Assessment of Surface Water Quality in the Podu Iloaiei Dam Lake (North-Eastern Romania): Potential Implications for Aquaculture Activities in the Area
Abstract
:1. Introduction
2. Materials and Methods
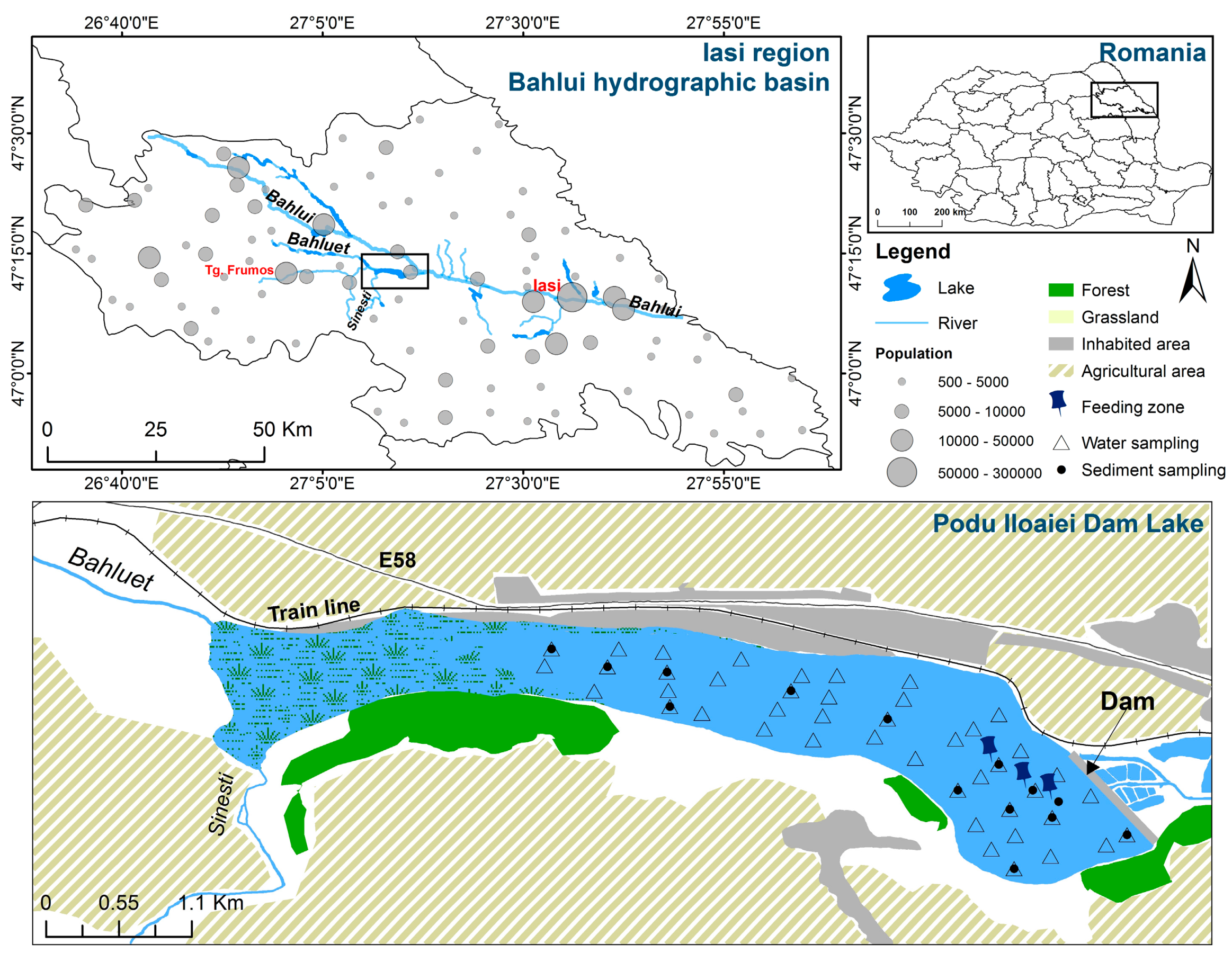
2.1. Study Area and Sampling Strategy
2.2. Materials and Methods
2.2.1. Water-Soluble Ions Analysis by Ion Chromatography
2.2.2. Water-Soluble Elements Analyses by Inductively Coupled Plasma Mass Spectrometry
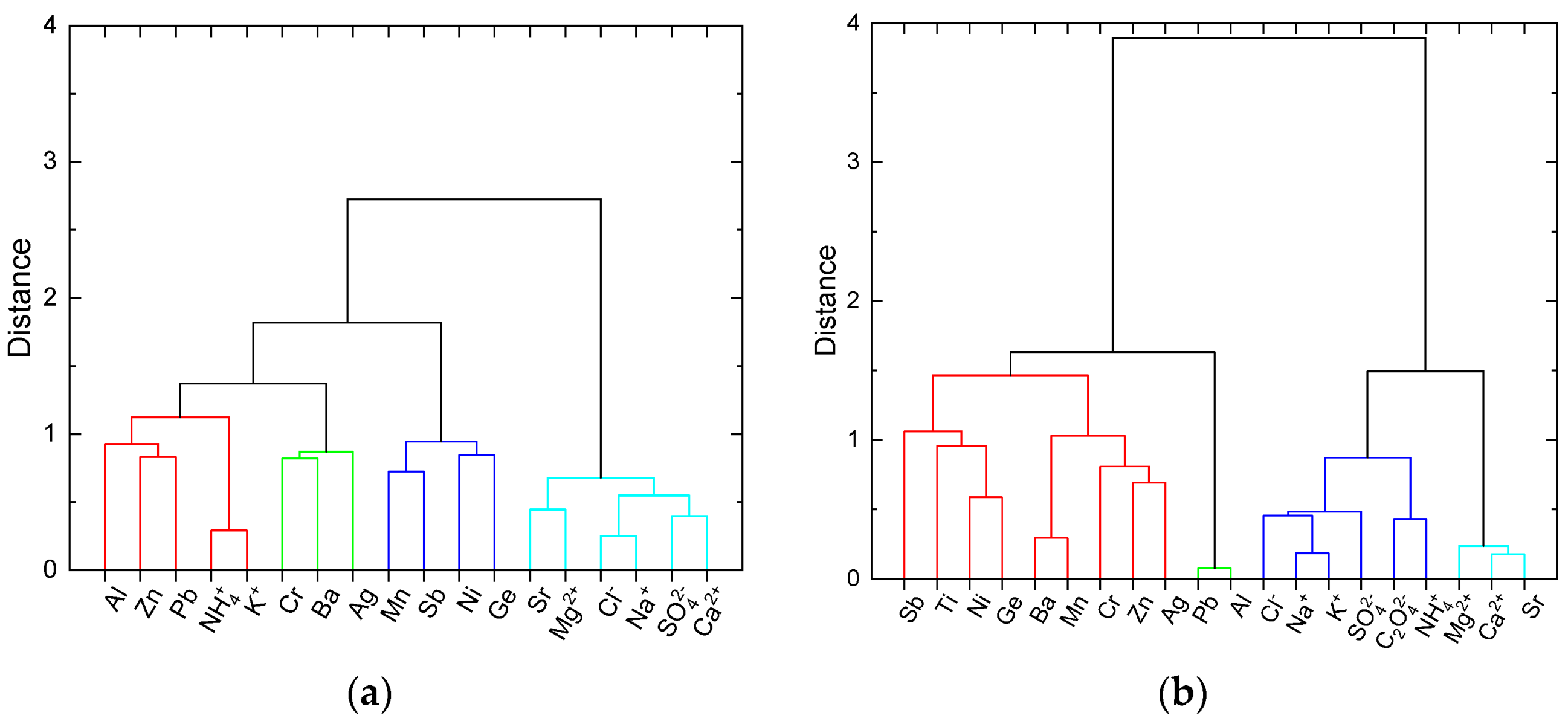
2.3. Statistical Analysis of the Data
3. Results
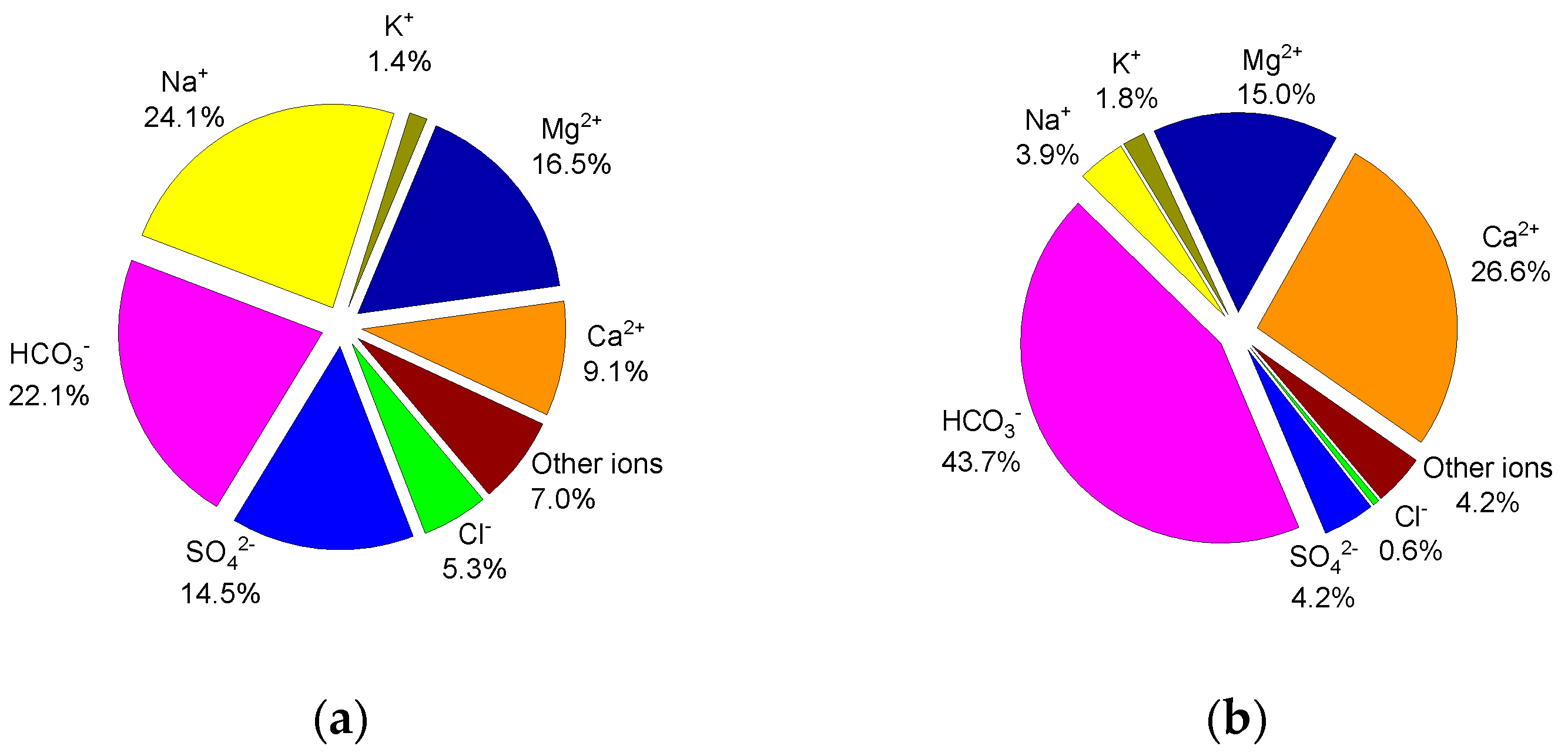
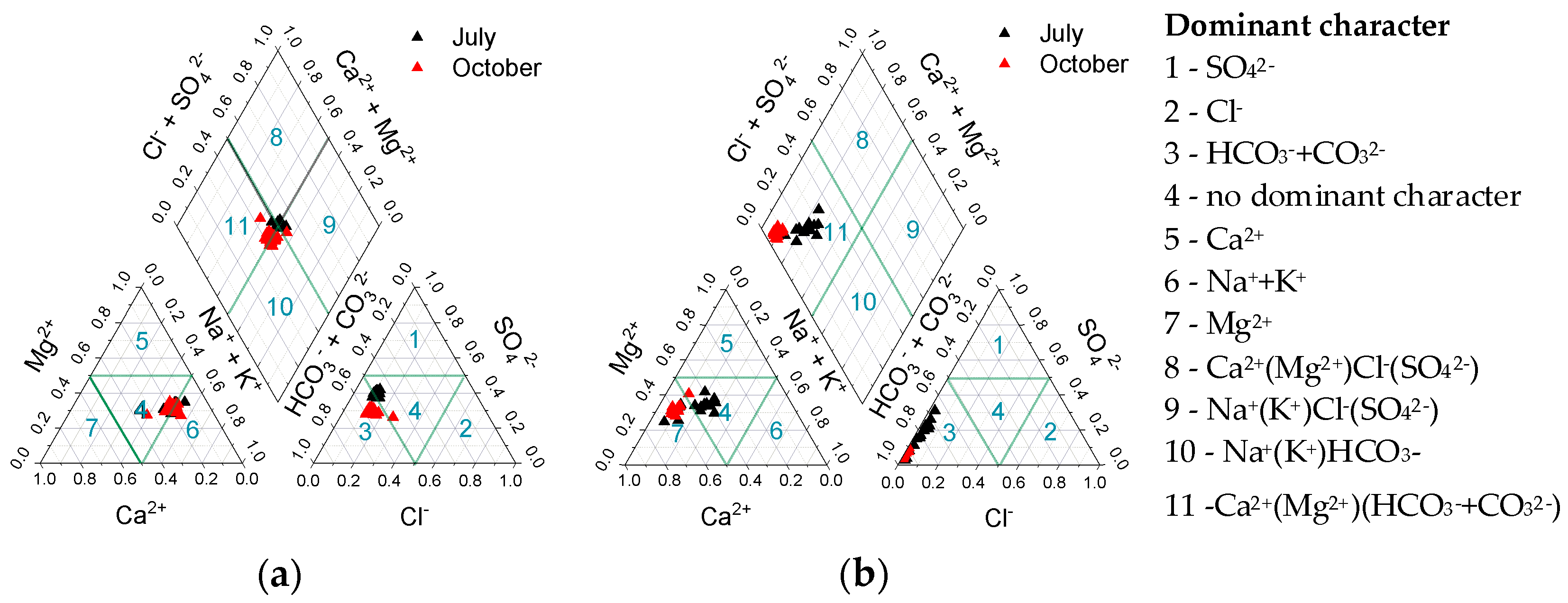
3.1. Water-Soluble Ions
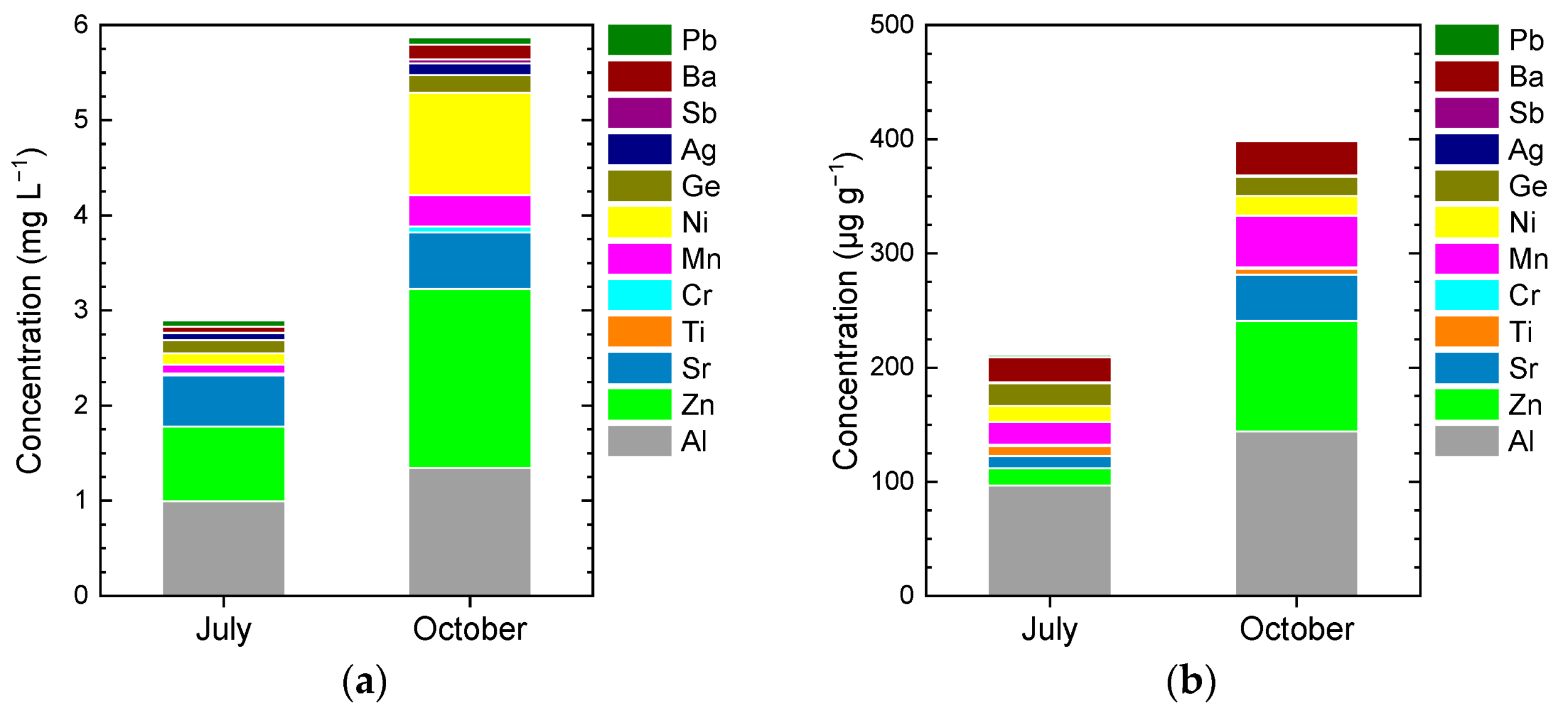
3.2. Water-Soluble Elements
4. Discussion
4.1. Water-Soluble Ions and Elements Related to Lake Water Typology
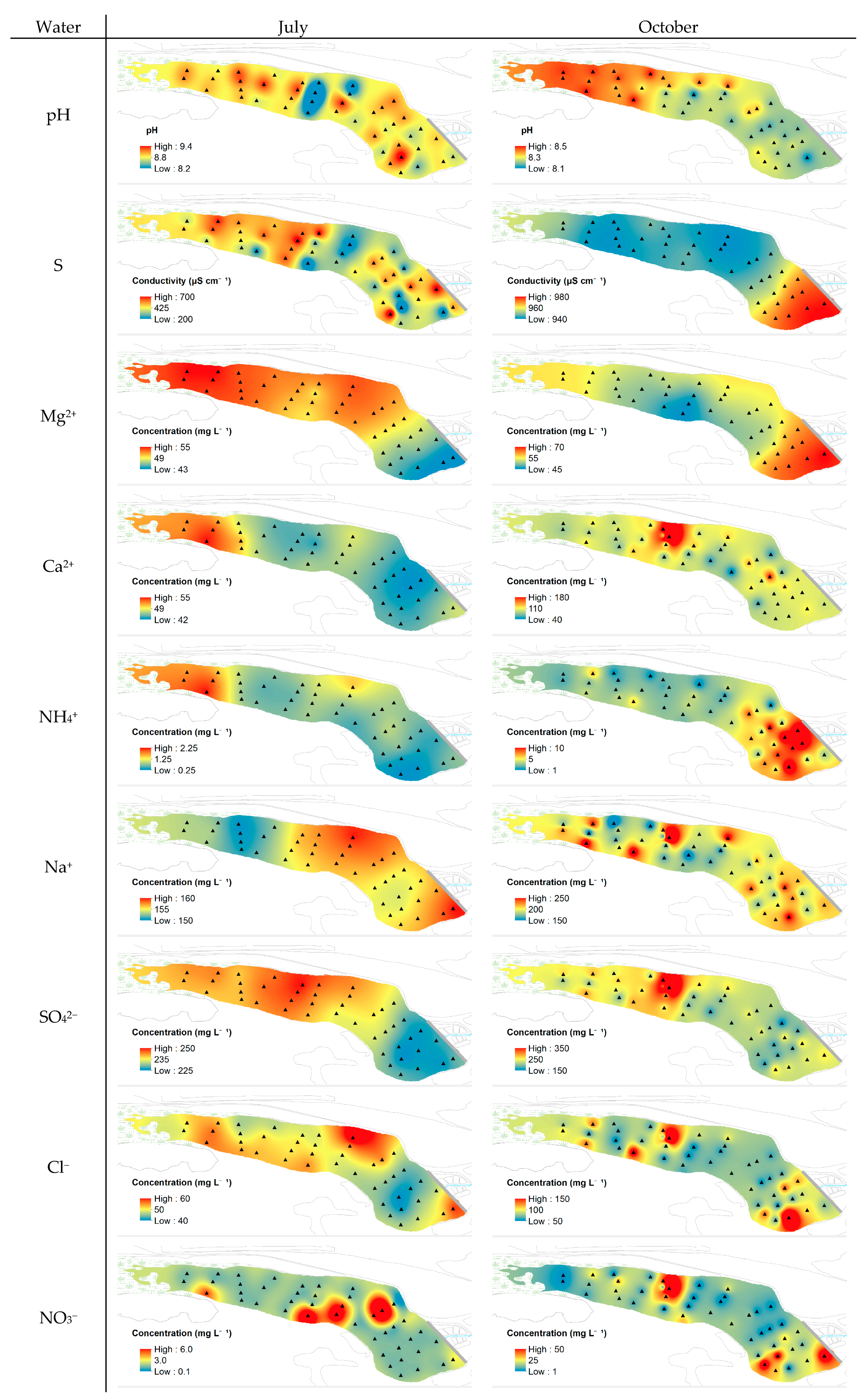
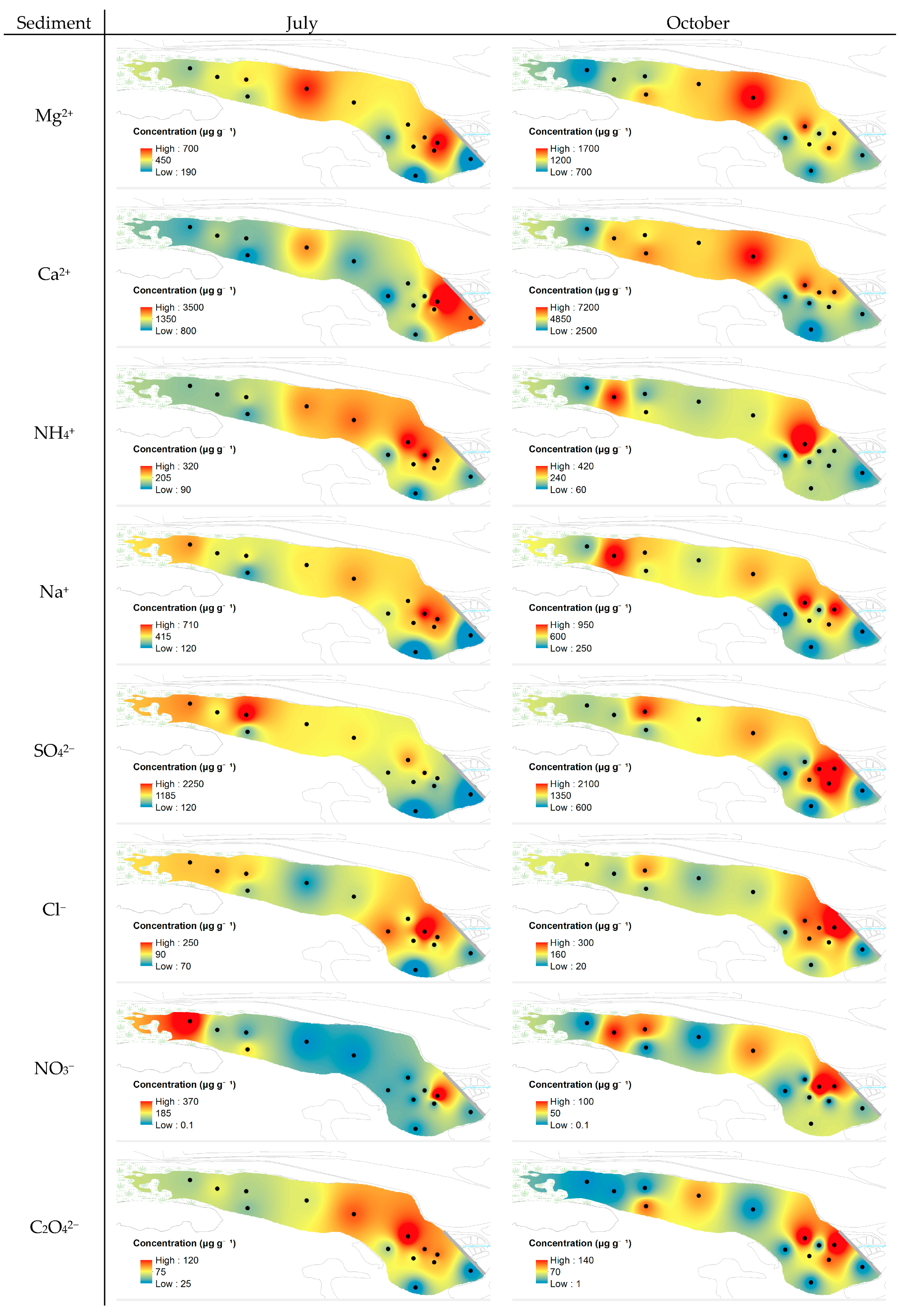
4.2. Distribution of Chemical Parameters in the Podu Iloaiei Dam Lake
4.3. Suitability of the Podu Iloaei Lake for Aquaculture Purposes and Potential Practical Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wurts, W.A.; Durborow, R.M. Southern Regional Aquaculture Center Interactions of pH, Carbon Dioxide, Alkalinity and Hardness in Fish Ponds; SRAC Publication No. 464; Southern Regional Aquaculture Center: Stoneville, MI, USA, 1992. [Google Scholar]
- Schuler, M.S.; Relyea, R.A. A Review of the Combined Threats of Road Salts and Heavy Metals to Freshwater Systems. Bioscience 2018, 68, 327–335. [Google Scholar] [CrossRef]
- Kalantzi, I.; Shimmield, T.M.; Pergantis, S.A.; Papageorgiou, N.; Black, K.D.; Karakassis, I. Heavy metals, trace elements and sediment geochemistry at four Mediterranean fish farms. Sci. Total Environ. 2013, 444, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Palanques, A.; Grimalt, J.; Belzunces, M.; Estrada, F.; Puig, P.; Guillén, J. Massive accumulation of highly polluted sedimentary deposits by river damming. Sci. Total Environ. 2014, 497–498, 369–381. [Google Scholar] [CrossRef]
- Ahmad, A.; Sheikh Abdullah, S.R.; Hasan, H.A.; Othman, A.R.; Ismail, N. Izzati Aquaculture industry: Supply and demand, best practices, effluent and its current issues and treatment technology. J. Environ. Manag. 2021, 287, 112271. [Google Scholar] [CrossRef] [PubMed]
- Boyd, C.E.; Massaut, L. Risks associated with the use of chemicals in pond aquaculture. Aquac. Eng. 1999, 20, 113–132. [Google Scholar] [CrossRef] [Green Version]
- Luo, S.; Wu, B.; Xiong, X.; Wang, J. Effects of total hardness and calcium: Magnesium ratio of water during early stages of rare minnows (Gobiocypris rarus). Comp. Med. 2016, 66, 181–187. [Google Scholar]
- Wendelaar Bonga, S.E.; Löwik, C.J.M.; van der Meij, J.C.A. Effects of external Mg2+ and Ca2+ on branchial osmotic water permeabiligy and prolactin secretion in the teleost fish Sarotherodon mossambicus. Gen. Comp. Endocrinol. 1983, 52, 222–231. [Google Scholar] [CrossRef]
- Boyd, C.E. Guidelines for aquaculture effluent management at the farm-level. Aquaculture 2003, 226, 101–112. [Google Scholar] [CrossRef]
- European Environment Agency. European Waters. Assessment of Status and Pressures 2018; European Environment Agency: Copenhagen, Denmark, 2018; ISBN 1977-8449. [Google Scholar]
- National Administration “Romanian Waters” Prut-Barlad Water Basin Administration, Annual Report 2016. Available online: http://prut-barlad.rowater.ro/wp-content/uploads/2021/07/Raport-activitate-2016.pdf (accessed on 25 July 2021).
- European Commission. Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for community action in the field of water policy. Off. J. Eur. Communities 2000, 327, 1–73. [Google Scholar]
- Ministry of Environment and Water Management. The Norm Regarding the Classification of Surface Water Quality in Order to Establish the Ecological Status of Water Bodies; Published in the Official Monitor no. 511 Bis of June 13, 2006, Approved by Order no. 161 of 16.02.2006 2006. Available online: http://legislatie.just.ro/Public/DetaliiDocumentAfis/74255 (accessed on 25 July 2021).
- Minea, I. The Evaluation of The Water Chemistry and Quality for The Lakes from the South of the Hilly Plain of Jijia (Bahlui Drainage Basin). Lakes Reserv. Ponds 2010, 4, 131–144. [Google Scholar]
- Popoiu, L.A. Cercetări Privind Calitatea Apelor din Lacurile de Acumulare Mici și Mijlocii din Zonele Colinare. Ph.D. Thesis, Gheorghe Asachi Technical University of Iaşi, Iași, Romania, 2017. [Google Scholar]
- Secu, C.V.; Breaban, I.G.; Juravle, D.T.; Lesenciuc, C.D. Human impact on urban soil properties from Bahlui Foodplain, Iasi, Romania. J. Environ. Prot. Ecol. 2016, 17, 154–163. [Google Scholar]
- Roșca, O.M.; Dippong, T.; Marian, M.; Mihali, C.; Mihalescu, L.; Hoaghia, M.A.; Jelea, M. Impact of anthropogenic activities on water quality parameters of glacial lakes from Rodnei mountains, Romania. Environ. Res. 2020, 182, 109–136. [Google Scholar] [CrossRef] [PubMed]
- Vione, D.; Falletti, G.; Maurino, V.; Minero, C.; Pelizzetti, E.; Malandrino, M.; Ajassa, R.; Olariu, R.I.; Arsene, C. Sources and sinks of hydroxyl radicals upon irradiation of natural water samples. Environ. Sci. Technol. 2006, 40, 3775–3781. [Google Scholar] [CrossRef]
- Vione, D.; Ravizzoli, B.; Rinaldi, E.; Pettinato, F.; Arsene, C.; Olariu, R.I. Studies regarding groundwater quality at rural sites: 1. Estimation of the anthropic factor impact by complementary chemical analyses. Rev. Chim. 2009, 60, 237–240. [Google Scholar]
- Vione, D.; Rinaldi, E.; Minero, C.; Maurino, V.; Olariu, R.I.; Arsene, C. Studies regarding groundwater quality at rural sites. 2. Photochemical generation of OH and NO2 radicals upon UV-A irradiation of nitrate-rich groundwater. Rev. Chim. 2009, 60, 551–554. [Google Scholar]
- Vione, D.; Lauri, V.; Minero, C.; Maurino, V.; Malandrino, M.; Carlotti, M.E.; Olariu, R.I.; Arsene, C. Photostability and photolability of dissolved organic matter upon irradiation of natural water samples under simulated sunlight. Aquat. Sci. 2009, 71, 34–45. [Google Scholar] [CrossRef] [Green Version]
- Vione, D.; Casanova, I.; Minero, C.; Duncianu, M.; Olariu, R.I.; Arsene, C. Assessing the potentiality of Romanian surface waters to produce hydroxyl and nitrite radicals. Rev. Chim. 2009, 60, 123–126. [Google Scholar]
- Vione, D.; Khanra, S.; Man, S.C.; Maddigapu, P.R.; Das, R.; Arsene, C.; Olariu, R.I.; Maurino, V.; Minero, C. Inhibition vs. enhancement of the nitrate-induced phototransformation of organic substrates by the •OH scavengers bicarbonate and carbonate. Water Res. 2009, 43, 4718–4728. [Google Scholar] [CrossRef]
- Diamantini, E.; Lutz, S.R.; Mallucci, S.; Majone, B.; Merz, R.; Bellin, A. Driver detection of water quality trends in three large European river basins. Sci. Total Environ. 2018, 612, 49–62. [Google Scholar] [CrossRef]
- National Agency for Environmental Protection. Report on the State of the Environment in Iasi County for 2018. Available online: http://www.anpm.ro/documents/839616/2486913/raport+anual+20181.pdf/7ca6f7fa-5823-4214-8c9e-a19d6fb88466 (accessed on 24 July 2021).
- WATER PIN Limited Liability Company. Securing the Podu Iloaiei Reservoir, on the Bahluet River, Iasi County, Synthesis Memoir. 2011. Available online: https://www.scribd.com/document/375298910/Memoriu-de-Sinteza-Ac-Podu-Iloaiei (accessed on 23 July 2021).
- Nicu, C.I.; Asandulesei, A. GIS-based evaluation of diagnostic areas in landslide susceptibility analysis of Bahluieț River Basin (Moldavian Plateau NE Romania). Are Neolithic sites in danger? Geomorphology 2018, 314, 27–41. [Google Scholar] [CrossRef]
- Ministry of Internal Affairs, Department for Emergency Situations and General Inspectorate for Emergency Situations. The Situation of the Accumulations under the Administration of D.A.Prut-S.G.A. from Iasi County, Annex No. 4. Available online: https://isujis.ro/wp-content/uploads/2016/04/anexa-4-SITUA%C5%A2IA-ACUMUL%C4%82RILOR.doc (accessed on 21 July 2021).
- Climate Change—Department for Waters Forests and Fisheries National Administration “Romanian Waters” Management-Bucharest, National Institute of Hydrology and Water, Prut-Barlad River Basin Management Plan, 2016–2021, In Accordance with cu Article No. 2015. Available online: https://vdocuments.mx/plan-management-prut-barlad-31032015.html (accessed on 21 July 2021).
- Arsene, C.; Olariu, R.I.; Mihalopoulos, N. Chemical composition of rainwater in the northeastern Romania, Iasi region (2003–2006). Atmos. Environ. 2007, 41, 9452–9467. [Google Scholar] [CrossRef]
- Ward, J.H., Jr. Hierarchical grouping to optimize an objective function. J. Am. Stat. Assoc. 1963, 58, 236–244. [Google Scholar] [CrossRef]
- Mathivanan, K.; Rajaram, R. Anthropogenic influences on toxic metals in water and sediment samples collected from industrially polluted Cuddalore coast, Southeast coast of India. Environ. Earth Sci. 2014, 72, 997–1010. [Google Scholar] [CrossRef]
- Rahman, M.S.; Saha, N.; Molla, A.H.; Al-Reza, S.M. Assessment of Anthropogenic Influence on Heavy Metals Contamination in the Aquatic Ecosystem Components: Water, Sediment, and Fish. Soil Sediment Contam. 2014, 23, 353–373. [Google Scholar] [CrossRef]
- Lin, Q.; Liu, E.; Zhang, E.; Li, K.; Shen, J. Spatial distribution, contamination and ecological risk assessment of heavy metals in surface sediments of Erhai Lake, a large eutrophic plateau lake in southwest China. Catena 2016, 145, 193–203. [Google Scholar] [CrossRef]
- Li, X.; Yang, H.; Zhang, C.; Zeng, G.; Liu, Y.; Xu, W.; Wu, Y.; Lan, S. Spatial distribution and transport characteristics of heavy metals around an antimony mine area in central China. Chemosphere 2017, 170, 17–24. [Google Scholar] [CrossRef]
- Kennedy, K.H. Introduction to 3D Data Modeling whit ArcGIS 3D AnalystT and Google Earth; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; ISBN 9780470548776. [Google Scholar]
- Johnston, K.; Krivoruchko, K.; Lucas, N. Using ArcGIS Geostatistical Analyst; Esri: West Redlands, CA, USA, 2001. [Google Scholar]
- Muranova, T.A.; Zinchenko, D.V.; Kononova, S.V.; Belova, N.A.; Miroshnikov, A.I. Plant protein hydrolysates as fish fry feed in aquaculture. Hydrolysis of rapeseed proteins by an enzyme complex from king crab hepatopancreas. Appl. Biochem. Microbiol. 2017, 53, 680–687. [Google Scholar] [CrossRef]
- Quinto, B.P.T.; Albuquerque, J.V.; Bezerra, R.S.; Peixoto, S.; Soares, R. Replacement of fishmeal by two types of fish protein hydrolysate in feed for postlarval shrimp Litopenaeus vannamei. Aquac. Nutr. 2018, 24, 768–776. [Google Scholar] [CrossRef]
- Daher, S.; Gülaçar, F. Geochemistry of carboxylic acids in the sediments from lake Cadagno (Switzerland). Arch. Sci. C. R. Seances Soc. 2005, 58, 25–42. [Google Scholar]
- Jonsson, A.; Ström, L.; Åberg, J. Composition and variations in the occurrence of dissolved free simple organic compounds of an unproductive lake ecosystem in northern Sweden. Biogeochemistry 2007, 82, 153–163. [Google Scholar] [CrossRef]
- Prychid, C.J.; Rudall, P.J. Calcium oxalate crystals in monocotyledons: A review of their structure and systematics. Ann. Bot. 1999, 84, 725–739. [Google Scholar] [CrossRef]
- Smith, R.L.; Oremland, R.S. Anaerobic Oxalate Degradation: Widespread Natural Occurrence in Aquatic Sediments. Appl. Environ. Microbiol. 1983, 46, 106–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Škrbić, B.; Durišić-Mladenović, N. Chemometric interpretation of heavy metal patterns in soils worldwide. Chemosphere 2010, 80, 1360–1369. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D. Water Quality Assessments—A Guide to Use of Biota, Sediments and Water in Environmental Monitoring, 2nd ed.; E&FN Spon: London, UK, 1996; ISBN 0419216006. [Google Scholar]
- Cristini, P.A.; Tietze, E.; De Francesco, C.G.; Martínez, D.E. Water geochemistry of shallow lakes from the southeastern Pampa plain, Argentina and their implications on mollusk shells preservation. Sci. Total Environ. 2017, 603–604, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Acosta, J.A.; Jansen, B.; Kalbitz, K.; Faz, A.; Martínez-Martínez, S. Salinity increases mobility of heavy metals in soils. Chemosphere 2011, 85, 1318–1324. [Google Scholar] [CrossRef] [PubMed]
- Roy, K.; Chari, M.S.; Gaur, S.R.; Thakur, A. Ecological dynamics and hydrobiological correlations in freshwater ponds-recent researches and application. Int. J. Environ. Biol. 2014, 4, 112–118. [Google Scholar] [CrossRef] [Green Version]
- Dodds, W.K.; Jones, J.R.; Welch, E.B. Suggested classification of stream trophic state: Distributions of temperate stream types by chlorophyll, total nitrogen, and phosphorus. Water Res. 1998, 32, 1455–1462. [Google Scholar] [CrossRef]


| Species/Parameter | Water | Sediment | ||
|---|---|---|---|---|
| July | October | July | October | |
| Cl− | 48.0 ± 5.2 (46.8) | 63.0 ± 17.7 (57.6) | 139 ± 39 (134) | 94.7 ± 64.8 (74.7) |
| SO42− | 234 ± 7 (235) | 181 ± 24 (179) | 1121 ± 546 (1155) | 1297 ± 484 (1170) |
| Na+ | 153 ± 4 (152) | 177± 9 (175) | 476 ± 169 (513) | 595 ± 195 (578) |
| K+ | 13.6 ± 8.3 (10.4) | 19.8 ± 15.3 (12.6) | 284 ± 98 (294) | 564 ± 133 (625) |
| Mg2+ | 56.7 ± 1.6 (56.6) | 62.9 ± 4.1 (62.7) | 550 ± 139 (538) | 1639 ± 320 (1728) |
| Ca2+ | 47.5 ± 6.7 (45.6) | 61.1 ± 17.8 (58.7) | 1338 ± 692 (1179) | 5041 ± 1350 (5305) |
| N-NO2− | 0.40 ± 0.27 (0.40) | 0.98 ± 1.12 (0.52) | 28.0 ± 40.1 (11.4) | <1.86 |
| N-NO3− | 0.56 ± 0.42 (0.47) | 1.56 ± 2.18 (0.92) | 18.8 ± 26.4 (6.2) | 7.92 ± 6.83 (8.29) |
| N-NH4+ | 0.65 ± 0.46 (0.55) | 2.74 ± 1.67 (2.28) | 142 ± 55 (138) | 106 ± 70 (87) |
| P-PO43− | <1.06 | 3.40 ± 2.20 (3.38) | 17.0 ± 10.4 (11.5) | 45.9 ± 16.7 (44.8) |
| H3C2O2− | 32.5 ± 30.5 (24.1) | 43.0 ± 43.0 (26.3) | 394 ± 462 (230) | 223 ± 118 (202) |
| HCO2− | 4.27 ± 0.74 (4.27) | 5.72 ± 6.57 (3.75) | 91.1 ± 66.3 (63.6) | 98.2 ± 47.4 (86.2) |
| C2O42− | <7.00 | <7.00 | 64.0 ± 24.2 (60.3) | 83.1 ± 35.9 (71.2) |
| pH | 9.09 ± 0.15 (9.11) | 8.25 ± 0.07 (8.24) | ||
| S | 474 ± 121 (491) | 956 ± 23 (948) | ||
| CaCO3 | 366 ± 19 (349) | 412 ± 58 (404) | ||
| ANC | 7.80 ± 0.52 (7.71) | 10.8 ± 0.8 (10.6) | ||
| Element | Water | Sediment | ||
|---|---|---|---|---|
| July | October | July | October | |
| Al | 1.00 ± 2.03 (0.48) | 1.35 ± 1.70 (0.68) | 96.5 ± 57.1 (79.6) | 143 ± 80 (120) |
| Ti | <0.60 | <0.60 | 8.67 ± 4.99 (7.60) | 5.36 ± 4.93 (2.94) |
| Cr | 0.019 ± 0.011 (0.019) | 0.064 ± 0.048 (0.051) | 1.21 ± 0.81 (0.79) | 1.13 ± 0.80 (0.84) |
| Mn | 0.097 ± 0.083 (0.072) | 0.33 ± 0.21 (0.34) | 20.0 ± 9.6 (18.7) | 45.4 ± 26.9 (38.2) |
| Ni | 0.12 ± 0.17 (0.09) | 1.08 ± 3.04 (0.41) | 14.4 ± 3.8 (14.2) | 17.2 ± 5.3 (17.2) |
| Zn | 0.79 ± 1.67 (0.12) | 1.88 ± 2.52 (1.27) | 15.3 ± 6.5 (17.7) | 97 ± 161 (37) |
| Ge | 0.14 ± 0.11 (0.13) | 0.19 ± 0.13 (0.15) | 19.6 ± 4.4 (20.3) | 16.8 ± 4.1 (16.9) |
| Sr | 0.54 ± 0.09 (0.52) | 0.60 ± 0.42 (0.52) | 10.6 ± 4.1 (9.4) | 40.6 ± 11.5 (39.5) |
| Ag | 0.075 ± 0.037 (0.073) | 0.13 ± 0.10 (0.10) | 0.61 ± 0.39 (0.45) | 0.95 ± 0.75 (0.63) |
| Sb | 0.006 ± 0.005 (0.005) | 0.04 ± 0.06 (0.02) | 0.26 ± 0.20 (0.18) | 0.24 ± 0.13 (0.25) |
| Ba | 0.059 ± 0.041 (0.054) | 0.16 ± 0.14 (0.12) | 22.1 ± 3.6 (21.7) | 30.3 ± 8.3 (31.5) |
| Pb | 0.071 ± 0.062 (0.062) | 0.08 ± 0.12 (0.05) | 1.93 ± 1.49 (1.52) | 1.20 ± 1.99 (0.62) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amarandei, C.; Negru, A.-G.; Soroaga, L.-V.; Cucu-Man, S.-M.; Olariu, R.-I.; Arsene, C. Assessment of Surface Water Quality in the Podu Iloaiei Dam Lake (North-Eastern Romania): Potential Implications for Aquaculture Activities in the Area. Water 2021, 13, 2395. https://doi.org/10.3390/w13172395
Amarandei C, Negru A-G, Soroaga L-V, Cucu-Man S-M, Olariu R-I, Arsene C. Assessment of Surface Water Quality in the Podu Iloaiei Dam Lake (North-Eastern Romania): Potential Implications for Aquaculture Activities in the Area. Water. 2021; 13(17):2395. https://doi.org/10.3390/w13172395
Chicago/Turabian StyleAmarandei, Cornelia, Alina-Giorgiana Negru, Laurentiu-Valentin Soroaga, Simona-Maria Cucu-Man, Romeo-Iulian Olariu, and Cecilia Arsene. 2021. "Assessment of Surface Water Quality in the Podu Iloaiei Dam Lake (North-Eastern Romania): Potential Implications for Aquaculture Activities in the Area" Water 13, no. 17: 2395. https://doi.org/10.3390/w13172395
APA StyleAmarandei, C., Negru, A.-G., Soroaga, L.-V., Cucu-Man, S.-M., Olariu, R.-I., & Arsene, C. (2021). Assessment of Surface Water Quality in the Podu Iloaiei Dam Lake (North-Eastern Romania): Potential Implications for Aquaculture Activities in the Area. Water, 13(17), 2395. https://doi.org/10.3390/w13172395






