Insights into the Use of Phytoremediation Processes for the Removal of Organic Micropollutants from Water and Wastewater; A Review
Abstract
:1. Introduction
2. Main Phytoremediation Mechanisms
- -
- Phytostabilization—the plant immobilizes inorganic impurities in its roots and does not eliminate them with binding occurring mainly through the mechanism of complexation;
- -
- Phytoacumulation or phytoextraction—pollutants accumulate in different parts of the plant. This process, which uses the mechanism of hyperaccumulation, is very significant in respect to heavy metal removal;
- -
- Phytodegradation or phytotransformation—plants metabolize, using the mechanism of degradation within the plant, organic pollutants taken from water or soil into, usually, less toxic substances;
- -
- Phytovolatilization—plants absorb organic and inorganic pollutants and then eliminate them through the processes of transpiration;
- -
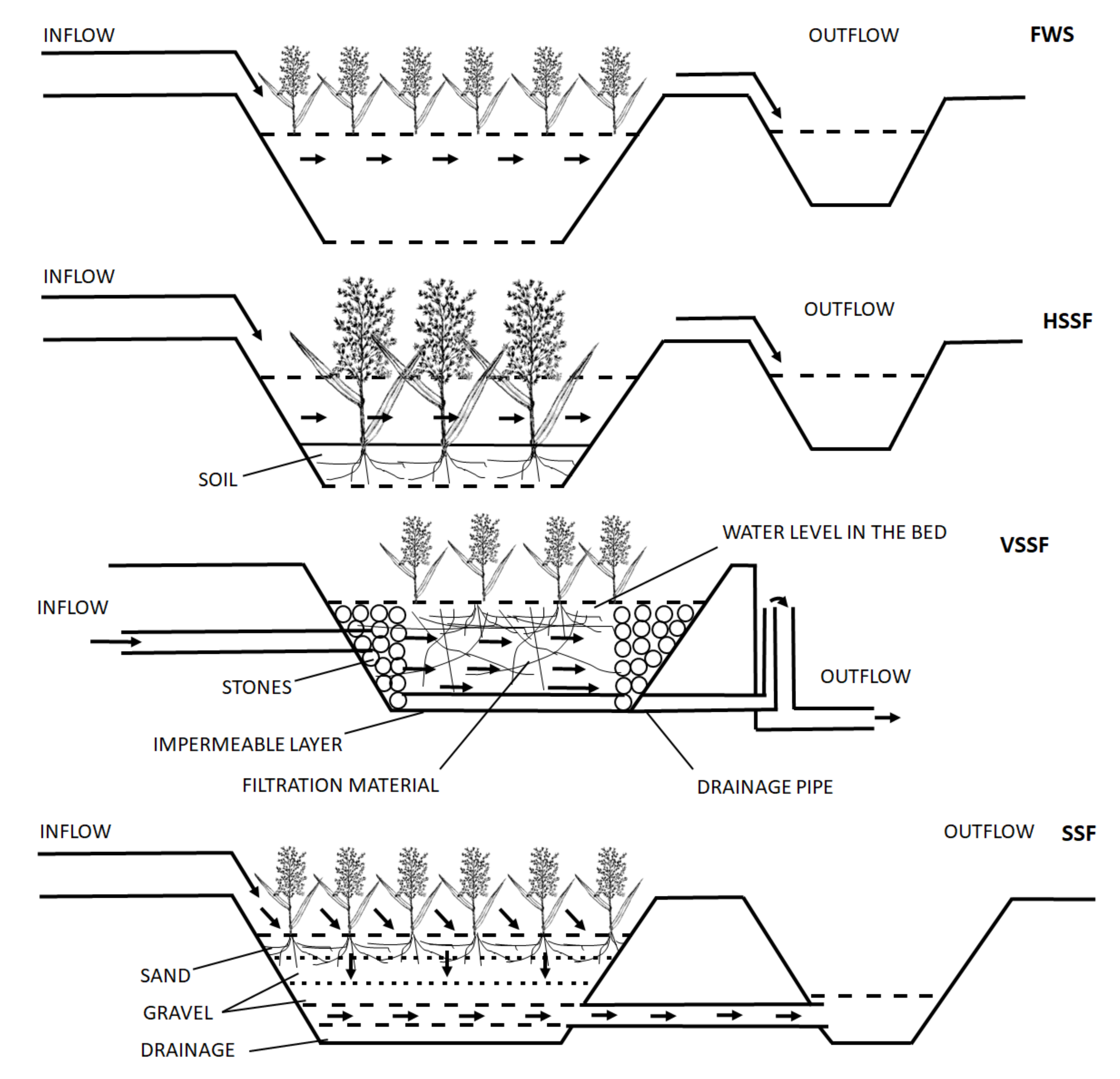
3. Characteristic of Phytoremediation Modules Used in Sewage Treatment Plants
- -
- Type of flow—within this category, it is possible to distinguish the following systems:
- -
- Type of plants used—this variable is very important, since some plants can accumulate pharmaceuticals while others may accumulate heavy metals;
- -
4. Plants Applied in the Remediation of Wastewater
- Surface macrophytes (helophytes)—grow on wetlands, develop above ground in boggy or fully submerged soil. Examples include Acorus calamus L., Carex rostrata Stokes, Phragmites australis, Schoenoplectus lacustris (L.) Palla, and Typha latifolia L.;
- Floating leaf macrophytes (rhizophytes)—their leaves float on or break the water’s, surface but they are rooted in soil. Examples include Nymphaea alba L. and Nuphar lutea;
- Submerged macrophytes—underwater plants that float in water but are rooted in soil. Examples include Myriophyllum spicatum, Ceratophyllum demersum L.;
- Freely floating macrophytes (pleustophytes)—plants that float on the water’s surface without being rooted in soil. Examples include Lemna minor L., Spirodela polyrhiza L., and Eichhornia crassipes [69].
5. Effectiveness of Micropollutant Removal by Plants
6. Toxic Effects of Micropollutants on Plants
7. Improving Overall Pollution Indicators
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Patel, M.; Kumar, R.; Kishor, K.; Mlsna, T.; Pittman, C.U.; Mohan, D. Pharmaceuticals of Emerging Concern in Aquatic Systems: Chemistry, Occurrence, Effects, and Removal Methods. Chem. Rev. 2019, 119, 3510–3673. [Google Scholar] [CrossRef] [Green Version]
- Jones, K.C. Persistent Organic Pollutants (POPs) and Related Chemicals in the Global Environment: Some Personal Reflections. Environ. Sci. Technol. 2021. [Google Scholar] [CrossRef]
- Richardson, S.D.; Kimura, S.Y. Water Analysis: Emerging Contaminants and Current Issues. Anal. Chem. 2019, 92, 473–505. [Google Scholar] [CrossRef]
- Kalogiouri, N.P.; Samanidou, V.F. Recent Advances in Miniaturized Microextraction Techniques for the Determination of Bisphenols in Environmental Samples: An Overview of the Last Two Decades. Curr. Anal. Chem. 2021, 17, 478–494. [Google Scholar] [CrossRef]
- Salgueiro-González, N.; Muniategui-Lorenzo, S.; López-Mahía, P.; Prada-Rodríguez, D. Trends in analytical methodologies for the determination of alkylphenols and bisphenol A in water samples. Anal. Chim. Acta 2017, 962, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Mansour, F.R.; Danielson, N.D. Solidification of floating organic droplet in dispersive liquid-liquid microextraction as a green analytical tool. Talanta 2017, 170, 22–35. [Google Scholar] [CrossRef]
- Sadutto, D.; Picó, Y. Sample Preparation to Determine Pharmaceutical and Personal Care Products in an All-Water Matrix: Solid Phase Extraction. Molecules 2020, 25, 5204. [Google Scholar] [CrossRef] [PubMed]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2: The Endocrine Society′s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, E1–E150. [Google Scholar] [CrossRef]
- Polianciuc, S.I.; Gurzău, A.E.; Kiss, B.; Ștefan, M.G.; Loghin, F. Antibiotics in the environment: Causes and consequences. Med. Pharm. Rep. 2020, 93, 231–240. [Google Scholar] [CrossRef]
- Marsala, R.Z.; Capri, E.; Russo, E.; Bisagni, M.; Colla, R.; Lucini, L.; Gallo, A.; Suciu, N. First evaluation of pesticides occurrence in groundwater of Tidone Valley, an area with intensive viticulture. Sci. Total. Environ. 2020, 736, 139730. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.-Q.; Liu, Y.-S.; Xiong, Q.; Cai, W.-W.; Ying, G.-G. Occurrence, toxicity and transformation of six typical benzotriazoles in the environment: A review. Sci. Total. Environ. 2019, 661, 407–421. [Google Scholar] [CrossRef]
- Kapelewska, J.; Kotowska, U.; Karpińska, J.; Kowalczuk, D.; Arciszewska, A.; Świrydo, A. Occurrence, removal, mass loading and environmental risk assessment of emerging organic contaminants in leachates, groundwaters and wastewaters. Microchem. J. 2018, 137, 292–301. [Google Scholar] [CrossRef]
- Petrie, B.; Barden, R.; Kasprzyk-Hordern, B. A review on emerging contaminants in wastewaters and the environment: Current knowledge, understudied areas and recommendations for future monitoring. Water Res. 2015, 72, 3–27. [Google Scholar] [CrossRef]
- Kanaujiya, D.K.; Paul, T.; Sinharoy, A.; Pakshirajan, K. Biological Treatment Processes for the Removal of Organic Micropollutants from Wastewater: A Review. Curr. Pollut. Rep. 2019, 5, 112–128. [Google Scholar] [CrossRef]
- Adeel, M.; Song, X.; Wang, Y.; Francis, D.; Yang, Y. Environmental impact of estrogens on human, animal and plant life: A critical review. Environ. Int. 2017, 99, 107–119. [Google Scholar] [CrossRef]
- Kim, S.; Choi, K. Occurrences, toxicities, and ecological risks of benzophenone-3, a common component of organic sunscreen products: A mini-review. Environ. Int. 2014, 70, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Swale, D.R.; Bloomquist, J.R. Is DEET a dangerous neurotoxicant? Pesticide Manag. Sci. 2019, 75, 2068–2070. [Google Scholar] [CrossRef]
- Prabhakara, A.; Nanjappa, D.P.; Babu, N.; Kalladka, K.; Chakraborty, A.; Chakraborty, G. Exposure to Mosquito Repellents Causes Profound Development Defects and Induces Oxidative Stress in Zebrafish. J. Heal. Allied Sci. NU 2020, 10, 122–127. [Google Scholar] [CrossRef]
- Godwin, A.D. Plasticizers. Appl. Plast. Eng. Handb. 2011, 487–501. [Google Scholar] [CrossRef]
- Bahnmüller, S.; Loi, C.H.; Linge, K.; von Gunten, U.; Canonica, S. Degradation rates of benzotriazoles and benzothiazoles under UV-C irradiation and the advanced oxidation process UV/H2O2. Water Res. 2015, 74, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Matthiessen, P.; Wheeler, J.; Weltje, L. A review of the evidence for endocrine disrupting effects of current-use chemicals on wildlife populations. Crit. Rev. Toxicol. 2017, 48, 195–216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pi, N.; Ng, J.; Kelly, B. Bioaccumulation of pharmaceutically active compounds and endocrine disrupting chemicals in aquatic macrophytes: Results of hydroponic experiments with Echinodorus horemanii and Eichhornia crassipes. Sci. Total. Environ. 2017, 601–602, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Geyer, H.J.; Rimkus, G.G.; Scheunert, I.; Kaune, A.; Schramm, K.-W.; Kettrup, A.; Zeeman, M.; Muir, D.C.G.; Hansen, L.G.; Mackay, D. Bioaccumulation and Occurrence of Endocrine-Disrupting Chemicals (EDCs), Persistent Organic Pollutants (POPs), and Other Organic Compounds in Fish and Other Organisms Including Human. In Bioaccumulation—New Aspects and Developments; The Handbook of Environmental Chemistry (Vol. 2 Series: Reactions and Processes); Beek, B., Ed.; Springer: Berlin, Heidelberg, 2000; Volume 2J. [Google Scholar]
- Diao, P.; Chen, Q.; Wang, R.; Sun, D.; Cai, Z.; Wu, H.; Duan, S. Phenolic endocrine-disrupting compounds in the Pearl River Estuary: Occurrence, bioaccumulation and risk assessment. Sci. Total. Environ. 2017, 584-585, 1100–1107. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, E.; Ladeira, C.; Viegas, S. EDCs Mixtures: A Stealthy Hazard for Human Health? Toxics 2017, 5, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wittassek, M.; Koch, H.M.; Angerer, J.; Brufcning, T. Assessing exposure to phthalates—The human biomonitoring approach. Mol. Nutr. Food Res. 2011, 55, 7–31. [Google Scholar] [CrossRef]
- Palanza, P.; Nagel, S.C.; Parmigiani, S.; Saal, F.S.V. Perinatal exposure to endocrine disruptors: Sex, timing and behavioral endpoints. Curr. Opin. Behav. Sci. 2016, 7, 69–75. [Google Scholar] [CrossRef] [Green Version]
- Langford, K.H.; Reid, M.J.; Fjeld, E.; Øxnevad, S.; Thomas, K.V. Environmental occurrence and risk of organic UV filters and stabilizers in multiple matrices in Norway. Environ. Int. 2015, 80, 1–7. [Google Scholar] [CrossRef]
- Net, S.; Dumoulin, D.; El-Osmani, R.; Rabodonirina, S.; Ouddane, B. Case study of PAHs, Me-PAHs, PCBs, phthalates and pesticides contamination in the Somme River water, France. Int. J. Environ. Res. 2014, 8, 1159–1170. [Google Scholar]
- Belfroid, A.; Van Velzen, M.; Van Der Horst, B.; Vethaak, D. Occurrence of bisphenol A in surface water and uptake in fish: Evaluation of field measurements. Chemosphere 2002, 49, 97–103. [Google Scholar] [CrossRef]
- Zhao, J.-L.; Ying, G.-G.; Wang, L.; Yang, J.-F.; Yang, X.-B.; Yang, L.-H.; Li, X. Determination of phenolic endocrine disrupting chemicals and acidic pharmaceuticals in surface water of the Pearl Rivers in South China by gas chromatography–negative chemical ionization–mass spectrometry. Sci. Total. Environ. 2009, 407, 962–974. [Google Scholar] [CrossRef]
- Kotowska, U.; Kapelewska, J.; Sturgulewska, J. Determination of phenols and pharmaceuticals in municipal wastewaters from Polish treatment plants by ultrasound-assisted emulsification–microextraction followed by GC–MS. Environ. Sci. Pollut. Res. 2013, 21, 660–673. [Google Scholar] [CrossRef] [Green Version]
- Baderna, D.; Maggioni, S.; Boriani, E.; Gemma, S.; Molteni, M.; Lombardo, A.; Colombo, A.; Bordonali, S.; Rotella, G.; Lodi, M.; et al. A combined approach to investigate the toxicity of an industrial landfill’s leachate: Chemical analyses, risk assessment and in vitro assays. Environ. Res. 2011, 111, 603–613. [Google Scholar] [CrossRef]
- Kapelewska, J.; Kotowska, U.; Wiśniewska, K. Determination of personal care products and hormones in leachate and groundwater from Polish MSW landfills by ultrasound-assisted emulsification microextraction and GC-MS. Environ. Sci. Pollut. Res. 2015, 23, 1642–1652. [Google Scholar] [CrossRef] [Green Version]
- Felis, E.; Kalka, J.; Sochacki, A.; Kowalska, K.; Bajkacz, S.; Harnisz, M.; Korzeniewska, E. Antimicrobial pharmaceuticals in the aquatic environment—Ocurrence and environmental implications. Eur. J. Pharmacol. 2020, 866, 172813. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.-C.; Chen, Y.Y.; Chiou, M.-R.; Chen, M.Y.; Fan, H.-J. Occurrence and treatment efficiency of pharmaceuticals in landfill leachates. Waste Manag. 2016, 55, 257–264. [Google Scholar] [CrossRef]
- Khan, N.A.; Khan, S.U.; Ahmed, S.; Farooqi, I.H.; Yousefi, M.; Mohammadi, A.A.; Changani, F. Recent trends in disposal and treatment technologies of emerging-pollutants—A critical review. TrAC Trends Anal. Chem. 2020, 122, 115744. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2018, 17, 145–155. [Google Scholar] [CrossRef]
- Weng, C.-H. Water pollution prevention and state of the art treatment technologies. Environ. Sci. Pollut. Res. 2020, 27, 34583–34585. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, P.; Giri, B.S.; Shukla, P.; Gupta, P. Recent advancement in remediation of synthetic organic antibiotics from environmental matrices: Challenges and perspective. Bioresour. Technol. 2021, 319, 124161. [Google Scholar] [CrossRef] [PubMed]
- Gmurek, M.; Olak-Kucharczyk, M.; Ledakowicz, S. Photochemical decomposition of endocrine disrupting compounds—A review. Chem. Eng. J. 2017, 310, 437–456. [Google Scholar] [CrossRef]
- Tominaga, F.K.; Silva, T.T.; Bolani, N.F.; Silva de Jesus, J.M.; Teixeira, A.C.S.C.; Borrely, S.I. Is ionizing radiation effective in removing pharmaceuticals from water? Environ. Sci. Pollut. Res. 2021, 28, 23975–23983. [Google Scholar] [CrossRef]
- Boczkaj, G.; Fernandes, A. Wastewater treatment by means of advanced oxidation processes at basic pH conditions: A review. Chem. Eng. J. 2017, 320, 608–633. [Google Scholar] [CrossRef]
- Pandey, N.; Chandra, J.; Xalxo, R.; Sahu, K. Concept and Types of Phytoremediat. In Approaches to the Remediation of Inorganic Pollutants, Hasanuzzaman, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2021; pp. 281–302. [Google Scholar] [CrossRef]
- Gatidou, G.; Oursouzidou, M.; Stefanatou, A.; Stasinakis, A.S. Removal mechanisms of benzotriazoles in duckweed Lemna minor wastewater treatment systems. Sci. Total. Environ. 2017, 596–597, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Gersberg, R.M.; Ng, W.J.; Tan, S.K. Removal of pharmaceuticals and personal care products in aquatic plant-based systems: A review. Environ. Pollut. 2014, 184, 620–639. [Google Scholar] [CrossRef] [PubMed]
- Justin, M.Z.; Pajk, N.; Zupanc, V.; Zupančič, M. Phytoremediation of landfill leachate and compost wastewater by irrigation of populous and Salix: Biomass and growth response. Waste Manag. 2010, 30, 1032–1042. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H. An overview of phytoremediation as a potentially promising technology for environmental pollution control. Biotechnol. Bioproc. Eng. 2013, 18, 431–439. [Google Scholar] [CrossRef]
- Chaudhary, E.; Sharma, P. Duckweed as eco-friendly tool for phytoremediation. IJSR 2014, 3, 1615–1617. [Google Scholar]
- Adhikari, U.; Harrigan, T.; Reinhold, D.M. Use of duckweed-based constructed wetlands for nutrient recovery and pollutant reduction from dairy wastewater. Ecol. Eng. 2015, 78, 6–14. [Google Scholar] [CrossRef]
- Fujita, M.; Mori, K.; Kodera, T. Nutrient removal and starch production through cultivation of Wolffia arrhiza. J. Biosci. Bioeng. 1999, 87, 194–198. [Google Scholar] [CrossRef]
- De Vasconcelos, V.M.; de Morais, E.R.C.; Faustino, S.J.B.; Hernandez, M.C.R.; Gaudêncio, H.R.S.C.; de Melo, R.R.; Bessa Junior, A.P. Floating aquatic macrophytes for the treatment of aquaculture effluents. Environ. Sci. Pollut. Res. 2020, 28(3), 2600–2607. [Google Scholar] [CrossRef]
- Ekperusi, A.O.; Sikoki, F.D.; Nwachukwu, E.O. Application of common duckweed (Lemna minor) in phytoremediation of chem-icals in the environment: State and future perspective. Chemosphere 2019, 223, 285–309. [Google Scholar] [CrossRef]
- Ansari, A.A.; Naeem, M.; Gill, S.S.; AlZuaibr, F.M. Phytoremediation of contaminated waters: An eco-friendly technology based on aquatic macrophytes application. Egypt. J. Aquat. Res. 2020, 46, 371–376. [Google Scholar] [CrossRef]
- Mustafa, H.M.; Hayder, G. Recent studies on applications of aquatic weed plants in phytoremediation of wastewater: A review article. Ain Shams Eng. J. 2021, 12, 355–365. [Google Scholar] [CrossRef]
- Gatliff, E.; Linton, P.J.; Riddle, D.J.; Thomas, P.R. Phytoremediation of soil and groundwater: Economic benefits over traditional methodologies. In Bioremediation and Bioeconomy; Prasad, M.N., Ed.; Elsevier: London, UK, 2016; pp. 589–608. [Google Scholar]
- Dhir, B.; Sharmila, P.; Saradhi, P.P. Potential of Aquatic Macrophytes for Removing Contaminants from the Environment. Crit. Rev. Environ. Sci. Technol. 2009, 39, 754–781. [Google Scholar] [CrossRef]
- Reichenauer, T.G.; Germida, J. Phytoremediation of Organic Contaminants in Soil and Groundwater. ChemSusChem 2008, 1, 708–717. [Google Scholar] [CrossRef]
- Carter, L.; Harris, E.; Williams, M.J.M.; Ryan, J.J.; Kookana, R.; Boxall, A.B.A. Fate and Uptake of Pharmaceuticals in Soil–Plant Systems. J. Agric. Food Chem. 2014, 62, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Q.; Gersberg, R.M.; Hua, T.; Zhu, J.; Goyal, M.K.; Ng, W.J.; Tan, S.K. Fate of pharmaceutical compounds in hydroponic mesocosms planted with Scirpus validus. Environ. Pollut. 2013, 181, 98–106. [Google Scholar] [CrossRef]
- Fatima, K.; Imran, A.; Naveed, M.; Afzal, M. Plant-bacteria synergism: An innovative approach for the remediation of crude oil-contaminated soils. Soil Environ. 2017, 36, 93–113. [Google Scholar] [CrossRef]
- Deng, Z.; Cao, L. Fungal endophytes and their interactions with plants in phytoremediation: A review. Chemosphere 2017, 168, 1100–1106. [Google Scholar] [CrossRef]
- Vymazal, J. Constructed Wetlands for Wastewater Treatment. Water 2010, 2, 530–549. [Google Scholar] [CrossRef] [Green Version]
- Constructed Wetlands Manual; UN-HABITAT: Nairobi, Kenya, 2008.
- Vymazal, J. Emergent plants used in free water surface constructed wetlands: A review. Ecol. Eng. 2013, 61, 582–592. [Google Scholar] [CrossRef]
- Zhang, D.; Gersberg, R.M.; Keat, T.S. Constructed wetlands in China. Ecol. Eng. 2009, 35, 1367–1378. [Google Scholar] [CrossRef]
- Verlicchi, P.; Zambello, E. How efficient are constructed wetlands in removing pharmaceuticals from untreated and treated urban wastewaters? A review. Sci. Total. Environ. 2014, 470–471, 1281–1306. [Google Scholar] [CrossRef]
- Sasmaz, M.; Obek, E.; Sasmaz, A. Bioaccumulation of Uranium and Thorium by Lemna minor and Lemna gibba in Pb-Zn-Ag Tailing Water. Bull. Environ. Contam. Toxicol. 2016, 97, 832–837. [Google Scholar] [CrossRef]
- Vymazal, J. Plants used in constructed wetlands with horizontal subsurface flow: A review. Hydrobiology 2011, 674, 133–156. [Google Scholar] [CrossRef]
- Mkandawire, M.; Dudel, E. Are Lemna spp. effective phytoremediation agents. Bioremed. Biodivers. Bioavailab. 2007, 1, 56–71. [Google Scholar]
- Appenroth, K.-J.; Sree, K.S.; Bog, M.; Ecker, J.; Seeliger, C.; Böhm, V.; Lorkowski, S.; Sommer, K.; Vetter, W.; Tolzin-Banasch, K.; et al. Nutritional Value of the Duckweed Species of the Genus Wolffia (Lemnaceae) as Human Food. Front. Chem. 2018, 6, 483. [Google Scholar] [CrossRef] [Green Version]
- Miranda, A.F.; Kumar, N.R.; Spangenberg, G.; Subudhi, S.; Lal, B.; Mouradov, A. Aquatic plants, landoltia punctata, and azolla filiculoides as bio-converters of wastewater to biofuel. Plants 2020, 9, 1–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muradov, N.; Taha, M.; Miranda, A.F.; Kadali, K.; Gujar, A.; Rochfort, S.; Stevenson, T.; Ball, A.S.; Mouradov, A. Dual application of duckweed and azolla plants for wastewater treatment and renewable fuels and petrochemicals production. Biotechnol. Biofuels 2014, 7, 30. [Google Scholar] [CrossRef] [Green Version]
- Toyama, T.; Hanaoka, T.; Tanaka, Y.; Morikawa, M.; Mori, K. Comprehensive evaluation of nitrogen removal rate and biomass, ethanol, and methane production yields by combination of four major duckweeds and three types of wastewater effluent. Bioresour. Technol. 2018, 250, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Huber, C.; Schröder, P. Fate of the sunscreen compound oxybenzone in Cyperus alternifolius based hydroponic culture: Uptake, biotransformation and phytotoxicity. Chemosphere 2017, 182, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Kotowska, U.; Karpińska, J.; Kapelewska, J.; Kowejsza, E.M.; Piotrowska-Niczyporuk, A.; Piekutin, J.; Kotowski, A. Removal of phthalates and other contaminants from municipal wastewater during cultivation of Wolffia arrhiza. Process. Saf. Environ. Prot. 2018, 120, 268–277. [Google Scholar] [CrossRef]
- Kaleniecka, A.; Zarzycki, P.K. Degradation Studies of Selected Bisphenols in the Presence of β-Cyclodextrin and/or Duckweed Water Plant. J. AOAC Int. 2020, 103, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rodriguez, A.; Matamoros, V.; Fontas, C.; Salvado, V. The influence of Lemna sp. and Spirogyra sp. on the removal of pharmaceuticals and endocrine disruptors in treated wastewaters. Int. J. Environ. Sci. Technol. 2014, 12, 2327–2338. [Google Scholar] [CrossRef] [Green Version]
- Dos Reis, A.R.; Tabei, K.; Sakakibara, Y. Oxidation mechanism and overall removal rates of endocrine disrupting chemicals by aquatic plants. J. Hazard. Mater. 2014, 265, 79–88. [Google Scholar] [CrossRef] [Green Version]
- Campos, J.M.; Queiroz, S.C.; Roston, D.M. Removal of the endocrine disruptors ethinyl estradiol, bisphenol A, and levonorgestrel by subsurface constructed wetlands. Sci. Total. Environ. 2019, 693, 133514. [Google Scholar] [CrossRef] [PubMed]
- Toro-Vélez, A.; Madera-Parra, C.; Peña, M.R.; Lee, W.; Cruz, J.B.; Walker, W.; Cárdenas-Henao, H.; Quesada-Calderón, S.; Garcia, H.; Lens, P. BPA and NP removal from municipal wastewater by tropical horizontal subsurface constructed wetlands. Sci. Total. Environ. 2016, 542, 93–101. [Google Scholar] [CrossRef]
- Eimoori, R.; Zolala, J.; Pourmohiabadi, H.; Noroozian, E.; Mansouri, H. Contribution of Azolla filiculoides to hydrazine elimination from water. Wetl. Ecol. Manag. 2020, 28, 439–447. [Google Scholar] [CrossRef]
- Masoudian, Z.; Salehi-Lisar, S.Y.; Norastehnia, A. Phytoremediation potential of Azolla filiculoides for sodium dodecyl benzene sulfonate (SDBS) surfactant considering some physiological responses, effects of operational parameters and biodegradation of surfactant. Environ. Sci. Pollut. Res. 2020, 27, 20358–20369. [Google Scholar] [CrossRef] [PubMed]
- Saleem, H.; Rehman, K.; Arslan, M.; Afzal, M. Enhanced degradation of phenol in floating treatment wetlands by plant-bacterial synergism. Int. J. Phytoremediat. 2018, 20, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Al-Baldawi, I.A. Removal of 1,2-Dichloroethane from real industrial wastewater using a sub-surface batch system with Typha angustifolia L. Ecotoxicol. Environ. Saf. 2018, 147, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Ogata, Y.; Toyama, T.; Yu, N.; Wang, X.; Sei, K.; Ike, M. Occurrence of 4-tert-butylphenol (4-t-BP) biodegradation in an aquatic sample caused by the presence of Spirodela polyrrhiza and isolation of a 4-t-BP-utilizing bacterium. Biodegradation 2012, 24, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Hänninen, O. Pentachlorophenol: Uptake/Elimination Kinetics and Metabolism in an Aquatic Plant, Eichhornia Crassipes. Environ. Toxicol. Chem. 1994, 13, 763. [Google Scholar] [CrossRef]
- Li, J.; Zhou, Q.; Campos, L.C. Removal of selected emerging PPCP compounds using greater duckweed (Spirodela polyrhiza) based lab-scale free water constructed wetland. Water Res. 2017, 126, 252–261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dosnon-Olette, R.; Couderchet, M.; El Arfaoui, A.; Sayen, S.; Eullaffroy, P. Influence of initial pesticide concentrations and plant population density on dimethomorph toxicity and removal by two duckweed species. Sci. Total. Environ. 2010, 408, 2254–2259. [Google Scholar] [CrossRef]
- Gorzerino, C.; Quemeneur, A.; Hillenweck, A.; Baradat, M.; Delous, G.; Ollitrault, M.; Azam, D.; Caquet, T.; Lagadic, L. Effects of diquat and fomesafen applied alone and in combination with a nonylphenol polyethoxylate adjuvant on Lemna minor in aquatic indoor microcosms. Ecotoxicol. Environ. Saf. 2009, 72, 802–810. [Google Scholar] [CrossRef]
- Gikas, G.D.; Pérez-Villanueva, M.E.; Tsioras, M.; Alexoudis, C.; Pérez-Rojas, G.; Masís-Mora, M.; Lizano-Fallas, V.; Rodríguez-Rodríguez, C.E.; Vryzas, Z.; Tsihrintzis, V.A. Low-cost approaches for the removal of terbuthylazine from agricultural wastewater: Constructed wetlands and biopurification system. Chem. Eng. J. 2018, 335, 647–656. [Google Scholar] [CrossRef]
- Yu, X.; Zhu, H.; Yan, B.; Xu, Y.; Bañuelos, G.; Shutes, B.; Wen, H.; Cheng, R. Removal of chlorpyrifos and its hydrolytic metabolite 3,5,6-trichloro-2-pyridinol in constructed wetland mesocosms under soda saline-alkaline conditions: Effectiveness and influencing factors. J. Hazard. Mater. 2019, 373, 67–74. [Google Scholar] [CrossRef]
- Liang, Y.; Zhu, H.; Bañuelos, G.; Shutes, B.; Yan, B.; Cheng, X. Removal of sulfamethoxazole from salt-laden wastewater in constructed wetlands affected by plant species, salinity levels and co-existing contaminants. Chem. Eng. J. 2018, 341, 462–470. [Google Scholar] [CrossRef]
- Iatrou, E.I.; Gatidou, G.; Damalas, D.; Thomaidis, N.S.; Stasinakis, A.S. Fate of antimicrobials in duckweed Lemna minor wastewater treatment systems. J. Hazard. Mater. 2017, 330, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Feng, G.; Gao, X.; Sun, C.; Guo, J.-S.; Zhu, Z. Removal of pharmaceutically active compounds (PhACs) and toxicological response of Cyperus alternifolius exposed to PhACs in microcosm constructed wetlands. J. Hazard. Mater. 2016, 301, 566–575. [Google Scholar] [CrossRef]
- Panja, S.; Sarkar, D.; Datta, R. Removal of tetracycline and ciprofloxacin from wastewater by vetiver grass (Chrysopogon zizanioides (L.) Roberty) as a function of nutrient concentrations. Environ. Sci. Pollut. Res. 2020, 27, 34951–34965. [Google Scholar] [CrossRef] [PubMed]
- Vo, H.N.P.; Koottatep, T.; Chapagain, S.K.; Panuvatvanich, A.; Polprasert, C.; Nguyen, T.M.H.; Chaiwong, C.; Nguyen, N.L. Removal and monitoring acetaminophen-contaminated hospital wastewater by vertical flow constructed wetland and peroxidase enzymes. J. Environ. Manag. 2019, 250, 109526. [Google Scholar] [CrossRef]
- Vymazal, J.; Březinová, T.D.; Koželuh, M.; Kule, L. Occurrence and removal of pharmaceuticals in four full-scale constructed wetlands in the Czech Republic—The first year of monitoring. Ecol. Eng. 2017, 98, 354–364. [Google Scholar] [CrossRef]
- Di Baccio, D.; Pietrini, F.; Bertolotto, P.; Pérez, S.; Barcelò, D.; Zacchini, M.; Donati, E. Response of Lemna gibba L. to high and environmentally relevant concentrations of ibuprofen: Removal, metabolism and morpho-physiological traits for biomonitoring of emerging contaminants. Sci. Total. Environ. 2017, 584–585, 363–373. [Google Scholar] [CrossRef]
- Liang, Z.; Lv, T.; Zhang, Y.; Stein, O.R.; Arias, C.A.; Brix, H.; Carvalho, P.N. Effects of constructed wetland design on ibuprofen removal—A mesocosm scale study. Sci. Total. Environ. 2017, 609, 38–45. [Google Scholar] [CrossRef]
- Sochacki, A.; Nowrotek, M.; Felis, E.; Kalka, J.; Ziembińska-Buczyńska, A.; Bajkacz, S.; Ciesielski, S.; Miksch, K. The effect of loading frequency and plants on the degradation of sulfamethoxazole and diclofenac in vertical-flow constructed wetlands. Ecol. Eng. 2018, 122, 187–196. [Google Scholar] [CrossRef]
- Shi, W.; Wang, L.; Rousseau, D.P.L.; Lens, P.N.L. Removal of estrone, 17α-ethinylestradiol, and 17-estradiol in algae and duckweed-based wastewater treatment systems. Environ. Sci. Pollut. Res. 2010, 17, 824–833. [Google Scholar] [CrossRef]
- Song, H.-L.; Nakano, K.; Taniguchi, T.; Nomura, M.; Nishimura, O. Estrogen removal from treated municipal effluent in small-scale constructed wetland with different depth. Bioresour. Technol. 2009, 100, 2945–2951. [Google Scholar] [CrossRef]
- Vymazal, J.; Březinová, T.; Koželuh, M. Occurrence and removal of estrogens, progesterone and testosterone in three constructed wetlands treating municipal sewage in the Czech Republic. Sci. Total. Environ. 2015, 536, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Fekete-Kertész, I.; Kunglné-Nagy, Z.; Gruiz, K.; Magyar; Farkas; Molnár, M. Assessing Toxicity of Organic Aquatic Micropollutants Based on the Total Chlorophyll Content of Lemna minor as a Sensitive Endpoint. Period. Polytech. Chem. Eng. 2015, 59, 262–271. [Google Scholar] [CrossRef] [Green Version]
- Richter, E.; Roller, E.; Kunkel, U.; Ternes, T.A.; Coors, A. Phytotoxicity of wastewater-born micropollutants—Characterisation of three antimycotics and a cationic surfactant. Environ. Pollut. 2016, 208, 512–522. [Google Scholar] [CrossRef]
- Nika, M.-C.; Ntaiou, K.; Elytis, K.; Thomaidi, V.; Gatidou, G.; Kalantzi, O.; Thomaidis, N.; Stasinakis, A. Wide-scope target analysis of emerging contaminants in landfill leachates and risk assessment using Risk Quotient methodology. J. Hazard. Mater. 2020, 394, 122493. [Google Scholar] [CrossRef] [PubMed]
- Basiglini, E.; Pintore, M.; Forni, C. Effects of treated industrial wastewaters and temperatures on growth and enzymatic activities of duckweed (Lemna minor L.). Ecotoxicol. Environ. Saf. 2018, 153, 54–59. [Google Scholar] [CrossRef]
- Pietrini, F.; Passatore, L.; Fischetti, E.; Carloni, S.; Ferrario, C.; Polesello, S.; Zacchini, M. Evaluation of morpho-physiological traits and contaminant accumulation ability in Lemna minor L. treated with increasing perfluorooctanoic acid (PFOA) concentrations under laboratory conditions. Sci. Total. Environ. 2019, 695, 133828. [Google Scholar] [CrossRef] [PubMed]
- Ceschin, S.; Crescenzi, M.; Iannelli, M.A. Phytoremediation potential of the duckweeds Lemna minuta and Lemna minor to remove nutrients from treated waters. Environ. Sci. Pollut. Res. 2020, 27, 15806–15814. [Google Scholar] [CrossRef]
- Mburu, N.; Tebitendwa, S.M.; Rousseau, D.P.L.; van Bruggen, J.J.A.; Lens, P.N.L. Performance Evaluation of Horizontal SubsurfaceFlow–Constructed Wetlands for the Treatmentof Domestic Wastewater in the Tropics. J. Environ. Eng. 2013, 139, 986–994. [Google Scholar] [CrossRef]
- Tabassum-Abbasi, P.P.; Abbasi, S.A. Ability of Indian pennywort Bacopa monnieri (L.) Pennell in the phytoremediation of sewage (greywater). Environ. Sci. Pollut. Res. 2020, 27, 6078–6087. [Google Scholar] [CrossRef]
- Adelodun, A.A.; Hassan, U.O.; Nwachuckwu, V.O. Environmental, mechanical, and biochemical benefits of water hyacinth (Eichhornia crassipes). Environ. Sci. Pollut. Res. 2020, 27, 30210–30221. [Google Scholar] [CrossRef] [PubMed]
- Valipour, A.; Raman, V.K.; Ahn, Y.-H. Effectiveness of Domestic Wastewater Treatment Using a Bio-Hedge Water Hyacinth Wetland System. Water 2015, 7, 329–347. [Google Scholar] [CrossRef] [Green Version]
- Weragoda, S.K.; Jinadasa, K.B.S.N.; Zhang, D.Q.; Gersberg, R.M.; Tan, S.K.; Tanaka, N.; Jern, N.W. Tropical Application of Floating Treatment Wetlands. Wetlands 2012, 32, 955–961. [Google Scholar] [CrossRef]
- Saeed, T.; Afrin, R.; Al Muyeed, A.; Sun, G. Treatment of tannery wastewater in a pilot-scale hybrid constructed wetland system in Bangladesh. Chemosphere 2012, 88, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhao, Y.; Tang, C.; Xu, L.; Morgan, D.; Liu, R. Role of macrophyte species in constructed wetland-microbial fuel cell for simultaneous wastewater treatment and bioenergy generation. Chem. Eng. J. 2020, 392, 123708. [Google Scholar] [CrossRef]
- Kumari, M.; Tripathi, B.D. Effect of aeration and mixed culture of Eichhornia crassipes and Salvinia natans on removal of wastewater pollutants. Ecol. Eng. 2014, 62, 48–53. [Google Scholar] [CrossRef]
- Ceschin, S.; Sgambato, V.; Ellwood, N.T.W.; Zuccarello, V. Phytoremediation performance of Lemna communities in a constructed wetland system for wastewater treatment. Environ. Exp. Bot. 2019, 162, 67–71. [Google Scholar] [CrossRef]
- Chance, L.M.G.; Majsztrik, J.C.; Bridges, W.C.; Willis, S.A.; Albano, J.P.; White, S.A. Comparative Nutrient Remediation by Monoculture and Mixed Species Plantings within Floating Treatment Wetlands. Environ. Sci. Technol. 2020, 54, 8710–8718. [Google Scholar] [CrossRef] [PubMed]
| Compounds | Water Sample | Concentration | References |
|---|---|---|---|
| Benzotriazoles | |||
| 4-methyl-1H-benzotriazole | Surface water | 1.44–4345 ng⋅dm−3 | [11] |
| 5-methyl-1H-benzotriazole | Surface water | 2–7181.4 ng⋅dm−3 | |
| 1H-benzotriazole | Surface water | <0.2–8529.8 ng⋅dm−3 | |
| UV filters | |||
| benzophenone | Landfill leachate | 0.15–16 μg⋅dm−3 | [12] |
| Groundwater from landfill | 0.028–0.492 μg⋅dm−3 | ||
| benzophenone 3 | Landfill leachate | <0.01–0.646 μg⋅dm−3 | [28] |
| Plasticizers | |||
| di-n-butylphthalate | Freshwater | 0.22–3.86 μg⋅dm−3 | [29] |
| dimethyl phthalate | Freshwater | 0.02–0.25 μg⋅dm−3 | [29] |
| bis(2-ethylhexyl) phthalate | Freshwater | 5.16–20.80 μg⋅dm−3 | [29] |
| Industrial chemicals | |||
| bisphenol A | Surface water | up to 330 ng⋅dm−3 | [30] |
| 2.2–1030 ng⋅dm−3 | [31] | ||
| 4-tert-octylphenol | Surface water | 1.0–2470 ng⋅dm−3 | |
| 4-nonylphenol | Surface water | 28.1–8890 ng⋅dm−3 | |
| Wastewater | 0.19–102.54 μg⋅dm−3 | [32] | |
| Insect repellents | |||
| N,N-diethyl-3-methylbenzamide | Landfill leachate | 73.7 ng⋅cm−3 | [33] |
| Groundwater from landfill | 0.019–16.901 μg⋅dm−3 | [34] | |
| Pesticides | |||
| atrazine | Surface water | 1.8–18 mg⋅dm−3 | [14] |
| dimethomorph | Groundwater | 0.002–0.03 μg⋅dm−3 | [10] |
| Antibacterial and antibiotics | |||
| sulfamethoxazole | Drinking water | 440 µg⋅dm−3 | [14] |
| ofloxacin | Drinking water | 11.1–1330 ng⋅dm−3 | [35] |
| Non-steroidal anti-inflammatory drugs | |||
| diclofenac | Surface water | 8.8–127 mg⋅dm−3 | [14] |
| Landfill leachates | 0.081–0.61 µg⋅dm−3 | [36] | |
| naproxen | Wastewater | 0.78–551.96 μg⋅dm−3 | [32] |
| ibuprofen | Drinking water | 50 µg⋅dm−3 | [14] |
| ketoprofen | Wastewater | 0.95–233.63 μg⋅dm−3 | [32] |
| Hormones | |||
| estrone | Wastewater | 0.2–10.1 ng⋅dm−3 | [14] |
| 17-β-estradiol | |||
| 17-α-ethinylestradiol | Ground water | 0.5–230 ng⋅dm−3 | [14] |
| estrone | Surface water | 0.7–75 ng⋅dm−3 | [31] |
| 17-β-estradiol | Surface water | 0.7–7.5 ng⋅dm−3 | [31] |
| Plants | Compounds | Analytes Concentrations | Time of Exposure | Type of Installation, Type of Medium | Removal | Remarks | Ref. | |
|---|---|---|---|---|---|---|---|---|
| Benzotriazoles | ||||||||
| Lemna minor | 1H-benzotriazole (BTR) | 150 µg·dm−3 | constant illumination | laboratory experiment, SIS medium | >99.9% | 36 days experiment | [45] | |
| 4-methyl-1H-benzotriazole (4TTR) | 48.2% | |||||||
| 5-methyl-1H-benzotriazole (5TTR) | >99.9% | |||||||
| xylytriazole | >99.9% | |||||||
| 5-chlorobenzotriazole (CBTR) | >99.9% | |||||||
| Cyperus alternifolius | oxybenzone | 5·10−6 mol·dm−3 | 12/12h light/darkness | laboratory experiment, Hoagland medium | 86.5% | 120h experiment | [75] | |
| 2,5·10−5 mol·dm−3 | 81.4% | |||||||
| Wolffia arrhiza | dibutyl phthalate | 100 µg·dm−3 | 16/8h light/darkness | laboratory experiment, non-treated wastewaters from WWTP and Hutner’s medium | 87.2% | [76] | ||
| bis(2-ethylhexyl) phthalate | 97.7% | |||||||
| Lemna minor L. | bisphenol A | 1 µg·dm−3 | 12/12h light/darkness | tap water | 62.5% | [77] | ||
| Lemna sp. | 100 µg·dm−3 | 12/12h light/darkness | secondary-treated wastewaters from WWTP | 96% | external experiment, natural conditions, Spain | [78] | ||
| Ceratophyllum demersum | 1–100 µg·dm−3 | 16/8h light/darkness | laboratory experiment, Hoagland medium | 99.2% | [79] | |||
| Riccia fluitans | 99.5% | |||||||
| Spirodela polyrhiza | 98.5% | |||||||
| Limnobium leavigatum | 89.9% | |||||||
| Cyperus isoclaudus | 20 µg·dm−3 | SSF-CW, synthetic wastewater | 85.9% | in 4 days | [80] | |||
| Eichhornia crassipes | 87.7% | |||||||
| Cyperus isoclaudus | 95.5% | gravel and bamboo charcoal as support medium | ||||||
| Phragmites australis | ~0.6 µg·dm−3 | HSF-CW, effluent of an anaerobic pond as primary treatment | 70.2% | [81] | ||||
| Helicionia pistacorum | 73.3% | |||||||
| Lemna minor L. | bisphenol S | 1 µg·dm−3 | 12/12h light/darkness | tap water | 72.2% | [77] | ||
| bisphenol B | 85.7% | |||||||
| Helicionia pistacorum | nonylphenol | HSF-CW, effluent of an anaerobic pond as primary treatment | 62.8% | [81] | ||||
| Phragmites australis | 52.1% | |||||||
| Azolla filiculoides | hydrazine | 16/8h light/darkness | Hoagland medium | 93% | 24h experiment | [82] | ||
| >99.9% | 96h experiment | |||||||
| Azolla filiculoides | sodium dodecyl benzene sulfonate | 10–40 mg·dm−3 | 16/8h light/darkness | nitrogen-free medium | 77.79% | [83] | ||
| Typha domingensis | phenol | 500 mg·dm−3 | tap water | 75% | 15 days experiment | [84] | ||
| Typha angustifolia L. | 1,2-dichloroethane | 390 mg·dm−3 | wastewater from a petrochemical industry; HSSF CWs | >99.9% | external experiment, natural conditions, Malaysia | [85] | ||
| Spirodela polyrhiza | 4-tert-butylphenol | 3.3·10−5 mol·dm−3 | 16/8h light/darkness | water samples from aquatic systems | 63% | external experiment, natural conditions, Japan | [86] | |
| Eichornia crassipes | pentachlorophenol | 2·10−6 mol·dm−3 | 12/12h light/darkness | distilled water | 47% | 12h experiment | [87] | |
| Spirodela polyrhiza | DEET | 25 mg·dm−3 | 14/10h light/darkness | laboratory experiment with synthetic wastewaters | 32.6% | [88] | ||
| Lemna minor | dimethomorph | 25 µg·dm−3 | laboratory experiment, nutrient medium | 36% | [89] | |||
| Spirodela polyrhiza | 32% | |||||||
| Lemna minor | 50 µg·dm−3 | 28% | ||||||
| Spirodela polyrhiza | 26% | |||||||
| Lemna minor | 600 µg·dm−3 | 19% | 96h experiment | |||||
| Spirodela polyrhiza | 14% | |||||||
| Lemna minor | 600 µg·dm−3 | 15% | plant density 0.05 g/flask | |||||
| 19% | plant density 0.10 g/flask | |||||||
| 20% | plant density 0.15 g/flask | |||||||
| 22% | plant density 0.20 g/flask | |||||||
| Spirodela polyrhiza | 600 µg·dm−3 | 8% | plant density 0.05 g/flask | |||||
| 14% | plant density 0.10 g/flask | |||||||
| 17% | plant density 0.15 g/flask | |||||||
| 18% | plant density 0.20 g/flask | |||||||
| Lemna minor | diquat | 44.4 µg·dm−3 | 14/10h light/darkness | dechlorinated tap water filtered through activated-charcoal cartridges | >99.9% | 16 days experiment | [90] | |
| 222.2 µg·dm−3 | >99.9% | |||||||
| fomesafen | 44.4 µg·dm−3 | 54.7% | ||||||
| 222.2 µg·dm−3 | 74.4% | |||||||
| Typha latifolia | terbuthylazine | 0.4 mg·dm−3 | HSF-CW | 58.4% | [91] | |||
| Phragmites australis | 73.7% | |||||||
| Canna indica | chlorpyrifos | 100 µg·dm−3 | SSF-CW | 88.07% | 24h experiment | [92] | ||
| 200 µg·dm−3 | 93.76% | |||||||
| 300 µg·dm−3 | 96.55% | |||||||
| Phragmites australis/ Typha oruentalis/ Vetiveria zizanioides/ Canna indica | sulfamethoxazole (SMX) | 2.3 μg·dm−3 | HSSF-CW, VSSF-CW | 70.1–76.3% | [93] | |||
| Lemna minor | 10 μg·dm−3 | 16/8h light/darkness | secondary biologically treated wastewater | 73% | 14 days experiment | [94] | ||
| Cyperus alternifolius | 10–500 µg·dm−3 | water simulating synthetic secondary sewage from WWTP | <80% | [95] | ||||
| Cyperus alternifolius | ofloxacin (OFX) | 90% | ||||||
| Cyperus alternifolius | roxithromycin (ROX) | 85% | ||||||
| Lemna minor | cefadroxil (CFD) | 10 μg·dm−3 | 16/8h light/darkness | secondary biologically treated wastewater | >99.9% | 14 days experiment | [94] | |
| Lemna minor | metronidazole (METRO) | 96% | ||||||
| Lemna minor | trimethoprim (TRI) | 59% | ||||||
| Chrysopogon zizanioides (L.) Roberty | ciprofloxacin (CIP) | 10 mg·dm−3 | wastewaters from WWTP | 60–94% | external experiment, natural conditions, USA | [96] | ||
| tetracycline (TTC) | 89->99.9% | |||||||
| Spirodela polyrhiza | paracetamol | 25 mg·dm−3 | 14/10h light/darkness | laboratory experiment with synthetic wastewaters | 97.7% | [88] | ||
| Salix alba+ Iris pseudacorus+ Juncus effusus+ Callitriche palustris+ Carex carophyllea | 10 mg·dm−3 | SVF-CW, wastewaters from hospital’s WWTP | 0->99.9% | external experiment, natural conditions, Thailand | [97] | |||
| Phragmites australis | HSF-CW, wastewaters from hospital’s WWTP | 20–69% | external experiment, natural conditions, Thailand | |||||
| Phragmites australis + Phalaris arundinacea | 86.2-99.6%. | |||||||
| Phragmites australis | 35 µg·dm−3 | 95% | ||||||
| 30 µg·dm−3 | 45% | |||||||
| Phragmites australis + Typha latifolia | 750 ng·dm−3 | 51.7–99% | ||||||
| Typha angustifolia+ Chrysopogon zizanioides+ Cyperus papyrus | 58.1% | |||||||
| Phragmites australis | 3–71 μg·dm−3 | HSSF-CW in the water- shed of a drinking water reservoir | 89–99.6% | external experiment, natural conditions, Czech Republic | [98] | |||
| Phalaris arundinacea | 0.35–180 μg·dm−3 | 86.2–88.7% | ||||||
| Lemna gibba L. | ibuprofen | 0.02–1 mg·dm−3 | 14/10h light/darkness | laboratory experiment, Murashige and Skoog medium | 89–92.5% | [99] | ||
| Juncus effusus/ Typha latifolia/ Berula erecta/ Phragmites australis/ Iris pseudacorus | 10 μg·dm−3 | water unsaturated CW, water saturated CW, aerated water saturated CW, tap water | 29–99% | [100] | ||||
| Lemna sp. | 100 µg·dm−3 | 12/12h light/darkness | secondary-treated wastewaters from WWTP | 93% | external experiment, natural conditions, Spain | [78] | ||
| Spirogyra sp. | 92% | |||||||
| Phragmites australis | 1.9–64 μg·dm−3 | HSSF-CW in the water- shed of a drinking water reservoir | 51.6–75% | external experiment, natural conditions, Czech Republic | [98] | |||
| Phragmites australis + Phalaris arundinacea | 6.6–36 μg·dm−3 | 45.6–49.4% | ||||||
| Phragmites australis | diclofenac | 140–5400 ng·dm−3 | 11.5–67.1% | |||||
| Phragmites australis + Phalaris arundinacea | 10–12000 ng·dm−3 | 29.3–58% | ||||||
| Phalaris arundinacea | 0.5 mg·dm−3 | 14/10h light/darkness | laboratory experiment, vertical-flow CW, synthetic wastewaters | 47.3% | the loading frequency 1 pulse per day | [101] | ||
| Phalaris arundinacea | 74.2% | the loading frequency 4 pulses per day | ||||||
| Spirogyra sp. | 100 µg·dm−3 | 12/12h light/darkness | secondary-treated wastewaters from WWTP | 54% | external experiment, natural conditions, Spain | [78] | ||
| Lemna sp. | 48% | |||||||
| Scripus validus | 0.5-2 mg·dm−3 | 12/12h light/darkness | laboratory experiment, Hoagland medium | 85–98% | [60] | |||
| Phragmites australis | ketoprofen | <10–6500 ng·dm−3 | HSSF-CW in the water- shed of a drinking water reservoir | 46.9–91.2% | external experiment, natural conditions, Czech Republic | [98] | ||
| Phragmites australis + Phalaris arundinacea | 19–2600 ng·dm−3 | 18.1% | ||||||
| Scripus validus | naproxen | 0.5–2 mg·dm−3 | 12/12h light/darkness | Hoagland medium | 90% | external experiment, natural conditions, Spain | [60] | |
| Lemna sp. | 17-α-ethinylestradiol | 100 µg·dm−3 | 12/12h light/darkness | secondary- treated wastewaters from WWTP | 94% | external experiment, natural conditions, Spain | [78] | |
| Spirogyra sp. | 94% | |||||||
| Ceratophyllum demersum, Riccia fluitans, Limnobium laevigatum, Spirodela polyrhiza | 1–100 µg·dm−3 | 16/8h light/darkness | laboratory experiment, Hoagland medium | >99.9% | addition of crude enzymes | [79] | ||
| Lemna species | 10 mg·dm−3 | 12/12h light/darkness | laboratory experiment, synthetic wastewater | >95% | 6 days experiment | [102] | ||
| Cyperus isoclaudus | 20 µg·dm−3 | SSF-CW, synthetic wastewater | 81.4% | [80] | ||||
| Eichhornia crassipes | 67.8% | |||||||
| Cyperus isoclaudus | >99.9% | gravel and bamboo charcoal as support medium | ||||||
| Phragmites australis | 0.94–3.62 ng·dm−3 | vertical-flow CW, effluent from WWTP | 75.3% | external experiment, natural conditions, Japan | [102] | |||
| Phragmites australis + Phalaris arundinacea | estrone | 5.9 ng·dm−3 | HSSF-CW, municipal wastewater | 85% | external experiment, natural conditions, Czech Republic | [103] | ||
| Ceratophyllum demersum, Riccia fluitans | 1–100 µg·dm−3 | 16/8h light/darkness | laboratory experiment, Hoagland medium | >99.9% | addition of crude enzymes | [79] | ||
| Limnobium laevigatum | 89.4% | |||||||
| Ceratophyllum demersum | 87.4% | |||||||
| Lemna species | 10 mg·dm−3 | 12/12h light/darkness | laboratory experiment, synthetic wastewater | >95% | 6 days experiment | [102] | ||
| Phragmites australis | 1.17–6.18 ng·dm−3 | vertical-flow CW, effluent from WWTP in Japan | 67.8% | [104] | ||||
| Ceratophyllum demersum, Riccia fluitans | 17-β-estradiol | 1–100 µg·dm−3 | 16/8h light/darkness | laboratory experiment, Hoagland medium | >99.9% | addition of crude enzymes | [79] | |
| Limnobium laevigatum | 95.5% | |||||||
| Ceratophyllum demersum | 95.7% | |||||||
| Lemna species | 10 mg·dm−3 | 12/12h light/darkness | laboratory experiment, synthetic wastewater | >95% | 6 days experiment | [102] | ||
| Phragmites australis | 2.94–4.65 ng·dm−3 | vertical-flow CW, effluent from WWTP in Japan | 84.0% | [104] | ||||
| Cyperus isoclaudus | levonorgestrel | 100 µg·dm−3 | SSF-CW, synthetic wastewater | >99.9% | [80] | |||
| Eichhornia crassipes | >99.9% | |||||||
| Cyperus isoclaudus | 99.8% | gravel and bamboo charcoal as support medium | ||||||
| Micropollutants | Plants | Symptoms of Abiotic Stress | Comments | Ref. |
|---|---|---|---|---|
| dimethomorph, C = 600 µg·dm−3 | Lemna minor | inhibition of growth by 21%, inhibition of photosynthesis | the increase in plant density caused a lower growth rate, reaching 26% at 0.20 g/E-flask | [89] |
| Spirodela polyrhiza | inhibition of growth by 19%, inhibition of photosynthesis | the increase in plant density caused a lower growth rate, reaching 24% at 0.20 g/E-flask | ||
| oxybenzone | Cyperus alternifolius | increased activity of antioxidant enzymes | 12/12 h light/darkness | [75] |
| hydrazine, C = 0–1000 µg·dm−3 | Azolla filiculoides | plant growth | hydrazine was a nitrogen source for the plant; 16/8 h light/darkness | [82] |
| sodium dodecyl benzene sulfonate, C = 0–40 mg·dm−3 | Azolla filiculoides | stunted growth, effect index increases with increasing concentration, the activity of antioxidant enzymes was stimulated at each concentration and increased with longer exposure to the surfactant, content of anthocyanins, H2O2, antioxidant activity, and electrolyte leakage were higher with the duration of the test, the total content of chlorophylls tested after 7 days decreased with increasing concentration | after 7 days, the total carotenoid content was reduced by surfactant at a concentration of 30 mg·dm−3 and 40 mg·dm−3; 16/8 h light/darkness | [83] |
| diquat, C = 44.4 µg·dm−3 | Lemna minor | 80% fading leaves, lower relative leaf number (FRG) and relative leaf area (RFA) | 14/10 h light/darkness | [90] |
| diquat, C = 222.2 µg·dm−3 | 100% leaf fading, much lower FRG and RFA | |||
| fomesafen, C = 44.4 µg·dm−3 | relative leaf number increase, RFA lower | |||
| fomesafen, C = 222.2 µg·dm−3 | inhibition RFN, much smaller RFA | |||
| diclofenac, C = 25 mg·dm−3 | Lemna minor | reduction in the amount of chlorophylls by 64% | diclofenac, nicotine, and 3,4-dichlorophenol inhibited chlorophyll content more than leaf number; 16/8 h light/darkness | [105] |
| bisphenol A | the total content of chlorophylls decreased with increasing concentration | |||
| 3,4-dichlorofenolu | ||||
| ketokonazol | Lemna minor | the size and number of leaves decreased and the color of the leaves changed | the plant had a hard time returning to previous state; 16/8 h light/darkness | [106] |
| benzyldimethyldodecylammonium chloride | ||||
| climbazole | ||||
| fluconazole | ||||
| mixture of micropollutants | Lemna minor | 100% inhibition of the growth rate | samples of wastewater contained bisphenol A, valsartan and 2-OH-benzothiazole | [107] |
| mixture of micropollutants | Lemna minor | plant’s growth inhibited by about 25% in the summer, no growth in the winter, in winter is more antioxidants | samples of sewage contained surfactants (heavy metals, hydrocarbons, nitrogen and phosphorus compounds) | [108] |
| perfluorooctanoic acid | Lemna minor | no change in the number of leaves, growth rate, chlorophyll content, and photosynthesis efficiency | any harmful effect on the plant; 16/8 h light/darkness | [109] |
| Plants | Pollution Indicators Changes | Ref. |
|---|---|---|
| Lemna minuta | DO ↓65%, NO3−-N ↓85% | [110] |
| Lemna minor | DO ↓30%, NO3−-N ↓60%, PO4–-P ↓70% | |
| Lemna minor | BOD ↓90%, COD ↓88% | [76] |
| Wolffia arrhiza | BOD ↓95%, COD ↓90%, TN ↓65–90%, TP ↓15–83% | |
| Cyperus alternifolius | COD ↓70–85%, NH4+-N ↓ 30–55%, TN ↓60–75% | [95] |
| Cyperus papyrus | TSS ↓75%, BOD ↓61%, COD ↓44%, NH4+-N ↓26%, NO3−-N ↓22%, TP ↓57% | [111] |
| Bacopa monnieri (L.) Pennell | TSS ↓90%, COD ↓76%, BOD ↓80%, P ↓55%, NO3−-N ↓65% | [112] |
| Eichhornia crassipes | NO3−-N ↓75%, PO4–-P ↓79%, TSS ↓, EC ↓, S ↓, pH ↑ | [113] |
| Eichhornia crassipes | NO3−-N ↓55%, PO4−-P ↓86%, TSS ↓, EC ↓, S ↓, pH ↓ | |
| COD ↓79%, BOD ↓86%, TN ↓76.61%, TP ↓44.84%, TSS ↓73.02%, PO4-–P ↓38.69%, NH4+-N ↓72.48% | [114] | |
| Salvinia molesta | TP ↓83%, NH4+-N ↓73%, COD ↓74.1% | [52] |
| Pistia stratiotes | TN ↓64.2% | |
| Typha domingenis | TOC ↓, BOD↓, COD↓, pH ↑ | [84] |
| Typha angustifolia | BOD ↓76%, NH4+-N ↓86%, NO3−-N ↓41%, PO4-–P ↓100% | [115] |
| Canna iridiflora | BOD ↓85%, NH4+-N ↓82%, NO3−-N ↓50%, PO4-–P ↓100% | |
| Phalaris arundinacea | DOC ↓95%, TKN ↓90–95%, NH4+-N ↓59–96%, PO4--P ↓25–31% | [101] |
| Phragmites australis | TSS ↓55%, BOD ↓98%, COD ↓ 98%, NH4+-N ↓86%, NO3−-N ↓50%, TP ↓87% | [116] |
| Scirpus validus | TSS ↓80%, NH4+-N ↓95%, TKN ↓65.3%, COD ↓88.1%, TN ↓22.5%, TP ↓67.1% | [97] |
| Spirodela polyrhiza | COD ↓79.3–89.3%, TOC ↓85.3–91.3% | [88] |
| Iris pseudacorus | NH4+-N ↓71.9%, NO3−-N ↓96.1% | [117] |
| Eichhornia crassipes + Salvinia natans | BOD ↓84.5%, COD ↓83.2%, TKN ↓53%, NO3−-N ↓26.6%, PO4--P ↓56.6% | [118] |
| several species of plants | DO ↓60–70%, NO3--N ↓80%, pH ↓ | [119] |
| several species of plants | DO ↓, pH ↓ | [120] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polińska, W.; Kotowska, U.; Kiejza, D.; Karpińska, J. Insights into the Use of Phytoremediation Processes for the Removal of Organic Micropollutants from Water and Wastewater; A Review. Water 2021, 13, 2065. https://doi.org/10.3390/w13152065
Polińska W, Kotowska U, Kiejza D, Karpińska J. Insights into the Use of Phytoremediation Processes for the Removal of Organic Micropollutants from Water and Wastewater; A Review. Water. 2021; 13(15):2065. https://doi.org/10.3390/w13152065
Chicago/Turabian StylePolińska, Weronika, Urszula Kotowska, Dariusz Kiejza, and Joanna Karpińska. 2021. "Insights into the Use of Phytoremediation Processes for the Removal of Organic Micropollutants from Water and Wastewater; A Review" Water 13, no. 15: 2065. https://doi.org/10.3390/w13152065
APA StylePolińska, W., Kotowska, U., Kiejza, D., & Karpińska, J. (2021). Insights into the Use of Phytoremediation Processes for the Removal of Organic Micropollutants from Water and Wastewater; A Review. Water, 13(15), 2065. https://doi.org/10.3390/w13152065








