Methane Production and Oxidation in Mangrove Soils Assessed by Stable Isotope Mass Balances
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Theoretical Considerations
2.3. Sampling Procedure
2.4. Isotopic Fractionation during Methanogenesis and Relative Contribution of Pathways
2.5. Mangrove Wetland Characterizations
2.6. Statistical Analyses
3. Results
3.1. Mangrove Forest and Soil Properties
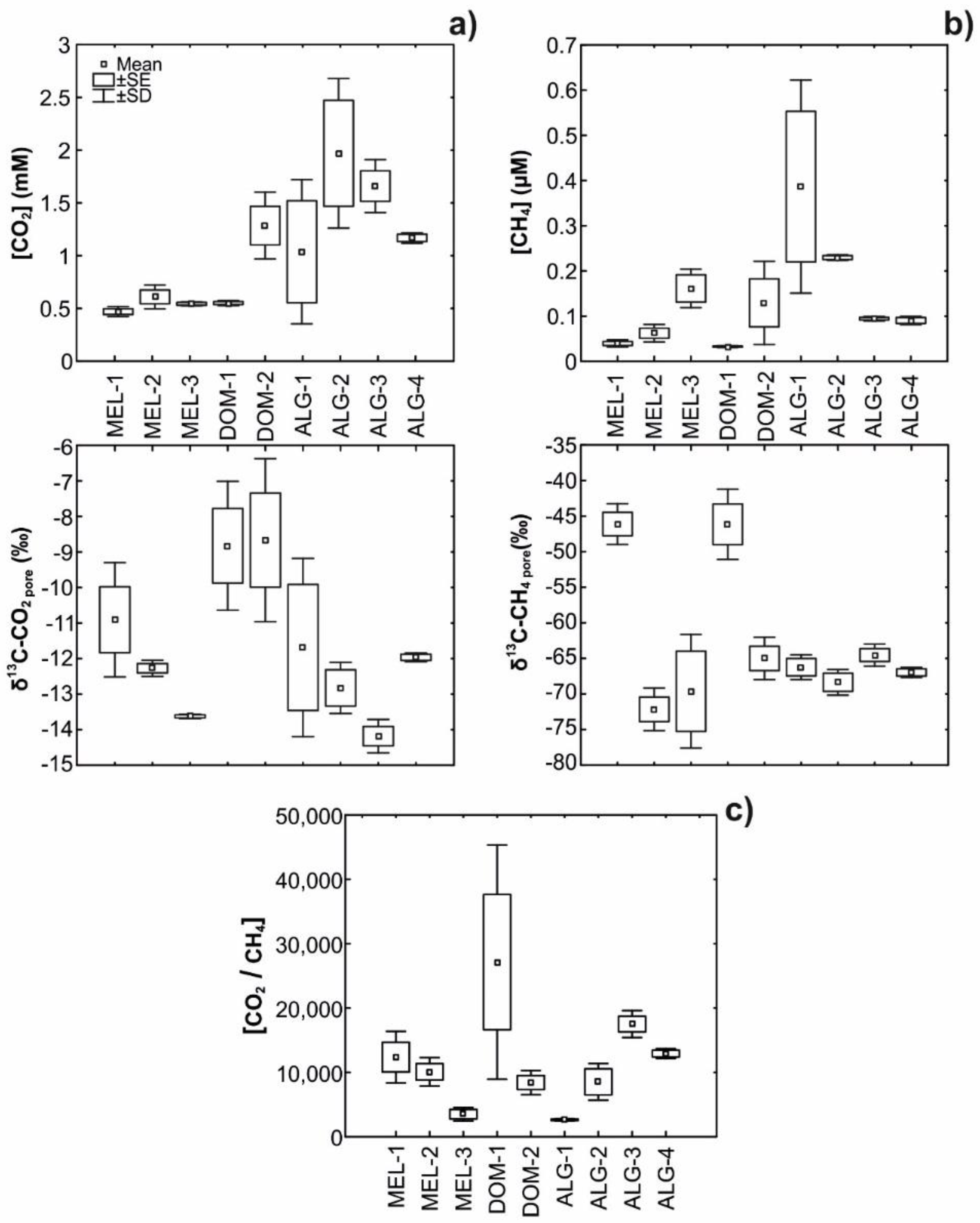
3.2. Field Pore Water CO2 and CH4 Concentrations and δ13C Signatures
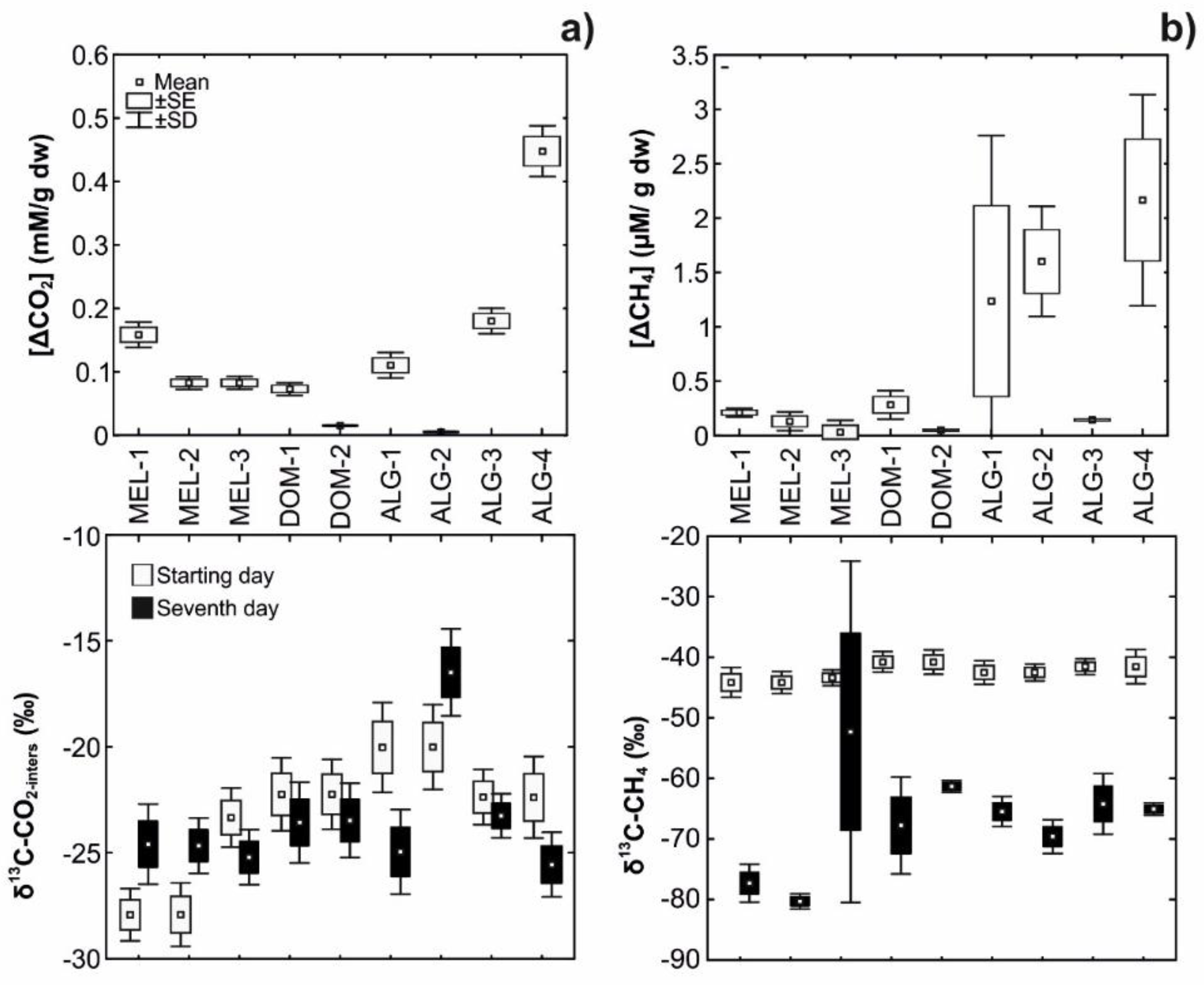
3.3. Laboratory Incubations
3.4. Isotope Mass Balances
3.5. Tidal, Forest Structure, and Water Quality Influences on Methane Production
4. Discussion
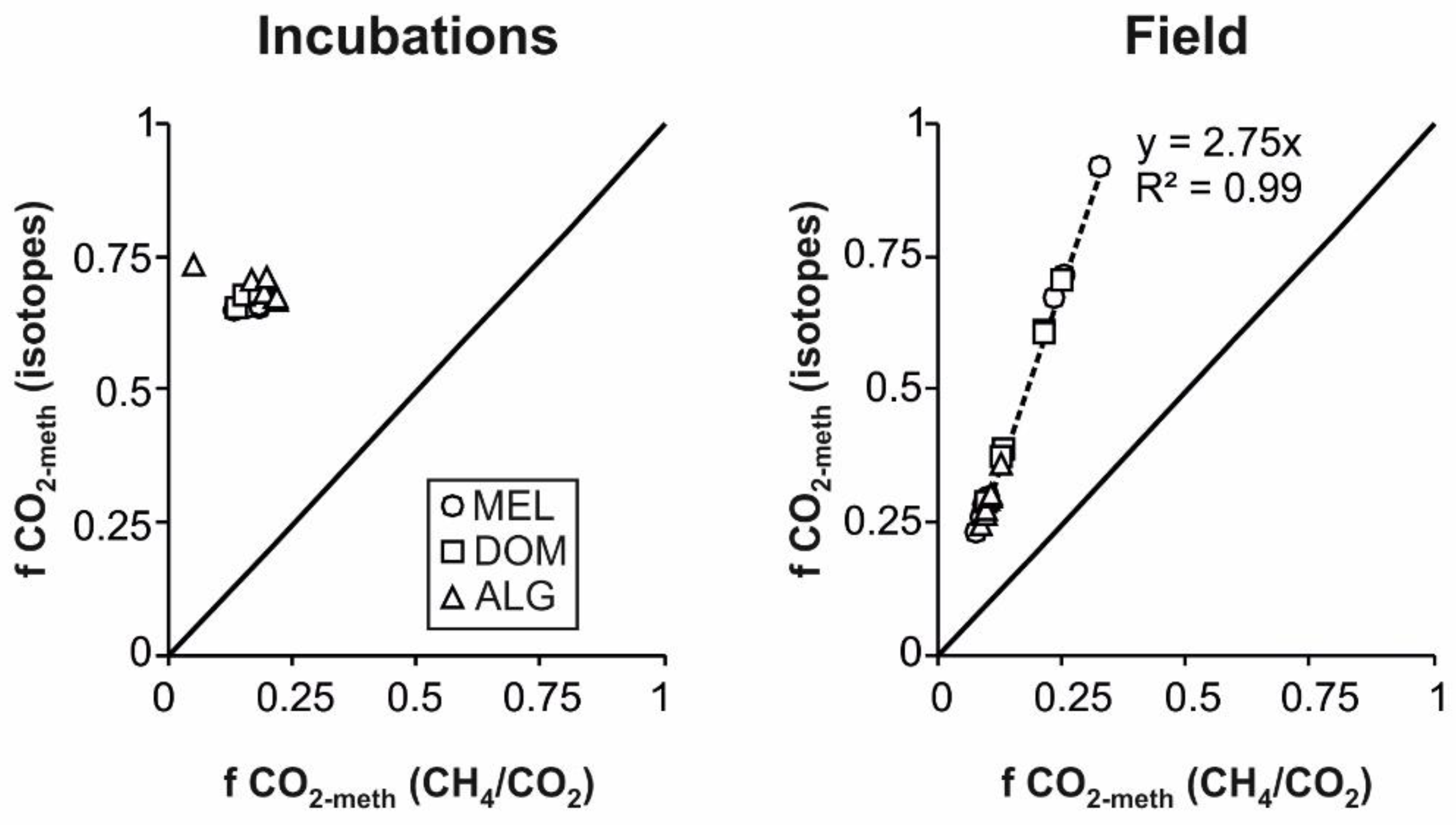
4.1. Validity of the Isotope–Mass Balance Approach to Estimate Methane Production in Mangroves
4.2. Dominance of Aerobic versus Anaerobic Respiration in Mangrove Soils
4.3. Methanogenesis Pathways and Shifts Derived by Shrimp-Farming
4.4. Alternative Non-Competitive Substrates for Methanogenesis in Mangroves
4.5. Sulfate Reduction Coupled to AOM in Mangroves
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Denman, K.L.; Brasseur, G.; Chidthaisong, A.; Ciais, P.; Cox, P.M.; Dickinson, R.E.; Hauglustaine, D.; Heinze, C.; Holland, E.; Jacob, D.; et al. Couplings Between Changes in the Climate System and Biogeochemistry. In Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Solomon, S., Qin, D., Manning, M., Chen, Z., Marquis, M., Averyt, K.B., Tignor, M., Miller, H.L., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 499–587. [Google Scholar]
- Chasar, L.S.; Chanton, J.P.; Glaser, P.H.; Siegel, D.I. Methane concentration and stable isotope distribution as evidence of rhizospheric processes: Comparison of a fen and bog in the Glacial Lake Agassiz Peatland Complex. Ann. Bot. 2000, 86, 655–663. [Google Scholar] [CrossRef] [Green Version]
- Segarra, K.E.A.; Comerford, C.; Slaughter, J.; Joye, S.B. Impact of electron acceptor availability on the anaerobic oxidation of methane in coastal freshwater and brackish wetland sediments. Geochim. Cosmochim. Ac. 2013, 115, 15–30. [Google Scholar] [CrossRef]
- Verma, A.; Subramanian, V.; Ramesh, R. Methane emissions from a coastal lagoon: Vembanad Lake, West Coast, India. Chemosphere 2002, 47, 883–889. [Google Scholar] [CrossRef]
- Kreuzwieser, J.; Buchholz, J.; Rennenberg, H. Emission of methane and nitrous oxide by Australian mangrove ecosystems. Plant Biol. 2003, 5, 423–431. [Google Scholar] [CrossRef]
- Giani, L.; Dittrich, K.; Marstfeld-hartmann, A.; Peters, G. Methanogenesis in salt marsh soils of the North Sea coast of Germany. Eur. J. Soil Sci. 1996, 47, 175–182. [Google Scholar] [CrossRef]
- Alongi, D.M.; Wattayakorn, G.; Pfitzner, J.; Tirendi, F.; Zagorskis, I.; Brunskill, G.J.; Davidson, A.; Clough, B.F. Organic carbon accumulation and metabolic pathways in sediments of mangrove forests in southern Thailand. Mar. Geol. 2001, 179, 85–103. [Google Scholar] [CrossRef]
- Furtado, A.L.S.; Casper, P.; Assis, F. Methanogenesis in an impacted and two dystrophic coastal lagoons (Macaé, Brazil). Braz. Arch. Biol. Technol. 2002, 45, 195–202. [Google Scholar] [CrossRef]
- Sotomayor, D.; Corredor, J.E.; Morell, J.M. Methane flux from mangrove sediments along the Southwestern coast of Puerto Rico. Estuaries 1994, 17, 140–147. [Google Scholar] [CrossRef]
- Allen, D.E.; Dalal, R.C.; Rennenberg, H.; Meyer, R.L.; Reeves, S.; Schmidt, S. Spatial and temporal variation of nitrous oxide and methane flux between subtropical mangrove sediments and the atmosphere. Soil Biol. Biochem. 2007, 39, 622–631. [Google Scholar] [CrossRef]
- Biswas, H.; Mukhopadhyay, S.K.; Sen, S.; Jana, T.K. Spatial and temporal patterns of methane dynamics in the tropical mangrove dominated estuary, NE coast of bay of Bengal, India. J. Mar. Sys. 2007, 68, 55–64. [Google Scholar] [CrossRef]
- Linto, N.; Barnes, J.; Ramachandran, R.; Divia, J.; Purvaja, R.; Upstill-Goddard, R.C. Carbon dioxide and methane emissions from mangrove-associated waters of the Andaman Islands, Bay of Bengal. Estuar. Coast. 2014, 37, 381–398. [Google Scholar] [CrossRef]
- Call, M.; Maher, D.T.; Santos, I.R.; Ruiz-Halpern, S.; Mangion, P.; Sanders, C.J.; Erler, D.V.; Oakes, J.M.; Rosentreter, J.; Murray, R.; et al. Spatial and temporal variability of carbon dioxide and methane fluxes over semi-diurnal and spring–neap–spring timescales in a mangrove creek. Geochim. Cosmochim. Ac. 2015, 150, 211–225. [Google Scholar] [CrossRef] [Green Version]
- Lee, R.; Porubsky, W.; Feller, I.; McKee, K.; Joye, S. Pore water biogeochemistry and soil metabolism in dwarf red mangrove habitats (Twin Cays, Belize). Biogeochemistry 2008, 87, 181–198. [Google Scholar] [CrossRef]
- Chuang, P.-C.; Young, M.B.; Dale, A.W.; Miller, L.G.; Herrera-Silveira, J.A.; Paytan, A. Methane and sulfate dynamics in sediments from mangrove-dominated tropical coastal lagoons, Yucatán, Mexico. Biogeosciences 2016, 13, 2981–3001. [Google Scholar] [CrossRef] [Green Version]
- Cui, M.; Ma, A.; Qi, H.; Zhuang, X.; Zhuang, G. Anaerobic oxidation of methane: An “active” microbial process. Microbiologyopen 2015, 4, 1–11. [Google Scholar] [CrossRef]
- Segarra, K.E.A.; Schubotz, F.; Samarkin, V.; Yoshinaga, M.Y.; Hinrichs, K.-U.; Joye, S.B. High rates of anaerobic methane oxidation in freshwater wetlands reduce potential atmospheric methane emissions. Nat. Commun. 2015, 6, 7477. [Google Scholar] [CrossRef]
- Chanton, J.P.; Chasar, L.; Glaser, P.H.; Siegel, D.I. Carbon and hydrogen isotopic effects in microbial methane from terrestrial environments. In Stable Isotopes and Biosphere—Atmosphere Interactions, Physiological Ecology Series; Flanagan, L.B., Ehleringer, J.R., Pataki, D.E., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 85–105. [Google Scholar]
- Corbett, J.E.; Tfaily, M.M.; Burdige, D.J.; Cooper, W.T.; Glaser, P.H.; Chanton, J.P. Partitioning pathways of CO2 production in peatlands with stable carbon isotopes. Biogeochemistry 2013, 114, 327–340. [Google Scholar] [CrossRef]
- Shoemaker, J.K.; Schrag, D.P. Subsurface characterization of methane production and oxidation from a New Hampshire wetland. Geobiology 2010, 8, 234–243. [Google Scholar] [CrossRef]
- Whiting, G.J.; Chanton, J.P. Primary production control of methane emission from wetlands. Nature 1993, 364, 794–795. [Google Scholar] [CrossRef]
- Coles, J.R.P.; Yavitt, J.B. Control of methane metabolism in a forested Northern Wetland, New York State, by aeration, substrates, and peat size fractions. Geomicrobiol. J. 2002, 19, 293–315. [Google Scholar] [CrossRef]
- Bastviken, D.; Cole, J.C.; Pace, M.; Tranvik, L. Methane emissions from lakes: Dependence of lake characteristics, two regional assessments, and a global estimate. Global Biogeochem. Cy. 2004, 18, GB4009. [Google Scholar] [CrossRef]
- West, W.E.; Creamer, K.P.; Jones, S.E. Productivity and depth regulate lake contributions to atmospheric methane. Limnol. Oceanogr. 2016, 61, S51–S61. [Google Scholar] [CrossRef] [Green Version]
- DelSontro, T.; Boutet, L.; StPierre, A.; del Giorgio, P.A.; Prairie, Y.T. Methane ebullition and diffusion from northern ponds and lakes regulated by the interaction between temperature and system productivity. Limnol. Oceanogr. 2016, 61, S62–S77. [Google Scholar] [CrossRef]
- DelSontro, T.; Beaulieu, J.J.; Downing, J.A. Greenhouse gas emissions from lakes and impoundments: Upscaling in the face of global change. Limnol. Oceanogr. Lett. 2018, 3, 64–75. [Google Scholar] [CrossRef]
- Kristensen, E.; Mangion, P.; Tang, M.; Flindt, M.R.; Holmer, M.; Ulomi, S. Microbial carbon oxidation rates and pathways in sediments of two Tanzanian mangrove forests. Biogeochemistry 2011, 103, 143–158. [Google Scholar] [CrossRef]
- Chen, G.C.; Tam, N.F.Y.; Wong, Y.S.; Ye, Y. Effect of wastewater discharge on greenhouse gas fluxes from mangrove soils. Atmos. Environ. 2011, 45, 1110–1115. [Google Scholar] [CrossRef]
- Chen, G.; Chen, B.; Yu, D.; Tam, N.F.Y.; Ye, Y.; Chen, S. Soil greenhouse gas emissions reduce the contribution of mangrove plants to the atmospheric cooling effect. Environ. Res. Lett. 2016, 11, 124019. [Google Scholar] [CrossRef]
- Casillas-Hernández, R.; Nolasco-Soria, H.; García-Galano, T.; Carrillo-Farnes, O.; Páez-Osuna, F. Water quality, chemical fluxes and production in semi-intensive Pacific white shrimp (Litopenaeus vannamei) culture ponds utilizing two different feeding strategies. Aquac. Eng. 2007, 36, 105–114. [Google Scholar] [CrossRef]
- Barraza-Guardado, R.H.; Arreola-Lizárraga, J.A.; López-Torres, M.A.; Casillas-Hernández, R.; Miranda-Baeza, A.; Magallón-Barrajas, F.; Ibarra-Gómez, C. Effluents of shrimp farms and its influence on the coastal ecosystems of Bahía de Kino Mexico. Sci. World J. 2013, 2013, 306370. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, J.M.; Pérez, M.; Romero, J. Effects of fish farm loadings on seagrass (Posidonia oceanica) distribution, growth and photosynthesis. Mar. Pollut. Bull. 2001, 42, 749–760. [Google Scholar] [CrossRef]
- Páez-Osuna, F.; Gracia, A.; Flores-Verdugo, F.; Lyle-Fritch, L.P.; Alonso-Rodríguez, R.; Roque, A.; Ruiz-Fernández, A.C. Shrimp aquaculture development and the environment in the Gulf of California ecoregion. Mar. Pollut. Bull. 2003, 46, 806–815. [Google Scholar] [CrossRef]
- Serrano-Grijalva, L.; Sánchez-Carrillo, S.; Angeler, D.G.; Sanchez-Andres, R.; Alvarez-Cobelas, M. Effects of shrimpfarm effluents on the food web structure in subtropical coastal lagoons. J. Exp. Mar. Biol. Ecol. 2011, 402, 65–74. [Google Scholar] [CrossRef] [Green Version]
- Aschenbroich, A.; Marchand, C.; Molnar, N.; Deborde, J.; Hubas, C.; Rybarczyk, H.; Meziane, T. Spatio-temporal variations in the composition of organic matter in surface sediments of a mangrove receiving shrimp farm effluents (New Caledonia). Sci. Total Environ. 2015, 512–513, 296–307. [Google Scholar] [CrossRef]
- Nóbrega, G.N.; Ferreira, T.O.; Siqueira, M.; Hermano, N.; Queiroz, M.; Artur, A.G.; Mendonça, E.D.S.; Silva, E.D.O.; Oterof, X.L. Edaphic factors controlling summer (rainy season) greenhouse gas emissions (CO2 and CH4) from semiarid mangrove soils (NE-Brazil). Sci. Total Environ. 2016, 542, 685–693. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Carrillo, S.; Sánchez-Andrés, R.; Alatorre, L.C.; Angeler, D.G.; Álvarez-Cobelas, M.; Arreola-Lizárraga, J.A. Nutrient fluxes in a semi-arid microtidal mangrove wetland in the Gulf of California. Est. Coast. Shelf Sci. 2009, 82, 654–662. [Google Scholar] [CrossRef]
- Steinbeck, J.; Ricketts, E.F. The Sea of Cortez. A Leisurely Journal of Travel and Research; Viking Press: New York, NY, USA, 1951. [Google Scholar]
- Almada-Bay, I. Sonora 2000 a Debate: Problemas y Soluciones, Riesgos y Oportunidades; El Colegio de Sonora: Mexico DF, México, 2000. [Google Scholar]
- Sánchez-Andrés, R.; Sánchez-Carrillo, S.; Alatorre, L.C.; Cirujano, S.; Álvarez-Cobelas, M. Litterfall dynamics and nutrient decomposition of arid mangroves in the Gulf of California: Their role sustaining ecosystem heterotrophy. Est. Coast. Shelf Sci. 2010, 89, 191–199. [Google Scholar] [CrossRef]
- Teh, Y.A.; Silver, W.L.; Conrad, M.E.; Borglin, S.E.; Carlson, C.M. Carbon isotope fractionation by methane-oxidizing bacteria in tropical rain forest soils. J. Geophys. Res. Biogeosci. 2006, 111, G02001. [Google Scholar] [CrossRef]
- Holler, T.; Wegener, G.; Knittel, K.; Boetius, A.; Brunner, B.; Kuypers, M.M.; Widdel, F. Substantial 13C/12C and D/H fractionation during anaerobic oxidation of methane by marine consortia enriched in vitro. Environ. Microbiol. Rep. 2009, 1, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Alperin, M.J.; Reeburgh, W.S.; Whiticar, M.J. Carbon and hydrogen isotope fractionation resulting from anaerobic methane oxidation. Global Biogeochem. Cy. 1988, 2, 279–288. [Google Scholar] [CrossRef]
- Grasshoff, K.; Kremlingl, K.; Ehrhardt, M. (Eds.) Methods of Seawater Analysis, 3rd ed.; Wiley-VCH: Weinheim, Germany, 1999. [Google Scholar]
- Jancsó, G. Interpretation of isotope effects on the solubility of gases. Nukleonika 2002, 243, 328–342. [Google Scholar] [CrossRef]
- Bacsik, Z.; Lopes, J.N.C.; Gomes, M.F.C.; Jancso, G.; Mink, J.; Padua, A.A.H. Solubility isotope effects in aqueous solutions of methane. J. Chem. Phys. 2002, 116, 10816–10824. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Tigreros Kodovska, F.; Sparrow, K.J.; Yvon-Lewis, S.A.; Paytan, A.; Dimova, N.T.; Lecher, A.; Kessler, J.D. Dissolved methane and carbon dioxide fluxes in Subarctic and Arctic regions: Assessing measurement techniques and spatial gradients. Earth Planet. Sc. Lett. 2016, 436, 43–55. [Google Scholar] [CrossRef] [Green Version]
- Knox, M.; Quay, P.D.; Wilbur, D. Kinetic isotopic fractionation during air-water gas transfer of O2, N2, CH4, and H2. J. Geophys. Res. 1992, 97, 20335–20343. [Google Scholar] [CrossRef]
- Conrad, R.; Chan, O.C.; Claus, P.; Casper, P. Characterization of methanogenic Archaea and stable isotope fractionation during methane production in the profundal sediment of an oligotrophic lake (Lake Stechlin, Germany). Limnol. Oceanogr. 2007, 52, 1393–1406. [Google Scholar] [CrossRef]
- Cintron, G.; Lugo, A.E.; Pool, D.J.; Morris, G. Mangroves of arid environments in Puerto Rico and adjacent islands. Biotropica 1978, 10, 110–121. [Google Scholar] [CrossRef]
- Holdridge, L.; Grenke, R.; Hathaway, W.C.; Liang, W.H.; Tosi, T. Forest Environments in Tropical Life Zone: A Pilot Study; Pergamon Press: New York, NY, USA, 1971. [Google Scholar]
- Howard, R.F.; Singer, M.J. Measuring forest soil bulk density using irregular hole, paraffin clod, and air permeability. Forest Sci. 1981, 27, 316–322. [Google Scholar]
- Bouyoucos, G.J. Hydrometer method improved for making particle size analyses of soils. Agron. J. 1962, 54, 464–465. [Google Scholar] [CrossRef]
- Kristensen, E.; Bouillon, S.; Dittmar, T.; Marchand, C. Organic carbon dynamics in mangrove ecosystems: A review. Aquat. Bot. 2008, 89, 201–219. [Google Scholar] [CrossRef] [Green Version]
- Jørgensen, B.B.; Kasten, S. Sulfur cycling and methane oxidation. In Marine Geochemistry; Schulz, H.D., Zabel, M., Eds.; Springer: Berlin, Germany, 2006; pp. 271–310. [Google Scholar]
- Keuskamp, J.A.; Feller, I.C.; Laanbroek, H.J.; Verhoeven, J.T.A.; Hefting, M.M. Short- and long-term effects of nutrient enrichment on microbial exoenzyme activity in mangrove peat. Soil Biol. Biogeochem. 2015, 81, 38–47. [Google Scholar] [CrossRef] [Green Version]
- Brye, K.R.; Rogers, C.W.; Smartt, A.D.; Norman, R.J. Soil texture effects on methane emissions from direct-seeded, delayed-flood rice production in Arkansas. Soil Sci. 2013, 178, 519–529. [Google Scholar] [CrossRef]
- Roversi, F.; van Maanen, B.; Colonna Rosman, P.C.; Freitas Neves, C.; Scudelari, A.C. N umerical modeling evaluation of the impacts of shrimp farming operations on long-term coastal lagoon morphodynamics. Estuar. Coast. 2020, 43, 1853–1872. [Google Scholar] [CrossRef]
- Villa, J.A.; Mitsch, W.J. Carbon sequestration in different wetland plant communities in Southwest Florida. Ecosyst. Serv. Managem. 2015, 11, 17–28. [Google Scholar]
- Pereyra, A.S.; Mitsch, W.J. Methane emissions from freshwater cypress (Taxodium distichum) swamp soils with natural and impacted hydroperiods in Southwest Florida. Ecol. Eng. 2018, 114, 46–56. [Google Scholar] [CrossRef]
- Hernández, M.E.; Junca-Gómez, D. Carbon stocks and greenhouse gas emissions (CH4 and N2O) in mangroves with different vegetation assemblies in the central coastal plain of Veracruz Mexico. Sci. Total Environ. 2020, 741, 140276. [Google Scholar] [CrossRef] [PubMed]
- Cabezas, A.; Mitsch, W.J.; MacDonnell, C.; Zhang, L.; Bydałeka, F.; Lasso, A. Methane emissions from mangrove soils in hydrologically disturbed and reference mangrove tidal creeks in Southwest Florida. Ecol. Eng. 2018, 114, 57–65. [Google Scholar] [CrossRef]
- Conrad, R.; Claus, P.; Casper, P. Characterization of stable isotope fractionation during methane production in the sediment of a eutrophic lake, Lake Dagow, Germany. Limnol. Oceanog. 2009, 54, 457–471. [Google Scholar] [CrossRef]
- Hornibrook, E.R.C.; Longstaffe, F.J.; Fyfe, W.S. Evolution of stable carbon isotope compositions for methane and carbon dioxide in freshwater wetlands and other anaerobic environments. Geochim. Cosmochim. Acta 2000, 64, 1013–1027. [Google Scholar] [CrossRef]
- Kumar, A.; Sharma, M.P. Estimation of green house gas emissions from Koteshwarhydropower reservoir, India. Environ. Monit. Assess. 2017, 189, 240. [Google Scholar] [CrossRef]
- Anaya-Rosas, R.E. Cultivo de Camarón Blanco, Litopenaeus Vannamei, Boone (1931), en Sistema Cerrado a Alta Densidad. MSc Thesis, CICESE, Ensenada, Mexico. 2005. Available online: https://cicese.repositorioinstitucional.mx/jspui/bitstream/1007/1144/1/167251.pdf (accessed on 3 July 2021).
- Otoshi, C.; Tang, L.R.; Dagdagan, D.V.; Holl, C.M.; Tallamy, C.J.; Moss, D.R.; Acre, S.M.; Moss, S.M. Super-intensive growout of the Pacific white shrimp Litopenaeus vannamei: Recent advances at the Oceanic Institute. In Proceedings of the 6th International Conference on Recirculating Aquaculture, Tokyo, Japan, 11–12 November 2021; Rakestraw, T., Douglas, L., Flick, G., Eds.; Virginia Polytechnic Institute and State University: Blacksburg, VA, USA, 2006; pp. 1–5. [Google Scholar]
- Beardsley, C.; Moss, S.; Malfatti, F.; Azam, F. Quantitative role of shrimp fecal bacteria in organic matter fluxes in a recirculating shrimp aquaculture system. FEMS Microbiol. Ecol. 2011, 77, 134–145. [Google Scholar] [CrossRef] [Green Version]
- Oremland, R.S.; Polcin, S. Methanogenesis and sulfate reduction—Competitive and non competitive substrates in estuarine sediments. Appl. Environ. Microb. 1982, 44, 1270–1276. [Google Scholar] [CrossRef] [Green Version]
- Parkes, R.J.; Brock, F.; Banning, N.; Hornibrook, E.R.C.; Roussel, E.G.; Weightman, A.J.; Fry, J.C. Changes in methanogenic substrate utilization and communities with depth in a salt-marsh, creek sediment in southern England. Estuar. Coast. Shelf. S. 2012, 96, 170–178. [Google Scholar] [CrossRef]
- Conrad, R.; Claus, P. Contribution of methanol to the production of methane and its C-13-isotopic signature in anoxic rice field soil. Biogeochemistry 2005, 73, 381–393. [Google Scholar] [CrossRef]
- Penger, J.; Conrad, R.; Blaser, M. Stable carbon isotope fractionation by methylotrophic methanogenic archaea. Appl. Environ. Microbiol. 2012, 78, 7596–7602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoehler, T.M.; Alperin, M.J.; Albert, D.B.; Martens, C.S. Apparent minimum free energy requirements for methanogenic Archaea and sulfate-reducing bacteria in an anoxic marine sediment. FEMS Microbiol. Ecol. 2001, 38, 33–41. [Google Scholar] [CrossRef]
- Nauhaus, K.; Treude, T.; Boetius, A.; Krüger, M. Environmental regulation of the anaerobic oxidation of methane: A comparison of ANME-1 and ANME-2 communities. Environ. Microbiol. 2005, 71, 98–106. [Google Scholar] [CrossRef]
- Meulepas, R.J.W.; Jagersma, C.G.; Zhang, Y.; Petrillo, M.; Cai, H.; Buisman, C.J.N.; Stams, A.J.M.; Lens, P.N.L. Trace methane oxidation and the methane-dependency of sulfate reduction in anaerobic granular sludge. FEMS Microbiol. Ecol. 2010, 72, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Wegener, G.; Niemann, H.; Elvert, M.; Hinrichs, K.-U.; Boetius, A. Assimilation of methane and inorganic carbon by microbial communities mediating the anaerobic oxidation of methane. Environ. Microbiol. 2008, 10, 2287–2298. [Google Scholar] [CrossRef] [PubMed]
- Meulepas, R.J.; Jagersma, C.G.; Khadem, A.F.; Stams, A.J.; Lens, P.N. Effect of methanogenic substrates on anaerobic oxidation of methane and sulfate reduction by an anaerobic methanotrophic enrichment. Appl. Microbiol. Biotechnol. 2010, 87, 1499–1506. [Google Scholar] [CrossRef] [Green Version]
- Valenzuela, E.I.; Avendaño, K.A.; Balagurusamy, N.; Arriaga, S.; Nieto-Delgado, C.; Thalasso, F.; Cervantes, F.J. Electron shuttling mediated by humic substances fuels anaerobic methane oxidation and carbon burial in wetland sediments. Sci. Total Environ. 2019, 650, 2674–2684. [Google Scholar] [CrossRef]
- Nielsen, L.P.; Risgaard-Petersen, N.; Fossing, H.; Christensen, P.B.; Sayama, M. Electric currents couple spatially separated biogeochemical processes in marine sediment. Nature 2010, 463, 1071–1074. [Google Scholar] [CrossRef]
- Valenzuela, E.I.; Prieto-Davó, A.; López-Lozano, N.E.; Hernández-Eligio, A.; Vega-Alvarado, L.; Juárez, K.; García-González, A.S.; López, M.G.; Cervantes, F.J. Anaerobic methane oxidation driven by microbial reduction of natural organic matter in a tropical wetland. Appl. Environ. Microbiol. 2017, 83, e00645-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| MEL-1 | MEL-2 | MEL-3 | DOM-1 | DOM-2 | ALG-1 | ALG-2 | ALG-3 | ALG-4 | |
|---|---|---|---|---|---|---|---|---|---|
| Tidal elev. (mAMSL) | 0.15 | 0.17 | 0.15 | 0.16 | 0.61 | 0.6 | 0.58 | 0.12 | 0.11 |
| Tidal freq. (tides d−1) | 1.5 | 1.5 | 1.5 | 1.4 | 4.3 | 4.1 | 4.1 | 1.2 | 1.2 |
| Distance to lagoon (m) | 2 | 2 | 2 | 2 | 50 | 55 | 55 | 2 | 2 |
| Distance to effluent (m) | 169 | 594 | 714 | 10 | 278 | No effluent | |||
| Mangrovesoil: | |||||||||
| Salinity (g kg−1) | 31.5 ± 0.4 | 40.2 ± 0.9 | 40.1 ± 0.5 | 30.4 ± 0.8 | 35.1 ± 0.4 | 34.7 ± 0.7 | 35.2 ± 0.1 | 34.7 ± 0.3 | 35.3 ± 0.5 |
| Total organic carbon (%) | 2.1 ± 0.06 | 1.5 ± 0.03 | 2.1 ± 0.07 | 2.2 ± 0.07 | 0.6 ± 0.002 | 2.4 ± 0.1 | 3.3 ± 0.12 | 1.6 ± 0.27 | 2.1 ± 0.03 |
| Total nitrogen (%) | 0.2 ± 0.004 | 0.1 ± 0.001 | 0.2 ± 0.007 | 0.2 ± 0.003 | 0.1 ± 0.0001 | 0.1 ± 0.002 | 0.2 ± 0.006 | 0.1 ± 0.13 | 0.2 ± 0.0003 |
| Total phosphorus (%) | 0.06 ± 0.008 | 0.05 ± 0.003 | 0.06 ± 0.009 | 0.06 ± 0.012 | 0.01 ± 0.002 | 0.01 ± 0.002 | 0.01 ± 0.002 | 0.03 ± 0.003 | 0.03 ± 0.006 |
| δ13C-Organic matter (‰) | −25 ± 0.04 | −25.2 ± 0.01 | −24.9 ± 0.06 | −24.5 ± 0.07 | −22 ± 0.11 | −25.1 ± 0.08 | −25.1 ± 0.07 | −24.5 ± 0.33 | −24.7 ± 0.09 |
| Bulk density (g cm−3) | 1.57 ± 0.01 | 1.51 ± 0.003 | 1.59 ± 0.01 | 1.69 ± 0.01 | 1.69 ± 0.002 | 1.09 ± 0.002 | 1.09 ± 0.002 | 1.15 ± 0.003 | 1.09 ± 0.01 |
| Sand/Silt/Clay (%) | 55/20/25 | 62/23/15 | 58/24/18 | 33/21/46 | 29/39/32 | 12/26/62 | 14/33/53 | 12/45/43 | 16/41/43 |
| Soilpore water: | |||||||||
| Eh redox potential (mV) | −117 to −172 | −119 to −179 | −121 to −175 | −121 to −169 | −118 to −166 | −121 to −165 | −125 to −168 | −129 to −172 | −128 to −177 |
| Ammonium (mg N L−1) | 0.53 | 0.41 | 0.39 | 0.22 | 0.05 | 0.06 | 0.08 | 0.11 | 0.14 |
| Nitrate (mg N L−1) | <0.01 | <0.01 | <0.01 | 0.05 | 0.08 | <0.01 | <0.01 | <0.01 | <0.01 |
| Total nitrogen (mg N L−1) | 2.74 | 2.62 | 2.83 | 0.74 | 0.64 | 1.16 | 0.85 | 0.69 | 0.92 |
| Total phosphorus (mg P L−1) | 0.63 | 0.39 | 0.41 | 0.2 | 0.12 | 0.09 | 0.06 | 0.06 | 0.05 |
| Sulfate (mM) | 26.5 | 27.6 | 28.4 | 25.1 | 25.7 | 26.2 | 26.8 | 28.9 | 29.1 |
| Mangroveforest stand: | |||||||||
| Tree density (tree ha−1) | 2600 | 3300 | 2900 | 3100 | 900 | 3800 | 4400 | ||
| Mean tree heigth (m) | 2.7 ± 0.5 | 3.1 ± 0.8 | 2.9 ± 0.8 | 2.7 ± 0.8 | 1.5 ± 0.7 | 3.9 ± 0.8 | 2.9 ± 0.7 | ||
| Basal area (m2 ha−1) | 7.4 | 50.8 | 32.8 | 15.6 | 2.5 | 58.5 | 77.8 | ||
| MEL-1 | MEL-2 | MEL-3 | DOM-1 | DOM-2 | ALG-1 | ALG-2 | ALG-3 | ALG-4 | |
|---|---|---|---|---|---|---|---|---|---|
| Field measurements: | |||||||||
| δ13C-OM (‰) | −25.1 ± 0.02 | −25.1 ± 0.03 | −25 ± 0.03 | −23.3 ± 0.04 | −23.1 ± 0.09 | −25.1 ± 0.02 | −25.1 ± 0.01 | −24.6 ± 0.05 | −24.7 ± 0.13 |
| δ13C-CH4 (‰) | −46.1 ± 2.86 | −72.2 ± 3.00 | −69.6 ± 7.98 | −53.4 ± 7.56 | −57.8 ± 15.1 | −66.2 ± 1.74 | −68.4 ± 1.81 | −64.5 ± 1.55 | −67 ± 0.71 |
| δ13C-CO2-pore water (‰) | −10.9 ± 1.61 | −12.3 ± 0.23 | −13.6 ± 0.06 | −7.9 ± 0.31 | −9.6 ± 2.55 | −11.7 ± 2.51 | −12.8 ± 0.72 | −14.2 ± 0.47 | −12 ± 0.12 |
| (δ13C-CO2-meth) | −4 ± 2.87 | 22 ± 3.05 | 19.5 ± 7.92 | 6.9 ± 7.64 | 11.7 ± 15.12 | 16 ± 1.69 | 18.3 ± 1.82 | 15.3 ± 1.48 | 17.6 ± 0.45 |
| (f CO2-meth) | 0.67 ± 0.13 | 0.27 ± 0.02 | 0.26 ± 0.05 | 0.53 ± 0.13 | 0.45 ± 0.22 | 0.33 ± 0.05 | 0.28 ± 0.03 | 0.26 ± 0.02 | 0.3 ± 0.001 |
| (f CO2-OM+AOM+other) | 0.24 ± 0.13 | 0.73 ± 0.02 | 0.74 ± 0.05 | 0.47 ± 0.13 | 0.55 ± 0.22 | 0.67 ± 0.05 | 0.72 ± 0.03 | 0.74 ± 0.02 | 0.7 ± 0.001 |
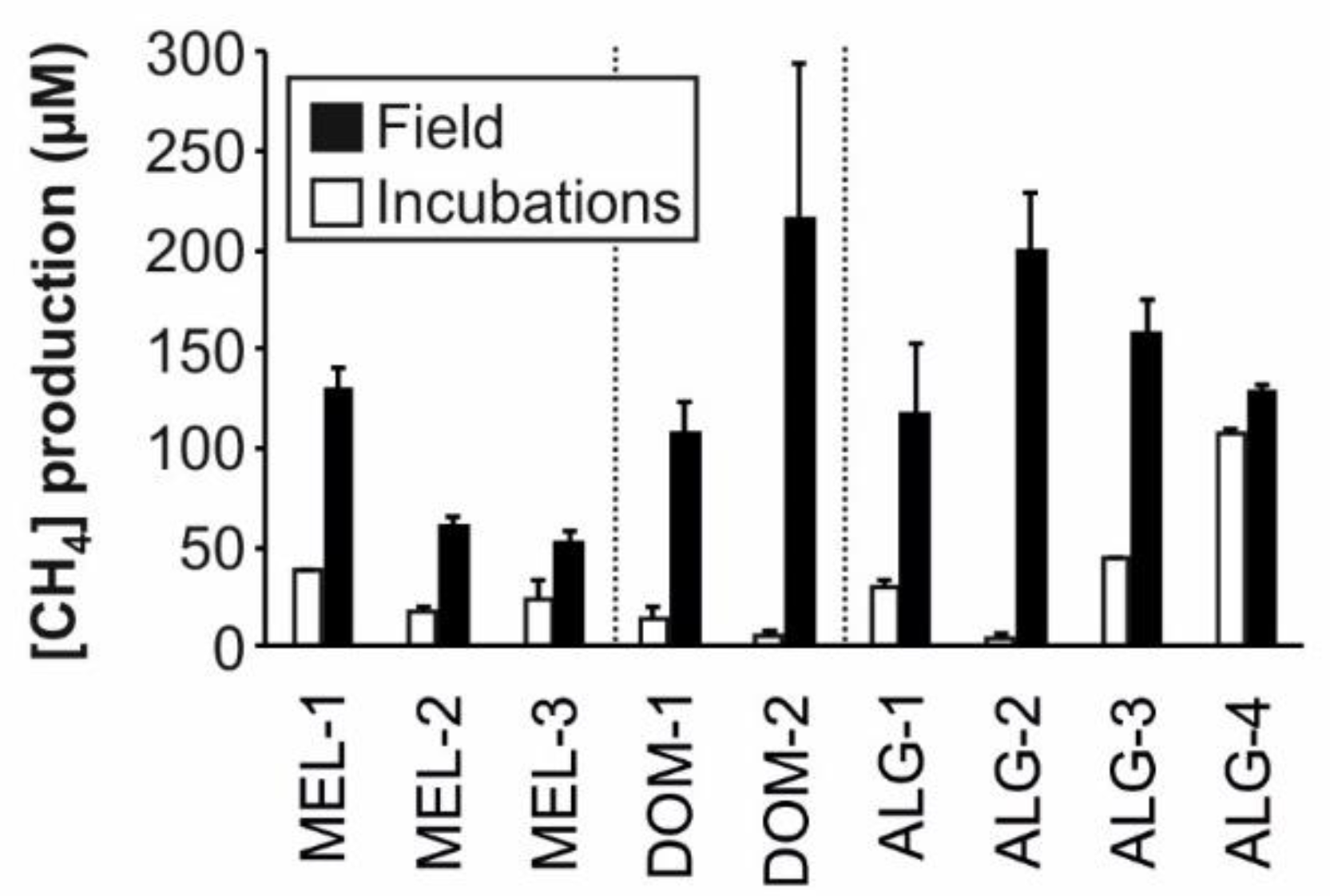
| [CH4]Prod (µM) | 129.9 ± 18.97 | 60.06 ± 9.37 | 51.65 ± 11.15 | 106.88 ± 28.76 | 215.17 ± 135.88 | 116.68 ± 62.62 | 199.03 ± 52.5 | 157.8 ± 29.11 | 127.76 ± 6.16 |
| [CH4]Meas (µM) | 0.04 ± 0.01 | 0.06 ± 0.02 | 0.16 ± 0.04 | 0.04 ± 0.01 | 0.12 ± 0.1 | 0.39 ± 0.24 | 0.23 ± 0.01 | 0.09 ± 0 | 0.09 ± 0.01 |
| [CH4]Fug (µM) | 129.86 ± 18.97 | 59.99 ± 9.35 | 51.49 ± 11.2 | 106.84 ± 28.77 | 215.05 ± 135.95 | 116.29 ± 62.39 | 198.8 ± 52.5 | 157.71 ± 29.11 | 127.66 ± 6.15 |
| (f CH4-Fug) | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Methanog. [CO2-eq (mM)] | 3.6 ± 0.53 | 1.67 ± 0.26 | 1.43 ± 0.31 | 2.97 ± 0.8 | 5.97 ± 3.77 | 3.24 ± 1.74 | 5.52 ± 1.46 | 4.38 ± 0.81 | 3.55 ± 0.17 |
| Laboratory incubations: | |||||||||
| δ13C-OM (‰) | −25.1 ± 0.03 | −25.1 ± 0.04 | −25.0 ± 0.03 | −23.3 ± 0.06 | −23 ± 0.08 | −25.1 ± 0.02 | −25.1 ± 0 | −24.6 ± 0.06 | −24.7 ± 0.13 |
| δ13C-CH4 (‰) | −77.4 ± 3.13 | −80.4 ± 1.27 | −72.3 ± 28.26 | −67.8 ± 8.03 | −61.3 ± 0.99 | −66.5 ± 1.41 | −69.2 ± 0.71 | −64.2 ± 5.03 | −65.1 ± 1.03 |
| δ13C-CO2-pore water (‰) | −24.5 ± 0.14 | −24.7 ± 0.03 | −25.7 ± 0.64 | −23.5 ± 0.12 | −23.6 ± 0.1 | −25 ± 0.02 | −18.3 ± 2.59 | −22.9 ± 0.51 | −25.7 ± 0.12 |
| (δ13C-CO2-meth) | 27.3 ± 3.18 | 30.2 ± 1.19 | 22.2 ± 28.32 | 21.3 ± 7.91 | 15.3 ± 0.82 | 16.3 ± 1.37 | 19 ± 0.71 | 15.1 ± 5.16 | 15.7 ± 1.29 |
| (f CO2-meth) | 0.65 ± 0.01 | 0.65 ± 0.001 | 0.66 ± 0.14 | 0.66 ± 0.02 | 0.68 ± 0.001 | 0.68 ± 0.001 | 0.72 ± 0.02 | 0.69 ± 0.02 | 0.67 ± 0.001 |
| (f CO2-rest) | 0.35 ± 0.01 | 0.35 ± 0.001 | 0.34 ± 0.14 | 0.34 ± 0.02 | 0.32 ± 0.001 | 0.32 ± 0.001 | 0.28 ± 0.02 | 0.31 ± 0.02 | 0.33 ± 0.001 |
| [CH4]Prod (µM) | 37.76 ± 0.44 | 17.56 ± 2.77 | 23.23 ± 10.69 | 13.39 ± 6.42 | 5.41 ± 2.23 | 29.84 ± 3.51 | 3.64 ± 3.13 | 43.93 ± 0.87 | 107.17 ± 2.11 |
| [CH4]Meas (µM) | 0.21 ± 0.02 | 0.13 ± 0.03 | 0.03 ± 0.04 | 0.28 ± 0.12 | 0.05 ± 0.01 | 1.24 ± 0.76 | 1.6 ± 0.31 | 0.15 ± 0.02 | 2.16 ± 1.24 |
| [CH4]AOM (µM) | 37.54 ± 0.43 | 17.42 ± 2.8 | 23.2 ± 10.73 | 13.1 ± 6.55 | 5.36 ± 2.22 | 28.6 ± 2.75 | 2.04 ± 3.44 | 43.79 ± 0.85 | 105.01 ± 3.35 |
| (f CH4-AOM) | 0.99 ± 0.001 | 0.99 ± 0.001 | 1 ± 0.001 | 0.97 ± 0.02 | 0.99 ± 0.001 | 0.96 ± 0.02 | 0.24 ± 0.74 | 1 ± 0.001 | 0.98 ± 0.01 |
| Methanog. [CO2-eq (mM)] | 1.39 ± 0.02 | 0.65 ± 0.1 | 0.85 ± 0.39 | 0.49 ± 0.24 | 0.2 ± 0.08 | 1.1 ± 0.13 | 0.13 ± 0.12 | 1.61 ± 0.03 | 3.94 ± 0.08 |
| δ13C-OM | δ13C-CH4-pore | δ13C-CH4-new | αCO2-CH4 (ε) | (f CH4-mc) | (f CH4-ma) | ||
|---|---|---|---|---|---|---|---|
| (‰) | (-) | (‰) | (-) | (-) | |||
| MEL-1 | −25.1 ± 0.03 | −77.4 ± 3.13 | −80.21 ± 3.6 | 1.036 | (−36 ± 4) | 0.62 ± 0.09 | 0.38 ± 0.91 |
| MEL-2 | −25.1 ± 0.04 | −80.4 ± 1.27 | −85.45 ± 0.15 | 1.039 | (−39 ± 1) | 0.76 ± 0 | 0.24 ± 1 |
| MEL-3 | −25 ± 0.03 | −72.3 ± 28.26 | −75.05 ± 3.89 | 1.010 | (−10 ± 30) | 0.49 ± 0.1 | 0.51 ± 0.9 |
| Mean | −25.06 ± 0.004 | −76.68 ± 3.96 | −80.24 ± 5.22 | 1.028 | (−28 ± 20) | 0.62 ± 0.13 | 0.38 ± 0.13 |
| DOM-1 | −23.3 ± 0.06 | −67.8 ± 8.03 | −69.4 ± 7.78 | 1.029 | (−29 ± 9) | 0.34 ± 0.2 | 0.66 ± 0.8 |
| DOM-2 | −23 ± 0.08 | −61.3 ± 0.99 | −63.65 ± 1.34 | 1.022 | (−22 ± 1) | 0.2 ± 0.03 | 0.8 ± 0.97 |
| Mean | −23.14 ± 0.007 | −64.58 ± 5.98 | −66.52 ± 5.64 | 1.026 | (−26 ± 7) | 0.27 ± 0.14 | 0.92 ± 0.03 |
| ALG-1 | −25.1 ± 0.02 | −66.5 ± 1.41 | −66.79 ± 1.26 | 1.026 | (−26 ± 2) | 0.28 ± 0.03 | 0.72 ± 0.97 |
| ALG-2 | −25.1 ± 0 | −69.2 ± 0.71 | −69.39 ± 0.76 | 1.029 | (−29 ± 1) | 0.34 ± 0.02 | 0.66 ± 0.98 |
| ALG-3 | −24.6 ± 0.06 | −64.2 ± 5.03 | −69.8 ± 5.42 | 1.024 | (−24 ± 6) | 0.35 ± 0.14 | 0.65 ± 0.86 |
| ALG-4 | −24.7 ± 0.13 | −65.1 ± 1.03 | −65.56 ± 1.31 | 1.025 | (−25 ± 1) | 0.25 ± 0.03 | 0.75 ± 0.97 |
| Mean | −24.86 ± 0.003 | −66.25 ± 2.84 | −67.89 ± 2.89 | 1.026 | (−26 ± 3) | 0.3 ± 0.07 | 0.7 ± 0.07 |
| Overall mean | −24.54 ± 0.8 | −69.36 ± 6.56 | −71.7 ± 7.48 | 1.027 | (−27 ± 11) | 0.4 ± 0.19 | 0.6 ± 0.19 |
| Field | Incubations | |||
|---|---|---|---|---|
| (f CO2-meth) | [CH4]prod (µM) | (f CO2-meth) | [CH4]prod (µM) | |
| Tidal and forest drivers: | ||||
| Tidal freq. (tides d−1) | −0.06 | 0.62 | −0.40 | −0.49 |
| Basal area (m2 ha−1) | −0.77 | 0.04 | −0.34 | 0.43 |
| Tree density (tree ha−1) | −0.53 | −0.14 | −0.16 | 0.46 |
| Soil fraction: | ||||
| Bulk density (g/cm3) | 0.31 | −0.14 | −0.45 | −0.37 |
| Total organic carbon (%) | 0.17 | −0.10 | 0.27 | −0.14 |
| Total nitrogen (%) | 0.22 | −0.17 | −0.22 | 0.00 |
| Total phosphorus (%) | 0.07 | -0.64 | −0.80 | 0.23 |
| Sand (%) | 0.05 | -0.56 | −0.86 | −0.20 |
| Silt (%) | −0.48 | 0.52 | 0.78 | 0.23 |
| Clay (%) | 0.23 | 0.33 | 0.69 | −0.09 |
| Porewater fraction: | ||||
| Salinity (mg/L) | −0.67 | −0.29 | −0.08 | −0.07 |
| Total solids (mg/L) | 0.11 | −0.48 | −0.60 | −0.48 |
| Total organic matter (mg/L) | 0.26 | −0.38 | −0.67 | −0.47 |
| Total inorganic matter (mg/L) | −0.33 | 0.22 | 0.48 | −0.07 |
| N-NO2− (mg N/L) | 0.26 | −0.39 | −0.90 | 0.07 |
| N-NH4+ (mg N/L) | 0.01 | −0.60 | −0.85 | 0.28 |
| N-NO3− (mg N/L) | 0.50 | 0.27 | 0.03 | −0.52 |
| Tot-N (mg N/L) | −0.13 | −0.68 | −0.64 | 0.27 |
| Tot-P (mg P/L) | 0.25 | −0.46 | −0.78 | −0.18 |
| SO42− (mM) | −0.73 | −0.13 | 0.08 | 0.62 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Carrillo, S.; Garatuza-Payan, J.; Sánchez-Andrés, R.; Cervantes, F.J.; Bartolomé, M.C.; Merino-Ibarra, M.; Thalasso, F. Methane Production and Oxidation in Mangrove Soils Assessed by Stable Isotope Mass Balances. Water 2021, 13, 1867. https://doi.org/10.3390/w13131867
Sánchez-Carrillo S, Garatuza-Payan J, Sánchez-Andrés R, Cervantes FJ, Bartolomé MC, Merino-Ibarra M, Thalasso F. Methane Production and Oxidation in Mangrove Soils Assessed by Stable Isotope Mass Balances. Water. 2021; 13(13):1867. https://doi.org/10.3390/w13131867
Chicago/Turabian StyleSánchez-Carrillo, Salvador, Jaime Garatuza-Payan, Raquel Sánchez-Andrés, Francisco J. Cervantes, María Carmen Bartolomé, Martín Merino-Ibarra, and Frederic Thalasso. 2021. "Methane Production and Oxidation in Mangrove Soils Assessed by Stable Isotope Mass Balances" Water 13, no. 13: 1867. https://doi.org/10.3390/w13131867
APA StyleSánchez-Carrillo, S., Garatuza-Payan, J., Sánchez-Andrés, R., Cervantes, F. J., Bartolomé, M. C., Merino-Ibarra, M., & Thalasso, F. (2021). Methane Production and Oxidation in Mangrove Soils Assessed by Stable Isotope Mass Balances. Water, 13(13), 1867. https://doi.org/10.3390/w13131867








