Review of Techniques to Reduce and Prevent Carbonate Scale. Prospecting in Water Treatment by Magnetism and Electromagnetism
Abstract
:1. Introduction
2. Methodology
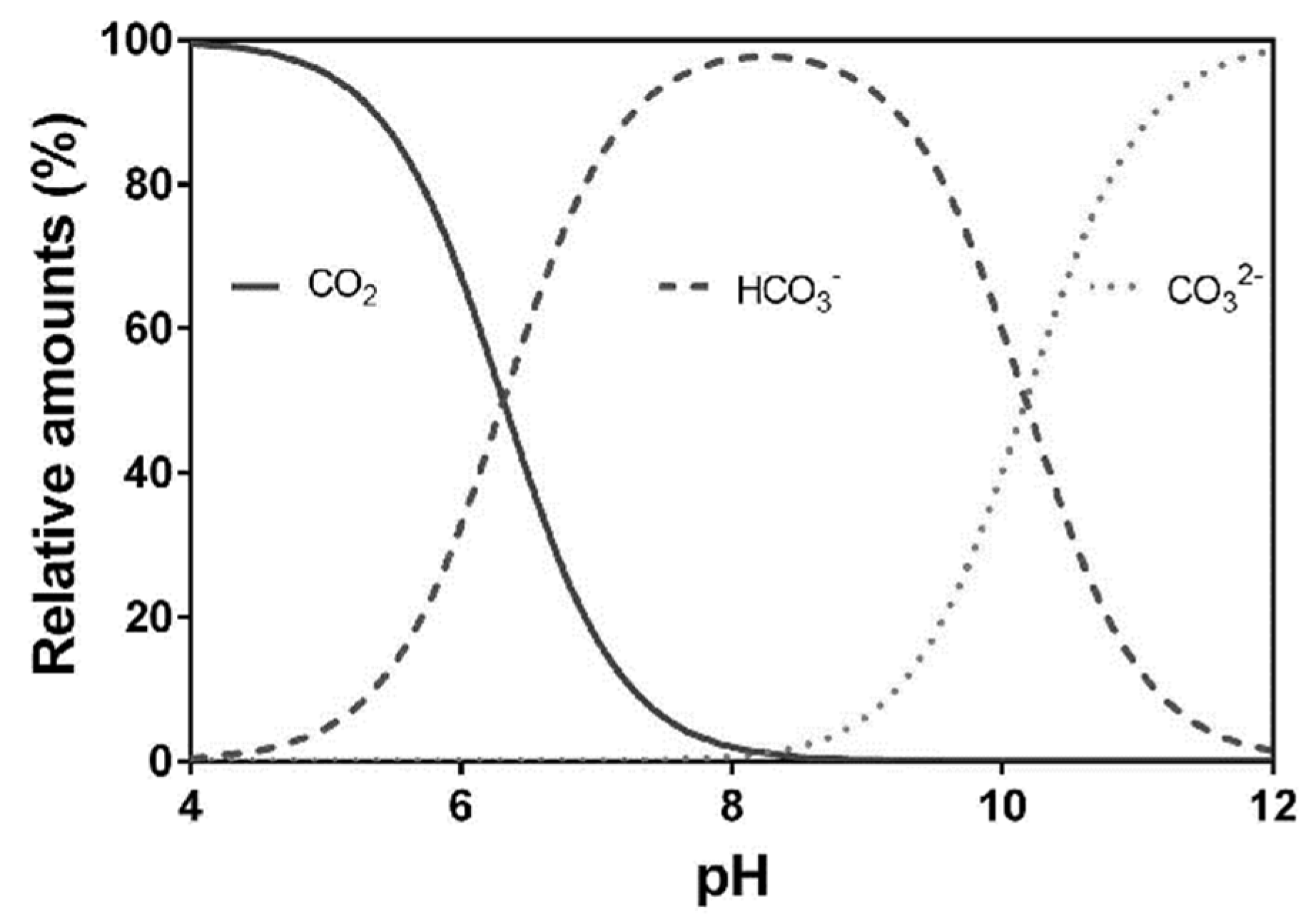
3. Calcium Carbonate and the CO2/HCO3−/CO32− System
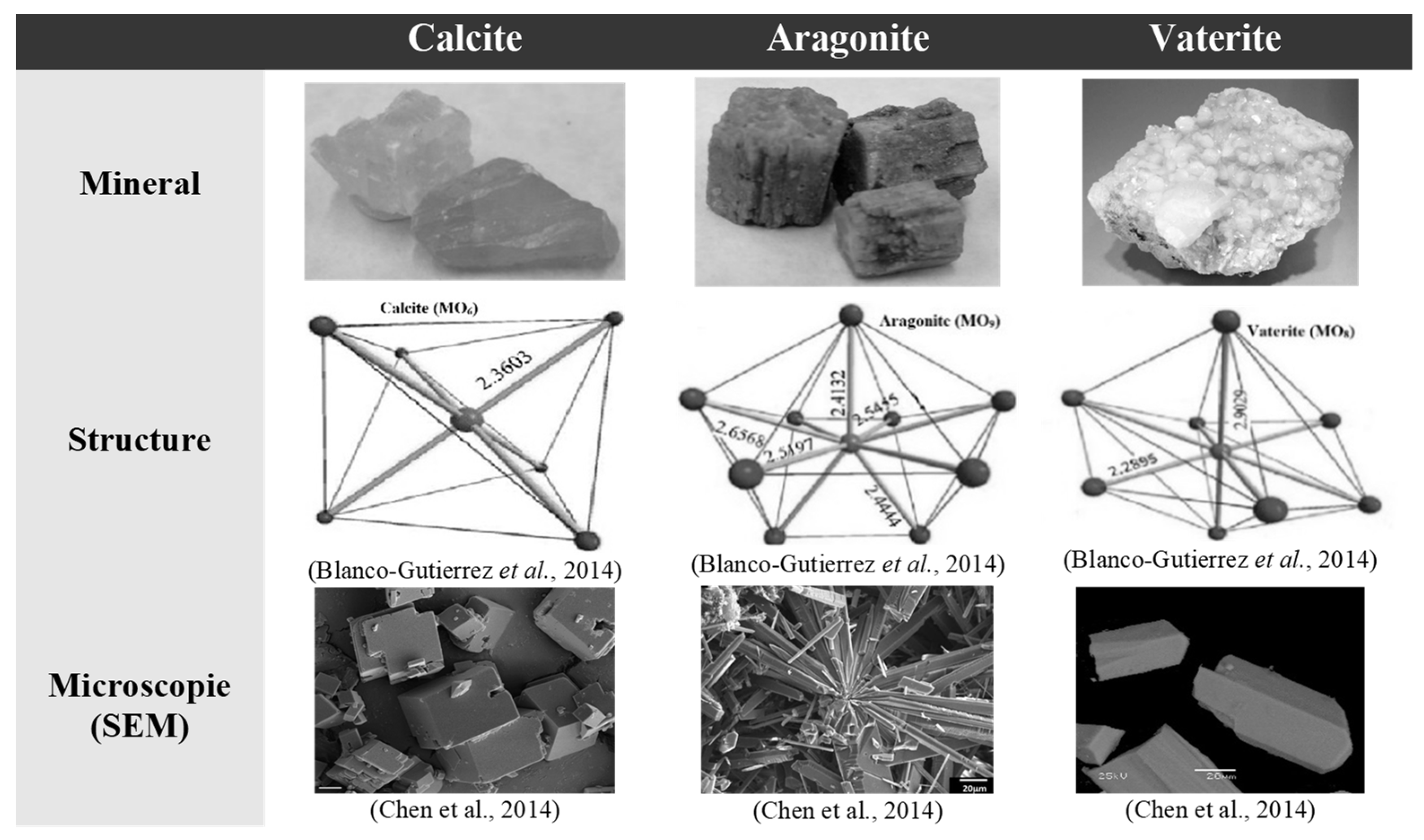
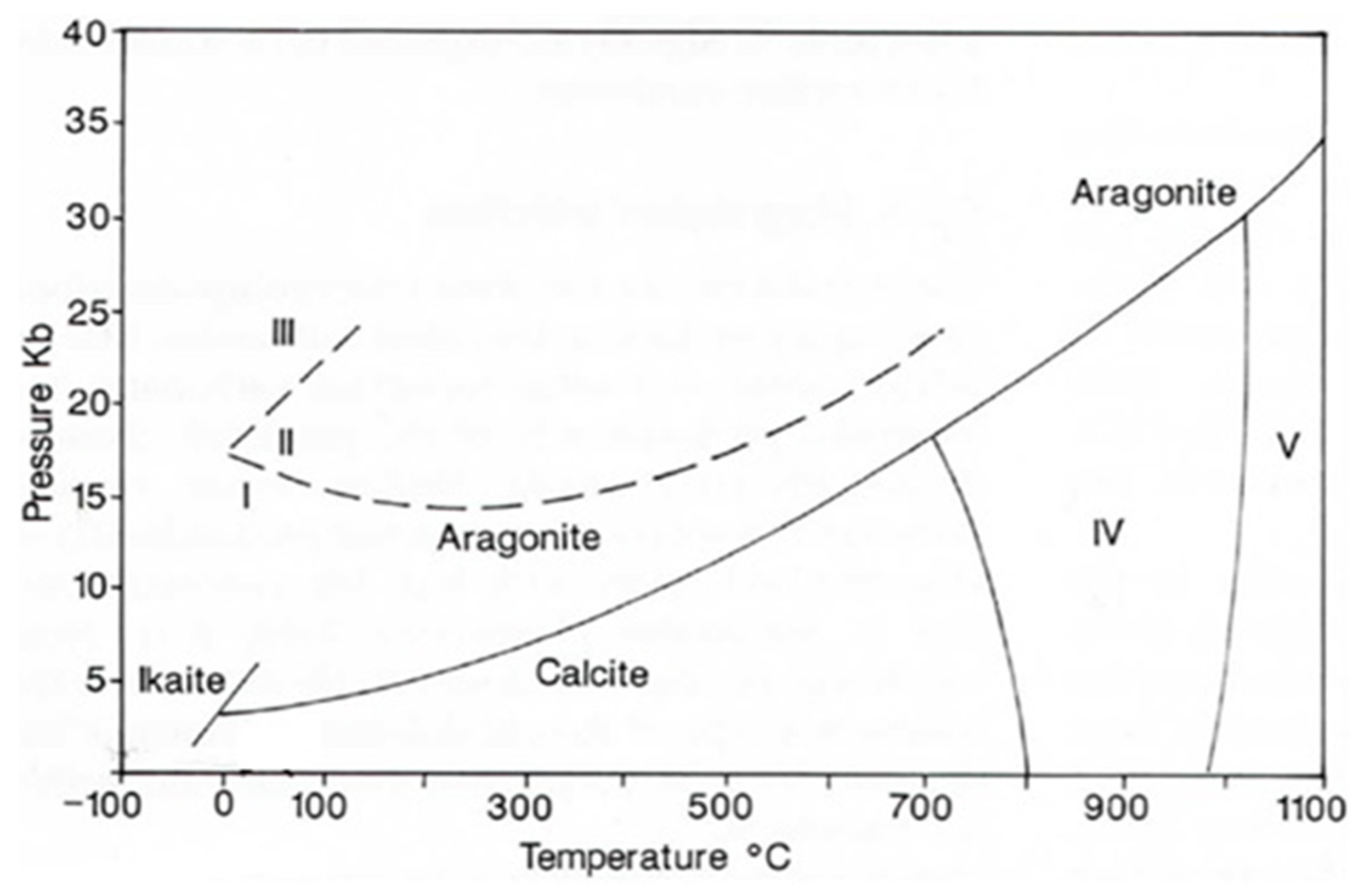
3.1. CaCO3 Polymorphs
3.2. Identification of Mineral Phases of Calcium Carbonate
- Secondary electrons. Produced from the emission of valence electrons. They are of very low energy so only the most superficial come out of the sample and provide information about the topography of the surface.
- Backscattered electrons. They have higher energy than secondary electrons, therefore they provide information on deeper parts of the sample and are sensitive to the composition of the sample.
- Characteristic X-rays. The energy of transition from one layer to another is characteristic of the element from which they come. Provides chemical information for the sample.
4. Treatments to Avoid Scale Formation
4.1. Chemical Precipitation
- Using calcium and sodium hydroxide ((7)–(10))
- Using sodium carbonate ((11) and (12))
4.2. Complexing Agents and Other Inhibitors of Precipitation
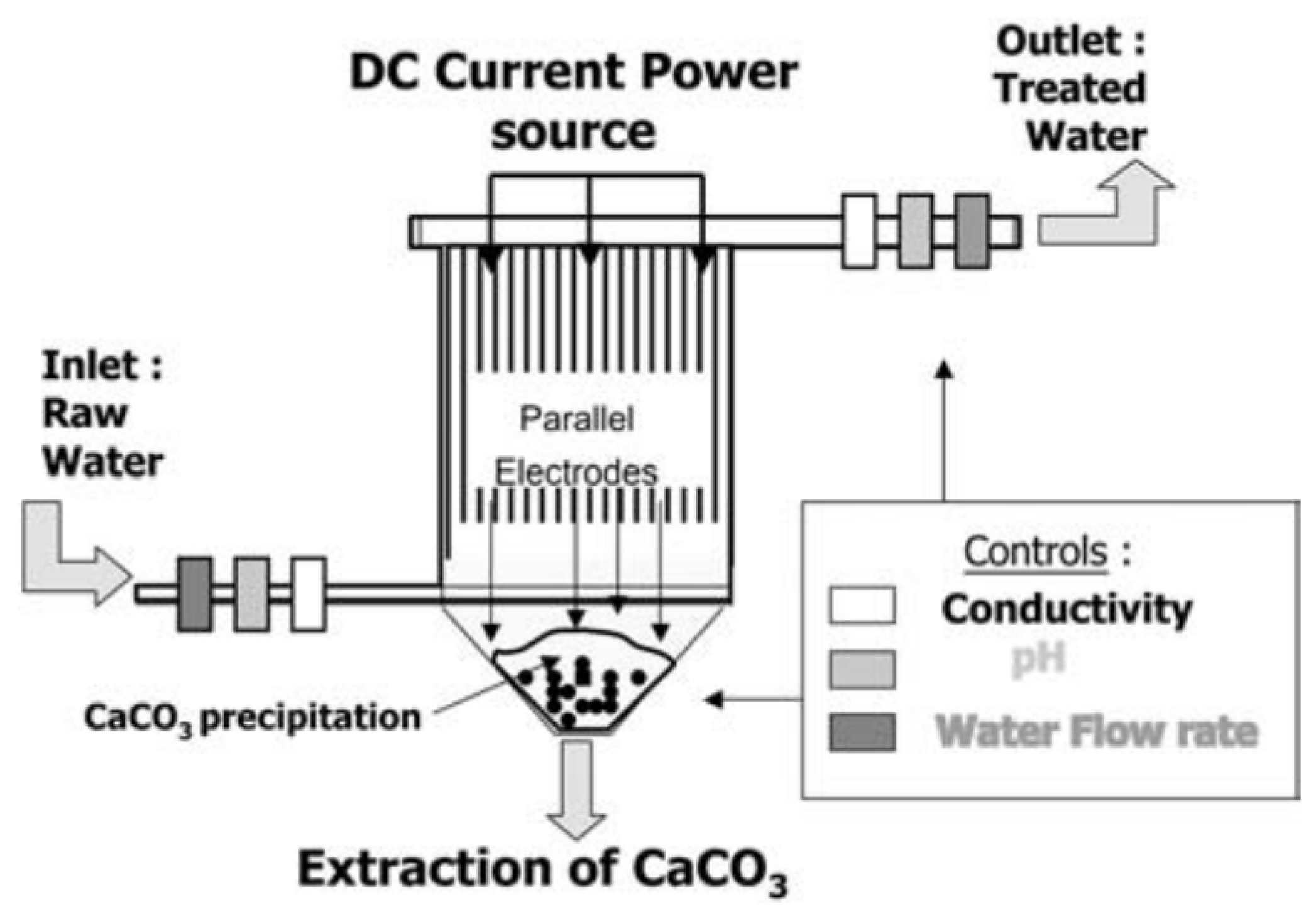
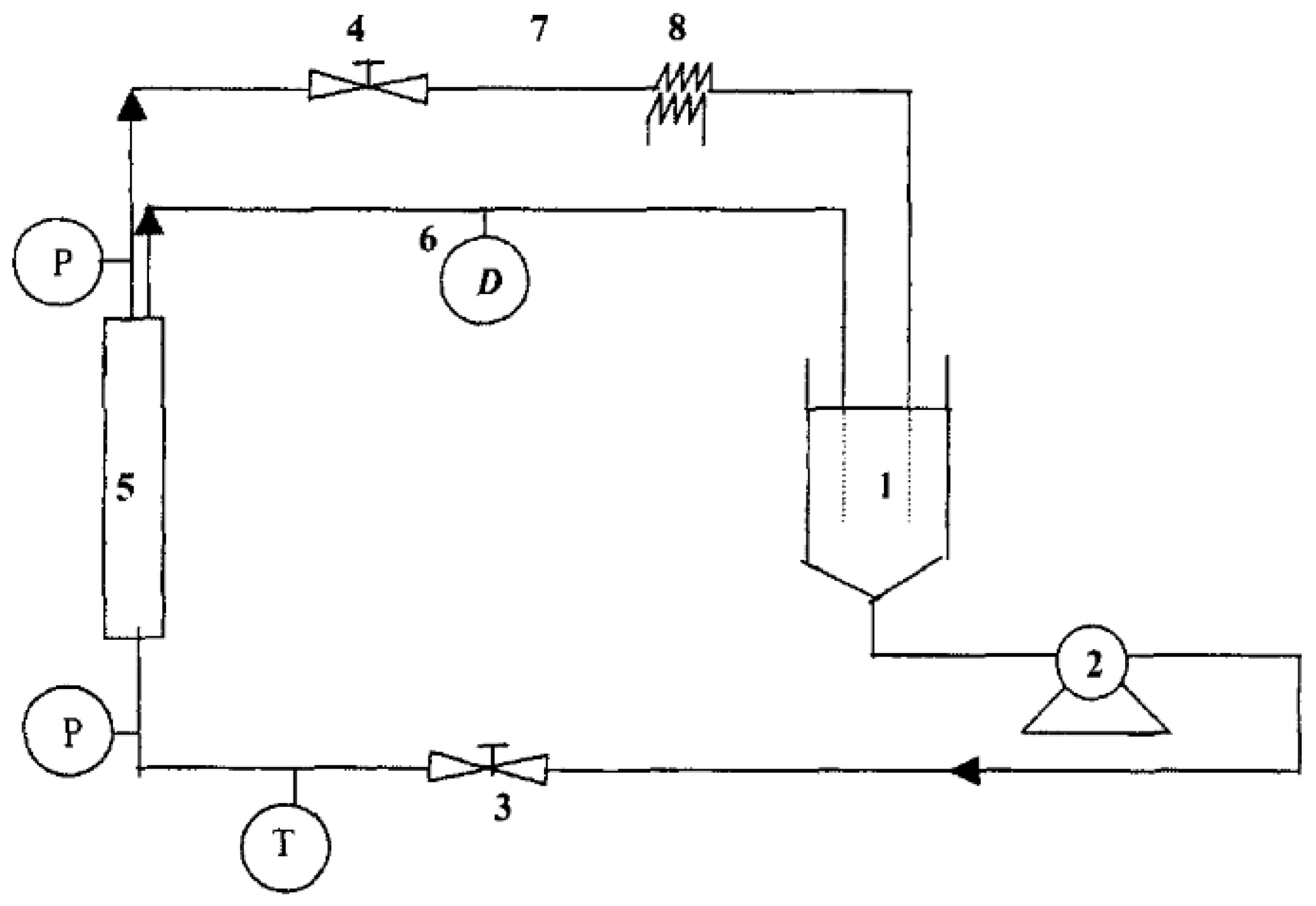
4.3. Electrochemical Treatments
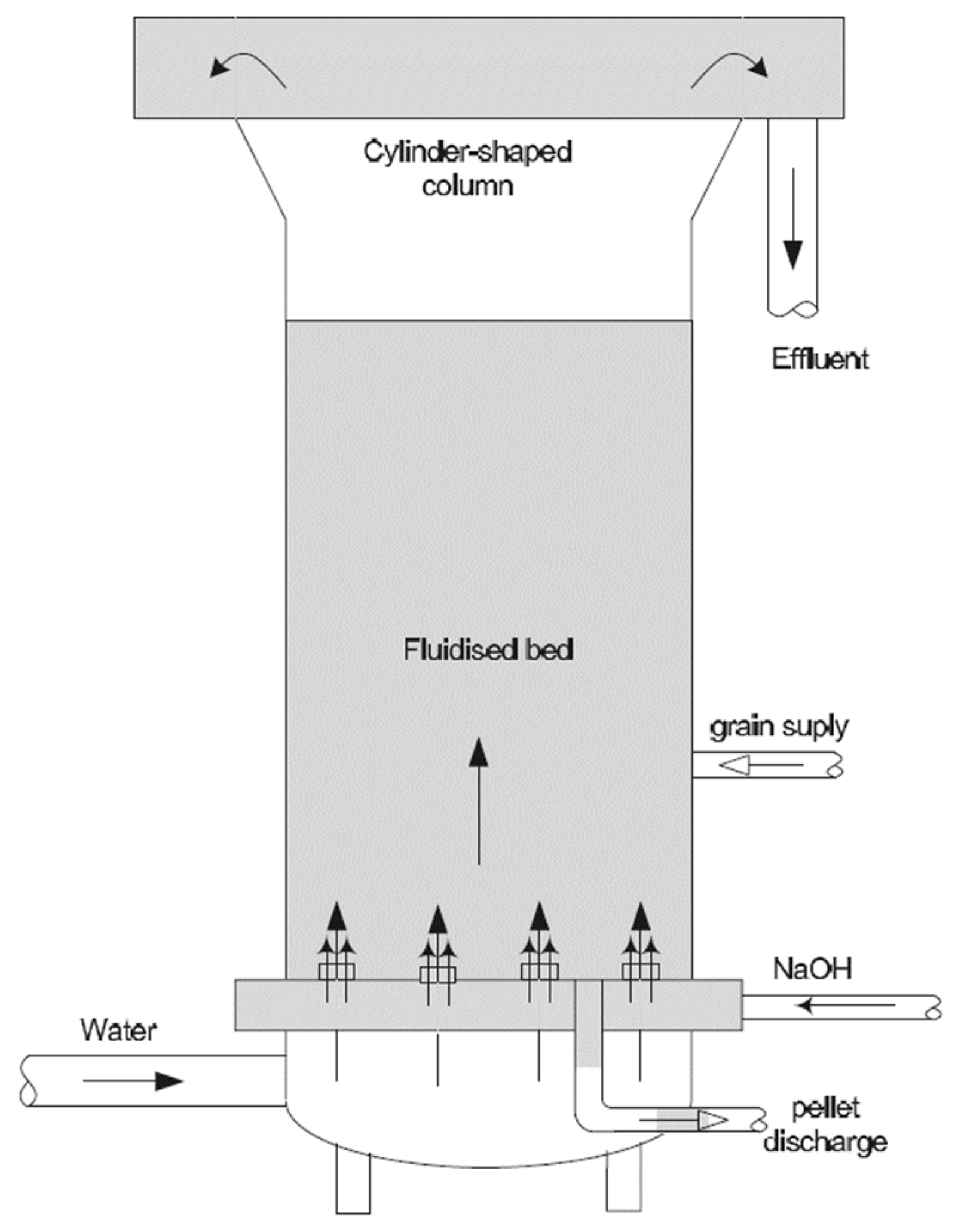
4.4. Cationic Exchange
4.5. Membrane Treatment
4.6. Other Treatments
4.7. Advantages and Disadvantages of Classical Water Softening Techniques and Comparative
5. Application of Magnetic and Electromagnetic Techniques to Prevent the Effects of Water Hardness
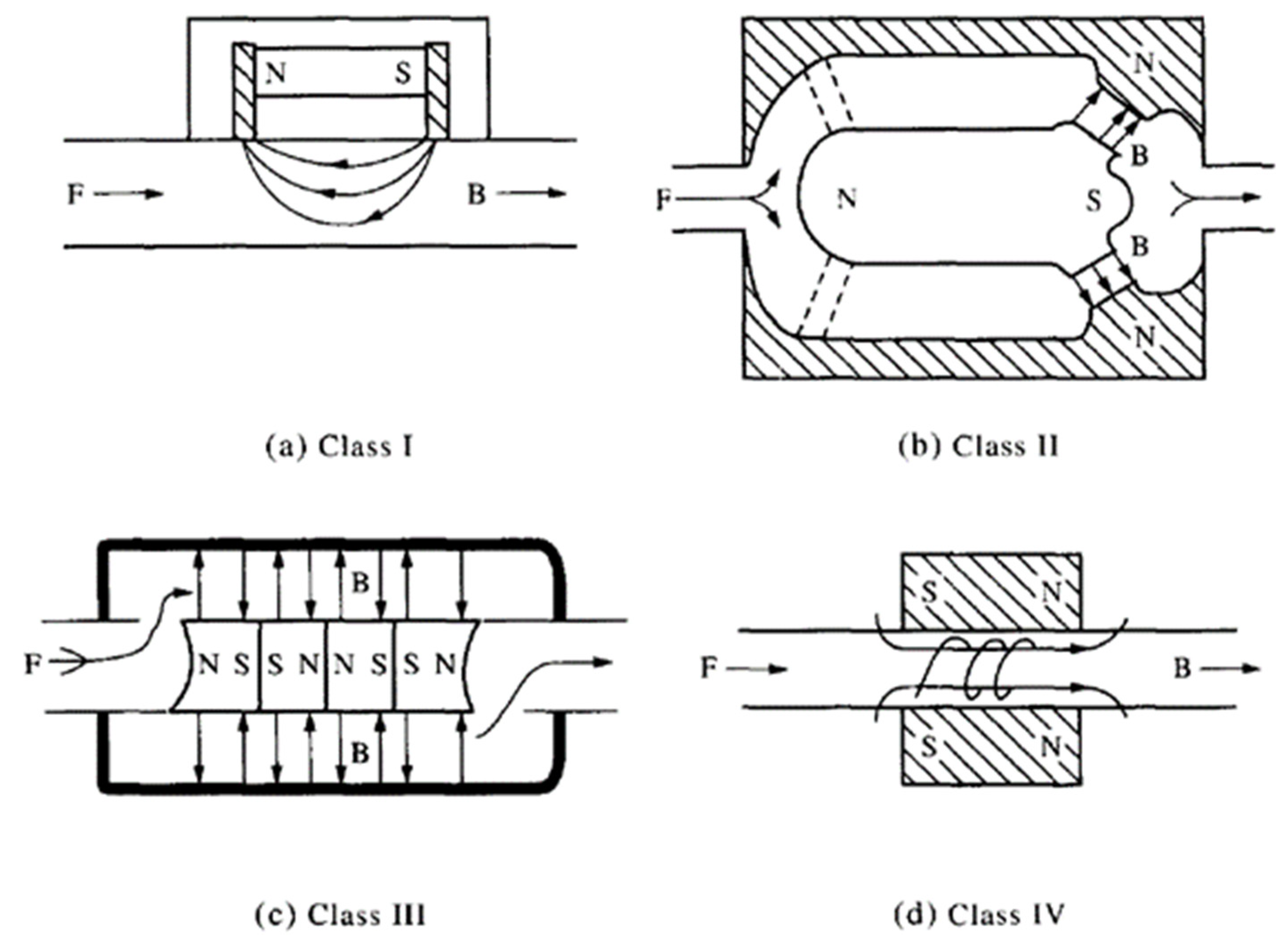
5.1. Magnetic Techniques
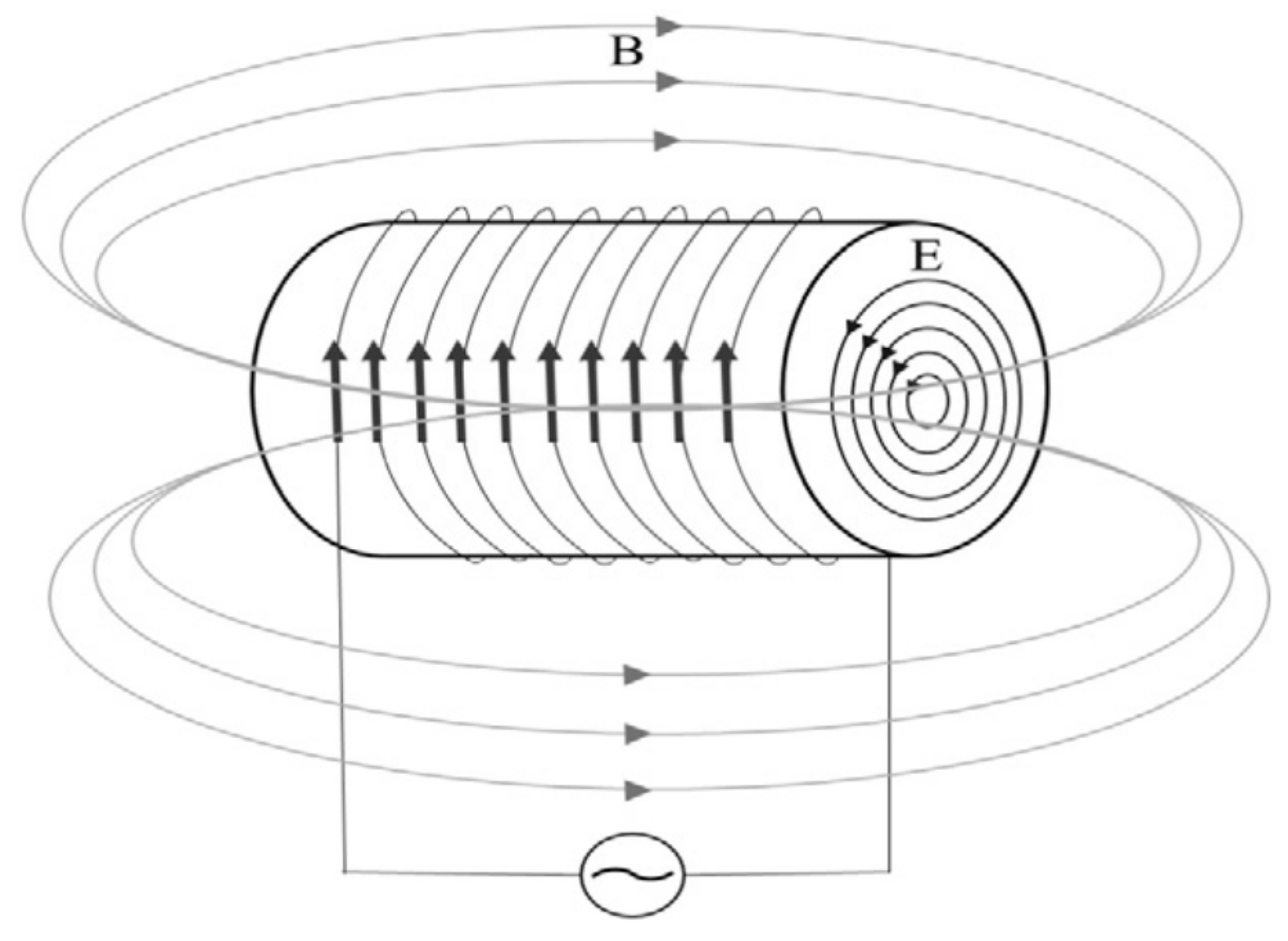
5.2. Electromagnetic Techniques
6. Studies of the Effects of Magnetic and Electromagnetic Fields on the Water Hardness
6.1. Identification of Crystalline Forms
6.1.1. Formation of Calcite and Aragonite in Scale
6.1.2. Reversion of Aragonite to Calcite
6.1.3. Formation of Calcite and Aragonite Induced by Evaporation
6.1.4. Orientation of Aragonite Crystals
6.2. Liquid Phase Studies
6.2.1. Crystal Formation within the Fluid
6.2.2. Effects on Water Properties
7. Other Magnetic and Electromagnetic Applications
7.1. Combining with Other Treatments
7.2. Microbiological Effects
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ghizellaoui, S.; Euvrard, M.; Ledion, J.; Chibani, A. Inhibition of scaling in the presence of copper and zinc by various chemical processes. Desalination 2007, 206, 185–197. [Google Scholar] [CrossRef]
- Eaton, A.D.; Clesceri, L.S.; Rice, E.W.; Greenberg, A.E.; Franson, A.M.H. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Associaton: Washington, DC, USA, 2005. [Google Scholar]
- Kirk, R.E.; Othmer, D.F.; Grayson, M.; Eckroth, D. Encyclopedia of Chemical Technology, 4th ed.; Wiley: New York, NY, USA, 1998. [Google Scholar]
- Kiely, G. Ingeniería Ambiental: Fundamentos, Entornos, Tecnologías y Sistemas de Gestión; McGraw-Hill/Interamericana de Espana, S.A.: Madrid, Spain, 1999; ISBN 9788448121518. [Google Scholar]
- Elsevier, B. Scopus Database. 2020. Available online: https://www.scopus.com/search/form.uri?display=basic#basic (accessed on 20 August 2021).
- Appelo, C.A.J.; Postma, D. Geochemistry, groundwater and pollution. Geochem. Groundw. Pollut. 1993. [Google Scholar] [CrossRef]
- de Leeuw, N.H.; Parker, S.C. Surface Structure and Morphology of Calcium Carbonate Polymorphs Calcite, Aragonite, and Vaterite: An Atomistic Approach. J. Phys. Chem. B 1998, 102, 2914–2922. [Google Scholar] [CrossRef]
- Pedersen, O.; Colmer, T.; Sand-Jensen, K. Underwater Photosynthesis of Submerged Plants–Recent Advances and Methods. Front. Plant Sci. 2013, 4, 140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gimeno Amorós, M.; Martínez Moya, S.; Boluda Botella, N. Tratamiento de Agua Por Electromagnetismo: Aplicación de Técnicas de Identificación de Calcita y Aragonito; Repositorio Institucional de la Universidad de Alicante: Alicante, Spain, 2020. [Google Scholar]
- Blanco-Gutierrez, V.; Demourges, A.; Jubera, V.; Gaudon, M. Eu(III)/Eu(II)-doped (Ca0.7Sr0.3)CO3 phosphors with vaterite/calcite/aragonite forms as shock/temperature detectors. J. Mater. Chem. 2014, 2, 9969–9977. [Google Scholar] [CrossRef]
- Chen, Z.; Xin, M.; Li, M.; Xu, J.; Li, X.; Chen, X. Biomimetic synthesis of coexistence of vaterite-calcite phases controlled by histidine-grafted-chitosan. J. Cryst. Growth 2014, 404, 107–115. [Google Scholar] [CrossRef]
- Tucker, M.; Wright, P. Carbonate Sedimentology; Blackwell Scientific Publications: Oxford, UK; London, UK, 1990. [Google Scholar]
- Wang, H.; Huang, W.; Han, Y. Diffusion-reaction compromise the polymorphs of precipitated calcium carbonate. Particuology 2013, 11, 301–308. [Google Scholar] [CrossRef]
- Coey, J.M.D.; Cass, S. Magnetic water treatment. J. Magn. Magn. Mater. 2000, 209, 71–74. [Google Scholar] [CrossRef] [Green Version]
- Raisen, E. The control of scale and corrosion in water systems using magnetic fields. In Proceedings of the International Corrosion Forum Devoted Exclusively to the Protection and Performance of Materials, New Orleans, LA, USA, 2–6 April 1984; National Association of Corrosion Engineers: New Orleans, USA, 1984. [Google Scholar]
- Herzog, R.E.; Shi, Q.; Patil, J.N.; Katz, J.L. Magnetic Water Treatment: The Effect of Iron on Calcium Carbonate Nucleation and Growth. Langmuir 1989, 5, 861–867. [Google Scholar] [CrossRef]
- Kobe, S.; Dražić, G.; Cefalas, A.C.; Sarantopoulou, E.; Stražišar, J. Nucleation and crystallization of CaCO3 in applied magnetic fields. Cryst. Eng. 2002, 5, 243–253. [Google Scholar] [CrossRef]
- Martínez Moya, S.; Boluda Botella, N.; García Quiles, J. Effect of electromagnetic water treatment on carbonate scale. Determination of calcite and aragonite by the Meigen test. In Proceedings of the 14th Mediterranean Congress of Chemical Engineering, Barcelona, Spain, 16–20 November 2020. [Google Scholar]
- Van, H.T.; Huong, L.; Nguyen, V.D.; Hoan, X. Characteristics and mechanisms of cadmium adsorption onto biogenic aragonite shells-derived biosorbent: Batch and column studies. J. Environ. Manag. 2019, 241, 535–548. [Google Scholar] [CrossRef] [PubMed]
- Sibony-nevo, O.; Pinkas, I.; Farstey, V.; Baron, H.; Addadi, L.; Weiner, S. The Pteropod Creseis acicula Forms Its Shell through a Disordered Nascent Aragonite Phase. Cryst. Growth Des. 2019, 19, 2564–2573. [Google Scholar] [CrossRef]
- Raz, S.; Hamilton, P.C.; Wilt, F.H.; Weiner, S.; Addadi, L. The transient phase of amorphous calcium carbonate in sea urchin larval spicules: The involvement of proteins and magnesium ions in its formation and stabilization. Adv. Funct. Mater. 2003, 13, 480–486. [Google Scholar] [CrossRef]
- Kong, J.; Liu, C.; Yang, D.; Yan, Y.; Chen, Y.; Huang, J.; Liu, Y.; Zheng, G.; Xie, L.; Zhang, R. Alv Protein Plays Opposite Roles in the Transition of Amorphous Calcium Carbonate to Calcite and Aragonite during Shell Formation. Cryst. Growth Des. 2018, 18, 3794–3804. [Google Scholar] [CrossRef]
- Friedman, G.M. Identification of Carbonate Minerals by Staining Methods. SEPM J. Sediment. Res. 1959, 29, 87–97. [Google Scholar] [CrossRef]
- Tamer, A. Chemical staining methods used in the identification of carbonate minerals. Bull. Miner. Res. Explor. Inst. Turk. 1965, 65, 133–147. [Google Scholar]
- Pueyo, J.J.; Alonso, R. Génesis de Formaciones Evaporíticas: Modelos Andinos e Ibéricos; Universitat de Barcelona: Barcelona, Spain, 1991; ISBN 84-7875-666-3. [Google Scholar]
- Togari, K.; Togari, S. Conditions Controlling the Crystal from of Calcium Carbonate Minerals (I) (On the Influences of the Temperature and the Presence of Magnesium Ion). J. Fac. Sci. Hokkaido Univ. 1955, 9, 55–65. [Google Scholar]
- Salzer, H. Eine einfache Reaktion zur U nterscheidung von Calcit und A ragonit. Speläologisches Jahrb. 1936, 76–79. Available online: http://www.xray.cz/xray/csca/kol2011/kurs/dalsi-cteni/connolly-2005/01-xrd-intro.pdf (accessed on 20 August 2021).
- Jirová, D. Modifications of CaCO3 in the shells of foraminifera. Časopis Pro Mineral. A Geol. 1956, 1, 101–104. [Google Scholar]
- Verdú, D. Efecto del Electromagnetismo Sobre la Dureza del Agua; Repositorio Institucional de la Universidad de Alicante: Alicante, Spain, 2016. [Google Scholar]
- Feigl, F. Qualitative Analysis by Spot Tests. III; Elsevier Publishing Company: New York, NY, USA, 1947. [Google Scholar]
- Goldstein, J.I. Scanning Electron Microscopy and X-ray Microanalysis; Springer: New York, NY, USA, 2013. [Google Scholar]
- Connolly, J.R. Introduction to X-ray Powder Diffraction. 2007, pp. 1–9. Available online: https://www.google.com.hk/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwjbzdS08NjyAhWHO3AKHSR_CW0QFnoECAIQAQ&url=http%3A%2F%2Fwww.xray.cz%2Fxray%2Fcsca%2Fkol2011%2Fkurs%2Fdalsi-cteni%2Fconnolly-2005%2F01-xrd-intro.pdf&usg=AOvVaw1PRd7YZi4ZI3dWNOKGmZZz (accessed on 20 August 2021).
- Bunaciu, A.A.; UdriŞTioiu, E.; Aboul-Enein, H. X-Ray Diffraction: Instrumentation and Applications. Crit. Rev. Anal. Chem. CRC 2015, 45. [Google Scholar] [CrossRef] [PubMed]
- Letterman, R.D. Calidad y Tratamiento del Agua; McGraw Hill: Madrid, Spain, 2002. [Google Scholar]
- Van Schagen, K.M.; Babuška, R.; Rietveld, L.C.; Veersma, A.M.J. Model-based dosing control of a pellet softening reactor. IFAC Proc. Vol. 2009, 7, 267–272. [Google Scholar] [CrossRef] [Green Version]
- Rahmani, K.; Jadidian, R.; Haghtalab, S. Evaluation of inhibitors and biocides on the corrosion, scaling and biofouling control of carbon steel and copper–nickel alloys in a power plant cooling water system. Desalination 2016, 393, 174–185. [Google Scholar] [CrossRef]
- Wang, C.; Li, S.; Li, T. Calcium carbonate inhibition by a phosphonate-terminated poly(maleic-co-sulfonate) polymeric inhibitor. Desalination 2009, 249, 1–4. [Google Scholar] [CrossRef]
- Ghizellaoui, S.; Boumagoura, M.; Rhouati, S.; Cheap-Charpentier, H.; Horner, O. Inhibition of CaCO3 growth in hard water by quercetin as green inhibitor. Water Environ. J. 2019, 1–10. [Google Scholar] [CrossRef]
- Karar, A.; Henni, A.; Namoune, F.; Rosei, F. Inhibition of nucleation and crystal growth of calcium carbonate in hard waters using Paronychia arabica in an arid desert region. Water Environ. J. 2020, 1–9. [Google Scholar] [CrossRef]
- Hu, R.; Huang, T.; Wang, T.; Wang, H.; Long, X. Application of chemical crystallization circulating pellet fluidized beds for softening and saving circulating water in thermal power plants. Int. J. Environ. Res. Public Health 2019, 16, 4576. [Google Scholar] [CrossRef] [Green Version]
- Touir, R.; Dkhireche, N.; Ebn Touhami, M.; Lakhrissi, M.; Lakhrissi, B.; Sfaira, M. Corrosion and scale processes and their inhibition in simulated cooling water systems by monosaccharides derivatives. Part I: EIS study. Desalination 2009, 249, 922–928. [Google Scholar] [CrossRef]
- Gabrielli, C.; Maurin, G.; Francy-Chausson, H.; Thery, P.; Tran, T.T.M.; Tlili, M. Electrochemical water softening: Principle and application. Desalination 2006, 201, 150–163. [Google Scholar] [CrossRef]
- Jin, H.; Yu, Y.; Zhang, L.; Yan, R.; Chen, X. Polarity reversal electrochemical process for water softening. Sep. Purif. Technol. 2019, 210, 943–949. [Google Scholar] [CrossRef]
- Sanjuán, I.; Benavente, D.; Expósito, E.; Montiel, V. Electrochemical water softening: Influence of water composition on the precipitation behaviour. Sep. Purif. Technol. 2019, 211, 857–865. [Google Scholar] [CrossRef]
- Hasson, D.; Shemer, H.; Semiat, R. Removal of scale-forming ions by a novel cation-exchange electrochemical system—A review. Desalin. Water Treat. 2016, 57, 23147–23161. [Google Scholar] [CrossRef]
- Clauwaert, P.; De Paepe, J.; Jiang, F.; Alonso-Fariñas, B.; Vaiopoulou, E.; Verliefde, A.; Rabaey, K. Electrochemical tap water softening: A zero chemical input approach. Water Res. 2020, 169. [Google Scholar] [CrossRef]
- Yu, Y.; Jin, H.; Quan, X.; Hong, B.; Chen, X. Continuous Multistage Electrochemical Precipitation Reactor for Water Softening. Ind. Eng. Chem. Res. 2019, 58, 461–468. [Google Scholar] [CrossRef]
- Zhi, S.L.; Zhang, K.Q. Hardness removal by a novel electrochemical method. Desalination 2016, 381, 8–14. [Google Scholar] [CrossRef]
- Birnhack, L.; Keller, O.; Tang, S.C.N.; Fridman-Bishop, N.; Lahav, O. A membrane-based recycling process for minimizing environmental effects inflicted by ion-exchange softening applications. Sep. Purif. Technol. 2019, 223, 24–30. [Google Scholar] [CrossRef]
- Al-Jaser, Z.A.; Hamoda, M.F. Removal of nickel and vanadium from desalination brines by ion-exchange resins. Desalin. Water Treat. 2019, 157, 148–156. [Google Scholar] [CrossRef] [Green Version]
- Weber, W.J.J. Control de la Calidad del Agua Procesos Fisicoquimicos; Reverte: Barcelona, Spain, 1979; ISBN 978-84-291-7522-6. [Google Scholar]
- Bekri-Abbes, I.; Bayoudh, S.; Baklouti, M. The removal of hardness of water using sulfonated waste plastic. Desalination 2008, 222, 81–86. [Google Scholar] [CrossRef]
- Brastad, K.S.; He, Z. Water softening using microbial desalination cell technology. Desalination 2013, 309, 32–37. [Google Scholar] [CrossRef]
- Wist, W.; Lehr, J.H.; McEachern, R. Water Softening with Potassium Chloride: Process, Health, and Environmental Benefits; Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Li, C.W.; Jian, J.C.; Liao, J.C. Integrating membrane filtration and a fluidized-bed pellet reactor for hardness removal. J. Am. Water Work. Assoc. 2004, 96, 151–158. [Google Scholar] [CrossRef]
- Schaep, J.; Van Der Bruggen, B.; Uytterhoeven, S.; Croux, R.; Vandecasteele, C.; Wilms, D.; Van Houtte, E.; Vanlerberghe, F. Remvoal of hardness from groundwater by nanofiltration. Desalination 1998, 119, 295–301. [Google Scholar] [CrossRef]
- Izadpanah, A.A.; Javidnia, A. The ability of a nanofiltration membrane to remove hardness and ions from diluted seawater. Water 2012, 4, 283–294. [Google Scholar] [CrossRef]
- Ghizellaoui, S.; Chibani, A.; Ghizellaoui, S. Use of nanofiltration for partial softening of very hard water. Desalination 2005, 179, 315–322. [Google Scholar] [CrossRef]
- Kianfar, F.; Kianfar, E. Synthesis of Isophthalic Acid/Aluminum Nitrate Thin Film Nanocomposite Membrane for Hard Water Softening. J. Inorg. Organomet. Polym. Mater. 2019, 29, 2176–2185. [Google Scholar] [CrossRef]
- Kumara, K.M.S.; Kularathne, K.A.M.; Ranathunga, P.G.K.B.; Upeka, S.A.C.; Herath, D.C.; Weragoda, S.K. Hardness and Fluoride Removal Efficiency of Groundwater: Use of Physiochemical Methods in Water Purification at Anuradhapura District Dry Zone in Sri Lanka. In ICSBE 2018; Dissanayake, R., Mendis, P., Eds.; Springer: Singapore, 2020; pp. 171–182. [Google Scholar]
- Yang, Y.; Gutsol, A.; Fridman, A.; Cho, Y.I. Removal of CaCO3 scales on a filter membrane using plasma discharge in water. Int. J. Heat Mass Transf. 2009, 52, 4901–4906. [Google Scholar] [CrossRef]
- Muniz, G.L.; Duarte, F.V.; Rakocevic, M. Assessment and optimization of carbonated hard water softening with moringa oleifera seeds. Desalin. Water Treat. 2020, 173, 156–165. [Google Scholar] [CrossRef]
- Mohammadian, M.; Sahraei, R.; Ghaemy, M. Synthesis and fabrication of antibacterial hydrogel beads based on modified-gum tragacanth/poly(vinyl alcohol)/Ag0 highly efficient sorbent for hard water softening. Chemosphere 2019, 225, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Entezari, M.H.; Tahmasbi, M. Water softening by combination of ultrasound and ion exchange. Ultrason. Sonochem. 2009, 16, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Gruber, C.E.; Carda, D.D. Performance Analysis of Permanent Magnet Type Water Treatment Devices; Final Report Issued to the Water Quality Association; South Dakota School of Mines and Technology: Rapid City, SD, USA, 1981. [Google Scholar]
- Alomari, A.A. Effect of magnetic treatment on temporary hardness of groundwater. Asian J. Chem. 2019, 31, 1017–1021. [Google Scholar] [CrossRef]
- Rajczykowski, K.; Loska, K. Stimulation of Heavy Metal Adsorption Process by Using a Strong Magnetic Field. Water Air Soil Pollut. 2018, 229. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Wei, H.; Li, Z. Effect of magnetic field on the physical properties of water. Results Phys. 2018, 8, 262–267. [Google Scholar] [CrossRef]
- Han, X.; Peng, Y.; Ma, Z. Effect of magnetic field on optical features of water and KCl solutions. Optik 2016, 127, 6371–6376. [Google Scholar] [CrossRef]
- Latva, M.; Inkinen, J.; Rämö, J.; Kaunisto, T.; Mäkinen, R.; Ahonen, M.; Matilainen, J.; Pehkonen, S. Studies on the magnetic water treatment in new pilot scale drinking water system and in old existing real-life water system. J. Water Process Eng. 2016, 9, 215–224. [Google Scholar] [CrossRef]
- Mahmoud, B.; Yosra, M.; Nadia, A. Effects of magnetic treatment on scaling power of hard waters. Sep. Purif. Technol. 2016, 171, 88–92. [Google Scholar] [CrossRef]
- Silva, I.B.; Queiroz Neto, J.C.; Petri, D.F.S. The effect of magnetic field on ion hydration and sulfate scale formation. Colloids Surf. A Physicochem. Eng. Asp. 2015, 465, 175–183. [Google Scholar] [CrossRef]
- Liu, C.Z.; Lin, C.H.; Yeh, M.S.; Chao, Y.M.; Shen, P. Surface Modification and Planar Defects of Calcium Carbonates by Magnetic Water Treatment. Nanoscale Res. Lett. 2010, 5, 1982–1991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tai, C.Y.; Wu, C.K.; Chang, M.C. Effects of magnetic field on the crystallization of CaCO3 using permanent magnets. Chem. Eng. Sci. 2008, 63, 5606–5612. [Google Scholar] [CrossRef]
- Wang, Y.; Babchin, J.; Chernyi, L.T.; Chow, R.S.; Sawatzky, R.P. Rapid onset of calcium carbonate crystallization under the influence of a magnetic field. Water Res. 1997, 31, 346–350. [Google Scholar] [CrossRef]
- Kronenberg, K.J. Experimental evidence for effects of magnetic fields on moving water. IEEE Trans. Magn. 1985, 21, 2059–2061. [Google Scholar] [CrossRef]
- Martynova, O.I.; Gusev, B.T.; Leont’ev, E.A. Concerning the mechanism of the influence of a magnetic field of aqueous solutions of salts. Uspekhi Fiz. Nauk 1969, 98, 195–199. [Google Scholar] [CrossRef]
- Martinez, S.; Boluda, N.; García, J. Técnicas electromagnéticas para el tratamiento de aguas y diseño de una panta piloto con dispositivo TK3K. In Congreso Nacional del Agua 2019: Innovación y Sostenibilidad; Universitat d’Alacant: Alacant, Spain, 2019; pp. 1003–1014. ISBN 978-84-1302-034-1. [Google Scholar]
- Kozic, V.; Krope, J.; Lipus, L.C.; Ticar, I. Magnetic field analysis on electromagnetic water treatment device. Hung. J. Ind. Chem. 2006, 34, 51–54. [Google Scholar] [CrossRef]
- Rouina, M.; Kariminia, H.R.; Mousavi, S.A.; Shahryari, E. Effect of electromagnetic field on membrane fouling in reverse osmosis process. Desalination 2016, 395, 41–45. [Google Scholar] [CrossRef]
- Jiang, W.; Xu, X.; Lin, L.; Wang, H.; Shaw, R.; Lucero, D.; Xu, P. A pilot study of an electromagnetic field for control of reverse osmosis membrane fouling and scaling during brackish groundwater desalination. Water 2019, 11, 1015. [Google Scholar] [CrossRef] [Green Version]
- Hachicha, M.; Kahlaoui, B.; Khamassi, N.; Misle, E.; Jouzdan, O. Effect of electromagnetic treatment of saline water on soil and crops. J. Saudi Soc. Agric. Sci. 2018, 17, 154–162. [Google Scholar] [CrossRef]
- Piyadasa, C.; Yeager, T.R.; Gray, S.R.; Stewart, M.B.; Ridgway, H.F.; Pelekani, C.; Orbell, J.D. The influence of electromagnetic fields from two commercially available water-treatment devices on calcium carbonate precipitation. Environ. Sci. Water Res. Technol. 2017, 3, 566–572. [Google Scholar] [CrossRef]
- Piyadasa, C.; Yeager, T.R.; Gray, S.R.; Stewart, M.B.; Ridgway, H.F.; Pelekani, C.; Orbell, J.D. Antimicrobial effects of pulsed electromagnetic fields from commercially available water treatment devices–controlled studies under static and flow conditions. J. Chem. Technol. Biotechnol. 2018, 93, 871–877. [Google Scholar] [CrossRef] [Green Version]
- Gonzàlvez, N. Efecto del Electromagnetismo Sobre la Dureza, Bacterias y Algas en Agua; Repositorio institucional de la Universidad de Alicante: Alicante, Spain, 2017. [Google Scholar]
- Mercier, A.; Bertaux, J.; Lesobre, J.; Gravouil, K.; Verdon, J.; Imbert, C.; Valette, E.; Héchard, Y. Characterization of biofilm formation in natural water subjected to low-frequency electromagnetic fields. Biofouling 2016, 32, 287–299. [Google Scholar] [CrossRef]
- Zhao, J.-D.; Liu, Z.-A.; Zhao, E.-J. Combined effect of constant high voltage electrostatic fi eld and variable frequency pulsed electromagnetic fi eld on the morphology of calcium carbonate scale in circulating cooling water systems. Water Sci. Technol. 2014, 1074–1082. [Google Scholar] [CrossRef] [Green Version]
- Xuefei, M.; Lan, X.; Jiapeng, C.; Zikang, Y.; Wei, H. Experimental study on calcium carbonate precipitation using electromagnetic field treatment. Water Sci. Technol. 2013, 67, 2784–2790. [Google Scholar] [CrossRef] [PubMed]
- Stojiljkovic, D.T.; Mitić, N.C.; Šmelcerović, A.A.; Kalićanin, B.M.; Tasić-Kostov, M.Z.; Djurović-Petrovi, M.D. Effect of variable frequency electromagnetic field on deposit formation in installations with geothermal water in Sijarinjska spa, Serbia. Therm. Sci. 2011, 15, 643–648. [Google Scholar] [CrossRef]
- Catli, Y. Studying Electric Field Effect on Water by Analysing the Instrument “Hydronic”; University of Trento: Trento, Italy, 2009; Available online: http://www.hydroniconline.it/pdf/tesi di master.pdf (accessed on 20 August 2021).
- Shahryari, A.; Pakshir, M. Influence of a modulated electromagnetic field on fouling in a double-pipe heat exchanger. J. Mater. Process. Technol. 2007, 3, 389–395. [Google Scholar] [CrossRef]
- Xiaokai, B.X.; Chongfang, M.; Yongchang, C. Investigation on the Electromagnetic Anti-Fouling Technology for Scale Prevention. Chem. Eng. Technol. 2005, 1540–1545. [Google Scholar] [CrossRef]
- Botello-Zubiate, M.E.; Alvarez, A.; Martínez-Villafañe, A.; Almeraya-Calderon, F.; Matutes-Aquino, J.A. Influence of magnetic water treatment on the calcium carbonate phase formation and the electrochemical corrosion behavior of carbon steel. J. Alloy. Compd. 2004, 369, 256–259. [Google Scholar] [CrossRef]
- Donaldson, J.D. Scale prevention and descaling. Tube Int. 1988, 7, 39–49. [Google Scholar]
- Ellingsen, F.T.; Vik, E.A. A revue of scale formation with emphasis on magnetic water treatment. In Proceedings of the 14th World Congress of the International Water Supply Association, Zurich, Switzerland, 6–10 September 1982; pp. 12–25. [Google Scholar]
- Zavaleta, M.; Rivas, E.; Mendoza, A.; Tinoco, S. Efectodel Campo Magnético en el Tratamiento de Aguas Duras. Inst. Fis. Glew Wataghim 1998, 8, 3–8. [Google Scholar]
- Busch, K.W.; Busch, M.A. Laboratory studies on magnetic water treatment and their relationship to a possible mechanism for scale reduction. Desalination 1997, 109, 131–148. [Google Scholar] [CrossRef]
- Parsons, S.A.; Wang, B.-L.; Judd, S.J.; Stephenson, T. Magnetic treatment of calcium carbonate scale. Effect o pH control. Water Res. 1997, 31, 339–342. [Google Scholar] [CrossRef]
- Guo, B.; Han, H.B.; Chai, F. Influence of magnetic field on microstructural and dynamic properties of sodium, magnesium and calcium ions. Trans. Nonferrous Met. Soc. China 2011, 21 (Suppl. 2), s494–s498. [Google Scholar] [CrossRef]


| Hardness | mg CaCO3/L |
|---|---|
| Soft | 0–75 |
| Moderately hard | 75–150 |
| Hard | 150–300 |
| Very hard | >300 |
| Treatment | Advantages | Disadvantages |
|---|---|---|
| Chemical softening and inhibitors |
|
|
| Electrochemical |
|
|
| Cation exchange |
|
|
| Membrane |
|
|
| Reference | Magnetic Equipment | Intensity (T) | T (°C) | Solution | Volume (L) | Velocity (m/s) | Effects Studied and Results |
|---|---|---|---|---|---|---|---|
| Alomari, 2019 [66] | Neodymium magnets | 0.5 | - | Groundwater | 20 | - | Hardness and scale formation (39% reduction in temporary hardness, 2% in permanent hardness and 9% in total hardness) |
| Rajczykowski & Loska, 2018 [67] | Neodymium magnets N38 | 0.517 | - | Heavy metal solutions | 0.4 | - | Adsorption of heavy metals on activated carbon (The effectiveness of the process with respect to copper is increased by more than 40% and a summary molar removal was increased by 11%) |
| Wang et al., 2018 [68] | MWD-1 magnetizing equipment | 0.28 | 25–70 | Tap water | - | 0.8 | Specific heat (Treatment can decrease it), evaporation (Increases by 39%), and boiling point (Reduction of around 2 °C) |
| Han et al., 2016 [69] | Permanent magnets | 0.08–0.18 | 20 | Purified water and KCl solutions | - | - | Properties of water and KCl solutions |
| Latva et al., 2016 [70] | Bauer Water Ltd.PJ-20iHS | 0.026 | 21.1–21.5 | Tap water | 60 | - | Calcium carbonate precipitation (Reduction by 15%) and corrosion of pipes (Corrosion rates of 1–50 mils/year were obtained using chemical inhibitors, with corrosion rates of 0.0 mils/year obtained for the magnetically treated water) |
| Mahmoud et al., 2016 [71] | Permanent magnets GMX 8000 | 0.8 | 27.3–27.9 | Hard water | 1 | - | Calcium carbonate precipitation with a rapid controlled precipitation test (Antiscaling efficiency was approximately 45%) |
| Silva et al., 2015 [72] | Permanent magnets | 1 | 24 | Ba, Sr, Ca, Ag and Na solutions | 0.05 | 0.992 | Control of sulfate scales (The particle size distributions obtained for the treated solutions are in a smaller range than the untreated ones) |
| Liu et al., 2010 [73] | Own equipment | 0.01–0.04 | 20 | Synthetic water | 60 | 0.5 | Effect on the structure of calcite and aragonite (Changes in morphology are observed after application of the treatment) |
| Tai et al., 2008 [74] | MagneGen 1000 and 100 | 0.007–0.102 | 25 | Na2CO3 and CaCl2 | - | 0.047 | Calcite and aragonite growth rates (Reductions of up to 37% were observed in the growth rate of calcite. When treating for 24 h before nucleation, more than 50% aragonite appeared. When treating for 48 h, the calcite disappeared, and the aragonite was the predominant form) |
| Coey and Cass, 2000 [14] | Ferrite ring magnets and Nd-Fe-B magnets | 0.1 | - | Groundwater and mineral water | 0.5–1 | 0.04–1.2 | Structure of calcium carbonate and persistence of the treatment (A lower amount of Aragonite (7%) is observed in the untreated samples and 54% in the treated samples. A memory effect of more than 200 h was observed) |
| Wang et al., 1997 [75] | Neodymium-iron-boron magnets | 0.2 | 25–30 | Na2CO3 and CaCl2 | 0.005 | - | Nucleation of a supersaturated solution (The application of the treatment generates a faster precipitation, with smaller and more irregular crystals) |
| Kronenberg, 1985 [76] | Ferrite type ring magnets | 0.1 | - | Tap water | 2 × 10−5–1 × 10−4 | - | Crystal morphology and formation of precipitates (An increase in crystallization nuclei and a reduction in crystals attached to the substrate are observed) |
| Martynova et al., 1969 [77] | Undetermined | 0.9 | - | CaSO4, CaSiO3, BaSO4, Ca(HCO3)2 solutions | - | 0.1 | Scale formation (The dimensions of the crystals decrease, and their number increases as the intensity of the magnetic field increases) |
| Reference | Electromagnetic Equipment | Frequency (kHz) | T (°C) | Solution | Volume (L) | Flow (L/h) | Velocity (m/s) | Effects Studied |
|---|---|---|---|---|---|---|---|---|
| Gimeno Amorós et al., 2019 [9] | TK3K equipment | 1 | 21–29 | Tap and synthetic water | 4 | 600 | - | Calcium carbonate precipitation and structure (The evaporation of water with temperature generates almost 100% aragonite in the samples. The Meigen test is useful for the identification of crystalline forms, but it has many interferents) |
| Jiang et al., 2019 [81] | HydroFLOW S38 and HS48 | 150 | 21–37 | Groundwater | - | 1362 | Scale in reverse osmosis membranes and effect on the permeate (The decreasing rate of the normalized water permeability with treatment improved by 38.3% and 14.3% compared to permeate flow without treatment) | |
| Hachicha et al., 2018 [82] | Aqua-4D system | 1–10 | - | Saline water | 0.005 | - | - | Effect of saline water on seed germination of corn and potato crop (The treatment of saline irrigation water increased the germination rate of corn seeds, absorption of the nutrients (K, N and P) by the potatoes and decreased the ion toxicity for the crop by decreasing the Na+ contents in the plants) |
| Piyadasa et al., 2017 [83] | Pulsed electromagnetic field (PEMF) devices | 100 | 27–40 | Na2CO3 and CaCl2 | 0.002–0.01 | - | - | Calcium carbonate precipitation and structure (Effects were observed on the CaCO3 precipitation profile and on the characteristics of the precipitate, but only with one of the two equipment analysed) |
| Piyadasa et al., 2018 [84] | Pulsed electromagnetic field (PEMF) devices | 100 | 25–27 | Water with bacteria | 2 | 5.52–27.6 | - | Effects on microorganisms (Negative and positive adaptive responses are observed in the growth rate after the application of the different equipment. These responses are highly dependent on the interaction of numerous factors and parameters) |
| Verdú, 2016 and Gonzàlvez, 2017 [29,85] | Antical RF equipment | 20–200 | 25–75 | Tap and pond water | 50 | 1000–500 | - | Calcium carbonate precipitation and effect on microorganisms (The Meigen test is useful for the identification of crystalline forms. Colony reduction percentages of up to 67% were observed in Escherichia coli and up to 78% in Enterococcus faecalis) |
| Mercier et al., 2016 [86] | Aqua-4D system | 1–10 | 20–25 | River water | 0.05 | 25 | - | Formation of biofilms (Reductions of up to 39.9% in maximum biofilm thickness were observed with treatment after 45 days) |
| Rouina et al., 2016 [80] | Copper solenoid with a flow of alternating electrical current | 0.05 | 28–35 | Calcium carbonate solution | 25 | - | 1.8–4.8 | Membrane performance, fouling and permeate (The rejection increased by 5.3% after the application of the electromagnetic fields. In the presence of the treatment, the permeate flow rate increased by 30% compared to the case in which no treatment was applied) |
| Zhao et al., 2014 [87] | Magnetoelectric coordinated water processor | 0.05–0.3 | 30–90 | Cooling water | - | - | 0.334–0.669 | Calcium carbonate precipitation and structure (The application of electromagnetic fields under favourable conditions can have a significant anti-scaling effect with 68.95% of anti-scaling ratio. It was also observed that the appearance and growth of aragonite is favoured and its transition to calcite is slowed) |
| Xuefei et al., 2013 [88] | Electromagnetic signal generator | 66–400 | 25 | Synthetic hard water | 40 | - | 0.8 | Cooling water scale (Conductivityit was reduced by 17% with treatment, compared to 31% for the untreated case. A smaller particle size was also observed with treatment, as well as looser structures, unlike without treatment, the crystalline structures were more compact) |
| Stojiljkovic et al., 2011 [89] | Solenoid around a pipe. | 0.2–4 | 60 | Geothermal water | - | 54 | - | Scale formation (A decrease in deposit of up to 78 times was achieved using a variable frequency electromagnetic field) |
| Catli, 2009 [90] | Hydronic equipment | 0.5–6 | 80 | Company’s water trial system | - | - | - | Modification of scale and effect on the water molecule (A modification of the crystalline phase is observed after the application of the treatment, favouring the formation of aragonite. The field strongly “polarizes” water, lining up the charges in the molecules) |
| Shahryari and Pakshir, 2007 [91] | MEF generating coil. | 1–5 | 30 | Cooling water | - | - | 0.5–1.3 | Effect on fouling of a heat exchanger (Ca2+ decreases in the water of 23%, 11.5% and 7% were observed at water velocities of 0.5, 0.8 and 1.3 ms−1, respectively) |
| Xiaokai et al., 2005 [92] | Electromagnetic anti-fouling device | 6–22 | - | Na2CO3 and CaCl2 | 50 | - | - | Crystal nucleation and fouling (The resistance to fouling obtained with the treatment was between 50–72% lower than that observed without treatment. It was also observed that the growth of the particles was promoted by a coagulation process and not by a nucleation growth.) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez Moya, S.; Boluda Botella, N. Review of Techniques to Reduce and Prevent Carbonate Scale. Prospecting in Water Treatment by Magnetism and Electromagnetism. Water 2021, 13, 2365. https://doi.org/10.3390/w13172365
Martínez Moya S, Boluda Botella N. Review of Techniques to Reduce and Prevent Carbonate Scale. Prospecting in Water Treatment by Magnetism and Electromagnetism. Water. 2021; 13(17):2365. https://doi.org/10.3390/w13172365
Chicago/Turabian StyleMartínez Moya, Sergio, and Nuria Boluda Botella. 2021. "Review of Techniques to Reduce and Prevent Carbonate Scale. Prospecting in Water Treatment by Magnetism and Electromagnetism" Water 13, no. 17: 2365. https://doi.org/10.3390/w13172365
APA StyleMartínez Moya, S., & Boluda Botella, N. (2021). Review of Techniques to Reduce and Prevent Carbonate Scale. Prospecting in Water Treatment by Magnetism and Electromagnetism. Water, 13(17), 2365. https://doi.org/10.3390/w13172365