Heterogeneous Photo-Fenton Catalytic Degradation of Practical Pharmaceutical Wastewater by Modified Attapulgite Supported Multi-Metal Oxides
Abstract
1. Introduction
2. Materials and Methods
2.1. Characteristics of Pharmaceutical Wastewater
2.2. Preparation of the Catalyst
2.3. The Experimental Methods
2.4. Analytical Methods
3. Results and Discussion
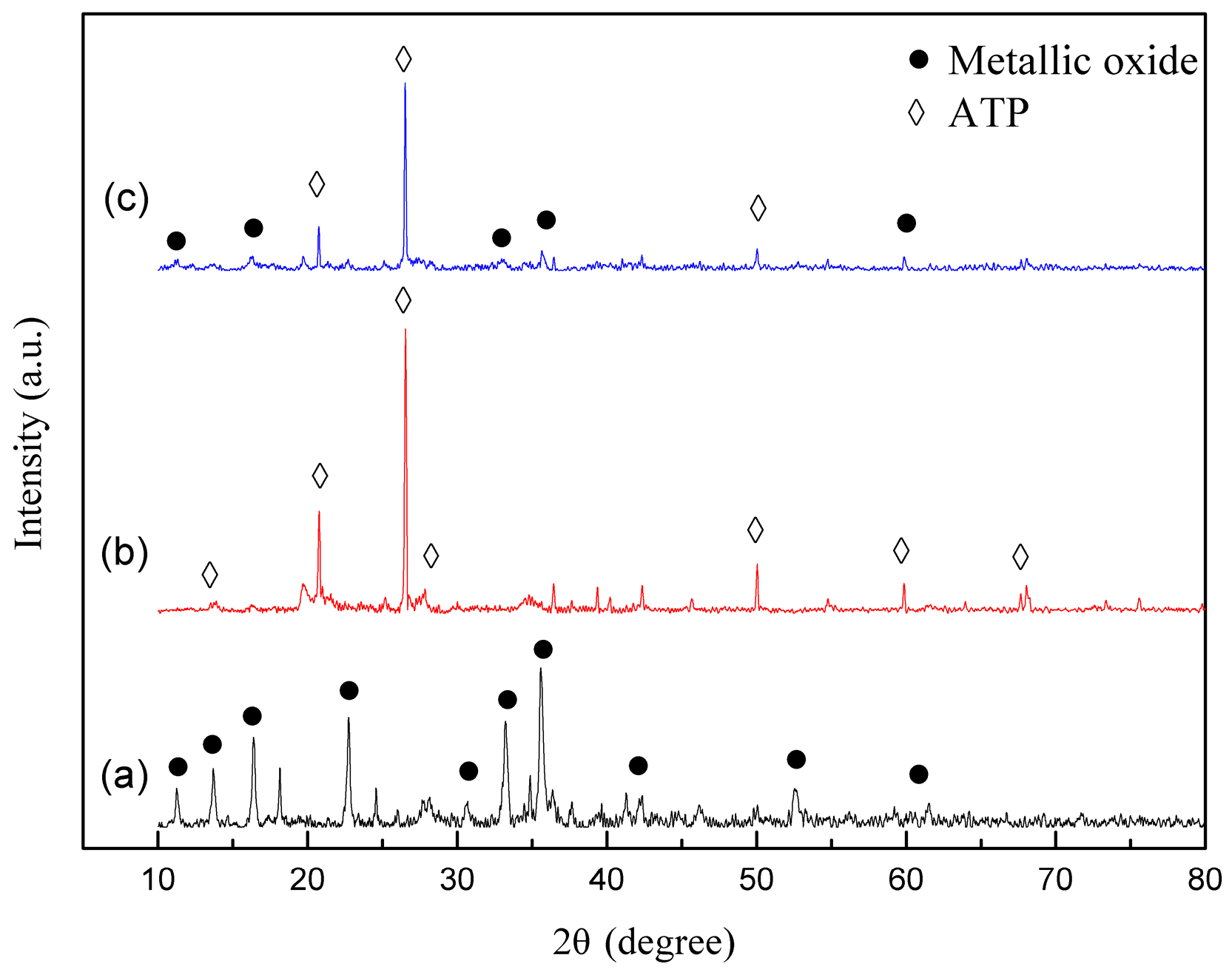
3.1. Characterization of Catalyst
3.2. Photo-Fenton Catalytic Degradation Performance
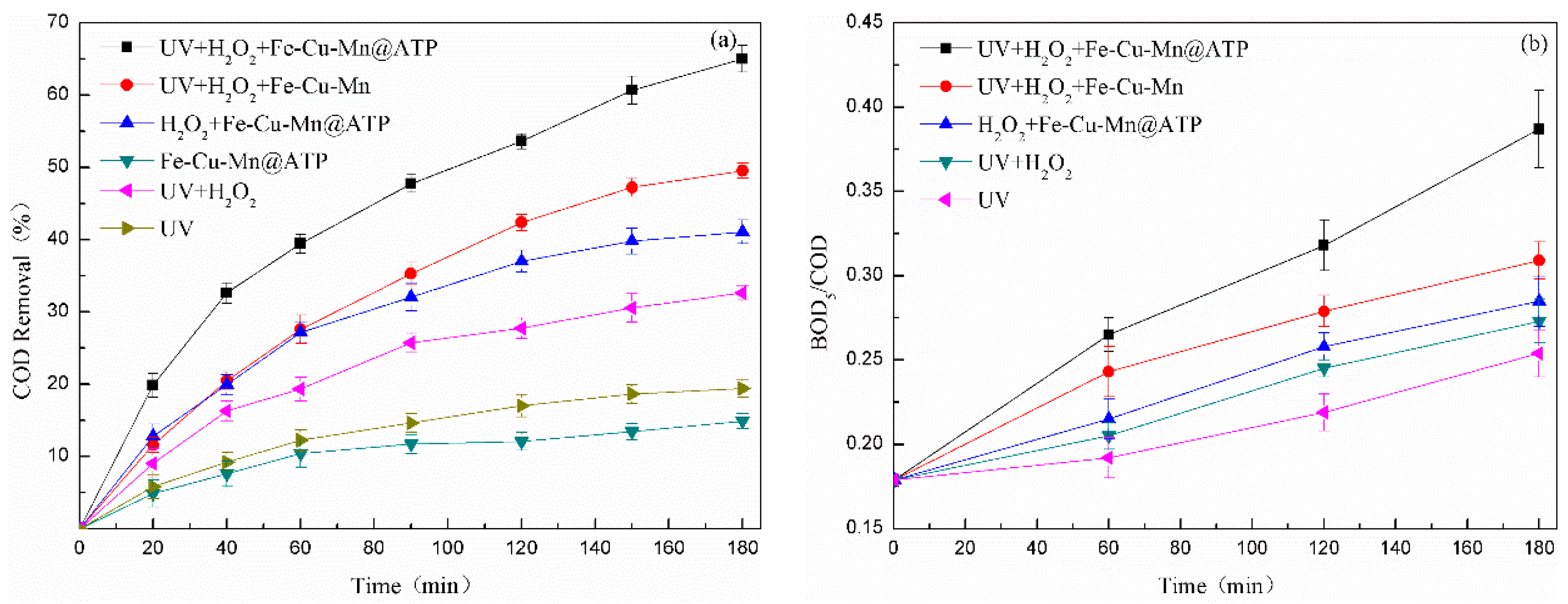
3.2.1. Effects of Different Treatment Systems
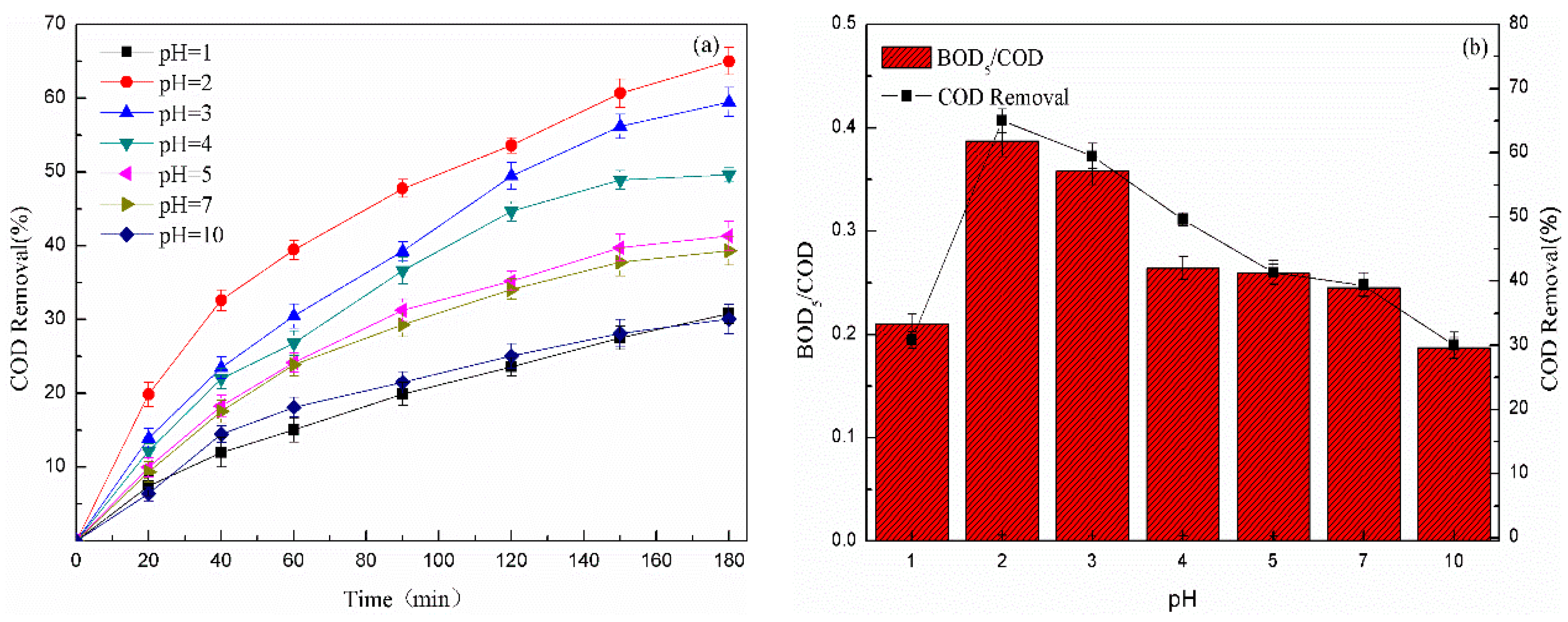
3.2.2. Effect of the Initial pH
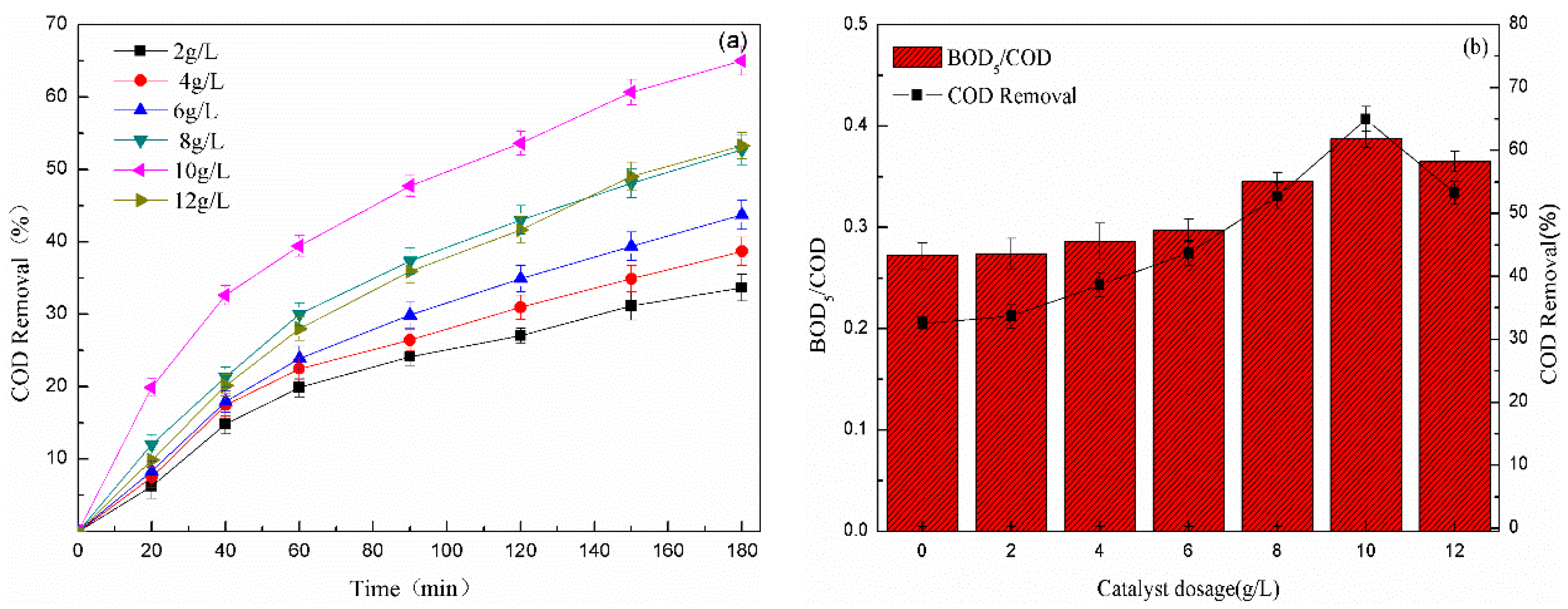
3.2.3. Effect of the Catalyst Dosage
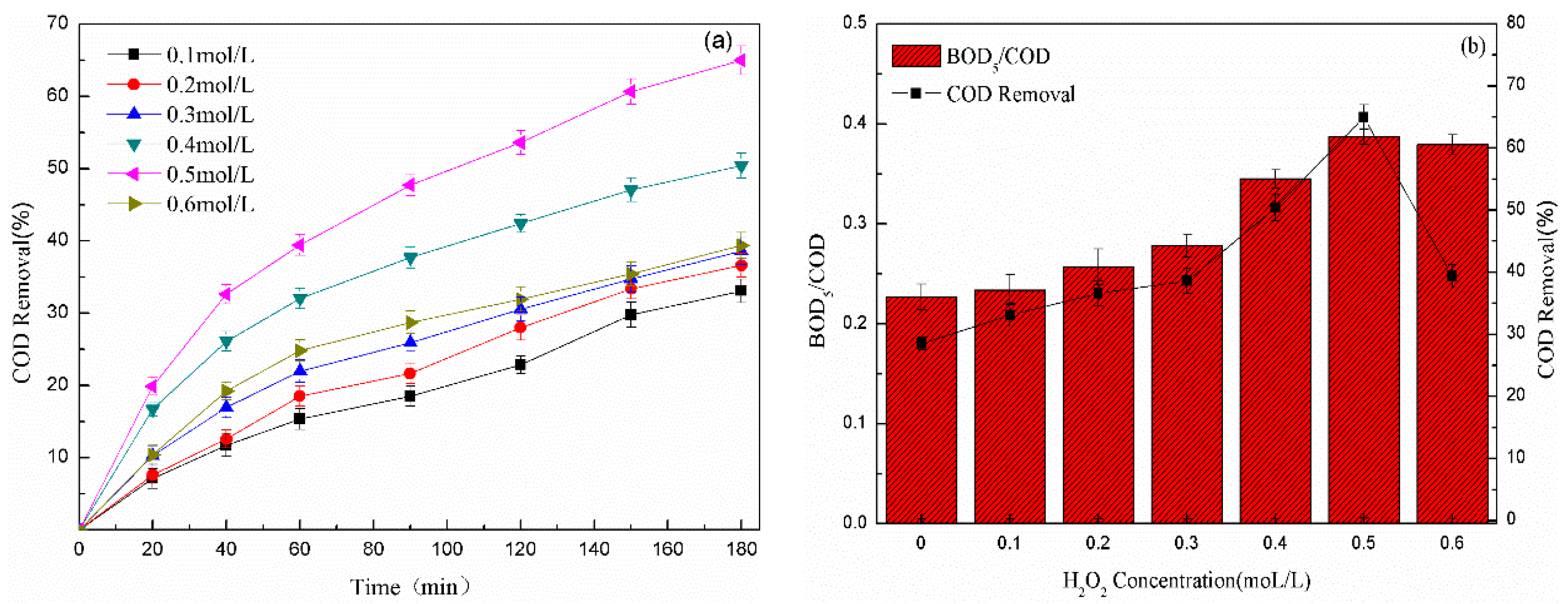
3.2.4. Effect of the Peroxide Concentration
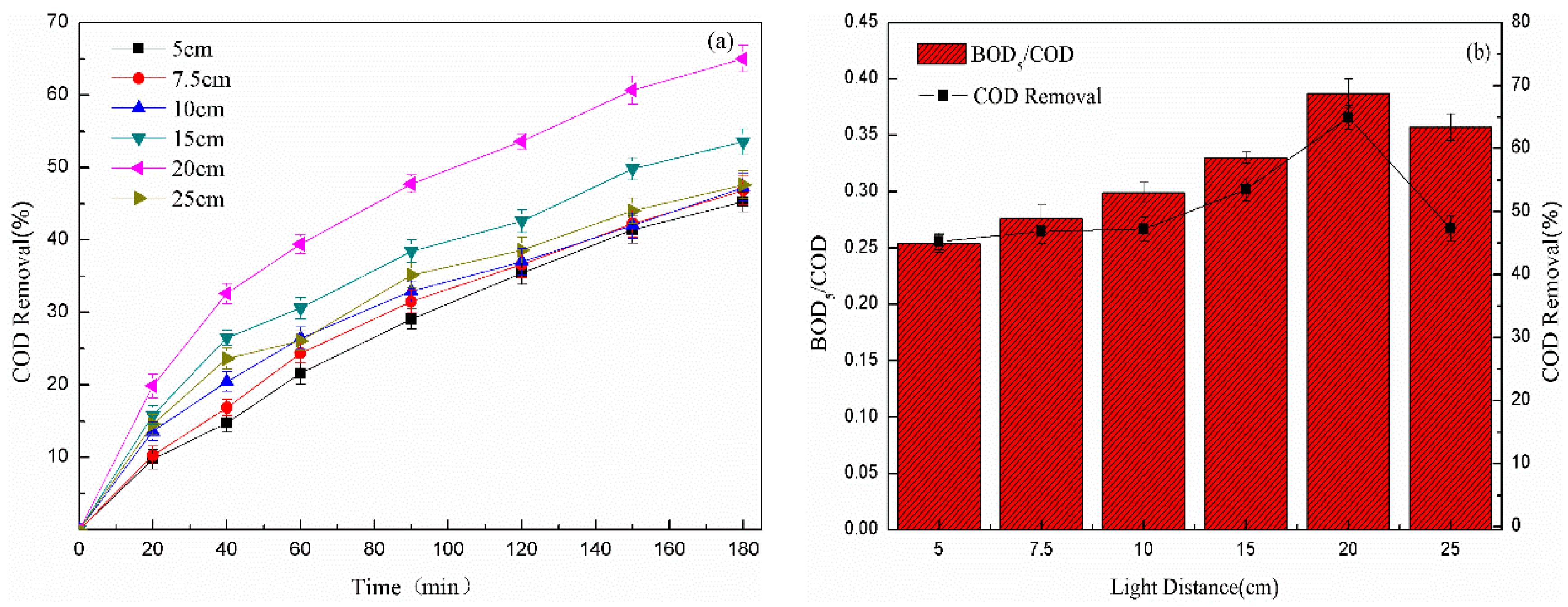
3.2.5. Effect of UV Light Distance
3.3. Variations of Organic Compounds
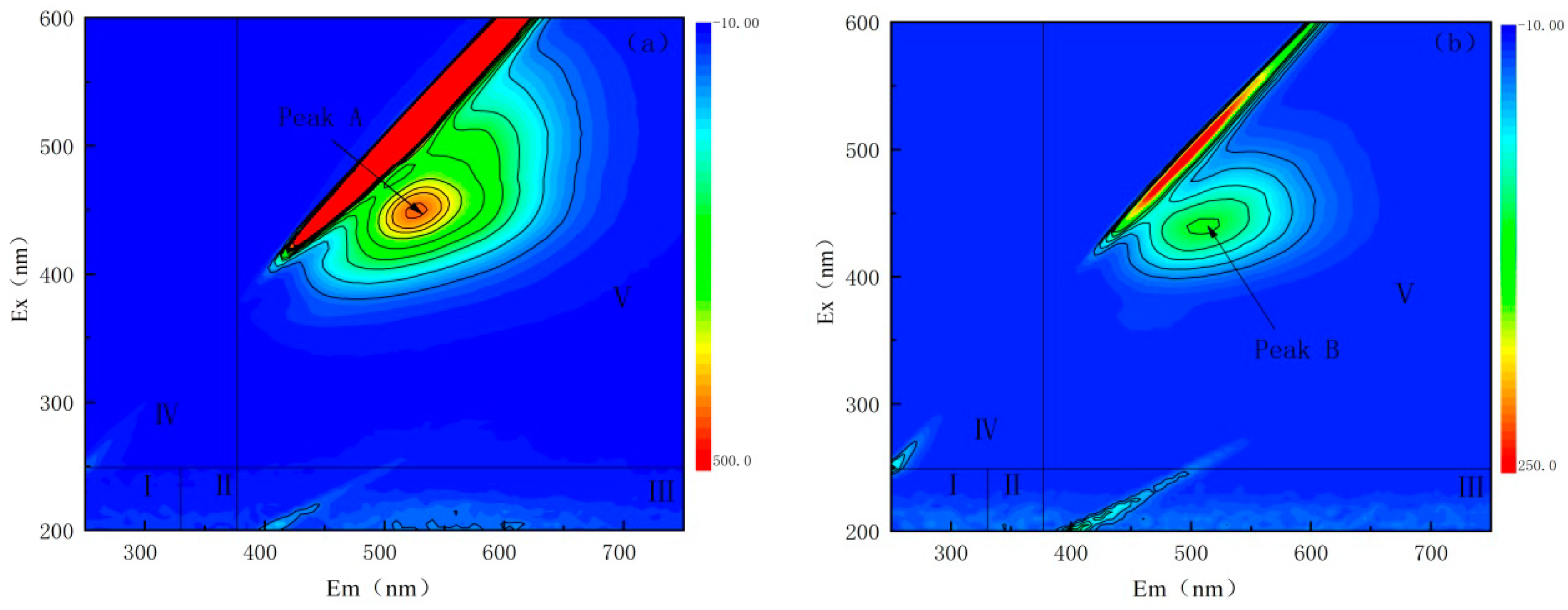
3.3.1. 3D-EEM the Fluorescence Spectrum Analysis
3.3.2. GC-MS Organic Compounds Change Analysis
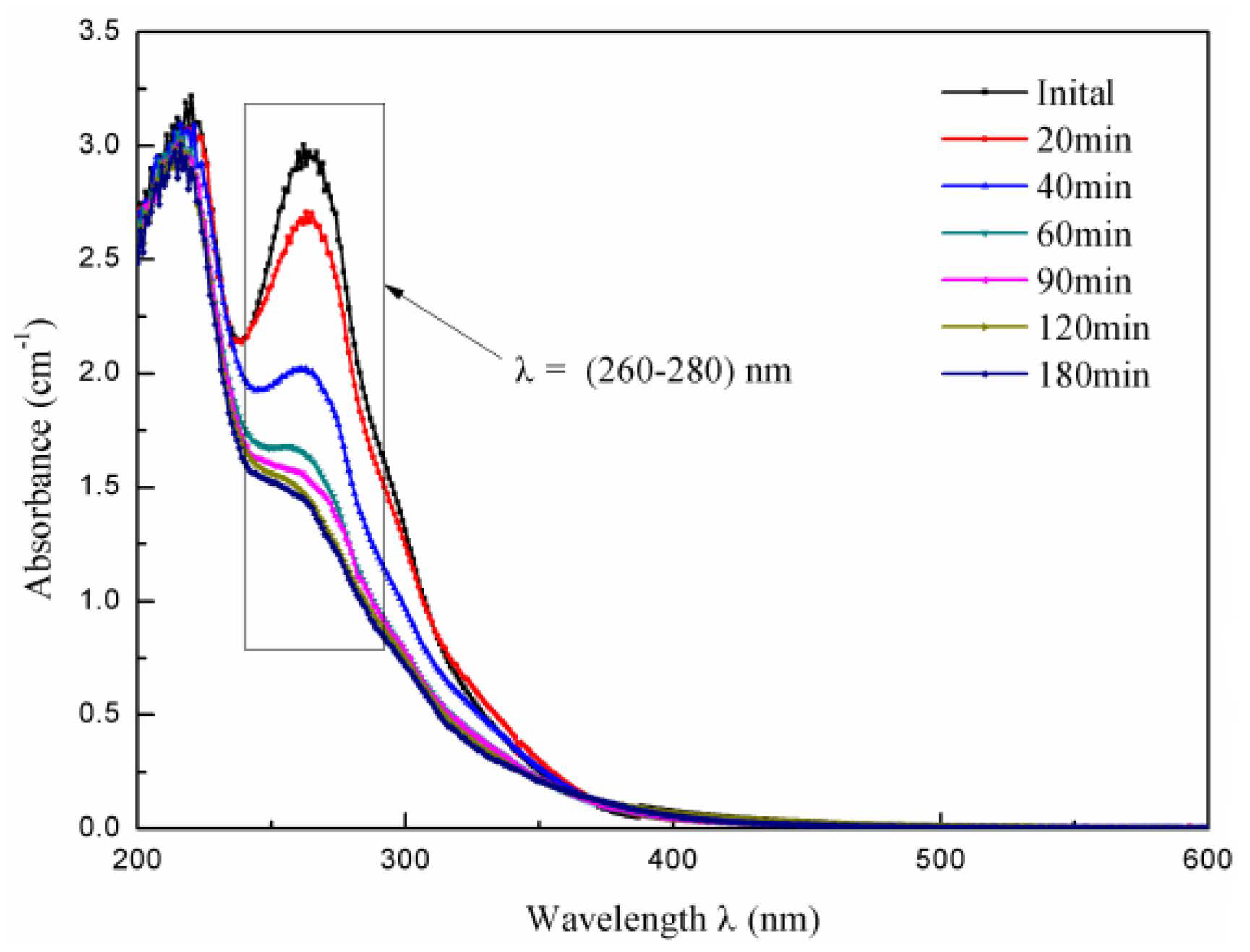
3.3.3. UV-Vis Absorption Spectrum Analysis
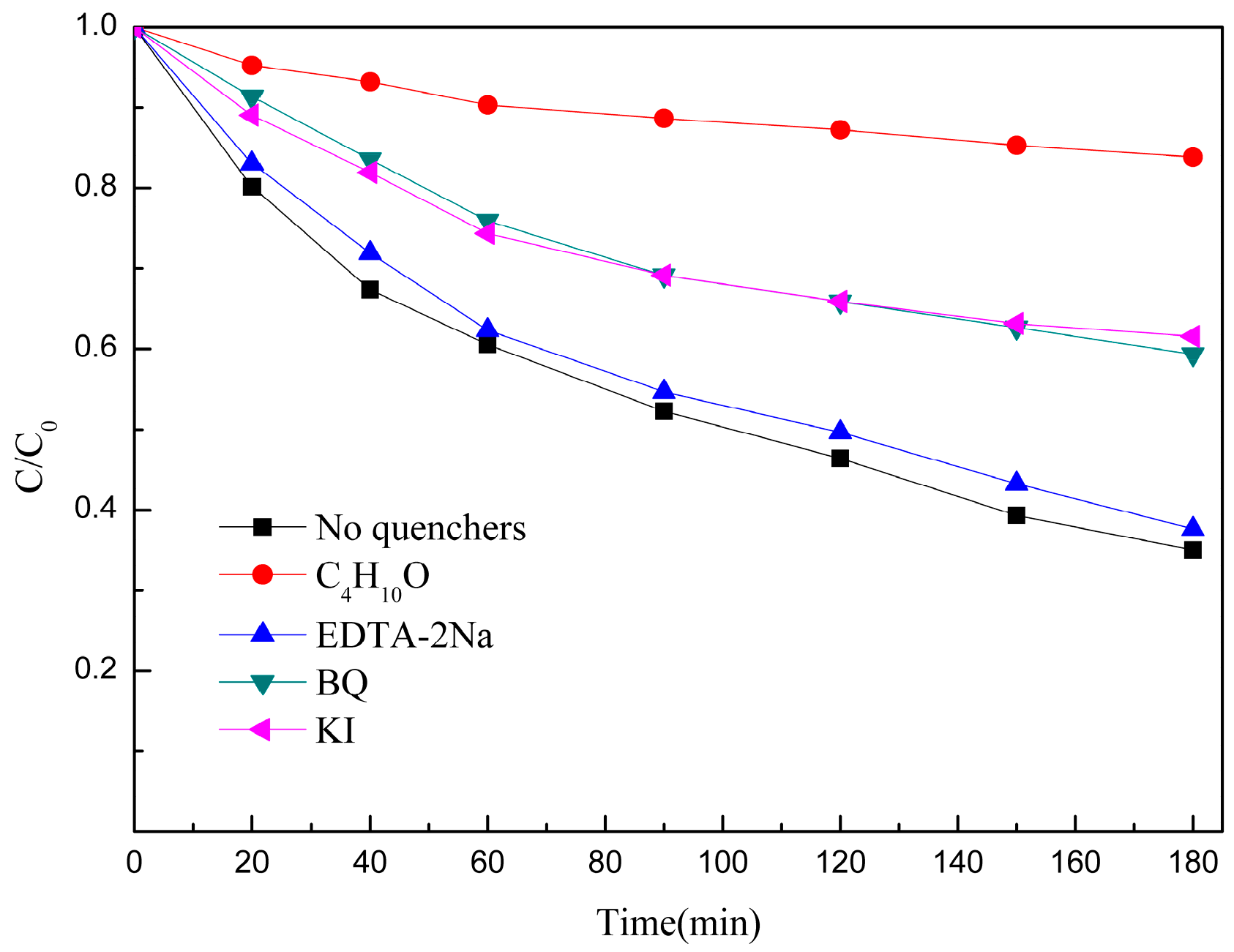
3.4. Degradation Mechanism of Photo-Fenton Catalytic Oxidation
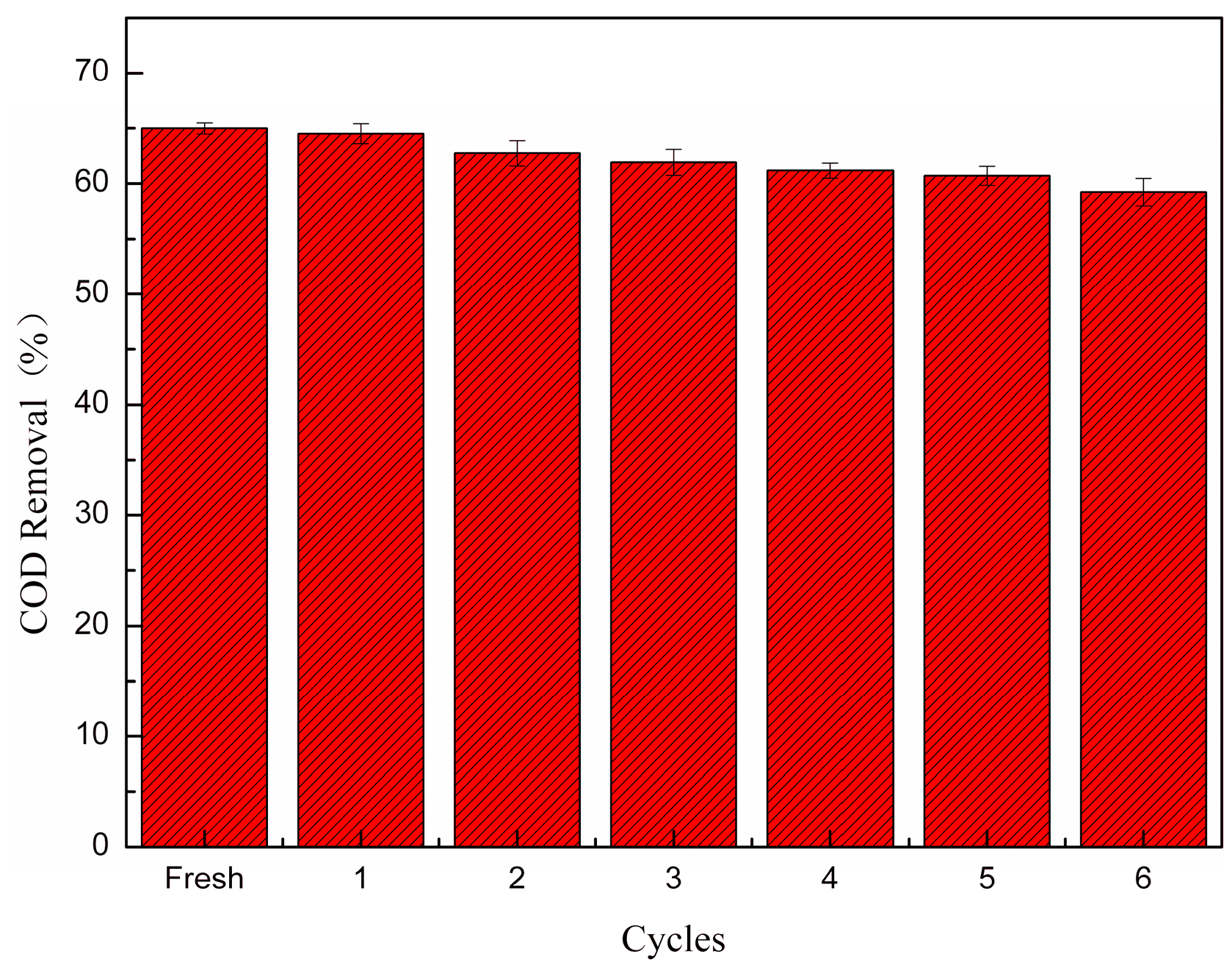
3.5. The Stability of the Catalyst
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Anliker, S.; Patrick, M.; Fenner, K.; Singer, H. Quantification of Active Ingredient Losses from Formulating Pharmaceutical Industries and Contribution to Wastewater Treatment Plant Emissions. Environ. Sci. Technol. 2020, 54, 15046–15056. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.L.; Zhang, X.T.; Wang, Q.; Yao, P.L. Metagenomic Analysis of Antibiotic Resistant Bacteria and Resistance Genes in a Pharmaceutical and Chemical Wastewater Treatment Plant. Huan Jing Ke Xue 2020, 41, 313–320. [Google Scholar]
- Gadipelly, C.; Pérez-González, A.; Yadav, G.D.; Ortiz, I.; Ibáñez, R.; Rathod, V.K.; Marathe, K.V. Pharmaceutical Industry Wastewater: Review of the Technologies for Water Treatment and Reuse. Ind. Eng. Chem. Res. 2014, 53, 11571–11592. [Google Scholar] [CrossRef]
- Behfar, R.D.R.; Heydari, R. Pharmaceutical Wastewater Chemical Oxygen Demand Reduction: Electro-Fenton, UV-enhanced Electro-Fenton and Activated Sludge. Int. J. Eng. 2019, 32, 1710–1715. [Google Scholar]
- Jhunjhunwala, A.; Pathak, U.; Sarkar, K.K.; Majee, S.; Mandal, D.D.; Mandal, T. Removal of levosulpiride from pharmaceutical wastewater using an advanced integrated treatment strategy comprising physical, chemical, and biological treatment. Environ. Prog. Sustain. Energy 2020. [Google Scholar] [CrossRef]
- Kirchon, A.; Zhang, P.; Li, J.; Joseph, E.A.; Chen, W.; Zhou, H.C. Effect of Isomorphic Metal Substitution on the Fenton and Photo-Fenton Degradation of Methylene Blue Using Fe-Based Metal-Organic Frameworks. ACS Appl. Mater. Interfaces 2020, 12, 9292–9299. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, C.; Xu, B.; Qi, F.; Chu, W. Degradation of benzotriazole by a novel Fenton-like reaction with mesoporous Cu/MnO2: Combination of adsorption and catalysis oxidation. Appl. Catal. B Environ. 2016, 199, 447–457. [Google Scholar] [CrossRef]
- Chong, S.; Zhang, G.; Zhang, N.; Liu, Y.; Huang, T.; Chang, H. Diclofenac degradation in water by FeCeOx catalyzed H2O2: Influencing factors, mechanism and pathways. J. Hazard. Mater. 2017, 334, 150–159. [Google Scholar] [CrossRef]
- Cen, H.; Nan, Z. Monodisperse Zn-doped Fe3O4 formation and photo-Fenton activity for degradation of rhodamine B in water. J. Phys. Chem. Solids 2018, 121, 1–7. [Google Scholar] [CrossRef]
- Ran, G.; Li, Q. Degradation of refractory organic compounds from dinitrodiazophenol containing industrial wastewater through UV/H(2)O(2) and UV/PS processes. Environ. Sci. Pollut. Res. Int. 2020, 27, 6042–6051. [Google Scholar] [CrossRef]
- Xu, Z.; Zheng, R.; Chen, Y.; Zhu, J.; Bian, Z. Ordered mesoporous Fe/TiO2 with light enhanced photo-Fenton activity. Chin. J. Catal. 2019, 40, 631–637. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, X.; Zhang, X.; Zhang, Y.; Wang, Y.; Sun, S.; Wu, W. Nanostructured Semiconductor Supported Iron Catalysts for Heterogeneous Photo-Fenton Oxidation: A Review. J. Mater. Chem. A 2020, 8, 15513–15546. [Google Scholar]
- Gogoi, A.; Navgire, M.; Sarma, K.C.; Gogoi, P. Fe3O4-CeO2 metal oxide nanocomposite as a Fenton-like heterogeneous catalyst for degradation of catechol. Chem. Eng. J. 2017, 311, 153–162. [Google Scholar] [CrossRef]
- Ahmad, I. Comparative study of metal (Al, Mg, Ni, Cu and Ag) doped ZnO/g-C3N4 composites: Efficient photocatalysts for the degradation of organic pollutants. Sep. Purif. Technol. 2020, 251, 117372. [Google Scholar] [CrossRef]
- Nichela, D.A.; Berkovic, A.M.; Costante, M.R.; Juliarena, M.P.; García Einschlag, F.S. Nitrobenzene degradation in Fenton-like systems using Cu(II) as catalyst. Comparison between Cu(II)- and Fe(III)-based systems. Chem. Eng. J. 2013, 228, 1148–1157. [Google Scholar] [CrossRef]
- Zhang, X.; Ding, Y.; Tang, H.; Han, X.; Zhu, L.; Wang, N. Degradation of bisphenol A by hydrogen peroxide activated with CuFeO2 microparticles as a heterogeneous Fenton-like catalyst: Efficiency, stability and mechanism. Chem. Eng. J. 2014, 236, 251–262. [Google Scholar] [CrossRef]
- Li, K.; Zhao, Y.; Song, C.; Guo, X. Magnetic ordered mesoporous Fe3O4/CeO2 composites with synergy of adsorption and Fenton catalysis. Appl. Surf. Sci. 2017, 425, 526–534. [Google Scholar] [CrossRef]
- Jauhar, S.; Singhal, S.; Dhiman, M. Manganese substituted cobalt ferrites as efficient catalysts for H2O2 assisted degradation of cationic and anionic dyes: Their synthesis and characterization. Appl. Catal. A Gen. 2014, 486, 210–218. [Google Scholar] [CrossRef]
- Su, Y.; Li, M.; Ge, M. Catalytic Degradation of Poly(Vinyl Alcohol) by FexMnyCuzOw/γ-Al2O3 Nano-particles Catalyst Using Box-Behnken Design. Fibers Polym. 2019, 20, 1774–1783. [Google Scholar] [CrossRef]
- Litter, M.I.; Slodowicz, M. An overview on heterogeneous Fenton and photoFenton reactions using zerovalent iron materials. J. Adv. Oxid. Technol. 2017, 20, 160–164. [Google Scholar] [CrossRef]
- Singh, L.; Rekha, P.; Chand, S. Cu-impregnated zeolite Y as highly active and stable heterogeneous Fenton-like catalyst for degradation of Congo red dye. Sep. Purif. Technol. 2016, 170, 321–336. [Google Scholar] [CrossRef]
- Viegas, R.M.C.; Mestre, A.S.; Mesquita, E.; Campinas, M.; Andrade, M.A.; Carvalho, A.P.; Rosa, M.J. Assessing the applicability of a new carob waste-derived powdered activated carbon to control pharmaceutical compounds in wastewater treatment. Sci. Total Environ. 2020, 743, 140791. [Google Scholar] [CrossRef] [PubMed]
- Liyanage, A.S.; Canaday, S.; Pittman, C.U., Jr.; Mlsna, T. Rapid remediation of pharmaceuticals from wastewater using magnetic Fe3O4/Douglas fir biochar adsorbents. Chemosphere 2020, 258, 127336. [Google Scholar] [CrossRef]
- Liu, Y.-X.; Zhong, H.; Li, X.-R.; Bao, Z.-L.; Cheng, Z.-P.; Zhang, Y.-J.; Li, C.-X. Fabrication of attapulgite-based dual responsive composite hydrogel and its efficient adsorption for methyl violet. Environ. Technol. 2020. [Google Scholar] [CrossRef]
- Zhang, T.; Nan, Z.-R. Decolorization of Methylene Blue and Congo Red by attapulgite-based heterogeneous Fenton catalyst. Desalination Water Treat. 2016, 57, 4633–4640. [Google Scholar] [CrossRef]
- He, Y.; Sun, X.; Zhang, P.; Wang, F.; Zhao, Z.; He, C. Cd(II) adsorption from aqueous solutions using modified attapulgite. Res. Chem. Intermed. 2020, 46, 4897–4908. [Google Scholar] [CrossRef]
- Guo, H.J.; Li, Q.L.; Zhang, H.R.; Peng, F.; Xiong, L.; Yao, S.M.; Huang, C.; Chen, X.D. CO2 hydrogenation over acid-activated Attapulgite/Ce0.75Zr0.25O2 nanocomposite supported Cu-ZnO based catalysts. Mol. Catal. 2019, 476, 14. [Google Scholar] [CrossRef]
- Eslami, H.; Ehrampoush, M.H.; Esmaeili, A.; Ebrahimi, A.A.; Ghaneian, M.T.; Falahzadeh, H.; Salmani, M.H. Synthesis of mesoporous Fe-Mn bimetal oxide nanocomposite by aeration co-precipitation method: Physicochemical, structural, and optical properties. Mater. Chem. Phys. 2019, 224, 65–72. [Google Scholar] [CrossRef]
- Association, C.; Washington, D. APHA: Standard methods for the examination of water and wastewater. American. Phys. Educ. Rev. 1995, 24, 481–486. [Google Scholar]
- Ma, C.; Yuan, P.; Jia, S.; Liu, Y.; Zhang, X.; Hou, S.; Zhang, H.; He, Z. Catalytic micro-ozonation by Fe3O4 nanoparticles @ cow-dung ash for advanced treatment of biologically pre-treated leachate. Waste Manag. 2019, 83, 23–32. [Google Scholar] [CrossRef]
- Li, Z.; Xin, Y.; Zhang, Z.; Wu, H.; Wang, P. Rational design of binder-free noble metal/metal oxide arrays with nanocauliflower structure for wide linear range nonenzymatic glucose detection. Sci. Rep. 2015, 5, 10617. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Xu, L.; Song, J.; Xing, R.; Xu, S.; Liu, D.; Song, H. Ultrasensitive non-enzymatic glucose sensor based on three-dimensional network of ZnO-CuO hierarchical nanocomposites by electrospinning. Sci. Rep. 2014, 4, 7382. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Li, Q.; Zhang, H.; Xiong, L.; Peng, F.; Yao, S.; Chen, X. Attapulgite supported Cu-Fe-Co based catalyst combination system for CO hydrogenation to lower alcohols. J. Fuel Chem. Technol. 2019, 47, 1346–1356. [Google Scholar] [CrossRef]
- Moradi, S.; Sobhgol, S.A.; Hayati, F.; Isari, A.A.; Kakavandi, B.; Bashardoust, P.; Anvaripour, B. Performance and reaction mechanism of MgO/ZnO/Graphene ternary nanocomposite in coupling with LED and ultrasound waves for the degradation of sulfamethoxazole and pharmaceutical wastewater. Sep. Purif. Technol. 2020, 251, 117373. [Google Scholar] [CrossRef]
- Mahouachi, L.; Rastogi, T.; Palm, W.U.; Ghorbel-Abid, I.; Ben Hassen Chehimi, D.; Kummerer, K. Natural clay as a sorbent to remove pharmaceutical micropollutants from wastewater. Chemosphere 2020, 258, 127213. [Google Scholar] [CrossRef]
- Naumczyk, J.; Prokurat, I.; Marcinowski, P. Landfill Leachates Treatment by H2O2/UV, O3/H2O2, Modified Fenton, and Modified Photo-Fenton Methods. Int. J. Photoenergy 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Yazici Guvenc, S.; Varank, G. Degradation of refractory organics in concentrated leachate by the Fenton process: Central composite design for process optimization. Front. Environ. Sci. Eng. 2020, 15, 2. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Z.; Wang, J. Fenton-like degradation of sulfamethoxazole in Cu0/Zn0-air system over a broad pH range: Performance, kinetics and mechanism. Chem. Eng. J. 2020, 403, 126320. [Google Scholar] [CrossRef]
- Zhang, Q.; Peng, Y.; Deng, F.; Wang, M.; Chen, D. Porous Z-scheme MnO2/Mn-modified alkalinized g-C3N4 heterojunction with excellent Fenton-like photocatalytic activity for efficient degradation of pharmaceutical pollutants. Sep. Purif. Technol. 2020, 246, 116890. [Google Scholar] [CrossRef]
- Ma, C.; He, Z.; Jia, S.; Zhang, X.; Hou, S. Treatment of stabilized landfill leachate by Fenton-like process using Fe3O4 particles decorated Zr-pillared bentonite. Ecotoxicol. Environ. Saf. 2018, 161, 489–496. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Le, G.H.; Le, C.H.; Nguyen, M.B.; Quan, T.T.T.; Pham, T.T.T.; Vu, T.A. Atomic implantation synthesis of Fe-Cu/SBA-15 nanocomposite as a heterogeneous Fenton-like catalyst for enhanced degradation of DDT. Mater. Res. Express 2018, 5, 115005. [Google Scholar] [CrossRef]
- Lou, Z.; Zhou, Z.; Zhang, W.; Zhang, X.; Hu, X.; Liu, P.; Zhang, H. Magnetized bentonite by Fe3O4 nanoparticles treated as adsorbent for methylene blue removal from aqueous solution: Synthesis, characterization, mechanism, kinetics and regeneration. J. Taiwan Inst. Chem. Eng. 2015, 49, 199–205. [Google Scholar] [CrossRef]
- Kasiri, M.; Khataee, A. Photooxidative decolorization of two organic dyes with different chemical structures by UV/H2O2 process: Experimental design. Desalination 2011, 270, 151–159. [Google Scholar] [CrossRef]
- Chen, W.; Westerhoff, P.; Leenheer, J.A.; Booksh, K. Fluorescence excitation-emission matrix regional integration to quantify spectra for dissolved organic matter. Environ. Sci. Technol. 2003, 37, 5701–5710. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-T.; Li, Y.; Zhang, X.-Q.; Li, J.-F.; Luo, Y.-N.; Wang, C.-W. Fabrication of a magnetically separable Cu2ZnSnS4/ZnFe2O4 p-n heterostructured nano-photocatalyst for synergistic enhancement of photocatalytic activity combining with photo-Fenton reaction. Appl. Surf. Sci. 2019, 479, 86–95. [Google Scholar] [CrossRef]
- Chen, T.; Zhu, Z.; Zhang, H.; Shen, X.; Qiu, Y.; Yin, D. Enhanced Removal of Veterinary Antibiotic Florfenicol by a Cu-Based Fenton-like Catalyst with Wide pH Adaptability and High Efficiency. ACS Omega 2019, 4, 1982–1994. [Google Scholar] [CrossRef]
- Abbasi Asl, E.; Haghighi, M.; Talati, A. Enhanced simulated sunlight-driven magnetic MgAl2O4-AC nanophotocatalyst for efficient degradation of organic dyes. Sep. Purif. Technol. 2020, 251, 117003. [Google Scholar] [CrossRef]
- Diao, Y.; Yan, Z.; Guo, M.; Wang, X. Magnetic multi-metal co-doped magnesium ferrite nanoparticles: An efficient visible light-assisted heterogeneous Fenton-like catalyst synthesized from saprolite laterite ore. J. Hazard. Mater. 2018, 344, 829–838. [Google Scholar] [CrossRef]
- Lv, X.; Ma, Y.; Li, Y.; Yang, Q. Heterogeneous Fenton-Like Catalytic Degradation of 2,4-Dichlorophenoxyacetic Acid by Nano-Scale Zero-Valent Iron Assembled on Magnetite Nanoparticles. Water 2020, 12, 2909. [Google Scholar] [CrossRef]
- di Luca, C.; Massa, P.; Grau, J.M.; Marchetti, S.G.; Fenoglio, R.; Haure, P. Highly dispersed Fe3+-Al2O3 for the Fenton-like oxidation of phenol in a continuous up-flow fixed bed reactor. Enhancing catalyst stability through operating conditions. Appl. Catal. B Environ. 2018, 237, 1110–1123. [Google Scholar] [CrossRef]

| Parameter | Unit | Concentration |
|---|---|---|
| pH | - | 1.0 ± 0.1 |
| COD | mg/L | 18,585 ± 265 |
| BOD5 | mg/L | 3350 ± 150 |
| BOD5/COD | - | 0.179 ± 0.015 |
| NH3-N | mg/L | 1554.9 ± 17.3 |
| TN | mg/L | 1780 ± 25 |
| NO3-N | mg/L | 88.5 ± 9 |
| NO2-N | mg/L | 10.2 ± 0.8 |
| Organic Compounds | Initial Values | Final Values | ||
|---|---|---|---|---|
| Peak Area | P (%) | Peak Area | P (%) | |
| Pyrimidine, 2-chloro | 134,337,798 | 18.86 | 112,957,014 | 31.23 |
| 2,6-Dichloropyrazine | 311,329,997 | 43.72 | 2,072,863 | 0.57 |
| Pyrimidine, 4,6-dichloro | 109,383,005 | 15.36 | 86,220,168 | 23.84 |
| Diethyl carbitol | 532,383 | 0.07 | N.D. | N.D. |
| 4-Amino-2,6-dichloropyridine | 180,401 | 0.03 | N.D. | N.D. |
| 2,4-Dihydroxybenzaldehyde, 2TMS derivative | 358,603 | 0.05 | 472,319 | 0.13 |
| Cyclo-penta-siloxane, deca-methyl | 3,375,447 | 0.47 | 2,703,467 | 0.75 |
| Pyrimidine, 2,4,6-trichloro | 145,047,498 | 20.37 | 21,144,132 | 5.85 |
| Benzoic acid, 2,6-dichloro-, 2-acetylphenyl ester | 158,224 | 0.02 | N.D. | N.D. |
| Benzene,1-chloro-3,5-difluoro- | 228,041 | 0.03 | N.D. | N.D. |
| Pyrimidine, tetrachloro | 371,995 | 0.05 | N.D. | N.D. |
| 4-Chloroquinoline | 300,677 | 0.04 | N.D. | N.D. |
| Cyclo-hexa-siloxane, dodeca-methyl | 4,558,815 | 0.64 | 3,698,006 | 1.02 |
| 2-Bromo-1,3-thiazole-5-carbaldehyde | 618,021 | 0.09 | N.D. | N.D. |
| Benzenamine, 2,3,4-trichloro | 166,015 | 0.02 | N.D. | N.D. |
| Cyclo-hepta-siloxane, tetra-deca-methyl | 800,715 | 0.11 | 1,155,308 | 0.32 |
| 2,4-Dichloroquinoline | 422,801 | 0.06 | N.D. | N.D. |
| 1,3-Dioxolane | N.D. | N.D. | 240,275 | 0.07 |
| 4-Methyl-2,4-bis(p-hydroxyphenyl) pent-1-ene, 2TMS derivative | N.D. | N.D. | 10,638,907 | 2.94 |
| Imidazo[1,5-a]pyridine, 3-phenyl | N.D. | N.D. | 357,153 | 0.10 |
| Pyrimidine, 4-chloro-2-methyl | N.D. | N.D. | 119,229 | 0.03 |
| N-(2,6-Diethylphenyl)-1,1,1- trifluoro-methane sulfonamide | N.D. | N.D. | 117,811,503 | 32.58 |
| Cyclotrisil-oxane, hexamethyl | N.D. | N.D. | 998,827 | 0.28 |
| Naphthalene, 2-chloro | N.D. | N.D. | 338,243 | 0.09 |
| 2H-imidazole-2-thione, 1,3-dihydro- 4-(2-methylpropyl) | N.D. | N.D. | 177,569 | 0.05 |
| 1-Nonadecanol, TMS derivative | N.D. | N.D. | 236,706 | 0.07 |
| Ethyne, 1-bromo-2-trimethylsilyl | N.D. | N.D. | 99,410 | 0.03 |
| 3,4-Dihydroxyphenylglycol, 4TMS derivative | N.D. | N.D. | 156,565 | 0.04 |
| 2,5,8-Triphenyl benzo-tristriazole | N.D. | N.D. | 63,392 | 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, M.; Wang, J.; Wang, Y.; He, Z. Heterogeneous Photo-Fenton Catalytic Degradation of Practical Pharmaceutical Wastewater by Modified Attapulgite Supported Multi-Metal Oxides. Water 2021, 13, 156. https://doi.org/10.3390/w13020156
Lu M, Wang J, Wang Y, He Z. Heterogeneous Photo-Fenton Catalytic Degradation of Practical Pharmaceutical Wastewater by Modified Attapulgite Supported Multi-Metal Oxides. Water. 2021; 13(2):156. https://doi.org/10.3390/w13020156
Chicago/Turabian StyleLu, Manjing, Jiaqi Wang, Yuzhong Wang, and Zhengguang He. 2021. "Heterogeneous Photo-Fenton Catalytic Degradation of Practical Pharmaceutical Wastewater by Modified Attapulgite Supported Multi-Metal Oxides" Water 13, no. 2: 156. https://doi.org/10.3390/w13020156
APA StyleLu, M., Wang, J., Wang, Y., & He, Z. (2021). Heterogeneous Photo-Fenton Catalytic Degradation of Practical Pharmaceutical Wastewater by Modified Attapulgite Supported Multi-Metal Oxides. Water, 13(2), 156. https://doi.org/10.3390/w13020156




