Partial Nitrification in a Sequencing Moving Bed Biofilm Reactor (SMBBR) with Zeolite as Biomass Carrier: Effect of Sulfide Pulses and Organic Matter Presence
Abstract
:1. Introduction
2. Methodology
2.1. Nitrifying Inoculum, Substrate, and Zeolite Characteristics
2.2. Experimental Setup and Experimental Design
2.3. Chemical Analyses
2.4. Efficiencies Removal Determination, Production Efficiencies and Statistical Analyses
3. Results and Discussion
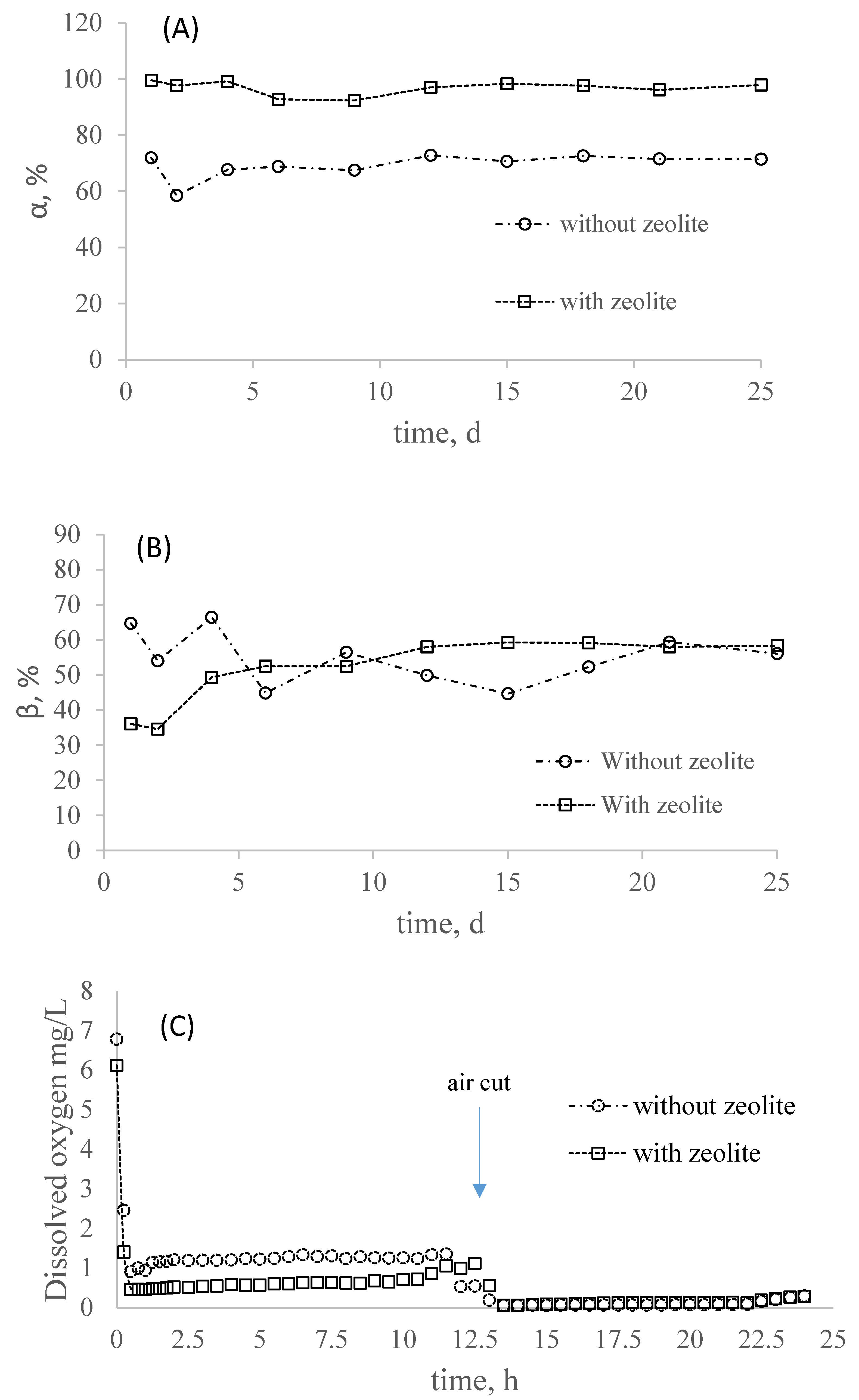
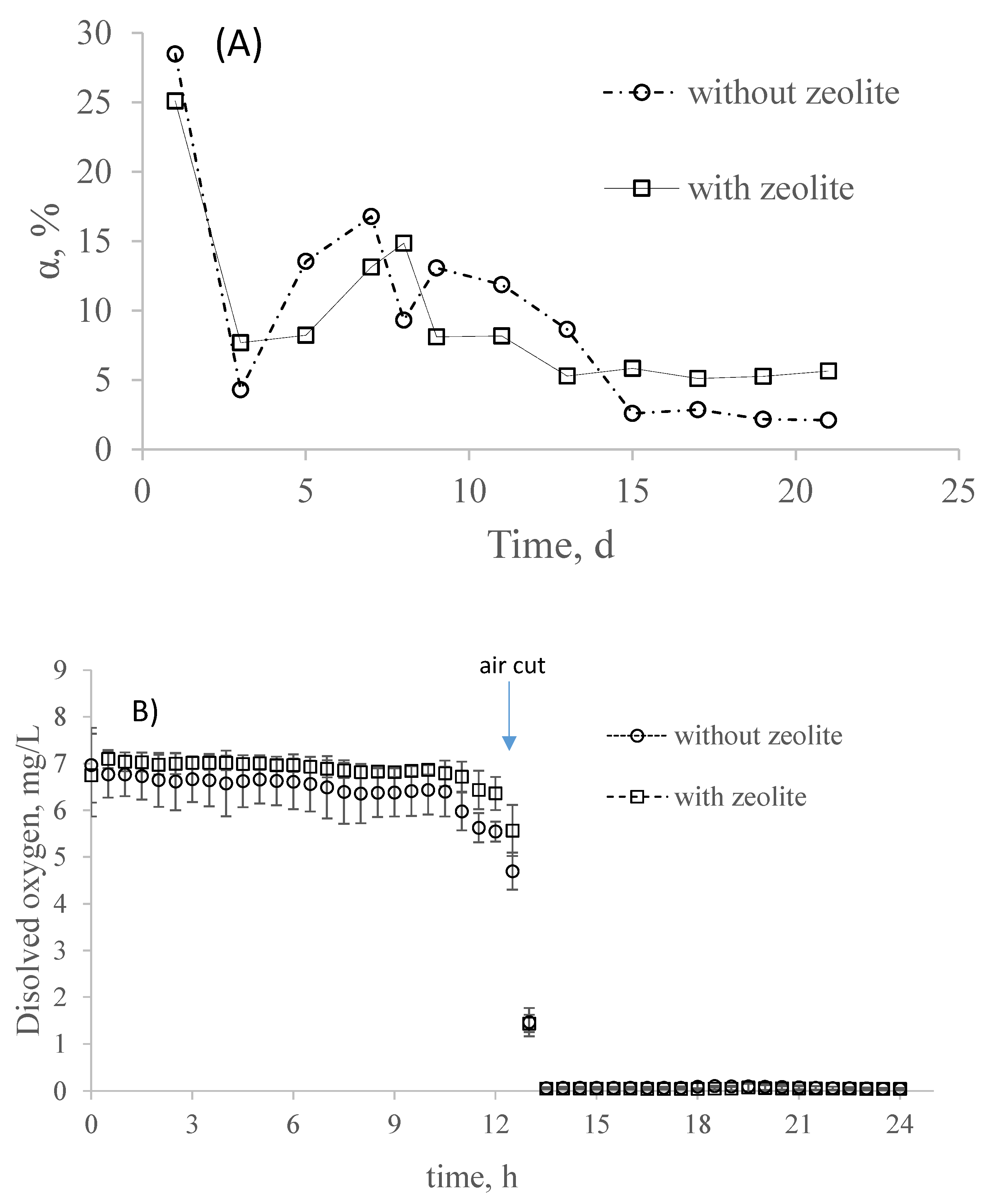
3.1. Partial Nitrification in SBR with and without Zeolite
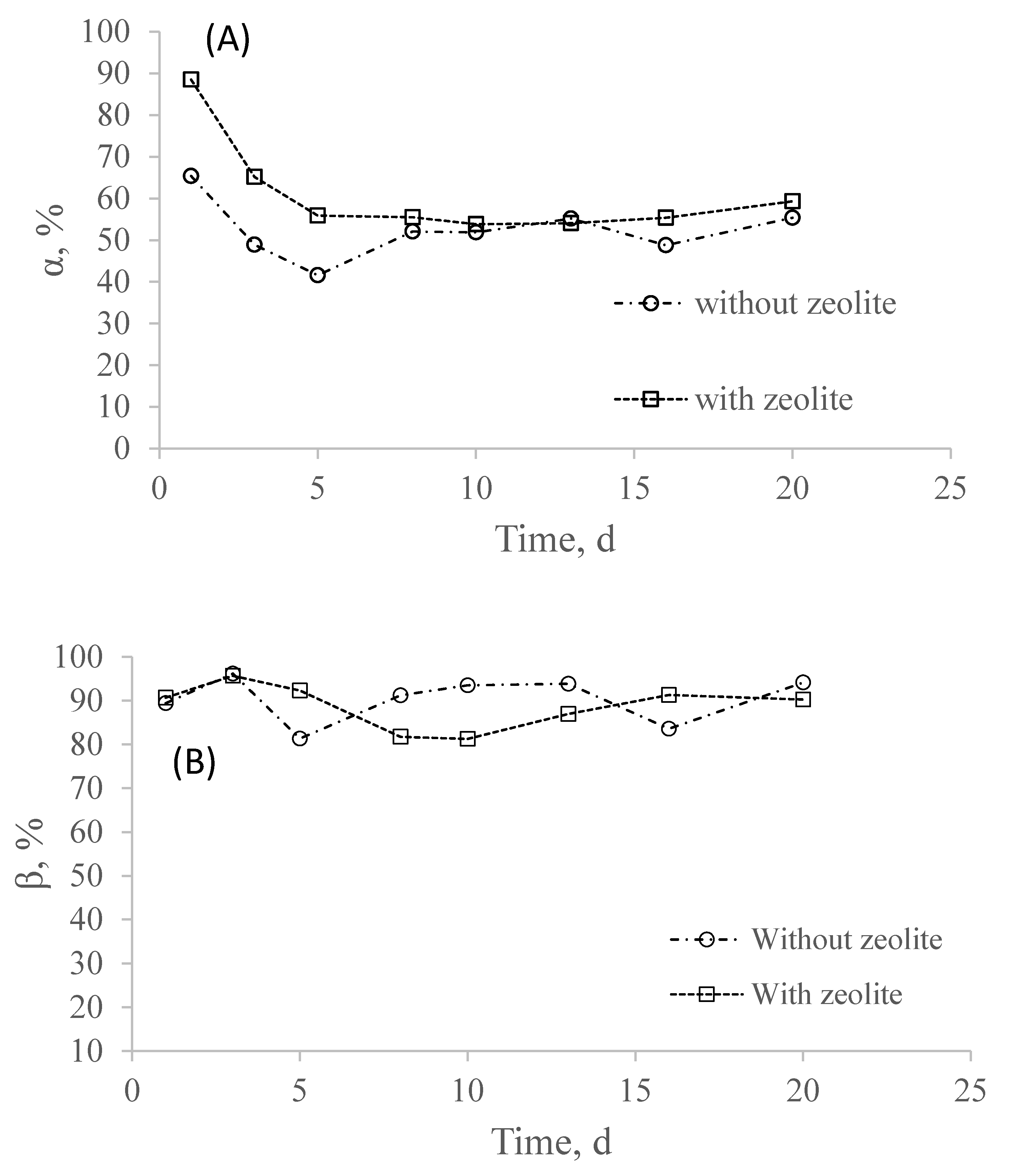
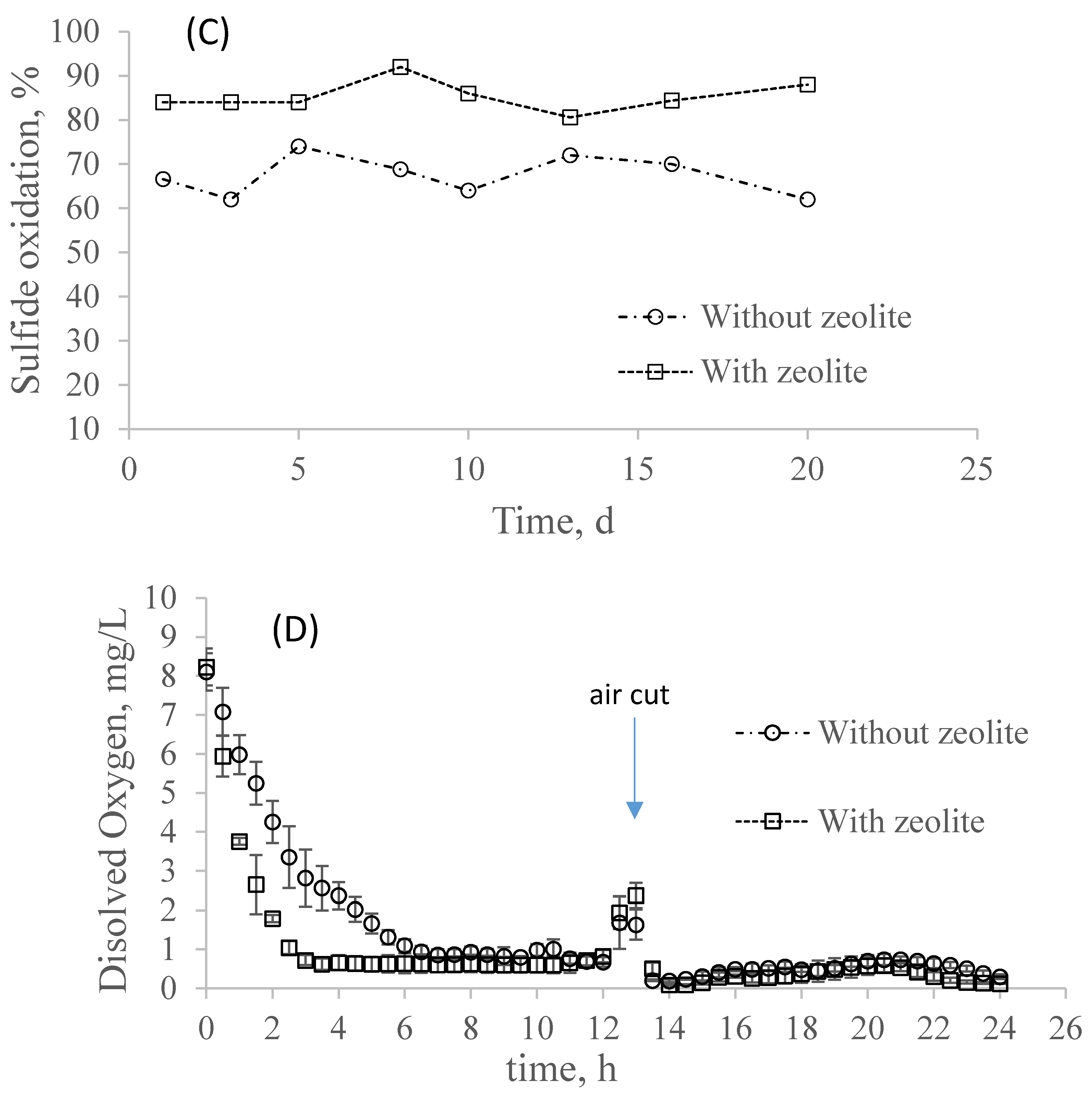
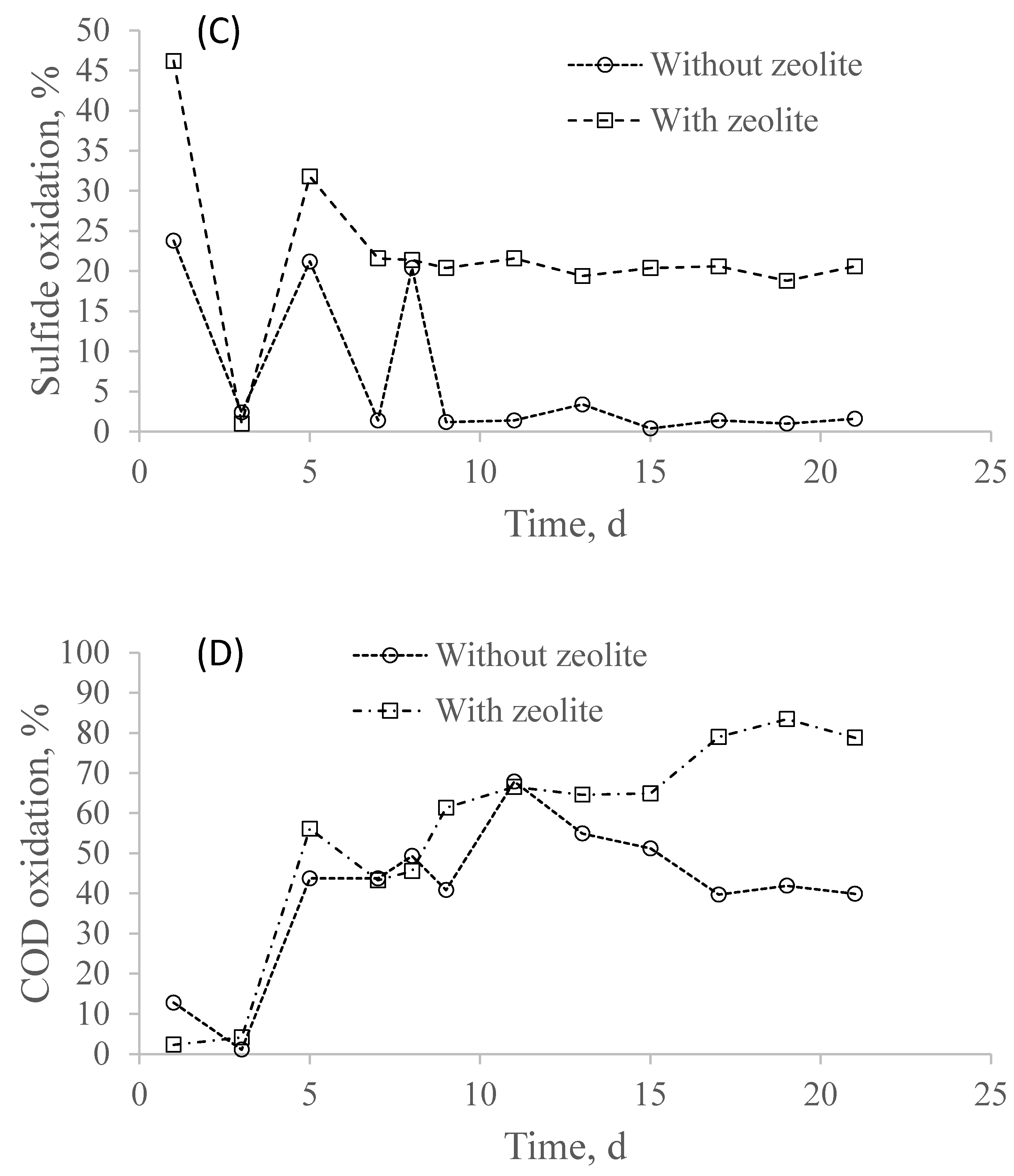
3.2. Effect of Sulfide Pulse on Partial Nitrification in the Systems with and without Zeolite
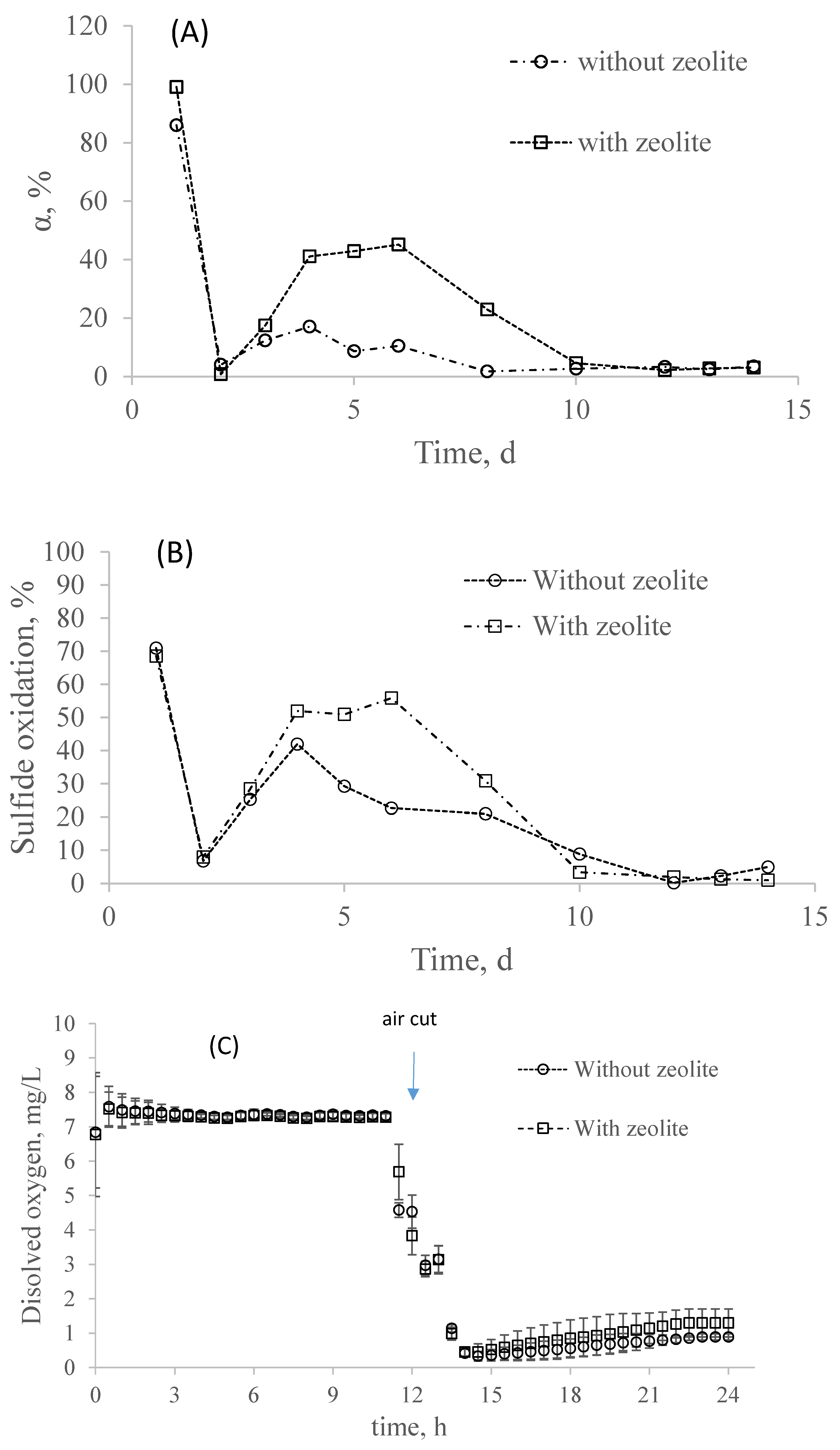
3.3. Simultaneous Effect of Sulfide and COD on Partial Nitrification in the System with and without Zeolite
3.4. Recommendations and Shortcomings of the Proposed System
4. Conclusions
- At low dissolved oxygen concentration (dissolved oxygen < 2.0 mg/L; vvm = 0.1), the system with zeolite was able to have higher ammonia oxidation; however, the nitrite accumulation (β) obtained similar values between the system with and without zeolite;
- The presence of sulfide improved the nitrite accumulation in both systems, with and without zeolite at HS− = 5 mg/L, where the system with zeolite was able to oxidize a higher percentage of sulfide. At HS− = 10 mg/L, both systems were strongly inhibited, although the system with zeolite was able to keep its activity longer; and
- The presence of sulfide and organic matter completely inhibited the nitrification process, with low nitrifying bacteria remaining active. Nevertheless, the system with zeolite was able to degrade higher percentages of organic matter and sulfide.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, W.Y.; She, Z.L.; Gao, M.C.; Wang, Q.; Jin, C.J.; Zhao, Y.G.; Guo, L. Effect of anaerobic/aerobic duration on nitrogen removal and microbial community in a simultaneous partial nitrification and denitrification system under low salinity. Sci. Total Environ. 2019, 651, 859–870. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.R.; Ye, L.; Wang, Q.L.; Zheng, M.; Lu, X.Y.; Wang, Z.Y.; Yuan, Z.G. Nitrite oxidizing bacteria (NOB) contained in influent deteriorate mainstream NOB suppression by sidestream inactivation. Water Res. 2019, 162, 331–338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz, G.; Jeison, D.; Chamy, R. Nitrification with high nitrite accumulation for the treatment of wastewater with high ammonia concentration. Water Res. 2003, 37, 1371–1377. [Google Scholar] [CrossRef]
- Erguder, T.H.; Boon, N.; Vlaeminck, S.E.; Verstraete, W. Partial Nitrification Achieved by Pulse Sulfide Doses in a Sequential Batch Reactor. Environ. Sci. Technol. 2008, 42, 8715–8720. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, D.I.B.; Thalasso, F.; Lopez, F.D.C.; Texier, A.C. Inhibitory effect of sulfide on the nitrifying respiratory process. J. Chem. Technol. Biotechnol. 2013, 88, 1344–1349. [Google Scholar] [CrossRef]
- Vela, J.D.; Dick, G.J.; Love, N.G. Sulfide inhibition of nitrite oxidation in activated sludge depends on microbial community composition. Water Res. 2018, 138, 241–249. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.G.; Kim, M.G.; Nakhla, G.; Andalib, M.; Fang, Y. Partial nitrification-reactor configurations, and operational conditions: Performance analysis. J. Environ. Chem. Eng. 2020, 8, 103984. [Google Scholar] [CrossRef]
- Arora, A.S.; Nawaz, A.; Qyyum, M.A.; Ismail, S.; Aslam, M.; Tawfik, A.; Yun, C.M.; Lee, M. Energy saving anammox technology-based nitrogen removal and bioenergy recovery from wastewater: Inhibition mechanisms, state-of-the-art control strategies, and prospects. Renew. Sustain. Energy Rev. 2021, 135, 110126. [Google Scholar] [CrossRef]
- Bassin, J.P.; Abbas, B.; Vilela, C.L.S.; Kleerebezem, R.; Muyzer, G.; Rosado, A.S.; van Loosdrecht, M.C.M.; Dezotti, M. Tracking the dynamics of heterotrophs and nitrifiers in moving- bed biofilm reactors operated at different COD/N ratios. Bioresour. Technol. 2015, 192, 131–141. [Google Scholar] [CrossRef]
- Montalvo, S.; Huilinir, C.; Borja, R.; Sanchez, E.; Herrmann, C. Application of zeolites for biological treatment processes of solid wastes and wastewaters—A review. Bioresour. Technol. 2020, 301, 122808. [Google Scholar] [CrossRef]
- Yang, Y.Y.; Chen, Z.G.; Wang, X.J.; Zheng, L.; Gu, X.Y. Partial nitrification performance and mechanism of zeolite biological aerated filter for ammonium wastewater treatment. Bioresour. Technol. 2017, 241, 473–481. [Google Scholar] [CrossRef]
- Chen, J.; Wang, R.X.; Wang, X.J.; Chen, Z.G.; Feng, X.H.; Qin, M.Z. Response of nitritation performance and microbial community structure in sequencing biofilm batch reactors filled with different zeolite and alkalinity ratio. Bioresour. Technol. 2019, 273, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.X.; Wang, X.J.; Deng, C.L.; Chen, Z.G.; Chen, Y.X.; Feng, X.H.; Zhong, Z. Partial nitritation performance and microbial community in sequencing batch biofilm reactor filled with zeolite under organics oppression and its recovery strategy. Bioresour. Technol. 2020, 305, 123031. [Google Scholar] [CrossRef]
- Gao, J.F.; Liu, X.H.; Fan, X.Y.; Dai, H.H. Effects of triclosan on performance, microbial community and antibiotic resistance genes during partial denitrification in a sequencing moving bed biofilm reactor. Bioresour. Technol. 2019, 281, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Huilinir, C.; Fuentes, V.; Esposito, G.; Montalvo, S.; Guerrero, L. Nitrification in the presence of sulfide and organic matter in a sequencing moving bed biofilm reactor (SMBBR) with zeolite as biomass carrier. J. Chem. Technol. Biotechnol. 2020, 95, 1614. [Google Scholar] [CrossRef] [Green Version]
- Rice, E.W.; Baird, R.B.; Eaton, A.D. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2017. [Google Scholar]
- Antileo, C.; Roeckel, M.; Wiesmann, U. High nitrite buildup during nitrification in a rotating disk reactor. Water Environ. Res. 2003, 75, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Huilinir, C.; Romero, R.; Munoz, C.; Bornhardt, C.; Roeckel, M.; Antileo, C. Dynamic modeling of partial nitrification in a rotating disk biofilm reactor: Calibration, validation and simulation. Biochem. Eng. J. 2010, 52, 7–18. [Google Scholar] [CrossRef]
- Arnalcios, M.; Amerlinck, Y.; Rehman, U.; Maere, T.; Van Hoey, S.; Naessens, W.; Nopens, I. From the affinity constant to the half-saturation index: Understanding conventional modeling concepts in novel wastewater treatment processes. Water Res. 2015, 70, 458–470. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, L.; Zhang, F.Z.; Jiang, H.; Ren, S.; Wang, W.; Peng, Y.Z. Nitrite accumulation in comammox-dominated nitrification-denitrification reactors: Effects of DO concentration and hydroxylamine addition. J. Hazard. Mater. 2020, 384, 121375. [Google Scholar] [CrossRef]
- Chen, Y.Z.; Zhao, Z.C.; Liu, H.; Ma, Y.H.; An, F.J.; Huang, J.M.; Shao, Z.W. Achieving stable two-stage mainstream partial-nitrification/anammox (PN/A) operation via intermittent aeration. Chemosphere 2020, 245, 125650. [Google Scholar] [CrossRef]
- Tao, C.; Hamouda, M.A. Steady-state modeling and evaluation of partial nitrification-anammox (PNA) for moving bed biofilm reactor and integrated fixed-film activated sludge processes treating municipal wastewater. J. Water Process Eng. 2019, 31, 100854. [Google Scholar] [CrossRef]
- Seuntjens, D.; Van Tendeloo, M.; Chatzigiannidou, I.; Carvajal-Arroyo, J.M.; Vandendriessche, S.; Vlaeminck, S.E.; Boon, N. Synergistic Exposure of Return-Sludge to Anaerobic Starvation, Sulfide, and Free Ammonia to Suppress Nitrite Oxidizing Bacteria. Environ. Sci. Technol. 2018, 52, 8725–8732. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Xing, C.; An, Y.; Hu, D.L.; Qiao, W.M.; Wang, L.C. Inhibitory effects of sulfide on nitrifying biomass in the anaerobic-anoxic-aerobic wastewater treatment process. J. Chem. Technol. Biotechnol. 2014, 89, 214–219. [Google Scholar] [CrossRef]
- Moradi, M.; Karimzadeh, R.; Moosavi, E.S. Modified and ion exchanged clinoptilolite for the adsorptive removal of sulfur compounds in a model fuel: New adsorbents for desulfurization. Fuel 2018, 217, 467–477. [Google Scholar] [CrossRef]
- Ortiz, D.I.B.; Jardines, M.A.M.; Lopez, F.D.C.; Texier, A.C. Biological ammonium and sulfide oxidation in a nitrifying sequencing batch reactor: Kinetic and microbial population dynamics assessments. Chemosphere 2020, 253, 126637. [Google Scholar] [CrossRef]
- Cueto, D.; Mora, M.; Gabriel, D. Evaluating and modeling biological sulfur production in the treatment of sulfide-laden streams containing ammonium. J. Chem. Technol. Biotechnol. 2021, 96, 439–447. [Google Scholar] [CrossRef]
- Sekine, M.; Akizuki, S.; Kishi, M.; Toda, T. Stable nitrification under sulfide supply in a sequencing batch reactor with a long fill period. J. Water Process Eng. 2018, 25, 190–194. [Google Scholar] [CrossRef]
- Mora, M.; Lopez, L.R.; Lafuente, J.; Perez, J.; Kleerebezem, R.; van Loosdrecht, M.C.M.; Gamisans, X.; Gabriel, D. Respirometric characterization of aerobic sulfide, thiosulfate and elemental sulfur oxidation by S-oxidizing biomass. Water Res. 2016, 89, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Namgung, H.G.; Cho, J.I.; Yoo, S.S.; Lee, B.J.; Ji, H.W. Removal of Hydrogen Sulfide in Septic Tanks for Treating Black Water via an Immobilized Media of Sulfur-Oxidizing Bacteria. Int. J. Environ. Res. Public Health 2020, 17, 684. [Google Scholar] [CrossRef] [Green Version]
- Sekine, M.; Akizuki, S.; Kishi, M.; Kurosawa, N.; Toda, T. Simultaneous biological nitrification and desulfurization treatment of ammonium and sulfide-rich wastewater: Effectiveness of a sequential batch operation. Chemosphere 2020, 244, 125381. [Google Scholar] [CrossRef]
- Abeling, U.; Seyfried, C.F. Anaerobic-aerobic treatment of high-strength ammonium waste-water—Nitrogen removal via nitrite. Water Sci. Technol. 1992, 26, 1007–1015. [Google Scholar]
- Guerrero, L.; Montalvo, S.; Huilinir, C.; Barahona, A.; Borja, R.; Cortes, A. Simultaneous nitrification-denitrification of wastewater: Effect of zeolite as a support in sequential batch reactor with step-feed strategy. Int. J. Environ. Sci. Technol. 2016, 13, 2325–2338. [Google Scholar] [CrossRef] [Green Version]
- Park, S.J.; Oh, J.W.; Yoon, T.I. The role of powdered zeolite and activated carbon carriers on nitrification in activated sludge with inhibitory materials. Process Biochem. 2003, 39, 211–219. [Google Scholar] [CrossRef]
- Moraes, B.S.; Orru, J.G.T.; Foresti, E. Nitrogen and sulfide removal from effluent of UASB reactor in a sequencing fed-batch biofilm reactor under intermittent aeration. J. Biotechnol. 2013, 164, 378–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Initial VSS Concentration, g/L | Final VSS Concentration g/L | VSS Percentage Change, % | Sulfide/VSS Ratio, mg S/g VSS * | ||
|---|---|---|---|---|---|
| Without sulfide and OM | With zeolite | 1.031 | 1.045 | +1.4 | - |
| Without zeolite | 1.102 | 1.075 | −2.5 | - | |
| With sulfide (Condition 1: HS− = 5 mg/L) | With zeolite | 1.045 | 1.435 | +37.3 | 3.48 |
| Without zeolite | 1.075 | 1.275 | +18.6 | 3.92 | |
| With sulfide (Condition 2: HS− = 10 mg/L) | With zeolite | 1.480 | 0.730 | −50.7 | 13.70 |
| Without zeolite | 1.470 | 0.700 | −52.4 | 14.30 | |
| With sulfide and OM (HS− = 5 mg/L and COD = 350 mg/L) | With Zeolite | 0.820 | 0.505 | −38.4 | 9.90 |
| Without zeolite | 0.810 | 0.545 | −32.7 | 9.17 | |
| Authors | HS− Concentration, mg/L | Phase of Dosage | Exposition Time, h | Process Studied |
|---|---|---|---|---|
| This work | 5–10 | anoxic | 12 | Partial nitrification |
| Seuntjenset al. [23] | 0–600 | anoxic | 1 h; 2 days | Partial nitrification |
| Erguder et al. [4] | 1.3–80 | anoxic | 23 | Partial nitrification |
| Huiliñir et al. [15] | 5–10 | aerobic | 3 | Simultaneous nitrification/sulfide oxidation |
| Ortiz et al. [26] | 2.5–20 | aerobic | Not reported | Simultaneous nitrification/sulfide oxidation |
| Cueto et al. [27] | 600 | aerobic | Not reported | Simultaneous nitrification/sulfide oxidation |
| Delgado-Vela et al. [6] | 2–35 | aerobic | Not reported | Simultaneous nitrification/sulfide oxidation |
| Sekine et al. [28] | 96 | aerobic | Not reported | Simultaneous nitrification/sulfide oxidation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huiliñir, C.; Fuentes, V.; Estuardo, C.; Antileo, C.; Pino-Cortés, E. Partial Nitrification in a Sequencing Moving Bed Biofilm Reactor (SMBBR) with Zeolite as Biomass Carrier: Effect of Sulfide Pulses and Organic Matter Presence. Water 2021, 13, 2484. https://doi.org/10.3390/w13182484
Huiliñir C, Fuentes V, Estuardo C, Antileo C, Pino-Cortés E. Partial Nitrification in a Sequencing Moving Bed Biofilm Reactor (SMBBR) with Zeolite as Biomass Carrier: Effect of Sulfide Pulses and Organic Matter Presence. Water. 2021; 13(18):2484. https://doi.org/10.3390/w13182484
Chicago/Turabian StyleHuiliñir, César, Vivian Fuentes, Carolina Estuardo, Christian Antileo, and Ernesto Pino-Cortés. 2021. "Partial Nitrification in a Sequencing Moving Bed Biofilm Reactor (SMBBR) with Zeolite as Biomass Carrier: Effect of Sulfide Pulses and Organic Matter Presence" Water 13, no. 18: 2484. https://doi.org/10.3390/w13182484
APA StyleHuiliñir, C., Fuentes, V., Estuardo, C., Antileo, C., & Pino-Cortés, E. (2021). Partial Nitrification in a Sequencing Moving Bed Biofilm Reactor (SMBBR) with Zeolite as Biomass Carrier: Effect of Sulfide Pulses and Organic Matter Presence. Water, 13(18), 2484. https://doi.org/10.3390/w13182484







