Hydrogeochemical Evolution of an Aquifer Regulated by Pyrite Oxidation and Organic Sediments
Abstract
:1. Introduction and Objectives
- To define the conceptual hydrogeological and hydrogeochemical model of the two aquifers of Ágreda.
- To identify the origin of the high sulphate content of carbonate and hydrogen sulphide quaternary aquifers, and their relationship to lithology and content of pyrites and peaty sediments, assessing the importance of pyrite oxidation in aerobic and anaerobic hydrogeological environments.
2. Materials and Methods
- A supply well for the town of Ágreda that we considered at the beginning of the flow line before entering the pyrite mineralized zone.
- The discharge area of this aquifer into Los Ojillos del Keyles springs at the edge of the Quaternary aquifer (and the recharge point of this Quaternary aquifer).
- The exit point of this last aquifer (hydrogen sulphide spring of Ágreda).
- Turbidity tube with Secchi disk at the bottom to determine the transparency of the water. Values had a variability of 1 cm.
- Hanna kit (HI 3810) to quantity dissolved oxygen in water, 0–10 mg/L range, variability 0.1 mg/L.
- Hanna kit (HI 3811) to assess the alkalinity of waters. Range 0–300 mg/L; variability 3 mg/L.
- Hach Kit (Nitraver) to assess the nitrate and nitrite content of the waters. Range 0–40 mg/L; variability 0.01 mg/L
- Hanna pH meter and other brands in measuring the pH of water. Variability of 0.2 pH units.
- Hanna conductivity meter for measuring the conductivity of water; 40 µS variability.
- An isotopic study of δ34S was performed on three water samples at the MAIMA Laboratory of the Barcelona University. Samples were analysed with a Carlo Erba 1108 Elemental Analyser linked to a Delta XP plus Finnigan Mat Isotope Ratio Mass Spectrometry (EA/IRMS) from Thermo Fisher.
- Identification in the field of pyrite mineralization and the pyrite content of the geological formations involved in the hydrogeology of the aquifers and sulphohydric springs of Ágreda and Débanos in the upper basin of the rivers Keyles and Añamaza, respectively. For this, the different geological formations in the field were walked on foot, and exhaustively for those that had an appreciable pyrite content. The pyrite content of these pyritic sedimentary formations was assessed in situ and visually, laterally observing variations in the density of the mineralization (lateral continuity). Two stratigraphic columns were also drawn in these formations or lithological columns from nearby boreholes that had been recognized.
- The cartography of the distribution of plant species that revealed the gypsum substrate in order to identify those interspersed layers with the Weald with a relative sulphate content.
- Use of PHREEQC version 3 (USGS, January 2020) software to determine the residence time in the aquifers, using the obtained field data as a starting point.
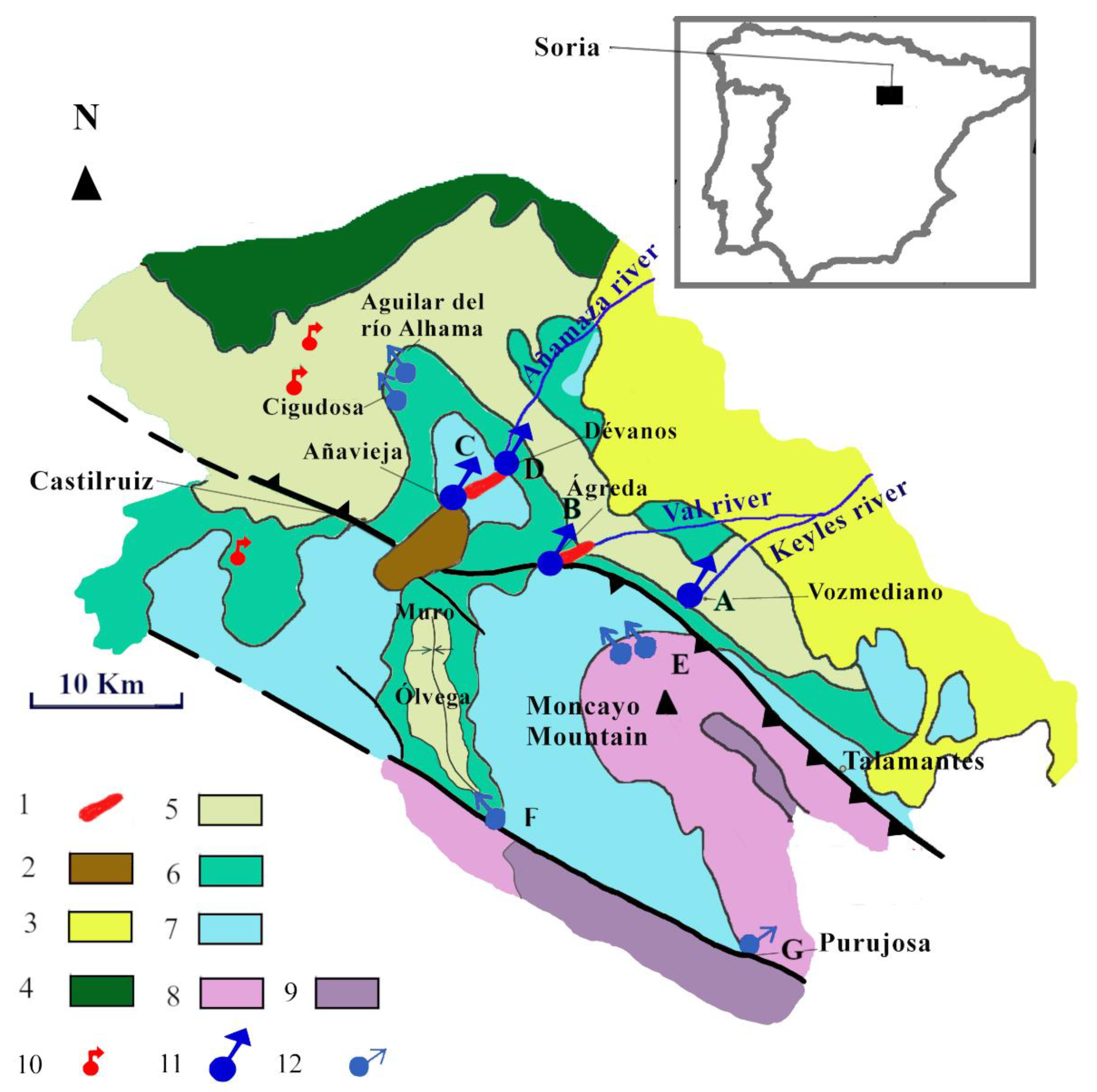
3. Site Geology
3.1. General Geological Characteristics of the Area
3.2. Stratigraphy of Purbeck-Weald Facies in the Ágreda Area
4. Results: Geological and Hydrogeological Characteristics of Aquifers
4.1. Pyrite Distribution
4.2. Gypsum Distribution
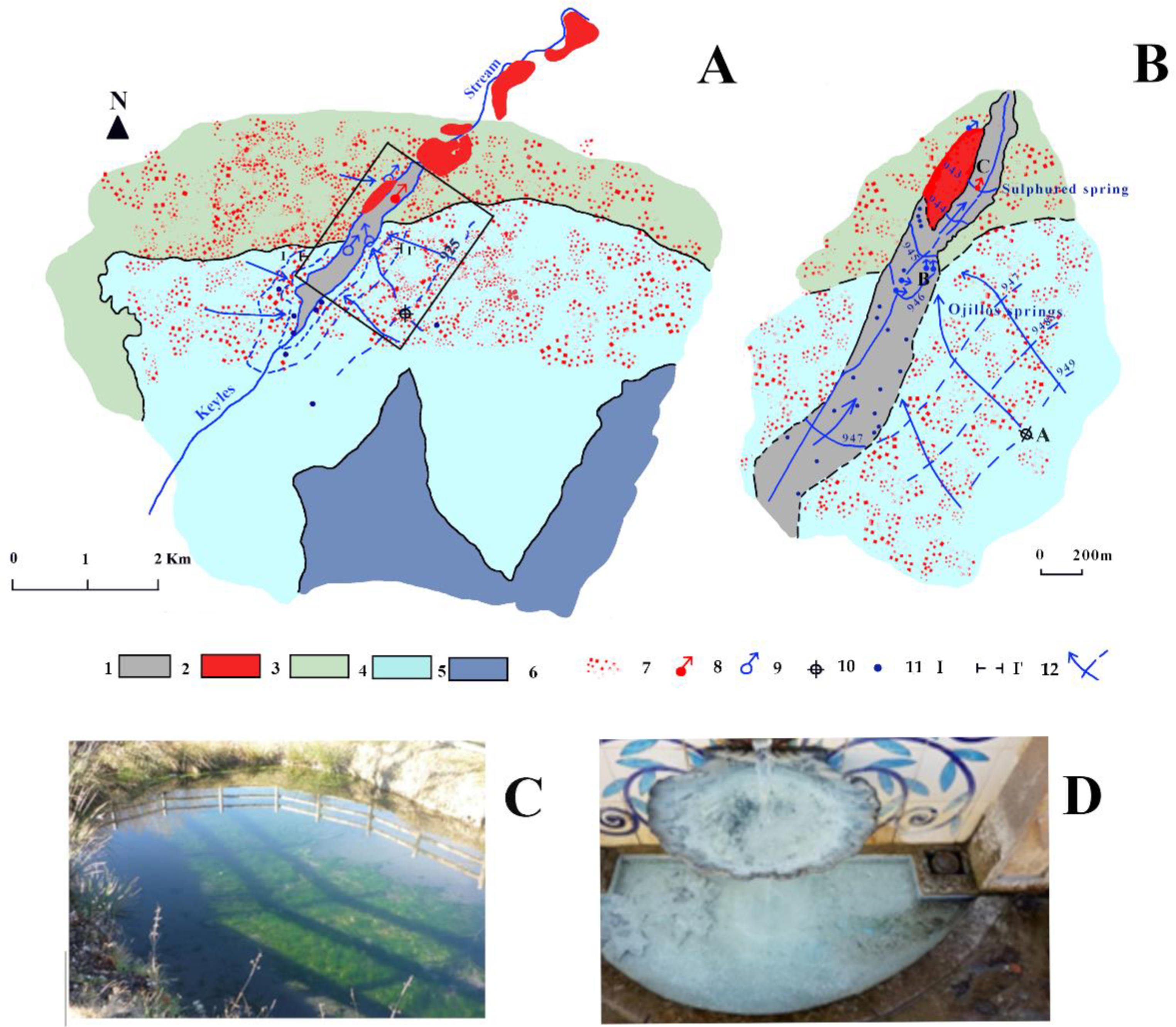
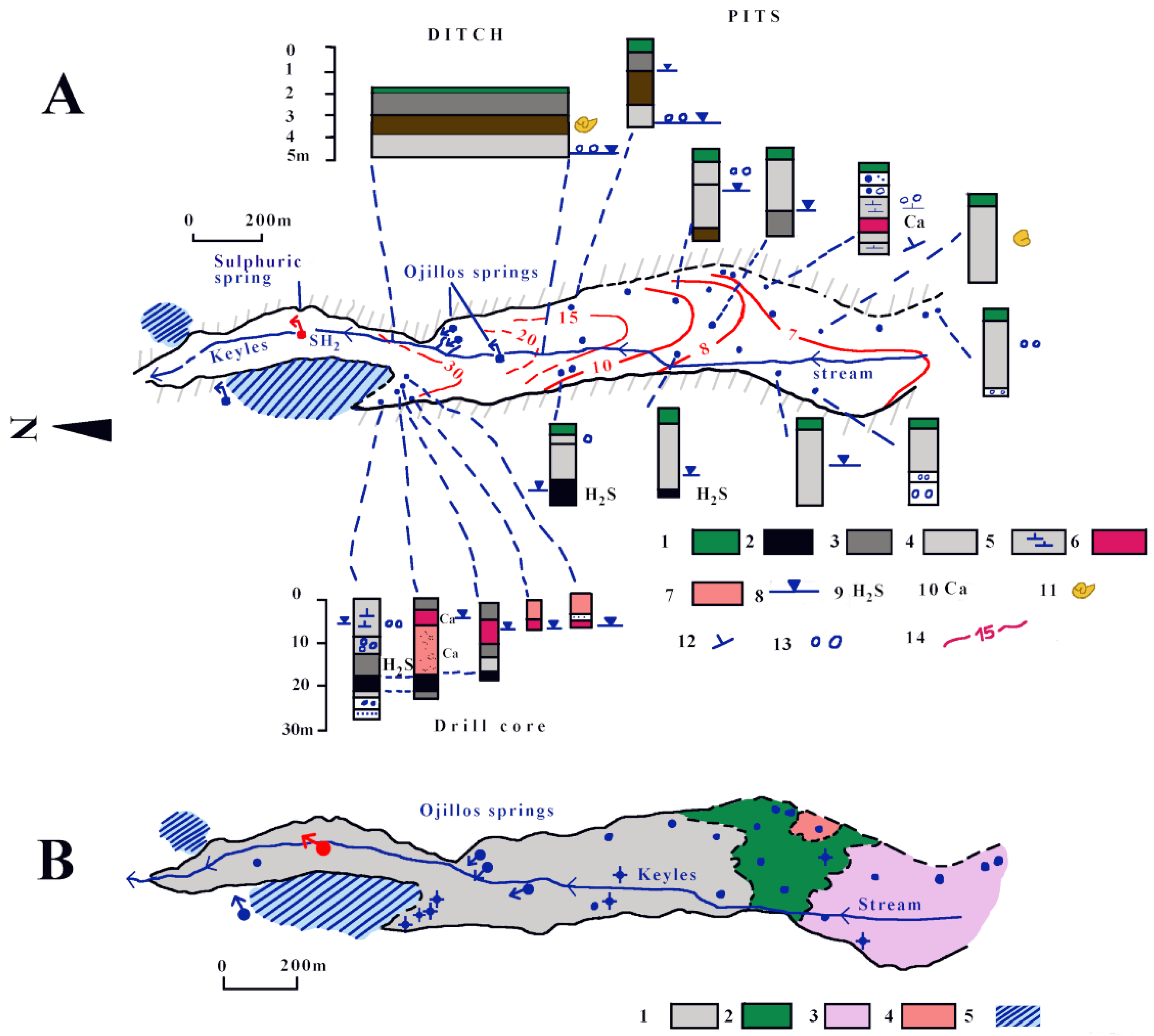
4.3. Hydrogeology
4.3.1. Añavieja and Débanos Springs Aquifer
4.3.2. Los Ojillos del Keyles’ Carbonate Aquifer
4.3.3. Quaternary Aquifer of Dehesa de Ágreda
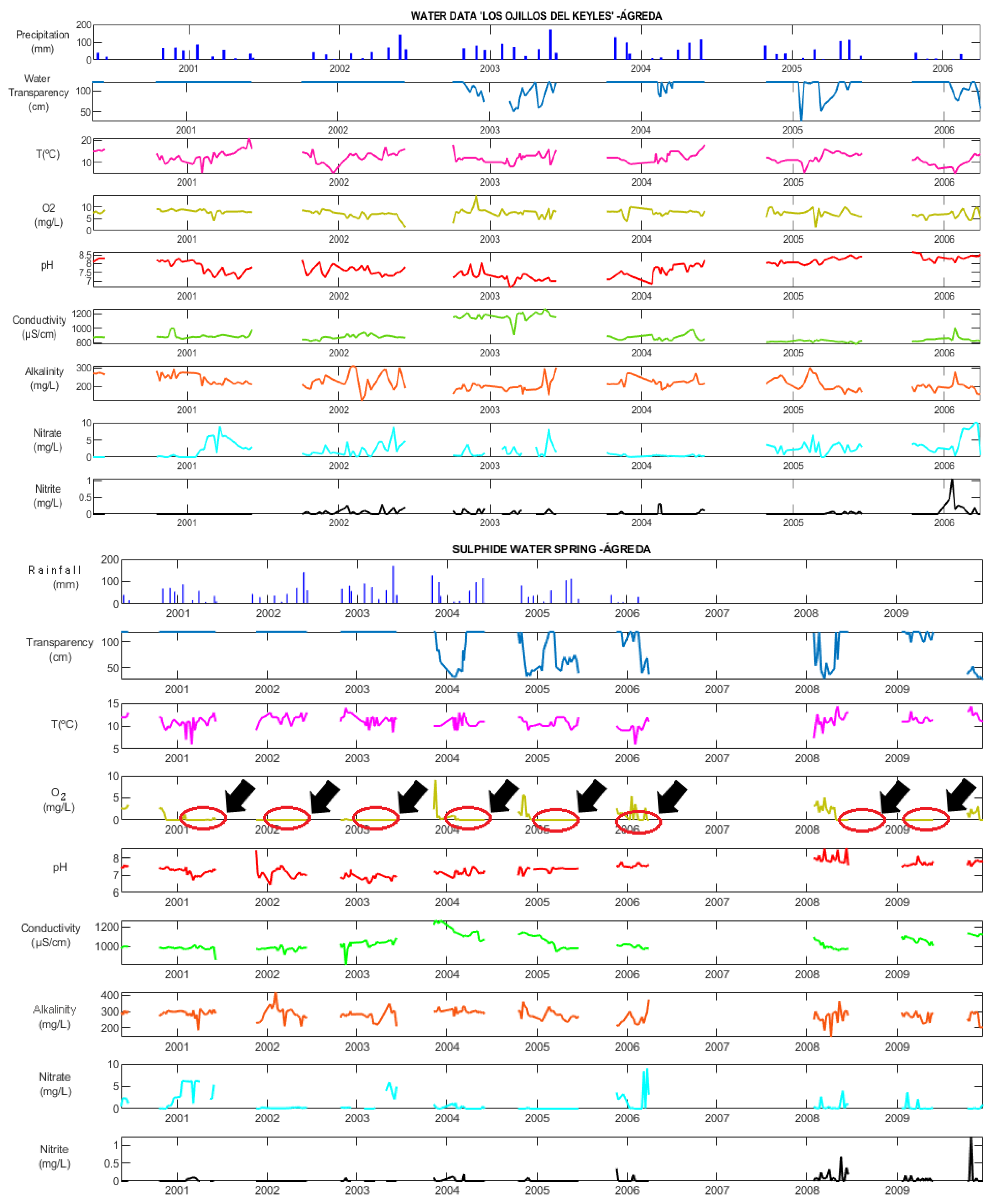
4.3.4. Hydrochemistry
5. Discussion
5.1. Distribution and Alteration (Oxidation) of Pyrites
- Loose and fragmented pyrites in alluvial deposits are the most susceptible to weathering because they are exposed to atmospheric oxidation [39]. This is the case of the pyrites contained in the gravel beds of the alluvial quaternary aquifer of Ágreda.
- The oxidation of pyrites depends on the permeability of the rock: it is greater in limestone and sandstone than in marls, shale, and compact black limestone. In the first group, they are almost always limonitised, while in the second group, they may appear slightly altered, with bright yellow colours. In the argillaceous rocks, they are limonitised on the surface due to the effect of weathering by desquamation caused by wet–dry cycles, but slopes on roads and railways are bright at a depth of less than 2 m. Permeability depends both on the type of lithology and the presence of fractures, which are more frequent in the competent rocks of the first group. In the environment of the fractures of cuttings, pyrites look more limonitised. In the limestones, and according to drill logs in the area of Añavieja, Servicio Geológico de Obras Públicas [36], oxidation depends on the depth to which karstification and groundwater have reached; in lithological column C of a borehole next to the spring of Añavieja (Figure 2), karstification reached 41 m depth, where only iron hydroxides remain in fractures from the oxidation of the pyrites. From 41 to 95 m, there are compact limestones where some water penetration can be seen because the very small pyrites are limonitised. Below and up to at least 151 m, they are also compact limestones in which small shiny pyrites abound without oxidation.
- Smaller pyrites with crystal asymmetry or low symmetry are more easily altered [39].
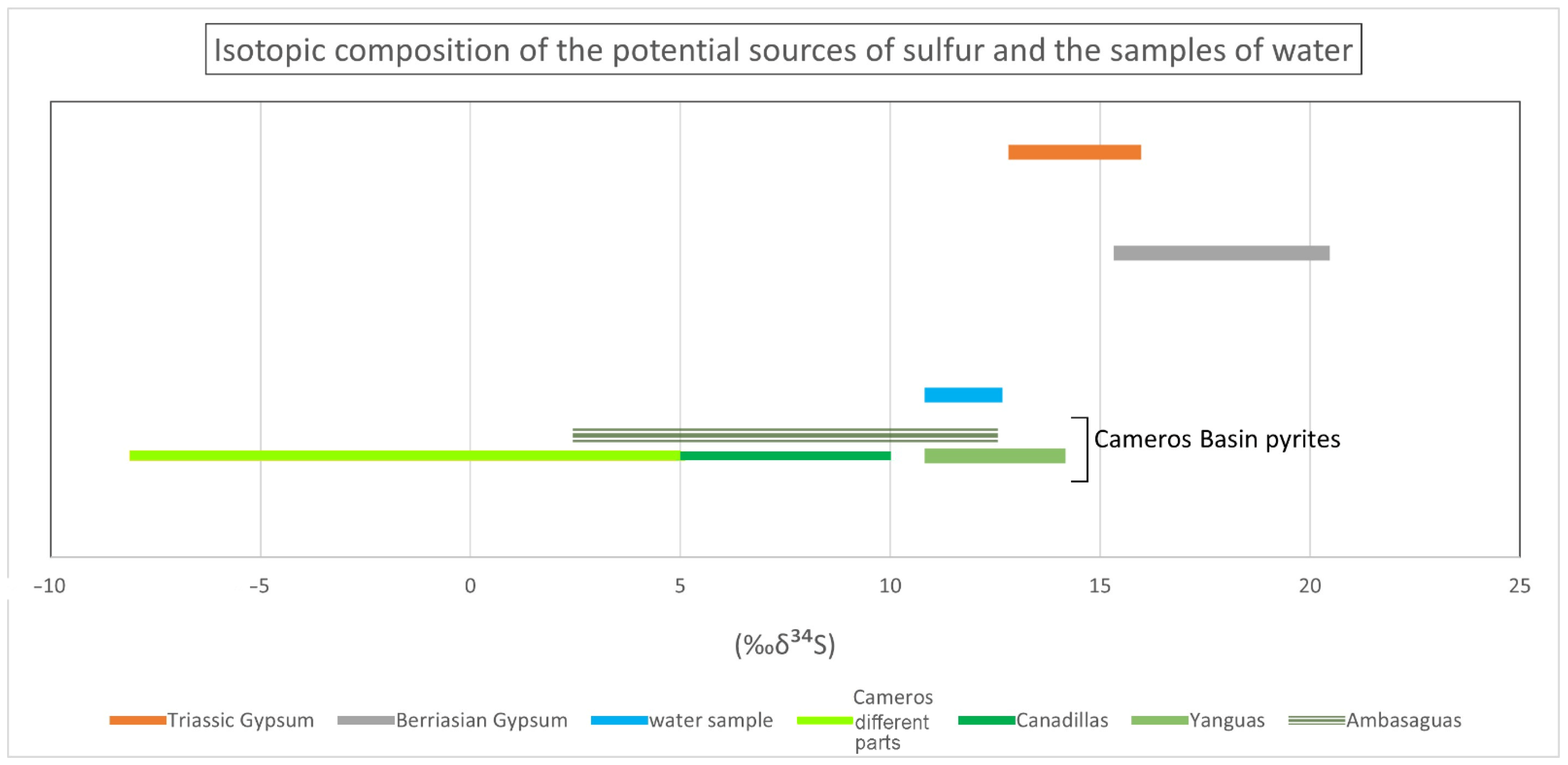
5.2. Hydrogeochemistry: Spatial Variations and Compositional Time Variations
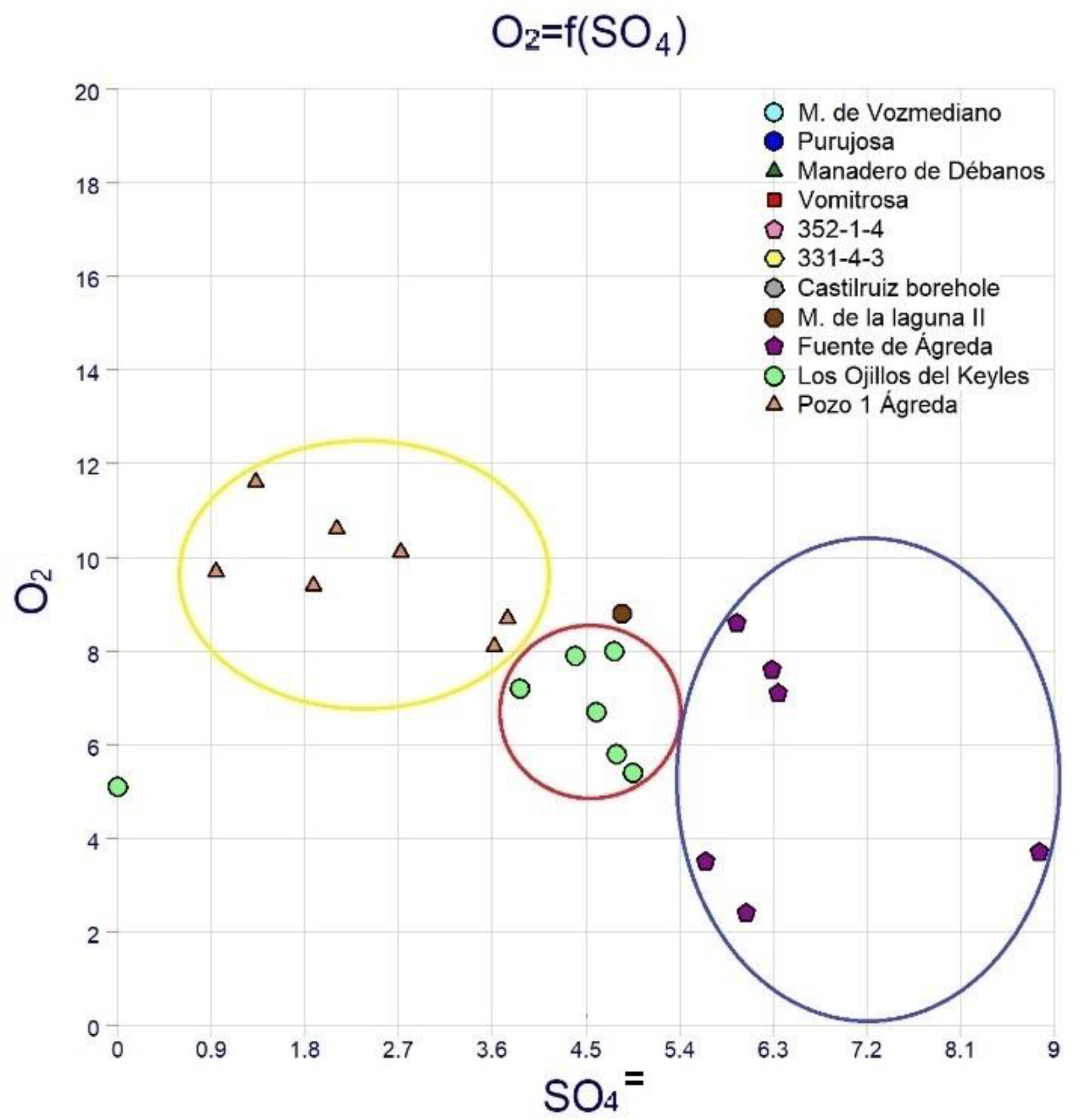
5.2.1. Spatial Variations
5.2.2. Compositional Time Variations
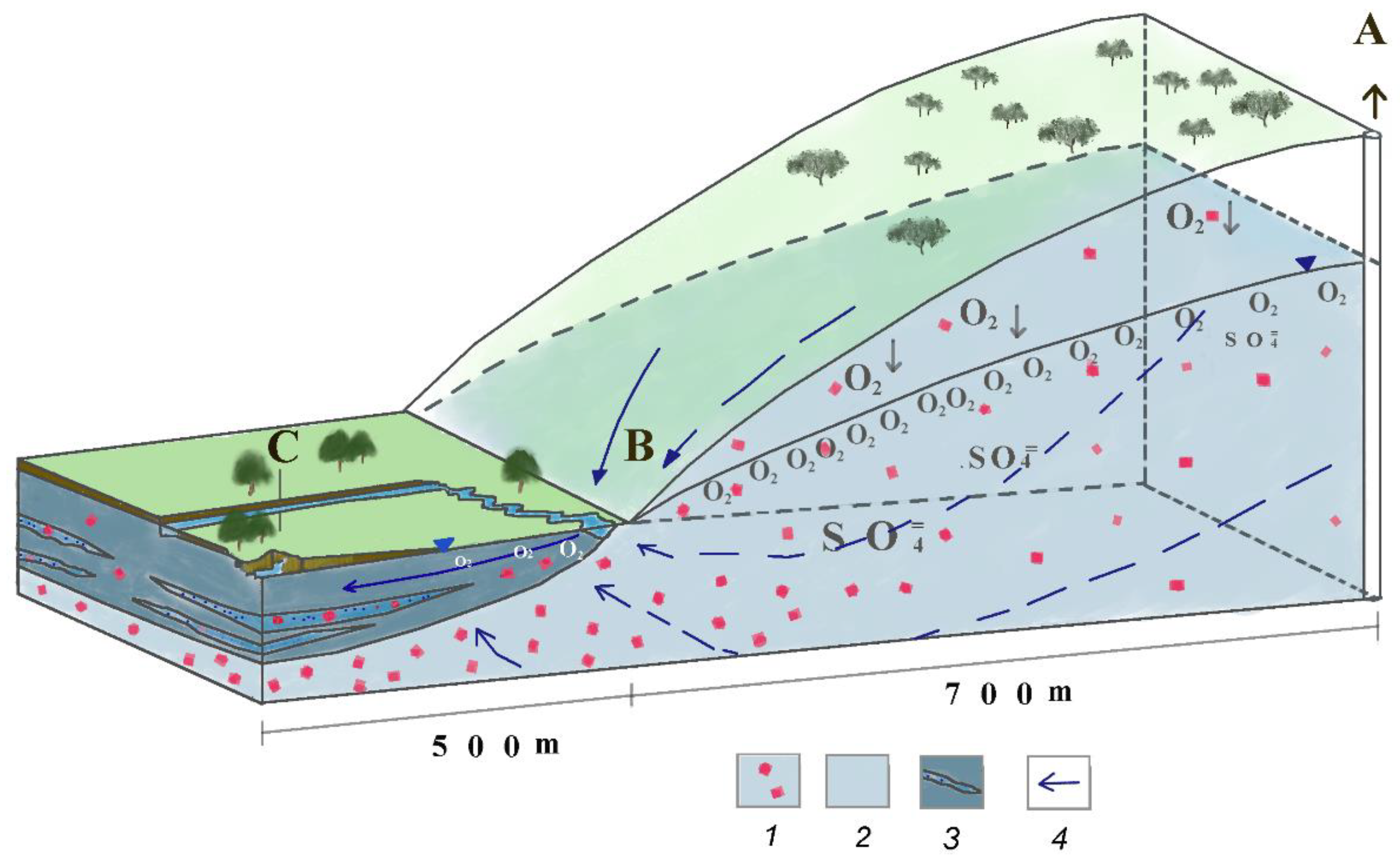
5.3. Conceptual Model of Hydrogeochemical Evolution
5.3.1. Hydrogeochemistry in the Carbonate Aquifer: Increase in Sulphates
5.3.2. Hydrogeochemistry in the Hydrogen Sulphide Aquifer of Ágreda
Hydrogeochemical Evolution: Pyrite Oxidation
Denitrification
Compositional Time Variations
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Starr, R.C.; Gillham, R.W. Denitrification and organic carbon availability in two aquifers. Ground Water 1993, 31, 934–947. [Google Scholar] [CrossRef]
- Tesoriero, A.J.; Puckett, L.J. O2 reduction and denitrification rates in shallow aquifers. Water Resour. Res. 2011, 47, 17. [Google Scholar] [CrossRef]
- Vermaat, J.E.; Harmsen, J.; Hellmann, F.A.; Van der Geest, H.G.; de Klein, J.J.; Kosten, S.; Smolders, A.J.; Verhoeven, J.T.; Mes, R.G.; Ouboter, M. Annual sulfate budgets for Dutch lowland peat polders: The soil is a major sulfate source through peat and pyrite oxidation. J. Hydrol. 2016, 533, 515–522. [Google Scholar] [CrossRef]
- Obermann, P. Hydrochemische/hydromechanische untersuchungen zum stoffgehalt von grun-waser bei landwirtschaftlicher nutzung. Bes. Mitt. Dt. Gewäss. Jahrb. 1982, 42, 217. [Google Scholar]
- Kleimeier, C.; Liu, H.; Rezanezhad, F.; Lennartz, B. Nitrate attenuation in degraded peat soil-based constructed wetlands. Water 2018, 10, 355. [Google Scholar] [CrossRef] [Green Version]
- Choi, H.; Oh, S. Abiotic transient nitrite occurrences from nitrate reduction through goethite-mediated Fe(III)/Fe(II) cycle with labile organic materials and ammonia. Water 2020, 12, 1202. [Google Scholar] [CrossRef] [Green Version]
- Postma, D.; Boesen, C.; Kristiansen, H.; Larsen, F. Nitrate reduction in an unconfined sandy aquifer: Water chemistry, reduction processes, and geochemical modeling. Water Resour. Res. 1991, 27, 2027–2045. [Google Scholar] [CrossRef]
- Tesoriero, A.J.; Liebscher, H.; Cox, S.E. Mechanism and rate of denitrification in an agricultural watershed: Electron and mass balance along groundwater flow paths. Water Resour. Res. 2000, 36, 1545–1559. [Google Scholar] [CrossRef]
- Visser, A.; Broers, H.P.; Heerdink, R.; Bierkens, M.F. Trends in pollutant concentrations in relation to time of recharge and reactive transport at the groundwater body scale. J. Hydrol. 2009, 369, 427–439. [Google Scholar] [CrossRef]
- Kohfahl, C.; Pekdeger, A. Rising groundwater tables in partly oxidized pyrite bearing dump-sediments: Column study and modelling approach. J. Hydrol. 2006, 331, 703–718. [Google Scholar] [CrossRef]
- Sebol, L.; Robertson, W.; Busenberg, E.; Plummer, L.; Ryan, M.C.; Schiff, S. Evidence of CFC degradation in groundwater under pyrite-oxidizing conditions. J. Hydrol. 2007, 347, 1–12. [Google Scholar] [CrossRef]
- Jha, D.; Bose, P. Use of pyrite for pH control during hydrogenotrophic denitrification using metallic iron as the ultimate electron donor. Chemosphere 2005, 61, 1020–1031. [Google Scholar] [CrossRef]
- Ge, Z.; Wei, D.; Zhang, J.; Hu, J.; Liu, Z.; Li, R. Natural pyrite to enhance simultaneous long-term nitrogen and phosphorus removal in constructed wetland: Three years of pilot study. Water Res. 2019, 148, 153–161. [Google Scholar] [CrossRef]
- Carrey, R.; Otero, N.; Soler, A.; Gómez-Alday, J.J.; Ayora, C. The role of lower cretaceous sediments in groundwater nitrate attenuation in central Spain: Column experiments. Appl. Geochem. 2013, 32, 142–152. [Google Scholar] [CrossRef]
- Schwientek, M.; Einsiedl, F.; Stichler, W.; Stögbauer, A.; Strauss, H.; Maloszewski, P. Evidence for denitrification regulated by pyrite oxidation in a heterogeneous porous groundwater system. Chem. Geol. 2008, 255, 60–67. [Google Scholar] [CrossRef]
- Dogramaci, S.; McLean, L.; Skrzypek, G. Hydrochemical and stable isotope indicators of pyrite oxidation in carbonate-rich environment; The Hamersley Basin, Western Australia. J. Hydrol. 2017, 545, 288–298. [Google Scholar] [CrossRef]
- Seibert, S.; Descourvieres, C.; Skrzypek, G.; Deng, H.; Prommer, H. Model-based analysis of δ34S signatures to trace sedimentary pyrite oxidation during managed aquifer recharge in a heterogeneous aquifer. J. Hydrol. 2017, 548, 368–381. [Google Scholar] [CrossRef]
- Strebel, O.; Böttcher, J.; Fritz, P. Use of isotope fractionation of sulfate-sulfur and sulfate-oxygen to assess bacterial desulfurication in a sandy aquifer. J. Hydrol. 1990, 121, 155–172. [Google Scholar] [CrossRef]
- Zhang, Y.-C.; Slomp, C.P.; Broers, H.P.; Passier, H.F.; Van Cappellen, P. Denitrification coupled to pyrite oxidation and changes in groundwater quality in a shallow sandy aquifer. Geochim. Cosmochim. Acta 2009, 73, 6716–6726. [Google Scholar] [CrossRef]
- Massmann, G.; Tichomirowa, M.; Merz, C.; Pekdeger, A. Sulfide oxidation and sulfate reduction in a shallow groundwater system (Oderbruch Aquifer, Germany). J. Hydrol. 2003, 278, 231–243. [Google Scholar] [CrossRef]
- Bohlke, J.K.; Wanty, R.; Tuttle, M.; Delin, G.; Landon, M. Denitrification in the recharge area and discharge area of a transient agricultural nitrate plume in a glacial outwash sand aquifer, Minnesota. Water Resour. Res. 2002, 38, 10–11. [Google Scholar] [CrossRef]
- Korom, S.F.; Schuh, W.M.; Tesfay, T.; Spencer, E.J. Aquifer denitrification and in situ mesocosms: Modeling electron donor contributions and measuring rates. J. Hydrol. 2012, 432–433, 112–126. [Google Scholar] [CrossRef]
- McMahon, P.; Chapelle, F. Redox processes and water quality of selected principal aquifer systems. Ground Water 2008, 46, 259–271. [Google Scholar] [CrossRef]
- Santofimia, E.; López-Pamo, E. The role of surface water and mine groundwater in the chemical stratification of an acidic pit lake (Iberian Pyrite Belt, Spain). J. Hydrol. 2013, 490, 21–31. [Google Scholar] [CrossRef]
- Sanz Pérez, E.; Pascual Arribas, C.; Sanz de Ojeda, M.; Menéndez-Pidal de Navascués, I. La antigua laguna de Ágreda. Celitberia 2015, 109, 177–191. [Google Scholar]
- CHE (Confederación Hidrográfica del Ebro). Análisis Químicos Aguas Subterráneas. Available online: http://www.chebro.es/contenido.visualizar.do?idContenido=13565&idMenu=2980 (accessed on 31 October 2020).
- Imaz, A.G. La Estructura de la Sierra de Cameros: Deformación Dúctil y Su Significado a Escala Cortical; Gobierno de la Rioja: Logroño, Spain, 2001. [Google Scholar]
- Mas, R.; García, A.; Salas, R.; Meléndez, A.; Alonso, A.; Aurell, M.; Bádenas, B.; Benito, M.I.; Carenas, B.; García-Hidalgo, J.F.; et al. Segunda fase de rifting: Jurásico superior-cretácico inferior. In Geología de España; Vera, J.A., Ed.; SGE-IGME: Madrid, Spain, 2004; pp. 503–510. [Google Scholar]
- González Acebrón, L. El Grupo Tera en el Sector Oriental de la Cuenca de Cameros: Ambientes Sedimentarios, Procedencia y Evolución Diagenética; Complutense de Madrid—Facultad de Ciencias Geológicas: Madrid, Spain, 2010. [Google Scholar]
- Meléndez, N.; Gómez-Fernández, J.C.; Gierlowski-Kordesch, E.H.; Kelts, K.R. Continental deposits of the eastern Cameros Basin (Northern Spain) during Tithonian-Berriasian time. In Lake Basins through Space and Time; American Association of Petroleum Geologists: Tulsa, OK, USA, 2000; pp. 263–277. [Google Scholar]
- Quijada, I.E. Evolución Sedimentaria y Paleogeografía de los Sistemas Costeros Siliciclásticos y Carbonático-Evaporíticos del Grupo Oncala (Berriasiense, Cuenca de Cameros Oriental). Ph.D. Thesis, Universidad Complutense de Madrid, Madrid, Spain, 12 December 2014. [Google Scholar]
- Alonso-Azcárate, J.; Rodas, M.; Botrell, S.H.; Mas, J.R. Los yacimientos de pirita de la cuenca de cameros. Zubia 2002, 14, 173–190. [Google Scholar]
- Alonso-Azcárate, J.; Rodas, M.; Mas, R.; Velasco, F. Origen de las piritas de la cuenca de cameros, (La Rioja). Geogaceta 1995, 18, 180–183. [Google Scholar]
- Esnaola, J.M.; Martin, M. Memoria del Mapa Geológico 1/50.000 no 351 (Ólvega); IGME: Madrid, Spain, 1973. [Google Scholar]
- SGOP. Estudio Hidrogeológico de la provincia de Soria; Servicio Geológico de Obras Públicas: Madrid, Spain, 1980. [Google Scholar]
- Zubizarreta, A.S.; Mateo, G.; Alonso, J.L.B. Catálogo Florístico de la Provincia de Soria, 2nd ed.; Diputación Provincial de Soria: Soria, Spain, 2000.
- Sanz, E. El Karst del Sur y Oeste del Moncayo; Universidad Complutense de Madrid: Madrid, Spain, 1981. [Google Scholar]
- Custodio, E.; Llamas, M.R. Hidrología Subterránea; Omega: Barcelona, Spain, 2001; p. 1194. [Google Scholar]
- Campo, M.P.M.; Osácar, M.C.; Aguayo, F.L. Diferenciación geoquímica de los materiales en facies “Weald” de la cuenca NE de Cameros (Soria-La Rioja). Geogaceta 1996, 2, 3. [Google Scholar]
- Yélamos, J.G.; Sanz Pérez, E. Hidrogeoquímica de los manantiales sulfhídricos y ferruginosos de las facies Purbeck-Weald del noroeste de la Cordillera Iberica (provincia de Soria). Estud. Geol. 1994, 50, 215–228. [Google Scholar] [CrossRef]
- Chivas, A.; Andrews, A.; Lyons, W.; Bird, M.; Donnelly, T. Isotopic constraints on the origin of salts in Australian playas. 1. Sulphur. Palaeogeogr. Palaeoclim. Palaeoecol. 1991, 84, 309–332. [Google Scholar] [CrossRef]
- Alonso-Azcárate, J.; Bottrell, S.; Mas, R. Synsedimentary versus metamorphic control of S, O and Sr isotopic compositions in gypsum evaporites from the Cameros Basin, Spain. Chem. Geol. 2006, 234, 46–57. [Google Scholar] [CrossRef]
- Alonso-Azcárate, J.; Rodas, M.; Bottrell, S.H.; Mas, J.R.; Raiswell, R. Estudio textural e isotópico de los sulfuros diseminados en los sedimentos de la cuenca de Cameros (La Rioja, España). Rev. Soc. Geol. Esp. 1999, 12, 241–249. [Google Scholar]
- Herczeg, A.L.; Rattray, K.J.; Dillon, P.J.; Pavelic, P.; Barry, K.E. Geochemical processes during five years of aquifer storage recovery. Ground Water 2004, 42, 438–445. [Google Scholar] [CrossRef]
- Hoffman, J. Geochemistry of acid mine drainage on the aquifers of Southwestern Wisconsin, and regulatory implications. In Impact Mining Ground Water, 4th ed.; NWWA: Denver, CO, USA, 1984; pp. 145–162. [Google Scholar]
- Apollaro, C.; Caracausi, A.; Paternoster, M.; Randazzo, P.; Aiuppa, A.; De Rosa, R.; Fuoco, I.; Mongelli, G.; Muto, F.; Vanni, E.; et al. Fluid geochemistry in a low-enthalpy geothermal field along a sector of southern Apennines chain (Italy). J. Geochem. Explor. 2020, 219, 106618. [Google Scholar] [CrossRef]
- Vasileiou, E.; Papazotos, P.; Dimitrakopoulos, D.; Perraki, M. Expounding the origin of chromium in groundwater of the Sarigkiol basin, Western Macedonia, Greece: A cohesive statistical approach and hydrochemical study. Environ. Monit. Assess. 2019, 191, 509. [Google Scholar] [CrossRef]


| Name | Date | pH | C (µS/cm) 25 °C/20 °C | Chemical-Analysis Concentrations (mg/L) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca2+ | Mg+ + | NH4+ | HCO3− | Cl− | SO42− | NO3− | NO2− | SiO2 | HS− | O2 | CO2 Free | TDS | ||||
| M. de Vozmediano 1 | 29 September 1981 | 7.5 | 558.56 | 104 | 37 | 220 | 14 | 188.4 | 15.3 | 0 | 587.7 | |||||
| M. de Vozmediano 2 | 21 April 1984 | 6.5 | 373 | 76.2 | 29 | 0 | 188 | 14 | 143.5 | 10.7 | 0 | 465.6 | ||||
| Purujosa | 21 April 1984 | 6.9 | 213 | 48.1 | 12 | 0 | 131 | 9.2 | 47.6 | 9.3 | 0 | 260.3 | ||||
| Manadero de Débanos | 21 April 1984 | 6.9 | 760 | 136 | 54 | 0 | 317 | 14 | 376.3 | 6.4 | 0 | 908.5 | ||||
| Vomitrosa | 20 September 1981 | 7.4 | 476.86 | 92.2 | 15 | 293 | 3.5 | 47.6 | 10.7 | 0 | 451.57 | |||||
| 352-1-4 | 24 September 1981 | 7.2 | 86.96 | 28.1 | 2.4 | 95.2 | 3.5 | 0 | 0 | 0 | 129.59 | |||||
| 331-4-3 (1) | 26 September 1981 | 6.9 | 40.8 | 4 | 2.4 | 12.2 | 3.5 | 2.7 | 5 | 0 | 30.35 | |||||
| 331-4-3 (2) | 21 April 1984 | 5.1 | 19 | 8 | 3.6 | 0 | 39.1 | 9 | 2.1 | 0 | 0 | 63.45 | ||||
| Castilruiz Borehole 2 | 5 May 2008 | 7.4 | * 794 | 147 | 19 | 272 | 8.7 | 216.8 | 7.48 | 690.36 | ||||||
| Castilruiz Borehole 1 | 30 July 2008 | 7.2 | 940 | 154 | 35 | 337 | 7.2 | 277.4 | 0.1 | |||||||
| M. de la laguna II | 7 April 2016 | 7.3 | 830 | 156 | 18 | <0.050 | 215 | 5.1 | 233 | 8.54 | 10 | 8.8 | 8 | |||
| Fuente de Ágreda (^) | 24 September 2015 | 7.3 | * 911 | 184 | 29.6 | 0.68 | 14.1 | 286 | 5.2 | 0.024 | 12.4 | 8.6 | 21 | |||
| Fuente de Ágreda (^) | 10 April 2013 | 7.2 | 1063 | 199 | 29.5 | 0.83 | 294 | 14.6 | 305 | 5.97 | 11.3 | 7.1 | 35 | |||
| Fuente de Ágreda (^) | 2 November 2011 | 7.1 | 1094 | 212 | 33.2 | 0.17 | 288.64 | 15.8 | 302 | 8.61 | 0.071 | 13.1 | 7.6 | 7 | ||
| Fuente de Ágreda (^) | 30 September 2009 | 7.2 | 1125 | 216 | 37.4 | <0.02 | 308 | 23.4 | 425 | 9.1 | 0.56 | 12.2 | 3.7 | |||
| Fuente de Ágreda (^) | 30 March 2006 | 6.8 | 195 | 28 | 0.79 | 263 | 18 | 271 | 4 | 0.017 | 12 | 3.5 | 34 | |||
| Fuente de Ágreda (^) | 3 April 2003 | 7.3 | 870 | 180 | 32 | 285 | 16 | 290 | 3 | 7.2 | 2.4 | 21 | ||||
| Fuente de Ágreda (+) | 12 March 1883 | 117 | 46 | 347 | 49 | 160 | 28 | 30 | ||||||||
| Min. | 6.8 | 870 | 117 | 28 | <0.02 | 263 | 14.1 | 160 | 3 | 0.017 | 7.2 | 30 | 2.4 | 7 | ||
| Max. | 7.3 | 1125 | 216 | 46 | 0.83 | 347 | 49 | 425 | 9.1 | 0.56 | 28 | 30 | 8.6 | 35 | ||
| Media | 7.15 | 969.67 | 186.14 | 33.67 | 0.62 | 297.61 | 19.29 | 291.13 | 5.98 | 0.168 | 13.74 | 30 | 5.48 | 23.6 | ||
| Los Ojillos del Keyles (^) | 26 November 2018 | 7.2 | 718 | 150 | 21.2 | 22.8 | 5.1 | |||||||||
| Los Ojillos del Keyles (^) | 24 September 2015 | 7.2 | * 724 | 145 | 21.7 | <0.13 | 11.5 | 221 | 26.0 | <0.02 | 10.6 | 6.7 | 11 | |||
| Los Ojillos del Keyles (^) | 28 January 2013 | 7.2 | 784 | 151 | 23.2 | <0.050 | 207 | 7.61 | 229 | 15.9 | 11.1 | 8.0 | 25 | |||
| Los Ojillos del Keyles (^) | 2 November 2011 | 875 | 140 | 22.1 | <0.050 | 206.64 | 7.57 | 211 | 16.6 | <0.001 | 9.97 | 7.9 | 10 | |||
| Los Ojillos del Keyles (^) | 30 September 2009 | 7.2 | 736 | 143 | 22 | <0.02 | 209 | 10.6 | 238 | 21.7 | 0.01 | 8.7 | 5.4 | |||
| Los Ojillos del Keyles (^) | 30 March 2006 | 7.0 | 156 | 20 | <0.046 | 212 | 9 | 186 | 17 | <0.01 | 11 | 7.2 | 26 | |||
| Los Ojillos del Keyles (^) | 3 April 2003 | 7.4 | 715 | 140 | 20 | 250 | 12 | 230 | 22 | 6.8 | 5.8 | 7 | ||||
| Los Ojillos del Keyles (**) | 10 May 1989 | 7.6 | 640 | 133 | 29 | 273 | 18 | 227 | 21 | 9.8 | ||||||
| Los Ojillos del Keyles (**) | 23 March 1983 | 7.2 | 797 | 132 | 24 | 262 | 7.1 | 206 | ||||||||
| Los Ojillos del Keyles (**) | 2 October 1981 | 7.8 | 721.1 | 132 | 46 | 293 | 14 | 285.2 | 12.2 | 0 | 792.1 | |||||
| Min. | 7.0 | 640 | 132 | 26 | <0.02 | 207 | 7.57 | 186 | 15.9 | 0 | 6.8 | 5.1 | 7 | 792.1 | ||
| Max. | 7.8 | 875 | 156 | 46 | <0.05 | 293 | 14 | 285.2 | 22.8 | <0.02 | 11.1 | 8.0 | 26 | 792.1 | ||
| Media | 7.31 | 745.57 | 142.2 | 24.92 | 239.08 | 10.82 | 225.91 | 19.47 | 0.002 | 9.71 | 6.59 | 15.8 | 792.1 | |||
| Pozo 1 Ágreda (^) | 28 March 2017 | 7.5 | * 670 | 109 | 14.7 | <0.050 | 171 | 7.29 | 131 | 15.6 | 8.0 | 10.1 | 4 | |||
| Pozo 1 Ágreda (^) | 7 April 2016 | 7.6 | 450 | 78.1 | 7.71 | <0.050 | 141 | 6.21 | 63.9 | 14.5 | <0.010 | 7.1 | 11.6 | 8 | ||
| Pozo 1 Ágreda (^) | 10 April 2013 | 7.4 | 367 | 69.9 | 6.7 | <0.050 | 128 | 8.3 | 45.2 | 20.7 | <0.001 | 6.2 | 9.7 | 9 | ||
| Pozo 1 Ágreda (^) | 27 November 2012 | 7.3 | 632 | 130 | 17.3 | <0.050 | 201 | 7.2 | 180 | 12.4 | <0.001 | 8.58 | 8.7 | 12 | ||
| Pozo 1 Ágreda (^) | 2 November 2011 | 7.2 | 697 | 139 | 19.2 | <0.050 | 205 | 7.71 | 174 | 13.2 | <0.001 | 7.91 | 8.1 | 6 | ||
| Pozo 1 Ágreda (^) | 15 April 2009 | 7.4 | 402 | 90.4 | 10.2 | <0.13 | 160 | 9.1 | 90.4 | 17.2 | <0.020 | 9.4 | 7 | |||
| Pozo 1 Ágreda (^) | 18 December 2007 | 6.9 | 99 | 19 | 0.077 | 204 | 11 | 101 | 21 | <0.01 | 9.3 | 10.6 | 13 | |||
| Min. | 6.9 | 367 | 69.9 | 6.7 | <0.05 | 141 | 6.21 | 45.2 | 12.4 | <0.001 | 6.2 | 8.1 | 4 | |||
| Max. | 7.6 | 697 | 139 | 19.2 | <0.13 | 205 | 11 | 180 | 21 | <0.01 | 9.3 | 11.6 | 13 | |||
| Media | 7.33 | 524.0 | 102.2 | 13.54 | 172.86 | 8.12 | 118.67 | 16.37 | 7.85 | 9.74 | 8.43 | |||||
| Layer | K (m/Day) | me | Vr (m/Day) | Time (Years) |
|---|---|---|---|---|
| Silt | 4.32 | 0.07 | 0.154 | 8.9 |
| Peat | 7.7 | 0.05 | 0.385 | 3.55 |
| Sample | mg/L C Organic | mg/L N |
|---|---|---|
| Dehesa de Agreda | <0.1 | 8.40 |
| Dehesa Park | <0.1 | <0.2 |
| Fuente de Ágreda (sulphurous source) | 0.74 | 3.30 |
| Los Ojillos | <0.1 | 5.68 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanz, E.; Bezares, C.; Pascual, C.; Menéndez Pidal, I.; Fonolla, C. Hydrogeochemical Evolution of an Aquifer Regulated by Pyrite Oxidation and Organic Sediments. Water 2021, 13, 2444. https://doi.org/10.3390/w13172444
Sanz E, Bezares C, Pascual C, Menéndez Pidal I, Fonolla C. Hydrogeochemical Evolution of an Aquifer Regulated by Pyrite Oxidation and Organic Sediments. Water. 2021; 13(17):2444. https://doi.org/10.3390/w13172444
Chicago/Turabian StyleSanz, Eugenio, Catalina Bezares, Carlos Pascual, Ignacio Menéndez Pidal, and Cristina Fonolla. 2021. "Hydrogeochemical Evolution of an Aquifer Regulated by Pyrite Oxidation and Organic Sediments" Water 13, no. 17: 2444. https://doi.org/10.3390/w13172444
APA StyleSanz, E., Bezares, C., Pascual, C., Menéndez Pidal, I., & Fonolla, C. (2021). Hydrogeochemical Evolution of an Aquifer Regulated by Pyrite Oxidation and Organic Sediments. Water, 13(17), 2444. https://doi.org/10.3390/w13172444






