Drinking Water Arsenic and Adverse Reproductive Outcomes in Men and Women: A Systematic PRISMA Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy and Study Selection
- Studies containing a relevant population:
- -
- Women of fertile age (15–51 years)
- -
- Men of fertile age (15–65 years)
- Studies assessing arsenic exposure from drinking water
- Studies containing a relevant control group
- Studies investigating at least one of the following outcomes:
- -
- Infertility (including extended TTP, ART, and menstrual disturbances)
- -
- Semen quality parameters
- -
- Pregnancy complications (i.e., spontaneous abortion)
- Original studies
- Studies with one of the following designs:
- -
- Cohort study
- -
- Case-control study
- Studies published in the English language
2.2. Data Extraction and Quality Assessment
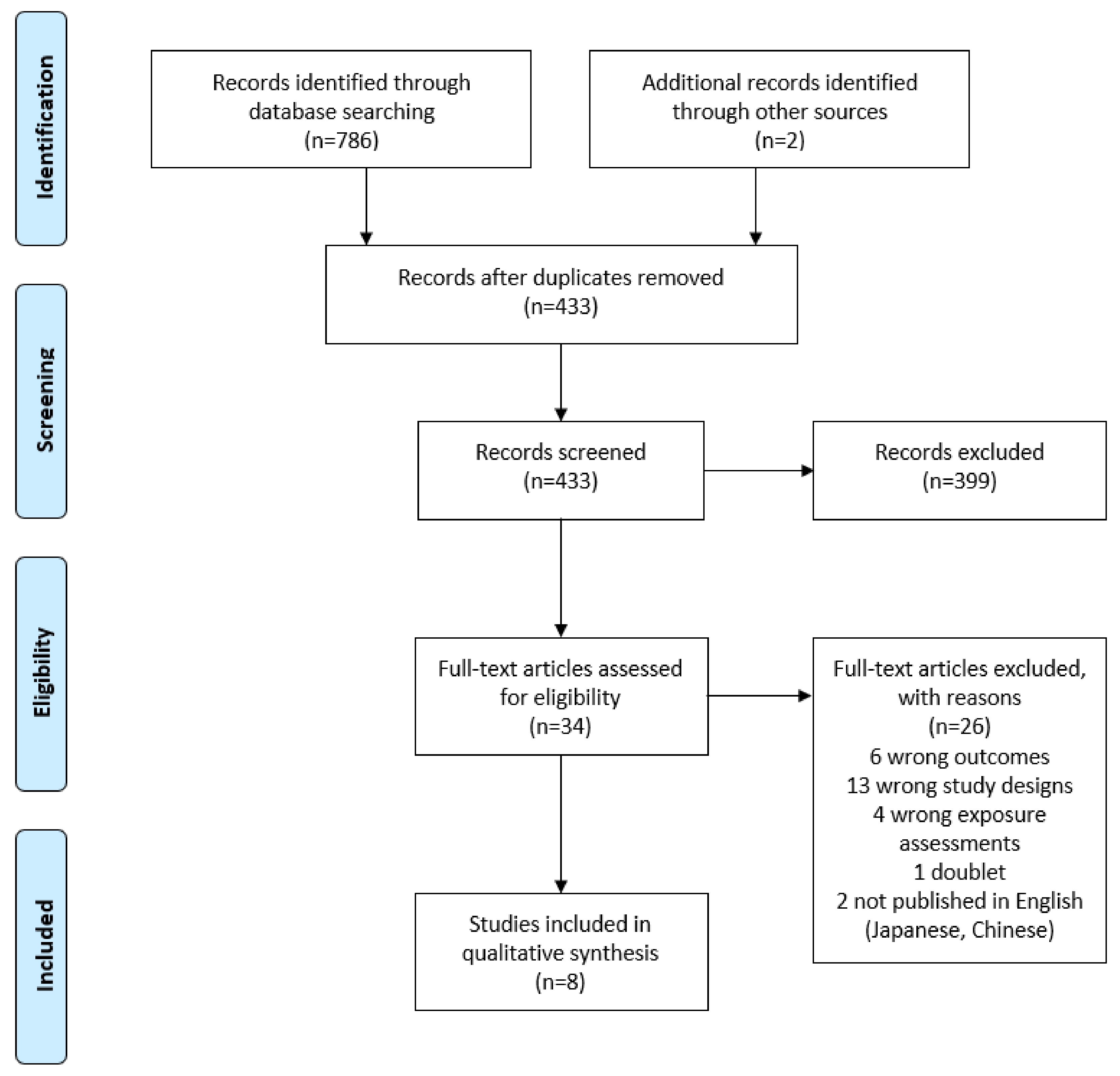
3. Results
3.1. Women
3.1.1. High-Level Arsenic Exposure (Above 10 µg/L)
3.1.2. Low to Moderate Arsenic Exposure (Below 10 µg/L)
3.2. Men
High-Level Arsenic Exposure (Above 10 µg/L)
4. Discussion
4.1. Strengths and Limitations
4.2. Spontaneous Abortion
4.3. Female Infertility and Fecundity
4.4. Male Infertility and Semen Quality Parameters
4.5. Assessment of Arsenic Exposure
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Juul, S.; Karmaus, W.; Olsen, J. Regional differences in waiting time to pregnancy: Pregnancy-based surveys from Denmark, France, Germany, Italy and Sweden. The European Infertility and Subfecundity Study Group. Hum. Reprod. 1999, 14, 1250–1254. [Google Scholar] [CrossRef] [PubMed]
- Ombelet, W.; Cooke, I.; Dyer, S.; Serour, G.; Devroey, P. Infertility and the provision of infertility medical services in developing countries. Hum. Reprod. Updat. 2008, 14, 605–621. [Google Scholar] [CrossRef] [PubMed]
- Weselak, M.; Arbuckle, T.E.; Walker, M.C.; Krewski, D. The influence of the environment and other exogenous agents on spontaneous abortion risk. J. Toxicol. Environ. Health Part B 2008, 11, 221–241. [Google Scholar] [CrossRef] [PubMed]
- IARC International Agency for Research on Cancer. Arsenic, Metals, Fibres and Dusts. A Review of Human Carcinogens. 2012. Available online: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono100C.pdf (accessed on 15 November 2019).
- Smedley, P.; Kinniburgh, D. A review of the source, behaviour and distribution of arsenic in natural waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines for Drinking-Water Quality, Fourth Edition, Geneva 2011. Available online: https://www.who.int/water_sanitation_health/dwq/chemicals/arsenic.pdf (accessed on 15 November 2019).
- Haque, R.; Mazumder, D.N.; Samanta, S.; Ghosh, N.; Kalman, D.; Smith, M.M.; Mitra, S.; Santra, A.; Lahiri, S.; Das, S.; et al. Arsenic in drinking water and skin lesions: Dose-response data from West Bengal, India. Epidemiology 2003, 14, 174–182. [Google Scholar] [CrossRef]
- Kuo, C.C.; Moon, K.A.; Wang, S.L.; Silbergeld, E.; Navas-Acien, A. The Association of Arsenic Metabolism with Cancer, Cardiovascular Disease, and Diabetes: A Systematic Review of the Epidemiological Evidence. Environ. Health Perspect. 2017, 125, 087001. [Google Scholar] [CrossRef]
- Moon, K.A.; Guallar, E.; Umans, J.G.; Devereux, R.B.; Best, L.G.; Francesconi, K.A.; Goessler, W.; Pollak, J.; Silbergeld, E.K.; Howard, B.V.; et al. Association between exposure to low to moderate arsenic levels and incident cardiovascular disease. A prospective cohort study. Ann. Intern. Med. 2013, 159, 649–659. [Google Scholar]
- Hamadani, J.D.; Tofail, F.; Nermell, B.; Gardner, R.; Shiraji, S.; Bottai, M.; Arifeen, S.E.; Huda, S.N.; Vahter, M. Critical windows of exposure for arsenic-associated impairment of cognitive function in pre-school girls and boys: A population-based cohort study. Int. J. Epidemiol. 2011, 40, 1593–1604. [Google Scholar] [CrossRef]
- Vahter, M. Effects of arsenic on maternal and fetal health. Annu. Rev. Nutr. 2009, 29, 381–399. [Google Scholar] [CrossRef]
- Quansah, R.; Armah, F.A.; Essumang, D.K.; Luginaah, I.; Clarke, E.; Marfoh, K.; C obbina, S.J.; Nketiah-Amponsah, E.; Namujju, P.B.; Obiri, S.; et al. Association of arsenic with adverse pregnancy outcomes/infant mortality: A systematic review and meta-analysis. Environ. Health Perspect. 2015, 123, 412–421. [Google Scholar] [CrossRef]
- He, W.; Greenwell, R.J.; Brooks, D.M.; Calderon-Garciduenas, L.; Beall, H.D.; Coffin, J.D. Arsenic exposure in pregnant mice disrupts placental vasculogenesis and causes spontaneous abortion. Toxicol. Sci. 2007, 99, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Navarro, P.A.; Liu, L.; Keefe, D.L. In vivo effects of arsenite on meiosis, preimplantation development, and apoptosis in the mouse. Biol. Reprod. 2004, 70, 980–985. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, F.I.; Hwang, T.S.; Hsieh, Y.C.; Lo, H.C.; Su, C.T.; Hsu, H.S.; Chiou, H.Y.; Chen, C.J. Risk of erectile dysfunction induced by arsenic exposure through well water consumption in Taiwan. Environ. Health Perspect. 2008, 116, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Bao, H.; Liu, F.; Liu, L.; Zhu, Y.G.; She, J.; Dong, S.; Cai, M.; Li, L.; Li, C.; et al. Environmental exposure to arsenic may reduce human semen quality: Associations derived from a Chinese cross-sectional study. Environ. Health 2012, 11, 46. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Rossano, M.G.; Protas, B.; Diamond, M.P.; Puscheck, E.; Daly, D.; Paneth, N.; Wirth, J.J. Cadmium, lead, and other metals in relation to semen quality: Human evidence for molybdenum as a male reproductive toxicant. Environ. Health Perspect. 2008, 116, 1473–1479. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, J.; Xu, W.; Huang, Q.; Liu, L.; Tian, M.; Xia, Y.; Zhang, W.; Shen, H. Low-level environmental arsenic exposure correlates with unexplained male infertility risk. Sci. Total. Environ. 2016, 571, 307–313. [Google Scholar] [CrossRef]
- Sengupta, M.; Deb, I.; Sharma, G.D.; Kar, K.K. Human sperm and other seminal constituents in male infertile patients from arsenic and cadmium rich areas of Southern Assam. Syst. Biol. Reprod. Med. 2013, 59, 199–209. [Google Scholar] [CrossRef]
- Oguri, T.; Yoshinaga, J.; Toshima, H.; Mizumoto, Y.; Hatakeyama, S.; Tokuoka, S. Urinary inorganic arsenic concentrations and semen quality of male partners of subfertile couples in Tokyo. J. Environ. Sci. Health Part A 2016, 51, 463–466. [Google Scholar] [CrossRef]
- Jeng, H.A.; Huang, Y.L.; Pan, C.H.; Diawara, N. Role of low exposure to metals as male reproductive toxicants. Int. J. Environ. Health Res. 2014, 25, 405–417. [Google Scholar] [CrossRef]
- Pant, N.; Murthy, R.C.; Srivastava, S.P. Male reproductive toxicity of sodium arsenite in mice. Hum. Exp. Toxicol. 2004, 23, 399–403. [Google Scholar] [CrossRef]
- Reddy, P.S.; Rani, G.P.; Sainath, S.B.; Meena, R.; Supriya, C. Protective effects of N-acetylcysteine against arsenic-induced oxidative stress and reprotoxicity in male mice. J. Trace Elem. Med. Biol. 2011, 25, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Sanghamitra, S.; Hazra, J.; Upadhyay, S.N.; Singh, R.K.; Amal, R.C. Arsenic induced toxicity on testicular tissue of mice. Indian J. Physiol. Pharmacol. 2008, 52, 84–90. [Google Scholar]
- Huang, Q.; Luo, L.; Alamdar, A.; Zhang, J.; Liu, L.; Tian, M.; Eqani, S.A.; Shen, H. Integrated proteomics and metabolomics analysis of rat testis: Mechanism of arsenic-induced male reproductive toxicity. Sci. Rep. 2016, 6, 32518. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- National Institute for Health Research. PROSPERO Protocol. Available online: https://www.crd.york.ac.uk/prospero/ (accessed on 15 November 2019).
- Wells, G.S.B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 15 November 2019).
- Susko, M.L.; Bloom, M.S.; Neamtiu, I.A.; Appleton, A.A.; Surdu, S.; Pop, C.; Fitzgerald, E.F.; Anastasiu, D.; Gurzau, E.S. Low-level arsenic exposure via drinking water consumption and female fecundity—A preliminary investigation. Environ. Res. 2017, 154, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.M.; Noble, B.N.; Joya, S.A.; Ibn Hasan, M.O.S.; Lin, P.I.; Rahman, M.L.; Mostofa, G.; Quamruzzaman, Q.; Rahman, M.; Christiani, D.C.; et al. A Prospective Cohort Study Examining the Associations of Maternal Arsenic Exposure With Fetal Loss and Neonatal Mortality. Am. J. Epidemiol. 2018, 188, 347–354. [Google Scholar] [CrossRef]
- Aschengrau, A.; Zierler, S.; Cohen, A. Quality of community drinking water and the occurrence of spontaneous abortion. Arch. Environ. Health Int. J. 1989, 44, 283–290. [Google Scholar] [CrossRef]
- Banu, S.A.; Kile, M.L.; Christiani, D.C.; Qumruzzaman, Q. Study of prenatal arsenic exposure and reproductive health outcome in Bangladesh. Bangladesh J. Obstet. Gynaecol. 2016, 28, 76–81. [Google Scholar] [CrossRef]
- Bloom, M.S.; Neamtiu, I.A.; Surdu, S.; Pop, C.; Lupsa, I.R.; Anastasium, D.; Fitzgerald, E.F.; Gurzau, E.S. Consumption of low-moderate level arsenic contaminated water does not increase spontaneous pregnancy loss: A case control study. Environ. Health 2014, 13, 1–10. [Google Scholar] [CrossRef]
- Mukherjee, S.C.; Saha, K.C.; Pati, S.; Dutta, R.N.; Rahman, M.M.; Sengupta, M.K.; Ahamed, S.; Lodh, D.; Das, B.; Hossain, M.; et al. Murshidabad—One of the nine groundwater arsenic-affected districts of West Bengal, India. Part II: Dermatological, neurological, and obstetric findings. Clin. Toxicol. 2005, 43, 835–848. [Google Scholar] [CrossRef]
- Rahman, A.; Vahter, M.; Ekström, E.C.; Rahman, M.; Golam Mustafa, A.H.M.; Wahed, M.A.; Yunus, M.; Persson, L.Å. Association of arsenic exposure during pregnancy with fetal loss and infant death: A cohort study in Bangladesh. Am. J. Epidemiol. 2007, 165, 1389–1396. [Google Scholar] [CrossRef]
- Laegehaandbogen. Spontan Abort 2019. Available online: https://www.sundhed.dk/sundhedsfaglig/laegehaandbogen/obstetrik/tilstande-og-sygdomme/aborter/spontan-abort/ (accessed on 31 October 2019).
- Prager, S.M.; Micks, E.; Dalton, V.K. Pregnancy Loss (Miscarriage): Risk Factors, Etiology, Clinical Manifestations, and Diagnostic Evaluation. UpToDate. 2019. Available online: https://www.uptodate.com/contents/pregnancy-loss-miscarriage-risk-factors-etiology-clinical-manifestations-and-diagnostic-evaluation (accessed on 24 November 2019).
- Liew, Z.; Olsen, J.; Cui, X.; Ritz, B.; Arah, O.A. Bias from conditioning on live birth in pregnancy cohorts: An illustration based on neurodevelopment in children after prenatal exposure to organic pollutants. Int. J. Epidemiol. 2015, 44, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Borzsonyi, M.; Bereczky, A.; Rudnai, P.; Csanady, M.; Horvath, A. Epidemiological studies on human subjects exposed to arsenic in drinking water in Southeast Hungary. Arch. Toxicol. 1992, 66, 77–78. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Fujino, Y.; Chai, J.; Wu, K.; Xia, Y.; Li, Y.; Lv, J.; Sun, Z.; Yoshimura, T. The prevalence of subjective symptoms after exposure to arsenic in drinking water in Inner Mongolia, China. J. Epidemiol. 2003, 13, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.A.; Salim Ullah Sayed, M.H.; Barua, S.; Haque Khan, M.; Faruquee, M.H.; Jalil, A.; Abdul Hadi, S.; Kabir Talukder, H. Arsenic in drinking water and pregnancy outcomes. Environ. Health Perspect. 2001, 109, 629. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, S.; Sengupta, M.K.; Mukherjee, S.C.; Pati, S.; Mukherjee, A.; Rahman, M.M.; Hossain, M.A.; Das, B.; Nayak, B.; Pal, A.; et al. An eight-year study report on arsenic contamination in groundwater and health effects in Eruani Village, Bangladesh and an approach for its mitigation. J. Health Popul. Nutr. 2006, 24, 129–141. [Google Scholar] [PubMed]
- Milton, A.H.; Smith, W.; Rahman, B.; Hasan, Z.; Kulsum, U.; Dear, K.; Rakibuddin, M.; Ali, A. Chronic arsenic exposure and adverse pregnancy outcomes in Bangladesh. Epidemiology 2005, 16, 82–86. [Google Scholar] [CrossRef]
- Chakraborti, D.; Rahman, M.M.; Ahamed, S.; Dutta, R.N.; Pati, S.; Mukherjee, S.C. Arsenic groundwater contamination and its health effects in Patna district (capital of Bihar) in the middle Ganga plain, India. Chemosphere 2016, 152, 520–529. [Google Scholar] [CrossRef]
- Chakraborti, D.; Rahman, M.M.; Das, B.; Chatterjee, A.; Das, D.; Nayak, B.; Pal, A.; Chowdhury, U.K.; Ahmed, S.; Biswas, B.K.; et al. Groundwater arsenic contamination and its health effects in India. Hydrogeol. J. 2017, 25, 1165–1181. [Google Scholar] [CrossRef]
- Von Ehrenstein, O.S.; Guha Mazumder, D.N.; Hira-Smith, M.; Ghosh, N.; Yuan, Y.; Windham, G.; Ghosh, A.; Haque, R.; Lahiri, S.; Kalman, D.; et al. Pregnancy outcomes, infant mortality, and arsenic in drinking water in West Bengal, India. Am. J. Epidemiol. 2006, 163, 662–669. [Google Scholar] [CrossRef]
- Sen, J.; Chaudhuri, A.B.D. Arsenic exposure through drinking water and its effect on pregnancy outcome in Bengali women. Arch. Ind. Hyg. Toxicol. 2008, 59, 271–275. [Google Scholar] [CrossRef]
- Carlson, M.D.; Morrison, R.S. Study design, precision, and validity in observational studies. J. Palliat. Med. 2009, 12, 77–82. [Google Scholar] [CrossRef]
- Shih, Y.H.; Islam, T.; Hore, S.K.; Sarwar, G.; Shahriar, M.H.; Yunus, M.; Graziano, J.H.; Harjes, J.; Baron, J.A.; Parvez, F.; et al. Associations between prenatal arsenic exposure with adverse pregnancy outcome and child mortality. Environ. Res. 2017, 158, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.; Persson, L.A.; Nermell, B.; El Arifeen, S.; Ekström, E.C.; Smith, A.H.; Vahter, M. Exposure and risk of spontaneous abortion, stillbirth, and infant mortality. Epidemiology 2010, 21, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Bloom, M.S.; Louis, G.M.; Sundaram, R.; Kostyniak, P.J.; Jain, J. Associations between blood metals and fecundity among women residing in New York State. Reprod. Toxicol. 2011, 31, 158–163. [Google Scholar] [CrossRef]
- Yang, W.H.; Guo, Z.W.; Han, X.H.; Xia, Y.J.; Wu, K.G.; Li, Y.H. Influence of arsenic exposure on menstruation. Chin. J. Endem. 2013, 32, 27–29. [Google Scholar]
- Mains, M. Arsenic in Danish Groundwater and Drinking Water with Focus on Human Exposure. Master’s Thesis, Institute of Geoscience, Aarhus University, Aarhus, Denmark, 2018. [Google Scholar]
- He, Y.; Zou, L.; Luo, W.; Yi, Z.; Yang, P.; Yu, S.; Liu, N.; Ji, J.; Guo, Y.; Liu, P.; et al. Heavy metal exposure, oxidative stress and semen quality: Exploring associations and mediation effects in reproductive-aged men. Chemosphere 2020, 244, 125498. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Feng, W.; Zhou, B.; Wang, Y.X.; He, X.S.; Yang, P.; You, L.; Yue, J.; Li, Y.F.; Lu, W.Q. Urinary metal concentrations in relation to semen quality: A cross-sectional study in China. Environ. Sci. Technol. 2015, 49, 5052–5059. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Liu, L.; Wu, Y.; Wang, X.; Luo, L.; Nan, B.; Zhang, J.; Tian, M.; Shen, H. Seminal plasma metabolites mediate the associations of multiple environmental pollutants with semen quality in Chinese men. Environ. Int. 2019, 132, 105066. [Google Scholar] [CrossRef]
- Bergamo, P.; Volpe, M.G.; Lorenzetti, S.; Mantovani, A.; Notari, T.; Cocca, E.; Cerullo, S.; Di Stasio, M.; Cerino, P.; Montano, L. Human semen as an early, sensitive biomarker of highly polluted living environment in healthy men: A pilot biomonitoring study on trace elements in blood and semen and their relationship with sperm quality and RedOx status. Reprod. Toxicol. 2016, 66, 1–9. [Google Scholar] [CrossRef]
- Sukhn, C.; Awwad, J.; Ghantous, A.; Zaatari, G. Associations of semen quality with non-essential heavy metals in blood and seminal fluid: Data from the Environment and Male Infertility (EMI) study in Lebanon. J. Assist. Reprod. Genet. 2018, 35, 1691–1701. [Google Scholar] [CrossRef]
- Wang, Y.X.; Wang, P.; Feng, W.; Liu, C.; Yang, P.; Chen, Y.J.; Sun, L.; Sun, Y.; Yue, J.; Gu, L.J.; et al. Relationships between seminal plasma metals/metalloids and semen quality, sperm apoptosis and DNA integrity. Environ. Pollut. 2017, 224, 224–234. [Google Scholar] [CrossRef]
- Liu, P.; Yuan, G.; Zhou, Q.; Liu, Y.; He, X.; Zhang, H.; Guo, Y.; Wen, Y.; Huang, S.; Ke, Y.; et al. The association between metal exposure and semen quality in Chinese males: The mediating effect of androgens. Environ. Pollut. 2020, 264, 113975. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhong, Y.; Jiang, X.; Wang, C.; Zuo, Z.; Sha, A. Seminal plasma metals concentration with respect to semen quality. Biol. Trace Elem. Res. 2012, 148, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Xu, W.; Zhang, J.; Chen, M.; Martin, F.L.; Xia, Y.; Liu, L.; Dong, S.; Zhu, Y.G. Urinary metabolic biomarkers link oxidative stress indicators associated with general arsenic exposure to male infertility in a han chinese population. Environ. Sci. Technol. 2013, 47, 8843–8851. [Google Scholar] [CrossRef]
- Eliasson, R. Biochemical analysis of human semen. Int. J. Androl. 1982, 128, 1144. [Google Scholar]
- Concha, G.; Vogler, G.; Nermell, B.; Vahter, M. Intra-individual variation in the metabolism of inorganic arsenic. Int. Arch. Occup. Environ. Health 2002, 75, 576–580. [Google Scholar] [CrossRef]
- Carlin, D.J.; Naujokas, M.F.; Bradham, K.D.; Cowden, J.; Heacock, M.; Henry, H.F.; Lee, J.S.; Thomas, D.J.; Thompson, C.; Tokar, E.J.; et al. Arsenic and Environmental Health: State of the Science and Future Research Opportunities. Environ. Health Perspect. 2016, 124, 890–899. [Google Scholar] [CrossRef]
- Gagnon, C. The role of environmental toxins in unexplained male infertility. Semin. Reprod. Med. 1988, 6, 369–376. [Google Scholar] [CrossRef]
- Concha, G.; Vogler, G.; Lezcano, D.; Nermell, B.; Vahter, M. Exposure to inorganic arsenic metabolites during early human development. Toxicol. Sci. 1998, 44, 185–190. [Google Scholar] [CrossRef]
- Fort, M.; Cosín-Tomás, M.; Grimalt, J.O.; Querol, X.; Casas, M.; Sunyer, J. Assessment of exposure to trace metals in a cohort of pregnant women from an urban center by urine analysis in the first and third trimesters of pregnancy. Environ. Sci. Pollut. Res. 2014, 21, 9234–9241. [Google Scholar] [CrossRef] [PubMed]
- Bloom, M.S.; Neamtiu, I.A.; Surdu, S.; Pop, C.; Anastasiu, D.; Appleton, A.A.; Fitzgerald, E.F.; Gurzau, E.S. Low level arsenic contaminated water consumption and birth outcomes in Romania-An exploratory study. Reprod. Toxicol. 2016, 59, 8–16. [Google Scholar] [CrossRef] [PubMed]
| Author, Year, Country | Study Design | Study Population (Size and Selection) | Exposure Assessment | Outcome | Confounding Control | Main Results 1 | Effect of Arsenic Exposure 2 | Total NOS-Score |
|---|---|---|---|---|---|---|---|---|
| Ahmed et. al., 2019, Bangladesh | Cohort study, prospectively collected information on exposure | Total: 1616 pregnant women | 1st quartile: 0.05–0.89 µg/L 2nd quartile: 0.09–2.00 µg/L 3rd quartile: 2.10–34.00 µg/L 4th quartile: 45.00–1400.00 µg/L | Fetal loss (a fetus that never showed signs of life outside the womb) | Maternal age (18–34 years and >35 years) Monthly family income Educational level Gravidity Clinic location | No significant difference in the occurrence of fetal loss amongst the four exposure groups (p-value = 0.208) | →/(↑) | 8 |
| A non-linear exposure-response relationship, varying across the pregnancy | ||||||||
| Case: 203 Women with fetal loss Comparison: Women with completed pregnancy | Urinary arsenic concentrations (not specified) | Gestational age 9–12 (lowest HR): HR = 0.81 (95%CI: 0.64–1.02) Gestational age 25–28 (highest HR): HR = 1.39 (95%CI: 1.01–1.75) | ||||||
| Similar results when maternal urinary arsenic concentration was used in the model | ||||||||
| Aschengrau et. al., 1989, USA | Case-control study | Total: 1677 | None (undetectable) Low: 0.8–1.3 µg/L High: 1.4–1.9 µg/L | Spontaneous abortion (<28 weeks) | Maternal age (<30 years and >30 years) Educational level History of prior abortions | Non-significant, positive association between arsenic levels and risk of spontaneous abortion. All subjects adjusted: Low: OR = 1.1 (95%CI: 0.6–1.8) High: OR = 1.5 (95%CI: 0.4–4.7) | (↑) | 9 |
| Case: 286 spontaneous abortions Control: 1391 completed pregnancies | ||||||||
| Banu et. al., 2013, Bangladesh | Cohort study, prospectively collected information on exposure | Total: 421 pregnant women | Average for all participants: 36.1 µg/L (SD = 83.7 and range ≤ 1–880 µg/L) | Spontaneous abortion or miscarriage (not specified) | None | 30 pregnancies resulted in spontaneous abortion or miscarriage (7.1%) | Not applicable | 4 |
| Case: 30 spontaneous abortions Comparison: None | ||||||||
| Bloom et. al., 2014, Romania | Case-control study | Total: 300 | Case: Average: 4.06 µg/L | Spontaneous abortion (<21 weeks). | Maternal age Smoking during pregnancy Educational level Prenatal vitamin use | No increased risk of spontaneous pregnancy loss in association with continuous inorganic arsenic exposure | → | 8 |
| Case: 150 spontaneous abortions Control: 150 ongoing pregnancies | Control: Average: 4.32 µg/L | Average inorganic arsenic levels: OR = 0.98 (95%CI: 0.96–1.01) | ||||||
| Mukherjee et. al., 2005, India | Cohort study, retrospectively collected information on exposure | Total: 24 | Case: Group A: 284–400 µg/L Group B: 401–1474 µg/L | Spontaneous abortion (not specified) | None | The number of spontaneous abortions increased with increasing arsenic levels in drinking water | Not applicable | 2 |
| Case: 17 (high arsenic exposure) Comparison: 7 (low arsenic exposure) | Comparison: Group C: <3 µg/L | Group A: 2 spontaneous abortion (95/1000 live births) Group B: 8 spontaneous abortion (182/1000 live births) Group C: 1 spontaneous abortion (55/1000 live births) | ||||||
| Rahman et. al., 2007, Bangladesh | Cohort study, prospectively collected information on exposure | Total: 29,134 pregnant women | 1st quintile (reference): <10 µg/L 2nd quintile: 10–166 µg/L 3rd quintile: 167–276 µg/L 4th quintile: 277–408 µg/L 5th quintile: ≥409 µg/L | Fetal loss (early < 28 weeks, late > 28 weeks) | Maternal age Parity Educational level Socioeconomic status | A tendency of increased risk of fetal loss for higher quintiles of water arsenic concentrations, although only significant for the 4th quintile (pooled results): 4th quintile: RR = 1.14 (95%CI: 1.01–1.30) If exposure was dichotomized below and above 50 µg/L: RR = 1.14 (CI95%: 1.04–1.25) | (↑) | 6 |
| Case: 2444 fetal losses Comparison: Not specified | ||||||||
| Susko et. al., 2017, Romania | Cohort study, retrospectively collected information on exposure | Total: 287 previously pregnant women | Planned: Average: 4.63 µg/L Peak: 5.55 µg/L Estimated daily: 7.29 µg/L | Time to pregnancy | Maternal age Cigarette smoking Educational level | No main effects for low-level drinking water arsenic on women’s fecundity | →/(↑) | 6 |
| Case: 94 planned pregnancies Comparison: 193 unplanned pregnancies | Unplanned: Average: 4.11 µg/L Peak: 4.59 µg/L Estimated daily: 5.75 µg/L | For women with a longer time to pregnancy, modestly lower pregnancy probabilities were observed with higher arsenic exposure |
| Author, Year, Country | Study Design | Study Population (Size and Selection) | Exposure Assessment | Outcome | Confounding Control (Smoking Status and Other (Alcohol Consumption, BMI, Age)) | Main Results | Effect of Arsenic Exposure 1 | Total NOS- Score |
|---|---|---|---|---|---|---|---|---|
| Sengupta, 2013, India | Case-control study | Total: 100 men | Mean drinking water arsenic concentrations: Cases: Azoospermia: 2585 µg/L Oligospermia: 2042 µg/L | Sperm concentration: <20 × 106/mL (azoospermia and oligospermia) | Smoking Alcohol Tobacco chewing Dietary habits | Mean drinking water arsenic and mean seminal arsenic content were both significantly higher amongst cases compared to controls (p < 0.05). | ↑ | 6 |
| Cases: 68 (sperm concentration <20 × 106/mL) Control: 32 (sperm concentration >20 × 106/mL) | Control: Normozoospermia: 149 µg/L | Mean drinking water arsenic levels were positively correlated (r = 0.867) with mean seminal plasma arsenic levels. | ||||||
| >20 × 106/mL (normozoospermia) | Arsenic content in drinking water and total sperm count was inversely related (r = −0.91) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barsøe, I.M.; Ebdrup, N.H.; Clausen, H.S.; Lyngsø, J.; Schullehner, J.; Ramlau-Hansen, C.H.; Bay, B.; Knudsen, U.B. Drinking Water Arsenic and Adverse Reproductive Outcomes in Men and Women: A Systematic PRISMA Review. Water 2021, 13, 1885. https://doi.org/10.3390/w13141885
Barsøe IM, Ebdrup NH, Clausen HS, Lyngsø J, Schullehner J, Ramlau-Hansen CH, Bay B, Knudsen UB. Drinking Water Arsenic and Adverse Reproductive Outcomes in Men and Women: A Systematic PRISMA Review. Water. 2021; 13(14):1885. https://doi.org/10.3390/w13141885
Chicago/Turabian StyleBarsøe, Ida M., Ninna H. Ebdrup, Hannah S. Clausen, Julie Lyngsø, Jörg Schullehner, Cecilia H. Ramlau-Hansen, Bjørn Bay, and Ulla B. Knudsen. 2021. "Drinking Water Arsenic and Adverse Reproductive Outcomes in Men and Women: A Systematic PRISMA Review" Water 13, no. 14: 1885. https://doi.org/10.3390/w13141885






