Response of Merlot Grapevine to Drought Is Associated to Adjustments of Growth and Nonstructural Carbohydrates Allocation in above and Underground Organs
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Modulated Drought Stress Treatment
2.2. Plant Water Status and Gas Exchange Monitoring
2.3. Morphometric and Biomass Measurements
2.4. Measurements of Non-Structural Carbohydrates
2.5. Statistical Analysis
3. Results
3.1. Water Plant Status and Use Efficiency
3.2. Biomass and Morphometric Traits in Different Organs in Response to Modulated Drought Stress
3.2.1. Morphometric Parameters in Leaf
3.2.2. Dry Matter Content and Morphometric Parameters in Shoot
3.2.3. Dry Matter Content and Morphometric Parameters in Root
3.2.4. Root-to-Shoot Ratio
3.3. Non-Structural Carbohydrate Content in Different Organs in Response to Modulated Drought Stress
3.3.1. NSC Content in Leaf Petiole
3.3.2. NSC Content in Shoot
3.3.3. NSC Content in Root
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cramer, W.; Guiot, J.; Fader, M.; Garrabou, J.; Gattuso, J.-P.; Iglesias, A.; Lange, M.A.; Lionello, P.; Llasat, M.C.; Paz, S.; et al. Climate Change and Interconnected Risks to Sustainable Development in the Mediterranean. Nat. Clim. Chang. 2018, 8, 972–980. [Google Scholar] [CrossRef]
- Santos, J.A.; Fraga, H.; Malheiro, A.C.; Moutinho-Pereira, J.; Dinis, L.-T.; Correia, C.; Moriondo, M.; Leolini, L.; Dibari, C.; Costafreda-Aumedes, S.; et al. A Review of the Potential Climate Change Impacts and Adaptation Options for European Viticulture. Appl. Sci. 2020, 10, 3092. [Google Scholar] [CrossRef]
- Masson-Delmotte, V.; Zhai, P.; Pörtner, H.-O.; Roberts, D.; Skea, J.; Shukla, P.R.; Pirani, A.; Moufouma-Okia, W.; Péan, C.; Pidcock, R.; et al. IPCC 2018: Summary for Policymakers. In Global Warming of 1.5 °C. An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty; Available online: https://www.ipcc.ch/sr15/ (accessed on 10 July 2021).
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant Drought Stress: Effects, Mechanisms and Management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef]
- Poni, S.; Gatti, M.; Palliotti, A.; Dai, Z.; Duchêne, E.; Truong, T.-T.; Ferrara, G.; Matarrese, A.M.S.; Gallotta, A.; Bellincontro, A.; et al. Grapevine Quality: A Multiple Choice Issue. Sci. Hortic. 2018, 234, 445–462. [Google Scholar] [CrossRef]
- Sorek, Y.; Greenstein, S.; Netzer, Y.; Shtein, I.; Jansen, S.; Hochberg, U. An Increase in Xylem Embolism Resistance of Grapevine Leaves during the Growing Season Is Coordinated with Stomatal Regulation, Turgor Loss Point and Intervessel Pit Membranes. New Phytol. 2021, 229, 1955–1969. [Google Scholar] [CrossRef]
- Perret, S.R.; Payen, S. Irrigation and the Environmental Tragedy: Pathways towards Sustainability in Agricultural Water Use. Irrig. Drain. 2020, 69, 263–271. [Google Scholar] [CrossRef]
- Tardieu, F. Variability among Species of Stomatal Control under Fluctuating Soil Water Status and Evaporative Demand: Modelling Isohydric and Anisohydric Behaviours. J. Exp. Bot. 1998, 49, 419–432. [Google Scholar] [CrossRef]
- Schultz, H.R. Differences in Hydraulic Architecture Account for Near-Isohydric and Anisohydric Behaviour of Two Field-Grown Vitis vinifera L. Cultivars during Drought: Hydraulic Conductance and Stomatal Behaviour. Plant Cell Environ. 2003, 26, 1393–1405. [Google Scholar] [CrossRef]
- Hochberg, U.; Rockwell, F.E.; Holbrook, N.M.; Cochard, H. Iso/Anisohydry: A Plant–Environment Interaction Rather Than a Simple Hydraulic Trait. Trends Plant Sci. 2018, 23, 112–120. [Google Scholar] [CrossRef]
- Villalobos-González, L.; Muñoz-Araya, M.; Franck, N.; Pastenes, C. Controversies in Midday Water Potential Regulation and Stomatal Behavior Might Result From the Environment, Genotype, and/or Rootstock: Evidence From Carménère and Syrah Grapevine Varieties. Front. Plant Sci. 2019, 10, 1522. [Google Scholar] [CrossRef]
- Gambetta, G.A.; Herrera, J.C.; Dayer, S.; Feng, Q.; Hochberg, U.; Castellarin, S.D. The Physiology of Drought Stress in Grapevine: Towards an Integrative Definition of Drought Tolerance. J. Exp. Bot. 2020, 71, 4658–4676. [Google Scholar] [CrossRef]
- Yadav, B.; Jogawat, A.; Rahman, M.S.; Narayan, O.P. Secondary Metabolites in the Drought Stress Tolerance of Crop Plants: A Review. Gene Rep. 2021, 23, 101040. [Google Scholar] [CrossRef]
- Degu, A.; Hochberg, U.; Wong, D.C.J.; Alberti, G.; Lazarovitch, N.; Peterlunger, E.; Castellarin, S.D.; Herrera, J.C.; Fait, A. Swift Metabolite Changes and Leaf Shedding Are Milestones in the Acclimation Process of Grapevine under Prolonged Water Stress. BMC Plant Biol. 2019, 19, 69. [Google Scholar] [CrossRef]
- Hochberg, U.; Degu, A.; Fait, A.; Rachmilevitch, S. Near Isohydric Grapevine Cultivar Displays Higher Photosynthetic Efficiency and Photorespiration Rates under Drought Stress as Compared with near Anisohydric Grapevine Cultivar. Physiol. Plant. 2013, 147, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Ledo, A.; Paul, K.I.; Burslem, D.F.R.P.; Ewel, J.J.; Barton, C.; Battaglia, M.; Brooksbank, K.; Carter, J.; Eid, T.H.; England, J.R.; et al. Tree Size and Climatic Water Deficit Control Root to Shoot Ratio in Individual Trees Globally. New Phytol. 2018, 217, 8–11. [Google Scholar] [CrossRef]
- Benjamin, J.G.; Nielsen, D.C.; Vigil, M.F.; Mikha, M.M.; Calderon, F. Water Deficit Stress Effects on Corn (Zea mays, L.) Root: Shoot Ratio. Open J. Soil Sci. 2014, 4, 151–160. [Google Scholar] [CrossRef]
- Anjum, S.A.; Ashraf, U.; Zohaib, A.; Tanveer, M.; Naeem, M.; Ali, I.; Tabassum, T.; Nazir, U. Growth and Developmental Responses of Crop Plants under Drought Stress: A Review. Zemdirb. Agric. 2017, 104, 267–276. [Google Scholar] [CrossRef]
- MacNeill, G.J.; Mehrpouyan, S.; Minow, M.A.A.; Patterson, J.A.; Tetlow, I.J.; Emes, M.J. Starch as a Source, Starch as a Sink: The Bifunctional Role of Starch in Carbon Allocation. J. Exp. Bot. 2017, 68, 4433–4453. [Google Scholar] [CrossRef]
- Dong, S.; Beckles, D.M. Dynamic Changes in the Starch-Sugar Interconversion within Plant Source and Sink Tissues Promote a Better Abiotic Stress Response. J. Plant Physiol. 2019, 234–235, 80–93. [Google Scholar] [CrossRef]
- Hochberg, U.; Bonel, A.G.; David-Schwartz, R.; Degu, A.; Fait, A.; Cochard, H.; Peterlunger, E.; Herrera, J.C. Grapevine Acclimation to Water Deficit: The Adjustment of Stomatal and Hydraulic Conductance Differs from Petiole Embolism Vulnerability. Planta 2017, 245, 1091–1104. [Google Scholar] [CrossRef] [PubMed]
- Fernández, M.D.; Hueso, J.J.; Cuevas, J. Water Stress Integral for Successful Modification of Flowering Dates in ‘Algerie’ Loquat. Irrig. Sci. 2010, 28, 127–134. [Google Scholar] [CrossRef]
- Gambetta, G.A.; Fei, J.; Rost, T.L.; Knipfer, T.; Matthews, M.A.; Shackel, K.A.; Walker, M.A.; McElrone, A.J. Water Uptake along the Length of Grapevine Fine Roots: Developmental Anatomy, Tissue-Specific Aquaporin Expression, and Pathways of Water Transport. Plant Physiol. 2013, 163, 1254–1265. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Harguindeguy, N.; Díaz, S.; Garnier, E.; Lavorel, S.; Poorter, H.; Jaureguiberry, P.; Bret-Harte, M.S.; Cornwell, W.K.; Craine, J.M.; Gurvich, D.E.; et al. New Handbook for Standardised Measurement of Plant Functional Traits Worldwide. Aust. J. Bot. 2013, 61, 167. [Google Scholar] [CrossRef]
- Falchi, R.; Petrussa, E.; Braidot, E.; Sivilotti, P.; Boscutti, F.; Vuerich, M.; Calligaro, C.; Filippi, A.; Herrera, J.C.; Sabbatini, P.; et al. Analysis of Non-Structural Carbohydrates and Xylem Anatomy of Leaf Petioles Offers New Insights in the Drought Response of Two Grapevine Cultivars. Int. J. Mol. Sci. 2020, 21, 1457. [Google Scholar] [CrossRef] [PubMed]
- Yemm, E.W.; Willis, A.J. The Estimation of Carbohydrates in Plant Extracts by Anthrone. Biochem. J. 1954, 57, 508–514. [Google Scholar] [CrossRef]
- Tomasella, M.; Petrussa, E.; Petruzzellis, F.; Nardini, A.; Casolo, V. The Possible Role of Non-Structural Carbohydrates in the Regulation of Tree Hydraulics. Int. J. Mol. Sci. 2019, 21, 144. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Fox, J.; Weisberg, S. An {R} Companion to Applied Regression; Sage: Thousand Oaks, CA, USA, 2019. [Google Scholar]
- Choné, X. Stem Water Potential Is a Sensitive Indicator of Grapevine Water Status. Ann. Bot. 2001, 87, 477–483. [Google Scholar] [CrossRef]
- Lovisolo, C.; Perrone, I.; Carra, A.; Ferrandino, A.; Flexas, J.; Medrano, H.; Schubert, A. Drought-Induced Changes in Development and Function of Grapevine (Vitis Spp.) Organs and in Their Hydraulic and Non-Hydraulic Interactions at the Whole-Plant Level: A Physiological and Molecular Update. Funct. Plant Biol. 2010, 37, 98. [Google Scholar] [CrossRef]
- Cochard, H.; Coll, L.; Le Roux, X.; Améglio, T. Unraveling the Effects of Plant Hydraulics on Stomatal Closure during Water Stress in Walnut. Plant Physiol. 2002, 128, 282–290. [Google Scholar] [CrossRef]
- Dayer, S.; Herrera, J.C.; Dai, Z.; Burlett, R.; Lamarque, L.J.; Delzon, S.; Bortolami, G.; Cochard, H.; Gambetta, G.A. The Sequence and Thresholds of Leaf Hydraulic Traits Underlying Grapevine Varietal Differences in Drought Tolerance. J. Exp. Bot. 2020, 71, 4333–4344. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.E.; Baeza, P. Relationships among Ambient Temperature and Vapor Pressure Deficit and Leaf and Stem Water Potentials of Fully Irrigated, Field-Grown Grapevines. Am. J. Enol. Vitic. 2007, 58, 173–181. [Google Scholar]
- Shellie, K.; Glenn, D.M. Wine Grape Response to Kaolin Particle Film Under Deficit and Well-Watered Conditions. Acta Hortic. 2008, 792, 587–591. [Google Scholar] [CrossRef]
- McDowell, N.; Pockman, W.T.; Allen, C.D.; Breshears, D.D.; Cobb, N.; Kolb, T.; Plaut, J.; Sperry, J.; West, A.; Williams, D.G.; et al. Mechanisms of Plant Survival and Mortality during Drought: Why Do Some Plants Survive While Others Succumb to Drought? New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef]
- Sade, N.; Gebremedhin, A.; Moshelion, M. Risk-Taking Plants: Anisohydric Behavior as a Stress-Resistance Trait. Plant Signal. Behav. 2012, 7, 767–770. [Google Scholar] [CrossRef]
- Sade, N.; Vinocur, B.J.; Diber, A.; Shatil, A.; Ronen, G.; Nissan, H.; Wallach, R.; Karchi, H.; Moshelion, M. Improving Plant Stress Tolerance and Yield Production: Is the Tonoplast Aquaporin SlTIP2;2 a Key to Isohydric to Anisohydric Conversion? New Phytol. 2009, 181, 651–661. [Google Scholar] [CrossRef]
- Gerzon, E.; Biton, I.; Yaniv, Y.; Zemach, H.; Netzer, Y.; Schwartz, A.; Fait, A.; Ben-Ari, G. Grapevine Anatomy as a Possible Determinant of Isohydric or Anisohydric Behavior. Am. J. Enol. Vitic. 2015, 66, 340–347. [Google Scholar] [CrossRef]
- Pellegrino, A.; Lebon, E.; Simonneau, T.; Wery, J. Towards a Simple Indicator of Water Stress in Grapevine (Vitis vinifera L.) Based on the Differential Sensitivities of Vegetative Growth Components. Aust. J. Grape Wine Res. 2005, 11, 306–315. [Google Scholar] [CrossRef]
- Cardoso, A.A.; Gori, A.; Da-Silva, C.J.; Brunetti, C. Abscisic Acid Biosynthesis and Signaling in Plants: Key Targets to Improve Water Use Efficiency and Drought Tolerance. Appl. Sci. 2020, 10, 6322. [Google Scholar] [CrossRef]
- Rosales, M.A.; Maurel, C.; Nacry, P. Abscisic Acid Coordinates Dose-Dependent Developmental and Hydraulic Responses of Roots to Water Deficit. Plant Physiol. 2019, 180, 2198–2211. [Google Scholar] [CrossRef]
- Brunner, I.; Herzog, C.; Dawes, M.A.; Arend, M.; Sperisen, C. How Tree Roots Respond to Drought. Front. Plant Sci. 2015, 6, 547. [Google Scholar] [CrossRef]
- Dry, P.R.; Loveys, B.R.; Düring, H. Partial Drying of the Rootzone of Grape. II. Changes in the Pattern of Root Development. Vitis 2000, 39, 4. [Google Scholar]
- Barrios-Masias, F.H.; Knipfer, T.; McElrone, A.J. Differential Responses of Grapevine Rootstocks to Water Stress Are Associated with Adjustments in Fine Root Hydraulic Physiology and Suberization. J. Exp. Bot. 2015, 66, 6069–6078. [Google Scholar] [CrossRef]
- Yıldırım, K.; Yağcı, A.; Sucu, S.; Tunç, S. Responses of Grapevine Rootstocks to Drought through Altered Root System Architecture and Root Transcriptomic Regulations. Plant Physiol. Biochem. 2018, 127, 256–268. [Google Scholar] [CrossRef]
- Moura, J.C.M.S.; Bonine, C.A.V.; De Oliveira Fernandes Viana, J.; Dornelas, M.C.; Mazzafera, P. Abiotic and Biotic Stresses and Changes in the Lignin Content and Composition in Plants. J. Integr. Plant Biol. 2010, 52, 360–376. [Google Scholar] [CrossRef]
- Steudle, E. Water uptake by plant roots: An integration of views. In Recent Advances of Plant Root Structure and Function; Gašparíková, O., Čiamporová, M., Mistrík, I., Baluška, F., Eds.; Springer: Dordrecht, The Netherlands, 2001; pp. 71–82. ISBN 978-90-481-5598-9. [Google Scholar]
- Vandeleur, R.K.; Mayo, G.; Shelden, M.C.; Gilliham, M.; Kaiser, B.N.; Tyerman, S.D. The Role of Plasma Membrane Intrinsic Protein Aquaporins in Water Transport through Roots: Diurnal and Drought Stress Responses Reveal Different Strategies between Isohydric and Anisohydric Cultivars of Grapevine. Plant Physiol. 2009, 149, 445–460. [Google Scholar] [CrossRef]
- Savi, T.; Bertuzzi, S.; Branca, S.; Tretiach, M.; Nardini, A. Drought-induced Xylem Cavitation and Hydraulic Deterioration: Risk Factors for Urban Trees under Climate Change? New Phytol. 2015, 205, 1106–1116. [Google Scholar] [CrossRef] [PubMed]
- Nardini, A.; Savi, T.; Losso, A.; Petit, G.; Pacilè, S.; Tromba, G.; Mayr, S.; Trifilò, P.; Lo Gullo, M.A.; Salleo, S. X-ray Microtomography Observations of Xylem Embolism in Stems of Laurus nobilis Are Consistent with Hydraulic Measurements of Percentage Loss of Conductance. New Phytol. 2017, 213, 1068–1075. [Google Scholar] [CrossRef] [PubMed]
- Eckert, C.; Sharmin, S.; Kogel, A.; Yu, D.; Kins, L.; Strijkstra, G.-J.; Polle, A. What Makes the Wood? Exploring the Molecular Mechanisms of Xylem Acclimation in Hardwoods to an Ever-Changing Environment. Forests 2019, 10, 358. [Google Scholar] [CrossRef]
- Falchi, R.; Petrussa, E.; Zancani, M.; Casolo, V.; Beraldo, P.; Nardini, A.; Sivilotti, P.; Calderan, A.; Herrera, J.C.; Peterlunger, E.; et al. Summer Drought Stress: Differential Effects on Cane Anatomy and Non-Structural Carbohydrate Content in Overwintering Cabernet Sauvignon and Syrah Vines. BIO Web Conf. 2019, 13, 03007. [Google Scholar] [CrossRef][Green Version]
- Jacobsen, A.L.; Rodriguez-Zaccaro, F.D.; Lee, T.F.; Valdovinos, J.; Toschi, H.S.; Martinez, J.A.; Pratt, R.B. Grapevine Xylem Development, Architecture, and Function. In Functional and Ecological Xylem Anatomy; Hacke, U., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 133–162. ISBN 978-3-319-15782-5. [Google Scholar]
- Munitz, S.; Netzer, Y.; Shtein, I.; Schwartz, A. Water Availability Dynamics Have Long-Term Effects on Mature Stem Structure in Vitis vinifera. Am. J. Bot. 2018, 105, 1443–1452. [Google Scholar] [CrossRef]
- Hochberg, U.; Degu, A.; Gendler, T.; Fait, A.; Rachmilevitch, S. The Variability in the Xylem Architecture of Grapevine Petiole and Its Contribution to Hydraulic Differences. Funct. Plant Biol. 2015, 42, 357. [Google Scholar] [CrossRef]
- Mitchell, P.J.; O’Grady, A.P.; Tissue, D.T.; White, D.A.; Ottenschlaeger, M.L.; Pinkard, E.A. Drought Response Strategies Define the Relative Contributions of Hydraulic Dysfunction and Carbohydrate Depletion during Tree Mortality. New Phytol. 2013, 197, 862–872. [Google Scholar] [CrossRef]
- Hochberg, U.; Albuquerque, C.; Rachmilevitch, S.; Cochard, H.; David-Schwartz, R.; Brodersen, C.R.; McElrone, A.; Windt, C.W. Grapevine Petioles Are More Sensitive to Drought Induced Embolism than Stems: Evidence from in Vivo MRI and Microcomputed Tomography Observations of Hydraulic Vulnerability Segmentation: Hydraulic Vulnerability Segmentation in Grapevine. Plant Cell Environ. 2016, 39, 1886–1894. [Google Scholar] [CrossRef]
- Petrussa, E.; Boscutti, F.; Vianello, A.; Casolo, V. ‘Last In-First Out’: Seasonal Variations of Non-Structural Carbohydrates, Glucose-6-Phosphate and ATP in Tubers of Two Arum Species. Plant Biol. 2018, 20, 346–356. [Google Scholar] [CrossRef]
- Chaves, M.M. Mechanisms Underlying Plant Resilience to Water Deficits: Prospects for Water-Saving Agriculture. J. Exp. Bot. 2004, 55, 2365–2384. [Google Scholar] [CrossRef]
- Gurrieri, L.; Merico, M.; Trost, P.; Forlani, G.; Sparla, F. Impact of Drought on Soluble Sugars and Free Proline Content in Selected Arabidopsis Mutants. Biology 2020, 9, 367. [Google Scholar] [CrossRef]
- Secchi, F.; Pagliarani, C.; Zwieniecki, M.A. The Functional Role of Xylem Parenchyma Cells and Aquaporins during Recovery from Severe Water Stress: Response of Xylem Parenchyma Cells to Embolism. Plant Cell Environ. 2017, 40, 858–871. [Google Scholar] [CrossRef] [PubMed]

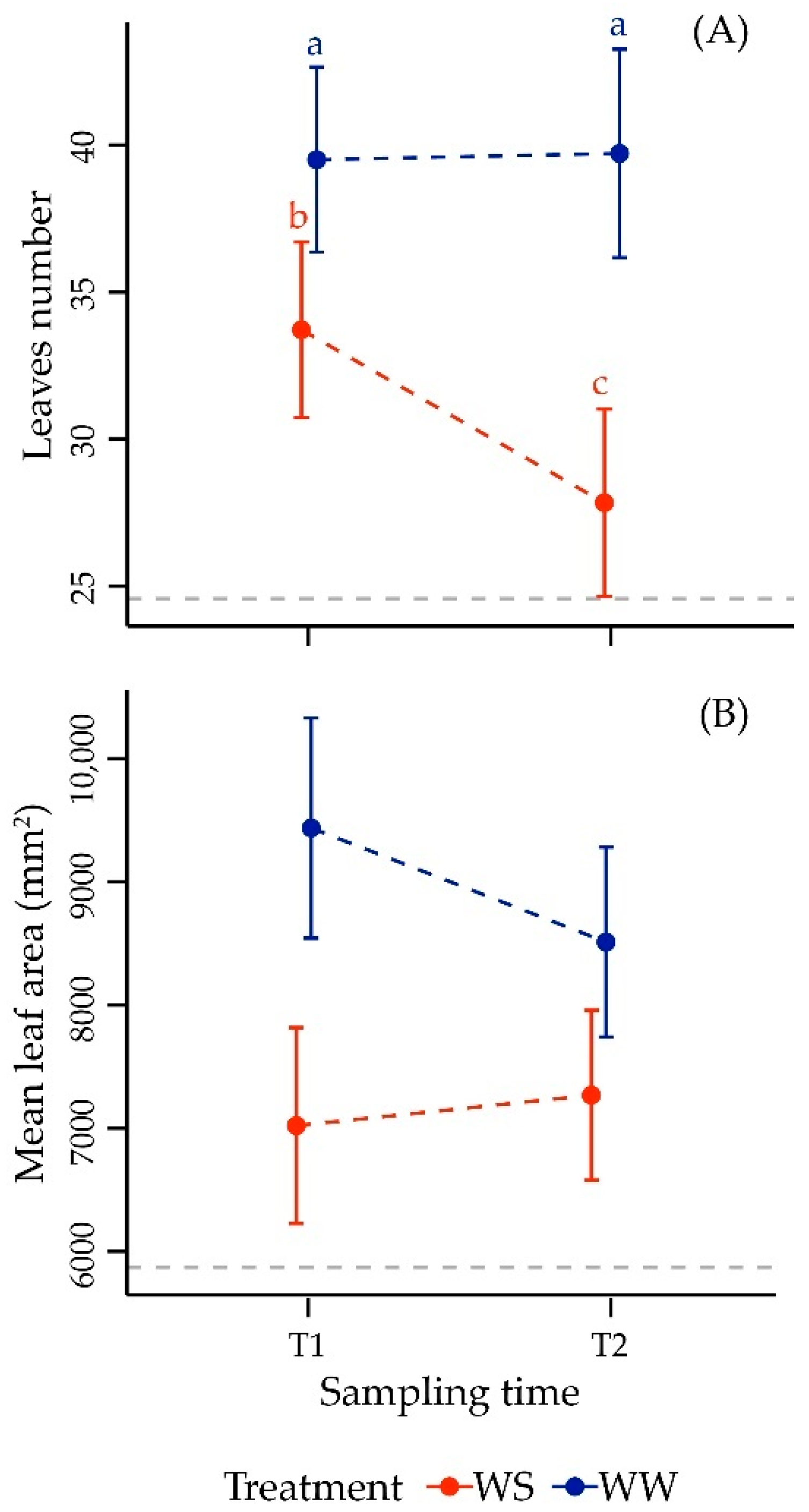
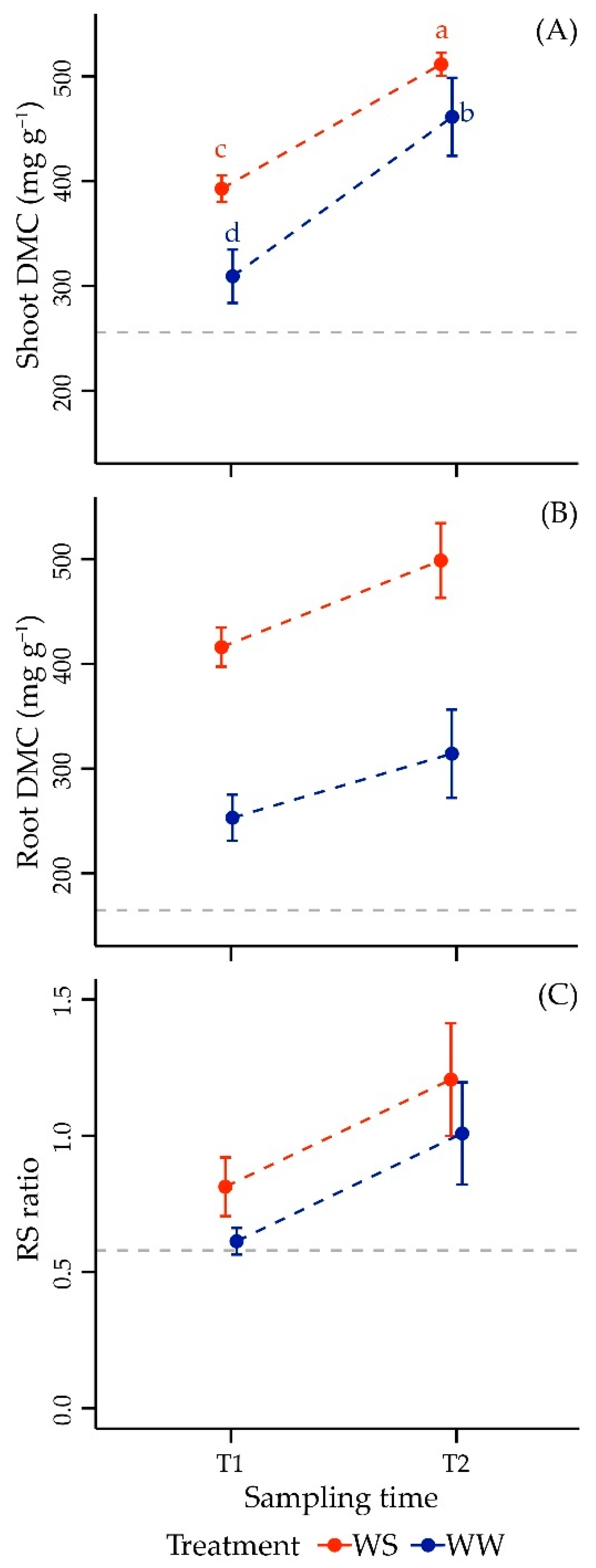
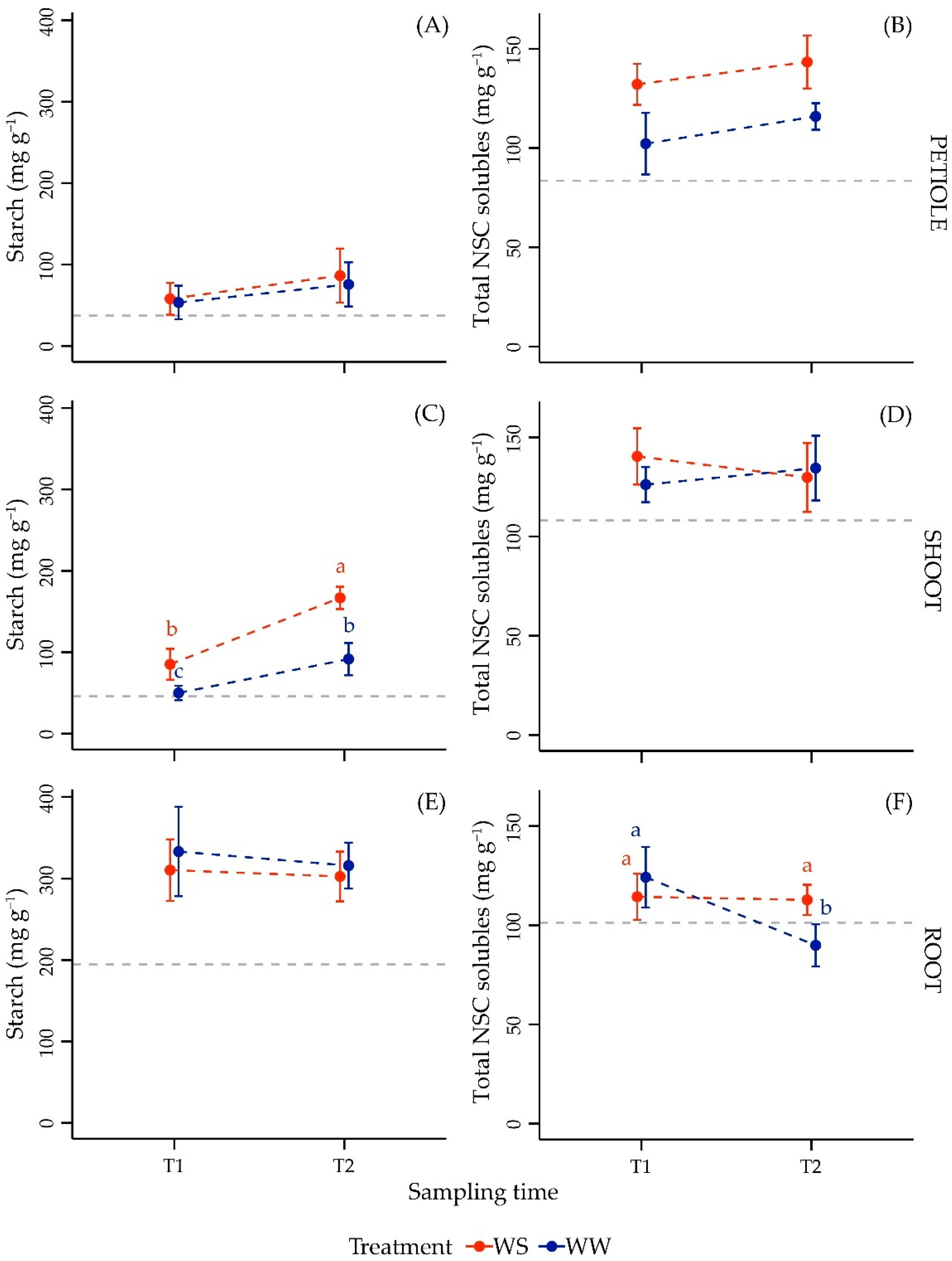
| Factor | Df | F Value | p-Value |
|---|---|---|---|
| Leaves number | |||
| Time | 1, 22 | 2.89 | 0.103 |
| Treatment | 1, 22 | 48.40 | <0.001 *** |
| Time × Treatment | 1, 22 | 5.76 | 0.025 * |
| Mean Leaf Area | |||
| Time | 1, 22 | 0.41 | 0.528 |
| Treatment | 1, 22 | 34.67 | <0.001 *** |
| Time × Treatment | 1, 22 | 3.56 | 0.072 |
| log(Shoot DMC) | |||
| Time | 1, 22 | 213.21 | <0.001 *** |
| Treatment | 1, 22 | 53.49 | <0.001 *** |
| Time × Treatment | 1, 22 | 8.17 | 0.009 ** |
| log(Root DMC) | |||
| Time | 1, 23 | 27.77 | <0.001 *** |
| Treatment | 1, 23 | 204.32 | <0.001 *** |
| Time × Treatment | 1, 23 | 0.22 | 0.639 |
| log(R/S ratio) | |||
| Time | 1, 24 | 60.35 | <0.001 *** |
| Treatment | 1, 24 | 16.60 | <0.001 *** |
| Time × Treatment | 1, 24 | 0.69 | 0.411 |
| Factor | Df | F Value | p-Value | |
|---|---|---|---|---|
| PETIOLE | log(Starch) | |||
| Time | 1, 17 | 6.14 | 0.024 * | |
| Treatment | 1, 17 | 0.47 | 0.504 | |
| Time × Treatment | 1, 17 | 0.02 | 0.883 | |
| PETIOLE | Total NSC solubles | |||
| Time | 1, 15 | 6.20 | 0.025 * | |
| Treatment | 1, 15 | 27.21 | <0.001 *** | |
| Time × Treatment | 1, 15 | 0.05 | 0.824 | |
| SHOOT | Starch | |||
| Time | 1, 24 | 104.48 | <0.001 *** | |
| Treatment | 1, 24 | 83.73 | <0.001 *** | |
| Time × Treatment | 1, 24 | 11.08 | 0.003 ** | |
| SHOOT | Total NSC solubles | |||
| Time | 1, 24 | 0.04 | 0.835 | |
| Treatment | 1, 24 | 0.75 | 0.395 | |
| Time × Treatment | 1, 24 | 2.96 | 0.098 | |
| ROOT | Starch | |||
| Time | 1, 21 | 0.41 | 0.528 | |
| Treatment | 1, 21 | 1.36 | 0.256 | |
| Time × Treatment | 1, 21 | 0.10 | 0.756 | |
| ROOT | Total NSC solubles | |||
| Time | 1, 23 | 15.91 | <0.001 *** | |
| Treatment | 1, 23 | 2.66 | 0.116 | |
| Time × Treatment | 1, 23 | 13.77 | 0.001 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vuerich, M.; Braidotti, R.; Sivilotti, P.; Alberti, G.; Casolo, V.; Braidot, E.; Boscutti, F.; Calderan, A.; Petrussa, E. Response of Merlot Grapevine to Drought Is Associated to Adjustments of Growth and Nonstructural Carbohydrates Allocation in above and Underground Organs. Water 2021, 13, 2336. https://doi.org/10.3390/w13172336
Vuerich M, Braidotti R, Sivilotti P, Alberti G, Casolo V, Braidot E, Boscutti F, Calderan A, Petrussa E. Response of Merlot Grapevine to Drought Is Associated to Adjustments of Growth and Nonstructural Carbohydrates Allocation in above and Underground Organs. Water. 2021; 13(17):2336. https://doi.org/10.3390/w13172336
Chicago/Turabian StyleVuerich, Marco, Riccardo Braidotti, Paolo Sivilotti, Giorgio Alberti, Valentino Casolo, Enrico Braidot, Francesco Boscutti, Alberto Calderan, and Elisa Petrussa. 2021. "Response of Merlot Grapevine to Drought Is Associated to Adjustments of Growth and Nonstructural Carbohydrates Allocation in above and Underground Organs" Water 13, no. 17: 2336. https://doi.org/10.3390/w13172336
APA StyleVuerich, M., Braidotti, R., Sivilotti, P., Alberti, G., Casolo, V., Braidot, E., Boscutti, F., Calderan, A., & Petrussa, E. (2021). Response of Merlot Grapevine to Drought Is Associated to Adjustments of Growth and Nonstructural Carbohydrates Allocation in above and Underground Organs. Water, 13(17), 2336. https://doi.org/10.3390/w13172336








