | 0 | 3 | 2.0 ± 0.8 2 | 1 | 3 | 14.6 ± 2.8 2 | 11 | 20 | |
| No 1 | 4 h | 24 h | 48 h | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| AVE ± SD | MIN | MAX | AVE ± SD | MIN | MAX | AVE ± SD | MIN | MAX | ||
| BY | 16–20 | 22.8 ± 4.2 | 9 | 28 | 22.4 ± 6.5 | 11 | 35 | 6.9 ± 5.1 | 0 | 17 |
| RL | 15–18 | 14.1 ± 4.3 2 | 7 | 22 | 15.1 ± 3.2 2 | 11 | 21 | 0 | 0 | 0 |
| PS | 10–12 | 12.6 ± 3.3 2 | 8 | 20 | 16.9 ± 2.8 2 | 14 | 23 | 0 | 0 | 0 |
| PET | 12–16 | 10.4 ± 2.8 2 | 7 | 16 | 15.4 ± 3.4 2 | 10 | 22 | 0 | 0 | 0 |
| PVC | 11–16 | 13.4 ± 5.4 2 | 4 | 23 | 6.2 ± 2.0 2 | 4 | 11 | 0 | 0 | 0 |
| PhR | 13–16 | 13.7 ± 2.7 2 | 9 | 19 | 13.7 ± 5.5 2 | 5 | 24 | 0 | 0 | 0 |
| BY + RL | 15–17 | 26.8 ± 7.1 | 17 | 39 | 19.9 ± 2.8 | 15 | 25 | 8.8 ± 5.5 | 0 | 15 |
| BY + PS | 13–27 | 24.4 ± 7.6 | 12 | 39 | 20.9 ± 4.4 | 14 | 31 | 8.1 ± 3.9 | 1 | 14 |
| BY + PET | 13–16 | 25.2 ± 6.9 | 15 | 36 | 20.8 ± 4.6 | 12 | 29 | 7.4 ± 3.4 | 0 | 12 |
| BY + PVC | 11–16 | 11.9 ± 3.1 2 | 7 | 19 | 14.7 ± 5.6 2 | 7 | 28 | 4.6 ± 1.7 2 | 3 | 8 |
| BY + PhR | 9–16 | 22.7 ± 7.5 | 7 | 36 | 26.3 ± 4.0 | 17 | 34 | 6.0 ± 3.2 | 1 | 12 |
| RL + PS | 14–15 | 11.5 ± 3.5 2 | 6 | 19 | 17.3 ± 6.3 2 | 1 | 25 | 4.9 ± 1.9 | 2 | 9 |
| RL + PVC | 15–16 | 6.4 ± 2.5 2 | 2 | 11 | 10.3 ± 4.4 2 | 4 | 24 | 0 | 0 | 0 |
| RL + PhR | 13–16 | 15.6 ± 3.9 | 10 | 24 | 16.3 ± 2.6 | 12 | 22 | 0 | 0 | 0 |
| No 1 | BY:MP = 1:3 | BY:MP = 1:1 | BY:MP = 3:1 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| AVE ± SD | MIN | MAX | AVE ± SD | MIN | MAX | AVE ± SD | MIN | MAX | ||
| BY + RL | 16–17 | 20.4 ± 3.1 | 16 | 27 | 19.9 ± 2.8 | 15 | 25 | 23.3 ± 3.7 | 16 | 28 |
| BY + PS | 14–27 | 21.0 ± 3.7 | 15 | 28 | 20.9 ± 4.4 | 14 | 31 | 20.6 ± 3.7 | 12 | 27 |
| BY + PET | 13–16 | 21.6 ± 3.3 | 17 | 29 | 20.8 ± 4.6 | 12 | 29 | 23.7 ± 5.1 | 10 | 32 |
| BY + PVC | 11–17 | 8.0 ± 3.3 2 | 4 | 17 | 14.7 ± 5.6 2 | 7 | 28 | 24.5 ± 4.7 | 17 | 32 |
| BY + PhR | 13–16 | 22.5 ± 4.4 | 14 | 34 | 26.3 ± 4.0 | 17 | 34 | 27.3 ± 4.5 | 21 | 37 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nałęcz-Jawecki, G.; Chojnacka, J.; Wawryniuk, M.; Drobniewska, A. Influence of Nano- and Small Microplastics on Ciliated Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water 2021, 13, 2857. https://doi.org/10.3390/w13202857
Nałęcz-Jawecki G, Chojnacka J, Wawryniuk M, Drobniewska A. Influence of Nano- and Small Microplastics on Ciliated Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water. 2021; 13(20):2857. https://doi.org/10.3390/w13202857
Chicago/Turabian StyleNałęcz-Jawecki, Grzegorz, Justyna Chojnacka, Milena Wawryniuk, and Agata Drobniewska. 2021. "Influence of Nano- and Small Microplastics on Ciliated Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835" Water 13, no. 20: 2857. https://doi.org/10.3390/w13202857
APA StyleNałęcz-Jawecki, G., Chojnacka, J., Wawryniuk, M., & Drobniewska, A. (2021). Influence of Nano- and Small Microplastics on Ciliated Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water, 13(20), 2857. https://doi.org/10.3390/w13202857






