Assessment of Arsenic Levels in Water, Sediment, and Human Hair around Ie Seu’um Geothermal Manifestation Area, Aceh, Indonesia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Tools and Materials
2.2. Sampling Technique
2.2.1. Water Sampling
2.2.2. Sediment Samplings
2.2.3. Hair Sampling
2.3. Sample Preparation
2.3.1. Sediment Sample Preparation
2.3.2. Hair Sample Preparation
2.4. Arsenic Analysis with GF-AAS
2.5. Method Validation
2.6. Calculation of the Standard Deviation of Concentration (Sc)
3. Result and Discussion
3.1. Water Sample Characteristics
3.2. Method Validation
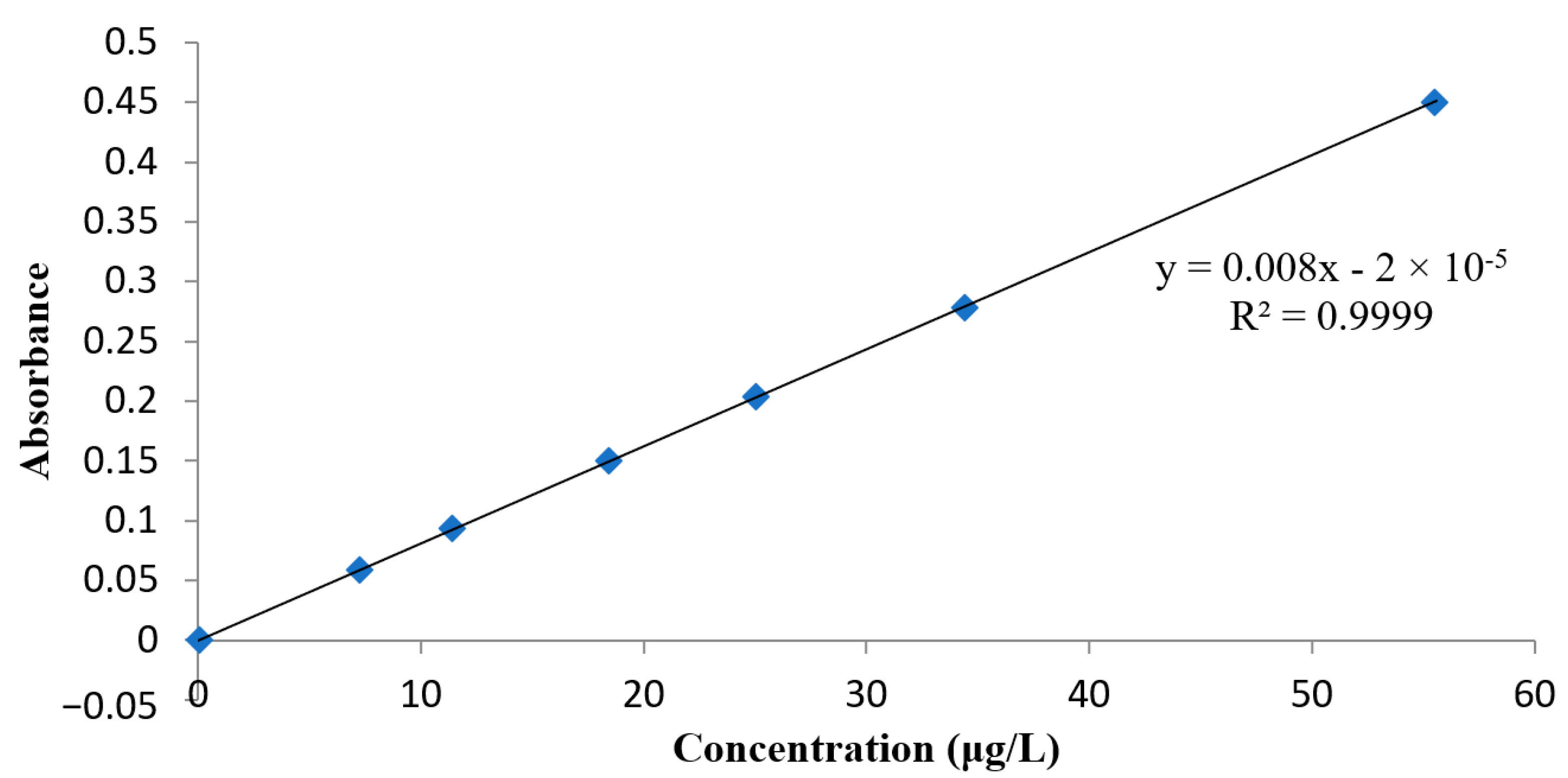
3.2.1. Calibration Curves and LoD/LoQ
3.2.2. Precision and %Recovery
3.3. Arsenic Analysis in Water Samples
3.4. Arsenic Analysis in Sediment Samples
3.5. Arsenic Analysis in Hair Samples
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, X.; Hu, Q. Development of Geothermal Resources in China: A Review. J. Earth Sci. 2018, 29, 452–467. [Google Scholar] [CrossRef]
- Idroes, R.; Yusuf, M.; Saiful, S.; Alatas, M.; Subhan, S.; Lala, A.; Muslem, M.; Suhendra, R.; Idroes, G.M.; Marwan, M.; et al. Geochemistry Exploration and Geothermometry Application in the North Zone of Seulawah Agam, Aceh Besar District, Indonesia. Energies 2019, 12, 4442. [Google Scholar] [CrossRef] [Green Version]
- Idroes, R.; Yusuf, M.; Alatas, M.; Lala, A.; Suhendra, R.; Idroes, G.M. Marwan Geochemistry of Sulphate spring in the Ie Jue geothermal areas at Aceh Besar district, Indonesia. In Proceedings of the Annual International Conferences, Banda Aceh, Indonesia, 12 September 2018; IOP Conference Series: Materials Science and Engineering. IOP Publishing: Bristol, UK, 2019; Volume 523, p. 012012. [Google Scholar] [CrossRef]
- Suhartono, E.; Noor, Z.; Edyson Budianto, W.Y.; Idroes, R. Effect of chronic lead exposure on bone using ATR-FTIR spectroscopy. In Proceeding of the International Conference on Bioinformatics and Nano-Medicine from Natural Resources for Biomedical Research, Malang, Indonesia, 21 November 2018; AIP Publishing: Melville, NY, USA, 2019; Volume 2108, p. 020025. [Google Scholar]
- Anawar, H.M.; Garcia-Sanchez, A.; Santa Regina, I. Evaluation of various chemical extraction methods to estimate plant-available arsenic in mine soils. Chemosphere 2008, 70, 1459–1467. [Google Scholar] [CrossRef] [PubMed]
- Hung, D.Q.; Nekrassova, O.; Compton, R.G. Analytical methods for inorganic arsenic in water: A review. Talanta 2004, 64, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Santra, B.K. Arsenic Contamination of Groundwater in West Bengal: Awareness for Health and Social Problems. Int. J. Appl. Sci. Eng. 2017, 5, 43. [Google Scholar] [CrossRef]
- ATSDR. Toxicological Profile for Arsenic. Departement of Health and Human Services; ATSDR CDC: Atlanta, GA, USA, 2007. [Google Scholar]
- Petrucci, R.H. Kimia Dasar; Terjemahan; Erlangga: Jakarta, Indonesia, 1982. [Google Scholar]
- Vogliardi, S.; Tucci, M.; Stocchero, G.; Ferrara, S.D.; Favretto, D. Sample preparation methods for determination of drugs of abuse in hair samples: A review. Anal. Chim. Acta 2015, 857, 1–27. [Google Scholar] [CrossRef]
- Guo, Q.; Planer-Friedrich, B.; Liu, M.; Yan, K.; Wu, G. Magmatic fluid input explaining the geochemical anomaly of very high arsenic in some southern Tibetan geothermal waters. Chem. Geol. 2019, 513, 32–43. [Google Scholar] [CrossRef]
- Herath, I.; Vithanage, M.; Bundschuh, J.; Maity, J.P.; Bhattacharya, P. Natural Arsenic in Global Groundwaters: Distribution and Geochemical Triggers for Mobilization. Curr. Pollut. Rep. 2016, 2, 68–89. [Google Scholar] [CrossRef] [Green Version]
- Birkle, P.; Bundschuh, J.; Sracek, O. Mechanisms of arsenic enrichment in geothermal and petroleum reservoirs fluids in Mexico. Water Res. 2010, 44, 5605–5617. [Google Scholar] [CrossRef] [PubMed]
- Yüksel, B.; Mergen, G.; Soylemezoglu, T. Assessment of Arsenic Levels in Human Hair by Hydride Generation Atomic Absorption Spectrometry: A Toxicological Application. At. Spectrosc.-Norwalk Connect. 2010, 31, 1–5. [Google Scholar]
- Yüksel, B.; Kayaalti, Z.; Soylemezoglu, T.; TURKSOY, V.; Tutkun, E. GFAAS Determination of Arsenic Levels in Biological Samples of Workers Occupationally Exposed to Metals: An Application in Analytical Toxicology. At. Spectrosc. 2015, 36, 171–176. [Google Scholar] [CrossRef]
- SNI 6989.8; Air Dan Limbah—Bagian 8: Cara Uji Timbal (Pb) Secara Spektrofotometri Serapan Atom (SSA); SNI: Jakarta, Indonesia, 2009.
- US EPA Operating Procedure. Sediment Sampling; EPA: Washington, DC, USA, 2010. [Google Scholar]
- SNI 6992.8; Sedimen—Bagian 8: Cara uji Seng (Zn) Secara Destruksi Asam Dengan Spektrofotometer Serapan Atom (SSA); SNI: Jakarta, Indonesia, 2017.
- SNI 4866; Cara Uji Cemaran Arsen dalam Makanan. Penerbit Badan Standarisasi Nasional; SNI: Jakarta, Indonesia, 1998.
- US EPA Method 7010; Graphite Furnace Atomic Absorption Spectrophotometry; EPA: Washington, DC, USA, 2007.
- Skoog, D.A. Fundamentals of Analytical Chemistry; Thomson: Hampshire, UK, 2004. [Google Scholar]
- Christian, G.D.; Dasgupta, P.K.; Schug, K.A. Analytical Chemistry; Wiley: Hoboken, NJ, USA, 2013. [Google Scholar]
- Menkes, R. Peraturan Menteri Kesehatan Republik Indonesia Nomor 492 Tahun 2010 Tentang Persyaratan Kualitas Air Minum; KEMENKES RI: Jakarta, Indonesia, 2010. [Google Scholar]
- Maity, J.P.; Ho, P.-R.; Huang, Y.-H.; Sun, A.-C.; Chen, C.-C.; Chen, C.-Y. The removal of arsenic from arsenic-bearing groundwater in In-situ and Ex-situ environment using novel natural magnetic rock material and synthesized magnetic material as adsorbent: A comparative assessment. Environ. Pollut. 2019, 253, 768–778. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, W. AOAC guidelines for single laboratory validation of chemical methods for dietary supplements and botanicals. Gaithersburg MD USA AOAC Int. 2002, 1, 12–19. [Google Scholar]
- Menkes, R. Peraturan Menteri Kesehatan Republik IndonesiaNomor 32 Tahun 2017 Tentang Standar Baku Mutu Kesehatan Lingkungan dan Persyaratan Kesehatan Air untuk Keperluan Higiene Sanitasi, Kolam Renang, Solus per Aqua, dan Pemandian Umum; KEMENKES RI: Jakarta, Indonesia, 2017. [Google Scholar]
- WHO Guidelines for Drinking Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011.
- Salim, N.A.A.; Wood, A.K.; Yusof, A.M.; Hamzah, M.S.; Elias, M.S.; Rahman, S.A. A study of arsenic and chromium contamination in sediments of freshwater bodies. Fresenius Environ. Bull. 2009, 18, 1618–1623. [Google Scholar]
- Wu, B.; Chen, T. Changes in hair arsenic concentration in a population exposed to heavy pollution: Follow-up investigation in Chenzhou City, Hunan Province, Southern China. J. Environ. Sci. 2010, 22, 283–289. [Google Scholar] [CrossRef]
- Ali, M.; Tarafdar, S.A. Arsenic in drinking water and in scalp hair by EDXRF A major recent health hazard in Bangladesh. J. Radioanal. Nucl. Chem. 2003, 256, 297–305. [Google Scholar] [CrossRef]
- Mayo Clinic Staff Detection of Nanocute Arsenic Expouse in Hair Specimens. Available online: https://neurology.testcatalog.org/show/ASHA (accessed on 23 June 2020).
- Paul, B.K. Arsenic contamination awareness among the rural residents in Bangladesh. Soc. Sci. Med. 2004, 59, 1741–1755. [Google Scholar] [CrossRef]
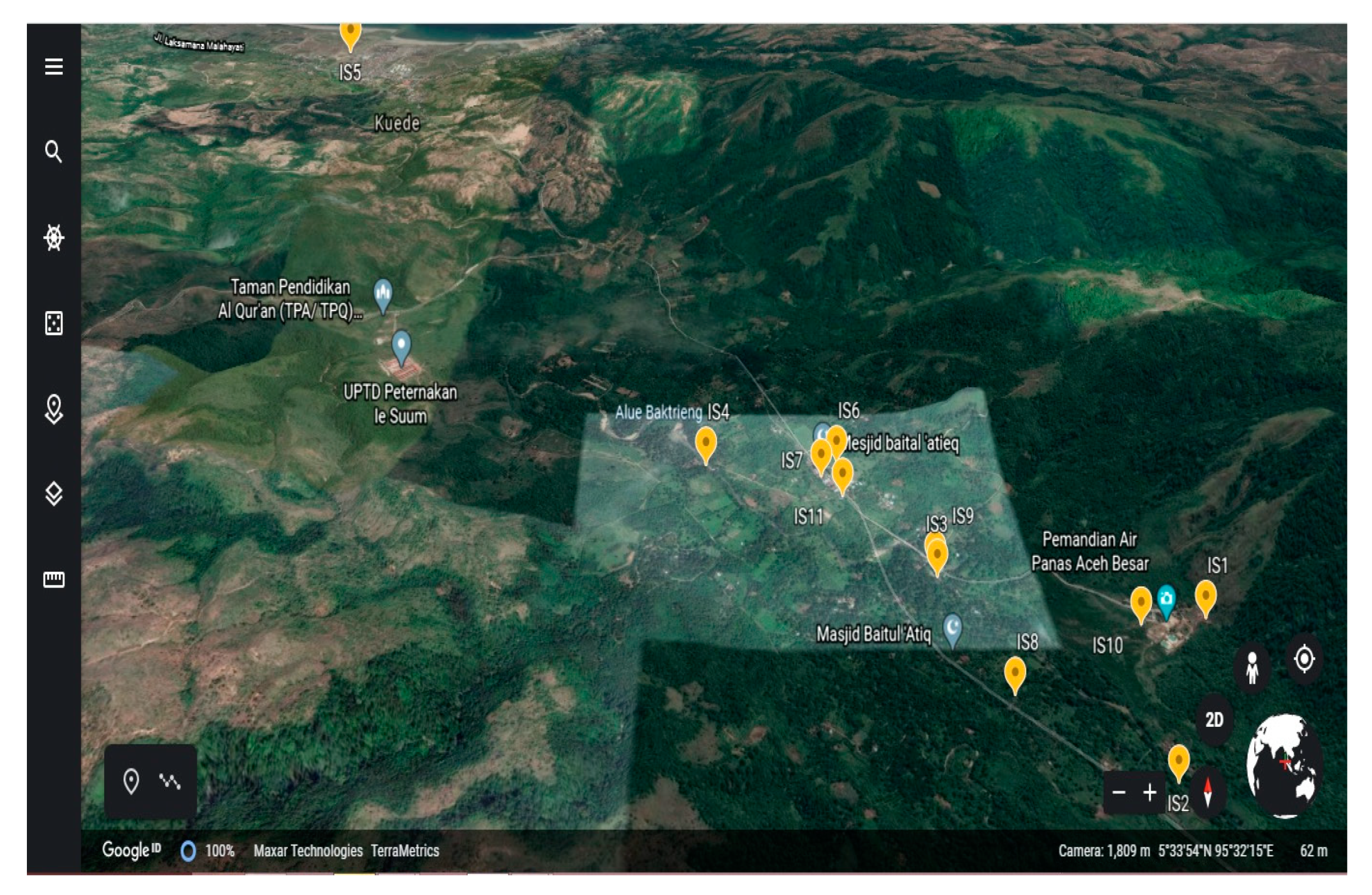
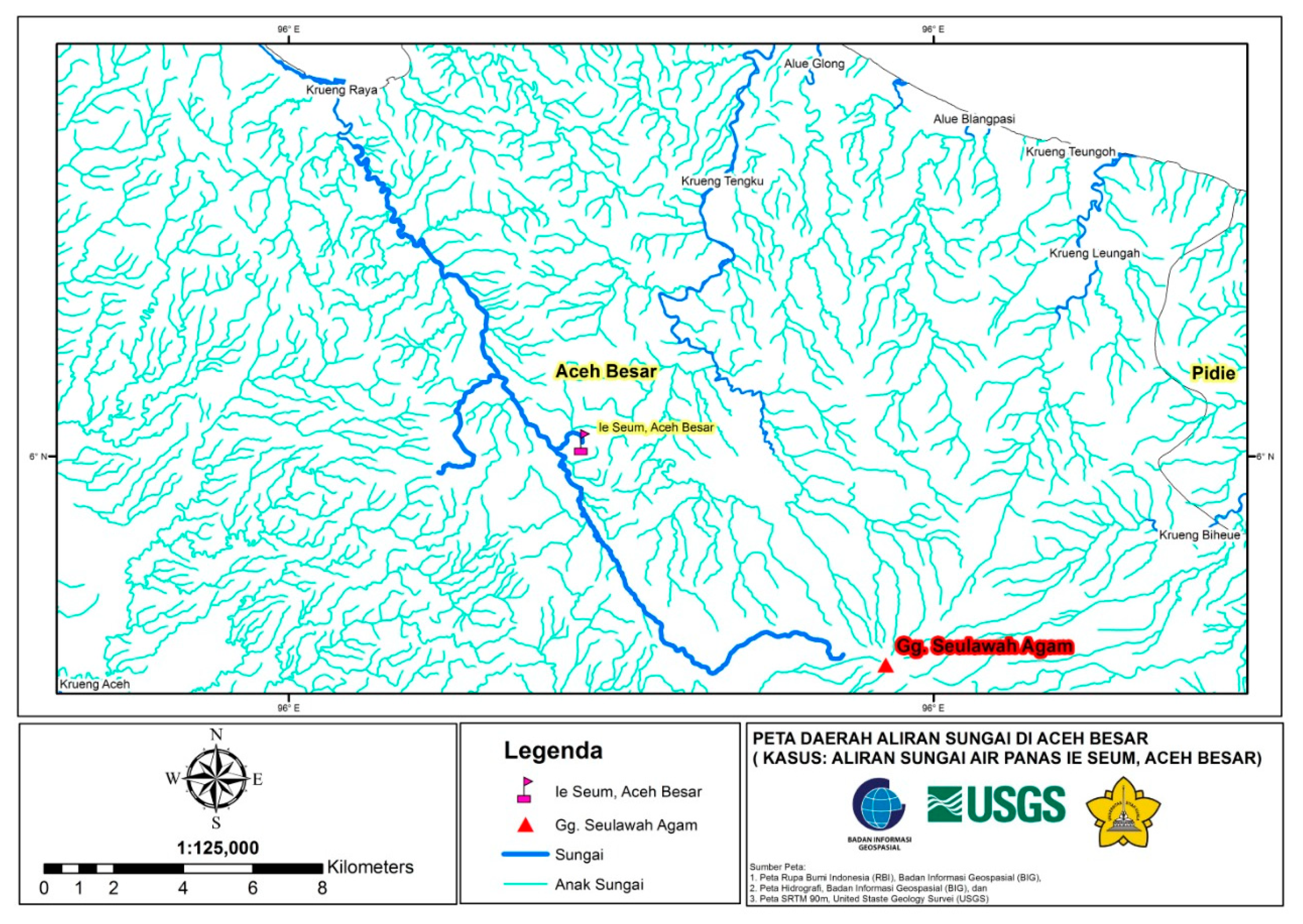
| Code | Locations | Coordinate |
|---|---|---|
| IS1 | Hot Spring | 5°32′49″ N; 95°32′55″ E |
| IS2 | River (Point 1) | 5°32′34″ N; 95°32′46″ E |
| IS3 | River (Point 2) | 5°32′55″ N; 95°32′31″ E |
| IS4 | River (Point 3) | 5°33′11″ N; 95°32′08″ E |
| IS5 | Estuary | 5°35′31″ N; 95°30′45″ E |
| IS6 | Borehole 1 | 5°33′11″ N; 95°32′23″ E |
| IS7 | Treatment Drinking Water | 5°33′09″ N; 95°32′21″ E |
| IS8 | Borehole 2 | 5°32′42″ N; 95°32′35″ E |
| IS9 | Borehole 3 | 5°32′56″ N; 95°32′31″ E |
| IS10 | Borehole 5 | 5°32′49″ N; 95°32′49″ E |
| IS11 | Borehole 4 | 5°33′06″ N; 95°32′23″ E |
| Name | Formula |
|---|---|
| Standard Deviation of Regression | |
| Standard Deviation of Slope | |
| Standard Deviation of Intercept | |
| Standard Deviation of the Concentration | |
| Limit of Detection Limit of Quantification |
| Code | pH ± SD | TDS ± SD (mg/L) | EC ± SD (μS/cm) | T ± SD (°C) | Salinity ± SD (‰) |
|---|---|---|---|---|---|
| IS1 | 6.65 ± 0.019 | 4534 ± 0.008 | 9686 ± 0.802 | 85.46 ± 0.330 | 17.8 ± 0.054 |
| IS2 | 7.57 ± 0.008 | 56.74 ± 0.559 | 117.6 ± 1.550 | 27.16 ± 0.015 | 0.12 ± 0.044 |
| IS3 | 7.58 ± 0.007 | 1614 ± 0.118 | 3892 ± 0.463 | 27.88 ± 0.028 | 0.94 ± 0.114 |
| IS4 | 7.64 ± 0.008 | 3232 ± 0.327 | 5086 ± 1.058 | 28.61 ± 0.017 | 0.58 ± 0.083 |
| IS5 | 7.52 ± 0.010 | 5564 ± 0.247 | 9536 ± 0.319 | 30.85 ± 0.030 | 19.08 ± 0.752 |
| IS6 | 7.49 ± 0.008 | 10.9 ± 1.466 | 26.66 ± 0.932 | 28.44 ± 0.011 | 0.14 ± 0.054 |
| IS7 | 6.15 ± 0.008 | 10.344 ± 0.307 | 24.94 ± 0.378 | 29.22 ± 0.016 | 0.12 ± 0.044 |
| IS8 | 6.66 ± 0.007 | 9.022 ± 0.813 | 13.52 ± 1.516 | 29.05 ± 0.020 | 0.12 ± 0.044 |
| IS9 | 6.63 ± 0.008 | 3.48 ± 0.080 | 5.946 ± 0.858 | 29.36 ± 0.012 | 0.16 ± 0.89 |
| IS10 | 6.55 ± 0.008 | 2270 ± 0.05 | 4526 ± 0.486 | 36.13 ± 0.008 | 8.46 ±0.320 |
| IS11 | 6.94 ± 0.011 | 117.94 ± 1.621 | 520 ± 1.224 | 29.88 ± 0.008 | 1.06 ± 0.054 |
| Name | Result |
|---|---|
| Slope | 0.00811667 |
| Intercept | –2.09278 × 10−5 |
| Standard Deviation of Slope (Sm) | 0.00000048 |
| Standard Deviation of Intercept (Sb) | 1.3402 × 10−5 |
| Coefficient of Determination (R2) | 0.99999998 |
| Standard Deviation of Regression (Sr) | 2.21382 × 10−5 |
| Sum of Square Regression | 0.13942563 |
| Sum of Square Residual | 2.45049 × 10−5 |
| %Recovery (Hair) | %Recovery (Sediment) | %RSD | ||
|---|---|---|---|---|
| Sample | Recovery (%) | Sample | Recovery (%) | |
| RS1 | 89.117 | SS1 | 105.175 | 1.067 |
| RS2 | 98.850 | SS2 | 89.815 | |
| RS3 | 101.027 | SS3 | 88.337 | |
| No. | Code | Concentration (μg/L) ± Sc |
|---|---|---|
| 1. | IS1 | 166.73 ± 0.008 |
| 2. | IS2 | 0.80 ± 0.003 |
| 3. | IS3 | 136.80 ± 0.007 |
| 4. | IS4 | 100.04 ± 0.006 |
| 5. | IS5 | 60.66 ± 0.004 |
| 6. | IS6 | 106.82 ± 0.006 |
| 7. | IS7 | 9.53 ± 0.004 |
| 8. | IS8 | 12.45 ± 0.004 |
| 9. | IS9 | 127.31 ± 0.007 |
| 10. | IS10 | 146.71 ± 0.007 |
| 11. | IS11 | 87.60 ± 0.005 |
| No | Code | Content ± Sc (mg/kg) |
|---|---|---|
| 1. | IS2 | 2.562 ± 0.003 |
| 2. | IS3 | 6.865 ± 0.005 |
| 3. | IS4 | 5.281 ± 0.004 |
| 4. | IS5 | 5.953 ± 0.004 |
| No. | Code | Age | Content ± Sc (mg/kg) |
|---|---|---|---|
| 1. | A1 | 27 | 54.59 ± 0.003 |
| 2. | A2 | 43 | 128.07 ± 0.005 |
| 3. | A3 | 23 | 115.44 ± 0.005 |
| 4. | A4 | 43 | 127.15 ± 0.005 |
| 5. | A5 | 22 | 98.69 ± 0.004 |
| 6. | A6 | 27 | n.d. |
| 7. | A7 | 20 | 66.47 ± 0.004 |
| 8. | A8 | 49 | 164.57 ± 0.006 |
| 9. | A9 | 22 | 110.13 ± 0.005 |
| 10. | A10 | 27 | 116.32 ± 0.005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irnawati, I.; Idroes, R.; Zulfiani, U.; Akmal, M.; Suhartono, E.; Idroes, G.M.; Muslem, M.; Lala, A.; Yusuf, M.; Saiful, S.; et al. Assessment of Arsenic Levels in Water, Sediment, and Human Hair around Ie Seu’um Geothermal Manifestation Area, Aceh, Indonesia. Water 2021, 13, 2343. https://doi.org/10.3390/w13172343
Irnawati I, Idroes R, Zulfiani U, Akmal M, Suhartono E, Idroes GM, Muslem M, Lala A, Yusuf M, Saiful S, et al. Assessment of Arsenic Levels in Water, Sediment, and Human Hair around Ie Seu’um Geothermal Manifestation Area, Aceh, Indonesia. Water. 2021; 13(17):2343. https://doi.org/10.3390/w13172343
Chicago/Turabian StyleIrnawati, Irnawati, Rinaldi Idroes, Utari Zulfiani, Muslim Akmal, Eko Suhartono, Ghazi Mauer Idroes, Muslem Muslem, Andi Lala, Muhammad Yusuf, Saiful Saiful, and et al. 2021. "Assessment of Arsenic Levels in Water, Sediment, and Human Hair around Ie Seu’um Geothermal Manifestation Area, Aceh, Indonesia" Water 13, no. 17: 2343. https://doi.org/10.3390/w13172343
APA StyleIrnawati, I., Idroes, R., Zulfiani, U., Akmal, M., Suhartono, E., Idroes, G. M., Muslem, M., Lala, A., Yusuf, M., Saiful, S., Suhud, K., Safitri, E., & Jalil, Z. (2021). Assessment of Arsenic Levels in Water, Sediment, and Human Hair around Ie Seu’um Geothermal Manifestation Area, Aceh, Indonesia. Water, 13(17), 2343. https://doi.org/10.3390/w13172343








