A Critical Review of Analytical Methods for Comprehensive Characterization of Produced Water
Abstract
1. Introduction
2. Water Composition
3. Tiered Analytical Approach
4. Field Sampling, Preservation, and Sample Pretreatment
4.1. Field Sampling and Preservation
4.2. Sample Preparation and Pretreatment
4.2.1. Liquid-Liquid Extraction and Solid-Liquid Extraction
4.2.2. Solid-Phase Extraction
4.2.3. Other Methods
5. Bulk Measurements and Basic Water Quality Parameters
6. Organic Analysis
6.1. Mass Spectrometry, Tandem Mass Spectrometry, and High-Resolution Mass Spectrometry
6.2. Volatile and Semi-Volatile Organic Analysis
6.3. Non-Volatile Organic Analysis
6.4. Three-Dimensional Excitation-Emission Matrix Fluorescence Spectroscopy
7. Inorganic Analysis
8. Microbiological Characterization
9. Analysis of Naturally Occurring Radioactive Material (NORM)
10. Summary, Knowledge Gap, and Research Needs
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| AEOs | Alkyl ethoxylates | LC-OCD | liquid chromatography coupled with organic carbon detection |
| APCI | Atmospheric pressure chemical ionization | LC-UV | liquid chromatography coupled with UV-diode array detector |
| ASE | Accelerated solvent extraction | LEAF | Leaching environmental assessment framework |
| AST | Above surface storage tank | LLE | Liquid-liquid extraction |
| BOD | Biochemical oxygen demand | MALDI | Matrix-assisted laser desorption/ionization |
| BSFTA | N, O-bis(trimethylsilyl)trifluoroacetamide | MDL | Minimum detection limit |
| BTEX | Benzene, toluene, ethylbenzene, and xylenes | MS | Mass spectroscopy |
| CI | Chemical ionization | MS/MS | Tandem mass spectrometry |
| COD | Chemical oxygen demand | NORM | Naturally occurring radioactive material |
| DCM | Dichloromethane | O&G | Oil and gas |
| DOC | Dissolved organic carbon | ORP | Oxidation-reduction potential |
| DOM | Dissolved organic matter | PAHs | Polycyclic aromatic hydrocarbons |
| EI | Electron ionization | PEGs | Polyethylene glycols |
| EPA | Environmental Protection Agency | PTFE | Polytetrafluoroethylene |
| ESI | Electrospray ionization | PW | Produced water |
| FEEMs | Fluorescence excitation-emission matrix | Q | Quadrupole mass analyzer |
| FID | Flame ionization detector | REEs | Rare earth elements |
| FPSE | Fabric phase sorptive extraction | SEM/EDX | Scanning electron microscopy/energy-dispersive X-ray spectroscopy |
| FT-ICR-MS | Fourier-transform ion cyclotron resonance mass spectrometry | SI | Supporting Information |
| FW | Flowback water | SLE | Solid-liquid extraction |
| GC | Gas chromatography | SPE | Solid-phase extraction |
| GC-FID | Gas chromatography coupled with flame ionization detector | SPLP | Synthetic precipitation leaching procedure |
| GC-MS | Gas chromatography-mass spectrometry | SPME | Solid-phase microextraction |
| GC-TCD | Gas chromatography coupled with thermal conductivity detector | SVOCs | Semi-volatile organic compounds |
| HDPE | High-density polyethylene | SWD | Salt water disposal |
| HF | Hydraulic fracturing | TCLP | Toxicity characteristic leaching procedure; |
| HPGe-GS | High-purity germanium gamma spectrometer | TDS | Total dissolved solids |
| HPLC | High performance liquid chromatography | TN | Total nitrogen |
| HPLC-MS | High performance liquid chromatography -mass spectroscopy | TOC | Total organic carbon |
| HPLC-UV | High performance liquid chromatography coupled with ultraviolet diode array detector | ToF | Time of flight mass analyzer |
| HRMS | High-resolution mass spectrometry | TPH | Total petroleum hydrocarbons |
| HSGC | Headspace gas chromatography | TSS | Total suspended solids |
| IC | Ion chromatography | UD | Unconventional oil and gas development |
| ICP-AES | Inductively coupled plasma-atomic emission spectroscopy | UIC | Underground injection control |
| ICP-MS | Inductively coupled plasma-mass spectroscopy | VOA | Volatile organic analysis |
| ICP-OES | Inductively coupled plasma-optical emission spectroscopy | VOCs | Volatile organic compounds |
| LC | Liquid chromatography | XRD | X-ray diffraction |
| LC-MS | liquid chromatography-mass spectroscopy |
References
- U.S. EIA. Annual Energy Outlook 2019: With Projections to 2050; U.S. Department of Energy: Washington, DC, USA, 2019.
- Zemlick, K.; Kalhor, E.; Thomson, B.M.; Chermak, J.M.; Graham, E.J.S.; Tidwell, V.C. Mapping the energy footprint of produced water management in New Mexico. Environ. Res. Lett. 2018, 13, 024008. [Google Scholar] [CrossRef]
- Scanlon, B.R.; Reedy, R.C.; Male, F.; Walsh, M. Water issues related to transitioning from conventional to unconventional oil production in the Permian Basin. Environ. Sci. Technol. 2017, 51, 10903–10912. [Google Scholar] [CrossRef] [PubMed]
- Fakhru’l-Razi, A.; Pendashteh, A.; Abdullah, L.C.; Biak, D.R.A.; Madaeni, S.S.; Abidin, Z.Z. Review of technologies for oil and gas produced water treatment. J. Hazard. Mater. 2009, 170, 530–551. [Google Scholar] [CrossRef]
- Ferrer, I.; Thurman, E.M. Chemical constituents and analytical approaches for hydraulic fracturing waters. Trends Environ. Anal. Chem. 2015, 5, 18–25. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, D.; Tsang, D.C.; Wang, L.; Ok, Y.S.; Feng, Y. A critical review of risks, characteristics, and treatment strategies for potentially toxic elements in wastewater from shale gas extraction. Environ. Int. 2019, 125, 452–469. [Google Scholar] [CrossRef]
- Vidic, R.D.; Brantley, S.L.; Vandenbossche, J.M.; Yoxtheimer, D.; Abad, J.D. Impact of shale gas development on regional water quality. Science 2013, 340, 1235009. [Google Scholar] [CrossRef]
- Danforth, C.; Chiu, W.A.; Rusyn, I.; Schultz, K.; Bolden, A.; Kwiatkowski, C.; Craft, E. An integrative method for identification and prioritization of constituents of concern in produced water from onshore oil and gas extraction. Environ. Int. 2020, 134, 105280. [Google Scholar] [CrossRef]
- U.S. EIA. Drilling Productivity Report; U.S. Department of Energy: Washington, DC, USA, 2020.
- GWPC. Produced Water Report: Regulations, Current Practices, and Research Needs. 2019. Available online: http://www.gwpc.org/producedwater (accessed on 3 October 2020).
- Kassotis, C.D.; Iwanowicz, L.R.; Akob, D.M.; Cozzarelli, I.M.; Mumford, A.C.; Orem, W.H.; Nagel, S.C. Endocrine disrupting activities of surface water associated with a West Virginia oil and gas industry wastewater disposal site. Sci. Total Environ. 2016, 557, 901–910. [Google Scholar] [CrossRef]
- Hildenbrand, Z.L.; Carlton, D.D., Jr.; Fontenot, B.E.; Meik, J.M.; Walton, J.L.; Taylor, J.T.; Thacker, J.B.; Korlie, S.; Shelor, C.P.; Henderson, D. A comprehensive analysis of groundwater quality in the Barnett Shale region. Environ. Sci. Technol. 2015, 49, 8254–8262. [Google Scholar] [CrossRef]
- He, Y.; Sun, C.; Zhang, Y.; Folkerts, E.J.; Martin, J.W.; Goss, G.G. Developmental toxicity of the organic fraction from hydraulic fracturing flowback and produced waters to early life stages of Zebrafish (Danio rerio). Environ. Sci. Technol. 2018, 52, 3820–3830. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, X.; Jha, A.N.; Rogers, H. Natural gas from shale formation–the evolution, evidences and challenges of shale gas revolution in United States. Renew. Sustain. Energy Rev. 2014, 30, 1–28. [Google Scholar] [CrossRef]
- Hornbach, M.J.; Jones, M.; Scales, M.; DeShon, H.R.; Magnani, M.B.; Frohlich, C.; Stump, B.; Hayward, C.; Layton, M. Ellenburger wastewater injection and seismicity in North Texas. Phys. Earth Planet. Inter. 2016, 261, 54–68. [Google Scholar] [CrossRef]
- Scanlon, B.R.; Reedy, R.C.; Xu, P.; Engle, M.; Nicot, J.P.; Yoxtheimer, D.; Yang, Q.; Ikonnikova, S. Can we beneficially reuse produced water from oil and gas extraction in the U.S.? Sci. Total Environ. 2020, 717, 137085. [Google Scholar] [CrossRef] [PubMed]
- Geza, M.; Ma, G.; Kim, H.; Cath, T.Y.; Xu, P. iDST: An integrated decision support tool for treatment and beneficial use of non-traditional water supplies—Part I. Methodology. J. Water Process Eng. 2018, 25, 236–246. [Google Scholar] [CrossRef]
- Ma, G.; Geza, M.; Cath, T.Y.; Drewes, J.E.; Xu, P. iDST: An integrated decision support tool for treatment and beneficial use of non-traditional water supplies—Part II. Marcellus and Barnett Shale case studies. J. Water Process Eng. 2018, 25, 258–268. [Google Scholar] [CrossRef]
- Rodriguez, A.Z.; Wang, H.; Hu, L.; Zhang, Y.; Xu, P. Treatment of Produced Water in the Permian Basin for Hydraulic Fracturing: Comparison of Different Coagulation Processes and Innovative Filter Media. Water 2020, 12, 770. [Google Scholar] [CrossRef]
- Khan, N.A.; Engle, M.; Dungan, B.; Holguin, F.O.; Xu, P.; Carroll, K.C. Volatile-organic molecular characterization of shale-oil produced water from the Permian Basin. Chemosphere 2016, 148, 126–136. [Google Scholar] [CrossRef]
- Santos, I.C.; Hildenbrand, Z.L.; Schug, K.A. A Review of Analytical Methods for Characterizing the Potential Environmental Impacts of Unconventional Oil and Gas Development. Anal. Chem. 2018, 91, 689–703. [Google Scholar] [CrossRef]
- PartĶ, C.T. Electronic Code of Federal Regulations. 2020. Available online: https://www.ecfr.gov/cgi-bin/text-idx?node=pt40.1.136 (accessed on 15 July 2020).
- Campisano, R.; Hall, K.; Griggs, J.; Willison, S.; Reimer, S.; Mash, H.; Magnuson, M.; Boczek, L.; Rhodes, E. Selected Analytical Methods for Environmental Remediation and Recovery (SAM); US Environmental Protection Agency: Washington, DC, USA, 2017.
- Kim, S.; Omur-Ozbek, P.; Dhanasekar, A.; Prior, A.; Carlson, K. Temporal analysis of flowback and produced water composition from shale oil and gas operations: Impact of frac fluid characteristics. J. Pet. Sci. Eng. 2016, 147, 202–210. [Google Scholar] [CrossRef]
- Oetjen, K.; Chan, K.E.; Gulmark, K.; Christensen, J.H.; Blotevogel, J.; Borch, T.; Spear, J.R.; Cath, T.Y.; Higgins, C.P. Temporal characterization and statistical analysis of flowback and produced waters and their potential for reuse. Sci. Total Environ. 2018, 619, 654–664. [Google Scholar] [CrossRef]
- Murali Mohan, A.; Hartsock, A.; Bibby, K.J.; Hammack, R.W.; Vidic, R.D.; Gregory, K.B. Microbial community changes in hydraulic fracturing fluids and produced water from shale gas extraction. Environ. Sci. Technol. 2013, 47, 13141–13150. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C.; Jack, R. Analysis of ions in hydraulic fracturing wastewaters using ion chromatography. In Hydraulic Fracturing: Environmental Issues; ACS Publications: Washington, DC, USA, 2015; pp. 135–150. [Google Scholar]
- Rosenblum, J.; Nelson, A.W.; Ruyle, B.; Schultz, M.K.; Ryan, J.N.; Linden, K.G. Temporal characterization of flowback and produced water quality from a hydraulically fractured oil and gas well. Sci. Total Environ. 2017, 596, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Mouser, P.J.; Borton, M.; Darrah, T.H.; Hartsock, A.; Wrighton, K.C. Hydraulic fracturing offers view of microbial life in the deep terrestrial subsurface. FEMS Microbiol. Ecol. 2016, 92, 11. [Google Scholar] [CrossRef] [PubMed]
- Hildenbrand, Z.L.; Santos, I.C.; Liden, T.; Carlton, D.D., Jr.; Varona-Torres, E.; Martin, M.S.; Reyes, M.L.; Mulla, S.R.; Schug, K.A. Characterizing variable biogeochemical changes during the treatment of produced oilfield waste. Sci. Total Environ. 2018, 634, 1519–1529. [Google Scholar] [CrossRef]
- Cluff, M.A.; Hartsock, A.; MacRae, J.D.; Carter, K.; Mouser, P.J. Temporal changes in microbial ecology and geochemistry in produced water from hydraulically fractured Marcellus Shale gas wells. Environ. Sci. Technol. 2014, 48, 6508–6517. [Google Scholar] [CrossRef] [PubMed]
- Vikram, A.; Lipus, D.; Bibby, K. Produced water exposure alters bacterial response to biocides. Environ. Sci. Technol. 2014, 48, 13001–13009. [Google Scholar] [CrossRef]
- Morono, Y.; Wishart, J.; Ito, M.; Ijiri, A.; Hoshino, T.; Torres, M.; Verba, C.; Terada, T.; Inagaki, F.; Colwell, F. Microbial Metabolism and Community Dynamics in Hydraulic Fracturing Fluids Recovered From Deep Hydrocarbon-Rich Shale. Front. Microbiol. 2019, 10, 376. [Google Scholar] [CrossRef]
- Lipus, D.; Roy, D.; Khan, E.; Ross, D.; Vikram, A.; Gulliver, D.; Hammack, R.; Bibby, K. Microbial communities in Bakken region produced water. Fems Microbiol. Lett. 2018, 365, fny107. [Google Scholar] [CrossRef]
- Liden, T.; Santos, I.C.; Hildenbrand, Z.L.; Schug, K.A. Treatment modalities for the reuse of produced waste from oil and gas development. Sci. Total Environ. 2018, 643, 107–118. [Google Scholar] [CrossRef]
- Lipus, D.; Vikram, A.; Ross, D.; Bain, D.; Gulliver, D.; Hammack, R.; Bibby, K. Predominance and metabolic potential of Halanaerobium spp. in produced water from hydraulically fractured Marcellus shale wells. Appl. Environ. Microbiol. 2017, 83. [Google Scholar] [CrossRef]
- Hull, N.M.; Rosenblum, J.S.; Robertson, C.E.; Harris, J.K.; Linden, K.G. Succession of toxicity and microbiota in hydraulic fracturing flowback and produced water in the Denver–Julesburg Basin. Sci. Total Environ. 2018, 644, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S. Sample Preparation Techniques in Analytical Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2004; Volume 237. [Google Scholar]
- Maguire-Boyle, S.J.; Barron, A.R. Organic compounds in produced waters from shale gas wells. Environ. Sci. Process. Impacts 2014, 16, 2237–2248. [Google Scholar] [CrossRef] [PubMed]
- Majors, R. Sample Preparation Fundamentals for Chromatography; Agilent Technologies Inc.: Mississauga, ON, Canada, 2013; pp. 55–181. [Google Scholar]
- Oetjen, K.; Giddings, C.G.; McLaughlin, M.; Nell, M.; Blotevogel, J.; Helbling, D.E.; Mueller, D.; Higgins, C.P. Emerging analytical methods for the characterization and quantification of organic contaminants in flowback and produced water. Trends Environ. Anal. Chem. 2017, 15, 12–23. [Google Scholar] [CrossRef]
- Riley, S.M.; Ahoor, D.C.; Regnery, J.; Cath, T.Y. Tracking oil and gas wastewater-derived organic matter in a hybrid biofilter membrane treatment system: A multi-analytical approach. Sci. Total Environ. 2018, 613, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Richter, B.E.; Jones, B.A.; Ezzell, J.L.; Porter, N.L.; Avdalovic, N.; Pohl, C. Accelerated solvent extraction: A technique for sample preparation. Anal. Chem. 1996, 68, 1033–1039. [Google Scholar] [CrossRef]
- Regnery, J.; Coday, B.D.; Riley, S.M.; Cath, T.Y. Solid-phase extraction followed by gas chromatography-mass spectrometry for the quantitative analysis of semi-volatile hydrocarbons in hydraulic fracturing wastewaters. Anal. Methods 2016, 8, 2058–2068. [Google Scholar] [CrossRef]
- Sørensen, L.; McCormack, P.; Altin, D.; Robson, W.J.; Booth, A.M.; Faksness, L.-G.; Rowland, S.J.; Størseth, T.R. Establishing a link between composition and toxicity of offshore produced waters using comprehensive analysis techniques-A way forward for discharge monitoring? Sci. Total Environ. 2019, 694, 133682. [Google Scholar] [CrossRef]
- Thacker, J.B.; Carlton, D.D.; Hildenbrand, Z.L.; Kadjo, A.F.; Schug, K.A. Chemical analysis of wastewater from unconventional drilling operations. Water 2015, 7, 1568–1579. [Google Scholar] [CrossRef]
- Almaraz, N.; Regnery, J.; Vanzin, G.F.; Riley, S.M.; Ahoor, D.C.; Cath, T.Y. Emergence and fate of volatile iodinated organic compounds during biological treatment of oil and gas produced water. Sci. Total Environ. 2020, 699, 134202. [Google Scholar] [CrossRef]
- Redman, A.; Parkerton, T.; Butler, J.; Letinski, D.; Frank, R.; Hewitt, L.; Bartlett, A.; Gillis, P.; Marentette, J.; Parrott, J. Application of the target lipid model and passive samplers to characterize the toxicity of bioavailable organics in oil sands process-affected water. Environ. Sci. Technol. 2018, 52, 8039–8049. [Google Scholar] [CrossRef]
- Kumar, R.; Malik, A.K.; Kabir, A.; Furton, K.G. Efficient analysis of selected estrogens using fabric phase sorptive extraction and high performance liquid chromatography-fluorescence detection. J. Chromatogr. A 2014, 1359, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Samanidou, V.; Galanopoulos, L.-D.; Kabir, A.; Furton, K.G. Fast extraction of amphenicols residues from raw milk using novel fabric phase sorptive extraction followed by high-performance liquid chromatography-diode array detection. Anal. Chim. Acta 2015, 855, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.D.; Ternes, T.A. Water analysis: Emerging contaminants and current issues. Anal. Chem. 2018, 90, 398–428. [Google Scholar] [CrossRef]
- Xu, F.; Zou, L.; Liu, Y.; Zhang, Z.; Ong, C.N. Enhancement of the capabilities of liquid chromatography–mass spectrometry with derivatization: General principles and applications. Mass Spectrom. Rev. 2011, 30, 1143–1172. [Google Scholar] [CrossRef] [PubMed]
- Kahrilas, G.A.; Blotevogel, J.; Corrin, E.R.; Borch, T. Downhole transformation of the hydraulic fracturing fluid biocide glutaraldehyde: Implications for flowback and produced water quality. Environ. Sci. Technol. 2016, 50, 11414–11423. [Google Scholar] [CrossRef]
- Ferrer, I.; Thurman, E.M. Analysis of hydraulic fracturing additives by LC/Q-TOF-MS. Anal. Bioanal. Chem. 2015, 407, 6417–6428. [Google Scholar] [CrossRef]
- Carlton, D.D., Jr.; Hildenbrand, Z.L.; Schug, K.A. Analytical Approaches for High-Resolution Environmental Investigations of Unconventional Oil and Gas Exploration. In Advances in Chemical Pollution, Environmental Management and Protection; Elsevier: Amsterdam, The Netherlands, 2017; Volume 1, pp. 193–226. [Google Scholar]
- Hickenbottom, K.L.; Hancock, N.T.; Hutchings, N.R.; Appleton, E.W.; Beaudry, E.G.; Xu, P.; Cath, T.Y. Forward osmosis treatment of drilling mud and fracturing wastewater from oil and gas operations. Desalination 2013, 312, 60–66. [Google Scholar] [CrossRef]
- Lyman, S.N.; Mansfield, M.L.; Tran, H.N.; Evans, J.D.; Jones, C.; O’Neil, T.; Bowers, R.; Smith, A.; Keslar, C. Emissions of organic compounds from produced water ponds I: Characteristics and speciation. Sci. Total Environ. 2018, 619, 896–905. [Google Scholar] [CrossRef]
- Sun, C.; Zhang, Y.; Alessi, D.S.; Martin, J.W. Nontarget profiling of organic compounds in a temporal series of hydraulic fracturing flowback and produced waters. Environ. Int. 2019, 131, 104944. [Google Scholar] [CrossRef]
- Liden, T.; Santos, I.C.; Hildenbrand, Z.L.; Schug, K.A. Analytical Methods for the Comprehensive Characterization of Produced Water. In Separation Science and Technology; Elsevier: Amsterdam, The Netherlands, 2019; Volume 11, pp. 199–217. [Google Scholar]
- Luek, J.L.; Gonsior, M. Organic compounds in hydraulic fracturing fluids and wastewaters: A review. Water Res. 2017, 123, 536–548. [Google Scholar] [CrossRef]
- Hoelzer, K.; Sumner, A.J.; Karatum, O.; Nelson, R.K.; Drollette, B.D.; O’Connor, M.P.; D’Ambro, E.L.; Getzinger, G.J.; Ferguson, P.L.; Reddy, C.M. Indications of transformation products from hydraulic fracturing additives in shale-gas wastewater. Environ. Sci. Technol. 2016, 50, 8036–8048. [Google Scholar] [CrossRef]
- De Vijlder, T.; Valkenborg, D.; Lemière, F.; Romijn, E.P.; Laukens, K.; Cuyckens, F. A tutorial in small molecule identification via electrospray ionization-mass spectrometry: The practical art of structural elucidation. Mass Spectrom. Rev. 2018, 37, 607–629. [Google Scholar] [CrossRef]
- Wahl, K.L.; Wunschel, S.C.; Jarman, K.H.; Valentine, N.B.; Petersen, C.E.; Kingsley, M.T.; Zartolas, K.A.; Saenz, A.J. Analysis of microbial mixtures by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Anal. Chem. 2002, 74, 6191–6199. [Google Scholar] [CrossRef]
- Thurman, E.M.; Ferrer, I.; Blotevogel, J.; Borch, T. Analysis of hydraulic fracturing flowback and produced waters using accurate mass: Identification of ethoxylated surfactants. Anal. Chem. 2014, 86, 9653–9661. [Google Scholar] [CrossRef] [PubMed]
- Thurman, E.M.; Ferrer, I.; Rosenblum, J.; Linden, K.; Ryan, J.N. Identification of polypropylene glycols and polyethylene glycol carboxylates in flowback and produced water from hydraulic fracturing. J. Hazard. Mater. 2017, 323, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Dass, C. Fundamentals of Contemporary Mass Spectrometry; John Wiley & Sons: Hoboken, NJ, USA, 2007; Volume 16. [Google Scholar]
- Hiraoka, K. Fundamental of Mass Spectrometry; Springer: Berlin, Germany, 2013. [Google Scholar]
- Hollender, J.; Schymanski, E.L.; Singer, H.P.; Ferguson, P.L. Nontarget Screening with High Resolution Mass Spectrometry in the Environment: Ready to Go? ACS Publications: Washington, DC, USA, 2017. [Google Scholar]
- Sitterley, K.A.; Linden, K.G.; Ferrer, I.; Thurman, E.M. Identification of proprietary amino ethoxylates in hydraulic fracturing wastewater using liquid chromatography/time-of-flight mass spectrometry with solid-phase extraction. Anal. Chem. 2018, 90, 10927–10934. [Google Scholar] [CrossRef] [PubMed]
- Schymanski, E.L.; Jeon, J.; Gulde, R.; Fenner, K.; Ruff, M.; Singer, H.P.; Hollender, J. Identifying Small Molecules via High Resolution Mass Spectrometry: Communicating Confidence; ACS Publications: Washington, DC, USA, 2014. [Google Scholar]
- NIST. National Institute of Standards and Technology. NIST20: Updates to the NIST Tandem and Electron Ionization Spectral Libraries. 2020. Available online: https://www.nist.gov/programs-projects/nist20-updates-nist-tandem-and-electron-ionization-spectral-libraries (accessed on 20 March 2020).
- Nell, M.; Helbling, D.E. Exploring matrix effects and quantifying organic additives in hydraulic fracturing associated fluids using liquid chromatography electrospray ionization mass spectrometry. Environ. Sci. Process. Impacts 2019, 21, 195–205. [Google Scholar] [CrossRef]
- EPA. Technical Overview of Volatile Organic Compounds; United States Environmental Protection Agency: Washington, DC, USA, 2016.
- Varona-Torres, E.; Carlton, D.D., Jr.; Hildenbrand, Z.L.; Schug, K.A. Matrix-effect-free determination of BTEX in variable soil compositions using room temperature ionic liquid co-solvents in static headspace gas chromatography mass spectrometry. Anal. Chim. Acta 2018, 1021, 41–50. [Google Scholar] [CrossRef]
- U.S. EPA. Clean Water Act Analytical Methods; U.S. EPA: Washington, DC, USA, 2019.
- McAdams, B.C.; Carter, K.E.; Blotevogel, J.; Borch, T.; Hakala, J.A. In situ transformation of hydraulic fracturing surfactants from well injection to produced water. Environ. Sci. Process. Impacts 2019, 21, 1777–1786. [Google Scholar] [CrossRef]
- Stoll, Z.A.; Forrestal, C.; Ren, Z.J.; Xu, P. Shale gas produced water treatment using innovative microbial capacitive desalination cell. J. Hazard. Mater. 2015, 283, 847–855. [Google Scholar] [CrossRef]
- Wang, H.; Lu, L.; Chen, X.; Bian, Y.; Ren, Z.J. Geochemical and microbial characterizations of flowback and produced water in three shale oil and gas plays in the central and western United States. Water Res. 2019, 164, 114942. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Mackay, E.; Ishkov, O.; Strachan, A. Predicted and observed evolution of produced-brine compositions and implications for scale management. Spe Prod. Oper. 2016, 31, 270–279. [Google Scholar] [CrossRef]
- Cantlay, T.; Eastham, J.L.; Rutter, J.; Bain, D.J.; Dickson, B.C.; Basu, P.; Stolz, J.F. Determining conventional and unconventional oil and gas well brines in natural samples I: Anion analysis with ion chromatography. J. Environ. Sci. Healthpart A 2020, 55, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Cantlay, T.; Bain, D.J.; Curet, J.; Jack, R.F.; Dickson, B.C.; Basu, P.; Stolz, J.F. Determining conventional and unconventional oil and gas well brines in natural sample II: Cation analyses with ICP-MS and ICP-OES. J. Environ. Sci. Healthpart A 2019, 55, 1–10. [Google Scholar] [CrossRef]
- Tanner, S.D.; Baranov, V.I.; Bandura, D.R. Reaction cells and collision cells for ICP-MS: A tutorial review. Spectrochim. Acta Part B At. Spectrosc. 2002, 57, 1361–1452. [Google Scholar] [CrossRef]
- O’Sullivan, J.E.; Watson, R.J.; Butler, E.C. An ICP-MS procedure to determine Cd, Co, Cu, Ni, Pb and Zn in oceanic waters using in-line flow-injection with solid-phase extraction for preconcentration. Talanta 2013, 115, 999–1010. [Google Scholar] [CrossRef]
- Balaram, V. Rare earth elements: A review of applications, occurrence, exploration, analysis, recycling, and environmental impact. Geosci. Front. 2019, 10, 1285–1303. [Google Scholar] [CrossRef]
- Weber, R.J.; Reisman, D.J. Rare Earth Elements: A Review of Production, Processing, Recycling, and Associated Environmental Issues; US EPA Region: Kansas, KS, USA, 2012. [Google Scholar]
- Meier, A.; Slowik, T.; Taggart, J. Rare earth elements by inductively coupled plasma–mass spectrometry. Us Geol. Surv. Open-File Rep. 2002, 2, 223. [Google Scholar]
- Quillinan, S.; Nye, C.; Engle, M.; Bartos, T.; Neupane, G.; Brant, J.; Bagdonas, D.; McLaughlin, J.F. Assessing REE Concentrations in Geothermal and Oil and Gas Produced Waters: A Potential Domestic Source of Strategic Mineral Commodities; University of Wyoming: Laramie, Wyoming, 2018. [Google Scholar]
- Xu, P.; Drewes, J.E.; Heil, D. Beneficial use of co-produced water through membrane treatment: Technical-economic assessment. Desalination 2008, 225, 139–155. [Google Scholar] [CrossRef]
- Palková, Z. Multicellular microorganisms: Laboratory versus nature. Embo Rep. 2004, 5, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Emerson, D.; Agulto, L.; Liu, H.; Liu, L. Identifying and characterizing bacteria in an era of genomics and proteomics. Bioscience 2008, 58, 925–936. [Google Scholar] [CrossRef]
- Mardis, E.R. The impact of next-generation sequencing technology on genetics. Trends Genet. 2008, 24, 133–141. [Google Scholar] [CrossRef]
- Peruski, A.H.; Peruski, L.F. Immunological methods for detection and identification of infectious disease and biological warfare agents. Clin. Diagn. Lab. Immunol. 2003, 10, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Hartzell, S.E.; Unger, M.A.; Vadas, G.G.; Yonkos, L.T. Evaluating porewater polycyclic aromatic hydrocarbon–related toxicity at a contaminated sediment site using a spiked field-sediment approach. Environ. Toxicol. Chem. 2018, 37, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Lay, J.O., Jr. MALDI-TOF mass spectrometry of bacteria. Mass Spectrom. Rev. 2001, 20, 172–194. [Google Scholar] [CrossRef]
- Santos, I.C.; Hildenbrand, Z.L.; Schug, K.A. Applications of MALDI-TOF MS in environmental microbiology. Analyst 2016, 141, 2827–2837. [Google Scholar] [CrossRef]
- Yang, Y.; Lin, Y.; Qiao, L. Direct MALDI-TOF MS identification of bacterial mixtures. Anal. Chem. 2018, 90, 10400–10408. [Google Scholar] [CrossRef]
- Santos, I.C.; Martin, M.S.; Reyes, M.L.; Carlton, D.D., Jr.; Stigler-Granados, P.; Valerio, M.A.; Whitworth, K.W.; Hildenbrand, Z.L.; Schug, K.A. Exploring the links between groundwater quality and bacterial communities near oil and gas extraction activities. Sci. Total Environ. 2018, 618, 165–173. [Google Scholar] [CrossRef]
- Zielinski, R.A.; Otton, J.K. Naturally Occurring Radioactive Materials (NORM) in Produced Water and Oil-field Equipment: An Issue for Energy Industry; US Department of the Interior, US Geological Survey: Washington, DC, USA, 1999.
- Warner, N.R.; Christie, C.A.; Jackson, R.B.; Vengosh, A. Impacts of shale gas wastewater disposal on water quality in western Pennsylvania. Environ. Sci. Technol. 2013, 47, 11849–11857. [Google Scholar] [CrossRef]
- Fisher, R.S. Geologic and geochemical controls on naturally occurring radioactive materials (NORM) in produced water from oil, gas, and geothermal operations. Environ. Geosci. 1998, 5, 139–150. [Google Scholar] [CrossRef]
- Brown, V.J. Radionuclides in Fracking Wastewater: Managing a Toxic Blend; National Institute of Environmental Health Sciences: Durham, NC, USA, 2014.
- Burden, S.; Fleming, M.; Frithsen, J.; Hills, L.; Klewicki, K.; Knightes, C. Hydraulic Fracturing for Oil and Gas: Impacts from the Hydraulic Fracturing Water Cycle on Drinking Water Resources in the United States; U.S. EPA: Washington, DC, USA, 2016.
- Akob, D.M.; Cozzarelli, I.M.; Dunlap, D.S.; Rowan, E.L.; Lorah, M.M. Organic and inorganic composition and microbiology of produced waters from Pennsylvania shale gas wells. Appl. Geochem. 2015, 60, 116–125. [Google Scholar] [CrossRef]
- Harto, C.B.; Smith, K.P.; Kamboj, S.; Quinn, J.J. Radiological Dose and Risk Assesssment of Landfill Disposal of Technologically Enhanced Naturally Occurring Radioactive Materials (Tenorm) in North Dakota; Argonne National Lab. (ANL): Argonne, IL, USA, 2014.
- Zhang, T.; Bain, D.; Hammack, R.; Vidic, R.D. Analysis of radium-226 in high salinity wastewater from unconventional gas extraction by inductively coupled plasma-mass spectrometry. Environ. Sci. Technol. 2015, 49, 2969–2976. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.W.; May, D.; Knight, A.W.; Eitrheim, E.S.; Mehrhoff, M.; Shannon, R.; Litman, R.; Schultz, M.K. Matrix complications in the determination of radium levels in hydraulic fracturing flowback water from Marcellus Shale. Environ. Sci. Technol. Lett. 2014, 1, 204–208. [Google Scholar] [CrossRef]
- Tasker, T.L.; Burgos, W.D.; Ajemigbitse, M.A.; Lauer, N.E.; Gusa, A.V.; Kuatbek, M.; May, D.; Landis, J.D.; Alessi, D.S.; Johnsen, A.M. Accuracy of methods for reporting inorganic element concentrations and radioactivity in oil and gas wastewaters from the Appalachian Basin, US based on an inter-laboratory comparison. Environ. Sci. Process. Impacts 2019, 21, 224–241. [Google Scholar] [CrossRef]
- Vanhaecke, F.; Degryse, P. Isotopic Analysis: Fundamentals and Applications Using ICP-MS; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Fan, W.; Hayes, K.F.; Ellis, B.R. Estimating Radium Activity in Shale Gas Produced Brine. Environ. Sci. Technol. 2018, 52, 10839–10847. [Google Scholar] [CrossRef] [PubMed]
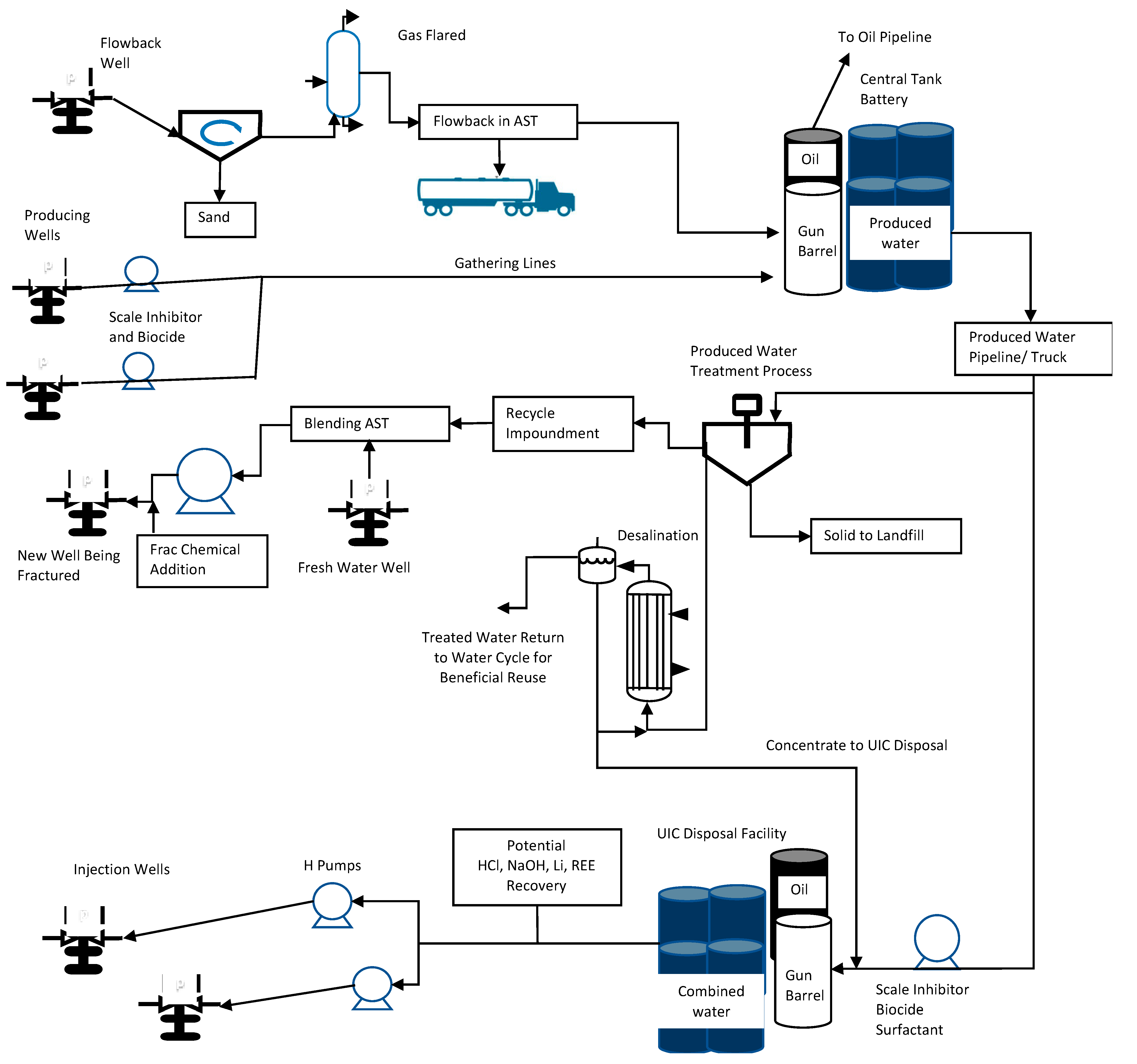

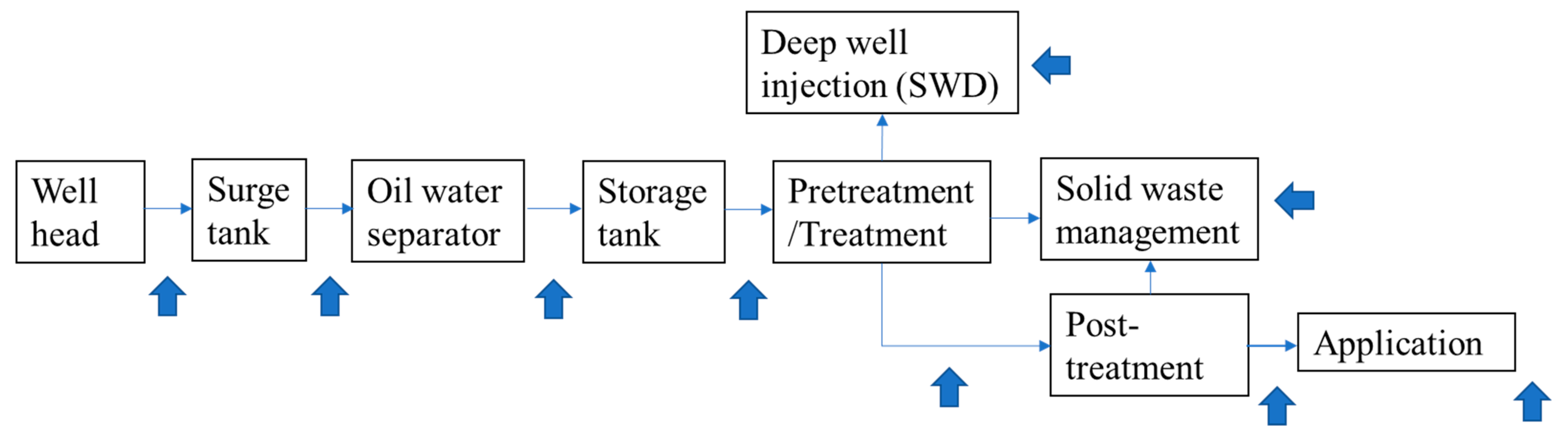
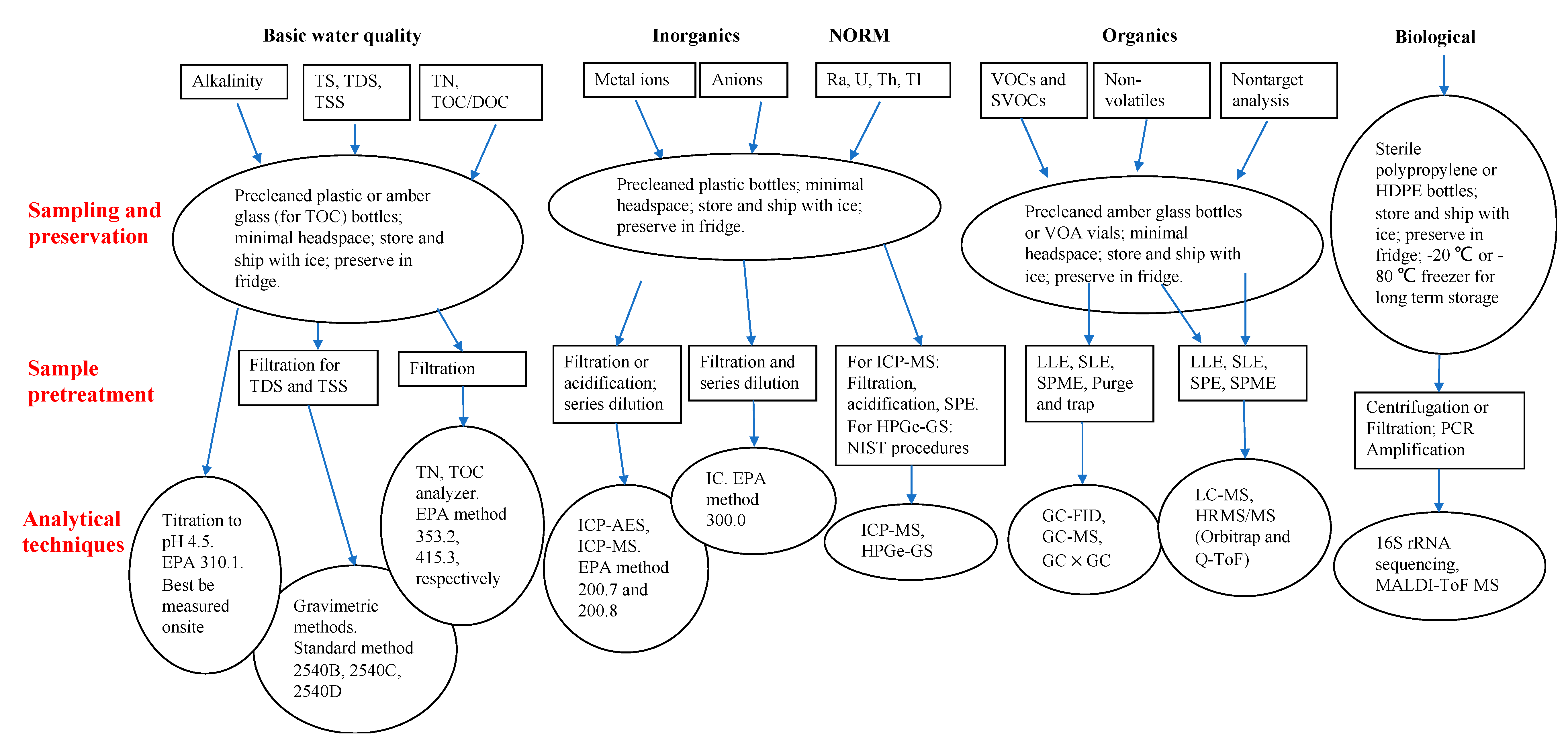
| Additive | Example of Chemical | Chemical Purpose | Concentration (of Total Fluid) |
|---|---|---|---|
| Acid | Hydrochloric acid | Help dissolve minerals and initiate cracks in the rock | 0.07–0.15% |
| Biocide | Glutaraldehyde | Eliminate bacteria that produce corrosive by-products | 0.075–0.06% |
| Breaker | Ammonium persulfate | Allow a delayed break down of the gel | 0.02–0.06% |
| Clay stabilizer | Choline chloride | Prevent clays from swelling or shifting | 0.05–0.2% |
| Corrosion inhibitor | Methanol | Product stabilizer and/or winterizing agent | 0.002–0.004% |
| Cross-linker | Petroleum distillate | Carrier fluid for borate or zirconate crosslinker | 0.007–0.032% |
| Friction reducer | Polyacrylamide | “Slick”, the water to minimize friction | 0.05–0.07% |
| Gelling agent | Guar gum | Thicken water to suspend the sand | 0.05–0.5% |
| Iron control | Citric acid | Prevent precipitation of metal oxides | 0.006 −0.011% |
| Non-emulsifier | Lauryl sulfate | Prevent the formation of emulsions in the fracture fluid | |
| pH adjusting agent | Sodium hydroxide | Adjust the pH of the fluid to maintain the effectiveness of other components, such as crosslinkers | 0.01–0.011% |
| Scale inhibitor | Sodium polycarboxylate | Prevent scale deposits in the pipe | 0.075–0.12% |
| Surfactant | Lauryl sulfate | Increase the viscosity of the fracture fluid | 0.05–0.1% |
| Level | Use | Description | Parameters | Frequency |
|---|---|---|---|---|
| Tier 1 | Continuous monitoring, bulk testing, KPI rapid analysis, process control | In-Line Sensors Field Parameters Filter Analysis | Flow, TSS, TDS, TOC, pH, ORP, iron, H2S, TPH, level sensing, carbonate | Realtime, continuous, and routine |
| Tier 2 | Detailed characterization, routine monitoring, and Tier 1 data verification | Conventional Lab Testing | Wet chemistry, ICP-OES, ICP-MS, GC, GC-MS, HPLC | Baseline, quarterly, when experiencing data excursions in Tier1. Proving up treatment efficacy and reliability, beneficial reuse investigation |
| Tier 3 | NPDES discharge compliance, modeling treatment technology; Waste disposal profile generation; Risk assessment and data capture for fate/transport modeling. | Unconventional Lab Testing; WET Testing | LC-MS, Gamma Spec, High Res GC-MS; Acute and chronic toxicity | When evaluating technology and management processes. As per permit/regulatory agency |
| Leachate Testing | TCLP, SPLP, LEAF testing of residual waste | |||
| Bio-mobility and accumulation testing | Tier 1,2,4 analysis of treated effluent on soil, plant, tissue samples | |||
| Tier 4 | Source apportionment, fingerprinting | SEM/EDX, XRD, FEEM, biomarker analysis, isotopic analysis | Evaluating technology and management process. Basic research for method development. Event response. Beneficial reuse investigations. | |
| Magnetic Sector | Quadrupole | Quadrupole Ion Trap | Time of Flight (ToF) | Orbitrap | FT-ICR | |
|---|---|---|---|---|---|---|
| Mass range (Da) | 15000 | 4000 | 4000 | Unlimited | >104 | >104 |
| Resolving power | 102–105 | 4000 | 103–104 | 15,000 | >105 | >106 |
| Mass accuracy (ppm) | 1–5 | 100 | 50–100 | 5–50 | 2–5 | 1–5 |
| Scan speed (Hz) | 0.1–20 | 1–20 | 1–30 | 101–106 | 10−1–101 | 10−2–101 |
| MS/MS | Excellent | Great | Great | Great | Great | Great |
| Cost | $$$$ | $ | $ | $$–$$$ | $$$ | $$$$ |
| Analytes | EPA-Approved Methods (Water Matrix) | Suitable for PW Analysis? |
|---|---|---|
| Basic water quality | ||
| Alkalinity | EPA Method 310.1 and 310.2 (Drinking, surface, and saline waters; domestic and industrial wastes) | EPA Method 310.1 is suitable for PW. |
| TS | Standard method 2540B (Drinking, surface, and saline waters; domestic and industrial wastewaters) | Yes, range up to 20,000 mg/L |
| TDS | Standard method 2540C (Drinking, surface, and saline waters; domestic and industrial wastewaters) | Yes, range up to 20,000 mg/L |
| TSS | Standard method 2540D (Drinking, surface, and saline waters; domestic and industrial wastewaters) | Yes, range up to 20,000 mg/L |
| TN | EPA Method 353.2: Inorganic nitrite and nitrate; EPA method 351.2 and 351.4: organic nitrogen and ammonia (drinking, surface, and saline waters; domestic and industrial wastes) | Yes, with proper sample preparation |
| TOC/DOC | EPA Method 415.3 or Standard methods 5310C (Source waters and drinking water) | Yes, with proper sample preparation |
| pH | EPA 150.1 (Drinking, surface, and saline waters; domestic and industrial wastes and acid rain) | Yes, with proper sample preparation |
| Inorganic | ||
| Metal ions | EPA 200.7: ICP-AES, EPA 200.8/6020 B: ICP-MS (Drinking, surface, and groundwater; wastewaters, sludges, and solid samples) | EPA methods can be used for PW with a series of dilutions to eliminate the impact of high Na+ concentration. ICP-AES and ICP-OES are reliable approaches. |
| Anions | EPA 300.0 (drinking water, surface water, mixed domestic and industrial wastewaters, groundwater, reagent waters, solids); EPA 300.1 (reagent water, surface water, groundwater, finished drinking water) | EPA 300.0 is suitable for PW with a series of dilutions to eliminate the impact of high Cl- concentration. IC is a reliable approach. |
| Organic | ||
| Non-Pesticide (120 parameters) | EPA 551, 601–625, 632, 1613B, etc. (drinking, ambient water, wastewater, sediment) | EPA methods based on GC may be suitable to analyze VOCs and SVOCs in PW with proper sample pretreatment, such as purge and trap, LLE, SPE, or SPME. However, the number of compounds is limited. |
| Pesticide (70 parameters) | EPA 553, 605, 610, etc. (drinking, ambient water, wastewater, sediment) | With proper sample pretreatment, such as LLE, SPE, or SPME, EPA methods based on LC may be suitable to analyze non-volatiles compounds in FPW. However, LC-HRMS/MS (Orbitrap and Q-ToF) would be more reliable approaches. |
| Nontarget analysis | No methods. | Nontarget analysis using HRMS/MS (confidence Levels 1 and 2) will be required to identify the unknown compounds in PW. |
| Biological Bacterial | EPA 1600, 1603, 1622, 1680, etc. (Wastewater and Sewage sludge, ambient water) | EPA methods detect limited types of bacteria. 16S rRNA sequencing and MALDI-ToF MS are reliable approaches for FPW. |
| NORM Ra, U, Th, Tl | EPA 900.0, 901.1, 903.0, 903.1. (Drinking water) | EPA methods cannot be used for PW. HPGe-GS (lower MDL and more accurate) and ICP-MS (more efficient) are reliable approaches. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, W.; Lin, L.; Xu, X.; Cheng, X.; Zhang, Y.; Hall, R.; Xu, P. A Critical Review of Analytical Methods for Comprehensive Characterization of Produced Water. Water 2021, 13, 183. https://doi.org/10.3390/w13020183
Jiang W, Lin L, Xu X, Cheng X, Zhang Y, Hall R, Xu P. A Critical Review of Analytical Methods for Comprehensive Characterization of Produced Water. Water. 2021; 13(2):183. https://doi.org/10.3390/w13020183
Chicago/Turabian StyleJiang, Wenbin, Lu Lin, Xuesong Xu, Xiaoxiao Cheng, Yanyan Zhang, Ryan Hall, and Pei Xu. 2021. "A Critical Review of Analytical Methods for Comprehensive Characterization of Produced Water" Water 13, no. 2: 183. https://doi.org/10.3390/w13020183
APA StyleJiang, W., Lin, L., Xu, X., Cheng, X., Zhang, Y., Hall, R., & Xu, P. (2021). A Critical Review of Analytical Methods for Comprehensive Characterization of Produced Water. Water, 13(2), 183. https://doi.org/10.3390/w13020183








